The Contribution of Host Cells to Pneumocystis Immunity: An Update
Abstract
1. Introduction
2. Pneumocystis Life Forms and Host Cells
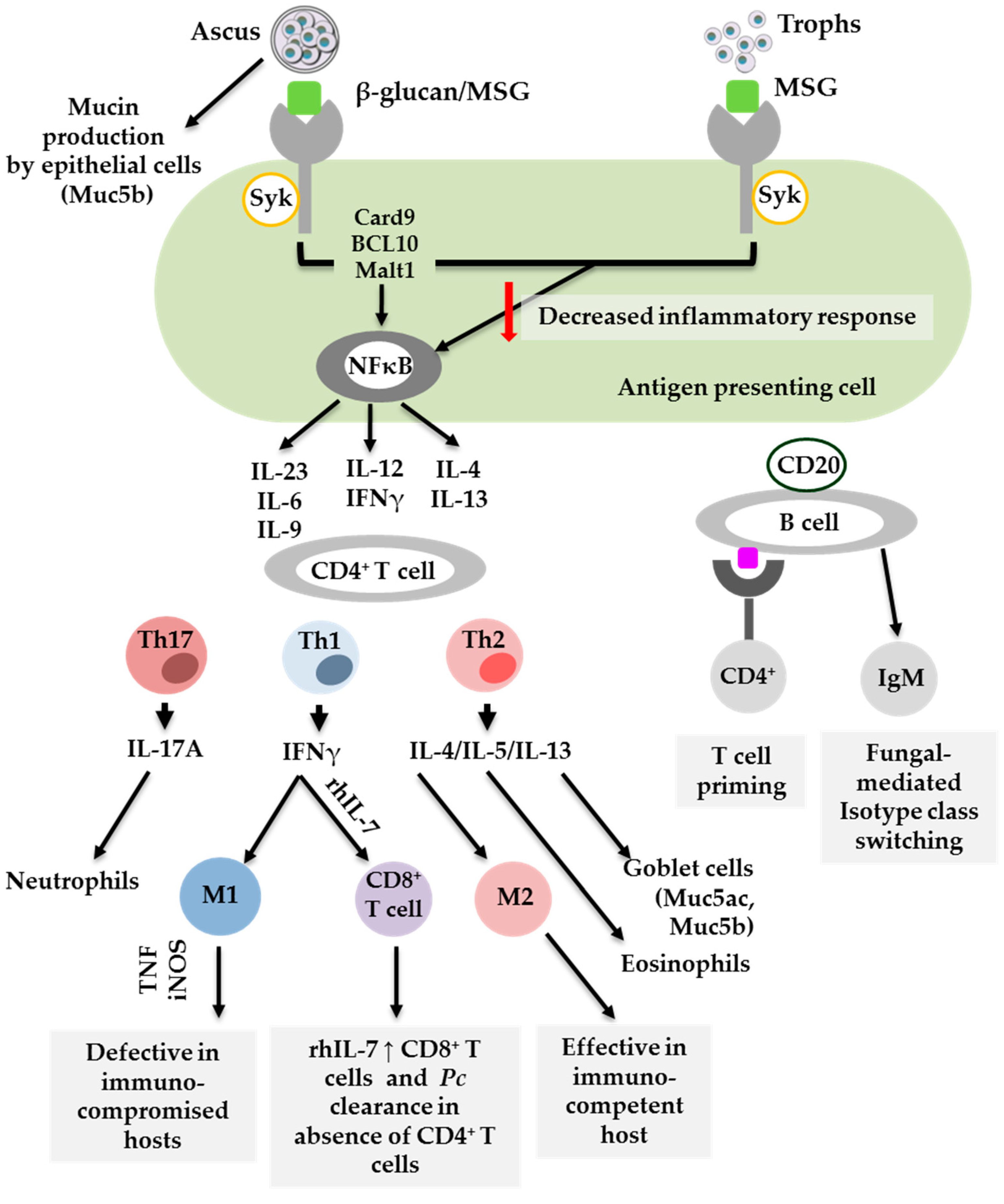
3. Dendritic Cells
4. Alveolar Macrophages
5. Lymphocytes: CD4+ T Cells, CD8+ T Cells and B Cells
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Morris, A.; Norris, K.A. Colonization by Pneumocystis jirovecii and its role in disease. Clin. Microbiol. Rev. 2012, 25, 297–317. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Cisse, O.H.; Kovacs, J.A. A Molecular Window into the Biology and Epidemiology of Pneumocystis spp. Clin. Microbiol. Rev. 2018, 31. [Google Scholar] [CrossRef] [PubMed]
- Buchacz, K.; Baker, R.K.; Palella, F.J., Jr.; Chmiel, J.S.; Lichtenstein, K.A.; Novak, R.M.; Wood, K.C.; Brooks, J.T.; HOPS Investigators. AIDS-defining opportunistic illnesses in US patients, 1994–2007: A cohort study. AIDS 2010, 24, 1549–1559. [Google Scholar] [CrossRef] [PubMed]
- Kelley, C.F.; Checkley, W.; Mannino, D.M.; Franco-Paredes, C.; Del Rio, C.; Holguin, F. Trends in hospitalizations for AIDS-associated Pneumocystis jirovecii Pneumonia in the United States (1986 to 2005). Chest 2009, 136, 190–197. [Google Scholar] [CrossRef] [PubMed]
- Kovacs, J.A.; Masur, H. Evolving health effects of Pneumocystis: One hundred years of progress in diagnosis and treatment. JAMA 2009, 301, 2578–2585. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, M.; Fishman, J.A. Prevention of infection due to Pneumocystis spp. in human immunodeficiency virus-negative immunocompromised patients. Clin. Microbiol. Rev. 2004, 17, 770–782. [Google Scholar] [CrossRef]
- Bienvenu, A.L.; Traore, K.; Plekhanova, I.; Bouchrik, M.; Bossard, C.; Picot, S. Pneumocystis pneumonia suspected cases in 604 non-HIV and HIV patients. Int. J. Infect. Dis. 2016, 46, 11–17. [Google Scholar] [CrossRef]
- Maini, R.; Henderson, K.L.; Sheridan, E.A.; Lamagni, T.; Nichols, G.; Delpech, V.; Phin, N. Increasing Pneumocystis pneumonia, England, UK, 2000–2010. Emerg. Infect. Dis. 2013, 19, 386–392. [Google Scholar] [CrossRef]
- Monnet, X.; Vidal-Petiot, E.; Osman, D.; Hamzaoui, O.; Durrbach, A.; Goujard, C.; Miceli, C.; Bourée, P.; Richard, C. Critical care management and outcome of severe Pneumocystis pneumonia in patients with and without HIV infection. Crit. Care 2008, 12, R28. [Google Scholar] [CrossRef]
- Wang, E.H.; Partovi, N.; Levy, R.D.; Shapiro, R.J.; Yoshida, E.M.; Greanya, E.D. Pneumocystis pneumonia in solid organ transplant recipients: Not yet an infection of the past. Transpl. Infect. Dis. 2012, 14, 519–525. [Google Scholar] [CrossRef]
- Fillatre, P.; Decaux, O.; Jouneau, S.; Revest, M.; Gacouin, A.; Robert-Gangneux, F.; Fresnel, A.; Guiguen, C.; Le Tulzo, Y.; Jégo, P.; et al. Incidence of Pneumocystis jiroveci pneumonia among groups at risk in HIV-negative patients. Am. J. Med. 2014, 127, 1242. [Google Scholar] [CrossRef]
- Su, Y.S.; Lu, J.J.; Perng, C.L.; Chang, F.Y. Pneumocystis jirovecii pneumonia in patients with and without human immunodeficiency virus infection. J. Microbiol. Immunol. Infect. 2008, 41, 478–482. [Google Scholar] [PubMed]
- Limper, A.H.; Offord, K.P.; Smith, T.F.; Martin, W.J., 2nd. Pneumocystis carinii pneumonia. Differences in lung parasite number and inflammation in patients with and without AIDS. Am. Rev. Respir. Dis. 1989, 140, 1204–1209. [Google Scholar] [CrossRef]
- Thomas, C.F., Jr.; Limper, A.H. Pneumocystis pneumonia. N. Engl. J. Med. 2004, 350, 2487–2498. [Google Scholar] [CrossRef] [PubMed]
- Festic, E.; Gajic, O.; Limper, A.H.; Aksamit, T.R. Acute respiratory failure due to pneumocystis pneumonia in patients without human immunodeficiency virus infection: Outcome and associated features. Chest 2005, 128, 573–579. [Google Scholar] [CrossRef]
- De Armas Rodriguez, Y.; Wissmann, G.; Muller, A.L.; Pederiva, M.A.; Brum, M.C.; Brackmann, R.L.; Capó de Paz, V.; Calderón, E.J. Pneumocystis jirovecii pneumonia in developing countries. Parasite 2011, 18, 219–228. [Google Scholar] [CrossRef]
- Wasserman, S.; Engel, M.E.; Griesel, R.; Mendelson, M. Burden of pneumocystis pneumonia in HIV-infected adults in sub-Saharan Africa: A systematic review and meta-analysis. BMC Infect. Dis. 2016, 16. [Google Scholar] [CrossRef]
- Chiliza, N.; Du Toit, M.; Wasserman, S. Outcomes of HIV-associated pneumocystis pneumonia at a South African referral hospital. PLoS ONE 2018, 13, e0201733. [Google Scholar] [CrossRef]
- Cushion, M.T.; Smulian, A.G.; Slaven, B.E.; Sesterhenn, T.; Arnold, J.; Staben, C.; Porollo, A.; Adamczak, R.; Meller, J. Transcriptome of Pneumocystis carinii during fulminate infection: Carbohydrate metabolism and the concept of a compatible parasite. PLoS ONE 2007, 2, e423. [Google Scholar] [CrossRef] [PubMed]
- Hauser, P.M. Genomic Insights into the Fungal Pathogens of the Genus Pneumocystis: Obligate Biotrophs of Humans and Other Mammals. PLoS Pathog. 2014, 10, e1004425. [Google Scholar] [CrossRef] [PubMed]
- Matsumura, Y.; Tsuchido, Y.; Yamamoto, M.; Nakano, S.; Nagao, M. Development of a fully automated PCR assay for the detection of Pneumocystis jirovecii using the GENECUBE system. Med. Mycol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Singh, Y.; Mirdha, B.R.; Guleria, R.; Kabra, S.K.; Mohan, A.; Chaudhry, R.; Kumar, L.; Dwivedi, S.N.; Agarwal, S.K. Genetic polymorphisms associated with treatment failure and mortality in pediatric Pneumocystosis. Sci. Rep. 2019, 9, 1192. [Google Scholar] [CrossRef]
- Cushion, M.T.; Ruffolo, J.J.; Walzer, P.D. Analysis of the developmental stages of Pneumocystis carinii, in vitro. Lab. Investig. J. Tech. Methods Pathol. 1988, 58, 324–331. [Google Scholar]
- Evans, H.M.; Bryant, G.L.; Garvy, B.A. The life cycle stages of Pneumocystis murina have opposing effects on the immune response to this opportunistic, fungal pathogen. Infect. Immun. 2016, 84, 3195–3205. [Google Scholar] [CrossRef] [PubMed]
- Kottom, T.J.; Hebrink, D.M.; Jenson, P.E.; Gudmundsson, G.; Limper, A.H. Evidence for Proinflammatory β-1,6 Glucans in the Pneumocystis carinii Cell Wall. Infect. Immun. 2015, 83, 2816–2826. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Evans, H.M.; Simpson, A.; Shen, S.; Stromberg, A.J.; Pickett, C.L.; Garvy, B.A. The Trophic Life Cycle Stage of the Opportunistic Fungal Pathogen Pneumocystis murina Hinders the Ability of Dendritic Cells to Stimulate CD4+ T Cell Responses. Infect. Immun. 2017, 85. [Google Scholar] [CrossRef]
- Evans, H.M.; Garvy, B.A. The trophic life cycle stage of Pneumocystis species induces protective adaptive responses without inflammation-mediated progression to pneumonia. Med. Mycol. 2018, 56, 994–1005. [Google Scholar] [CrossRef]
- Kottom, T.J.; Hebrink, D.M.; Limper, A.H. Binding of Pneumocystis carinii to the lung epithelial cell receptor HSPA5 (GRP78). J. Med. Microbiol. 2018, 67, 1772–1777. [Google Scholar] [CrossRef]
- Perez-Nazario, N.; Rangel-Moreno, J.; O’Reilly, M.A.; Pasparakis, M.; Gigliotti, F.; Wright, T.W. Selective ablation of lung epithelial IKK2 impairs pulmonary Th17 responses and delays the clearance of Pneumocystis. J. Immunol. 2013, 191, 4720–4730. [Google Scholar] [CrossRef]
- Eddens, T.; Campfield, B.T.; Serody, K.; Manni, M.L.; Horne, W.; Elsegeiny, W.; McHugh, K.J.; Pociask, D.; Chen, K.; Zheng, M.; et al. A Novel CD4+ T Cell-Dependent Murine Model of Pneumocystis-driven Asthma-like Pathology. Am. J. Respir. Crit. Care Med. 2016, 194, 807–820. [Google Scholar] [CrossRef]
- Iturra, P.A.; Rojas, D.A.; Pérez, F.J.; Méndez, A.; Ponce, C.A.; Bonilla, P.; Bustamante, R.; Rodríguez, H.; Beltrán, C.J.; Vargas, S.L. Progression of type 2 helper T cell–type inflammation and airway remodeling in a rodent model of naturally acquired subclinical primary pneumocystis infection. Am. J. Pathol. 2018, 188, 417–431. [Google Scholar] [CrossRef]
- Rojas, D.A.; Iturra, P.A.; Méndez, A.; Ponce, C.A.; Bustamante, R.; Gallo, M.; Bórquez, P.; Vargas, S.L. Increase in secreted airway mucins and partial Muc5b STAT6/FoxA2 regulation during Pneumocystis primary infection. Sci. Rep. 2019, 14, 2078. [Google Scholar] [CrossRef]
- McAllister, F.; Mc Allister, F.; Steele, C.; Zheng, M.; Young, E.; Shellito, J.E.; Marrero, L.; Kolls, J.K. T cytotoxic-1 CD8+ T cells are effector cells against pneumocystis in mice. J. Immunol. 2004, 172, 1132–1138. [Google Scholar] [CrossRef]
- Ruan, S.; Samuelson, D.R.; Assouline, B.; Morre, M.; Shellito, J.E. Treatment with Interleukin-7 Restores Host Defense against Pneumocystis in CD4+ T-Lymphocyte-Depleted Mice. Infect. Immun. 2016, 84, 108–119. [Google Scholar] [CrossRef]
- Carmona, E.M.; Vassallo, R.; Vuk-Pavlovic, Z.; Standing, J.E.; Kottom, T.J.; Limper, A.H. Pneumocystis cell wall beta-glucans induce dendritic cell costimulatory molecule expression and inflammatory activation through a Fas-Fas ligand mechanism. J. Immunol. 2006, 177, 459–467. [Google Scholar] [CrossRef] [PubMed]
- Carmona, E.M.; Kottom, T.J.; Hebrink, D.M.; Moua, T.; Singh, R.-D.; Pagano, R.E.; Limper, A.H. Glycosphingolipids mediate pneumocystis cell wall β-glucan activation of the IL-23/IL-17 axis in human dendritic cells. Am. J. Respir. Cell Mol. Biol. 2012, 47, 50–59. [Google Scholar] [CrossRef]
- Sassi, M.; Kutty, G.; Ferreyra, G.A.; Bishop, L.R.; Liu, Y.; Qiu, J.; Huang, D.W.; Kovacs, J.A. The Major Surface Glycoprotein of Pneumocystis murina Does Not Activate Dendritic Cells. J. Infect. Dis. 2018, 218, 1631–1640. [Google Scholar] [CrossRef]
- Limper, A.H.; Hoyte, J.S.; Standing, J.E. The role of alveolar macrophages in Pneumocystis carinii degradation and clearance from the lung. J. Clin. Investig. 1997, 99, 2110–2117. [Google Scholar] [CrossRef] [PubMed]
- Leopold Wager, C.M.; Wormley, F.L. Classical versus alternative macrophage activation: The Ying and the Yang in host defense against pulmonary fungal infections. Mucosal. Immunol. 2014, 7, 1023–1035. [Google Scholar] [CrossRef]
- Nandakumar, V.; Hebrink, D.; Jenson, P.; Kottom, T.; Limper, A.H. Differential Macrophage Polarization from Pneumocystis in Immunocompetent and Immunosuppressed Hosts: Potential Adjunctive Therapy during Pneumonia. Infect. Immun. 2017, 85. [Google Scholar] [CrossRef]
- Bhagwat, S.P.; Gigliotti, F.; Wang, J.; Wang, Z.; Notter, R.H.; Murphy, P.S.; Rivera-Escalera, F.; Malone, J.; Jordan, M.B.; Elliott, M.R.; et al. Intrinsic Programming of Alveolar Macrophages for Protective Antifungal Innate Immunity against Pneumocystis Infection. Front. Immunol. 2018, 9, 2131. [Google Scholar] [CrossRef] [PubMed]
- Rudner, X.L.; Happel, K.I.; Young, E.A.; Shellito, J.E. Interleukin-23 (IL-23)-IL-17 cytokine axis in murine Pneumocystis carinii infection. Infect. Immun. 2007, 75, 3055–3061. [Google Scholar] [CrossRef] [PubMed]
- Ripamonti, C.; Bishop, L.R.; Kovacs, J.A. Pulmonary Interleukin-17-Positive Lymphocytes Increase during Pneumocystis murina Infection but Are Not Required for Clearance of Pneumocystis. Infect. Immun. 2017, 85. [Google Scholar] [CrossRef] [PubMed]
- Elyaman, W.; Bradshaw, E.M.; Uyttenhove, C.; Dardalhon, V.; Awasthi, A.; Imitola, J.; Bettelli, E.; Oukka, M.; van Snick, J.; Renauld, J.-C.; et al. IL-9 induces differentiation of TH17 cells and enhances function of FoxP3+ natural regulatory T cells. Proc. Natl. Acad. Sci. USA 2009, 106, 12885–12890. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Rong, H.-M.; Zhang, C.; Zhai, K.; Tong, Z.-H. IL-9 Deficiency Promotes Pulmonary Th17 Response in Murine Model of Pneumocystis Infection. Front. Immunol. 2018, 9, 1118. [Google Scholar] [CrossRef] [PubMed]
- Kolls, J.K.; Habetz, S.; Shean, M.K.; Vazquez, C.; Brown, J.A.; Lei, D.; Schwarzenberger, P.; Ye, P.; Nelson, S.; Summer, W.R.; et al. IFN-γ and CD8+ T Cells Restore Host Defenses Against Pneumocystis carinii in Mice Depleted of CD4+ T Cells. J. Immunol. 1999, 162, 2890–2894. [Google Scholar] [PubMed]
- Myers, R.C.; Dunaway, C.W.; Nelson, M.P.; Trevor, J.L.; Morris, A.; Steele, C. STAT4-dependent and -independent Th2 responses correlate with protective immunity against lung infection with Pneumocystis murina. J. Immunol. 2013, 190, 6287–6294. [Google Scholar] [CrossRef] [PubMed]
- Eddens, T.; Elsegeiny, W.; Nelson, M.P.; Horne, W.; Campfield, B.T.; Steele, C.; Kolls, J.K. Eosinophils Contribute to Early Clearance of Pneumocystis murina Infection. J. Immunol. 2015, 195, 185–193. [Google Scholar] [CrossRef] [PubMed]
- Vargas, S.L.; Ponce, C.A.; Gallo, M.; Pérez, F.; Astorga, J.F.; Bustamante, R.; Chabé, M.; Durand-Joly, I.; Iturra, P.; Miller, R.F.; et al. Near-universal prevalence of Pneumocystis and associated increase in mucus in the lungs of infants with sudden unexpected death. Clin. Infect. Dis. 2013, 2, 171–179. [Google Scholar] [CrossRef]
- Hernandez-Novoa, B.; Bishop, L.; Logun, C.; Munson, P.J.; Elnekave, E.; Rangel, Z.G.; Barb, J.; Danner, R.L.; Kovacs, J.A. Immune responses to Pneumocystis murina are robust in healthy mice but largely absent in CD40 ligand-deficient mice. J. Leukoc. Biol. 2008, 84, 420–430. [Google Scholar] [CrossRef]
- Gigliotti, F.; Crow, E.L.; Bhagwat, S.P.; Wright, T.W. Sensitized CD8+ T cells fail to control organism burden but accelerate the onset of lung injury during Pneumocystis carinii pneumonia. Infect. Immun. 2006, 74, 6310–6316. [Google Scholar] [CrossRef] [PubMed]
- De la Rua, N.M.; Samuelson, D.R.; Charles, T.P.; Welsh, D.A.; Shellito, J.E. CD4+ T-Cell-Independent Secondary Immune Responses to Pneumocystis Pneumonia. Mucosal. Immun. 2016, 178. [Google Scholar] [CrossRef]
- Kolls, J.K. An Emerging Role of B Cell Immunity in Susceptibility to Pneumocystis Pneumonia. Am. J. Respir. Cell Mol. Biol. 2017, 56, 279–280. [Google Scholar] [CrossRef] [PubMed]
- Lund, F.E.; Hollifield, M.; Schuer, K.; Lines, J.L.; Randall, T.D.; Garvy, B.A. B cells are required for generation of protective effector and memory CD4 cells in response to Pneumocystis lung infection. J. Immunol. 2006, 176, 6147–6154. [Google Scholar] [CrossRef] [PubMed]
- Opata, M.M.; Hollifield, M.L.; Lund, F.E.; Randall, T.D.; Dunn, R.; Garvy, B.A.; Feola, D.J. B Lymphocytes Are Required during the Early Priming of CD4+ T Cells for Clearance of Pneumocystis Infection in Mice. J. Immunol. 2015, 195, 611–620. [Google Scholar] [CrossRef]
- Rapaka, R.R.; Ricks, D.M.; Alcorn, J.F.; Chen, K.; Khader, S.A.; Zheng, M.; Plevy, S.; Bengtén, E.; Kolls, J.K. Conserved natural IgM antibodies mediate innate and adaptive immunity against the opportunistic fungus Pneumocystis murina. J. Exp. Med. 2010, 207, 2907–2919. [Google Scholar] [CrossRef]
- Elsegeiny, W.; Eddens, T.; Chen, K.; Kolls, J.K. Anti-CD20 Antibody Therapy and Susceptibility to Pneumocystis Pneumonia. Infect. Immun. 2015, 83, 2043–2052. [Google Scholar] [CrossRef] [PubMed]
- Gigliotti, F.; Haidaris, C.G.; Wright, T.W.; Harmsen, A.G. Passive intranasal monoclonal antibody prophylaxis against murine Pneumocystis carinii pneumonia. Infect. Immun. 2002, 70, 1069–1074. [Google Scholar] [CrossRef] [PubMed]
- Zheng, M.; Shellito, J.E.; Marrero, L.; Zhong, Q.; Julian, S.; Ye, P.; Wallace, V.; Schwarzenberger, P.; Kolls, J.K. CD4+ T cell-independent vaccination against Pneumocystis carinii in mice. J. Clin. Investig. 2001, 108, 1469–1474. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Otieno-Odhiambo, P.; Wasserman, S.; Hoving, J.C. The Contribution of Host Cells to Pneumocystis Immunity: An Update. Pathogens 2019, 8, 52. https://doi.org/10.3390/pathogens8020052
Otieno-Odhiambo P, Wasserman S, Hoving JC. The Contribution of Host Cells to Pneumocystis Immunity: An Update. Pathogens. 2019; 8(2):52. https://doi.org/10.3390/pathogens8020052
Chicago/Turabian StyleOtieno-Odhiambo, Patricia, Sean Wasserman, and J. Claire Hoving. 2019. "The Contribution of Host Cells to Pneumocystis Immunity: An Update" Pathogens 8, no. 2: 52. https://doi.org/10.3390/pathogens8020052
APA StyleOtieno-Odhiambo, P., Wasserman, S., & Hoving, J. C. (2019). The Contribution of Host Cells to Pneumocystis Immunity: An Update. Pathogens, 8(2), 52. https://doi.org/10.3390/pathogens8020052




