2.2.1. Surface Charge and Hydrophobicity
It is important to remember that microorganisms have different mechanisms of adhesion and retention, influenced by the substrata, nutrients, ionic strength, pH values and temperatures, and also their phenotype and genotype [
40]. The ability of microorganisms to attach to the surfaces is crucial for the beginning of colonization. The process of adhesion of microorganisms to surfaces is very complex, and is affected by many variables. The hydrophobicity and the charge of the cell surface, the presence of bacterial adhesins (e.g., fimbriae, flagella and pili), and particularly the quantity and composition of generated EPS, are the main factors that influence both the rate and degree of microbial adhesion [
41]. Indeed, the surface properties of microbial cells have a major impact on adhesion to a substratum. The surface charge of the cells is often determined as its zeta potential, which is measured from the mobility of cells in the presence of an electrical field under well-known conditions (pH and salt concentrations) [
42,
43]. At physiologic conditions, most of the microorganisms are negatively charged due to the presence of anionic groups, such as carboxyl and phosphate, in their membranes [
42].
Table 2 shows the several variations of zeta potential of cells in the presence of the different phytochemicals.
Table 2.
Zeta potential (mV) results of suspensions of E. coli and S. aureus exposed to the phytochemicals at their MIC.
Table 2.
Zeta potential (mV) results of suspensions of E. coli and S. aureus exposed to the phytochemicals at their MIC.
| | Zeta Potencial (mV) |
|---|
| | E. coli CECT 434 | S. aureus CECT 976 |
|---|
| Control | −13.0 ± 1.4 | −29.8 ± 1.3 |
| 7-HC | −13.5 ± 1.8 | −26.0 ± 6.2 |
| I3C | −21.0 ± 5.7 | −27.3 ± 6.3 |
| SA | −1.80 ± 0.3 | 1.80 ± 0.5 |
| SP | −12.7 ± 1.6 | −19.4 ± 2.2 |
Both bacterial strains present a negative surface charge: −13.0 mV for
E. coli and −29.8 mV for
S. aureus. 7-HC and SP have no significant effect in the charge of
E. coli (
p > 0.05). In contrast, I3C and SA seem to change the
E. coli surface charge (
p < 0.05) for more and less negative values, respectively. The charge of
S. aureus surface become less negative when exposed to all compounds, but significant alteration was observed when in contact with SA and SP (
p < 0.05). The first one was able to make the membrane surface positive, while the exposure to SP changed the surface charge of cells to less negative values. Other authors have also studied the effects of phenolic compounds in cell surface charge with the same bacteria [
32]. In accordance with our results, they showed that
E. coli and
S. aureus has a negative surface charge. The interaction between bacteria and phenolics (gallic and ferulic acids) change the surface charge of cells to less negative values especially for
E. coli. In the case of
S. aureus, the zeta potential values were similar to the control. In a study performed by Abreu
et al. [
7], the zeta potential was studied for
E. coli and
S. aureus when in contact with a glucosinolate (phenyl isothiocyanate). It was verified that the values of surface charge of the cells become less negative for both bacteria. In our study, this was particularly verified in the case of
S. aureus.
As mentioned above, the hydrophobicity has been characterized as one important aspect in bacterial adhesion [
44]. The hydrophobicity can be calculated through the energy of hydrophobic attraction (∆
GTOT). If ∆
GTOT < 0 mJ/cm
2, the interaction between the surface molecules is attractive, which means that molecules have less affinity for water than among themselves, and the surface is considered hydrophobic. If ∆
GTOT > 0, the surface is considered hydrophilic, and the interaction between the surface molecules is repulsive. Therefore, the more negative the value of ∆
GTOT, more hydrophobic is the surface; and the more positive the value of Δ
GTOT, more hydrophilic is the surface [
44,
45].
Table 3 presents the results of hydrophobicity obtained for
E. coli and
S. aureus cells in the presence of the four phytochemicals. Regarding the several parameters, the apolar component (γ
LW) was particularly changed when
E. coli is treated with I3C and SP (
p < 0.05), making their surface less apolar. The same phytochemicals were also able to change the γ
AB component making the cells more polar (
p < 0.05). However, in the presence of 7-HC and mainly SA,
E. coli become less polar. The electron donor component increased with the application of I3C and SP and decreased with 7-HC and SP. In the case of
S. aureus, the treatment with the several phytochemicals did not permit variations on the polarity of surface molecules (
p > 0.05). Except in the case of SA, the value of γ
AB was lower than the observed in the control, which means that the surface of molecules was less hydrophilic. Regarding their electron donor capacity, 7-HC and SA varied significantly (
p < 0.05), compared with the values obtained when
S. aureus was not treated with phytochemicals. These phytochemicals decreased the capacity to donate electrons,
i.e., when
S. aureus was in contact with 7-HC and SA the cell lost its ability to give electrons (γ
− decreased).
The ΔG
TOT values obtained for
E. coli and
S. aureus, before exposure to the phytochemicals, show that they have hydrophilic character (ΔG
TOT > 0 mJ/m
2). It is possible to observe changes in the bacterial membrane physicochemical character of
E. coli with the application of all compounds particularly with SA and SP (
p < 0.05). The application of 7-HC, I3C and SP promoted the decrease of hydrophilic properties of
E. coli. However, with SA the cell surface of this bacterium become more hydrophilic. The opposite effect was observed for
S. aureus with SA. This phytochemical induced a cell surface hydrophobic character (
p < 0.05). Similar result was obtained with 7-HC. However, I3C increased bacterial hydrophilic character (
p < 0.05). In general, the results obtained demonstrated that the selected phytochemicals significantly interact with bacterial surface constituents, modifying their physicochemical properties. In other work [
7], the interaction of a GHP, phenyl isothiocyanate, with bacterial cells was assessed. The results demonstrated that the interaction caused an alteration of cell surface hydrophobicity. The interaction with the phytochemical promoted the increase of their hydrophilic character. Even if the cell membrane is the first contact point between the microorganism and the phytochemical, to our knowledge there are no relevant studies on the effects of phytochemicals on microbial surface properties.
Table 3.
Hydrophobicity (∆GTOT), and apolar (γLW) and polar (γAB) components of the surface tension of untreated (control) and phytochemical treated cells. The means ± SDs are illustrated.
Table 3.
Hydrophobicity (∆GTOT), and apolar (γLW) and polar (γAB) components of the surface tension of untreated (control) and phytochemical treated cells. The means ± SDs are illustrated.
| Bacteria | Phytochemical | Surface Tension Parameters (mJ/m2) | Hydrophobicity (mJ/m2) |
|---|
| γLW | γAB | γ
+ | γ
− | ∆GTOT |
|---|
| E. coli CECT 434 | Control | 33.6 ± 5.0 | 22.4 ± 5.4 | 2.6 ± 0.5 | 52.0 ± 4.8 | 28.9 ± 7.1 |
| 7-HC | 30.7 ± 4.8 | 20.8 ± 4.7 | 1.50 ± 0.3 | 44.9 ± 7.1 | 21.0 ± 5.1 |
| I3C | 20.2 ± 4.8 | 37.1 ± 7.3 | 7.80 ± 1.9 | 55.8 ± 6.7 | 20.6 ± 3.8 |
| SA | 31.0 ± 5.2 | 2.07 ± 6.0 | 1.50 ± 0.3 | 59.7 ± 11 | 37.6 ± 18 |
| SP | 21.0 ± 1.8 | 40.1 ± 3.1 | 7.80 ± 1.3 | 52.1 ± 2.8 | 19.7 ± 3.3 |
| S. aureus CECT 976 | Control | 35.4 ± 5.4 | 19.7 ± 4.6 | 2.00 ± 0.4 | 53.5 ± 3.8 | 30.2 ± 3.2 |
| 7-HC | 36.2 ± 3.4 | 21.1 ± 3.8 | 2.70 ± 0.3 | 47.8 ± 4.2 | 22.4 ± 4.8 |
| I3C | 34.5 ± 4.2 | 20.4 ± 4.8 | 2.20 ± 0.5 | 55.4 ± 5.1 | 32.2 ± 7.3 |
| SA | 37.4 ± 3.0 | 15.2 ± 3.5 | 1.50 ± 0.3 | 44.8 ± 7.7 | 22.6 ± 5.6 |
| SP | 36.1 ± 4.4 | 18.3 ± 4.4 | 2.1 ± 0.5 | 54.4 ± 1.5 | 30.4 ± 2.9 |
Bacterial adhesion to a surface is a complex process that can be influenced by several factors: physicochemical properties of the cell (hydrophobicity and surface charge), material surface properties and environmental factors (temperature, pH, exposure time, concentration of bacteria, chemical treatment or the presence of antimicrobials and fluid flow conditions) [
40]. Behind these conditions, biological properties of bacteria also influence the attachment to surface, such as: presence of fimbriae and flagella, and the production of EPS [
40].
The polystyrene (PS) microtiter plates are usually used as the standard bioreactor system for adhesion and bacterial biofilm formation. According to the PS physicochemical surface properties, this material was characterized for being hydrophobic and present negative surface charge (∆
GTOT = −44 mJ/m
2; zeta potential = −32 ± 2 mV) [
40]. Therefore, in order to predict the ability of microorganisms to adhere to PS surfaces, the free energy of interaction between the bacterial surface and the PS surface was assessed according to a thermodynamic approach (
Table 4). The thermodynamic theory of adhesion permits the quantification of the free energy of adhesion and predicts the possibility of establishment of an interface between a surface and the microorganism. Analyzing the free energy of adhesion, it is possible to conclude that both
S. aureus and
E. coli have no theoretical thermodynamic capacity to adhere to PS (
![]()
> 0 mJ/m
2). This was significantly reversed when
S. aureus was treated with SA (
![]()
< 0 mJ/m
2). Besides, a decrease in the free energy of adhesion was found for
S. aureus and
E. coli when exposed to 7-HC, and for
S. aureus exposed to SP. An increase in the
![]()
value (less favorable adhesion) was found for
E. coli when exposed to I3C, SA and SP. A similar effect was observed for
S. aureus with I3C, where there was an increase in the free energy of adhesion. These distinct values of
![]()
found after exposure to phytochemicals is apparently related to the distinct physicochemical cell surface properties (
Table 3). The interaction of the different compounds tested with bacterial cells, appears to depend on the bacteria tested and on the molecule used. Several authors explained that increasing the hydrophobicity can cause an increasing extension of adhesion [
46,
47,
48]. Although it is known that hydrophobicity plays an important role in the adhesion phenomena, the results obtained in this work suggest that this phenomenon is not only influenced by physicochemical surface properties and is governed by other factors. Indeed, the thermodynamic theory of adhesion not do consider the electrostatic interactions and biological aspects of adhesion [
44].
Table 4.
Free energy of adhesion (∆GTOTbws) of E. coli and S. aureus to polystyrene (PS), with and without exposure to the phytochemicals.
Table 4.
Free energy of adhesion (∆GTOTbws) of E. coli and S. aureus to polystyrene (PS), with and without exposure to the phytochemicals.
| Strain | Phytochemical | Free Energy of Adhesion— ![]() (mJ/m2) (mJ/m2) |
|---|
| E. coli CECT 434 | Control | 4.4 ± 1.2 |
| 7-HC | 3.5 ± 0.5 |
| I3C | 15.3 ± 3.3 |
| SA | 10.3 ± 2.1 |
| SP | 13.5 ± 2.1 |
| S. aureus CECT 976 | Control | 5.7 ± 1.2 |
| 7-HC | 1.4 ± 0.2 |
| I3C | 6.4 ± 1.2 |
| SA | −3.2 ± 0.4 |
| SP | 5.4 ± 0.5 |
Opposite effects were obtained, when comparing the
![]()
for the Gram-negative (the free energy of adhesion increased), except with 7-HC, and the Gram-positive bacterium (the free energy of adhesion decreased), except with I3C. Therefore, it is expected that I3C, SA and SP can hinder the adhesion of
E. coli to PS. In addition, I3C may also impair the adhesion of
S. aureus. In previous studies [
49,
50,
51,
52] the anti-adhesive properties of some polyphenolics was described. In these studies, the anti-adhesive tests were performed mainly with
Streptococcus mutans using glass [
52] and saliva-coated hydroxyapatite [
49,
50,
51,
52] as adhesion surfaces. Moreover, Borges
et al. [
18,
23], demonstrated that some phenolic acids (gallic and ferulic acids) and GHP (allylisothiocyanate and 2-phenylethilisotiocyanate) reduced the potential of adhesion to PS of some pathogenic bacteria, including
E. coli and
S. aureus. In other work performed by Luis and coworkers [
53], gallic acid was also able to influence the adhesion properties of methicillin-resistant
S. aureus (MRSA) to PS. Similar results were obtained by Lemos
et al. [
54], with SA against
Bacillus cereus.
2.2.2. Cell Motility
Motility is one of the most important features in microbial physiology. Bacteria show different ways of motility. Swimming and swarming motilities are two forms of surface flagella-dependent motility existing in
E. coli [
23,
54,
55]. These types of motility contribute to the virulence of pathogens through adhesion and biofilm formation on biotic and abiotic surfaces [
23].
S. aureus is a non-flagellated bacterium with a kind of motility defined as sliding or colony spreading [
23]. This sliding motility is produced by the expansive forces of a growing colony in combination with reduced surface tension [
23].
The phytochemicals at their MIC were tested for their ability to interfere with swimming and swarming motilities of
E. coli, and sliding motility of
S. aureus. The results obtained are presented in
Table 5.
E. coli showed an increasing in swimming and swarming motilities over time. Swimming and sliding motility were mostly affected when I3C was added (
p < 0.05). However, I3C did not influence swarming motility (24 to 72 h). SA was also able to promote a decrease in swimming and sliding motilities but not swarming motility, despite being very low in the first 24 h, increased in the last 48 h. Probably,
E. coli was able to adapt to SA after a long period of exposure. The application of SP caused an increase in swarming and sliding motilities in the first hours, but after a long period of exposure, both motilities decreased. Finally, 7-HC influenced swarming and swimming motilities but it was not able to change sliding motility. These results may be important because changes in motility can be associated with a reduced ability of bacteria to form biofilms. Likewise, other reports shown that, many natural compounds (extracts or pure products) have capability to interfere with bacterial motility of several microorganisms. Methanolic extracts of
Cuminum cyminum, which contain methyl eugenol, inhibited swimming and swarming motility of
P. aeruginosa,
P. mirabilis and
Serratia marcescens [
56]. Cinnamaldehyde and eugenol from
Cinnamomum cassia affected the biofilm formation of
E. coli, through interference with their swimming motility [
57].
Table 5.
Motility results for bacteria with and without phytochemicals. The drop baseline was 6 mm, which was subtracted from the results presented.
Table 5.
Motility results for bacteria with and without phytochemicals. The drop baseline was 6 mm, which was subtracted from the results presented.
| Time/Phytochemical | E. coli CECT 434 | S. aureus CECT 976 |
|---|
| Swimming (mm) | Swarming (mm) | Sliding (mm) |
|---|
| 24 h | | | |
| Control | 79.0 ± 1.2 | 8.70 ± 0.6 | 7.00 ± 0.0 |
| 7-HC | 7.00 ± 1.0 | 7.70 ± 1.5 | 5.00 ± 0.0 |
| I3C | 4.70 ± 0.6 | 7.70 ± 2.9 | 0.0 ± 0.0 |
| SA | 3.30 ± 0.9 | 2.00 ± 0.9 | 7.70 ± 0.6 |
| SP | 80.0 ± 0.0 | 56.0 ± 2.0 | 84.0 ± 0.0 |
| 48 h | | | |
| Control | 84.7 ± 0.6 | 13.7 ± 3.8 | 8.00 ± 1.0 |
| 7-HC | 43.3 ± 2.9 | 8.70 ± 1.2 | 8.30 ± 0.6 |
| I3C | 0.0 ± 0.0 | 10.0 ± 7.8 | 0.0 ± 0.0 |
| SA | 0.0 ± 0.0 | 55.0 ± 8.7 | 0.0 ± 0.0 |
| SP | 84.0 ± 0.0 | 61.7 ± 9.1 | 56.7 ± 5.8 |
| 72 h | | | |
| Control | 84.0 ± 0.0 | 64.3 ± 7.6 | 7.70 ± 0.6 |
| 7-HC | 51.3 ± 2.3 | 8.30 ± 0.6 | 8.30 ± 0.6 |
| I3C | 0.0 ± 0.0 | 8.70 ± 5.5 | 0.0 ± 0.0 |
| SA | 0.0 ± 0.0 | 54.3 ± 6.4 | 2.00 ± 0.6 |
| SP | 84.0 ± 0.0 | 13.3 ± 3.2 | 55.0 ± 8.7 |
In a recent study performed by Borges
et al. [
23], two phenolic acids (gallic acid and ferulic acid), demonstrated potential to inhibit bacterial motility of four pathogenic bacteria (
E. coli,
P. aeruginosa,
S. aureus and
L. monocytogenes). These authors found similar results with two isothiocyanates (allylisothiocyanate and 2-phenylethylisotiocyanate). In another study, it was found that ferulic acid and SA can inhibit the swimming motility of
Bacillus cereus and
Pseudomonas fluorescens [
23,
54].
A relationship between cells surface motility and biofilm formation has been reported, especially in the case of swarming motility. Both processes, biofilm formation and swarming, require production of flagella and surface polysaccharides [
23]. The major role of swarming motility in biofilm development is to promote initial attachment, probably because the force-generating motion helps to overcome electrostatic repulsive forces between bacterium and the substratum, improving the initial interactions between the two surfaces [
58]. Therefore, this demonstrates their important function on the early stages of biofilm formation [
59,
60]. Several authors have reported mutants with altered swarming motility that made it difficult to form biofilm, concluding that they can play a crucial role in biofilm development [
58,
61].
2.2.3. Quorum-Sensing
QS is a mechanism by which a bacterial population senses its cell density [
62]. This mechanism influences bacterial biofilm growth and development and it is related to cell-cell interactions [
63]. This cell-cell communication system is dependent on several factors: synthesis, exchange and perception of small signal molecules between bacteria [
62]. A disc diffusion assay was performed for QSI screening using the biosensor strain
Chromobacterium violaceum (CV12472).
C. violaceum synthesizes the violet pigment as a result of their autoinducers
N-acyl homoserine lactones (AHLs) based QS systems CviI/CviR (homologs of LuxI/LuxR systems), which sense and responds to autoinducers C6-AHL and C4-AHL [
64]. The phytochemicals were tested as QS inhibitors, at several concentrations.
Table 6 shows the results obtained.
Table 6.
Quorum-sensing results for several phytochemicals at different concentrations.
Table 6.
Quorum-sensing results for several phytochemicals at different concentrations.
| | 7-HC | I3C | SA | SP |
|---|
| Conc. (µg/mL) | QSI pigm. | Inhibition halo (mm) | QS halo (mm) | QSI pigm. | Inhibition halo (mm) | QS halo (mm) | QSI pigm. | Inhibition halo (mm) | QS halo (mm) | QSI pigm. | Inhibition halo (mm) | QS halo (mm) |
| 250 | +/− | 12 | n.d. | + | 11 | n.d. | + | 14 | n.d. | + | 10 | n.d. |
| 500 | +/− | 11 | n.d. | +/− | 11 | 5 | + | 14 | n.d. | + | 11 | n.d. |
| 750 | +/− | 10 | n.d. | +/− | 11 | 6 | + | 19 | n.d. | + | 12 | n.d. |
| 1000 | +/− | 11 | 7 | +/− | 11 | 14 | + | 16 | n.d. | + | 14 | n.d. |
| 1500 | +/− | 11 | 7 | +/− | 12 | 15 | +/− | 14 | 6 | + | 9 | n.d. |
| 2000 | +/− | 10 | 5 | − | 16 | 9 | − | 16 | 8 | + | 10 | n.d. |
| 3000 | +/− | 10 | 8 | − | 20 | 5 | − | 16 | 8 | + | 10 | n.d. |
| 4000 | +/− | 12 | 10 | − | 20 | 11 | − | 15 | 12 | + | 10 | n.d. |
| 5000 | +/− | 11 | 5 | − | 25 | 9 | − | 18 | 9 | + | 11 | n.d. |
The MIC of the four phytochemicals tested against
C. violaceum CV12472 ranged from 25 to 3200 µg/mL. This means that phytochemicals were able to inhibit bacterial growth. QS results showed the effects of the phytochemicals in bacterial growth (inhibition halo) and also the effect of phytochemical in QS, through the detection of pigment inhibition (QS halo). Inhibition of pigment production was detected with some phytochemicals at different concentrations. Of all phytochemicals tested, inhibition of pigment production was detected with 7-HC, I3C and SA with zones of pigment inhibition ranging between 5 to 19 mm. No effect on pigment production was observed with SP at the concentrations tested. The I3C was the most effective to inhibit pigment production. At 500 µg/mL of IC3, inhibition was low, but increasing the concentration, the zone of pigment inhibition also increased. Regarding 7-HC and SA at low concentrations, there was no inhibition of pigment production; but for 1000 and 1500 µg/mL of 7-HC and SA, respectively, QSI halos were detected. Although, SP showed antimicrobial activity, it was not possible to observe an effect on pigment inhibition at the concentrations tested. Regarding QS halos, the concentration of I3C, 7-HC and SA influenced significantly the quantity of pigment production. Therefore, inhibition of QS activity by phytochemicals is concentration dependent, as reported by other authors [
62,
64,
65]. The same authors identified one compound (clove oil) able to inhibit pigment production with 19 mm of pigment inhibition zone against
C. violaceum (CV12472). In addition, cinnamon, peppermint and lavender caused pigment inhibition against the same bacterium [
62]. It has been reported that
Tecoma capensis,
Sonchus oleraceus,
Pityriasis alba,
Pinus nigra,
Jasminum sambac,
Rosmarinus officinalis,
Lavandula angustifolia and
Laurus nobilis are great sources of antimicrobial compounds and QS inhibitors [
66]. Other study demonstrated that isothiocyanates, like allylisothiocyanate, benzylisothiocyanate and 2-phenylethylisothiocyanate, have capacity for QSI by modulation of the activity and synthesis of autoinducers, particularly
N-acyl homoserine lactones (AHLs), interfering with the QS systems of
C. violaceum (CV12472) [
32]. SA has been described as QS inhibitor for diverse bacteria, including
P. aeruginosa [
54,
67].
As QS is an important event that is related with the different steps of bacterial biofilm formation and differentiation, QS inhibitors can be useful in biofilm eradication [
16,
64]. Moreover, QS inhibitors can help to overcome the selective pressure created by antibiotic use [
64]. Therefore, the results obtained in this simple screening assay suggest that the selected phytochemicals can inhibit QS phenomena. In addition, these results reinforce the potential of phytochemical products as QS inhibitors.
The phytochemicals and other compounds that affect QS can act at different levels: inhibition of signal biosynthesis or inhibition of activity of AHL-producing enzymes, enzymatic signal degradation and inhibition of reception signal molecules [
62]. More tests would be necessary in order to be conclusive about the specific mechanisms causing this QSI.
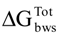 > 0 mJ/m2). This was significantly reversed when S. aureus was treated with SA (
> 0 mJ/m2). This was significantly reversed when S. aureus was treated with SA ( 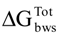 < 0 mJ/m2). Besides, a decrease in the free energy of adhesion was found for S. aureus and E. coli when exposed to 7-HC, and for S. aureus exposed to SP. An increase in the
< 0 mJ/m2). Besides, a decrease in the free energy of adhesion was found for S. aureus and E. coli when exposed to 7-HC, and for S. aureus exposed to SP. An increase in the 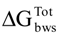 value (less favorable adhesion) was found for E. coli when exposed to I3C, SA and SP. A similar effect was observed for S. aureus with I3C, where there was an increase in the free energy of adhesion. These distinct values of
value (less favorable adhesion) was found for E. coli when exposed to I3C, SA and SP. A similar effect was observed for S. aureus with I3C, where there was an increase in the free energy of adhesion. These distinct values of 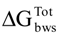 found after exposure to phytochemicals is apparently related to the distinct physicochemical cell surface properties (Table 3). The interaction of the different compounds tested with bacterial cells, appears to depend on the bacteria tested and on the molecule used. Several authors explained that increasing the hydrophobicity can cause an increasing extension of adhesion [46,47,48]. Although it is known that hydrophobicity plays an important role in the adhesion phenomena, the results obtained in this work suggest that this phenomenon is not only influenced by physicochemical surface properties and is governed by other factors. Indeed, the thermodynamic theory of adhesion not do consider the electrostatic interactions and biological aspects of adhesion [44].
found after exposure to phytochemicals is apparently related to the distinct physicochemical cell surface properties (Table 3). The interaction of the different compounds tested with bacterial cells, appears to depend on the bacteria tested and on the molecule used. Several authors explained that increasing the hydrophobicity can cause an increasing extension of adhesion [46,47,48]. Although it is known that hydrophobicity plays an important role in the adhesion phenomena, the results obtained in this work suggest that this phenomenon is not only influenced by physicochemical surface properties and is governed by other factors. Indeed, the thermodynamic theory of adhesion not do consider the electrostatic interactions and biological aspects of adhesion [44].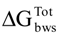 (mJ/m2)
(mJ/m2)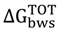 < 0 mJ/m2—thermodynamic favorable adhesion;
< 0 mJ/m2—thermodynamic favorable adhesion; 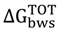 > 0 mJ/m2—thermodynamic unfavorable adhesion.
> 0 mJ/m2—thermodynamic unfavorable adhesion.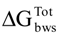 for the Gram-negative (the free energy of adhesion increased), except with 7-HC, and the Gram-positive bacterium (the free energy of adhesion decreased), except with I3C. Therefore, it is expected that I3C, SA and SP can hinder the adhesion of E. coli to PS. In addition, I3C may also impair the adhesion of S. aureus. In previous studies [49,50,51,52] the anti-adhesive properties of some polyphenolics was described. In these studies, the anti-adhesive tests were performed mainly with Streptococcus mutans using glass [52] and saliva-coated hydroxyapatite [49,50,51,52] as adhesion surfaces. Moreover, Borges et al. [18,23], demonstrated that some phenolic acids (gallic and ferulic acids) and GHP (allylisothiocyanate and 2-phenylethilisotiocyanate) reduced the potential of adhesion to PS of some pathogenic bacteria, including E. coli and S. aureus. In other work performed by Luis and coworkers [53], gallic acid was also able to influence the adhesion properties of methicillin-resistant S. aureus (MRSA) to PS. Similar results were obtained by Lemos et al. [54], with SA against Bacillus cereus.
for the Gram-negative (the free energy of adhesion increased), except with 7-HC, and the Gram-positive bacterium (the free energy of adhesion decreased), except with I3C. Therefore, it is expected that I3C, SA and SP can hinder the adhesion of E. coli to PS. In addition, I3C may also impair the adhesion of S. aureus. In previous studies [49,50,51,52] the anti-adhesive properties of some polyphenolics was described. In these studies, the anti-adhesive tests were performed mainly with Streptococcus mutans using glass [52] and saliva-coated hydroxyapatite [49,50,51,52] as adhesion surfaces. Moreover, Borges et al. [18,23], demonstrated that some phenolic acids (gallic and ferulic acids) and GHP (allylisothiocyanate and 2-phenylethilisotiocyanate) reduced the potential of adhesion to PS of some pathogenic bacteria, including E. coli and S. aureus. In other work performed by Luis and coworkers [53], gallic acid was also able to influence the adhesion properties of methicillin-resistant S. aureus (MRSA) to PS. Similar results were obtained by Lemos et al. [54], with SA against Bacillus cereus.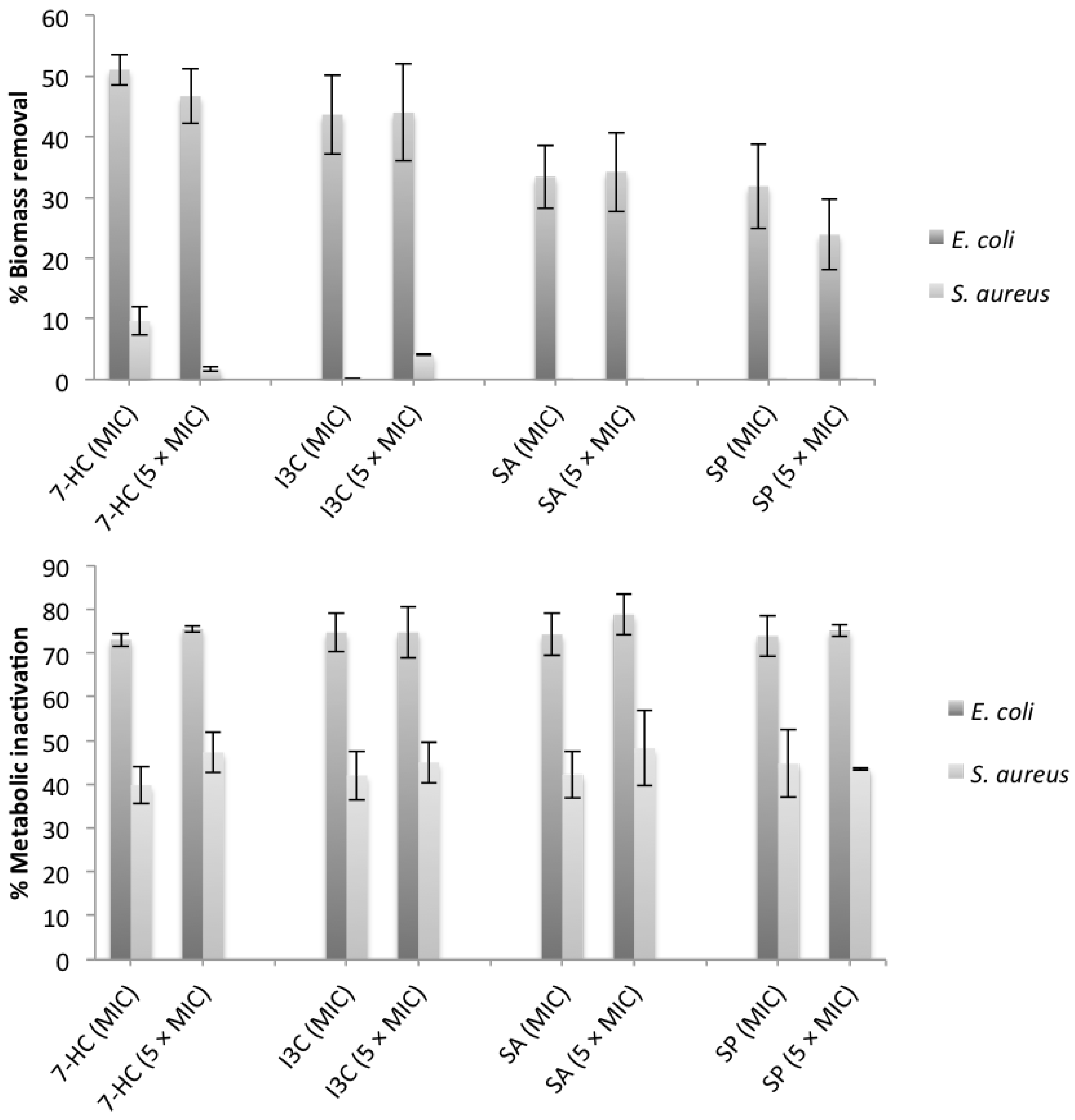

 .
.
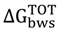 ) is studied by the interaction between one bacteria (b) and a substratum (s) that are immersed or dissolved in water (w) and is expressed by the interfacial tension components:
) is studied by the interaction between one bacteria (b) and a substratum (s) that are immersed or dissolved in water (w) and is expressed by the interfacial tension components:




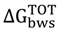 if are negative or positive, respectively [40].
if are negative or positive, respectively [40].








