Multivariate Assessment of Microbiological and Incubation Data from an Experimental Trial Evaluating Essential Oil–Based Sanitizers and Formaldehyde on Hatching Eggs
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Source
2.2. Multivariate Analysis
3. Results and Discussion
3.1. Principal Component Analysis
3.2. Canonical Analysis
3.3. Discriminant Analysis
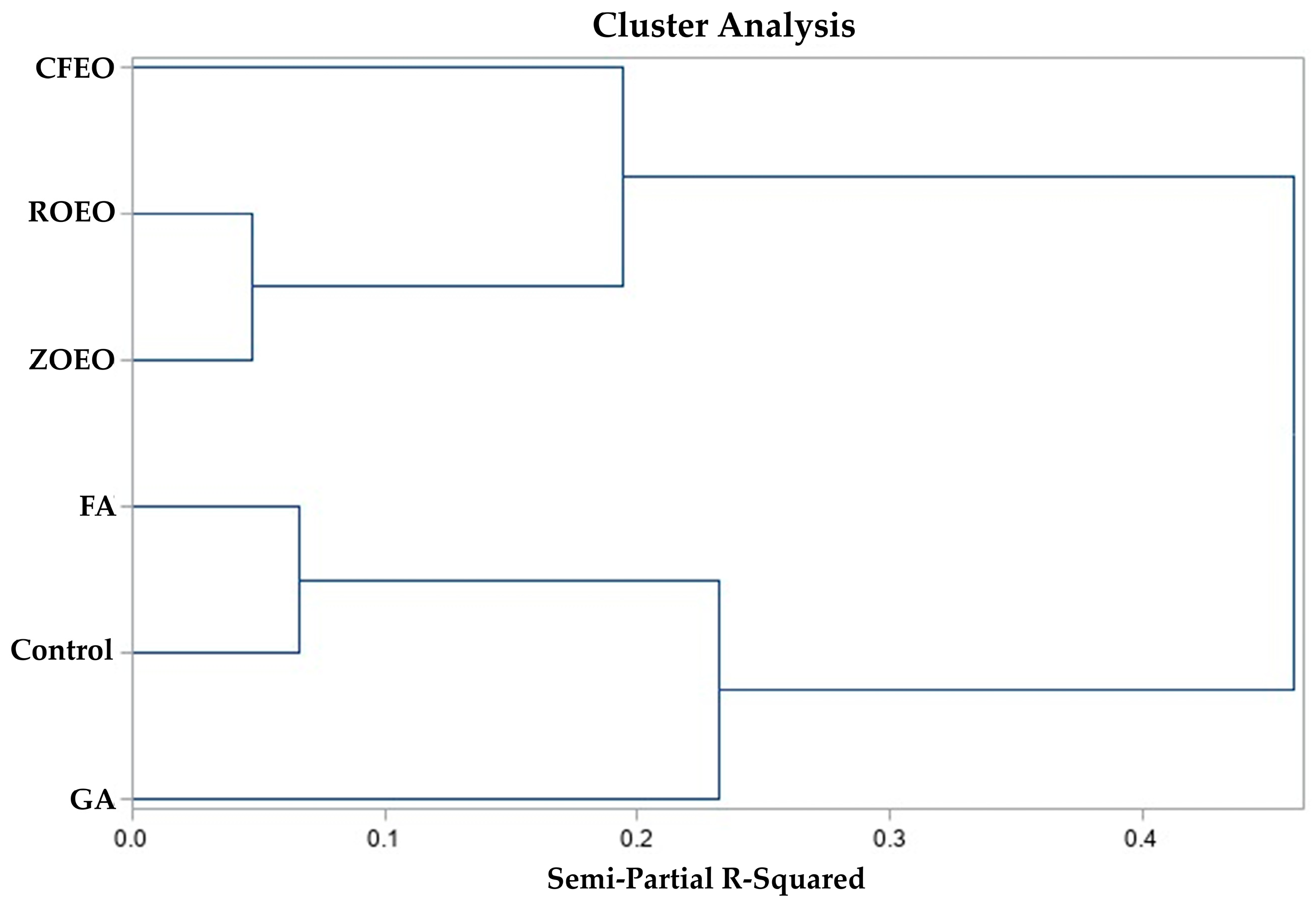
3.4. Hierarchical Cluster Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Oliveira, G.d.S.; McManus, C.; dos Santos, V.M. Multivariate Analysis of Microbiological and Incubation Parameters in Hatching Eggs Sanitized With or Without Essential Oils. Vet. Sci. 2025, 12, 600. [Google Scholar] [CrossRef]
- Melo, E.F.; Clímaco, W.L.S.; Triginelli, M.V.; Vaz, D.P.; de Souza, M.R.; Baião, N.C.; Pompeu, M.A.; Lara, L.J.C. An Evaluation of Alternative Methods for Sanitizing Hatching Eggs. Poult. Sci. 2019, 98, 2466–2473. [Google Scholar] [CrossRef]
- Coulibaly, F.; Onbaşılar, E.E.; Bakır, B.; Sarıçam İnce, S. The effects of using UV light instead of formaldehyde in disinfection of hatching eggs on shell microbial load, embryo development, hatchability, and chick characteristics. Int. J. Environ. Health Res. 2024, 34, 2852–2862. [Google Scholar] [CrossRef]
- Wlazlo, L.; Drabik, K.; Al-Shammari, K.I.A.; Batkowska, J.; Nowakowicz-Debek, B.; Gryzińska, M. Use of Reactive Oxygen Species (Ozone, Hydrogen Peroxide) for Disinfection of Hatching Eggs. Poult. Sci. 2020, 99, 2478–2484. [Google Scholar] [CrossRef]
- Badran, A.M.M.; Osman, A.M.R.; Yassein, D.M.M. Comparative Study of the Effect of Some Disinfectants on Embryonic Mortality, Hatchability, and Some Blood Components. Egypt. Poult. Sci. J. 2018, 38, 1069–1081. [Google Scholar] [CrossRef]
- Batkowska, J.; Al-Shammari, K.I.A.; Lukasz, W.; Nowakowicz-Debek, B.; Gryzinska, M. Evaluation of Propolis Extract as a Disinfectant of Japanese Quail (Coturnix coturnix Japonica) Hatching Eggs. Poult. Sci. 2018, 97, 2372–2377. [Google Scholar] [CrossRef] [PubMed]
- Fouad, W.; Abdelfattah, M.G.; Abdelnabi, M.A. Effect of Spraying Hatching Eggs by Different Levels of Vinegar on Embryological Development, Hatchability and Physiological Performance of Dandarwi Chicks. Egypt. Poult. Sci. J. 2019, 39, 291–309. [Google Scholar] [CrossRef]
- Gholami-Ahangaran, M.; Shahzamani, S.; Yazdkhasti, M. Comparison of Virkon S and Formaldehyde on Hatchability and Survival Rate of Chicks in Disinfection of Fertile Eggs. Rev. Med. Vet. 2016, 167, 45–49. [Google Scholar]
- Hrnčár, C.; Hanusová, E.; Hanus, A.; Arpášová, H.; Kokoszyński, D.; Bujko, J. The Effect of Various Disinfectants on Hatching Results in Chickens. Sci. Pap. Anim. Sci. Biotechnol. 2021, 54, 193–196. [Google Scholar]
- El-Soufi, A.; Al Khatib, A.; Khazaal, S.; El Darra, N.; Raafat, K. Evaluation of essential oils as natural antibacterial agents for eggshell sanitization and quality preservation. Processes 2025, 13, 224. [Google Scholar] [CrossRef]
- Soares, C.E.D.S.; Leite, C.E.C.; Dahlke, F.; Maiorka, A.; Miotto, M.; Scussel, V.; Lindner, J.D.D. Antifungal Action of Ozone on Chicken Eggshell Cuticles: A Preliminary Study. Ozone Sci. Eng. 2022, 44, 407–412. [Google Scholar] [CrossRef]
- Oliveira, G.D.S.; McManus, C.; Santos, P.H.G.D.S.; de Sousa, D.E.R.; Jivago, J.L.D.P.R.; de Castro, M.B.; dos Santos, V.M. Hatching egg sanitizers based on essential oils: Microbiological parameters, hatchability, and poultry health. Antibiotics 2024, 13, 1066. [Google Scholar] [CrossRef] [PubMed]
- Touazi, L.; Aberbour, A.; Djenadi, K.; Benberkane, A.; Aissanou, S.; Boumenir, M.; Iguer-Ouada, M.; Hornick, J.-L.; Moula, N. Rosemary Essential Oil as a Natural Disinfectant in Japanese Quail Eggs (Coturnix coturnix Japonica): Effect on the Bacterial Load and Hatchability Characteristics. Int. J. Vet. Sci. Med. 2025, 13, 1. [Google Scholar]
- Noori, S.; Zeynali, F.; Almasi, H. Antimicrobial and antioxidant efficiency of nanoemulsion-based edible coating containing ginger (Zingiber officinale) essential oil and its effect on safety and quality attributes of chicken breast fillets. Food Control 2018, 84, 312–320. [Google Scholar] [CrossRef]
- Oliveira, G.D.S.; McManus, C.; Pires, P.G.D.S.; dos Santos, V.M. Combination of Cassava Starch Biopolymer and Essential Oils for Coating Table Eggs. Front. Sustain. Food Syst. 2022, 6, 957229. [Google Scholar] [CrossRef]
- Hosseinzadeh, S.; Shariatmadari, F.; Karimi Torshizi, M.A.; Ahmadi, H.; Scholey, D. Plectranthus amboinicus and rosemary (Rosmarinus officinalis L.) essential oils effects on performance, antioxidant activity, intestinal health, immune response, and plasma biochemistry in broiler chickens. Food Sci. Nutr. 2023, 11, 3939–3948. [Google Scholar] [CrossRef] [PubMed]
- dos Santos, V.M.; Oliveira, G.D.S.; Santos, P.H.G.D.S.; Cerqueira, L.D.A.; Jivago, J.L.D.P.R.; Paixão, S.S.R.M.; de Castro, M.B.; McManus, C. Measuring Contamination Levels and Incubation Results of Hatching Eggs Sanitized with Essential Oils. Antibiotics 2025, 14, 1076. [Google Scholar] [CrossRef]
- El-Kashef, M.M.A.; El Sabry, M.I. Egg sanitation with ginger and garlic solutions affects embryonic development, hatchability, blood parameters and post-hatch performance of Japanese quail. Trop. Anim. Health Prod. 2025, 57, 338. [Google Scholar] [CrossRef]
- Batkowska, J.; Al-Shammari, K.I.A.; Gryzinska, M.M.; Brodacki, A.; Wlazlo, L.; Nowakowicz-Debek, B. Effect of Using Colloidal Silver in the Disinfection of Hatching Eggs on Some Microbial, Hatchability and Performance Traits in Japanese Quail (Coturnix Cot. Japonica). Eur. Poult. Sci. 2017, 81, 211. [Google Scholar] [CrossRef]
- Fouad, W.; Abdel-Hafez, M.S. Effect of Spraying Hatching Eggs of Japanese Quails by Live Yeast on Physiological Changes in the Embryonic Development, Hatchability and Total Bacterial Count. Egypt. Poult. Sci. J. 2017, 37, 1303–1321. [Google Scholar] [CrossRef]
- Bekhet, G.; Khalifa, A.Y.Z. Essential Oil Sanitizers to Sanitize Hatching Eggs. J. Appl. Anim. Res. 2022, 50, 695–701. [Google Scholar] [CrossRef]
- Zeweil, H.S.; Rizk, R.E.; Bekhet, G.M.; Ahmed, M.R. Comparing the Effectiveness of Egg Disinfectants against Bacteria and Mitotic Indices of Developing Chick Embryos. J. Basic Appl. Zool. 2015, 70, 1–15. [Google Scholar] [CrossRef]
- Iraqi, E.E.; El-Sahn, A.A.; El-Barbary, A.M.; Ahmed, M.M.; Elkomy, A.E. Antimicrobial activity of tea tree and lavender essential oils and their effects on hatching performance and eggshell bacterial count of Japanese quail eggs. BMC Vet. Res. 2025, 21, 176. [Google Scholar] [CrossRef] [PubMed]
- Messens, W.; Grijspeerdt, K.; De Reu, K.; De Ketelaere, B.; Mertens, K.; Bamelis, F.; Kemps, B.; De Baerdemaeker, J.; Decuypere, E.; Herman, L. Eggshell penetration of various types of hens’ eggs by Salmonella enterica serovar Enteritidis. J. Food Prot. 2007, 70, 623–628. [Google Scholar] [CrossRef] [PubMed]
- Falleh, H. Demystifying the power of essential oils: A review of their antibacterial properties and potential as natural food preservatives. EXCLI J. 2025, 24, 828–850. [Google Scholar] [PubMed]
- McDonnell, G.; Russell, A.D. Antiseptics and Disinfectants: Activity, Action, and Resistance. Clin. Microbiol. Rev. 1999, 12, 147–179. [Google Scholar] [CrossRef]
- Valverde-Santiago, M.; Pontel, L.B. Emerging Mechanisms Underlying Formaldehyde Toxicity and Response. Mol. Cell 2025, 85, 2068–2079. [Google Scholar] [CrossRef]



| Variables | Treatments | ||||||
|---|---|---|---|---|---|---|---|
| Control | GA | FA | ZOEO | CFEO | ROEO | p Value | |
| EWL (%) | 12.87 ± 0.95 | 11.59 ± 0.18 | 12.82 ± 0.61 | 12.24 ± 0.61 | 12.77 ± 1.07 | 12.34 ± 0.43 | ns |
| CW (g) | 45.97 ± 0.67 ab | 46.28 ± 0.92 a | 44.94 ± 0.42 b | 46.32 ± 0.33 a | 46.50 ± 0.57 a | 46.14 ± 0.31 ab | 0.0156 |
| HI (%) | 90.36 ± 4.88 | 92.02 ± 3.30 | 90.29 ± 2.28 | 93.48 ± 3.60 | 93.16 ± 1.06 | 94.66 ± 2.04 | ns |
| EM (%) | 2.98 ± 2.06 | 1.79 ± 1.54 | 3.27 ± 1.14 | 1.79 ± 1.19 | 1.49 ± 1.14 | 1.79 ± 2.06 | ns |
| IM (%) | 0.00 ± 0.00 | 0.30 ± 0.60 | 0.30 ± 0.60 | 0.60 ± 1.19 | 0.30 ± 0.60 | 0.00 ± 0.00 | ns |
| LM (%) | 3.57 ± 5.05 | 5.36 ± 2.06 | 4.17 ± 2.06 | 3.27 ± 2.03 | 3.57 ± 1.68 | 2.98 ± 1.54 | ns |
| CE (%) | 2.08 ± 2.45 | 0.00 ± 0.00 | 1.19 ± 1.37 | 0.30 ± 0.60 | 0.60 ± 1.19 | 0.00 ± 0.00 | ns |
| EGM (log10 CFU/mL) | 1.60 ± 0.30 a | 1.13 ± 0.25 a | <1 b | <1 b | <1 b | <1 b | <0.0001 |
| YSM (log10 CFU/mL) | 2.58 ± 0.39 a | 1.66 ± 0.48 ab | 1.99 ± 0.10 ab | 1.06 ± 0.20 b | 1.13 ± 0.99 b | 1.00 ± 0.35 b | 0.0137 |
| CW | HI | EM | IM | LM | CE | EGM | YSM | |
|---|---|---|---|---|---|---|---|---|
| EWL | −0.23 | −0.39 | 0.17 | 0.13 | 0.09 | 0.34 | −0.08 | −0.29 |
| CW | −0.02 | −0.29 | 0.10 | 0.19 | −0.04 | −0.01 | −0.24 | |
| HI | −0.52 * | −0.29 | −0.57 * | −0.41 * | −0.06 | −0.34 | ||
| EM | 0.12 | −0.22 | 0.39 | 0.06 | −0.01 | |||
| IM | −0.00 | 0.08 | −0.26 | −0.11 | ||||
| LM | −0.34 | 0.03 | 0.29 | |||||
| CE | 0.12 | 0.21 | ||||||
| EGM | 0.45 |
| Variables | R2 Partial | p < F | p < Lambda | p < ASCC |
|---|---|---|---|---|
| EGM | 0.8464 | 0.0002 | 0.0002 | 0.0002 |
| CW | 0.6440 | 0.0263 | <0.0001 | 0.0014 |
| YSM | 0.5348 | 0.1230 | <0.0001 | 0.0050 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
dos Santos, V.M.; Oliveira, G.d.S.; McManus, C. Multivariate Assessment of Microbiological and Incubation Data from an Experimental Trial Evaluating Essential Oil–Based Sanitizers and Formaldehyde on Hatching Eggs. Pathogens 2026, 15, 426. https://doi.org/10.3390/pathogens15040426
dos Santos VM, Oliveira GdS, McManus C. Multivariate Assessment of Microbiological and Incubation Data from an Experimental Trial Evaluating Essential Oil–Based Sanitizers and Formaldehyde on Hatching Eggs. Pathogens. 2026; 15(4):426. https://doi.org/10.3390/pathogens15040426
Chicago/Turabian Styledos Santos, Vinícius Machado, Gabriel da Silva Oliveira, and Concepta McManus. 2026. "Multivariate Assessment of Microbiological and Incubation Data from an Experimental Trial Evaluating Essential Oil–Based Sanitizers and Formaldehyde on Hatching Eggs" Pathogens 15, no. 4: 426. https://doi.org/10.3390/pathogens15040426
APA Styledos Santos, V. M., Oliveira, G. d. S., & McManus, C. (2026). Multivariate Assessment of Microbiological and Incubation Data from an Experimental Trial Evaluating Essential Oil–Based Sanitizers and Formaldehyde on Hatching Eggs. Pathogens, 15(4), 426. https://doi.org/10.3390/pathogens15040426







