CMGC Kinases in Viral Infection and Human Disease
Abstract
1. Introduction
1.1. Structural and Functional Organization of the CMGC Kinases
1.1.1. Cyclin-Dependent Kinases (CDKs)
1.1.2. Dual-Specificity Tyrosine Phosphorylation-Regulated Kinases (DYRKs)
1.1.3. Mitogen-Activated Protein Kinases (MAPKs)
1.1.4. Glycogen Synthase Kinases (GSKs)
1.1.5. CDC-like Kinases (CLKs)
1.1.6. Serine/Arginine-Rich Protein Kinases (SRPKs)
1.1.7. ROS-Cross-Hybridizing Kinases (RCKs)
1.1.8. CDK-like Kinases (CDKL)
1.2. CMGC Signaling and Cross-Talk Among CMGC Kinases
1.3. CMGC Kinases in Viral Infections
1.4. CMGCs and DNA Viruses
1.5. CMGC Kinases and RNA Viruses
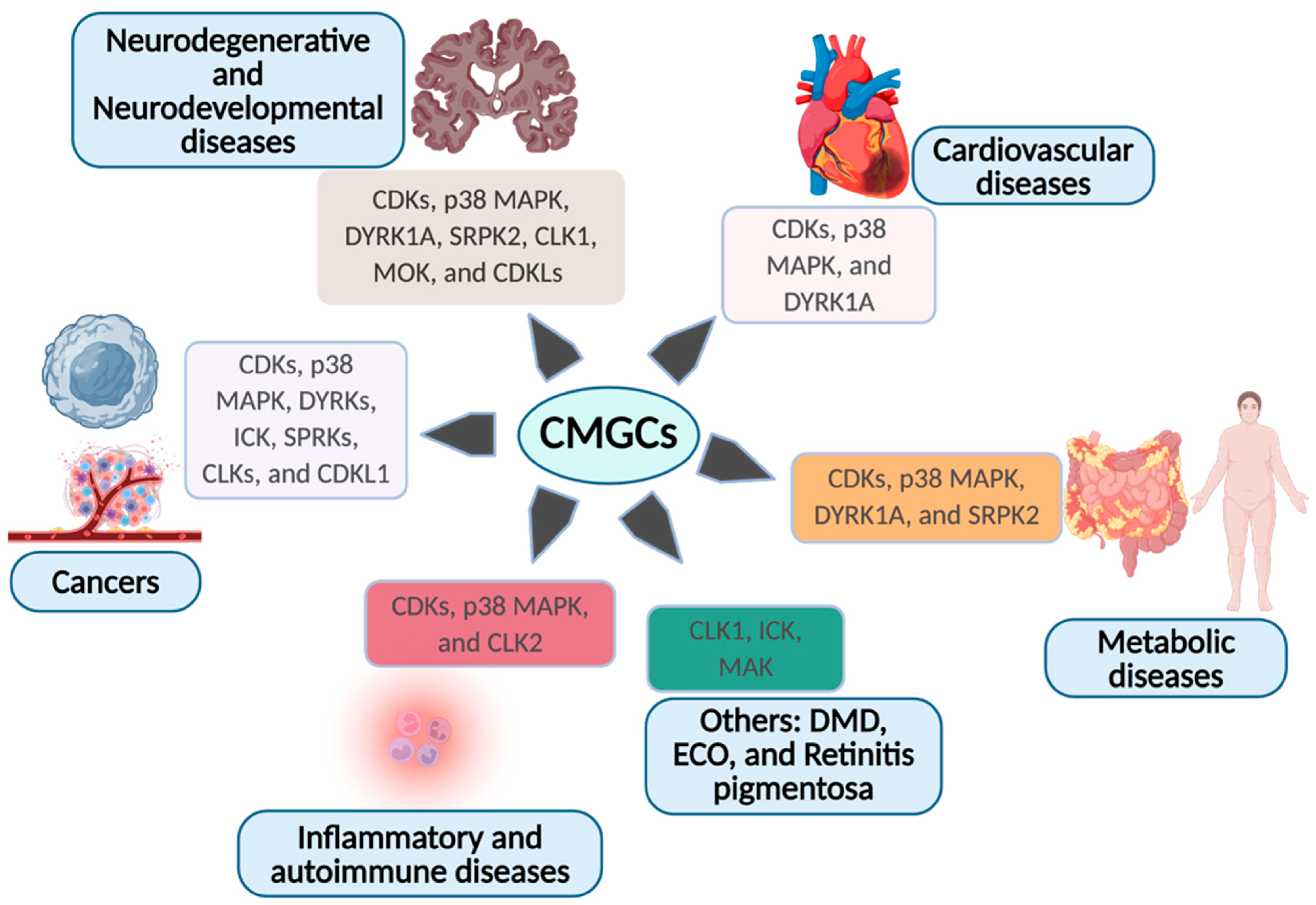
1.6. CMGCs in Other Human Diseases
1.6.1. CDKs: Context-Dependent Oncogenes and Systemic Regulators
1.6.2. p38 MAPK and DYRKs: Stress, Degeneration, and Metabolism
1.6.3. RNA Processing Kinases: SRPKs and CLKs
1.6.4. RCKs and CDKLs: Emerging Disease Axes
2. Conclusions and Future Perspectives
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AKT | Protein kinase B |
| ASK1 | Apoptosis signal-regulating kinase 1 |
| ATF2 | Activating transcription factor 2 |
| Bax | Bcl-2-associated X protein |
| CDKi | Cyclin-dependent kinase inhibitor |
| CHIKV | Chikungunya virus |
| CHOP | C/EBP homologous protein |
| CREB | cAMP response element-binding protein |
| CTD | C-terminal domain |
| DENV | Dengue virus |
| DLK | Dual leucine zipper kinase |
| DREAM | DP, RB-like, E2F, and MuvB complex |
| E2F | E2F transcription factor |
| EBV | Epstein–Barr virus |
| EGFR | Epidermal growth factor receptor |
| GPCR | G protein-coupled receptor |
| HBV | Hepatitis B virus |
| HBeAg | Hepatitis B e antigen |
| HCMV | Human cytomegalovirus |
| HCV | Hepatitis C virus |
| HIV | Human immunodeficiency virus |
| HPV | Human papillomavirus |
| HSV | Herpes simplex virus |
| HTLV | Human T-cell leukemia virus |
| IAV | Influenza A virus |
| ICK | Intestinal Cell Kinase, recently renamed CILK1 |
| IRF3 | Interferon regulatory factor 3 |
| JEV | Japanese encephalitis virus |
| JNK | c-Jun N-terminal kinase |
| KSHV | Kaposi’s sarcoma-associated herpesvirus |
| LRP | Low-density lipoprotein receptor-related protein |
| MAK | male germ cell-associated kinase |
| MAP2K | Mitogen-activated protein kinase kinase |
| MAP3K | Mitogen-activated protein kinase kinase kinase |
| MEK | MAPK/ERK kinase |
| MEKK3 | MEK kinase 3 |
| MEKK4 | MEK kinase 4 |
| MHV | Mouse hepatitis virus |
| MK2/3 | MAPK-activated protein kinase 2/3 |
| Mnk1/2 | MAPK-interacting kinase 1/2 |
| MOK | MAPK/MAK/MRK-overlapping kinase |
| NAPA | N-terminal autophosphorylation accessory region |
| NF-κB | Nuclear factor kappa-light-chain-enhancer of activated B cells |
| NLK | Nemo-like kinase |
| PEDV | Porcine epidemic diarrhea virus |
| PEST | Proline, glutamate, serine, threonine-rich sequence |
| PI3K | Phosphoinositide 3-kinase |
| PPAR | Peroxisome proliferator-activated receptor |
| RAF | Rapidly accelerated fibrosarcoma kinase |
| RAS | Rat sarcoma small GTPase |
| RS domain | Arginine-serine-rich domain |
| RSV | Respiratory syncytial virus |
| RTKs | Receptor tyrosine kinases |
| Rb | Retinoblastoma protein |
| RdRp | RNA-dependent RNA polymerase |
| SAMHD1 | SAM domain and HD domain-containing protein 1 |
| SHVV | Severe acute respiratory syndrome coronavirus 2 |
| STE20 | Serine-glutamate-glycine motif |
| STAT3 | Signal transducer and activator of transcription 3 |
| TAB1 | TGF-β-activated kinase 1-binding protein 1 |
| TBEV | Tick-borne encephalitis virus |
| TCR | T-cell receptor |
| TDY motif | Threonine-aspartate-tyrosine motif |
| TEY motif | Threonine-glutamate-tyrosine motif |
| TQE motif | Threonine-glutamine-glutamate motif |
| TRAF | TNF receptor-associated factor |
| TXY motif | Threonine-any amino acid-tyrosine motif |
| VEGFR | Vascular endothelial growth factor receptor |
| VZV | Varicella-zoster virus |
| ZAP70 | Zeta-chain-associated protein kinase 70 |
| ZIKV | Zika virus |
| cMET | Mesenchymal–epithelial transition factor |
| cccDNA | Covalently closed circular DNA |
| dNTP | Deoxynucleotide triphosphate |
| eIF2B | Eukaryotic initiation factor 2B |
| mTOR | Mechanistic target of rapamycin |
| pgRNA | Pregenomic RNA |
| STE20 | Sterile 20 Kinase |
References
- Cohen, P. The role of protein phosphorylation in human health and disease. The Sir Hans Krebs Medal Lecture. Eur. J. Biochem. 2001, 268, 5001–5010. [Google Scholar]
- Manning, G.; Whyte, D.B.; Martinez, R.; Hunter, T.; Sudarsanam, S. The protein kinase complement of the human genome. Science 2002, 298, 1912–1934. [Google Scholar] [CrossRef]
- Aranda, S.; Laguna, A.; Luna, S.d.l. DYRK family of protein kinases: Evolutionary relationships, biochemical properties, and functional roles. FASEB J. 2011, 25, 449–462. [Google Scholar] [CrossRef]
- Li, C.; Liu, Y.; Luo, S.; Yang, M.; Li, L.; Sun, L. A review of CDKL: An underestimated protein kinase family. Int. J. Biol. Macromol. 2024, 277, 133604. [Google Scholar] [CrossRef] [PubMed]
- Kannan, N.; Neuwald, A.F. Evolutionary constraints associated with functional specificity of the CMGC protein kinases MAPK, CDK, GSK, SRPK, DYRK, and CK2α. Protein Sci. 2004, 13, 2059–2077. [Google Scholar] [CrossRef] [PubMed]
- Malumbres, M. Cyclin-dependent kinases. Genome Biol. 2014, 15, 122. [Google Scholar] [CrossRef]
- Lorca, T.; Labbé, J.C.; Devault, A.; Fesquet, D.; Capony, J.P.; Cavadore, J.C.; Le Bouffant, F.; Dorée, M. Dephosphorylation of cdc2 on threonine 161 is required for cdc2 kinase inactivation and normal anaphase. EMBO J. 1992, 11, 2381–2390. [Google Scholar] [CrossRef]
- Poon, R.Y.; Hunter, T. Dephosphorylation of Cdk2 Thr160 by the cyclin-dependent kinase-interacting phosphatase KAP in the absence of cyclin. Science 1995, 270, 90–93. [Google Scholar] [CrossRef]
- Yang, J.; Kornbluth, S. All aboard the cyclin train: Subcellular trafficking of cyclins and their CDK partners. Trends Cell Biol. 1999, 9, 207–210. [Google Scholar] [CrossRef]
- Veiga-Fernandes, H.; Rocha, B. High expression of active CDK6 in the cytoplasm of CD8 memory cells favors rapid division. Nat. Immunol. 2004, 5, 31–37. [Google Scholar] [CrossRef]
- An, X.; Ng, S.S.; Xie, D.; Zeng, Y.-X.; Sze, J.; Wang, J.; Chen, Y.C.; Chow, B.K.; Lu, G.; Poon, W.S. Functional characterisation of cell cycle-related kinase (CCRK) in colorectal cancer carcinogenesis. Eur. J. Cancer 2010, 46, 1752–1761. [Google Scholar] [CrossRef]
- Boni, J.; Rubio-Perez, C.; López-Bigas, N.; Fillat, C.; de la Luna, S. The DYRK family of kinases in cancer: Molecular functions and therapeutic opportunities. Cancers 2020, 12, 2106. [Google Scholar] [CrossRef]
- Laham, A.J.; Saber-Ayad, M.; El-Awady, R. DYRK1A: A down syndrome-related dual protein kinase with a versatile role in tumorigenesis. Cell. Mol. Life Sci. 2021, 78, 603–619. [Google Scholar] [CrossRef]
- Qile, M.; Ji, Y.; Houtman, M.J.; Veldhuis, M.; Romunde, F.; Kok, B.; Van der Heyden, M.A. Identification of a PEST sequence in vertebrate KIR2. 1 that modifies rectification. Front. Physiol. 2019, 10, 863. [Google Scholar] [CrossRef] [PubMed]
- Kinstrie, R.; Luebbering, N.; Miranda-Saavedra, D.; Sibbet, G.; Han, J.; Lochhead, P.A.; Cleghon, V. Characterization of a domain that transiently converts class 2 DYRKs into intramolecular tyrosine kinases. Sci. Signal. 2010, 3, ra16. [Google Scholar] [CrossRef]
- Singh, R.; Lauth, M. Emerging roles of DYRK kinases in embryogenesis and hedgehog pathway control. J. Dev. Biol. 2017, 5, 13. [Google Scholar] [CrossRef]
- Wippich, F.; Bodenmiller, B.; Trajkovska, M.G.; Wanka, S.; Aebersold, R.; Pelkmans, L. Dual Specificity Kinase DYRK3 Couples Stress Granule Condensation/Dissolution to mTORC1 Signaling. Cell 2013, 152, 791–805. [Google Scholar] [CrossRef]
- Ng, G.Y.Q.; Loh, Z.W.; Fann, D.Y.; Mallilankaraman, K.; Arumugam, T.V.; Hande, M.P. Role of Mitogen-Activated Protein (MAP) Kinase Pathways in Metabolic Diseases. Genome Integr. 2024, 15, e20230003. [Google Scholar] [CrossRef]
- Elmadbouh, O.H.M.; Pandol, S.J.; Edderkaoui, M. Glycogen Synthase Kinase 3β: A True Foe in Pancreatic Cancer. Int. J. Mol. Sci. 2022, 23, 14133. [Google Scholar] [CrossRef] [PubMed]
- Nagini, S.; Sophia, J.; Mishra, R. Glycogen synthase kinases: Moonlighting proteins with theranostic potential in cancer. Semin. Cancer Biol. 2019, 56, 25–36. [Google Scholar] [CrossRef] [PubMed]
- Beurel, E.; Grieco, S.F.; Jope, R.S. Glycogen synthase kinase-3 (GSK3): Regulation, actions, and diseases. Pharmacol. Ther. 2015, 148, 114–131. [Google Scholar] [CrossRef]
- Shin, S.H.; Lee, E.J.; Chun, J.; Hyun, S.; Kim, Y.I.; Kang, S.S. The nuclear localization of glycogen synthase kinase 3β is required its putative PY-nuclear localization sequences. Mol. Cells 2012, 34, 375–382. [Google Scholar] [CrossRef]
- Soniat, M.; Chook, Y.M. Nuclear localization signals for four distinct karyopherin-β nuclear import systems. Biochem. J. 2015, 468, 353–362. [Google Scholar] [CrossRef]
- Azoulay-Alfaguter, I.; Yaffe, Y.; Licht-Murava, A.; Urbanska, M.; Jaworski, J.; Pietrokovski, S.; Hirschberg, K.; Eldar-Finkelman, H. Distinct Molecular Regulation of Glycogen Synthase Kinase-3α Isozyme Controlled by Its N-terminal Region: FUNCTIONAL ROLE IN CALCIUM/CALPAIN SIGNALING. J. Biol. Chem. 2011, 286, 13470–13480. [Google Scholar] [CrossRef]
- Song, M.; Pang, L.; Zhang, M.; Qu, Y.; Laster, K.V.; Dong, Z. Cdc2-like kinases: Structure, biological function and therapeutic targets for diseases. Signal Transduct. Target. Ther. 2023, 8, 148. [Google Scholar] [CrossRef] [PubMed]
- Jiang, K.; Patel, N.A.; Watson, J.E.; Apostolatos, H.; Kleiman, E.; Hanson, O.; Hagiwara, M.; Cooper, D.R. Akt2 regulation of Cdc2-like kinases (Clk/Sty), serine/arginine-rich (SR) protein phosphorylation, and insulin-induced alternative splicing of PKCβII messenger ribonucleic acid. Endocrinology 2009, 150, 2087–2097. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Carter, G.; Romero, J.; Gower, K.M.; Watson, J.; Patel, N.A.; Cooper, D.R. Clk/STY (cdc2-like kinase 1) and Akt regulate alternative splicing and adipogenesis in 3T3-L1 pre-adipocytes. PLoS ONE 2013, 8, e53268. [Google Scholar] [CrossRef] [PubMed]
- ElHady, A.K.; Abdel-Halim, M.; Abadi, A.H.; Engel, M. Development of selective Clk1 and-4 inhibitors for cellular depletion of cancer-relevant proteins. J. Med. Chem. 2017, 60, 5377–5391. [Google Scholar] [CrossRef]
- Bullock, A.N.; Das, S.; Debreczeni, J.E.; Rellos, P.; Fedorov, O.; Niesen, F.H.; Guo, K.; Papagrigoriou, E.; Amos, A.L.; Cho, S. Kinase domain insertions define distinct roles of CLK kinases in SR protein phosphorylation. Structure 2009, 17, 352–362. [Google Scholar] [CrossRef]
- Zahler, A.M.; Lane, W.S.; Stolk, J.A.; Roth, M.B. SR proteins: A conserved family of pre-mRNA splicing factors. Genes Dev. 1992, 6, 837–847. [Google Scholar] [CrossRef]
- Ding, J.-H.; Zhong, X.-Y.; Hagopian, J.C.; Cruz, M.M.; Ghosh, G.; Feramisco, J.; Adams, J.A.; Fu, X.-D. Regulated Cellular Partitioning of SR Protein-specific Kinases in Mammalian Cells. Mol. Biol. Cell 2006, 17, 876–885. [Google Scholar] [CrossRef] [PubMed]
- Fu, Z.; Schroeder, M.J.; Shabanowitz, J.; Kaldis, P.; Togawa, K.; Rustgi, A.K.; Hunt, D.F.; Sturgill, T.W. Activation of a Nuclear Cdc2-Related Kinase within a Mitogen-Activated Protein Kinase-Like TDY Motif by Autophosphorylation and Cyclin-Dependent Protein Kinase-Activating Kinase. Mol. Cell. Biol. 2005, 25, 6047–6064. [Google Scholar] [CrossRef]
- Wang, L.Y.; Kung, H.J. Male germ cell-associated kinase is overexpressed in prostate cancer cells and causes mitotic defects via deregulation of APC/CCDH1. Oncogene 2012, 31, 2907–2918. [Google Scholar] [CrossRef]
- Canovas, B.; Nebreda, A.R. Diversity and versatility of p38 kinase signalling in health and disease. Nat. Rev. Mol. Cell Biol. 2021, 22, 346–366. [Google Scholar] [CrossRef]
- De Nicola, G.F.; Martin, E.D.; Chaikuad, A.; Bassi, R.; Clark, J.; Martino, L.; Verma, S.; Sicard, P.; Tata, R.; Atkinson, R.A. Mechanism and consequence of the autoactivation of p38α mitogen-activated protein kinase promoted by TAB1. Nat. Struct. Mol. Biol. 2013, 20, 1182–1190. [Google Scholar] [CrossRef]
- Salvador, J.M.; Mittelstadt, P.R.; Guszczynski, T.; Copeland, T.D.; Yamaguchi, H.; Appella, E.; Fornace, A.J., Jr.; Ashwell, J.D. Alternative p38 activation pathway mediated by T cell receptor–proximal tyrosine kinases. Nat. Immunol. 2005, 6, 390–395. [Google Scholar] [CrossRef]
- Ma, Y.-T.; Li, C.; Shen, Y.; You, W.-H.; Han, M.-X.; Mu, Y.-F.; Han, F.-J. Mechanisms of the JNK/p38 MAPK signaling pathway in drug resistance in ovarian cancer. Front. Oncol. 2025, 15, 1533352. [Google Scholar] [CrossRef]
- Whitaker, R.H.; Cook, J.G. Stress Relief Techniques: P38 MAPK Determines the Balance of Cell Cycle and Apoptosis Pathways. Biomolecules 2021, 11, 1444. [Google Scholar] [CrossRef]
- Casanovas, O.; Miró, F.; Estanyol, J.M.; Itarte, E.; Agell, N.; Bachs, O. Osmotic stress regulates the stability of cyclin D1 in a p38SAPK2-dependent manner. J. Biol. Chem. 2000, 275, 35091–35097. [Google Scholar] [CrossRef] [PubMed]
- Thornton, T.M.; Rincon, M. Non-classical p38 map kinase functions: Cell cycle checkpoints and survival. Int. J. Biol. Sci. 2008, 5, 44. [Google Scholar] [CrossRef]
- Gubern, A.; Joaquin, M.; Marquès, M.; Maseres, P.; Garcia-Garcia, J.; Amat, R.; González-Nuñez, D.; Oliva, B.; Real, F.X.; de Nadal, E. The N-terminal phosphorylation of RB by p38 bypasses its inactivation by CDKs and prevents proliferation in cancer cells. Mol. Cell 2016, 64, 25–36. [Google Scholar] [CrossRef] [PubMed]
- Bulavin, D.V.; Amundson, S.A.; Fornace, A.J., Jr. p38 and Chk1 kinases: Different conductors for the G2/M checkpoint symphony. Curr. Opin. Genet. Dev. 2002, 12, 92–97. [Google Scholar] [CrossRef]
- Sequeira, R.C.; Godad, A. Understanding glycogen synthase kinase-3: A novel avenue for Alzheimer’s disease. Mol. Neurobiol. 2024, 61, 4203–4221. [Google Scholar] [PubMed]
- Maurer, U.; Preiss, F.; Brauns-Schubert, P.; Schlicher, L.; Charvet, C. GSK-3–at the crossroads of cell death and survival. J. Cell Sci. 2014, 127, 1369–1378. [Google Scholar] [CrossRef]
- Pap, M.; Cooper, G.M. Role of glycogen synthase kinase-3 in the phosphatidylinositol 3-Kinase/Akt cell survival pathway. J. Biol. Chem. 1998, 273, 19929–19932. [Google Scholar] [CrossRef]
- Bijur, G.N.; Davis, R.E.; Jope, R.S. Rapid activation of heat shock factor-1 DNA binding by H2O2 and modulation by glutathione in human neuroblastoma and Alzheimer’s disease cybrid cells. Mol. Brain Res. 1999, 71, 69–77. [Google Scholar]
- Yoshida, S.; Yoshida, K. New insights into the roles for DYRK family in mammalian development and congenital diseases. Genes Dis. 2023, 10, 758–770. [Google Scholar] [CrossRef]
- Ashford, A.L.; Oxley, D.; Kettle, J.; Hudson, K.; Guichard, S.; Cook, S.J.; Lochhead, P.A. A novel DYRK1B inhibitor AZ191 demonstrates that DYRK1B acts independently of GSK3β to phosphorylate cyclin D1 at Thr286, not Thr288. Biochem. J. 2014, 457, 43–56. [Google Scholar] [CrossRef]
- Litovchick, L.; Florens, L.A.; Swanson, S.K.; Washburn, M.P.; DeCaprio, J.A. DYRK1A protein kinase promotes quiescence and senescence through DREAM complex assembly. Genes Dev. 2011, 25, 801–813. [Google Scholar] [CrossRef]
- Mercer, S.E.; Ewton, D.Z.; Deng, X.; Lim, S.; Mazur, T.R.; Friedman, E. Mirk/Dyrk1B mediates survival during the differentiation of C2C12Myoblasts. J. Biol. Chem. 2005, 280, 25788–25801. [Google Scholar] [CrossRef] [PubMed]
- Deng, X.; Mercer, S.E.; Shah, S.; Ewton, D.Z.; Friedman, E. The cyclin-dependent kinase inhibitor p27Kip1 is stabilized in G0 by Mirk/dyrk1B kinase. J. Biol. Chem. 2004, 279, 22498–22504. [Google Scholar] [CrossRef]
- Lara-Chica, M.; Correa-Sáez, A.; Jiménez-Izquierdo, R.; Garrido-Rodríguez, M.; Ponce, F.J.; Moreno, R.; Morrison, K.; Di Vona, C.; Arató, K.; Jiménez-Jiménez, C. A novel CDC25A/DYRK2 regulatory switch modulates cell cycle and survival. Cell Death Differ. 2022, 29, 105–117. [Google Scholar] [CrossRef]
- Taira, N.; Nihira, K.; Yamaguchi, T.; Miki, Y.; Yoshida, K. DYRK2 is targeted to the nucleus and controls p53 via Ser46 phosphorylation in the apoptotic response to DNA damage. Mol. Cell 2007, 25, 725–738. [Google Scholar] [CrossRef] [PubMed]
- Taira, N.; Mimoto, R.; Kurata, M.; Yamaguchi, T.; Kitagawa, M.; Miki, Y.; Yoshida, K. DYRK2 priming phosphorylation of c-Jun and c-Myc modulates cell cycle progression in human cancer cells. J. Clin. Investig. 2012, 122, 859–872. [Google Scholar] [CrossRef] [PubMed]
- Owusu, M.; Bannauer, P.; da Silva, J.F.; Mourikis, T.P.; Jones, A.; Májek, P.; Caldera, M.; Wiedner, M.; Lardeau, C.-H.; Mueller, A.C. Mapping the human kinome in response to DNA damage. Cell Rep. 2019, 26, 555–563.e556. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.; Zou, Y.; Friedman, E. The Transcriptional Activator Mirk/Dyrk1B Is Sequestered by p38α/β MAP Kinase. J. Biol. Chem. 2002, 277, 49438–49445. [Google Scholar] [CrossRef]
- Song, W.J.; Song, E.A.; Jung, M.S.; Choi, S.H.; Baik, H.H.; Jin, B.K.; Kim, J.H.; Chung, S.H. Phosphorylation and inactivation of glycogen synthase kinase 3β (GSK3β) by dual-specificity tyrosine phosphorylation-regulated kinase 1A (Dyrk1A). J. Biol. Chem. 2015, 290, 2321–2333. [Google Scholar] [CrossRef]
- Xiong, Y.; Kuppuswamy, D.; Li, Y.; Livanos, E.M.; Hixon, M.; White, A.; Beach, D.; Tlsty, T.D. Alteration of cell cycle kinase complexes in human papillomavirus E6- and E7-expressing fibroblasts precedes neoplastic transformation. J. Virol. 1996, 70, 999–1008. [Google Scholar] [CrossRef]
- Fischer, M.; Uxa, S.; Stanko, C.; Magin, T.M.; Engeland, K. Human papilloma virus E7 oncoprotein abrogates the p53-p21-DREAM pathway. Sci. Rep. 2017, 7, 2603. [Google Scholar] [CrossRef]
- Ding, Q.; Li, L.; Whyte, P. Human papillomavirus 18 E1^E4 protein interacts with cyclin A/CDK 2 through an RXL motif. Mol. Cell. Biochem. 2013, 373, 29–40. [Google Scholar] [CrossRef]
- Zydek, M.; Hagemeier, C.; Wiebusch, L. Cyclin-Dependent Kinase Activity Controls the Onset of the HCMV Lytic Cycle. PLoS Pathog. 2010, 6, e1001096. [Google Scholar] [CrossRef]
- Schütz, M.; Wangen, C.; Sommerer, M.; Kögler, M.; Eickhoff, J.; Degenhart, C.; Klebl, B.; Naing, Z.; Egilmezer, E.; Hamilton, S.T.; et al. Cytomegalovirus cyclin-dependent kinase ortholog vCDK/pUL97 undergoes regulatory interaction with human cyclin H and CDK7 to codetermine viral replication efficiency. Virus Res. 2023, 335, 199200. [Google Scholar] [CrossRef]
- Schütz, M.; Cordsmeier, A.; Wangen, C.; Horn, A.H.C.; Wyler, E.; Ensser, A.; Sticht, H.; Marschall, M. The Interactive Complex between Cytomegalovirus Kinase vCDK/pUL97 and Host Factors CDK7–Cyclin H Determines Individual Patterns of Transcription in Infected Cells. Int. J. Mol. Sci. 2023, 24, 17421. [Google Scholar] [CrossRef]
- Obergfäll, D.; Wild, M.; Sommerer, M.; Barillas Dahm, M.; Kicuntod, J.; Tillmanns, J.; Kögler, M.; Lösing, J.; Dhotre, K.; Müller, R.; et al. Cyclin-Dependent Kinase 8 Represents a Positive Regulator of Cytomegalovirus Replication and a Novel Host Target for Antiviral Strategies. Pharmaceutics 2024, 16, 1238. [Google Scholar] [CrossRef]
- Orlando Joseph, S.; Astor Todd, L.; Rundle Scott, A.; Schaffer Priscilla, A. The Products of the Herpes Simplex Virus Type 1 Immediate-Early US1/US1.5 Genes Downregulate Levels of S-Phase-Specific Cyclins and Facilitate Virus Replication in S-Phase Vero Cells. J. Virol. 2006, 80, 4005–4016. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Obergfäll, D.; Hahn, F.; Kicuntod, J.; Wangen, C.; Kögler, M.; Wagner, S.; Kaufer, B.; Marschall, M. New Assay Systems to Characterize the Broad-Spectrum Antiherpesviral and Non-Herpesviral Activity of Cyclin-Dependent Kinase (CDK) 8 Inhibitors. Pharmaceuticals 2025, 18, 1560. [Google Scholar] [CrossRef] [PubMed]
- Jiang, L.; Yu, Y.; Li, Z.; Gao, Y.; Zhang, H.; Zhang, M.; Cao, W.; Peng, Q.; Chen, X. BMS-265246, a Cyclin-Dependent Kinase Inhibitor, Inhibits the Infection of Herpes Simplex Virus Type 1. Viruses 2023, 15, 1642. [Google Scholar] [CrossRef]
- Whisnant Adam, W.; Dyck Dionisi, O.; Salazar Sanchez, V.; Rappold Julia, M.; Djakovic, L.; Grothey, A.; Marante Ana, L.; Fischer, P.; Peng, S.; Wolf, K.; et al. Herpes simplex virus 1 inhibits phosphorylation of RNA polymerase II CTD serine-7. J. Virol. 2024, 98, e01178-24. [Google Scholar] [CrossRef]
- Knight Jason, S.; Robertson Erle, S. Epstein-Barr Virus Nuclear Antigen 3C Regulates Cyclin A/p27 Complexes and Enhances Cyclin A-Dependent Kinase Activity. J. Virol. 2004, 78, 1981–1991. [Google Scholar] [CrossRef]
- Iwahori, S.; Murata, T.; Kudoh, A.; Sato, Y.; Nakayama, S.; Isomura, H.; Kanda, T.; Tsurumi, T. Phosphorylation of p27Kip1 by Epstein-Barr Virus Protein Kinase Induces Its Degradation through SCFSkp2 Ubiquitin Ligase Actions during Viral Lytic Replication. J. Biol. Chem. 2009, 284, 18923–18931. [Google Scholar] [CrossRef]
- Tursiella, M.L.; Bowman, E.R.; Wanzeck, K.C.; Throm, R.E.; Liao, J.; Zhu, J.; Sample, C.E. Epstein-Barr Virus Nuclear Antigen 3A Promotes Cellular Proliferation by Repression of the Cyclin-Dependent Kinase Inhibitor p21WAF1/CIP1. PLoS Pathog. 2014, 10, e1004415. [Google Scholar] [CrossRef]
- Hollyoake, M.; Stühler, A.; Farrell, P.; Gordon, J.; Sinclair, A. The Normal Cell Cycle Activation Program Is Exploited during the Infection of Quiescent B Lymphocytes by Epstein-Barr Virus1. Cancer Res. 1995, 55, 4784–4787. [Google Scholar]
- Cayrol, C.; Flemington, E.K. The Epstein-Barr virus bZIP transcription factor Zta causes G0/G1 cell cycle arrest through induction of cyclin-dependent kinase inhibitors. EMBO J. 1996, 15, 2748–2759. [Google Scholar] [CrossRef]
- Moffat, J.F.; Greenblatt, R.J. Effects of varicella-zoster virus on cell cycle regulatory pathways. In Varicella-Zoster Virus; Springer: Berlin/Heidelberg, Germany, 2010; pp. 67–77. [Google Scholar]
- Leisenfelder, S.A.; Moffat, J.F. Varicella-zoster virus infection of human foreskin fibroblast cells results in atypical cyclin expression and cyclin-dependent kinase activity. J. Virol. 2006, 80, 5577–5587. [Google Scholar] [CrossRef] [PubMed]
- Jones, T.; Ramos da Silva, S.; Bedolla, R.; Ye, F.; Zhou, F.; Gao, S. Viral Cyclin promotes KSHV-induced cellular transformation and tumorigenesis by overriding contact inhibition. Cell Cycle 2014, 13, 845–858. [Google Scholar] [CrossRef] [PubMed]
- Godden-Kent, D.; Talbot, S.J.; Boshoff, C.; Chang, Y.; Moore, P.; Weiss, R.A.; Mittnacht, S. The cyclin encoded by Kaposi’s sarcoma-associated herpesvirus stimulates cdk6 to phosphorylate the retinoblastoma protein and histone H1. J. Virol. 1997, 71, 4193–4198. [Google Scholar] [CrossRef]
- Chang, P.-C.; Li, M. Kaposi’s Sarcoma-Associated Herpesvirus K-Cyclin Interacts with Cdk9 and Stimulates Cdk9-Mediated Phosphorylation of p53 Tumor Suppressor. J. Virol. 2008, 82, 278–290. [Google Scholar] [CrossRef]
- Jung, J.K.; Arora, P.; Pagano, J.S.; Jang, K.L. Expression of DNA Methyltransferase 1 Is Activated by Hepatitis B Virus X Protein via a Regulatory Circuit Involving the p16INK4a-Cyclin D1-CDK 4/6-pRb-E2F1 Pathway. Cancer Res. 2007, 67, 5771–5778. [Google Scholar] [CrossRef]
- Lee, S.G.; Rho, H.M. Transcriptional repression of the human p53 gene by hepatitis B viral X protein. Oncogene 2000, 19, 468–471. [Google Scholar] [CrossRef]
- Liang, Y.J.; Chang, H.S.; Wang, C.Y.; Yu, W.C. DYRK1A stabilizes HPV16E7 oncoprotein through phosphorylation of the threonine 5 and threonine 7 residues. Int. J. Biochem. Cell Biol. 2008, 40, 2431–2441. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.S.; Lin, C.H.; Yang, C.H.; Yen, M.S.; Lai, C.R.; Chen, Y.R.; Liang, Y.J.; Yu, W.C. Increased expression of Dyrk1a in HPV16 immortalized keratinocytes enable evasion of apoptosis. Int. J. Cancer 2007, 120, 2377–2385. [Google Scholar] [CrossRef]
- Hutterer, C.; Milbradt, J.; Hamilton, S.; Zaja, M.; Leban, J.; Henry, C.; Vitt, D.; Steingruber, M.; Sonntag, E.; Zeitträger, I.; et al. Inhibitors of dual-specificity tyrosine phosphorylation-regulated kinases (DYRK) exert a strong anti-herpesviral activity. Antivir. Res. 2017, 143, 113–121. [Google Scholar] [CrossRef]
- Egilmezer, E.; Hamilton, S.T.; Lauw, G.; Follett, J.; Sonntag, E.; Schütz, M.; Marschall, M.; Rawlinson, W.D. Human Cytomegalovirus Dysregulates Cellular Dual-Specificity Tyrosine Phosphorylation-Regulated Kinases and Sonic Hedgehog Pathway Proteins in Neural Astrocyte and Placental Models. Viruses 2024, 16, 918. [Google Scholar] [CrossRef] [PubMed]
- Pastor, F.; Charles, E.; Di Vona, C.; Chapelle, M.; Rivoire, M.; Passot, G.; Chabot, B.; de la Luna, S.; Lucifora, J.; Durantel, D.; et al. The dual-specificity kinase DYRK1A interacts with the Hepatitis B virus genome and regulates the production of viral RNA. PLoS ONE 2024, 19, e0311655. [Google Scholar] [CrossRef]
- Xu, J.; Zeng, X.; Huang, J.; Ma, S.; Li, K.; Yang, S.; Naz, W.; Yousaf, T.; Yuan, S.; Liu, Y.; et al. Dual-specificity tyrosine-regulated kinase 4 modulates the STAT3-FOS signaling axis to inhibit hepatitis B virus replication via autophagy. Int. J. Biol. Sci. 2025, 21, 2415–2429. [Google Scholar] [CrossRef]
- Wang, C.; Hu, R.; Wang, T.; Duan, L.; Hou, Q.; Wang, J.; Yang, Z. A bivalent β-carboline derivative inhibits macropinocytosis-dependent entry of pseudorabies virus by targeting the kinase DYRK1A. J. Biol. Chem. 2023, 299, 104605. [Google Scholar] [CrossRef]
- Liu, X.; Cohen Jeffrey, I. Epstein-Barr Virus (EBV) Tegument Protein BGLF2 Promotes EBV Reactivation through Activation of the p38 Mitogen-Activated Protein Kinase. J. Virol. 2015, 90, 1129–1138. [Google Scholar] [CrossRef]
- Adamson, A.L.; Jeffus, D.; Davis, A.; Greengrove, E. Epstein-Barr virus lytic replication activates and is dependent upon MAPK-interacting kinase 1/2 in a cell-type dependent manner. Virology 2022, 572, 72–85. [Google Scholar] [CrossRef]
- Adamson Amy, L.; Darr, D.; Holley-Guthrie, E.; Johnson Robert, A.; Mauser, A.; Swenson, J.; Kenney, S. Epstein-Barr Virus Immediate-Early Proteins BZLF1 and BRLF1 Activate the ATF2 Transcription Factor by Increasing the Levels of Phosphorylated p38 and c-Jun N-Terminal Kinases. J. Virol. 2000, 74, 1224–1233. [Google Scholar] [CrossRef] [PubMed]
- Johnson, R.A.; Huong, S.M.; Huang, E.S. Activation of the mitogen-activated protein kinase p38 by human cytomegalovirus infection through two distinct pathways: A novel mechanism for activation of p38. J. Virol. 2000, 74, 1158–1167. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Pan, H.; Xie, J.; Ye, F.; Gao, S.-J. Modulation of Kaposi’s Sarcoma-Associated Herpesvirus Infection and Replication by MEK/ERK, JNK, and p38 Multiple Mitogen-Activated Protein Kinase Pathways during Primary Infection. J. Virol. 2006, 80, 5371–5382. [Google Scholar] [CrossRef]
- Xie, J.; Ajibade, A.O.; Ye, F.; Kuhne, K.; Gao, S.-J. Reactivation of Kaposi’s sarcoma-associated herpesvirus from latency requires MEK/ERK, JNK and p38 multiple mitogen-activated protein kinase pathways. Virology 2008, 371, 139–154. [Google Scholar] [CrossRef]
- Tang, Q.; Luan, F.; Yuan, A.; Sun, J.; Rao, Z.; Wang, B.; Liu, Y.; Zeng, N. Sophoridine Suppresses Herpes Simplex Virus Type 1 Infection by Blocking the Activation of Cellular PI3K/Akt and p38 MAPK Pathways. Front. Microbiol. 2022, 13, 872505. [Google Scholar] [CrossRef]
- McLean, T.I.; Bachenheimer, S.L. Activation of cJUN N-terminal kinase by herpes simplex virus type 1 enhances viral replication. J. Virol. 1999, 73, 8415–8426. [Google Scholar] [CrossRef]
- Su, A.-R.; Qiu, M.; Li, Y.-L.; Xu, W.-T.; Song, S.-W.; Wang, X.-H.; Song, H.-Y.; Zheng, N.; Wu, Z.-W. BX-795 inhibits HSV-1 and HSV-2 replication by blocking the JNK/p38 pathways without interfering with PDK1 activity in host cells. Acta Pharmacol. Sin. 2017, 38, 402–414. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Zhang, Q.; Zhang, X.; You, L.; Wang, W.; Liu, W.; Han, Y.; Ma, C.; Xu, W.; Chen, J.; et al. HoxA10 Facilitates SHP-1-Catalyzed Dephosphorylation of p38 MAPK/STAT3 To Repress Hepatitis B Virus Replication by a Feedback Regulatory Mechanism. J. Virol. 2019, 93, 7. [Google Scholar] [CrossRef]
- Li, Y.-X.; Ren, Y.-L.; Fu, H.-J.; Zou, L.; Yang, Y.; Chen, Z. Hepatitis B Virus Middle Protein Enhances IL-6 Production via p38 MAPK/NF-κB Pathways in an ER Stress-Dependent Manner. PLoS ONE 2016, 11, e0159089. [Google Scholar] [CrossRef] [PubMed]
- Casaday Rebecca, J.; Bailey Justin, R.; Kalb Suzanne, R.; Brignole Edward, J.; Loveland Amy, N.; Cotter Robert, J.; Gibson, W. Assembly Protein Precursor (pUL80.5 Homolog) of Simian Cytomegalovirus Is Phosphorylated at a Glycogen Synthase Kinase 3 Site and Its Downstream “Priming” Site: Phosphorylation Affects Interactions of Protein with Itself and with Major Capsid Protein. J. Virol. 2004, 78, 13501–13511. [Google Scholar] [CrossRef][Green Version]
- Fujimuro, M.; Hayward, S.D. The Latency-Associated Nuclear Antigen of Kaposi’s Sarcoma-Associated Herpesvirus Manipulates the Activity of Glycogen Synthase Kinase-3β. J. Virol. 2003, 77, 8019–8030. [Google Scholar] [CrossRef] [PubMed]
- Qin, D.; Feng, N.; Fan, W.; Ma, X.; Yan, Q.; Lv, Z.; Zeng, Y.; Zhu, J.; Lu, C. Activation of PI3K/AKT and ERK MAPK signal pathways is required for the induction of lytic cycle replication of Kaposi’s sarcoma-associated herpesvirus by herpes simplex virus type 1. BMC Microbiol. 2011, 11, 240. [Google Scholar] [CrossRef]
- Mody, P.H.; Marvin, K.N.; Hynds, D.L.; Hanson, L.K. Cytomegalovirus infection induces Alzheimer’s disease-associated alterations in tau. J. NeuroVirol. 2023, 29, 400–415. [Google Scholar] [CrossRef]
- Piacentini, R.; Li Puma, D.D.; Ripoli, C.; Marcocci, M.E.; De Chiara, G.; Garaci, E.; Palamara, A.T.; Grassi, C. Herpes Simplex Virus type-1 infection induces synaptic dysfunction in cultured cortical neurons via GSK-3 activation and intraneuronal amyloid-β protein accumulation. Sci. Rep. 2015, 5, 15444. [Google Scholar] [CrossRef]
- Lin, Y.; Zhu, Y.; Jing, L.; Chen, Y.; Xiao, X.; Lei, X.; Xie, Z. GSK3A promotes human adenovirus replication and phosphorylates viral L4-22K protein. Life Sci. Alliance 2025, 8, 9. [Google Scholar] [CrossRef] [PubMed]
- Nishitsuji, H.; Naito, Y.; Murakami, Y.; Sugiyama, M.; Mizokami, M.; Shoji, I.; Murata, T.; Shimotohno, K. Identification of glycogen synthase kinase 3alpha/beta as a host factor required for HBV transcription using high-throughput screening. Hepatology 2025, 82, 1551–1564. [Google Scholar] [CrossRef] [PubMed]
- Yomoda, J.; Muraki, M.; Kataoka, N.; Hosoya, T.; Suzuki, M.; Hagiwara, M.; Kimura, H. Combination of Clk family kinase and SRp75 modulates alternative splicing of Adenovirus E1A. Genes Cells 2008, 13, 233–244. [Google Scholar] [CrossRef]
- Lim, Z.; Mohd-Ismail, N.K.B.; Png, E.; Sze, C.W.; Lin, Q.; Hong, W.; Lim, S.G.; Tan, Y.-J.; Gunaratne, J. Phosphoproteomics Unravel HBV Triggered Rewiring of Host Phosphosignaling Events. Int. J. Mol. Sci. 2022, 23, 5127. [Google Scholar] [CrossRef] [PubMed]
- Faizo, A.A.A.; Bellward, C.; Hernandez-Lopez, H.R.; Stevenson, A.; Gu, Q.; Graham, S.V. The splicing factor kinase, SR protein kinase 1 (SRPK1) is essential for late events in the human papillomavirus life cycle. PLoS Pathog. 2025, 21, e1012697. [Google Scholar] [CrossRef]
- Bell, I.; Martin, A.; Roberts, S. The E1^E4 Protein of Human Papillomavirus Interacts with the Serine-Arginine-Specific Protein Kinase SRPK1. J. Virol. 2007, 81, 5437–5448. [Google Scholar] [CrossRef]
- Prescott Emma, L.; Brimacombe Claire, L.; Hartley, M.; Bell, I.; Graham, S.; Roberts, S. Human Papillomavirus Type 1 E1^E4 Protein Is a Potent Inhibitor of the Serine-Arginine (SR) Protein Kinase SRPK1 and Inhibits Phosphorylation of Host SR Proteins and of the Viral Transcription and Replication Regulator E2. J. Virol. 2014, 88, 12599–12611. [Google Scholar] [CrossRef]
- Mole, S.; Faizo, A.A.A.; Hernandez-Lopez, H.; Griffiths, M.; Stevenson, A.; Roberts, S.; Graham, S.V. Human papillomavirus type 16 infection activates the host serine arginine protein kinase 1 (SRPK1)—Splicing factor axis. J. Gen. Virol. 2020, 101, 523–532. [Google Scholar] [CrossRef]
- Sandri-Goldin, R.M. The many roles of the regulatory protein ICP27 during herpes simplex virus infection. Front. Biosci. 2008, 13, 5241–5256. [Google Scholar] [CrossRef] [PubMed]
- Sciabica, K.S.; Dai, Q.J.; Sandri-Goldin, R.M. ICP27 interacts with SRPK1 to mediate HSV splicing inhibition by altering SR protein phosphorylation. EMBO J. 2003, 22, 1608–1619. [Google Scholar] [CrossRef]
- Ote, I.; Lebrun, M.; Vandevenne, P.; Bontems, S.; Medina-Palazon, C.; Manet, E.; Piette, J.; Sadzot-Delvaux, C. Varicella-Zoster Virus IE4 Protein Interacts with SR Proteins and Exports mRNAs through the TAP/NXF1 Pathway. PLoS ONE 2009, 4, e7882. [Google Scholar] [CrossRef]
- Duarte, M.; Wang, L.; Calderwood, M.A.; Adelmant, G.; Ohashi, M.; Roecklein-Canfield, J.; Marto, J.A.; Hill, D.E.; Deng, H.; Johannsen, E. An RS Motif within the Epstein-Barr Virus BLRF2 Tegument Protein Is Phosphorylated by SRPK2 and Is Important for Viral Replication. PLoS ONE 2013, 8, e53512. [Google Scholar] [CrossRef][Green Version]
- Chen, C.; Wang, J.C.-Y.; Zlotnick, A. A Kinase Chaperones Hepatitis B Virus Capsid Assembly and Captures Capsid Dynamics in vitro. PLoS Pathog. 2011, 7, e1002388. [Google Scholar] [CrossRef]
- Zheng, Y.; Fu, X.D.; Ou, J.H. Suppression of hepatitis B virus replication by SRPK1 and SRPK2 via a pathway independent of the phosphorylation of the viral core protein. Virology 2005, 342, 150–158. [Google Scholar] [CrossRef][Green Version]
- Yip, R.P.H.; Kwok, D.C.Y.; Lai, L.T.F.; Ho, S.M.; Wong, I.C.K.; Chan, C.P.; Lau, W.C.Y.; Ngo, J.C.K. SRPK2 Mediates HBV Core Protein Phosphorylation and Capsid Assembly via Docking Interaction. PLoS Pathog. 2024, 20, e1011978. [Google Scholar] [CrossRef]
- Daub, H.; Blencke, S.; Habenberger, P.; Kurtenbach, A.; Dennenmoser, J.; Wissing, J.; Ullrich, A.; Cotten, M. Identification of SRPK1 and SRPK2 as the Major Cellular Protein Kinases Phosphorylating Hepatitis B Virus Core Protein. J. Virol. 2002, 76, 8124–8137. [Google Scholar] [CrossRef]
- Surjit, M.; Liu, B.; Chow, V.T.K.; Lal, S.K. The Nucleocapsid Protein of Severe Acute Respiratory Syndrome-Coronavirus Inhibits the Activity of Cyclin-Cyclin-dependent Kinase Complex and Blocks S Phase Progression in Mammalian Cells. J. Biol. Chem. 2006, 281, 10669–10681. [Google Scholar] [CrossRef] [PubMed]
- Bouhaddou, M.; Memon, D.; Meyer, B.; White, K.M.; Rezelj, V.V.; Correa Marrero, M.; Polacco, B.J.; Melnyk, J.E.; Ulferts, S.; Kaake, R.M.; et al. The Global Phosphorylation Landscape of SARS-CoV-2 Infection. Cell 2020, 182, 685–712.e619. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; Lei, X.; Chang, Y.; Zhao, J.; Wang, J.; Dong, X.; Liu, Q.; Zhang, Z.; Wang, L.; Yi, D.; et al. SARS-CoV-2 hijacks cellular kinase CDK2 to promote viral RNA synthesis. Signal Transduct. Target. Ther. 2022, 7, 400. [Google Scholar] [CrossRef]
- Gupta, R.K.; Mlcochova, P. Cyclin D3 restricts SARS-CoV-2 envelope incorporation into virions and interferes with viral spread. EMBO J. 2022, 41, e111653. [Google Scholar] [CrossRef]
- He, Y.; Xu, K.; Keiner, B.; Zhou, J.; Czudai, V.; Li, T.; Chen, Z.; Liu, J.; Klenk, H.-D.; Shu Yue, L.; et al. Influenza A Virus Replication Induces Cell Cycle Arrest in G0/G1 Phase. J. Virol. 2010, 84, 12832–12840. [Google Scholar] [CrossRef]
- Zhao, L.; Yan, Y.; Dai, Q.; Wang, Z.; Yin, J.; Xu, Y.; Wang, Z.; Guo, X.; Li, W.; Cao, R.; et al. The CDK1 inhibitor, Ro-3306, is a potential antiviral candidate against influenza virus infection. Antivir. Res. 2022, 201, 105296. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Lian, X.; Gao, Y.; Jiang, L.; Li, Z.; Zhang, H.; Su, Y.; Peng, Q.; Chen, X. LDC000067, a CDK9 inhibitor, unveils promising results in suppressing influenza virus infections in vitro and in vivo. Antimicrob. Agents Chemother. 2024, 69, e01172-24. [Google Scholar] [CrossRef] [PubMed]
- Chiu, H.-C.; Huang, W.-R.; Liao, T.-L.; Chi, P.-I.; Nielsen, B.L.; Liu, J.-H.; Liu, H.-J. Mechanistic insights into avian reovirus p17-modulated suppression of cell cycle CDK–cyclin complexes and enhancement of p53 and cyclin H interaction. J. Biol. Chem. 2018, 293, 12542–12562. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, T.; Hanada, T.; Tokuhisa, T.; Kosai, K.-I.; Sata, M.; Kohara, M.; Yoshimura, A. Activation of STAT3 by the Hepatitis C Virus Core Protein Leads to Cellular Transformation. J. Exp. Med. 2002, 196, 641–653. [Google Scholar] [CrossRef]
- Ohkawa, K.; Ishida, H.; Nakanishi, F.; Hosui, A.; Ueda, K.; Takehara, T.; Hori, M.; Hayashi, N. Hepatitis C Virus Core Functions as a Suppressor of Cyclin-dependent Kinase-activating Kinase and Impairs Cell Cycle Progression. J. Biol. Chem. 2004, 279, 11719–11726. [Google Scholar] [CrossRef]
- Bahnassy, A.A.; Zekri, A.-R.N.; Loutfy, S.A.; Mohamed, W.S.; Moneim, A.A.; Salem, S.E.; Sheta, M.M.; Omar, A.; Al-Zawahry, H. The role of cyclins and cyclin dependent kinases in development and progression of hepatitis C virus-genotype 4-associated hepatitis and hepatocellular carcinoma. Exp. Mol. Pathol. 2011, 91, 643–652. [Google Scholar] [CrossRef]
- Shackel, N.A.; McGuinness, P.H.; Abbott, C.A.; Gorrell, M.D.; McCaughan, G.W. Insights into the pathobiology of hepatitis C virus-associated cirrhosis: Analysis of intrahepatic differential gene expression. Am. J. Pathol. 2002, 160, 641–654. [Google Scholar] [CrossRef]
- Wagayama, H.; Shiraki, K.; Yamanaka, T.; Sugimoto, K.; Ito, T.; Fujikawa, K.; Takase, K.; Nakano, T. p21WAF1/CTP1 expression and hepatitis virus type. Dig. Dis. Sci. 2001, 46, 2074–2079. [Google Scholar] [CrossRef] [PubMed]
- Dove, B.; Brooks, G.; Bicknell, K.; Wurm, T.; Hiscox Julian, A. Cell Cycle Perturbations Induced by Infection with the Coronavirus Infectious Bronchitis Virus and Their Effect on Virus Replication. J. Virol. 2006, 80, 4147–4156. [Google Scholar] [CrossRef]
- Chen, C.-J.; Makino, S. Murine Coronavirus Replication Induces Cell Cycle Arrest in G0/G1 Phase. J. Virol. 2004, 78, 5658–5669. [Google Scholar] [CrossRef]
- Hu, X.; Chen, Z.; Wu, X.; Ding, Z.; Huang, Y.; Fu, Q.; Chen, Z.; Wu, H. Phosphorylation of VP1 Mediated by CDK1-Cyclin B1 Facilitates Infectious Bursal Disease Virus Replication. J. Virol. 2023, 97, e01941-22. [Google Scholar] [CrossRef]
- Rychlowska, M.; Agyapong, A.; Weinfeld, M.; Schang Luis, M. Zika Virus Induces Mitotic Catastrophe in Human Neural Progenitors by Triggering Unscheduled Mitotic Entry in the Presence of DNA Damage While Functionally Depleting Nuclear PNKP. J. Virol. 2022, 96, e00333-22. [Google Scholar] [CrossRef]
- Sui, L.; Wang, W.; Guo, X.; Zhao, Y.; Tian, T.; Zhang, J.; Wang, H.; Xu, Y.; Chi, H.; Xie, H.; et al. Multi-protomics analysis identified host cellular pathways perturbed by tick-borne encephalitis virus infection. Nat. Commun. 2024, 15, 10435. [Google Scholar] [CrossRef]
- Tokunaga, M.; Miyamoto, Y.; Suzuki, T.; Otani, M.; Inuki, S.; Esaki, T.; Nagao, C.; Mizuguchi, K.; Ohno, H.; Yoneda, Y.; et al. Novel anti-flavivirus drugs targeting the nucleolar distribution of core protein. Virology 2020, 541, 41–51. [Google Scholar] [CrossRef] [PubMed]
- Honrubia, J.M.; Valverde, J.R.; Muñoz-Santos, D.; Ripoll-Gómez, J.; de la Blanca, N.; Izquierdo, J.; Villarejo-Torres, M.; Marchena-Pasero, A.; Rueda-Huélamo, M.; Nombela, I.; et al. Interaction between SARS-CoV PBM and Cellular PDZ Domains Leading to Virus Virulence. Viruses 2024, 16, 1214. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharyya, S.; Tobacman, J.K. SARS-CoV-2 spike protein-ACE2 interaction increases carbohydrate sulfotransferases and reduces N-acetylgalactosamine-4-sulfatase by p38 MAPK. Signal Transduct. Target. Ther. 2024, 9, 39. [Google Scholar] [CrossRef] [PubMed]
- Higgins Christina, A.; Nilsson-Payant Benjamin, E.; Bonaventure, B.; Kurland Andrew, P.; Ye, C.; Yaron Tomer, M.; Johnson Jared, L.; Adhikary, P.; Golynker, I.; Panis, M.; et al. SARS-CoV-2 hijacks p38β/MAPK11 to promote virus replication. mBio 2023, 14, e01007-23. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.; Jiao, L.; Chen, J.; Chen, P.; Zhou, F.; Zhang, J.; Wang, M.; Wu, Q.; Cao, S.; Lu, H.; et al. Duck Tembusu virus infection activates the MKK3/6-p38 MAPK signaling pathway to promote virus replication. Vet. Microbiol. 2024, 288, 109951. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Li, P.; Chen, A.; Li, H.; Liu, Z.; Yu, L.; Hou, X. Quantitative proteomic analysis shows involvement of the p38 MAPK pathway in bovine parainfluenza virus type 3 replication. Virol. J. 2022, 19, 116. [Google Scholar] [CrossRef]
- Mudaliar, P.; Pradeep, P.; Abraham, R.; Sreekumar, E. Targeting cap-dependent translation to inhibit Chikungunya virus replication: Selectivity of p38 MAPK inhibitors to virus-infected cells due to autophagy-mediated down regulation of phospho-ERK. J. Gen. Virol. 2021, 102, 001629. [Google Scholar] [CrossRef]
- Nayak, T.K.; Mamidi, P.; Sahoo, S.S.; Kumar, P.S.; Mahish, C.; Chatterjee, S.; Subudhi, B.B.; Chattopadhyay, S.; Chattopadhyay, S. P38 and JNK mitogen-activated protein kinases interact with chikungunya virus non-structural protein-2 and regulate TNF induction during viral infection in macrophages. Front. Immunol. 2019, 10, 786. [Google Scholar] [CrossRef]
- Barbachano-Guerrero, A.; Endy, T.P.; King, C.A. Dengue virus non-structural protein 1 activates the p38 MAPK pathway to decrease barrier integrity in primary human endothelial cells. J. Gen. Virol. 2020, 101, 484–496. [Google Scholar] [CrossRef]
- Sreekanth, G.P.; Chuncharunee, A.; Sirimontaporn, A.; Panaampon, J.; Noisakran, S.; Yenchitsomanus, P.-t.; Limjindaporn, T. SB203580 Modulates p38 MAPK Signaling and Dengue Virus-Induced Liver Injury by Reducing MAPKAPK2, HSP27, and ATF2 Phosphorylation. PLoS ONE 2016, 11, e0149486. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Yip, A.; Seah, P.G.; Blasco, F.; Shi, P.-Y.; Hervé, M. Modulation of inflammation and pathology during dengue virus infection by p38 MAPK inhibitor SB203580. Antivir. Res. 2014, 110, 151–157. [Google Scholar] [CrossRef]
- Choi, M.S.; Heo, J.; Yi, C.M.; Ban, J.; Lee, N.J.; Lee, N.R.; Kim, S.W.; Kim, N.J.; Inn, K.S. A novel p38 mitogen activated protein kinase (MAPK) specific inhibitor suppresses respiratory syncytial virus and influenza A virus replication by inhibiting virus-induced p38 MAPK activation. Biochem. Biophys. Res. Commun. 2016, 477, 311–316. [Google Scholar] [CrossRef]
- Zhan, Y.; Yu, S.; Yang, S.; Qiu, X.; Meng, C.; Tan, L.; Song, C.; Liao, Y.; Liu, W.; Sun, Y.; et al. Newcastle Disease virus infection activates PI3K/Akt/mTOR and p38 MAPK/Mnk1 pathways to benefit viral mRNA translation via interaction of the viral NP protein and host eIF4E. PLoS Pathog. 2020, 16, e1008610. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, A.; Lin, Q.F.; Eaton, H.E.; Shmulevitz, M. p38 Mitogen-Activated Protein Kinase Signaling Enhances Reovirus Replication by Facilitating Efficient Virus Entry, Capsid Uncoating, and Postuncoating Steps. J. Virol. 2023, 97, e0000923. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.; Sun, F.; Wang, L.; Gao, M.; Xie, Y.; Sun, Y.; Liu, H.; Yuan, Y.; Yi, W.; Huang, Z. Virus-induced p38 MAPK activation facilitates viral infection. Theranostics 2020, 10, 12223. [Google Scholar] [CrossRef] [PubMed]
- Tsutsumi, T.; Suzuki, T.; Moriya, K.; Shintani, Y.; Fujie, H.; Miyoshi, H.; Matsuura, Y.; Koike, K.; Miyamura, T. Hepatitis C Virus Core Protein Activates Erk and P38 Mapk in Cooperation With Ethanol in Transgenic Mice. Hepatology 2003, 38, 820–828. [Google Scholar] [CrossRef]
- Yang, S.-H.; Geun Lee, C.; Woo Lee, C.; Choi, E.-J.; Kew Yoon, S.; Seog Ahn, K.; Chul Sung, Y. Hepatitis C Virus Core Inhibits the Fas-mediated p38 Mitogen Activated Kinase Signaling Pathway in Hepatocytes. Mol. Cells 2002, 13, 452–462. [Google Scholar] [CrossRef] [PubMed]
- Ji, W.T.; Lee, L.H.; Lin, F.L.; Wang, L.; Liu, H.J. AMP-activated protein kinase facilitates avian reovirus to induce mitogen-activated protein kinase (MAPK) p38 and MAPK kinase 3/6 signalling that is beneficial for virus replication. J. Gen. Virol. 2009, 90, 3002–3009. [Google Scholar] [CrossRef] [PubMed]
- Hui, K.P.; Lee, S.M.; Cheung, C.Y.; Ng, I.H.; Poon, L.L.; Guan, Y.; Ip, N.Y.; Lau, A.S.; Peiris, J.S. Induction of proinflammatory cytokines in primary human macrophages by influenza A virus (H5N1) is selectively regulated by IFN regulatory factor 3 and p38 MAPK. J. Immunol. 2009, 182, 1088–1098. [Google Scholar] [CrossRef]
- Börgeling, Y.; Schmolke, M.; Viemann, D.; Nordhoff, C.; Roth, J.; Ludwig, S. Inhibition of p38 mitogen-activated protein kinase impairs influenza virus-induced primary and secondary host gene responses and protects mice from lethal H5N1 infection. J. Biol. Chem. 2014, 289, 13–27. [Google Scholar] [CrossRef]
- Hirasawa, K.; Kim, A.; Han, H.S.; Han, J.; Jun, H.S.; Yoon, J.W. Effect of p38 mitogen-activated protein kinase on the replication of encephalomyocarditis virus. J. Virol. 2003, 77, 5649–5656. [Google Scholar] [CrossRef]
- Rixon, H.W.M.; Brown, G.; Murray, J.T.; Sugrue, R.J. The respiratory syncytial virus small hydrophobic protein is phosphorylated via a mitogen-activated protein kinase p38-dependent tyrosine kinase activity during virus infection. J. Gen. Virol. 2005, 86, 375–384. [Google Scholar] [CrossRef]
- Marchant, D.; Singhera Gurpreet, K.; Utokaparch, S.; Hackett Tillie, L.; Boyd John, H.; Luo, Z.; Si, X.; Dorscheid Delbert, R.; McManus Bruce, M.; Hegele Richard, G. Toll-Like Receptor 4-Mediated Activation of p38 Mitogen-Activated Protein Kinase Is a Determinant of Respiratory Virus Entry and Tropism. J. Virol. 2010, 84, 11359–11373. [Google Scholar] [CrossRef]
- Peng, H.; Shi, M.; Zhang, L.; Li, Y.; Sun, J.; Zhang, L.; Wang, X.; Xu, X.; Zhang, X.; Mao, Y.; et al. Activation of JNK1/2 and p38 MAPK signaling pathways promotes enterovirus 71 infection in immature dendritic cells. BMC Microbiol. 2014, 14, 147. [Google Scholar] [CrossRef]
- Fitzpatrick, C.J.; Mudhasani, R.R.; Altamura, L.A.; Campbell, C.E.; Tran, J.P.; Beitzel, B.F.; Narayanan, A.; De la Fuente, C.L.; Kehn-Hall, K.; Smith, J.M. Junin virus activates p38 MAPK and HSP27 upon entry. Front. Cell. Infect. Microbiol. 2022, 12, 798978. [Google Scholar] [CrossRef] [PubMed]
- Jia, X.; Chen, J.; Li, C.; Li, J.; Su, M.; Yang, K.; Zhang, Y.; Li, Z. Glycogen synthase kinase 3 promotes the proliferation of porcine epidemic diarrhoea virus by phosphorylating the nucleocapsid protein. Virulence 2025, 16, 2506504. [Google Scholar] [CrossRef]
- Qin, X.; Zhang, Y.A.; Tu, J. p38MAPK- and GSK3-Mediated Phosphorylation of Snakehead Vesiculovirus Phosphoprotein at Threonine 160 Facilitates Viral Replication. J. Virol. 2023, 97, e0040423. [Google Scholar] [CrossRef] [PubMed]
- Rana, A.K.; Rahmatkar, S.N.; Kumar, A.; Singh, D. Glycogen synthase kinase-3: A putative target to combat severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) pandemic. Cytokine Growth Factor Rev. 2021, 58, 92–101. [Google Scholar] [CrossRef]
- Johnson, B.A.; Zhou, Y.; Lokugamage, K.G.; Vu, M.N.; Bopp, N.; Crocquet-Valdes, P.A.; Kalveram, B.; Schindewolf, C.; Liu, Y.; Scharton, D.; et al. Nucleocapsid mutations in SARS-CoV-2 augment replication and pathogenesis. PLoS Pathog. 2022, 18, e1010627. [Google Scholar] [CrossRef]
- Yun, J.S.; Song, H.; Kim, N.H.; Cha, S.Y.; Hwang, K.H.; Lee, J.E.; Jeong, C.H.; Song, S.H.; Kim, S.; Cho, E.S.; et al. Glycogen Synthase Kinase-3 Interaction Domain Enhances Phosphorylation of SARS-CoV-2 Nucleocapsid Protein. Mol. Cells 2022, 45, 911–922. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.-H.; Yeh, S.-H.; Tsay, Y.-G.; Shieh, Y.-H.; Kao, C.-L.; Chen, Y.-S.; Wang, S.-H.; Kuo, T.-J.; Chen, D.-S.; Chen, P.-J. Glycogen Synthase Kinase-3 Regulates the Phosphorylation of Severe Acute Respiratory Syndrome Coronavirus Nucleocapsid Protein and Viral Replication. J. Biol. Chem. 2009, 284, 5229–5239. [Google Scholar] [CrossRef]
- Ishikawa, C.; Mori, N. A New Strategy for Adult T-Cell Leukemia Treatment Targeting Glycogen Synthase Kinase-3β. Eur. J. Haematol. 2024, 113, 852–862. [Google Scholar] [CrossRef]
- Sarhan, M.A.; Abdel-Hakeem, M.S.; Mason, A.L.; Tyrrell, D.L.; Houghton, M. Glycogen synthase kinase 3β inhibitors prevent hepatitis C virus release/assembly through perturbation of lipid metabolism. Sci. Rep. 2017, 7, 2495. [Google Scholar] [CrossRef]
- Butt, A.M.; Feng, D.; Nasrullah, I.; Tahir, S.; Idrees, M.; Tong, Y.; Lu, J. Computational identification of interplay between phosphorylation and O-β-glycosylation of human occludin as potential mechanism to impair hepatitis C virus entry. Infect. Genet. Evol. 2012, 12, 1235–1245. [Google Scholar] [CrossRef]
- Saleh, M.; Rüschenbaum, S.; Welsch, C.; Zeuzem, S.; Moradpour, D.; Gouttenoire, J.; Lange, C.M. Glycogen synthase kinase 3β enhances hepatitis C virus replication by supporting miR-122. Front. Microbiol. 2018, 9, 2949. [Google Scholar] [CrossRef]
- Petitdemange, C.; Maucourant, C.; Tarantino, N.; Rey, J.; Vieillard, V. Glycogen synthetase kinase 3 inhibition drives MIC-A/B to promote cytokine production by human natural killer cells in Dengue virus type 2 infection. Eur. J. Immunol. 2020, 50, 342–352. [Google Scholar] [CrossRef] [PubMed]
- Jones, T.; Raman, R.; Puhl, A.C.; Lane, T.R.; Riabova, O.; Kazakova, E.; Makarov, V.; Ekins, S. Discovery of Dual Targeting GSK-3β/HIV-1 Reverse Transcriptase Inhibitors as Neuroprotective Antiviral Agents. ACS Chem. Neurosci. 2025, 16, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Guendel, I.; Iordanskiy, S.; Van Duyne, R.; Kehn-Hall, K.; Saifuddin, M.; Das, R.; Jaworski, E.; Sampey Gavin, C.; Senina, S.; Shultz, L.; et al. Novel Neuroprotective GSK-3β Inhibitor Restricts Tat-Mediated HIV-1 Replication. J. Virol. 2014, 88, 1189–1208. [Google Scholar] [CrossRef]
- Yuan, J.; Zhang, J.; Wong, B.W.; Si, X.; Wong, J.; Yang, D.; Luo, H. Inhibition of glycogen synthase kinase 3β suppresses coxsackievirus-induced cytopathic effect and apoptosis via stabilization of β-catenin. Cell Death Differ. 2005, 12, 1097–1106. [Google Scholar] [CrossRef] [PubMed]
- Müller, K.H.; Kakkola, L.; Nagaraj, A.S.; Cheltsov, A.V.; Anastasina, M.; Kainov, D.E. Emerging cellular targets for influenza antiviral agents. Trends Pharmacol. Sci. 2012, 33, 89–99. [Google Scholar] [CrossRef]
- Jheng, J.-R.; Hsieh, C.-F.; Chang, Y.-H.; Ho, J.-Y.; Tang, W.-F.; Chen, Z.-Y.; Liu, C.-J.; Lin, T.-J.; Huang, L.-Y.; Chern, J.-H.; et al. Rosmarinic acid interferes with influenza virus A entry and replication by decreasing GSK3β and phosphorylated AKT expression levels. J. Microbiol. Immunol. Infect. 2022, 55, 598–610. [Google Scholar] [CrossRef]
- Hirata, N.; Suizu, F.; Matsuda-Lennikov, M.; Edamura, T.; Bala, J.; Noguchi, M. Inhibition of Akt kinase activity suppresses entry and replication of influenza virus. Biochem. Biophys. Res. Commun. 2014, 450, 891–898. [Google Scholar] [CrossRef]
- Kim, J.Y.; Park, S.Y.; Lyoo, H.R.; Koo, E.S.; Kim, M.S.; Jeong, Y.S. Extended stability of cyclin D1 contributes to limited cell cycle arrest at G1-phase in BHK-21 cells with Japanese encephalitis virus persistent infection. J. Microbiol. 2015, 53, 77–83. [Google Scholar] [CrossRef]
- Fu, Z.; Xiang, Y.; Fu, Y.; Su, Z.; Tan, Y.; Yang, M.; Yan, Y.; Baghaei Daemi, H.; Shi, Y.; Xie, S.; et al. DYRK1A is a multifunctional host factor that regulates coronavirus replication in a kinase-independent manner. J. Virol. 2023, 98, e01239-23. [Google Scholar] [CrossRef]
- Strine, M.S.; Cai, W.L.; Wei, J.; Alfajaro, M.M.; Filler, R.B.; Biering, S.B.; Sarnik, S.; Chow, R.D.; Patil, A.; Cervantes, K.S.; et al. DYRK1A promotes viral entry of highly pathogenic human coronaviruses in a kinase-independent manner. PLoS Biol. 2023, 21, e3002097. [Google Scholar] [CrossRef]
- An, T.; Li, S.; Pan, W.; Tien, P.; Zhong, B.; Shu, H.-B.; Wu, S. DYRK2 Negatively Regulates Type I Interferon Induction by Promoting TBK1 Degradation via Ser527 Phosphorylation. PLoS Pathog. 2015, 11, e1005179. [Google Scholar] [CrossRef]
- Booiman, T.; Loukachov, V.V.; van Dort, K.A.; van ‘t Wout, A.B.; Kootstra, N.A. DYRK1A Controls HIV-1 Replication at a Transcriptional Level in an NFAT Dependent Manner. PLoS ONE 2015, 10, e0144229. [Google Scholar] [CrossRef] [PubMed]
- Kisaka, J.K.; Ratner, L.; Kyei, G.B. The Dual-Specificity Kinase DYRK1A Modulates the Levels of Cyclin L2 To Control HIV Replication in Macrophages. J. Virol. 2020, 94, 6. [Google Scholar] [CrossRef] [PubMed]
- Nekhai, S.; Zhou, M.; Fernandez, A.; Lane, W.S.; Lamb, N.J.C.; Brady, J.; Kumar, A. HIV-1 Tat-associated RNA polymerase C-terminal domain kinase, CDK2, phosphorylates CDK7 and stimulates Tat-mediated transcription. Biochem. J. 2002, 364, 649–657. [Google Scholar] [CrossRef]
- Kim, Y.K.; Bourgeois, C.F.; Pearson, R.; Tyagi, M.; West, M.J.; Wong, J.; Wu, S.Y.; Chiang, C.M.; Karn, J. Recruitment of TFIIH to the HIV LTR is a rate-limiting step in the emergence of HIV from latency. EMBO J. 2006, 25, 3596–3604. [Google Scholar] [CrossRef] [PubMed]
- Pauls, E.; Ruiz, A.; Badia, R.; Permanyer, M.; Gubern, A.; Riveira-Muñoz, E.; Torres-Torronteras, J.; Álvarez, M.; Mothe, B.; Brander, C.; et al. Cell Cycle Control and HIV-1 Susceptibility Are Linked by CDK6-Dependent CDK2 Phosphorylation of SAMHD1 in Myeloid and Lymphoid Cells. J. Immunol. 2014, 193, 1988–1997. [Google Scholar] [CrossRef]
- Pak, V.; Eifler, T.T.; Jäger, S.; Krogan, N.J.; Fujinaga, K.; Peterlin, B.M. CDK11 in TREX/THOC Regulates HIV mRNA 3′ End Processing. Cell Host Microbe 2015, 18, 560–570. [Google Scholar] [CrossRef]
- Berro, R.; Pedati, C.; Kehn-Hall, K.; Wu, W.; Klase, Z.; Even, Y.; Genevière, A.-M.; Ammosova, T.; Nekhai, S.; Kashanchi, F. CDK13, a New Potential Human Immunodeficiency Virus Type 1 Inhibitory Factor Regulating Viral mRNA Splicing. J. Virol. 2008, 82, 7155–7166. [Google Scholar] [CrossRef]
- Cereseto, A.; Parks, R.W.; Rivadeneira, E.; Franchini, G. Limiting amounts of p27Kip1 correlates with constitutive activation of cyclin E-CDK2 complex in HTLV-I-transformed T-cells. Oncogene 1999, 18, 2441–2450. [Google Scholar] [CrossRef]
- Kibler Karen, V.; Jeang, K.-T. CREB/ATF-Dependent Repression of Cyclin A by Human T-Cell Leukemia Virus Type 1 Tax Protein. J. Virol. 2001, 75, 2161–2173. [Google Scholar] [CrossRef]
- Baydoun, H.H.; Pancewicz, J.; Bai, X.; Nicot, C. HTLV-I p30 inhibits multiple S phase entry checkpoints, decreases cyclin E-CDK2 interactions and delays cell cycle progression. Mol. Cancer 2010, 9, 302. [Google Scholar] [CrossRef]
- Grassmann, R.; Aboud, M.; Jeang, K.-T. Molecular mechanisms of cellular transformation by HTLV-1 Tax. Oncogene 2005, 24, 5976–5985. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Li, H.; Tsai, M.D. Direct binding of the N-terminus of HTLV-1 tax oncoprotein to cyclin-dependent kinase 4 is a dominant path to stimulate the kinase activity. Biochemistry 2003, 42, 6921–6928. [Google Scholar] [CrossRef]
- Karakama, Y.; Sakamoto, N.; Itsui, Y.; Nakagawa, M.; Tasaka-Fujita, M.; Nishimura-Sakurai, Y.; Kakinuma, S.; Oooka, M.; Azuma, S.; Tsuchiya, K.; et al. Inhibition of hepatitis C virus replication by a specific inhibitor of serine-arginine-rich protein kinase. Antimicrob. Agents Chemother. 2010, 54, 3179–3186. [Google Scholar] [CrossRef]
- Yaron, T.M.; Heaton, B.E.; Levy, T.M.; Johnson, J.L.; Jordan, T.X.; Cohen, B.M.; Kerelsky, A.; Lin, T.Y.; Liberatore, K.M.; Bulaon, D.K.; et al. Host protein kinases required for SARS-CoV-2 nucleocapsid phosphorylation and viral replication. Sci. Signal 2022, 15, eabm0808. [Google Scholar] [CrossRef]
- Molinero, M.; Gómez, S.; Benítez, I.D.; Vengoechea, J.; González, J.; Polanco, D.; Gort-Paniello, C.; Moncusí-Moix, A.; García-Hidalgo, M.C.; Perez-Pons, M. Multiplex protein profiling of bronchial aspirates reveals disease-, mortality-and respiratory sequelae-associated signatures in critically ill patients with ARDS secondary to SARS-CoV-2 infection. Front. Immunol. 2022, 13, 942443. [Google Scholar] [CrossRef]
- Fukuhara, T.; Hosoya, T.; Shimizu, S.; Sumi, K.; Oshiro, T.; Yoshinaka, Y.; Suzuki, M.; Yamamoto, N.; Herzenberg, L.A.; Herzenberg, L.A.; et al. Utilization of host SR protein kinases and RNA-splicing machinery during viral replication. Proc. Natl. Acad. Sci. USA 2006, 103, 11329–11333. [Google Scholar] [CrossRef] [PubMed]
- Dahal, S.; Clayton, K.; Been, T.; Fernet-Brochu, R.; Ocando, A.V.; Balachandran, A.; Poirier, M.; Maldonado, R.K.; Shkreta, L.; Boligan, K.F.; et al. Opposing roles of CLK SR kinases in controlling HIV-1 gene expression and latency. Retrovirology 2022, 19, 18. [Google Scholar] [CrossRef] [PubMed]
- Takamatsu, Y.; Krähling, V.; Kolesnikova, L.; Halwe, S.; Lier, C.; Baumeister, S.; Noda, T.; Biedenkopf, N.; Becker, S. Serine-Arginine Protein Kinase 1 Regulates Ebola Virus Transcription. mBio 2020, 11, 1. [Google Scholar] [CrossRef]
- Wong, R.; Balachandran, A.; Mao, A.Y.Q.; Dobson, W.; Gray-Owen, S.; Cochrane, A. Differential effect of CLK SR Kinases on HIV-1 gene expression: Potential novel targets for therapy. Retrovirology 2011, 8, 47. [Google Scholar] [CrossRef]
- Wong, R.W.; Balachandran, A.; Ostrowski, M.A.; Cochrane, A. Digoxin Suppresses HIV-1 Replication by Altering Viral RNA Processing. PLoS Pathog. 2013, 9, e1003241. [Google Scholar] [CrossRef]
- An, J.; Nakajima, T.; Shibata, H.; Arimura, T.; Yasunami, M.; Kimura, A. A novel link of HLA locus to the regulation of immunity and infection: NFKBIL1 regulates alternative splicing of human immune-related genes and influenza virus M gene. J. Autoimmun. 2013, 47, 25–33. [Google Scholar] [CrossRef]
- Artarini, A.; Meyer, M.; Shin, Y.J.; Huber, K.; Hilz, N.; Bracher, F.; Eros, D.; Orfi, L.; Keri, G.; Goedert, S.; et al. Regulation of influenza A virus mRNA splicing by CLK1. Antivir. Res. 2019, 168, 187–196. [Google Scholar] [CrossRef] [PubMed]
- Bojkova, D.; Reus, P.; Panosch, L.; Bechtel, M.; Rothenburger, T.; Kandler, J.D.; Pfeiffer, A.; Wagner, J.U.G.; Shumliakivska, M.; Dimmeler, S.; et al. Identification of novel antiviral drug candidates using an optimized SARS-CoV-2 phenotypic screening platform. iScience 2023, 26, 105944. [Google Scholar] [CrossRef]
- Thinwa, J.W.; Zou, Z.; Parks, E.; Sebti, S.; Hui, K.; Wei, Y.; Goodarzi, M.; Singh, V.; Urquhart, G.; Jewell, J.L.; et al. CDKL5 regulates p62-mediated selective autophagy and confers protection against neurotropic viruses. J. Clin. Investig. 2024, 134, e168544. [Google Scholar] [CrossRef]
- Charman, M.; Weitzman, M.D. Replication Compartments of DNA Viruses in the Nucleus: Location, Location, Location. Viruses 2020, 12, 151. [Google Scholar] [CrossRef] [PubMed]
- Choi, K.H. Viral polymerases. Adv. Exp. Med. Biol. 2012, 726, 267–304. [Google Scholar] [CrossRef] [PubMed]
- Romanelli, M.G.; Pignatti, P.F. Herpes simplex virus replication in the presence of DNA polymerase alpha inhibitors. Virus Res. 1988, 11, 293–302. [Google Scholar] [CrossRef]
- Louten, J. Chapter 4—Virus Replication. In Essential Human Virology; Academic Press: Cambridge, MA, USA, 2016; pp. 49–70. [Google Scholar] [CrossRef]
- Lo, S.Y.; Lai, M.J.; Yang, C.H.; Li, H.C. Unveiling the Connection: Viral Infections and Genes in dNTP Metabolism. Viruses 2024, 16, 1412. [Google Scholar] [CrossRef]
- Rampersad, S.; Tennant, P. Chapter 3—Replication and Expression Strategies of Viruses. In Viruses; Academic Press: Cambridge, MA, USA, 2018; pp. 55–82. [Google Scholar] [CrossRef]
- Lei, C.-Q.; Zhong, B.; Zhang, Y.; Zhang, J.; Wang, S.; Shu, H.-B. Glycogen Synthase Kinase 3β Regulates IRF3 Transcription Factor-Mediated Antiviral Response via Activation of the Kinase TBK1. Immunity 2010, 33, 878–889. [Google Scholar] [CrossRef] [PubMed]
- Fujimuro, M.; Hayward, S.D. Manipulation of glycogen-synthase kinase-3 activity in KSHV-associated cancers. J. Mol. Med. 2004, 82, 223–231. [Google Scholar] [CrossRef]
- Payne, S. Introduction to RNA Viruses. In Viruses; Academic Press: Cambridge, MA, USA, 2017; pp. 97–105. [Google Scholar] [CrossRef]
- Smith, J.A.; Schmechel, S.C.; Raghavan, A.; Abelson, M.; Reilly, C.; Katze, M.G.; Kaufman, R.J.; Bohjanen, P.R.; Schiff, L.A. Reovirus induces and benefits from an integrated cellular stress response. J. Virol. 2006, 80, 2019–2033. [Google Scholar] [CrossRef]
- Mohanty, S.; Harhaj, E.W. Mechanisms of Oncogenesis by HTLV-1 Tax. Pathogens 2020, 9, 543. [Google Scholar] [CrossRef]
- Pellarin, I.; Dall’Acqua, A.; Favero, A.; Segatto, I.; Rossi, V.; Crestan, N.; Karimbayli, J.; Belletti, B.; Baldassarre, G. Cyclin-dependent protein kinases and cell cycle regulation in biology and disease. Signal Transduct. Target. Ther. 2025, 10, 11. [Google Scholar] [CrossRef]
- Fassl, A.; Geng, Y.; Sicinski, P. CDK4 and CDK6 kinases: From basic science to cancer therapy. Science 2022, 375, eabc1495. [Google Scholar] [CrossRef]
- Liu, X.; Wu, H.; Liu, Z. An integrative human pan-cancer analysis of cyclin-dependent kinase 1 (CDK1). Cancers 2022, 14, 2658. [Google Scholar] [CrossRef]
- Karst, A.M.; Jones, P.M.; Vena, N.; Ligon, A.H.; Liu, J.F.; Hirsch, M.S.; Etemadmoghadam, D.; Bowtell, D.D.; Drapkin, R. Cyclin E1 deregulation occurs early in secretory cell transformation to promote formation of fallopian tube–derived high-grade serous ovarian cancers. Cancer Res. 2014, 74, 1141–1152. [Google Scholar] [CrossRef] [PubMed]
- Malumbres, M.; Barbacid, M. To cycle or not to cycle: A critical decision in cancer. Nat. Rev. Cancer 2001, 1, 222–231. [Google Scholar] [CrossRef]
- AbuHammad, S.; Cullinane, C.; Martin, C.; Bacolas, Z.; Ward, T.; Chen, H.; Slater, A.; Ardley, K.; Kirby, L.; Chan, K.T. Regulation of PRMT5–MDM4 axis is critical in the response to CDK4/6 inhibitors in melanoma. Proc. Natl. Acad. Sci. USA 2019, 116, 17990–18000. [Google Scholar] [CrossRef] [PubMed]
- Ding, X.; Sharko, A.C.; McDermott, M.S.; Schools, G.P.; Chumanevich, A.; Ji, H.; Li, J.; Zhang, L.; Mack, Z.T.; Sikirzhytski, V. Inhibition of CDK8/19 Mediator kinase potentiates HER2-targeting drugs and bypasses resistance to these agents in vitro and in vivo. Proc. Natl. Acad. Sci. USA 2022, 119, e2201073119. [Google Scholar] [CrossRef]
- Liu, H.; Liu, K.; Dong, Z. Targeting CDK12 for cancer therapy: Function, mechanism, and drug discovery. Cancer Res. 2021, 81, 18–26. [Google Scholar] [CrossRef]
- Kollmann, K.; Heller, G.; Schneckenleithner, C.; Warsch, W.; Scheicher, R.; Ott, R.G.; Schäfer, M.; Fajmann, S.; Schlederer, M.; Schiefer, A.-I. A kinase-independent function of CDK6 links the cell cycle to tumor angiogenesis. Cancer Cell 2013, 24, 167–181. [Google Scholar] [CrossRef]
- Malumbres, M.; Barbacid, M. Cell cycle, CDKs and cancer: A changing paradigm. Nat. Rev. Cancer 2009, 9, 153–166. [Google Scholar] [CrossRef]
- Maciejowski, J.; de Lange, T. Telomeres in cancer: Tumour suppression and genome instability. Nat. Rev. Mol. Cell Biol. 2017, 18, 175–186. [Google Scholar] [CrossRef]
- Matsuura, I.; Denissova, N.G.; Wang, G.; He, D.; Long, J.; Liu, F. Cyclin-dependent kinases regulate the antiproliferative function of Smads. Nature 2004, 430, 226–231. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhao, Y.; Yiminniyaze, R.; Zhu, N.; Zhang, Y.; Wumaier, G.; Xia, J.; Dong, L.; Zhou, D.; Wang, J. CDK10 suppresses metastasis of lung adenocarcinoma through inhibition of the ETS2/c-Raf/p-MEK/p-ERK signaling loop. Mol. Carcinog. 2024, 63, 61–74. [Google Scholar] [CrossRef]
- Nguyen, M.D.; Larivière, R.C.; Julien, J.-P. Deregulation of Cdk5 in a mouse model of ALS: Toxicity alleviated by perikaryal neurofilament inclusions. Neuron 2001, 30, 135–148. [Google Scholar] [CrossRef]
- Stearns, T. Centrosome duplication: A centriolar pas de deux. Cell 2001, 105, 417–420. [Google Scholar] [CrossRef] [PubMed]
- Fredi, B.M.; De Labio, R.W.; Rasmussen, L.T.; Chagas, E.F.B.; Chen, E.S.; Turecki, G.; Smith, M.d.A.C.; Payão, S.L.M. CDK10, CDK11, FOXO1, and FOXO3 gene expression in Alzheimer’s disease encephalic samples. Cell. Mol. Neurobiol. 2023, 43, 2953–2962. [Google Scholar] [CrossRef] [PubMed]
- Chaput, D.; Kirouac, L.; Stevens, S.M., Jr.; Padmanabhan, J. Potential role of PCTAIRE-2, PCTAIRE-3 and P-Histone H4 in amyloid precursor protein-dependent Alzheimer pathology. Oncotarget 2016, 7, 8481. [Google Scholar]
- Chung, H.-l.; Mao, X.; Wang, H.; Park, Y.-J.; Marcogliese, P.C.; Rosenfeld, J.A.; Burrage, L.C.; Liu, P.; Murdock, D.R.; Yamamoto, S. De novo variants in CDK19 are associated with a syndrome involving intellectual disability and epileptic encephalopathy. Am. J. Hum. Genet. 2020, 106, 717–725. [Google Scholar] [CrossRef] [PubMed]
- Bajić, V.P.; Su, B.; Lee, H.-G.; Kudo, W.; Siedlak, S.L.; Živković, L.; Spremo-Potparević, B.; Djelic, N.; Milicevic, Z.; Singh, A.K. Mislocalization of CDK11/PITSLRE, a regulator of the G2/M phase of the cell cycle, in Alzheimer disease. Cell. Mol. Biol. Lett. 2011, 16, 359–372. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Mohamed, T.M.; Ang, Y.-S.; Radzinsky, E.; Zhou, P.; Huang, Y.; Elfenbein, A.; Foley, A.; Magnitsky, S.; Srivastava, D. Regulation of cell cycle to stimulate adult cardiomyocyte proliferation and cardiac regeneration. Cell 2018, 173, 104–116.e112. [Google Scholar] [CrossRef]
- Aguilar, V.; Fajas, L. Cycling through metabolism. EMBO Mol. Med. 2010, 2, 338–348. [Google Scholar] [CrossRef] [PubMed]
- Abella, A.; Dubus, P.; Malumbres, M.; Rane, S.G.; Kiyokawa, H.; Sicard, A.; Vignon, F.; Langin, D.; Barbacid, M.; Fajas, L. Cdk4 promotes adipogenesis through PPARγ activation. Cell Metab. 2005, 2, 239–249. [Google Scholar] [CrossRef]
- Nonomura, Y.; Nagasaka, K.; Hagiyama, H.; Sekine, C.; Nanki, T.; Tamamori-Adachi, M.; Miyasaka, N.; Kohsaka, H. Direct modulation of rheumatoid inflammatory mediator expression in retinoblastoma protein–dependent and–independent pathways by cyclin-dependent kinase 4/6. Arthritis Rheum. 2006, 54, 2074–2083. [Google Scholar] [CrossRef]
- Yik, J.H.; Hu, Z.a.; Kumari, R.; Christiansen, B.A.; Haudenschild, D.R. Cyclin-Dependent Kinase 9 Inhibition protects cartilage from the catabolic effects of proinflammatory cytokines. Arthritis Rheumatol. 2014, 66, 1537–1546. [Google Scholar] [CrossRef]
- Cho, Y.-Y.; Tang, F.; Yao, K.; Lu, C.; Zhu, F.; Zheng, D.; Pugliese, A.; Bode, A.M.; Dong, Z. Cyclin-dependent Kinase-3–mediated c-Jun phosphorylation at Ser63 and Ser73 enhances cell transformation. Cancer Res. 2009, 69, 272–281. [Google Scholar] [CrossRef]
- Hu, J.H.; Malloy, C.; Tabor, G.T.; Gutzmann, J.J.; Liu, Y.; Abebe, D.; Karlsson, R.-M.; Durell, S.; Cameron, H.A.; Hoffman, D.A. Activity-dependent isomerization of Kv4.2 by Pin1 regulates cognitive flexibility. Nat. Commun. 2020, 11, 1567. [Google Scholar] [CrossRef]
- Chung, S.H.; Biswas, S.; Selvaraj, V.; Liu, X.B.; Sohn, J.; Jiang, P.; Chen, C.; Chmilewsky, F.; Marzban, H.; Horiuchi, M.; et al. The p38α mitogen-activated protein kinase is a key regulator of myelination and remyelination in the CNS. Cell Death Dis. 2015, 6, e1748. [Google Scholar] [CrossRef]
- Kheiri, G.; Dolatshahi, M.; Rahmani, F.; Rezaei, N. Role of p38/MAPKs in Alzheimer’s disease: Implications for amyloid beta toxicity targeted therapy. Rev. Neurosci. 2018, 30, 9–30. [Google Scholar] [CrossRef]
- Lin, X.; Wang, M.; Zhang, J.; Xu, R. p38 MAPK: A Potential Target of Chronic Pain. Curr. Med. Chem. 2014, 21, 4405–4418. [Google Scholar] [CrossRef]
- Romero-Becerra, R.; Santamans, A.M.; Folgueira, C.; Sabio, G. p38 MAPK Pathway in the Heart: New Insights in Health and Disease. Int. J. Mol. Sci. 2020, 21, 7412. [Google Scholar] [CrossRef]
- Arabacilar, P.; Marber, M. The case for inhibiting p38 mitogen-activated protein kinase in heart failure. Front. Pharmacol. 2015, 6, 102. [Google Scholar] [CrossRef]
- Bengal, E.; Aviram, S. p38α MAPK Regulation of Energy Metabolism in Skeletal Muscle Offers a Therapeutic Path for Type 2 Diabetes. Cells 2025, 14, 1277. [Google Scholar] [CrossRef]
- Manieri, E.; Sabio, G. Stress kinases in the modulation of metabolism and energy balance. J. Mol. Endocrinol. 2015, 55, R11–R22. [Google Scholar] [CrossRef]
- Rammohan, M.; Harris, E.; Bhansali, R.S.; Zhao, E.; Li, L.S.; Crispino, J.D. The chromosome 21 kinase DYRK1A: Emerging roles in cancer biology and potential as a therapeutic target. Oncogene 2022, 41, 2003–2011. [Google Scholar] [CrossRef] [PubMed]
- Feki, A.; Hibaoui, Y. DYRK1A protein, a promising therapeutic target to improve cognitive deficits in Down syndrome. Brain Sci. 2018, 8, 187. [Google Scholar] [CrossRef] [PubMed]
- Altafaj, X.; Dierssen, M.; Baamonde, C.; Martí, E.; Visa, J.; Guimerà, J.; Oset, M.; González, J.R.; Flórez, J.; Fillat, C. Neurodevelopmental delay, motor abnormalities and cognitive deficits in transgenic mice overexpressing Dyrk1A (minibrain), a murine model of Down’s syndrome. Hum. Mol. Genet. 2001, 10, 1915–1923. [Google Scholar] [CrossRef] [PubMed]
- Arbones, M.L.; Thomazeau, A.; Nakano-Kobayashi, A.; Hagiwara, M.; Delabar, J.M. DYRK1A and cognition: A lifelong relationship. Pharmacol. Ther. 2019, 194, 199–221. [Google Scholar] [CrossRef] [PubMed]
- Deboever, E.; Fistrovich, A.; Hulme, C.; Dunckley, T. The Omnipresence of DYRK1A in Human Diseases. Int. J. Mol. Sci. 2022, 23, 9355. [Google Scholar] [CrossRef] [PubMed]
- Hille, S.; Dierck, F.; Kühl, C.; Sosna, J.; Adam-Klages, S.; Adam, D.; Lüllmann-Rauch, R.; Frey, N.; Kuhn, C. Dyrk1a regulates the cardiomyocyte cell cycle via D-cyclin-dependent Rb/E2f-signalling. Cardiovasc. Res. 2016, 110, 381–394. [Google Scholar] [CrossRef]
- Pozo, N.; Zahonero, C.; Fernández, P.; Liñares, J.M.; Ayuso, A.; Hagiwara, M.; Pérez, A.; Ricoy, J.R.; Hernández-Laín, A.; Sepúlveda, J.M. Inhibition of DYRK1A destabilizes EGFR and reduces EGFR-dependent glioblastoma growth. J. Clin. Investig. 2013, 123, 2475–2487. [Google Scholar] [CrossRef]
- Yang, C.; Ji, D.; Weinstein, E.J.; Choy, E.; Hornicek, F.J.; Wood, K.B.; Liu, X.; Mankin, H.; Duan, Z. The kinase Mirk is a potential therapeutic target in osteosarcoma. Carcinogenesis 2010, 31, 552–558. [Google Scholar] [CrossRef]
- Kim, J.; Siverly, A.N.; Chen, D.; Wang, M.; Yuan, Y.; Wang, Y.; Lee, H.; Zhang, J.; Muller, W.J.; Liang, H. Ablation of miR-10b suppresses oncogene-induced mammary tumorigenesis and metastasis and reactivates tumor-suppressive pathways. Cancer Res. 2016, 76, 6424–6435. [Google Scholar] [CrossRef]
- Tufail, M.; Wu, C. SRPKs: A promising therapeutic target in cancer. Clin. Exp. Med. 2023, 23, 3093–3112. [Google Scholar] [CrossRef]
- Li, Q.; Zeng, C.; Liu, H.; Yung, K.W.Y.; Chen, C.; Xie, Q.; Zhang, Y.; Wan, S.W.C.; Mak, B.S.W.; Xia, J.; et al. Protein-protein interaction inhibitor of SRPKs alters the splicing isoforms of VEGF and inhibits angiogenesis. iScience 2021, 24, 102423. [Google Scholar] [CrossRef]
- Li, G.; Chen, H.; Shen, F.; Smithson, S.B.; Shealy, G.L.; Ping, Q.; Liang, Z.; Han, J.; Adams, A.C.; Li, Y.; et al. Targeting hepatic serine-arginine protein kinase 2 ameliorates alcohol-associated liver disease by alternative splicing control of lipogenesis. Hepatology 2023, 78, 1506–1524. [Google Scholar] [CrossRef]
- Bustos, F.; Segarra-Fas, A.; Nardocci, G.; Cassidy, A.; Antico, O.; Davidson, L.; Brandenburg, L.; Macartney, T.J.; Toth, R.; Hastie, C.J.; et al. Functional Diversification of SRSF Protein Kinase to Control Ubiquitin-Dependent Neurodevelopmental Signaling. Dev. Cell 2020, 55, 629–647.e7. [Google Scholar] [CrossRef] [PubMed]
- Chan, C.B.; Ye, K. Serine-arginine protein kinases: New players in neurodegenerative diseases? Rev. Neurosci. 2013, 24, 401–413. [Google Scholar] [CrossRef]
- Chowdhury, I.; Dashi, G.; Keskitalo, S. CMGC kinases in health and cancer. Cancers 2023, 15, 3838. [Google Scholar] [CrossRef]
- Tucker, B.A.; Scheetz, T.E.; Mullins, R.F.; DeLuca, A.P.; Hoffmann, J.M.; Johnston, R.M.; Jacobson, S.G.; Sheffield, V.C.; Stone, E.M. Exome sequencing and analysis of induced pluripotent stem cells identify the cilia-related gene male germ cell-associated kinase (MAK) as a cause of retinitis pigmentosa. Proc. Natl. Acad. Sci. USA 2011, 108, E569–E576. [Google Scholar] [CrossRef]
- Chen, T.-M.; Huang, Y.-T.; Wang, G.-C. Outcome of colon cancer initially presenting as colon perforation and obstruction. World J. Surg. Oncol. 2017, 15, 164. [Google Scholar] [CrossRef] [PubMed]
- Lahiry, P.; Wang, J.; Robinson, J.F.; Turowec, J.P.; Litchfield, D.W.; Lanktree, M.B.; Gloor, G.B.; Puffenberger, E.G.; Strauss, K.A.; Martens, M.B. A multiplex human syndrome implicates a key role for intestinal cell kinase in development of central nervous, skeletal, and endocrine systems. Am. J. Hum. Genet. 2009, 84, 134–147. [Google Scholar] [CrossRef]
- Pérez-Cabello, J.A.; Silvera-Carrasco, L.; Franco, J.M.; Capilla-González, V.; Armaos, A.; Gómez-Lima, M.; García-García, R.; Yap, X.W.; Leal-Lasarte, M.; Lall, D.; et al. MAPK/MAK/MRK overlapping kinase (MOK) controls microglial inflammatory/type-I IFN responses via Brd4 and is involved in ALS. Proc. Natl. Acad. Sci. USA 2023, 120, e2302143120. [Google Scholar] [CrossRef] [PubMed]
- Kilstrup-Nielsen, C.; Rusconi, L.; La Montanara, P.; Ciceri, D.; Bergo, A.; Bedogni, F.; Landsberger, N. What we know and would like to know about CDKL5 and its involvement in epileptic encephalopathy. Neural Plast. 2012, 2012, 728267. [Google Scholar] [CrossRef] [PubMed]
- Pestana Knight, E.M.; Olson, H.E. CDKL5 Deficiency Disorder: Some Lessons Learned 20 Years After the First Description. Am. J. Intellect. Dev. Disabil. 2024, 129, 101–109. [Google Scholar] [CrossRef]
- Bereshneh, A.H.; Andrews, J.C.; Eberl, D.F.; Bademci, G.; Borja, N.A.; Bivona, S.; Chung, W.K.; Yamamoto, S.; Wangler, M.F.; McKee, S.; et al. De novo variants in CDKL1 and CDKL2 are associated with neurodevelopmental symptoms. Am. J. Hum. Genet. 2025, 112, 846–862. [Google Scholar] [CrossRef]
- Qin, C.; Ren, L.; Ji, M.; Lv, S.; Wei, Y.; Zhu, D.; Lin, Q.; Xu, P.; Chang, W.; Xu, J. CDKL1 promotes tumor proliferation and invasion in colorectal cancer. OncoTargets Ther. 2017, 2017, 1613–1624. [Google Scholar] [CrossRef]
- Santamaría, D.; Barrière, C.; Cerqueira, A.; Hunt, S.; Tardy, C.; Newton, K.; Cáceres, J.F.; Dubus, P.; Malumbres, M.; Barbacid, M. Cdk1 is sufficient to drive the mammalian cell cycle. Nature 2007, 448, 811–815. [Google Scholar] [CrossRef]
- Diril, M.K.; Ratnacaram, C.K.; Padmakumar, V.C.; Du, T.; Wasser, M.; Coppola, V.; Tessarollo, L.; Kaldis, P. Cyclin-dependent kinase 1 (Cdk1) is essential for cell division and suppression of DNA re-replication but not for liver regeneration. Proc. Natl. Acad. Sci. USA 2012, 109, 3826–3831. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Ollikainen, M.; Sipilä, P.; Mustelin, L.; Wang, X.; Su, S.; Huan, T.; Levy, D.; Wilson, J.; Snieder, H. Genetic and environmental effects on gene expression signatures of blood pressure: A transcriptome-wide twin study. Hypertension 2018, 71, 457–464. [Google Scholar] [CrossRef] [PubMed]
- Sopić, M.; Ninić, A.; Ostanek, B.; Bojanin, D.; Milenković, T.; Munjas, J.; Mihajlović, M.; Vekić, J.; Marc, J.; Spasojević-Kalimanovska, V. Downregulation of mapk/mak/mrk overlapping kinase 1 in peripheral blood mononuclear cells of pediatric patients with type 1 diabetes mellitus. J. Med. Biochem. 2022, 41, 282. [Google Scholar] [CrossRef]
- Tsai, J.M.; Nowak, R.P.; Ebert, B.L.; Fischer, E.S. Targeted protein degradation: From mechanisms to clinic. Nat. Rev. Mol. Cell Biol. 2024, 25, 740–757. [Google Scholar] [CrossRef] [PubMed]


| Virus(es) | CMGC Kinase Involved | Context-Dependent Function | Mechanism | Experimental System Used |
|---|---|---|---|---|
| HPV (DNA virus) | Cyclin/CDK | Likely proviral |
| In vitro |
| Likely proviral |
| In vitro | ||
| Likely proviral |
| In vitro | ||
| HCMV (DNA virus) | Cyclin/CDK | Proviral |
| In vitro |
| Likely proviral |
| In vitro | ||
| Proviral |
| In vitro | ||
| Proviral |
| In vitro | ||
| HSV (DNA virus) | Cyclin/CDK | Antiviral |
| In vitro |
| Proviral |
| In vitro | ||
| Proviral |
| In vitro | ||
| Antiviral |
| In vitro | ||
| EBV (DNA virus) | Cyclin/CDK | Likely proviral |
| In vitro |
| Proviral |
| In vitro | ||
| Likely proviral |
| In vitro and In vivo | ||
| Likely proviral |
| In vitro | ||
| Likely antiviral |
| In vitro | ||
| VZV (DNA virus) | Cyclins | Likely proviral |
| In vitro |
| KSHV (DNA virus) | Cyclin/CDK | Likely proviral |
| In vitro and In vivo |
| Likely proviral |
| In vitro | ||
| Hepatitis B virus (DNA virus, but uses an RNA intermediate to replicate its genome) | Cyclin/CDK | Likely proviral |
| In vitro |
| Likely proviral |
| In vitro | ||
| HPV (DNA virus) | DYRK | Likely proviral |
| In vitro |
| Likely proviral |
| In vitro and clinical samples | ||
| Herpesviruses (DNA virus) | DYRK | Proviral |
| In vitro |
| Likely proviral |
| In vitro | ||
| Hepatitis B virus (DNA virus) | DYRK | Proviral |
| In vitro |
| Antiviral |
| In vitro | ||
| Pseudorabies virus (DNA virus) | DYRK | Proviral |
| In vitro and In vivo |
| EBV (DNA virus) | p38 MAPK | Proviral |
| In vitro |
| Proviral |
| In vitro | ||
| Proviral |
| In vitro | ||
| HCMV (DNA virus) | p38 MAPK | Likely proviral |
| In vitro |
| KSHV (DNA virus) | p38 MAPK | Proviral |
| In vitro |
| Proviral |
| In vitro | ||
| HSV-1 (DNA virus) | p38 MAPK | Proviral |
| In vitro |
| Proviral |
| In vitro | ||
| Proviral |
| In vitro | ||
| HBV (DNA virus) | p38 MAPK | Proviral |
| Clinical study |
| Likely proviral |
| In vitro | ||
| Simian Cytomegalovirus (DNA virus) | GSK3 | Likely proviral |
| In vitro |
| KHSV (DNA virus) | GSK3 | Likely proviral |
| In vitro |
| KHSV (DNA virus) | GSK3 | Antiviral |
| In vitro |
| HSV and HCMV (DNA virus) | GSK3 | Likely proviral |
| In vitro |
| Likely proviral |
| Ex vivo | ||
| Human adenovirus (DNA virus) | GSK3 | Proviral |
| In vitro |
| HBV (DNA virus) | GSK3 | Proviral |
| In vitro |
| Adenovirus (DNA virus) | CLK | Likely proviral |
| In vitro |
| HBV (DNA virus) | CLK | Proviral |
| In vitro |
| HPV16 (DNA virus) | SRPK | Proviral |
| In vitro |
| Likely antiviral |
| In vitro | ||
| Likely proviral |
| In vitro | ||
| HSV-1 (DNA virus) | SRPK | Likely antiviral |
| In vitro |
| VZV (DNA virus) | SRPK | Proviral |
| In vitro |
| EBV (DNA virus) | SRPK | Proviral |
| In vitro |
| HBV (DNA virus) | SRPK | Proviral |
| In vitro |
| Antiviral |
| In vitro | ||
| Proviral |
| In vitro | ||
| Proviral |
| In vitro | ||
| SARS-CoV2 (RNA virus) | Cyclin/CDKs | Likely antiviral |
| In vitro |
| Likely proviral |
| In vitro | ||
| Proviral |
| In vitro | ||
| Antiviral |
| In vitro | ||
| Influenza virus (RNA virus) | Cyclin/CDKs | Likely proviral |
| In vitro |
| Proviral |
| In vitro and In vivo | ||
| Avian reovirus p17 protein (RNA virus) | Cyclin/CDKs | Likely antiviral |
| In vitro |
| HCV (RNA virus) | Cyclin/CDKs | Likely proviral |
| In vitro and In vivo |
| Likely antiviral |
| In vitro | ||
| Likely proviral |
| Clinical study | ||
| Likely antiviral |
| Clinical study | ||
| Coronavirus Infectious Bronchitis Virus (RNA virus) | Cyclin | Likely antiviral |
| In vitro |
| MHV (RNA virus) | Cyclin | Likely antiviral |
| In vitro |
| Infectious Bursal Disease Virus (RNA virus) | Cyclin | Proviral |
| In vitro |
| ZIKV (RNA virus) | Cyclin/CDKs | Likely proviral |
| In vitro |
| TBEV (RNA virus) | Cyclin/CDKs | Likely proviral |
| In vitro and In vivo |
| JEV (RNA virus) | Cyclin/CDKs | Proviral |
| In vitro |
| SARS-CoV-2 (RNA virus) | p38 MAPK | Proviral |
| In vitro |
| Likely proviral |
| In vitro and In vivo | ||
| Likely proviral |
| In vitro and In vivo | ||
| Proviral |
| In vitro | ||
| Duck Tembusu virus (RNA virus) | p38 MAPK | Proviral |
| In vitro |
| Bovine parainfluenza virus type 3 (RNA virus) | p38 MAPK | Likely proviral |
| In vitro |
| CHIKV (RNA virus) | p38 MAPK | Proviral |
| In vitro |
| Proviral |
| In vitro | ||
| DENV (RNA virus) | p38 MAPK | Likely proviral |
| In vitro |
| Likely proviral |
| In vivo | ||
| Likely antiviral |
| In vitro and In vivo | ||
| RSV and IAV (RNA virus) | p38 MAPK | Proviral |
| In vitro |
| Newcastle disease virus (RNA virus) | p38 MAPK | Proviral |
| In vitro |
| Reovirus (RNA virus) | p38 MAPK | Proviral |
| In vitro |
| HCV (RNA virus) | p38 MAPK | Proviral |
| In vitro and clinical samples |
| Likely proviral |
| In vivo | ||
| Likely antiviral |
| In vitro and In vivo | ||
| Avian Reovirus (RNA virus) | p38 MAPK | Proviral |
| In vitro |
| Influenza virus (RNA virus) | p38 MAPK | Likely antiviral |
| In vitro |
| Likely proviral |
| In vitro and In vivo | ||
| Encephalomyocarditis (EMC) virus (RNA virus) | p38 MAPK | Proviral |
| In vitro |
| RSV (RNA virus) | p38 MAPK | Proviral |
| In vitro |
| Respiratory viruses (RNA virus) | p38 MAPK | Proviral |
| In vitro |
| Enterovirus (RNA virus) | p38 MAPK | Proviral |
| In vitro |
| Junin Virus (RNA virus) | p38 MAPK | Proviral |
| In vitro |
| Porcine epidemic diarrhea virus (PEDV) (RNA virus) | GSK3 | Proviral |
| In vitro |
| Snakehead vesiculovirus (SHVV) (RNA virus) | GSK3 | Proviral |
| In vitro |
| SARS-CoV-2 (RNA virus) | GSK3 | Proviral |
| In vitro and In vivo |
| Proviral |
| In vitro | ||
| Proviral |
| In vitro | ||
| HTLV (RNA virus) | GSK3 | Proviral |
| In vitro |
| HCV (RNA virus) | GSK3 | Proviral |
| In vitro |
| Likely antiviral |
| In silico | ||
| Proviral |
| In vitro | ||
| DENV (RNA virus) | GSK3 | Possibly antiviral |
| In vitro |
| HIV (RNA virus) | GSK3 | Proviral |
| In vitro |
| Proviral |
| In vitro | ||
| Coxsackievirus (RNA virus) | GSK3 | Proviral |
| In vitro |
| Influenza virus (RNA virus) | GSK3 | Proviral |
| In vitro and In vivo |
| Proviral |
| In vitro | ||
| Japanese encephalitis virus (RNA virus) | GSK3 | Proviral |
| In vitro |
| Coronaviruses (RNA virus) | DYRK | Proviral |
| In vitro |
| Proviral |
| In vitro | ||
| Sendai Virus (RNA virus) | DYRK | Possibly proviral |
| In vitro |
| HIV (RNA virus) | DYRK | Antiviral |
| In vitro |
| Antiviral |
| In vitro | ||
| HIV (RNA virus) | Cyclin/CDK | Proviral |
| In vitro |
| Proviral |
| In vitro | ||
| Proviral |
| In vitro | ||
| Proviral |
| In vitro | ||
| Proviral |
| In vitro | ||
| HTLV (RNA virus) | Cyclin/CDK | Likely proviral |
| In vitro |
| Likely antiviral |
| In vitro | ||
| Likely antiviral |
| In vitro | ||
| Likely proviral |
| In vitro | ||
| Likely proviral |
| In vitro | ||
| HCV (RNA virus) | SRPK | Proviral |
| In vitro |
| SARS-CoV-2 (RNA virus) | SRPK | Proviral |
| In vitro |
| Likely proviral |
| Clinical study | ||
| HIV and Sindbis virus (RNA virus) | SRPK | Proviral |
| In vitro |
| EBOLA (RNA virus) | SRPK | Proviral |
| In vitro |
| HIV (RNA virus) | CLK | Antiviral |
| In vitro |
| Proviral |
| In vitro | ||
| Proviral |
| In vitro | ||
| Proviral |
| Ex vivo | ||
| Influenza virus (RNA virus) | CLK | Proviral |
| In vitro and In vivo |
| SARS-CoV-2 (RNA virus) | CLK | Likely proviral |
| In vitro |
| Sindbis virus (RNA virus) | CDKL5 | Antiviral |
| In vitro and In vivo |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Amusan, O.T.; Guo, H. CMGC Kinases in Viral Infection and Human Disease. Pathogens 2026, 15, 366. https://doi.org/10.3390/pathogens15040366
Amusan OT, Guo H. CMGC Kinases in Viral Infection and Human Disease. Pathogens. 2026; 15(4):366. https://doi.org/10.3390/pathogens15040366
Chicago/Turabian StyleAmusan, Oluwamuyiwa T., and Hongyan Guo. 2026. "CMGC Kinases in Viral Infection and Human Disease" Pathogens 15, no. 4: 366. https://doi.org/10.3390/pathogens15040366
APA StyleAmusan, O. T., & Guo, H. (2026). CMGC Kinases in Viral Infection and Human Disease. Pathogens, 15(4), 366. https://doi.org/10.3390/pathogens15040366






