Comparative Efficacy of a Novel Topical Formulation with Antimicrobial Peptides and Encapsulated Plant Extracts Versus Conventional Therapies for Canine Otitis Externa
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Ethics Statement
2.2. Diagnosis and OTIS-3
2.3. Pruritus Evaluation
2.4. pH Measurement
2.5. Cytological Evaluation
2.6. Microbiological Assays
2.7. Statistical Analysis
3. Results
3.1. Dogs Included in the Present Study
3.2. Evaluation of Efficacy and pH Evaluation
3.3. Microbiological Identification
3.4. Statistical Analysis of Clinical Parameters, Pruritus, pH and Cytology
3.4.1. Comparison of Time Points Between Groups
3.4.2. Comparison of Time Points Within Groups
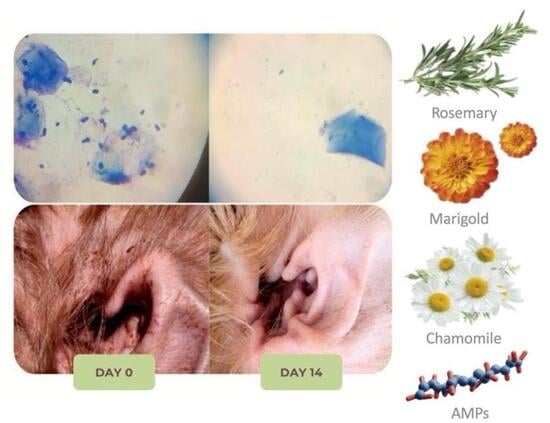
3.5. Clinical Cases
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| OE | Otitis externa |
| AMR | Antimicrobial resistance |
| AD | Atopic dermatitis |
| AMP(s) | Antimicrobial peptide(s) |
| OTIS-3 | Otitis Index Scoring System |
| pVAS | Pruritus Visual Analog Scale |
| PUCPR | Pontifícia Universidade Católica do Paraná |
| GA | Group A (Therapy A: antimicrobial peptides + plant extracts) |
| GB | Group B (Therapy B: gentamicin + betamethasone valerate + clotrimazole) |
| MSD | Merck Sharp & Dohme Animal Health |
| MALDI-TOF MS | Matrix-Assisted Laser Desorption/Ionization–Time of Flight Mass Spectrometry |
| CLSI | Clinical and Laboratory Standards Institute |
| CNPq | Conselho Nacional de Desenvolvimento Científico e Tecnológico (Brazilian National Council for Scientific and Technological Development) |
| MDR | Multidrug resistant |
References
- Perry, L.R.; MacLennan, B.; Korven, R.; Rawlings, T.A. Epidemiological study of dogs with otitis externa in Cape Breton, Nova Scotia. Can. Vet. J. 2017, 58, 168–174. [Google Scholar] [PubMed]
- Rougier, S.; Borell, D.; Pheulpin, S.; Woehrlé, F.; Boisramé, B. A comparative study of two antimicrobi-al/anti-inflammatory formulations in the treatment of canine otitis externa. Vet. Dermatol. 2005, 16, 299–307. [Google Scholar] [CrossRef]
- Angus, J.C.; Lichtensteiger, C.; Campbell, L.; Schaeffer, D.J. Breed variations in histopathologic features of chronic severe otitis externa in dogs: 80 cases (1995–2001). J. Am. Vet. Med. Assoc. 2002, 221, 1000–1006. [Google Scholar] [CrossRef]
- Chan, W.Y.; Khazandi, M.; Hickey, E.E.; Page, S.W.; Trott, D.J.; Hill, P.B. In vitro antimicrobial activity of seven adjuvants against common pathogens associated with canine otitis externa. Vet. Dermatol. 2019, 30, 133–138. [Google Scholar] [CrossRef]
- Nuttall, T. Managing recurrent otitis externa in dogs: What have we learned and what can we do better? J. Am. Vet. Med. Assoc. 2023, 261, S10–S22. [Google Scholar] [CrossRef] [PubMed]
- Santoro, D.; Marsella, R.; Pucheu-Haston, C.M.; Eisenschenk, M.N.C.; Nuttall, T.; Bizikova, P. Review: Pathogenesis of canine atopic dermatitis: Skin barrier and host–micro-organism interaction. Vet. Dermatol. 2015, 26, 84-e25. [Google Scholar] [CrossRef]
- Korbelik, J.; Singh, A.; Rousseau, J.; Weese, J.S. Characterization of the otic bacterial microbiota in dogs with otitis externa compared to healthy individuals. Vet. Dermatol. 2019, 30, 228–270. [Google Scholar] [CrossRef] [PubMed]
- Bajwa, J. Canine otitis externa–Treatment and complications. Can. Vet. J. 2019, 60, 97–99. [Google Scholar]
- Hensel, P.; Austel, M.; Wooley, R.E.; Keys, D.; Ritchie, B.W. In vitro and in vivo evaluation of a potentiated miconazole aural solution in chronic Malassezia otitis externa in dogs. Vet. Dermatol. 2009, 20, 429–434. [Google Scholar] [CrossRef]
- Cain, C.L. Antimicrobial resistance in staphylococci in small animals. Vet. Clin. North. Am. Small Anim. Pract. 2013, 43, 19–40. [Google Scholar] [CrossRef]
- Murray, C.J.; Ikuta, K.S.; Sharara, F.; Swetschinski, L.; Aguilar, G.R.; Gray, A.; Tasak, N. Global burden of bacterial antimicrobial resistance in 2019: A systematic analysis. Lancet 2022, 399, 629–655. [Google Scholar] [CrossRef]
- Beever, L.; Bond, R.; Graham, P.A.; Jackson, B.; Lloyd, D.H.; Loeffler, A. Increasing antimicrobial resistance in clinical isolates of Staphylococcus intermedius group bacteria and emergence of MRSP in the UK. Vet. Rec. 2015, 176, 172. [Google Scholar] [CrossRef] [PubMed]
- Lozano, C.; Rezusta, A.; Ferrer, I.; Pérez-Laguna, V.; Zarazaga, M.; Ruiz-Ripa, L.; Torres, C. Staphylococcus pseudintermedius human infection cases in Spain: Dog-to-human transmission. Vector Borne Zoonotic Dis. 2017, 17, 268–270. [Google Scholar] [CrossRef]
- Akhtar, G.; Faried, H.N.; Razzaq, K.; Ullah, S.; Wattoo, F.M.; Shehzad, M.A.; Sajjad, Y.; Ahsan, M.; Javed, T.; Dessoky, E.S.; et al. Chitosan-induced physiological and biochemical regulations confer drought tolerance in pot marigold (Calendula officinalis L.). Agronomy 2022, 12, 474. [Google Scholar] [CrossRef]
- Dawid-Pać, R. Medicinal plants used in treatment of inflammatory skin diseases. Postepy Dermatol. Alergol. 2013, 30, 170–177. [Google Scholar] [CrossRef]
- Weber, N.; Biehler, K.; Schwabe, K.; Haarhaus, B.; Quirin, K.W.; Frank, U.; Wölfle, U. Hop extract acts as an antioxidant with antimicrobial effects against Propionibacterium acnes and Staphylococcus aureus. Molecules 2019, 24, 223. [Google Scholar] [CrossRef]
- Ibrahim, N.; Abbas, H.; El-Sayed, N.S.; Gad, H.A. Rosmarinus officinalis L. hexane extract: Phytochemical analysis, nanoencapsulation, and in silico, in vitro, and in vivo anti-photoaging potential evaluation. Sci. Rep. 2022, 12, 13102. [Google Scholar] [CrossRef]
- Santoro, D.; Marsella, R.; Bunick, D.; Graves, T.K.; Campbell, K.L. Expression and distribution of canine anti-microbial peptides in the skin of healthy and atopic beagles. Vet. Immunol. Immunopathol. 2011, 144, 382–388. [Google Scholar] [CrossRef] [PubMed]
- Santoro, D. Comparison of the quantity and antimicrobial activity of host defence peptides in ear canals between healthy and atopic dogs: A preliminary study. Vet. Dermatol. 2023, 34, 452–459. [Google Scholar] [CrossRef]
- Li, T.; Ren, X.; Luo, X.; Wang, Z.; Li, Z.; Luo, X.; Cheng, F. A foundation model identifies broad-spectrum antimicrobial peptides against drug-resistant bacterial infection. Nat. Commun. 2024, 15, 7538. [Google Scholar] [CrossRef] [PubMed]
- Nuttall, T.; Bensignor, E. A pilot study to develop an objective clinical score from canine otitis externa. Vet. Dermatol. 2014, 25, 530–537. [Google Scholar] [CrossRef]
- Hill, P.B.; Lau, P.; Rybnicek, J. Development of an owner-assessed scale to measure the severity of pruritus in dogs. Vet. Dermatol. 2007, 18, 301–308. [Google Scholar] [CrossRef]
- Rybnicek, J.; Lau-Gillard, P.J.; Harvey, R.; Hill, P.B. Further validation of a pruritus severity scale for use in dogs. Vet. Dermatol. 2009, 20, 115–122. [Google Scholar] [CrossRef] [PubMed]
- Young, A.J.; Torres, S.M.F.; Koch, S.N.; Eisenschenk, M.N.C.; Rendahl, A.K. Canine Pruritus Visual Analog. Scale: How does it capture owners’ perception of their pet’s itching level? Vet. Dermatol. 2019, 30, 377–381. [Google Scholar] [CrossRef] [PubMed]
- Bae, S.; Jin, Y.; Oh, T. In vivo and in vitro efficacy of florfenicol, terbinafine and mometasone furoate topical otic solution for treatment of canine otitis externa. Korean J. Vet. Res. 2021, 61, 37. [Google Scholar] [CrossRef]
- Diaz-Campos, D.V.; Lubbers, B.V.; Schwarz, S.; Bowden, R.; Burbick, C.R.; Govendir, M.; Scott, B.; Killian, S.B.; Lawhonm, S.D.; Li, X.-Z.; et al. Performance Standards for Antimicrobial Disk and Dilution Susceptibility Tests for Bacteria Isolated from Animals, 7th ed; CLSI Supplement VET01S; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2024. [Google Scholar]
- Ludwig, L.; Tsukui, T.; Kageyama, M.; Farias, M. Evaluation of sensitization to the crude extract of Dermatophagoides farinae and its derived allergens, Der f 2 and Zen 1, in dogs with atopic dermatitis in Southern Brazil. Vet. Immunol. Immunopathol. 2021, 234, 110199. [Google Scholar] [CrossRef]
- Favrot, C.; Steffan, J.; Seewald, W.; Picco, F. A prospective study on the clinical features of chronic canine atopic dermatitis and its diagnosis. Vet. Dermatol. 2010, 21, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Tresch, M.; Mevissen, M.; Ayrle, H.; Melzig, M.; Roosje, P.; Walkenhorst, M. Medicinal plants as therapeutic options for topical treatment in canine dermatology? A systematic review. BMC Vet. Res. 2019, 15, 174. [Google Scholar] [CrossRef]
- Farsi, S.; Chaudhry, S.; Khan, A.; Gardner, J.; Ogwo, M.; Ofori, B.; Cervantes, J. Antimicrobial effect of chamomile-containing over-the-counter ear and eye drops. J. Investig. Med. 2024, 72, 305–311. [Google Scholar] [CrossRef]
- Nogueira, J.C.R.; Diniz, M.F.M.; Lima, E.O. In vitro antimicrobial activity of plants in Acute Otitis Externa. Rev. Bras. Otorrinolaringol. 2008, 74, 118–124. [Google Scholar] [CrossRef]
- Vercelli, C.; Pasquetti, M.; Giovannetti, G.; Visioni, S.; Re, G.; Giorgi, M.; Peano, A. In vitro and in vivo evaluation of a new phytotherapic blend to treat acute externa otitis in dogs. J. Vet. Pharmacol. Ther. 2021, 44, 910–918. [Google Scholar] [CrossRef] [PubMed]
- Gmyterco, V.C.; Luciano, F.B.; Ludwig, L.A.; Evangelista, A.G.; Ferreira, T.S.; Borek, F.; de Farias, M.R. Comparative study of a commercial formula containing natural antimicrobials versus oral cephalexin or topical chlorhexidine-miconazole therapies for treating superficial pyoderma in dogs. Vet. Dermatol. 2025, 36, 137–147. [Google Scholar] [CrossRef]
- Lee, J.; Jung, E.; Koh, J.; Kim, Y.S.; Park, D. Effect of rosmarinic acid on atopic dermatitis. J. Dermatol. 2008, 35, 768–771. [Google Scholar] [CrossRef] [PubMed]
- Matousek, J.L.; Campbell, K.L. A comparative review of cutaneous pH. Vet. Dermatol. 2002, 13, 293–300. [Google Scholar] [CrossRef]
- Mittal, A.; Kumar, S. Role of pH of External Auditory Canal in Acute Otitis Externa. Indian. J. Otolaryngol. Head. Neck Surg. 2014, 66, 86–91. [Google Scholar] [CrossRef]
- Angelova-Fischer, I.; Dapic, I.; Hoek, A.K.; Jakasa, I.; Fischer, T.W.; Zillikens, D.; Kezic, S. Skin barrier integrity and natural moisturising factor levels after cumulative dermal exposure to alkaline agents in atopic dermatitis. Acta Derm. Venereol. 2014, 94, 640–644. [Google Scholar] [CrossRef]
- Ferreira, T.C.; Ferreira, T.M.V.; Pinheiro, B.Q.; Verde, J.F.L.; Júnior, J.A.S.; Nunes-Pinheiro, D.C.S. Terapia otológica induz imunotoxicidade em cão. Cienc. Anim. 2023, 29, 155. [Google Scholar]
- Penna, B.; Varges, R.; Medeiros, L.; Martins, G.M.; Martins, R.R.; Lilenbaum, W. Species distribution and antimicrobial susceptibility of staphylococci isolated from canine otitis externa. Vet. Dermatol. 2010, 21, 292–296. [Google Scholar] [CrossRef]
- Weese, J.S. The canine and feline skin microbiome in health and disease. Vet. Dermatol. 2013, 24, 137–145. [Google Scholar] [CrossRef]
- Igimi, S.; Takahashi, E.; Mitsuoka, T. Staphylococcus schleiferi subsp. coagulans subsp. nov., isolated from the external auditory meatus of dogs with external ear otitis. Int. J. Syst. Bacteriol. 1990, 40, 409–411. [Google Scholar] [CrossRef] [PubMed]
- May, E.R.; Hnilica, K.A.; Frank, L.A.; Jones, R.D.; Bemis, D.A. Isolation of Staphylococcus schleiferi from healthy dogs and dogs with otitis, pyoderma, or both. J. Am. Vet. Med. Assoc. 2005, 227, 928–931. [Google Scholar] [CrossRef]
- Yamashita, K.; Shimizu, A.; Kawano, J.; Uchida, E.; Haruna, A.; Igimi, S. Isolation and characterization of staphylococci from external auditory meatus of dogs with or without otitis externa with special reference to Staphylococcus schleiferi subsp. coagulans isolates. J. Vet. Med. Sci. 2005, 67, 263–268. [Google Scholar] [CrossRef] [PubMed]
- Palomino-Farfán, J.A.; Alvarez, L.V.; Siuce, M.J.; Calle, S.E. Antimicrobial resistance in coagulase-positive staphylococci (CoPS) isolated from dogs with external otitis. Rev. Investig. Vet. Peru 2020, 31, e17558. [Google Scholar] [CrossRef]
- Madhaiyan, M.; Wirth, J.S.; Saravanan, V.S. Phylogenomic analyses of the Staphylococcaceae family suggest the reclassification of five species within the genus Staphylococcus as heterotypic synonyms, the promotion of five subspecies to novel species, the taxonomic reassignment of five Staphylococcus species to Mammaliicoccus gen. nov., and the formal assignment of Nosocomiicoccus to the family Staphylococcaceae. Int. J. Syst. Evol. Microbiol. 2020, 70, 5926–5936. [Google Scholar] [CrossRef]
- Sewid, A.H.; Kania, S.A. Distinguishing characteristics of Staphylococcus schleiferi and Staphylococcus coagulans of human and canine origin. PLoS ONE 2024, 19, e0296850. [Google Scholar] [CrossRef]
- Jones, R.D.; Kania, S.A.; Rohrbach, B.W.; Frank, L.A.; Bemis, D.A. Prevalence of oxacillin- and multidrug-resistant staphylococci in clinical samples from dogs: 1772 samples (2001–2005). J. Am. Vet. Med. Assoc. 2007, 230, 221–227. [Google Scholar] [CrossRef]
- Cain, C.L.; Morris, D.O.; O’Shea, K.; Rankin, S.C. Genotypic relatedness and phenotypic characterization of Staphylococcus schleiferi subspecies in clinical samples from dogs. Am. J. Vet. Res. 2011, 72, 96–102. [Google Scholar] [CrossRef]
- Nguyen, A.D.K.; Moran, D.; Eland, C.L.; Wilks, K. Staphylococcus schleiferi subspecies coagulans septic shock in an immunocompetent male following canine otitis externa. Turk. J. Emerg. Med. 2023, 23, 184–187. [Google Scholar] [CrossRef]
- Petrov, V.; Mihaylov, G.; Tsachev, I.; Zhelev, G.; Marutsov, P.; Koev, K. Otitis externa in dogs: Microbiology and antimicrobial susceptibility. Rev. Med. Vet. 2013, 164, 18–22. [Google Scholar]
- Devriese, L.A.; Vancanneyt, M.; Baele, M.; Vaneechoutte, M.; De Graef, E.; Snauwaert, C.; Haesebrouck, F. Staphylococcus pseudintermedius sp. nov., a coagulase-positive species from animals. Int. J. Syst. Evol. Microbiol. 2005, 55, 1569–1573. [Google Scholar] [CrossRef] [PubMed]
- Bannoehr, J.; Guardabassi, L. Staphylococcus pseudintermedius in the dog: Taxonomy, diagnostics, ecology, epidemiology and pathogenicity. Vet. Dermatol. 2012, 23, 253–266. [Google Scholar] [CrossRef] [PubMed]
- Lilenbaum, W.; Veras, M.; Blum, E.; Souza, G.N. Antimicrobial susceptibility of staphylococci isolated from otitis externa in dogs. Lett. Appl. Microbiol. 2000, 31, 42–45. [Google Scholar] [CrossRef]
- Oliveira, L.C.; Leite, C.A.L.; Brilhante, C.B.M.; Carvalho, C.B.M. Etiology of canine otitis media and antimicrobial susceptibility of coagulase-positive staphylococci in Fortaleza City, Brazil. Braz. J. Microbiol. 2006, 37, 144–147. [Google Scholar] [CrossRef][Green Version]
- Lyskova, P.; Vydrzalova, M.; Mazurova, J. Identification and antimicrobial susceptibility of bacteria and yeasts isolated from healthy dogs and dogs with otitis externa. J. Vet. Med. A Physiol. Pathol. Clin. Med. 2007, 54, 559–563. [Google Scholar] [CrossRef] [PubMed]



| ID | Group | Breed | Sex | Age | Surgically Sterilized | Ear Affected |
|---|---|---|---|---|---|---|
| C-01 | A | Lhasa Apso | Male | 14 | Yes | Both |
| C-02 | A | Lhasa Apso | Female | 16 | Yes | Right |
| C-03 | A | Lhasa Apso | Female | 7 | Yes | Both |
| C-04 | B | Beagle | Male | 7 | Yes | Left |
| C-05 | A | German Shephard | Female | 7 | No | Both |
| C-06 | B | Shih Tzu | Male | 8 | Yes | Both |
| C-07 | B | Lhasa Apso | Female | 6 | Yes | Left |
| C-08 | B | Shih Tzu | Male | 7 | Yes | Both |
| C-09 | A | Golden Retriever | Female | 3 | Yes | Both |
| C-10 | B | Golden Retriever | Male | 1 | Yes | Both |
| C-11 | A | Pit Bull | Female | 3 | No | Both |
| C-12 | B | Mixed | Male | 12 | Yes | Both |
| C-13 | B | Shar-Pei | Female | 2 | Yes | Both |
| C-14 | B | Shih Tzu | Female | 4 | Yes | Both |
| C-15 | B | Lhasa Apso | Male | 10 | Yes | Both |
| C-17 | A | Mixed | Female | 3 | Yes | Left |
| C-18 | A | Mixed | Male | 3 | Yes | Both |
| C-19 | A | Schnauzer | Female | 5 | Yes | Right |
| C-20 | B | Golden Retriever | Female | 4 | Yes | Right |
| C-21 | A | Dachshund | Female | 10 | Yes | Both |
| C-22 | B | Shih Tzu | Male | 10 | Yes | Both |
| PG-01 | A | Shih Tzu | Female | 5 | Yes | Both |
| PG-02 | A | Lhasa Apso | Female | 4 | Yes | Both |
| PG-03 | B | Poodle | Female | 7 | Yes | Both |
| PG-04 | A | Lhasa Apso | Male | 8 | Yes | Both |
| PG-05 | A | Mixed | Male | 6 | Yes | Both |
| PG-06 | A | Lhasa Apso | Male | 1 | Yes | Both |
| PG-07 | B | Mixed | Female | 6 | Yes | Both |
| PG-09 | A | French bulldog | Male | 2 | Yes | Both |
| PG-09 | B | Chihuahua | Female | 3 | Yes | Both |
| PG-10 | A | Mixed | Female | 3 | Yes | Both |
| PG-11 | B | Lhasa | Male | 8 | Yes | Both |
| PG-12 | B | Male | Male | 9 | Yes | Both |
| PG-13 | B | Labrador | Male | 3 | Yes | Both |
| PG-14 | B | Mixed | Male | 4 | Yes | Both |
| PG-15 | A | Lhasa Apso | Male | 2 | Yes | Both |
| PG-16 | B | Mixed | Female | 8 | Yes | Both |
| PG-17 | A | Mixed | Male | 5 | Yes | Both |
| Microorganism | Number of Isolates | Proportion (%) |
|---|---|---|
| Bacillus cereus | 3 | 5.0 |
| Bacillus megaterium | 2 | 3.33 |
| Bacillus pumillus | 2 | 3.33 |
| Enterococcus canintestini | 1 | 1.67 |
| Enterococcus faecalis | 1 | 1.67 |
| Escherichia coli | 2 | 3.33 |
| Klebsiella planticola | 1 | 1.67 |
| Klebsiella variicola | 1 | 1.67 |
| Leclercia adecarboxylata | 1 | 1.67 |
| Malassezia pachydermatis | 3 | 5.0 |
| Paenibacillus lautus | 1 | 1.67 |
| Paenibacillus nematophilus | 1 | 1.67 |
| Proteus mirabilis | 9 | 15.0 |
| Pseudomonas aeruginosa | 4 | 6.67 |
| Pseudomonas koreensis | 1 | 1.67 |
| Psychrobacter lutiphocae | 1 | 1.67 |
| Raoultella (K.) ornithinolytica | 1 | 1.67 |
| Staphylococcus coagulans | 10 | 16.7 |
| Staphylococcus intermedius | 1 | 1.67 |
| Staphylococcus pseudintermedius | 6 | 10.0 |
| Staphylococcus schleiferi | 4 | 6.67 |
| Staphylococcus saprophyticus subsp. saprophyticus | 1 | 1.67 |
| Staphylococcus warneri | 1 | 1.67 |
| Weissella cibaria | 1 | 1.67 |
| Weissella confusa | 1 | 1.67 |
| Total | 60 | 100.0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bannach, T.C.; Mongruel, A.C.B.; Evangelista, A.G.; de Souza, V.B.M.; Voi, R.; Otuki, M.F.; de Farias, M.R.; Luciano, F.B. Comparative Efficacy of a Novel Topical Formulation with Antimicrobial Peptides and Encapsulated Plant Extracts Versus Conventional Therapies for Canine Otitis Externa. Pathogens 2025, 14, 1112. https://doi.org/10.3390/pathogens14111112
Bannach TC, Mongruel ACB, Evangelista AG, de Souza VBM, Voi R, Otuki MF, de Farias MR, Luciano FB. Comparative Efficacy of a Novel Topical Formulation with Antimicrobial Peptides and Encapsulated Plant Extracts Versus Conventional Therapies for Canine Otitis Externa. Pathogens. 2025; 14(11):1112. https://doi.org/10.3390/pathogens14111112
Chicago/Turabian StyleBannach, Tatiana Charello, Anna Claudia Baumel Mongruel, Alberto Gonçalves Evangelista, Vitória Brigida Mielnik de Souza, Renata Voi, Michel Fleith Otuki, Marconi Rodrigues de Farias, and Fernando Bittencourt Luciano. 2025. "Comparative Efficacy of a Novel Topical Formulation with Antimicrobial Peptides and Encapsulated Plant Extracts Versus Conventional Therapies for Canine Otitis Externa" Pathogens 14, no. 11: 1112. https://doi.org/10.3390/pathogens14111112
APA StyleBannach, T. C., Mongruel, A. C. B., Evangelista, A. G., de Souza, V. B. M., Voi, R., Otuki, M. F., de Farias, M. R., & Luciano, F. B. (2025). Comparative Efficacy of a Novel Topical Formulation with Antimicrobial Peptides and Encapsulated Plant Extracts Versus Conventional Therapies for Canine Otitis Externa. Pathogens, 14(11), 1112. https://doi.org/10.3390/pathogens14111112