Epidemiological Survey of the Main Tick-Borne Pathogens Infecting Dogs from the Republic of Moldova
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Sampling
2.2. Molecular Techniques for Vector-Borne Pathogen Identification
2.3. Statistical Analysis
3. Results
3.1. Study Population
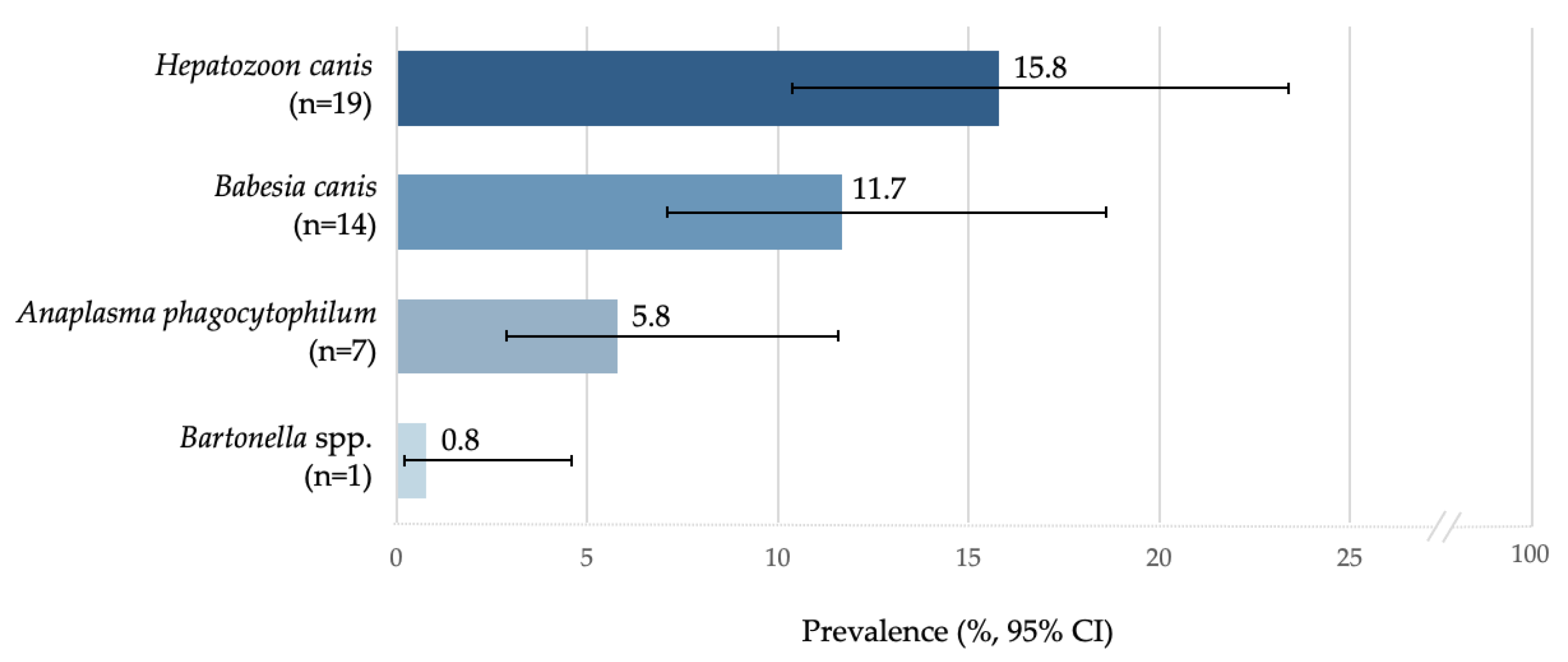
3.2. Molecular Analysis Outcomes
3.3. Risk Factors for Tick-Borne Pathogens
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- de la Fuente, J.; Estrada-Pena, A.; Venzal, J.M.; Kocan, K.M.; Sonenshine, D.E. Overview: Ticks as vectors of pathogens that cause disease in humans and animals. Front. Biosci. 2008, 13, 6938–6946. [Google Scholar] [CrossRef]
- Rochlin, I.; Toledo, A. Emerging tick-borne pathogens of public health importance: A mini-review. J. Med. Microbiol. 2020, 69, 781–791. [Google Scholar] [CrossRef]
- Boulanger, N.; Boyer, P.; Talagrand-Reboul, E.; Hansmann, Y. Ticks and tick-borne diseases. Med. Mal. Infect. 2019, 49, 87–97. [Google Scholar] [CrossRef]
- Jaenson, T.G.; Hjertqvist, M.; Bergström, T.; Lundkvist, A. Why is tick-borne encephalitis increasing? A review of the key factors causing the increasing incidence of human TBE in Sweden. Parasit. Vectors 2012, 5, 184. [Google Scholar] [CrossRef]
- Cayol, C.; Koskela, E.; Mappes, T.; Siukkola, A.; Kallio, E.R. Temporal dynamics of the tick Ixodes ricinus in northern Europe: Epidemiological implications. Parasit. Vectors 2017, 10, 166. [Google Scholar] [CrossRef] [PubMed]
- Černý, J.; Lynn, G.; Hrnková, J.; Golovchenko, M.; Rudenko, N.; Grubhoffer, L. Management Options for Ixodes ricinus-Associated Pathogens: A Review of Prevention Strategies. Int. J. Environ. Res. Public Health 2020, 17, 1830. [Google Scholar] [CrossRef] [PubMed]
- Johnson, N.; Phipps, L.P.; Hansford, K.M.; Folly, A.J.; Fooks, A.R.; Medlock, J.M.; Mansfield, K.L. One Health Approach to Tick and Tick-Borne Disease Surveillance in the United Kingdom. Int. J. Environ. Res. Public Health 2022, 19, 5833. [Google Scholar] [CrossRef]
- Mac, S.; da Silva, S.R.; Sander, B. The economic burden of Lyme disease and the cost-effectiveness of Lyme disease interventions: A scoping review. PLoS ONE 2019, 14, e0210280. [Google Scholar] [CrossRef] [PubMed]
- Estrada-Peña, A.; Salman, M. Current Limitations in the Control and Spread of Ticks that Affect Livestock: A Review. Agriculture 2013, 3, 221–235. [Google Scholar] [CrossRef]
- Rubel, F.; Brugger, K.; Pfeffer, M.; Chitimia-Dobler, L.; Didyk, Y.M.; Leverenz, S.; Dautel, H.; Kahl, O. Geographical distribution of Dermacentor marginatus and Dermacentor reticulatus in Europe. Ticks Tick Borne Dis. 2016, 7, 224–233. [Google Scholar] [CrossRef]
- Cunze, S.; Glock, G.; Kochmann, J.; Klimpel, S. Ticks on the move-climate change-induced range shifts of three tick species in Europe: Current and future habitat suitability for Ixodes ricinus in comparison with Dermacentor reticulatus and Dermacentor marginatus. Parasitol. Res. 2022, 121, 2241–2252. [Google Scholar] [CrossRef] [PubMed]
- Rigaud, E.; Jaulhac, B.; Garcia-Bonnet, N.; Hunfeld, K.P.; Féménia, F.; Huet, D.; Goulvestre, C.; Vaillant, V.; Deffontaines, G.; Abadia-Benoist, G. Seroprevalence of seven pathogens transmitted by the Ixodes ricinus tick in forestry workers in France. Clin. Microbiol. Infect. 2016, 22, e731–e739. [Google Scholar] [CrossRef] [PubMed]
- Capligina, V.; Salmane, I.; Keišs, O.; Vilks, K.; Japina, K.; Baumanis, V.; Ranka, R. Prevalence of tick-borne pathogens in ticks collected from migratory birds in Latvia. Ticks Tick Borne Dis. 2014, 5, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Morozov, A.; Tischenkov, A.; Silaghi, C.; Proka, A.; Toderas, I.; Movila, A.; Frickmann, H.; Poppert, S. Prevalence of Bacterial and Protozoan Pathogens in Ticks Collected from Birds in the Republic of Moldova. Microorganisms 2022, 10, 1111. [Google Scholar] [CrossRef]
- Movila, A.; Gatewood, A.; Toderas, I.; Duca, M.; Papero, M.; Uspenskaia, I.; Conovalov, J.; Fish, D. Prevalence of Borrelia burgdorferi sensu lato in Ixodes ricinus and I. lividus ticks collected from wild birds in the Republic of Moldova. Int. J. Med. Microbiol. 2008, 298, 149–153. [Google Scholar] [CrossRef]
- Movila, A.; Rolain, J.M.; Podavalenko, A.; Toderas, I.; Tkachenco, L.; Naglov, V.; Raoult, D. Detection of spotted fever group Rickettsiae and family Anaplasmataceae in Ixodes ricinus ticks from Republic of Moldova and Eastern Ukraine. Clin. Microbiol. Infect. 2009, 15 (Suppl. 2), 32–33. [Google Scholar] [CrossRef][Green Version]
- Skotarczak, B. The role of companion animals in the environmental circulation of tick-borne bacterial pathogens. Ann. Agric. Environ. Med. 2018, 25, 473–480. [Google Scholar] [CrossRef]
- Hodžić, A.; Alić, A.; Fuehrer, H.P.; Harl, J.; Wille-Piazzai, W.; Duscher, G.G. A molecular survey of vector-borne pathogens in red foxes (Vulpes vulpes) from Bosnia and Herzegovina. Parasit. Vectors 2015, 8, 88. [Google Scholar] [CrossRef]
- Barbour, A.G.; Maupin, G.O.; Teltow, G.J.; Carter, C.J.; Piesman, J. Identification of an uncultivable Borrelia species in the hard tick Amblyomma americanum: Possible agent of a Lyme disease-like illness. J. Infect. Dis. 1996, 173, 403–409. [Google Scholar] [CrossRef]
- Clark, K.L.; Leydet, B.; Hartman, S. Lyme borreliosis in human patients in Florida and Georgia, USA. Int. J. Med. Sci. 2013, 10, 915–931. [Google Scholar] [CrossRef]
- Regnery, R.L.; Spruill, C.L.; Plikaytis, B.D. Genotypic identification of rickettsiae and estimation of intraspecies sequence divergence for portions of two rickettsial genes. J. Bacteriol. 1991, 173, 1576–1589. [Google Scholar] [CrossRef] [PubMed]
- Norman, A.F.; Regnery, R.; Jameson, P.; Greene, C.; Krause, D.C. Differentiation of Bartonella-like isolates at the species level by PCR-restriction fragment length polymorphism in the citrate synthase gene. J. Clin. Microbiol. 1995, 33, 1797–1803. [Google Scholar] [CrossRef] [PubMed]
- Sjöstedt, A.; Eriksson, U.; Berglund, L.; Tärnvik, A. Detection of Francisella tularensis in ulcers of patients with tularemia by PCR. J. Clin. Microbiol. 1997, 35, 1045–1048. [Google Scholar] [CrossRef]
- Alberti, A.; Zobba, R.; Chessa, B.; Addis, M.F.; Sparagano, O.; Pinna Parpaglia, M.L.; Cubeddu, T.; Pintori, G.; Pittau, M. Equine and canine Anaplasma phagocytophilum strains isolated on the island of Sardinia (Italy) are phylogenetically related to pathogenic strains from the United States. Appl. Environ. Microbiol. 2005, 71, 6418–6422. [Google Scholar] [CrossRef] [PubMed]
- Murphy, G.L.; Ewing, S.A.; Whitworth, L.C.; Fox, J.C.; Kocan, A.A. A molecular and serologic survey of Ehrlichia canis, E. chaffeensis, and E. ewingii in dogs and ticks from Oklahoma. Vet. Parasitol. 1998, 79, 325–339. [Google Scholar] [CrossRef]
- Siarkou, V.I.; Mylonakis, M.E.; Bourtzi-Hatzopoulou, E.; Koutinas, A.F. Sequence and phylogenetic analysis of the 16S rRNA gene of Ehrlichia canis strains in dogs with clinical monocytic ehrlichiosis. Vet. Microbiol. 2007, 125, 304–312. [Google Scholar] [CrossRef]
- Epi Info™ 3.5.1. Available online: https://www.cdc.gov/epiinfo/support/downloads/prevversions.html (accessed on 7 October 2022).
- Andersson, M.O.; Tolf, C.; Tamba, P.; Stefanache, M.; Waldenström, J.; Dobler, G.; Chițimia-Dobler, L. Canine tick-borne diseases in pet dogs from Romania. Parasit. Vectors 2017, 10, 155. [Google Scholar] [CrossRef] [PubMed]
- Pacifico, L.; Braff, J.; Buono, F.; Beall, M.; Neola, B.; Buch, J.; Sgroi, G.; Piantedosi, D.; Santoro, M.; Tyrrell, P.; et al. Hepatozoon canis in hunting dogs from Southern Italy: Distribution and risk factors. Parasitol. Res. 2020, 119, 3023–3031. [Google Scholar] [CrossRef]
- Hamel, D.; Röhrig, E.; Pfister, K. Canine vector-borne disease in travelled dogs in Germany—A retrospective evaluation of laboratory data from the years 2004–2008. Vet. Parasitol. 2011, 181, 31–36. [Google Scholar] [CrossRef]
- Vojta, L.; Mrljak, V.; Curković, S.; Zivicnjak, T.; Marinculić, A.; Beck, R. Molecular epizootiology of canine hepatozoonosis in Croatia. Int. J. Parasitol. 2009, 39, 1129–1136. [Google Scholar] [CrossRef]
- Cimpan, A.A.; Nachum-Biala, Y.; Ben-Shitrit, B.; Miron, L.; Baneth, G. Epidemiological Study of Canine Babesiosis and Hepatozoonosis in the South of Romania. Acta Parasitol. 2020, 65, 669–678. [Google Scholar] [CrossRef] [PubMed]
- Hornok, S.; Tánczos, B.; Fernández de Mera, I.G.; de la Fuente, J.; Hofmann-Lehmann, R.; Farkas, R. High prevalence of Hepatozoon-infection among shepherd dogs in a region considered to be free of Rhipicephalus sanguineus. Vet. Parasitol. 2013, 196, 189–193. [Google Scholar] [CrossRef]
- Cassini, R.; Zanutto, S.; Frangipane di Regalbono, A.; Gabrielli, S.; Calderini, P.; Moretti, A.; Tampieri, M.P.; Pietrobelli, M. Canine piroplasmosis in Italy: Epidemiological aspects in vertebrate and invertebrate hosts. Vet. Parasitol. 2009, 165, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Ebani, V.V.; Nardoni, S.; Fognani, G.; Mugnaini, L.; Bertelloni, F.; Rocchigiani, G.; Papini, R.A.; Stefani, F.; Mancianti, F. Molecular detection of vector-borne bacteria and protozoa in healthy hunting dogs from Central Italy. Asian Pac. J. Trop. Biomed. 2015, 5, 108–112. [Google Scholar] [CrossRef]
- Baneth, G.; Bourdeau, P.; Bourdoiseau, G.; Bowman, D.; Breitschwerdt, E.; Capelli, G.; Cardoso, L.; Dantas-Torres, F.; Day, M.; Dedet, J.-P. Vector-borne diseases-constant challenge for practicing veterinarians: Recommendations from the CVBD World Forum. Parasit. Vectors 2012, 5, 55. [Google Scholar] [CrossRef]
- Talambuta, N.; Oleg, C.; Dumitru, E.; Ştefan, R.; Galina, M.; Maria, Z.; Elena, G.; Tudor, A. Diversitatea parazitofaunei la Canis familiaris din ecosistemul urban, Chişinău. In Actual Problems of Zoology and Parasitology: Achievements and Prospects, Chișinău, Republic of Moldova, 13 October 2017; Institutul de Zoologie: Chișinău, Republic of Moldova, 2017; pp. 212–219. [Google Scholar]
- Murata, T.; Inoue, M.; Taura, Y.; Nakama, S.; Abe, H.; Fujisaki, K. Detection of Hepatozoon canis oocyst from ticks collected from the infected dogs. J. Vet. Med. Sci. 1995, 57, 111–112. [Google Scholar] [CrossRef] [PubMed]
- de Miranda, R.L.; de Castro, J.R.; Olegário, M.M.; Beletti, M.E.; Mundim, A.V.; O’Dwyer, L.H.; Eyal, O.; Talmi-Frank, D.; Cury, M.C.; Baneth, G. Oocysts of Hepatozoon canis in Rhipicephalus (Boophilus) microplus collected from a naturally infected dog. Vet. Parasitol. 2011, 177, 392–396. [Google Scholar] [CrossRef] [PubMed]
- Dantas-Torres, F. Biology and ecology of the brown dog tick, Rhipicephalus sanguineus. Parasit. Vectors 2010, 3, 26. [Google Scholar] [CrossRef] [PubMed]
- Schäfer, I.; Müller, E.; Nijhof, A.M.; Aupperle-Lellbach, H.; Loesenbeck, G.; Cramer, S.; Naucke, T.J. First evidence of vertical Hepatozoon canis transmission in dogs in Europe. Parasit. Vectors 2022, 15, 296. [Google Scholar] [CrossRef] [PubMed]
- Baneth, G.; Harrus, S.; Gal, A.; Aroch, I. Canine vector-borne co-infections: Ehrlichia canis and Hepatozoon canis in the same host monocytes. Vet. Parasitol. 2015, 208, 30–34. [Google Scholar] [CrossRef] [PubMed]
- Kovačević Filipović, M.M.; Beletić, A.D.; Ilić Božović, A.V.; Milanović, Z.; Tyrrell, P.; Buch, J.; Breitschwerdt, E.B.; Birkenheuer, A.J.; Chandrashekar, R. Molecular and Serological Prevalence of Anaplasma phagocytophilum, A. platys, Ehrlichia canis, E. chaffeenses, E. ewingii, Borrelia burgdorferi, Babesia canis, B. gibsoni and B. vogeli among Clinically Healthy Outdoor Dogs in Serbia. Vet. Parasitol. Reg. Stud. Rep. 2018, 14, 117–122. [Google Scholar] [CrossRef] [PubMed]
- Welc-Faleciak, R.; Rodo, A.; Siński, E.; Bajer, A. Babesia canis and other tick-borne infections in dogs in Central Poland. Vet. Parasitol. 2009, 166, 191–198. [Google Scholar] [CrossRef] [PubMed]
- Imre, M.; Farkas, R.; Ilie, M.S.; Imre, K.; Dărăbuş, G. Survey of babesiosis in symptomatic dogs from Romania: Occurrence of Babesia gibsoni associated with breed. Ticks Tick Borne Dis. 2013, 4, 500–502. [Google Scholar] [CrossRef]
- Solano-Gallego, L.; Sainz, Á.; Roura, X.; Estrada-Peña, A.; Miró, G. A review of canine babesiosis: The European perspective. Parasit. Vectors 2016, 9, 336. [Google Scholar] [CrossRef] [PubMed]
- Paulauskas, A.; Radzijevskaja, J.; Karvelienė, B.; Grigonis, A.; Aleksandravičienė, A.; Zamokas, G.; Babickaitė, L.; Sabūnas, V.; Petkevičius, S. Detection and molecular characterization of canine babesiosis causative agent Babesia canis in the naturally infected dog in Lithuania. Vet. Parasitol. 2014, 205, 702–706. [Google Scholar] [CrossRef] [PubMed]
- Leica, L.; Mitrea, I.L.; Ionita, M. Clinical Occurrence of Canine Babesiosis in the Coastal Area of the Black Sea (Dobrogea) in Southeastern Romania and Associated Epidemiological Implications. J. Parasitol. 2019, 105, 491–496. [Google Scholar] [CrossRef] [PubMed]
- Máthé, A.; Vörös, K.; Papp, L.; Reiczigel, J. Clinical manifestations of canine babesiosis in Hungary (63 cases). Acta Vet. Hung. 2006, 54, 367–385. [Google Scholar] [CrossRef]
- Mircean, V.; Dumitrache, M.O.; Györke, A.; Pantchev, N.; Jodies, R.; Mihalca, A.D.; Cozma, V. Seroprevalence and geographic distribution of Dirofilaria immitis and tick-borne infections (Anaplasma phagocytophilum, Borrelia burgdorferi sensu lato, and Ehrlichia canis) in dogs from Romania. Vector Borne Zoonotic Dis. 2012, 12, 595–604. [Google Scholar] [CrossRef]
- Enache, D.; Imre, M.; Ilie, M.S.; Coprean, D. Seroprevalence of Anaplasma phagocytophilum in dogs from Constanţa county. J. Biotechnol. 2015, 208, S95. [Google Scholar] [CrossRef]
- Majláthová, V.; Majláth, I.; Víchová, B.; Gul’ová, I.; Derdáková, M.; Sesztáková, E.; Pet’ko, B. Polymerase chain reaction confirmation of Babesia canis canis and Anaplasma phagocytophilum in dogs suspected of babesiosis in Slovakia. Vector Borne Zoonotic Dis. 2011, 11, 1447–1451. [Google Scholar] [CrossRef]
- Ebani, V.V.; Bertelloni, F.; Turchi, B.; Cerri, D. Serological and molecular survey of Anaplasma phagocytophilum in Italian hunting dogs. Ann. Agric. Environ. Med. 2013, 20, 289–292. [Google Scholar] [PubMed]
- de la Fuente, J.; Torina, A.; Naranjo, V.; Nicosia, S.; Alongi, A.; La Mantia, F.; Kocan, K.M. Molecular characterization of Anaplasma platys strains from dogs in Sicily, Italy. BMC Vet. Res. 2006, 2, 24. [Google Scholar] [CrossRef] [PubMed]
- Alberti, A.; Addis, M.F.; Sparagano, O.; Zobba, R.; Chessa, B.; Cubeddu, T.; Parpaglia, M.L.; Ardu, M.; Pittau, M. Anaplasma phagocytophilum, Sardinia, Italy. Emerg. Infect. Dis. 2005, 11, 1322–1324. [Google Scholar] [CrossRef]
- Hornok, S.; Dénes, B.; Meli, M.L.; Tánczos, B.; Fekete, L.; Gyuranecz, M.; de la Fuente, J.; de Mera, I.G.; Farkas, R.; Hofmann-Lehmann, R. Non-pet dogs as sentinels and potential synanthropic reservoirs of tick-borne and zoonotic bacteria. Vet. Microbiol. 2013, 167, 700–703. [Google Scholar] [CrossRef] [PubMed]
- Skotarczak, B.; Adamska, M.; Rymaszewska, A.; Suproń, M.; Sawczuk, M.; Maciejewska, A. Anaplasma phagocytophila and protozoans of Babesia genus in dogs from endemic areas of Lyme disease in north-western Poland. Wiad. Parazytol. 2004, 50, 555–561. [Google Scholar] [PubMed]
- Rymaszewska, A.; Adamska, M. Molecular evidence of vector-borne pathogens coinfecting dogs from Poland. Acta Vet. Hung. 2011, 59, 215–223. [Google Scholar] [CrossRef]
- Hamel, D.; Shukullari, E.; Rapti, D.; Silaghi, C.; Pfister, K.; Rehbein, S. Parasites and vector-borne pathogens in client-owned dogs in Albania. Blood pathogens and seroprevalences of parasitic and other infectious agents. Parasitol. Res. 2016, 115, 489–499. [Google Scholar] [CrossRef]
- Kybicová, K.; Schánilec, P.; Hulínská, D.; Uherková, L.; Kurzová, Z.; Spejchalová, S. Detection of Anaplasma phagocytophilum and Borrelia burgdorferi sensu lato in dogs in the Czech Republic. Vector Borne Zoonotic Dis. 2009, 9, 655–661. [Google Scholar] [CrossRef]
- Movila, A.; Toderas, I.; Uspenskaia, I.; Conovalov, J. Molecular detection of tick-borne pathogens in Ixodes ricinus from Moldova collected in 1960. Ticks Tick Borne Dis. 2013, 4, 359–361. [Google Scholar] [CrossRef] [PubMed]
- Morozov, A.C.; Tischenkov, A.A.; Proka, A.A.; Silaghi, C.; Toderas, I.K.; Movila, A.A.; Poppert, S. Prevalence of tick-borne pathogens in ticks from migratory birds in Republic of Moldova. Parasit. Vectors 2014, 7, P4. [Google Scholar] [CrossRef]
- Dumler, J.S.; Barbet, A.F.; Bekker, C.P.; Dasch, G.A.; Palmer, G.H.; Ray, S.C.; Rikihisa, Y.; Rurangirwa, F.R. Reorganization of genera in the families Rickettsiaceae and Anaplasmataceae in the order Rickettsiales: Unification of some species of Ehrlichia with Anaplasma, Cowdria with Ehrlichia and Ehrlichia with Neorickettsia, descriptions of six new species combinations and designation of Ehrlichia equi and ‘HGE agent’ as subjective synonyms of Ehrlichia phagocytophila. Int. J. Syst. Evol. Microbiol. 2001, 51, 2145–2165. [Google Scholar] [CrossRef] [PubMed]
- Roura, X.; Santamarina, G.; Tabar, M.D.; Francino, O.; Altet, L. Polymerase chain reaction detection of Bartonella spp. in dogs from Spain with blood culture-negative infectious endocarditis. J. Vet. Cardiol. 2018, 20, 267–275. [Google Scholar] [CrossRef] [PubMed]
- Diniz, P.P.; Billeter, S.A.; Otranto, D.; De Caprariis, D.; Petanides, T.; Mylonakis, M.E.; Koutinas, A.F.; Breitschwerdt, E.B. Molecular documentation of Bartonella infection in dogs in Greece and Italy. J. Clin. Microbiol. 2009, 47, 1565–1567. [Google Scholar] [CrossRef]
- Torrejón, E.; Sanches, G.S.; Moerbeck, L.; Santos, L.; André, M.R.; Domingos, A.; Antunes, S. Molecular survey of Bartonella species in stray cats and dogs, humans, and questing ticks from Portugal. Pathogens 2022, 11, 749. [Google Scholar] [CrossRef]
- CDC 2020. Available online: https://www.cdc.gov/bartonella/veterinarians/ (accessed on 2 October 2022).
- Álvarez-Fernández, A.; Breitschwerdt, E.B.; Solano-Gallego, L. Bartonella infections in cats and dogs including zoonotic aspects. Parasit. Vectors 2018, 11, 624. [Google Scholar] [CrossRef] [PubMed]
- Sainz, Á.; Roura, X.; Miró, G.; Estrada-Peña, A.; Kohn, B.; Harrus, S.; Solano-Gallego, L. Guideline for veterinary practitioners on canine ehrlichiosis and anaplasmosis in Europe. Parasit. Vectors 2015, 8, 75. [Google Scholar] [CrossRef] [PubMed]
- Springer, A.; Glass, A.; Topp, A.K.; Strube, C. Zoonotic Tick-Borne Pathogens in Temperate and Cold Regions of Europe-A Review on the Prevalence in Domestic Animals. Front. Vet. Sci. 2020, 7, 604910. [Google Scholar] [CrossRef] [PubMed]
- Sanches, G.S.; Couto, J.; Silva-Pedrosa, R.; Ferrolho, J.; Santos, A.S.; Santos-Silva, M.M.; Antunes, S.; Domingos, A. Molecular heterogeneity of Rhipicephalus sanguineus sensu lato and screening for Ehrlichia canis in mainland Portugal. Ticks Tick Borne Dis. 2018, 9, 1383–1390. [Google Scholar] [CrossRef]
- Atif, F.A.; Mehnaz, S.; Qamar, M.F.; Roheen, T.; Sajid, M.S.; Ehtisham-Ul-Haque, S.; Kashif, M.; Ben Said, M. Epidemiology, Diagnosis, and Control of Canine Infectious Cyclic Thrombocytopenia and Granulocytic Anaplasmosis: Emerging Diseases of Veterinary and Public Health Significance. Vet. Sci. 2021, 8, 312. [Google Scholar] [CrossRef]
| Pathogen | Target Gene | Product Size (bp) | Forward Primer | Reverse Primer | References |
|---|---|---|---|---|---|
| Piroplasmids and Hepatozoon spp. (Nested PCR) | 18S rRNA | 561–620 | BTH-1 F: CCT GAG AAA CGG CTA CCA CAT CT | BTH-1R: TTG CGA CCA TAC TCC CCC CA | [18] |
| GF2: GTC TTG TAA TTG GAA TGA TGG | GR2: CCA AAG ACT TTG ATT TCT CTC | ||||
| Borrelia spp. (Nested PCR) | flaB | 350 | FlaLL: ACA TAT TCA GAT GCA GAC AGA GGT | FlaRL: TGT TAG ACG TTA CCG ATA CTA ACG | [19,20] |
| FlaLS: AAC AGC TGA AGA GCT TGG AAT G | FlaRS: CGA TAA TCT TAC TAT TCA CTA GTT TC | ||||
| SFG Rickettsia | gltA | 381 | Rsfg877: GGG GGC CTG CTC ACG GCG G | Rsfg1258: ATT GCA AAA AGT ACA GTG AAC A | [21] |
| Bartonella spp. | gltA | 380–400 | bart781: GGG GAC CAG CTC ATG GTG G | bart1137: AAT GCA AAA AGA ACA GTA AAC A | [22] |
| Francisella tularensis | 17-kDa lipoprotein gene | 400 | TUL4-435: GCT GTA TCA TCA TTT AAT AAA CTG CTG | TUL4-863: TTG GGA AGC TTG TAT CAT GGC ACT | [23] |
| Anaplasma phagocytophilum (Heminested PCR) | groEL | 570 | EphplgroEL(569)F: ATG GTA TGC AGT TTG ATC GC | EphplgroEL(1193)R: TCT ACT CTG TCT TTG CGT TC | [24] |
| EphgroEL(1142)R: TTG AGT ACA GCA ACA CCA CCG GAA | |||||
| Anaplasma platys | 16S rRNA | 349 | EPLAT5: TTT GTC GTA GCT TGC TAT GAT | EPLAT3: CTT CTG TGG GTA CCG TC | [25] |
| Ehrlichia canis (Nested PCR) | 16S rRNA | 389 | ECC: AGA ACG AAC GCT GGC GGC AAG CC | ECB: CGT ATT ACC GCG GCT GCT GGC A | [26] |
| «canis»: CAA TTA TTT ATA GCC TCT GGC TAT AGG A | HE3: TAT AGG TAC CGT CAT TAT CTT CCC TAT |
| Category | Sampled Dogs (N = 120) n (%) |
|---|---|
| Age (years) | |
| 0 to 1 | 11 (9.2) |
| 1 to 8 | 99 (82.5) |
| >8 | 10 (8.3) |
| Sex | |
| Female | 70 (58.3) |
| Male | 50 (41.7) |
| Breed | |
| Pure breed | 11 (9.2) |
| Mixed | 109 (90.8) |
| Location | |
| Chișinău | 78 (65.0) |
| Cahul | 42 (35.0) |
| Origin | |
| Shelter | 90 (75) |
| Clinic | 30 (25) |
| Category | Hepatozoon canis | Babesia canis | Anaplasma phagocytophilum | Bartonella spp. |
|---|---|---|---|---|
| Age, n (%) | ||||
| 0 to 1, N = 11 | 2 (18.2) | 2 (18.2) | 2 (18.2) | 0 (0.0) |
| 1 to 8, N = 99 | 17 (17.2) | 11 (11.1) | 4 (4.0) | 1 (1.0) |
| >8, N = 10 | 0 (0.0) | 1 (10.0) | 1 (10.0) | 0 (0.0) |
| p-value | 0.26 | 0.5 | 0.8 | - |
| OR (95% CI) | 0.5 (0.1–1.7) | 0.6 (0.1–2.7) | 0.8 (0.1–5.1) | - |
| Sex; n (%) | ||||
| Female, N = 70 | 11 (15.7) | 6 (8.6) | 4 (5.7) | 0 (0.0) |
| Male, N = 50 | 8 (16.0) | 8 (16.0) | 3 (6.0) | 1 (2.0) |
| p-value | 0.7 | 0.2 | 0.6 | - |
| OR (95% CI) | 0.8 (0.3–2.4) | 2.1 (0.6–6.7) | 1.6 (0.2–10.3) | - |
| Breed, n (%) | ||||
| Pure breed, N = 11 | 0 (0.0) | 0 (0.0) | 3 (27.3) | 0 (0.0) |
| Mixed, N = 109 | 19 (17.4) | 14 (12.8) | 4 (3.7) | 1 (0.9) |
| p-value | 1.0 | 1.0 | 0.1 | - |
| OR (95% CI) | 0.0 (0.0–>1.0 × 10 12) | 0.0 (0.0–>1.0 × 1012) | 6.4 (0.5–74.8) | - |
| Location, n (%) | ||||
| Chișinău, N = 78 | 15 (19.2) | 9 (11.5) | 4 (5.1) | 0 (0.0) |
| Cahul, N = 42 | 4 (9.5) | 5 (11.9) | 3 (7.1) | 1 (2.4) |
| p-value | 0.03 | 0.8 | 0.06 | - |
| OR (95% CI) | 3.9 (1.1–13.4) | 1.2 (0.4–4.2) | 0.04 (0.002–1.2) | - |
| Origin, n (%) | ||||
| Shelter, N = 90 | 18 (20.0) | 13 (14.4) | 2 (2.2) | 1 (1.1) |
| Vet Clinic, N = 30 | 1 (3.3) | 1 (3.3) | 5 (16.7) | 0 (0.0) |
| p-value | 0.1 | 0.3 | 0.03 | - |
| OR (95% CI) | 0.2 (0.02–1.4) | 0.3 (0.04–3.0) | 27.0 (1.4–521.9) | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
D’Amico, G.; Ionică, A.M.; Györke, A.; Dumitrache, M.O. Epidemiological Survey of the Main Tick-Borne Pathogens Infecting Dogs from the Republic of Moldova. Pathogens 2022, 11, 1267. https://doi.org/10.3390/pathogens11111267
D’Amico G, Ionică AM, Györke A, Dumitrache MO. Epidemiological Survey of the Main Tick-Borne Pathogens Infecting Dogs from the Republic of Moldova. Pathogens. 2022; 11(11):1267. https://doi.org/10.3390/pathogens11111267
Chicago/Turabian StyleD’Amico, Gianluca, Angela Monica Ionică, Adriana Györke, and Mirabela Oana Dumitrache. 2022. "Epidemiological Survey of the Main Tick-Borne Pathogens Infecting Dogs from the Republic of Moldova" Pathogens 11, no. 11: 1267. https://doi.org/10.3390/pathogens11111267
APA StyleD’Amico, G., Ionică, A. M., Györke, A., & Dumitrache, M. O. (2022). Epidemiological Survey of the Main Tick-Borne Pathogens Infecting Dogs from the Republic of Moldova. Pathogens, 11(11), 1267. https://doi.org/10.3390/pathogens11111267








