Investigation of Long COVID Prevalence and Its Relationship to Epstein-Barr Virus Reactivation
Abstract
1. Introduction
2. Results
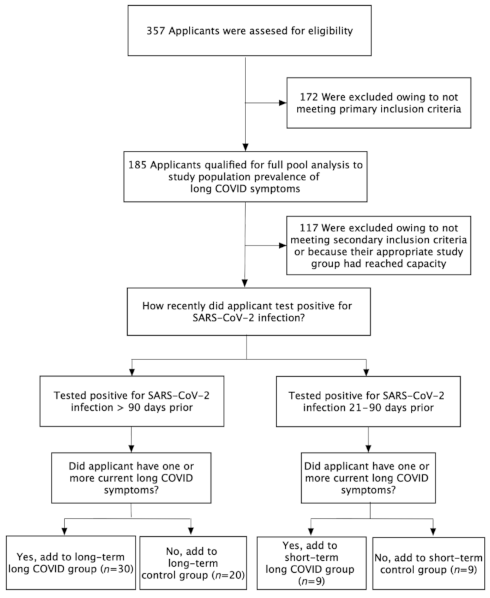
2.1. Long COVID Prevalence
2.2. Analysis of Primary (Long-Term) Study Participants
2.3. Analysis of Secondary (Short-Term) Study Participants
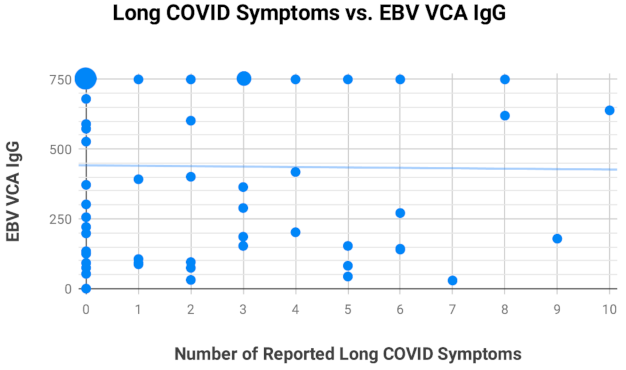
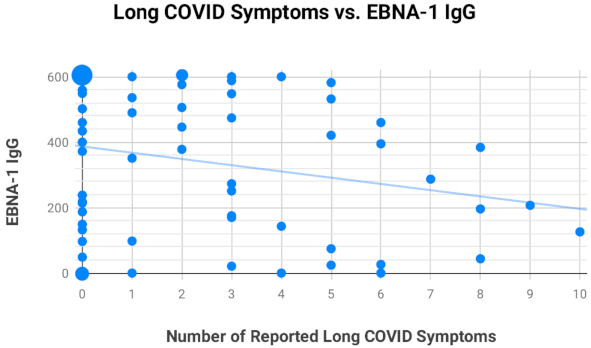
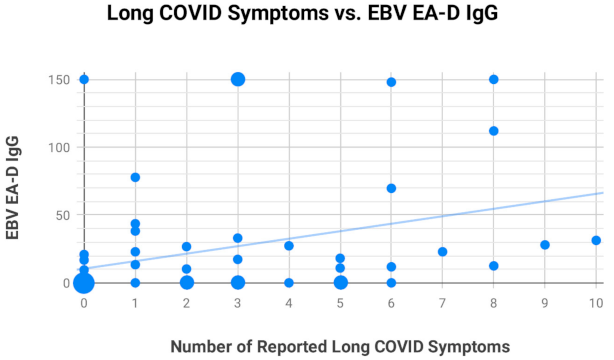
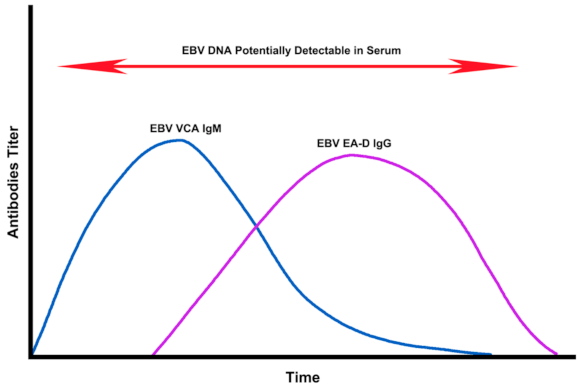
2.4. Assessment of EBV EA-D IgG, EBV VCA IgG, and EBNA-1 IgG Titers in All Subjects
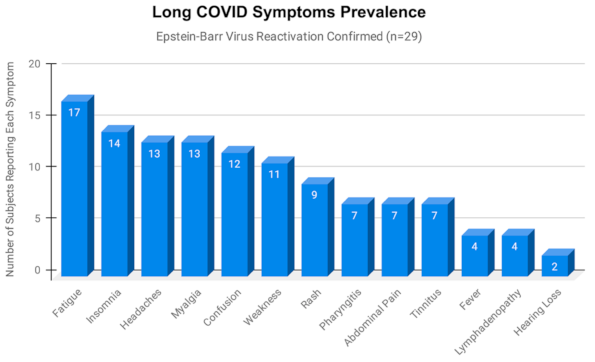
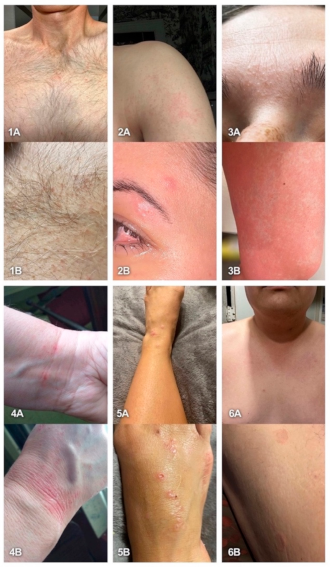
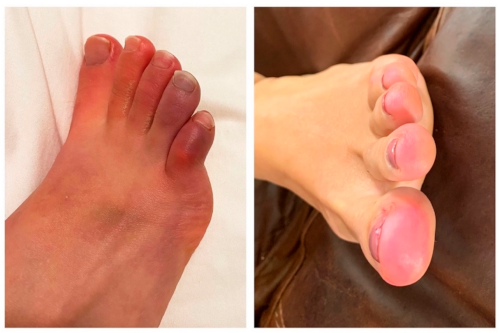
2.5. Most Frequently Reported Symptoms
3. Discussion
4. Materials and Methods
4.1. Study Design
4.2. Patient Recruitment
4.3. Primary and Secondary Study Groups
4.4. Assessments
4.5. Review of Photographs
4.6. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A. Epstein–Barr Virus Antibody Panel Results
| ID | EBV EA-D IgG | EBV VCA IgM | EBV VCA IgG | EBNA-1 IgG |
|---|---|---|---|---|
| 06966 | - | 81.4 | >750 | >600 |
| 13444 | 148 | - | 143 | 460 |
| 13459 | - | - | 202 | 143 |
| 15170 | - | - | >750 | 532 |
| 15366 | 10.2 | - | 401 | 576 |
| 15994 | - | - | >750 | 548 |
| 16370 | 69.7 | - | >750 | - |
| 16381 | - | - | 153 | 74.4 |
| 16968 | 22.9 | - | >750 | >600 |
| 16979 | >150 | - | >750 | 273 |
| 17050 | - | - | >750 | 351 |
| 17113 | 77.8 | 69.1 | 106 | 490 |
| 17332 | 13.4 | - | 87.2 | 97.8 |
| 17706 | - | - | 31.1 | 378 |
| 35143 | 18.1 | - | 81.9 | 582 |
| 35145 | >150 | - | 620 | 196 |
| 35188 | 26.7 | - | 602 | 446 |
| 35946 | 112 | - | >750 | 384 |
| 83584 | - | 67.1 | 364 | 588 |
| 83821 | >150 | - | 153 | 21 |
| 83860 | - | - | >750 | >600 |
| 83893 | 28 | - | 179 | 207 |
| 83925 | - | - | 95 | 506 |
| 83929 | 10.9 | - | 43.4 | 24.1 |
| 83938 | 31.3 | - | 639 | 126 |
| 87629 | - | - | >750 | 251 |
| 87634 | - | - | 74.4 | >600 |
| 87987 | 17.3 | - | 289 | 175 |
| 87999 | 33 | - | >750 | 474 |
| 94332 | 22.9 | - | 29.1 | 287 |
| ID | EBV EA-D IgG | EBV VCA IgM | EBV VCA IgG | EBNA-1 IgG |
|---|---|---|---|---|
| 13481 | - | - | 198 | 372 |
| 16169 | - | - | 134 | 149 |
| 16199 | 16.8 | - | >750 | 502 |
| 16710 | - | - | 680 | >600 |
| 16715 | - | - | 573 | 187 |
| 16721 | - | - | 125 | >600 |
| 16755 | - | - | - | - |
| 16832 | - | - | 198 | 48.7 |
| 17783 | - | - | 91.9 | >600 |
| 17804 | - | - | 302 | >600 |
| 83740 | - | - | >750 | 460 |
| 83859 | - | - | 53 | 559 |
| 83911 | - | - | >750 | 400 |
| 83924 | - | - | 74.6 | 132 |
| 83966 | - | - | >750 | 589 |
| 84549 | - | - | 129 | 434 |
| 85068 | 9.51 | - | >750 | >600 |
| 87612 | - | - | >750 | 96.6 |
| 87994 | >150 | - | >750 | >600 |
| 93070 | - | - | >750 | 238 |
| ID | EBV EA-D IgG | EBV VCA IgM | EBV VCA IgG | EBNA-1 IgG |
|---|---|---|---|---|
| 13963 | - | - | >750 | 421 |
| 16378 | 43.6 | - | 92.3 | 536 |
| 16555 | - | - | 271 | 26.2 |
| 16717 | 38.2 | - | 392 | - |
| 16750 | 27.3 | - | 418 | - |
| 17106 | - | - | >750 | >600 |
| 17323 | >150 | - | 186 | 170 |
| 17387 | 12.5 | 47.5 | >750 | 43.7 |
| 17390 | 11.8 | - | 140 | 395 |
| ID | EBV EA-D IgG | EBV VCA IgM | EBV VCA IgG | EBNA-1 IgG |
|---|---|---|---|---|
| 07506 | - | - | 221 | 214 |
| 09092 | - | - | >750 | 549 |
| 15509 | 20.9 | - | 372 | >600 |
| 16406 | - | - | >750 | - |
| 16642 | - | - | 590 | 218 |
| 16770 | - | - | 256 | >600 |
| 92742 | - | - | >750 | - |
| 93074 | - | - | 527 | >600 |
| 94266 | - | - | >750 | - |
References
- Logue, J.K.; Franko, N.M.; McCulloch, D.J.; McDonald, D.; Magedson, A.; Wolf, C.R.; Chu, H.Y. Sequelae in Adults at 6 Months After COVID-19 Infection. JAMA Netw. Open 2021, 4, e210830. [Google Scholar] [CrossRef] [PubMed]
- Greenhalgh, T.; Knight, M.; A’Court, C.; Buxton, M.; Husain, L. Management of post-acute covid-19 in primary care. BMJ 2020, 370, m3026. [Google Scholar] [CrossRef] [PubMed]
- Al-Jahdhami, I.; Al-Naamani, K.; Al-Mawali, A. The Post-acute COVID-19 Syndrome (Long COVID). Oman Med. J. 2021, 36, e220. [Google Scholar] [CrossRef] [PubMed]
- Garrigues, E.; Janvier, P.; Kherabi, Y.; Le Bot, A.; Hamon, A.; Gouze, H.; Doucet, L.; Berkani, S.; Oliosi, E.; Mallart, E.; et al. Post-discharge persistent symptoms and health-related quality of life after hospitalization for COVID-19. J. Infect. 2020, 81, e4–e6. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Huang, L.; Wang, Y.; Li, X.; Ren, L.; Gu, X.; Kang, L.; Guo, L.; Liu, M.; Zhou, X.; et al. 6-month consequences of COVID-19 in patients discharged from hospital: A cohort study. Lancet 2021, 397, 220–232. [Google Scholar] [CrossRef]
- Callard, F.; Perego, E. How and why patients made Long Covid. Soc. Sci. Med. 2021, 268, 113426. [Google Scholar] [CrossRef]
- Neves, M.; Marinho-Dias, J.; Ribeiro, J.; Sousa, H. Epstein-Barr virus strains and variations: Geographic or disease-specific variants? J. Med. Virol. 2017, 89, 373–387. [Google Scholar] [CrossRef]
- Kanda, T.; Yajima, M.; Ikuta, K. Epstein-Barr virus strain variation and cancer. Cancer Sci. 2019, 110, 1132–1139. [Google Scholar] [CrossRef]
- Misko, I.S.; Cross, S.M.; Khanna, R.; Elliott, S.L.; Schmidt, C.; Pye, S.J.; Silins, S.L. Crossreactive recognition of viral, self, and bacterial peptide ligands by human class I-restricted cytotoxic T lymphocyte clonotypes: Implications for molecular mimicry in autoimmune disease. Proc. Natl. Acad. Sci. USA 1999, 96, 2279–2284. [Google Scholar] [CrossRef]
- Houen, G.; Trier, N.H. Epstein-Barr Virus and Systemic Autoimmune Diseases. Front. Immunol. 2020, 11, 587380. [Google Scholar] [CrossRef]
- Straus, S.E.; Tosato, G.; Armstrong, G.; Lawley, T.; Preble, O.T.; Henle, W.; Davey, R.; Pearson, G.; Epstein, J.; Brus, I.; et al. Persisting illness and fatigue in adults with evidence of Epstein-Barr virus infection. Ann. Intern. Med. 1985, 102, 7–16. [Google Scholar] [CrossRef]
- Stowe, R.P.; Pierson, D.L.; Feeback, D.L.; Barrett, A.D. Stress-induced reactivation of Epstein-Barr virus in astronauts. Neuroimmunomodulation 2000, 8, 51–58. [Google Scholar] [CrossRef]
- Schaade, L.; Kleines, M.; Hausler, M. Application of virus-specific immunoglobulin M (IgM), IgG, and IgA antibody detection with a polyantigenic enzyme-linked immunosorbent assay for diagnosis of Epstein-Barr virus infections in childhood. J. Clin. Microbiol. 2001, 39, 3902–3905. [Google Scholar] [CrossRef][Green Version]
- Smatti, M.K.; Al-Sadeq, D.W.; Ali, N.H.; Pintus, G.; Abou-Saleh, H.; Nasrallah, G.K. Epstein-Barr Virus Epidemiology, Serology, and Genetic Variability of LMP-1 Oncogene Among Healthy Population: An Update. Front. Oncol. 2018, 8, 211. [Google Scholar] [CrossRef]
- Baeck, M.; Herman, A. COVID toes: Where do we stand with the current evidence? Int. J. Infect. Dis. 2021, 102, 53–55. [Google Scholar] [CrossRef]
- Kasl, S.V.; Evans, A.S.; Niederman, J.C. Psychosocial risk factors in the developmental of infectious mononucleosis. Psychosom. Med. 1979, 41, 445–466. [Google Scholar] [CrossRef]
- Glaser, R.; Rice, J.; Sheridan, J.; Fertel, R.; Stout, J.; Speicher, C.; Pinsky, D.; Kotur, M.; Post, A.; Beck, M.; et al. Stress-related immune suppression: Health implications. Brain Behav. Immun. 1987, 1, 7–20. [Google Scholar] [CrossRef]
- Glaser, R.; Kiecolt-Glaser, J. Stress-associated immune modulation and its implications for reactivation of latent herpesviruses. Hum. Herpesvirus Infect. 1994, 13, 245. [Google Scholar]
- Chen, T.; Song, J.; Liu, H.; Zheng, H.; Chen, C. Positive Epstein-Barr virus detection in coronavirus disease 2019 (COVID-19) patients. Sci. Rep. 2021, 11, 10902. [Google Scholar] [CrossRef]
- Paolucci, S.; Cassaniti, I.; Novazzi, F.; Fiorina, L.; Piralla, A.; Comolli, G.; Bruno, R.; Maserati, R.; Gulminetti, R.; Novati, S.; et al. EBV DNA increase in COVID-19 patients with impaired lymphocyte subpopulation count. Int. J. Infect. Dis. 2020, 104, 315–319. [Google Scholar] [CrossRef]
- Simonnet, A.; Engelmann, I.; Moreau, A.S.; Garcia, B.; Six, S.; Kalioubie, A.E.; Robriquet, L.; Hober, D.; Jourdain, M. High incidence of Epstein-Barr virus, cytomegalovirus, and human-herpes virus-6 reactivations in critically-ill patients with Covid-19. Infect. Dis. Now 2021, 51, 296–299. [Google Scholar] [CrossRef] [PubMed]
- Lehner, G.F.; Klein, S.J.; Zoller, H.; Peer, A.; Bellmann, R.; Joannidis, M. Correlation of interleukin-6 with Epstein-Barr virus levels in COVID-19. Crit. Care 2020, 24, 657. [Google Scholar] [CrossRef] [PubMed]
- Singh, V.; Upadhyay, P.; Reddy, J.; Granger, J. SARS-CoV-2 Respiratory Co-Infections: Incidence of Viral and Bacterial Co-Pathogens. Int. J. Infect. Dis. 2021, 105, 617–620. [Google Scholar] [CrossRef]
- Dowd, J.B.; Palermo, T.; Brite, J.; McDade, T.W.; Aiello, A. Seroprevalence of Epstein-Barr virus infection in U.S. children ages 6–19, 2003–2010. PLoS ONE 2013, 8, e64921. [Google Scholar] [CrossRef] [PubMed]
- Bauer, G. Simplicity through complexity: Immunoblot with recombinant antigens as the new gold standard in Epstein-Barr virus serology. Clin. Lab. 2001, 47, 223–230. [Google Scholar]
- Lam, W.K.J.; Jiang, P.; Chan, K.C.A.; Cheng, S.H.; Zhang, H.; Peng, W.; Tse, O.Y.O.; Tong, Y.K.; Gai, W.; Zee, B.C.Y.; et al. Sequencing-based counting and size profiling of plasma Epstein-Barr virus DNA enhance population screening of nasopharyngeal carcinoma. Proc. Natl. Acad. Sci. USA 2018, 115, E5115–E5124. [Google Scholar] [CrossRef] [PubMed]
- Chan, K.C.A.; Woo, J.K.S.; King, A.; Zee, B.C.Y.; Lam, W.K.J.; Chan, S.L.; Chu, S.W.I.; Mak, C.; Tse, I.O.L.; Leung, S.Y.M.; et al. Analysis of Plasma Epstein-Barr Virus DNA to Screen for Nasopharyngeal Cancer. N. Engl. J. Med. 2017, 377, 513–522. [Google Scholar] [CrossRef]
- Odumade, O.A.; Hogquist, K.A.; Balfour, H.H., Jr. Progress and problems in understanding and managing primary Epstein-Barr virus infections. Clin. Microbiol. Rev. 2011, 24, 193–209. [Google Scholar] [CrossRef]
- Drago, F.; Herzum, A.; Ciccarese, G.; Parodi, A. May syphilis protect against human papillomavirus infection? An example of heterologous immunity. G. Ital. Dermatol. Venereol. 2019, 154, 719–721. [Google Scholar] [CrossRef]
- Spencer, S.A.; Fenske, N.A.; Espinoza, C.G.; Hamill, J.R.; Cohen, L.E.; Espinoza, L.R. Granuloma annulare-like eruption due to chronic Epstein-Barr virus infection. Arch. Dermatol. 1988, 124, 250–255. [Google Scholar] [CrossRef]
- Goodlad, J. Epstein-Barr Virus Associated Lymphoproliferative Disorders in the Skin. Available online: https://bit.ly/goodladj1 (accessed on 15 June 2021).
- Fiorini, G.F.; Sinico, R.A.; Winearls, C.; Custode, P.; De Giuli-Morghen, C.; D’Amico, G. Persistent Epstein-Barr virus infection in patients with type II essential mixed cryoglobulinemia. Clin. Immunol. Immunopathol. 1988, 47, 262–269. [Google Scholar] [CrossRef]
- Ichinose, K.; Origuchi, T.; Tashiro, N.; Kawashiri, S.Y.; Iwamoto, N.; Fujikawa, K.; Aramaki, T.; Arima, K.; Tamai, M.; Yamasaki, S.; et al. An elderly patient with chronic active Epstein-Barr virus infection with mixed cryoglobulinemia and review of the literature. Mod. Rheumatol. 2013, 23, 1022–1028. [Google Scholar] [CrossRef]
- Rabinowitz, H.K. Infectious mononucleosis presenting as Raynaud’s phenomenon. J. Fam. Pract. 1989, 29, 311–315. [Google Scholar]
- Blereau, R.P. Idiopathic Raynaud Disease. Consultant360 2013, 194. Available online: https://www.consultant360.com/article/idiopathic-raynaud-disease (accessed on 15 June 2021).
- Nirenberg, M.S.; Herrera, M. Foot manifestations in a patient with COVID-19 and Epstein-Barr virus: A case study. Foot 2020, 46, 101707. [Google Scholar] [CrossRef]
- Arslan, F.; Karagz, E.; Bekz, H.S.; Ceylan, B.; Mert, A. Epstein-Barr virus-associated haemophagocytic lymphohistiocytosis presenting with acute sensorineural hearing loss: A case report and review of the literature. Infez. Med. 2017, 25, 277–280. [Google Scholar]
- Williams, L.L.; Lowery, H.W.; Glaser, R. Sudden hearing loss following infectious mononucleosis: Possible effect of altered immunoregulation. Pediatrics 1985, 75, 1020–1027. [Google Scholar]
- Sun, R.; Liu, H.; Wang, X. Mediastinal Emphysema, Giant Bulla, and Pneumothorax Developed during the Course of COVID-19 Pneumonia. Korean J. Radiol. 2020, 21, 541–544. [Google Scholar] [CrossRef]
- Fidan, V. New type of corona virus induced acute otitis media in adult. Am. J. Otolaryngol. 2020, 41, 102487. [Google Scholar] [CrossRef]
- Lechien, J.R.; Chiesa-Estomba, C.M.; Place, S.; Van Laethem, Y.; Cabaraux, P.; Mat, Q.; Huet, K.; Plzak, J.; Horoi, M.; Hans, S.; et al. Clinical and epidemiological characteristics of 1420 European patients with mild-to-moderate coronavirus disease 2019. J. Intern. Med. 2020, 288, 335–344. [Google Scholar] [CrossRef]
- Yang, Y.; Gao, F. Clinical characteristics of primary and reactivated Epstein-Barr virus infection in children. J. Med. Virol. 2020, 92, 3709–3716. [Google Scholar] [CrossRef] [PubMed]
- Chimenti, C.; Verardo, R.; Grande, C.; Francone, M.; Frustaci, A. Infarct-like myocarditis with coronary vasculitis and aneurysm formation caused by Epstein-Barr virus infection. ESC Heart Fail. 2020, 7, 938–941. [Google Scholar] [CrossRef] [PubMed]
- Chimenti, C.; Russo, A.; Pieroni, M.; Calabrese, F.; Verardo, R.; Thiene, G.; Russo, M.A.; Maseri, A.; Frustaci, A. Intramyocyte detection of Epstein-Barr virus genome by laser capture microdissection in patients with inflammatory cardiomyopathy. Circulation 2004, 110, 3534–3539. [Google Scholar] [CrossRef] [PubMed]
- Binkley, P.F.; Cooke, G.E.; Lesinski, A.; Taylor, M.; Chen, M.; Laskowski, B.; Waldman, W.J.; Ariza, M.E.; Williams, M.V., Jr.; Knight, D.A.; et al. Evidence for the role of Epstein Barr Virus infections in the pathogenesis of acute coronary events. PLoS ONE 2013, 8, e54008. [Google Scholar] [CrossRef]
- Jaumally, B.A.; Salem, A.; Robinett, K. Epstein-Barr Virus-Related Multisystem Failure in an Immunocompetent Young Man. Crit. Care Med. 2018, 46, 323. [Google Scholar] [CrossRef]
- Young, L.S.; Yap, L.F.; Murray, P.G. Epstein-Barr virus: More than 50 years old and still providing surprises. Nat. Rev. Cancer 2016, 16, 789–802. [Google Scholar] [CrossRef]
- Hoshino, Y.; Katano, H.; Zou, P.; Hohman, P.; Marques, A.; Tyring, S.K.; Follmann, D.; Cohen, J.I. Long-term administration of valacyclovir reduces the number of Epstein-Barr virus (EBV)-infected B cells but not the number of EBV DNA copies per B cell in healthy volunteers. J. Virol. 2009, 83, 11857–11861. [Google Scholar] [CrossRef]
- Verma, D.; Thompson, J.; Swaminathan, S. Spironolactone blocks Epstein-Barr virus production by inhibiting EBV SM protein function. Proc. Natl. Acad. Sci. USA 2016, 113, 3609–3614. [Google Scholar] [CrossRef]
- Kotfis, K.; Lechowicz, K.; Drozdzal, S.; Niedzwiedzka-Rystwej, P.; Wojdacz, T.K.; Grywalska, E.; Biernawska, J.; Wisniewska, M.; Parczewski, M. COVID-19-The Potential Beneficial Therapeutic Effects of Spironolactone during SARS-CoV-2 Infection. Pharmaceuticals 2021, 14, 71. [Google Scholar] [CrossRef]
- Mareev, V.Y.; Orlova, Y.A.; Plisyk, A.G.; Pavlikova, E.P.; Matskeplishvili, S.T.; Akopyan, Z.A.; Seredenina, E.M.; Potapenko, A.V.; Agapov, M.A.; Asratyan, D.A.; et al. Results of Open-Label non-Randomized Comparative Clinical Trial: “BromhexIne and Spironolactone for Coronsmall a, CyrillicvirUs Infection requiring hospiTalization (BISCUIT). Kardiologiia 2020, 60, 4–15. [Google Scholar] [CrossRef]
- Liu, J.; Zhang, S.; Wu, Z.; Shang, Y.; Dong, X.; Li, G.; Zhang, L.; Chen, Y.; Ye, X.; Du, H.; et al. Clinical outcomes of COVID-19 in Wuhan, China: A large cohort study. Ann. Intensive Care 2020, 10, 99. [Google Scholar] [CrossRef]
| Characteristic | Long-Term Long COVID Group | Long-Term Control Group |
|---|---|---|
| Mean Age | 43.8 ± 13.4 | 43.9 ± 13.7 |
| ≤40 years—no. (%) | 14 (46.7) | 9 (45.0) |
| >40 years—no. (%) | 16 (53.3) | 11 (55.0) |
| Female—no. (%) | 23 (76.7) | 14 (70.0) |
| Male—no. (%) | 7 (23.3) | 6 (30.0) |
| United States Census Region | Long-Term Long COVID Group | Long-Term Control Group |
|---|---|---|
| Region 1: Northeast—no. | 10 | 4 |
| Region 2: Midwest—no. | 5 | 3 |
| Region 3: South—no. | 5 | 6 |
| Region 4: West—no. | 10 | 7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gold, J.E.; Okyay, R.A.; Licht, W.E.; Hurley, D.J. Investigation of Long COVID Prevalence and Its Relationship to Epstein-Barr Virus Reactivation. Pathogens 2021, 10, 763. https://doi.org/10.3390/pathogens10060763
Gold JE, Okyay RA, Licht WE, Hurley DJ. Investigation of Long COVID Prevalence and Its Relationship to Epstein-Barr Virus Reactivation. Pathogens. 2021; 10(6):763. https://doi.org/10.3390/pathogens10060763
Chicago/Turabian StyleGold, Jeffrey E., Ramazan A. Okyay, Warren E. Licht, and David J. Hurley. 2021. "Investigation of Long COVID Prevalence and Its Relationship to Epstein-Barr Virus Reactivation" Pathogens 10, no. 6: 763. https://doi.org/10.3390/pathogens10060763
APA StyleGold, J. E., Okyay, R. A., Licht, W. E., & Hurley, D. J. (2021). Investigation of Long COVID Prevalence and Its Relationship to Epstein-Barr Virus Reactivation. Pathogens, 10(6), 763. https://doi.org/10.3390/pathogens10060763





