Microstructure Evolution in Super Duplex Stainless Steels Containing σ-Phase Investigated at Low-Temperature Using In Situ SEM/EBSD Tensile Testing
Abstract
1. Introduction
2. Materials and Methods
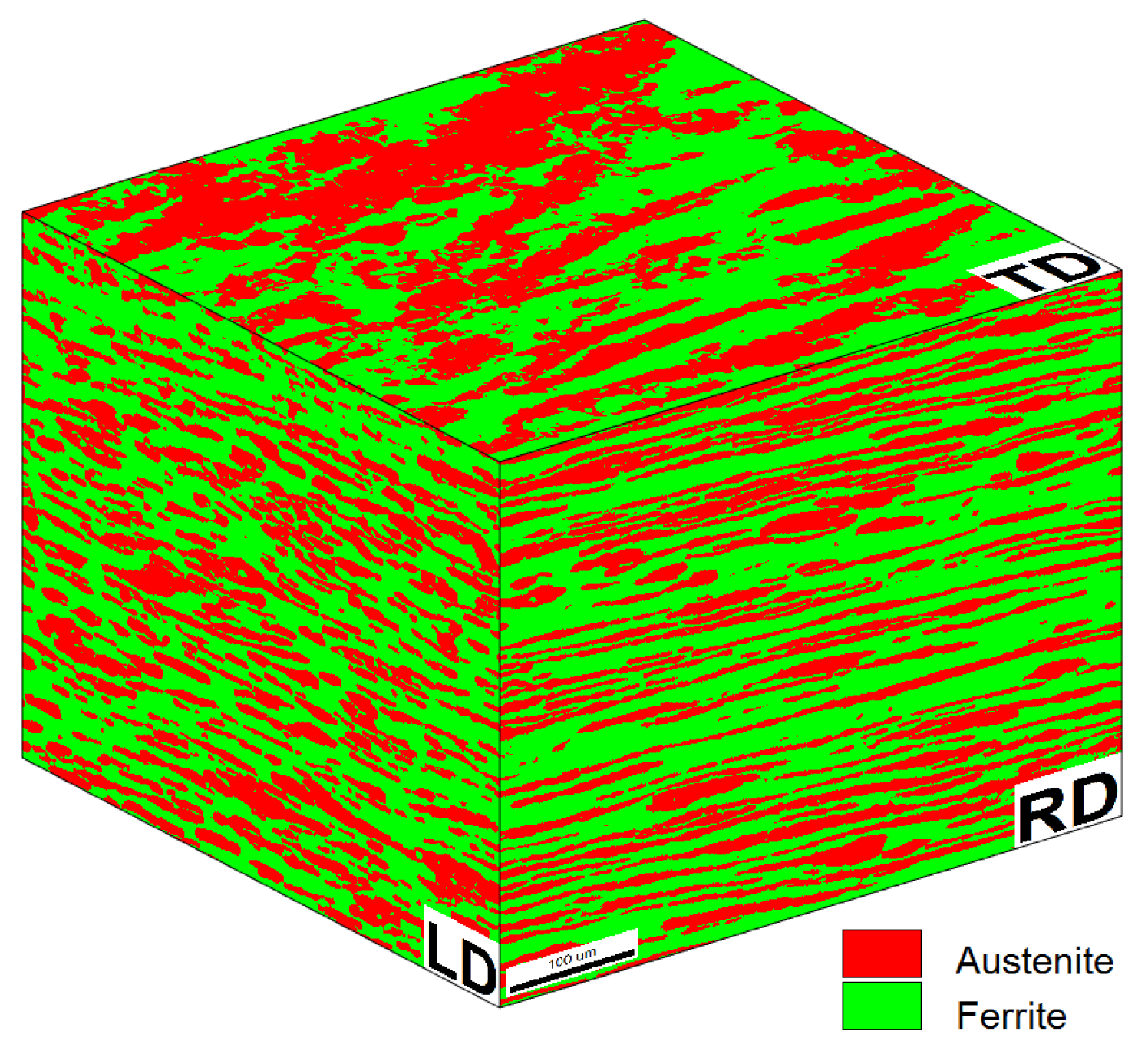
2.1. Material and Heat Treatments
2.2. Materials Characterization
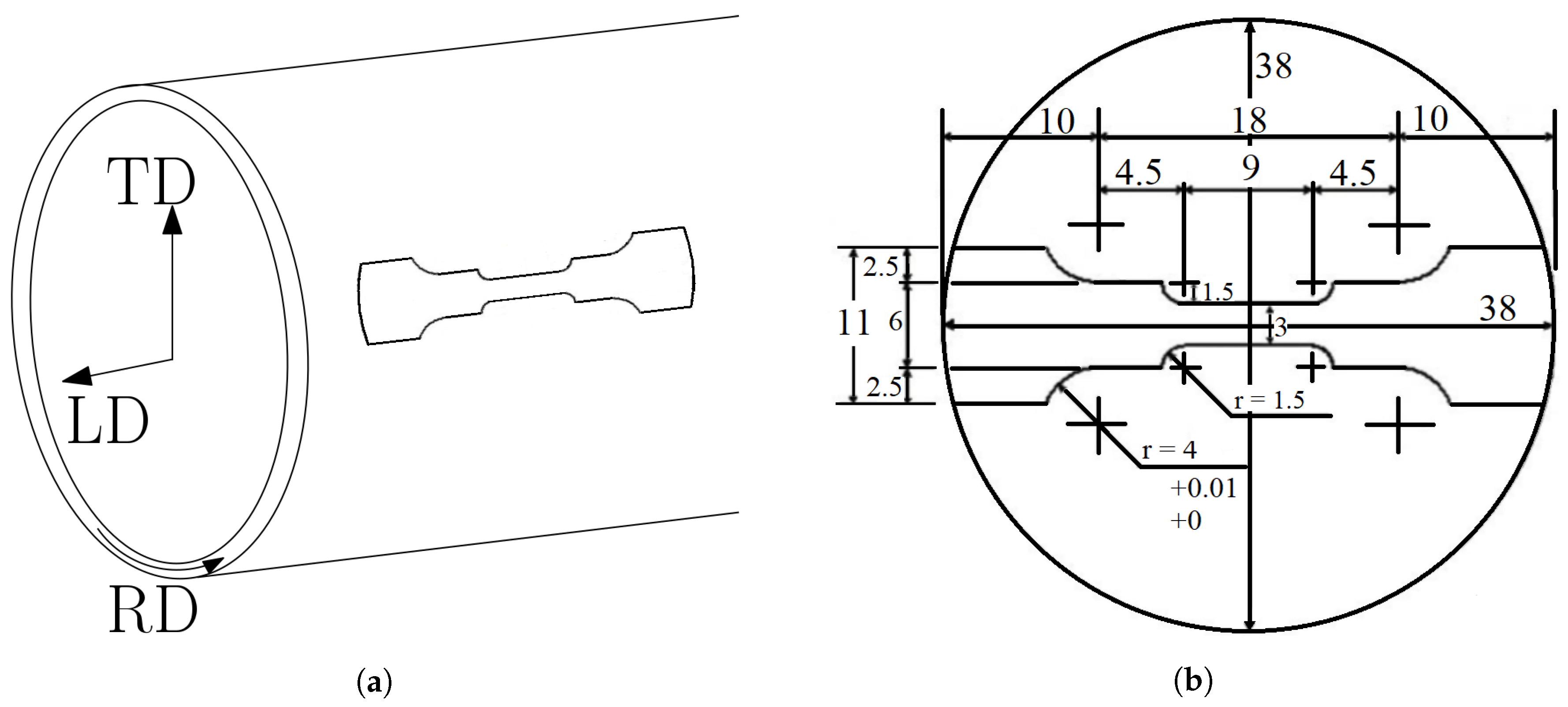
2.3. Tensile Testing
3. Results
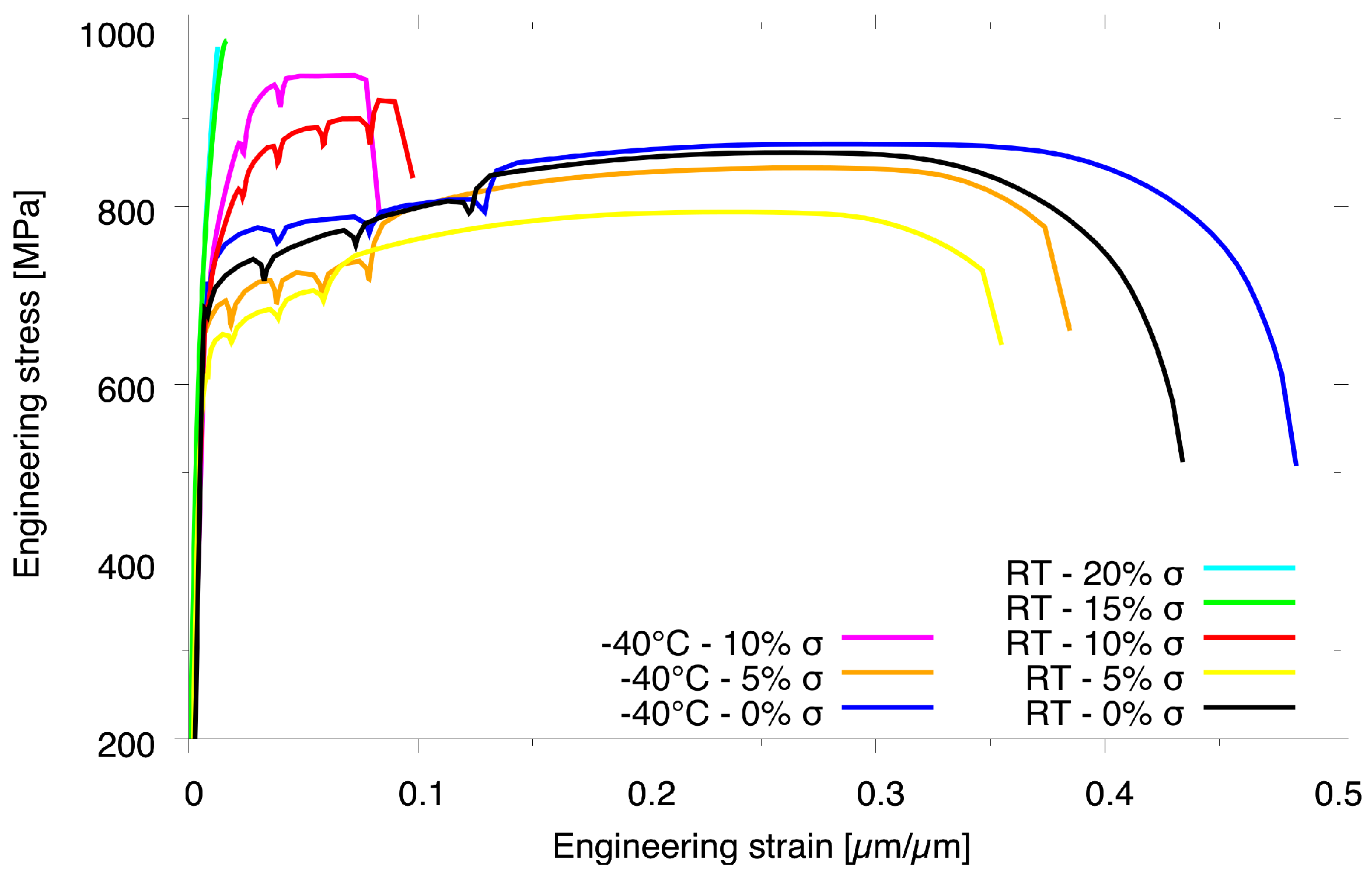
3.1. Tensile Properties and Fracture Surfaces
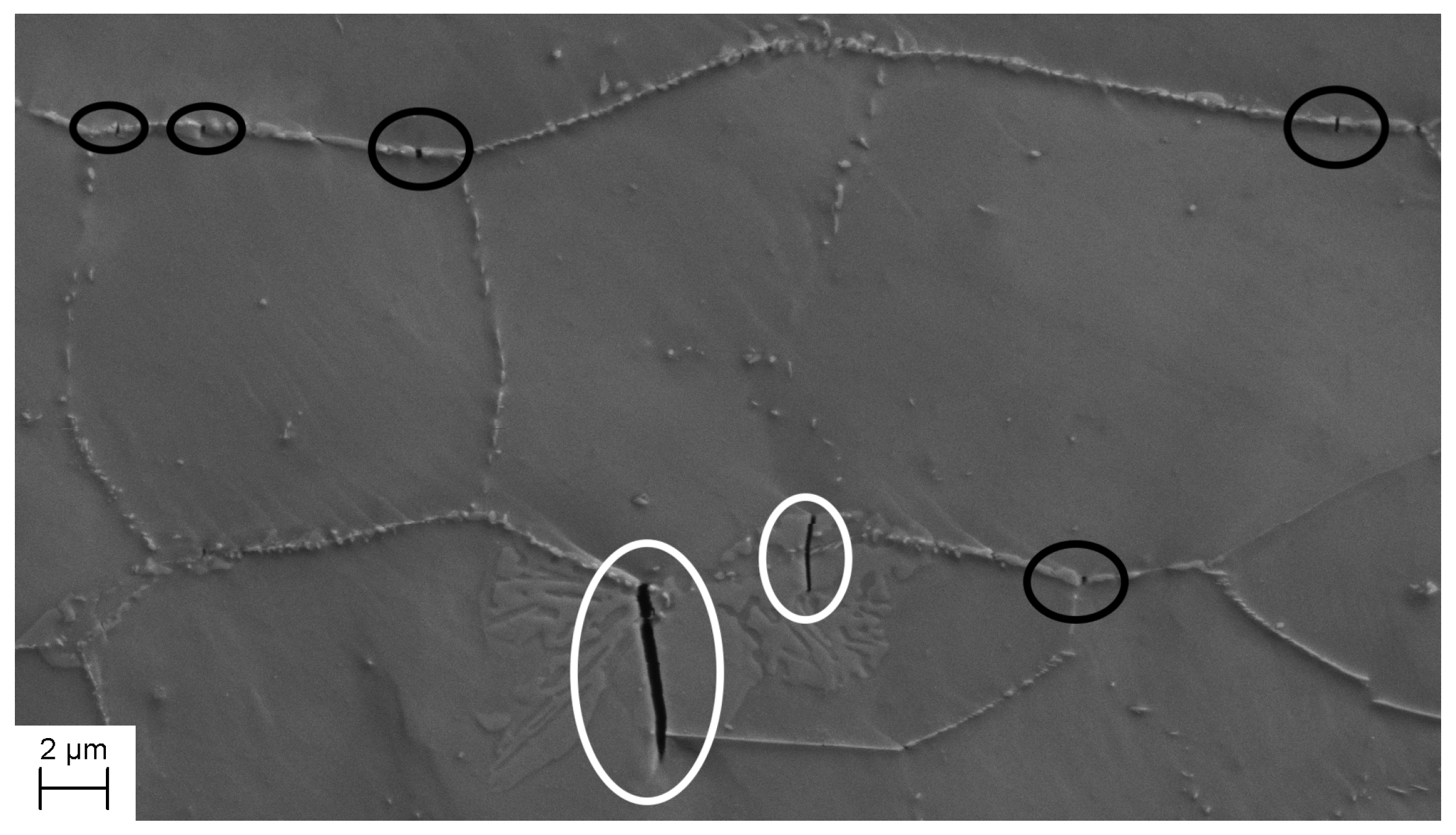
3.2. Microstructure Evolution
4. Discussion
5. Conclusions
- The cracks in -phase contribute towards a lower flow stress but were not of critical size concerning a large reduction in tensile ductility. The specimens with small amounts of -phase and -phase still retained a ductility of 35%.
- Visible cracks start to form after 3–4% strain, regardless of -phase content and they all form perpendicular to the tensile direction.
- During the initial stages of deformation, the cracks are constrained by the ferrite/austenite matrix. However, during the later stages, these cracks start to propagate through the material and coalesce. This occurs moments before fracture.
- The ferrite accommodates more deformation than austenite at room temperature tests; however, during low-temperature tests, both phases have a more equal behavior during deformation.
- At low temperature, with -present, the material had slightly higher flow stress and lower ductility. However, the amount of -phase present is the most important aspect when it comes to duplex steels. It alters the phase balance of ferrite and austenite and deteriorates the mechanical properties.
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| CASA | Centre for Advanced Structural Analysis |
| DSS | Duplex Stainless Steel |
| EBSD | Electron Backscatter Diffraction |
| GOS | Grain Orientation Spread |
| IQ | Image Quality |
| LD | Longitudinal Direction |
| NTNU | Norwegian University of Science and Technology |
| RD | Radial Direction |
| RT | Room Temperature |
| SDSS | Super Duplex Stainless Steel |
| SEM | Scanning Electron Microscope |
| TD | Transverse Direction |
| UTS | Ultimate Tensile Strength |
References
- NORSOK Standard. Materials Selection. 2004. Available online: http://www.standard.no/pagefiles/1174/m-dp-001r1.pdf (accessed on 11 June 2018).
- Børvik, T.; Lange, H.; Marken, L.A.; Langseth, M.; Hopperstad, O.S.; Aursand, M.; Rørvik, G. Pipe fittings in duplex stainless steel with deviation in quality caused by sigma phase precipitation. Mater. Sci. Eng. A 2010, 527, 6945–6955. [Google Scholar] [CrossRef]
- Børvik, T.; Marken, L.A.; Langseth, M.; Rørvik, G.; Hopperstad, O.S. Influence of sigma-phase precipitation on the impact behaviour of duplex stainless steel pipe fittings. Ships Offshore Struct. 2016, 11, 25–37. [Google Scholar] [CrossRef]
- Lee, Y.H.; Kim, K.T.; Lee, Y.D.; Kim, K.Y. Effects of W substitution on ς and χ phase precipitation and toughness in duplex stainless steels. Mater. Sci. Technol. 1998, 14, 757–764. [Google Scholar] [CrossRef]
- Kim, S.B.; Paik, K.W.; Kim, Y.G. Effect of Mo substitution by W on high temperature embrittlement characteristics in duplex stainless steels. Mater. Sci. Eng. A 1998, 247, 67–74. [Google Scholar] [CrossRef]
- Lopez, N.; Cid, M.; Puiggali, M. Influence of σ-phase on mechanical properties and corrosion resistance of duplex stainless steels. Corros. Sci. 1999, 41, 1615–1631. [Google Scholar] [CrossRef]
- Chen, T.H.; Yang, J.R. Effects of solution treatment and continuous cooling on σ-phase precipitation in a 2205 duplex stainless steel. Mater. Sci. Eng. A 2001, 311, 28–41. [Google Scholar] [CrossRef]
- Chen, T.H.; Weng, K.L.; Yang, J.R. The effect of high-temperature exposure on the microstructural stability and toughness property in a 2205 duplex stainless steel. Mater. Sci. Eng. A 2002, 338, 259–270. [Google Scholar] [CrossRef]
- Zucato, I.; Moreira, M.C.; Machado, I.F.; Lebrão, S.M.G. Microstructural characterization and the effect of phase transformations on toughness of the UNS S31803 duplex stainless steel aged treated at 850 °C. Mater. Res. 2002, 5, 385–389. [Google Scholar] [CrossRef]
- Cvijović, Z.; Radenković, G. Microstructure and pitting corrosion resistance of annealed duplex stainless steel. Corros. Sci. 2006, 48, 3887–3906. [Google Scholar] [CrossRef]
- Michalska, J.; Sozańska, M. Qualitative and quantitative analysis of σ and χ phases in 2205 duplex stainless steel. Mater. Charact. 2006, 56, 355–362. [Google Scholar] [CrossRef]
- Souza, C.M.; Abreu, H.F.G.; Tavares, S.S.M.; Rebello, J.M.A. The σ phase formation in annealed UNS S31803 duplex stainless steel: Texture aspects. Mater. Charact. 2008, 59, 1301–1306. [Google Scholar] [CrossRef]
- Pohl, M.; Storz, O.; Glogowski, T. Effect of intermetallic precipitations on the properties of duplex stainless steel. Mater. Charact. 2007, 58, 65–71. [Google Scholar] [CrossRef]
- Calliari, I.; Zanesco, M.; Ramous, E. Influence of isothermal aging on secondary phases precipitation and toughness of a duplex stainless steel SAF 2205. J. Mater. Sci. 2006, 41, 7643–7649. [Google Scholar] [CrossRef]
- Escriba, D.; Materna-Morris, E.; Plaut, R.; Padilha, A. Chi-phase precipitation in a duplex stainless steel. Mater. Charact. 2009, 60, 1214–1219. [Google Scholar] [CrossRef]
- Elstad, K.R. In Situ Tensile Testing During Continuous EBSD Mapping of Super Duplex Stainless Steel Containing Sigma Phase. 2016. Available online: http://hdl.handle.net/11250/2418016 (accessed on 25 May 2018).
- Stradomski, Z.; Dyja, D. Sigma Phase Precipitation in Duplex Phase Stainless Steels. 2009. Available online: http://www.ysesm.ing.unibo.it/Abstract/57Dyja.pdf (accessed on 25 May 2018).
- Padilha, A.F.; Rios, P.R. Decomposition of Austenite in Austenitic Stainless Steels. ISIJ Int. 2002, 42, 325–327. [Google Scholar] [CrossRef]
- Cahn, R.W.; Haasen, P.; Kramer, E.J. Materials Science and Technology: A Comprehensive Treatment—Volume 1: Structure of Solids; Wiley-VCH: Weinheim, Germnay, 2005. [Google Scholar]
- Cahn, R.W.; Haasen, P.; Kramer, E.J. Materials Science and Technology: A Comprehensive Treatment—Volume 7: Constitution and Properties of Steel; Wiley-VCH: Weinheim, Germnay, 2005. [Google Scholar]
- Kim, S.K.; Kang, K.Y.; Kim, M.S.; Lee, J.M. Low-temperature mechanical behavior of super duplex stainless steel with sigma precipitation. Metals 2015, 5, 1732–1745. [Google Scholar] [CrossRef]
- Karlsen, M.; Hjelen, J.; Grong, Ø.; Rørvik, G.; Chiron, R.; Schubert, U.; Nilsen, E. SEM/EBSD based in situ studies of deformation induced phase transformations in supermartensitic stainless steels. Mater. Sci. Technol. 2008, 24, 64–72. [Google Scholar] [CrossRef]
- Karlsen, M.; Grong, Ø.; Søfferud, M.; Hjelen, J.; Rørvik, G.; Chiron, R. Scanning Electron Microscopy/Electron Backscatter Diffraction—Based Observations of Martensite Variant Selection and Slip Plane Activity in Supermartensitic Stainless Steels during Plastic Deformation at Elevated, Ambient, and Subzero Temperatures. Metall. Mater. Trans. A 2009, 40, 310–320. [Google Scholar] [CrossRef]
- Jorge-Badiola, D.; Iza-Mendia, A.; Gutiérrez, I. Study by EBSD of the development of the substructure in a hot deformed 304 stainless steel. Mater. Sci. Eng. A 2005, 394, 445–454. [Google Scholar] [CrossRef]
- Mitsche, S.; Poelt, P.; Sommitsch, C. Recrystallization behaviour of the nickel-based alloy 80 A during hot forming. J. Microsc. 2007, 227, 267–274. [Google Scholar] [CrossRef] [PubMed]
- Byun, T.; Hashimoto, N.; Farrell, K. Temperature dependence of strain hardening and plastic instability behaviors in austenitic stainless steels. Acta Mater. 2004, 52, 3889–3899. [Google Scholar] [CrossRef]
- Lee, K.J.; Chun, M.S.; Kim, M.H.; Lee, J.M. A new constitutive model of austenitic stainless steel for cryogenic applications. Comput. Mater. Sci. 2009, 46, 1152–1162. [Google Scholar] [CrossRef]
- Park, W.S.; Yoo, S.W.; Kim, M.H.; Lee, J.M. Strain-rate effects on the mechanical behavior of the AISI 300 series of austenitic stainless steel under cryogenic environments. Mater. Des. 2010, 31, 3630–3640. [Google Scholar] [CrossRef]






| Element | C | Si | Mn | P | S | Cr | Mo | Ni | Cu | W | N |
|---|---|---|---|---|---|---|---|---|---|---|---|
| wt% | 0.018 | 0.42 | 0.52 | 0.017 | 0.001 | 25.55 | 3.46 | 8.28 | 0.72 | 0.52 | 0.25 |
| Average | Ferrite | Austenite | Overall |
|---|---|---|---|
| Composition | 56.3% | 43.7% | 100% |
| Grain size | 9 µm | 6.5 µm | 7.9 µm |
| Electrolyte | Struers A2 |
|---|---|
| Voltage [V] | 20 |
| Time [s] | 15 |
| Temperature [°C] | 22 |
| Temperature [°C] | Time [min] | Amount -Phase [%] |
|---|---|---|
| - | - | 0 |
| 850 | 12 | 5 |
| 850 | 15 | 10 |
| 850 | 20 | 15 |
| 850 | 25 | 20 |
| Acceleration Voltage [kV] | 20 |
|---|---|
| Working distance [mm] | 24.6–25.4 |
| Tilt angle [°] | 70 |
| Aperture size [µm] | 300 |
| Probe current [nA] | 65–70 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paulsen, C.O.; Broks, R.L.; Karlsen, M.; Hjelen, J.; Westermann, I. Microstructure Evolution in Super Duplex Stainless Steels Containing σ-Phase Investigated at Low-Temperature Using In Situ SEM/EBSD Tensile Testing. Metals 2018, 8, 478. https://doi.org/10.3390/met8070478
Paulsen CO, Broks RL, Karlsen M, Hjelen J, Westermann I. Microstructure Evolution in Super Duplex Stainless Steels Containing σ-Phase Investigated at Low-Temperature Using In Situ SEM/EBSD Tensile Testing. Metals. 2018; 8(7):478. https://doi.org/10.3390/met8070478
Chicago/Turabian StylePaulsen, Christian Oen, Runar Larsen Broks, Morten Karlsen, Jarle Hjelen, and Ida Westermann. 2018. "Microstructure Evolution in Super Duplex Stainless Steels Containing σ-Phase Investigated at Low-Temperature Using In Situ SEM/EBSD Tensile Testing" Metals 8, no. 7: 478. https://doi.org/10.3390/met8070478
APA StylePaulsen, C. O., Broks, R. L., Karlsen, M., Hjelen, J., & Westermann, I. (2018). Microstructure Evolution in Super Duplex Stainless Steels Containing σ-Phase Investigated at Low-Temperature Using In Situ SEM/EBSD Tensile Testing. Metals, 8(7), 478. https://doi.org/10.3390/met8070478






