Simultaneous Improvement of Glass-Forming Ability and Ductility in Co-Based BMGs Through Si/Fe Microalloying
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. Composition Design of Co-Based BMGs with Enhanced GFA
3.2. Thermal Properties
3.3. Mechanical Properties
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Inoue, A.; Shen, B.L.; Koshiba, H.; Kato, H.; Yavari, A.R. Cobalt-based bulk glassy alloy with ultrahigh strength and soft magnetic properties. Nat. Mater. 2003, 2, 661–663. [Google Scholar] [CrossRef]
- Zhang, G.L.; Wang, Q.Q.; Yuan, C.C.; Yang, W.M.; Zhou, J.; Xue, L.; Hu, F.; Sun, B.A.; Shen, B.L. Effects of Cu additions on mechanical and soft-magnetic properties of CoFeBSiNb bulk metallic glasses. J. Alloys Compd. 2018, 737, 815–820. [Google Scholar] [CrossRef]
- Taghvaei, A.H.; Stoica, M.; Prashanth, K.G.; Eckert, J. Fabrication and characterization of bulk glassy Co40Fe22Ta8B30 alloy with high thermal stability and excellent soft magnetic properties. Acta Mater. 2013, 61, 6609–6621. [Google Scholar] [CrossRef]
- Huang, D.W.; Li, Y.H.; Yang, Y.P.; Zhu, Z.W.; Zhang, W. Soft magnetic Co-based Co-Fe-B-Si-P bulk metallic glasses with high saturation magnetic flux density of over 1.2 T. J. Alloys Compd. 2020, 843, 154862. [Google Scholar] [CrossRef]
- Ma, H.R.; Mu, C.Y.; Yang, M.S.; Mao, F.X.; He, A.N.; Dong, Y.Q.; Man, Q.K.; Li, J.W. Effect of structural heterogeneity on stable pit growth of Co-based metallic glasses. Corros. Sci. 2024, 232, 112032. [Google Scholar] [CrossRef]
- Zhang, G.Y.; Zhang, H.; Yue, S.Q.; Cheng, R.J.; Wang, A.D.; He, A.N.; Dong, Y.Q.; Ni, H.W.; Liu, C.T. Preparation of non-magnetic and ductile Co-based bulk metallic glasses with high GFA and hardness. Intermetallics 2019, 107, 47–52. [Google Scholar] [CrossRef]
- Liang, X.Y.; Li, Y.H.; Bao, F.; Zhu, Z.W.; Zhang, H.F.; Zhang, W. Roles of Y and Fe contents on glass-forming ability, thermal stability, and magnetic properties of Co-based CoFeYB bulk metallic glasses. Intermetallics 2021, 132, 107135. [Google Scholar] [CrossRef]
- Wang, J.F.; Li, R.; Hua, N.B.; Zhang, T. Co-based ternary bulk metallic glasses with ultrahigh strength and plasticity. J. Mater. Res. 2011, 26, 2072–2079. [Google Scholar] [CrossRef]
- Ma, X.; Li, Q.; Xie, L.; Chang, C.T.; Li, H.X. Effect of Ni addition on the properties of CoMoPB bulk metallic glasses. J. Non-Cryst. Solids 2022, 587, 121573. [Google Scholar] [CrossRef]
- Murali, P.; Narasimhan, R.; Guo, T.F.; Zhang, Y.W.; Gao, H.J. Shear bands mediate cavitation in brittle metallic glasses. Scr. Mater. 2013, 68, 567–570. [Google Scholar] [CrossRef]
- Bie, L.Y.; Li, Q.; Cao, D.; Li, H.X.; Zhang, J.J.; Chang, C.T.; Sun, Y.F. Preparation and properties of quaternary CoMoPB bulk metallic glasses. Intermetallics 2016, 71, 7–11. [Google Scholar] [CrossRef]
- Wang, J.; Kaban, I.; Levytskyi, V.; Li, R.; Han, J.; Stoica, M.; Gumeniuk, R.; Nielsch, K. Ultra-high strength Co-Ta-B bulk metallic glasses: Glass formation, thermal stability and crystallization. J. Alloys Compd. 2021, 860, 158398. [Google Scholar] [CrossRef]
- Zhang, T.; Yang, Q.; Ji, Y.F.; Li, R.; Pang, S.J.; Wang, J.F.; Xu, T. Centimeter-scale-diameter Co-based bulk metallic glasses with fracture strength exceeding 5000 MPa. Chin. Sci. Bull. 2011, 56, 3972–3977. [Google Scholar] [CrossRef]
- Inoue, A.; Shen, B.L.; Chang, C.T. Super-high strength of over 4000 MPa for Fe-based bulk glassy alloys in ((Fe1−xCox)0.75B0.2Si0.05)96Nb4 system. Acta Mater. 2004, 52, 4093–4099. [Google Scholar] [CrossRef]
- Inoue, A. Recent progress of Zr-based bulk amorphous alloys. Sci. Rep. RITU 1996, A 42, 1–11. [Google Scholar]
- Zhang, J.Y.; Ma, S.; Wang, H.; Kubota, T.; Li, Y.H.; Kato, H.; Umetsu, R.Y.; Yao, M.; Zhang, W. Effects and mechanisms of Nb and Fe additions on glass-forming ability and magnetic properties of a Co71.5Gd3.5B25 metallic glass. J. Alloys Compd. 2023, 968, 171875. [Google Scholar] [CrossRef]
- Ren, J.P.; Li, Y.H.; Liang, X.Y.; Kato, H.; Zhang, W. Role of Fe substitution for Co on thermal stability and glass-forming ability of soft magnetic Co-based Co-Fe-B-P-C metallic glasses. Intermetallics 2022, 147, 107598. [Google Scholar] [CrossRef]
- Zhao, Y.; Shang, B.S.; Zhang, B.; Tong, X.; Ke, H.B.; Bai, H.Y.; Wang, W.H. Ultrastable metallic glass by room temperature aging. Sci. Adv. 2022, 8, eabn3623. [Google Scholar] [CrossRef]
- Lu, J.; Li, Y.H.; Ma, S.; Li, W.P.; Bao, F.; Zhu, Z.W.; Zeng, Q.S.; Zhang, H.F.; Yao, M.; Zhang, W. Novel Soft Magnetic Co-Based Ternary Co-Er-B Bulk Metallic Glasses. Acta Metall. Sin. (Engl. Lett.) 2024, 37, 1633–1642. [Google Scholar] [CrossRef]
- Ma, S.; Ran, Y.Z.; Liang, X.Y.; Jiang, L.; Li, Y.H.; Wang, X.D.; Yao, M.; Zhang, W. Unveiling the role of Y content in glass-forming ability and soft magnetic properties of Co-Y-B metallic glasses by experiment and ab initio molecular dynamics simulations. J. Alloys Compd. 2022, 902, 163637. [Google Scholar] [CrossRef]
- Ma, S.; Lu, J.; Wang, Y.Y.; Li, Y.H.; Wang, X.D.; Zeng, Q.S.; Zhang, W.; Yao, M. Role of rare earth elements addition in enhancing glass-forming ability and magnetic softness of a Co75B25 metallic glass: Theoretical prediction and experimental verification. J. Mater. Res. Technol. 2023, 26, 6193–6204. [Google Scholar] [CrossRef]
- Li, R.; Pang, S.; Ma, C.; Zhang, T. Influence of similar atom substitution on glass formation in (La–Ce)–Al–Co bulk metallic glasses. Acta Mater. 2007, 55, 3719–3726. [Google Scholar] [CrossRef]
- Kwon, S.; Kim, S.; Choi-Yim, H.; Lee, D.; Kwon, S.; Kim, K. Effects of Fe substitution for Co on the thermal, magnetic, and mechanical properties of the Co-Fe-B-Si-Mo alloy system. J. Korean Phys. Soc. 2018, 72, 171–176. [Google Scholar] [CrossRef]
- Zhao, Y.M.; Li, X.; Liu, X.B.; Bi, J.Z.; Wu, Y.; Xiao, R.J.; Li, R.; Zhang, T. Balancing benefits of strength, plasticity and glass-forming ability in Co-based metallic glasses. J. Mater. Sci. Technol. 2021, 86, 110–116. [Google Scholar] [CrossRef]
- Jia, S.K.; Jiang, Y.H.; Chen, S.W.; Han, X.J. Enhancing glass forming ability and magnetic properties of Co-Fe-Si-B metallic glasses by similar element substitution: Experimental and theoretical investigations. Comput. Mater. Sci. 2022, 213, 111639. [Google Scholar] [CrossRef]
- Yuan, C.C.; Lv, Z.W.; Pang, C.M.; Wu, X.L.; Lan, S.; Lu, C.Y.; Wang, L.G.; Yu, H.B.; Luan, J.H.; Zhu, W.W.; et al. Atomic-scale heterogeneity in large-plasticity Cu-doped metallic glasses. J. Alloys Compd. 2019, 798, 517–522. [Google Scholar] [CrossRef]
- Dun, C.C.; Liu, H.S.; Shen, B.L. Enhancement of plasticity in Co-Nb-B ternary bulk metallic glasses with ultrahigh strength. J. Non-Cryst. Solids 2012, 358, 3060–3064. [Google Scholar] [CrossRef]
- Zhang, B.; Wang, R.J.; Zhao, D.Q.; Pan, M.X.; Wang, W.H. Superior glass-forming ability through microalloying in cerium-based alloys. Phys. Rev. B 2006, 73, 092201. [Google Scholar] [CrossRef]
- Guo, G.Q.; Yang, L.; Wu, S.Y.; Zeng, Q.S.; Sun, C.J.; Wang, Y.G. Structure-induced microalloying effect in multicomponent alloys. Mater. Des. 2016, 103, 308–314. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhang, B.; Sato, K.; Wang, W.H. Microalloying suppresses the formation of ultrastable Ce-based bulk metallic glasses. Appl. Phys. Lett. 2025, 126, 191903. [Google Scholar] [CrossRef]
- Zhao, Y.; Sato, K. Mechanism of glass-forming ability enhancement upon micro solute addition for Ce-Ga-Cu-Ni bulk metallic glasses. Intermetallics 2019, 114, 106603. [Google Scholar] [CrossRef]
- Yue, L.Q.; Sun, Q.J.; Wang, Y.H.; Wang, Y.; Xue, R.J.; Li, X.; Yang, G.J.; Yi, J.J. Si-alloying effect on glass-forming ability and crystallization behavior of Zr72.5Al10Fe17.5 metallic glass. J. Non-Cryst. Solids 2022, 580, 121391. [Google Scholar] [CrossRef]
- Shao, L.L.; Xue, L.; Wang, Q.Q.; Ma, K.L.; Huang, J.D.; Shen, B.L. Effects of Si addition on glass-forming ability and crystallization behavior of DyCoAl bulk metallic glass. J. Alloys Compd. 2021, 874, 159964. [Google Scholar] [CrossRef]
- Li, X.; Shi, Z.G.; Zhang, T. Effect of similar element substitution on Fe-B-Si-Mo bulk metallic glasses studied by experiment and ab initio molecular dynamics simulation. J. Alloys Compd. 2019, 784, 1139–1144. [Google Scholar] [CrossRef]
- Wang, Q.; Zhu, C.L.; Dong, C.; Qiang, J.B.; Zhang, W.; Inoue, A. Formation and soft magnetic properties of Co (-Fe)-Si-B-Nb bulk metallic glasses in relation to clusters. In Proceedings of the 13th Conference on Liquid and Amorphous Metals, Ekaterinburg, Russia, 8–14 July 2007; p. 98. [Google Scholar]
- Inoue, A. High Strength Bulk Amorphous Alloys with Low Critical Cooling Rates (Overview). Mater. Trans. 1994, 35, 85–92. [Google Scholar] [CrossRef]
- Hosko, J.; Janotova, I.; Svec, P.; Matko, I.; Janickovic, D.; Svec, P., Sr. Structure analysis of CoFeBSiNb(Ga) pseudobulk metallic glasses. Appl. Surf. Sci. 2013, 269, 77–80. [Google Scholar] [CrossRef]
- Hosko, J.; Janotova, I.; Svec, P.; Janickovic, D.; Vlasak, G.; Illekova, E.; Matko, I.; Svec, P., Sr. Preparation of thin ribbon and bulk glassy alloys in CoFeBSiNb(Ga) using planar flow casting and suction casting methods. J. Non-Cryst. Solids 2012, 358, 1545–1549. [Google Scholar] [CrossRef]
- Takeuchi, A.; Inoue, A. Classification of Bulk Metallic Glasses by Atomic Size Difference, Heat of Mixing and Period of Constituent Elements and Its Application to Characterization of the Main Alloying Element (Overview). Mater. Trans. 2005, 46, 2817–2829. [Google Scholar] [CrossRef]
- Stein, F.; Jiang, D.; Palm, M.; Sauthoff, G.; Grüner, D.; Kreiner, G. Experimental reinvestigation of the Co–Nb phase diagram. Intermetallics 2008, 16, 785–792. [Google Scholar] [CrossRef]
- Du, Y.; Schuster, J.; Chang, Y.A.; Jin, Z.; Huang, B.Y. A thermodynamic description of the B–Co system: Modeling and experiment. Int. J. Mater. Res. 2002, 93, 1157–1163. [Google Scholar] [CrossRef]
- Cai, A.H.; Xiong, X.; Liu, Y.; Li, J.H.; Luo, Y. Characteristics of near-eutectic and off-eutectic Zr–Al–Ni–Cu glass forming alloys. Mater. Sci. Eng. A 2009, 516, 100–102. [Google Scholar] [CrossRef]
- Sun, Y.J.; Qu, D.D.; Huang, Y.J.; Liss, K.D.; Wei, X.S.; Xing, D.W.; Shen, J. Zr–Cu–Ni–Al bulk metallic glasses with superhigh glass-forming ability. Acta Mater. 2009, 57, 1290–1299. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhang, B. Evaluating the correlation between liquid fragility and glass-forming ability in the extremely strong Ce-based bulk metallic glasses. J. Appl. Phys. 2017, 122, 115107. [Google Scholar] [CrossRef]
- Sun, B.A.; Yu, H.B.; Jiao, W.; Bai, H.Y.; Zhao, D.Q.; Wang, W.H. Plasticity of Ductile Metallic Glasses: A Self-Organized Critical State. Phys. Rev. Lett. 2012, 109, 189904(E). [Google Scholar] [CrossRef]
- Shen, B.; Inoue, A.; Chang, C. Superhigh strength and good soft-magnetic properties of (Fe,Co)-B-Si-Nb bulk glassy alloys with high glass-forming ability. Appl. Phys. Lett. 2011, 85, 4911–4913. [Google Scholar] [CrossRef]
- Ramasamy, P.; Stoica, M.; Bera, S.; Calin, M.; Eckert, J. Effect of replacing Nb with (Mo and Zr) on glass forming ability, magnetic and mechanical properties of FeCoBSiNb bulk metallic glass. J. Alloys Compd. 2017, 707, 78–81. [Google Scholar] [CrossRef]
- Inoue, A.; Shen, B.L. Formation and applications of bulk glassy alloys in late transition metal base system. In Proceedings of the 2nd International Conference on Flow Dynamics, Sendai, Japan, 16–18 November 2005; Volume 832, p. 11. [Google Scholar]
- Inoue, A.; Shen, B.L. Formation and soft magnetic properties of Co-Fe-Si-B-Nb bulk glassy alloys. Mater. Trans. 2002, 43, 1230–1234. [Google Scholar] [CrossRef]
- Schnabel, V.; Köhler, M.; Music, D.; Bednarcik, J.; Clegg, W.J.; Raabe, D.; Schneider, J.M. Ultra-stiff metallic glasses through bond energy density design. J. Phys.-Condens. Mater. 2017, 29, 265502. [Google Scholar] [CrossRef]
- Lai, L.M.; He, R.G.; Ding, K.L.; Liu, T.H.; Liu, R.Y.; Chen, Y.X.; Guo, S.F. Ternary Co-Mo-B bulk metallic glasses with ultrahigh strength and good ductility. J. Non-Cryst. Solids 2019, 524, 119657. [Google Scholar] [CrossRef]
- Lai, L.M.; Ding, K.L.; Liu, T.H.; Chen, Y.X.; Guo, S.F. Ternary Co-W-B bulk metallic glasses with ultrahigh strength. J. Non-Cryst. Solids 2020, 544, 120194. [Google Scholar] [CrossRef]
- Aihemaiti, N.; Li, Q.; Li, M.; Chang, C.; Li, H.; Ma, X. Preparation of CoMoPC bulk metallic glasses using fluxing treatment and J-quenching technique. Mater. Sci. Technol. 2020, 36, 1756–1761. [Google Scholar] [CrossRef]
- Wang, Q.Q.; Zhang, G.L.; Zhou, J.; Yuan, C.C.; Shen, B.L. Effects of Ni substitution for Fe/Co on mechanical and magnetic properties of Co-based bulk metallic glasses. J. Alloys Compd. 2020, 820, 153105. [Google Scholar] [CrossRef]
- Li, L.; Sun, H.J.; Fang, Y.Z.; Zheng, J.L. Co-based soft magnetic bulk glassy alloys optimized for glass-forming ability and plasticity. Bull. Mater. Sci. 2016, 39, 691–695. [Google Scholar] [CrossRef]
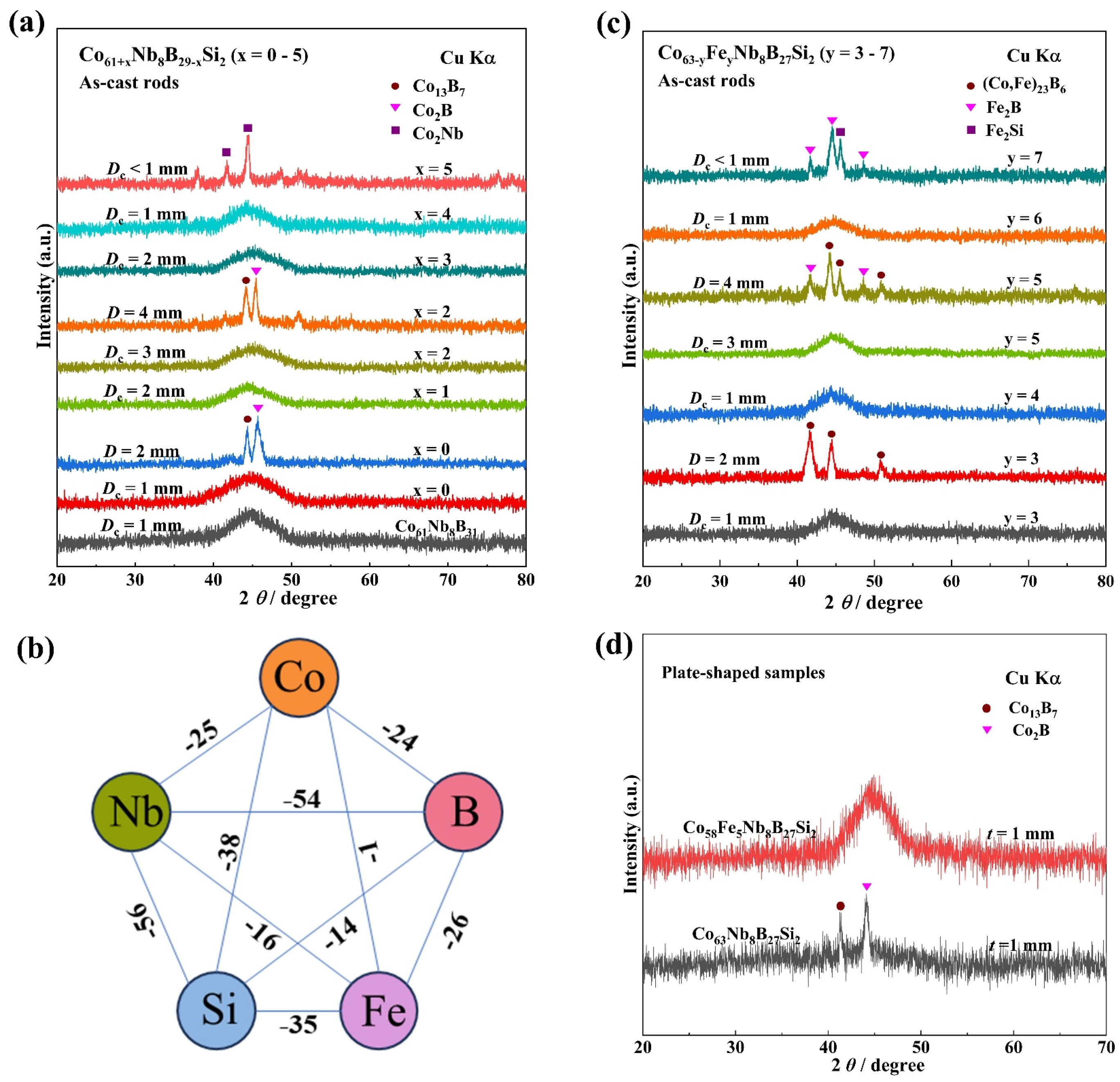
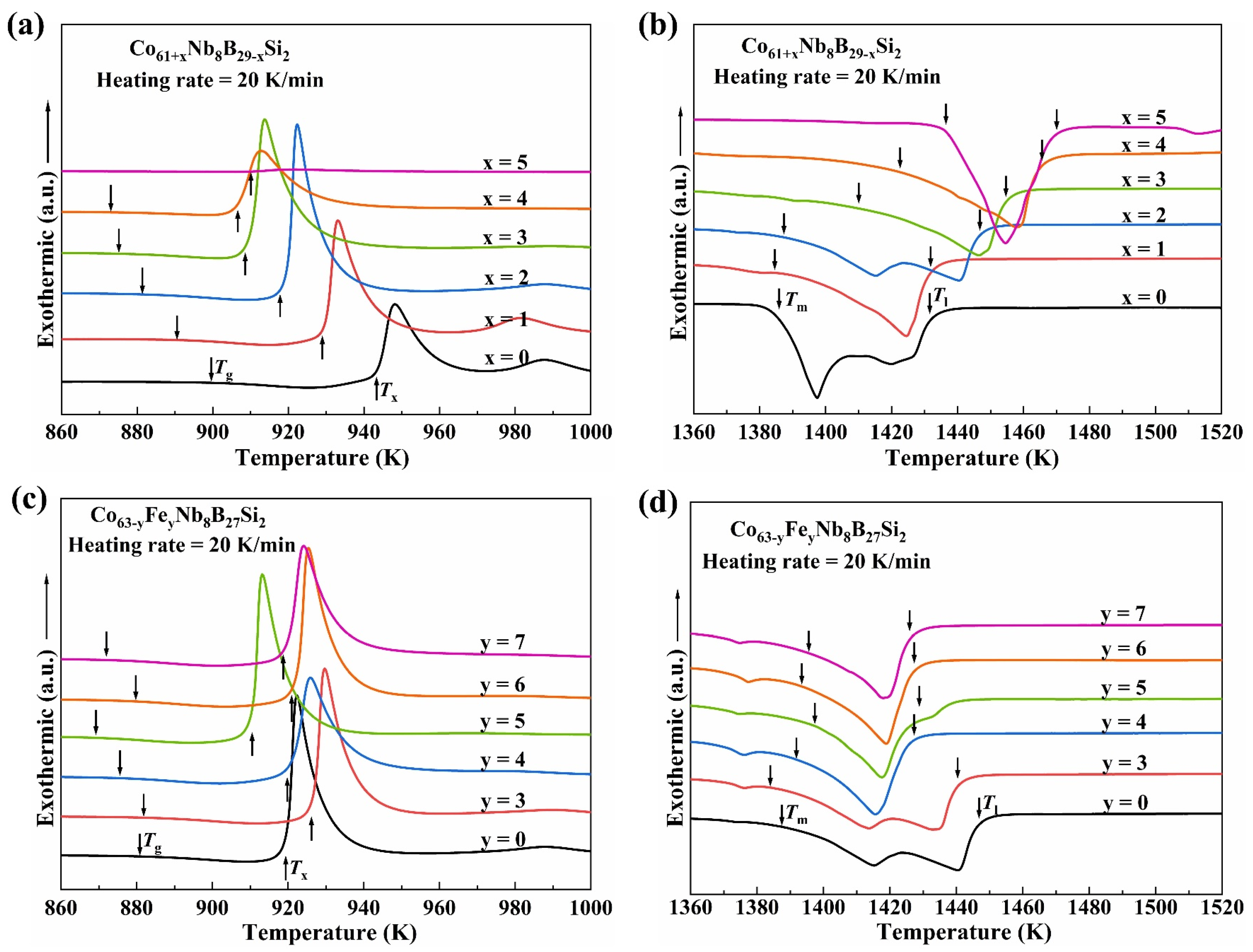
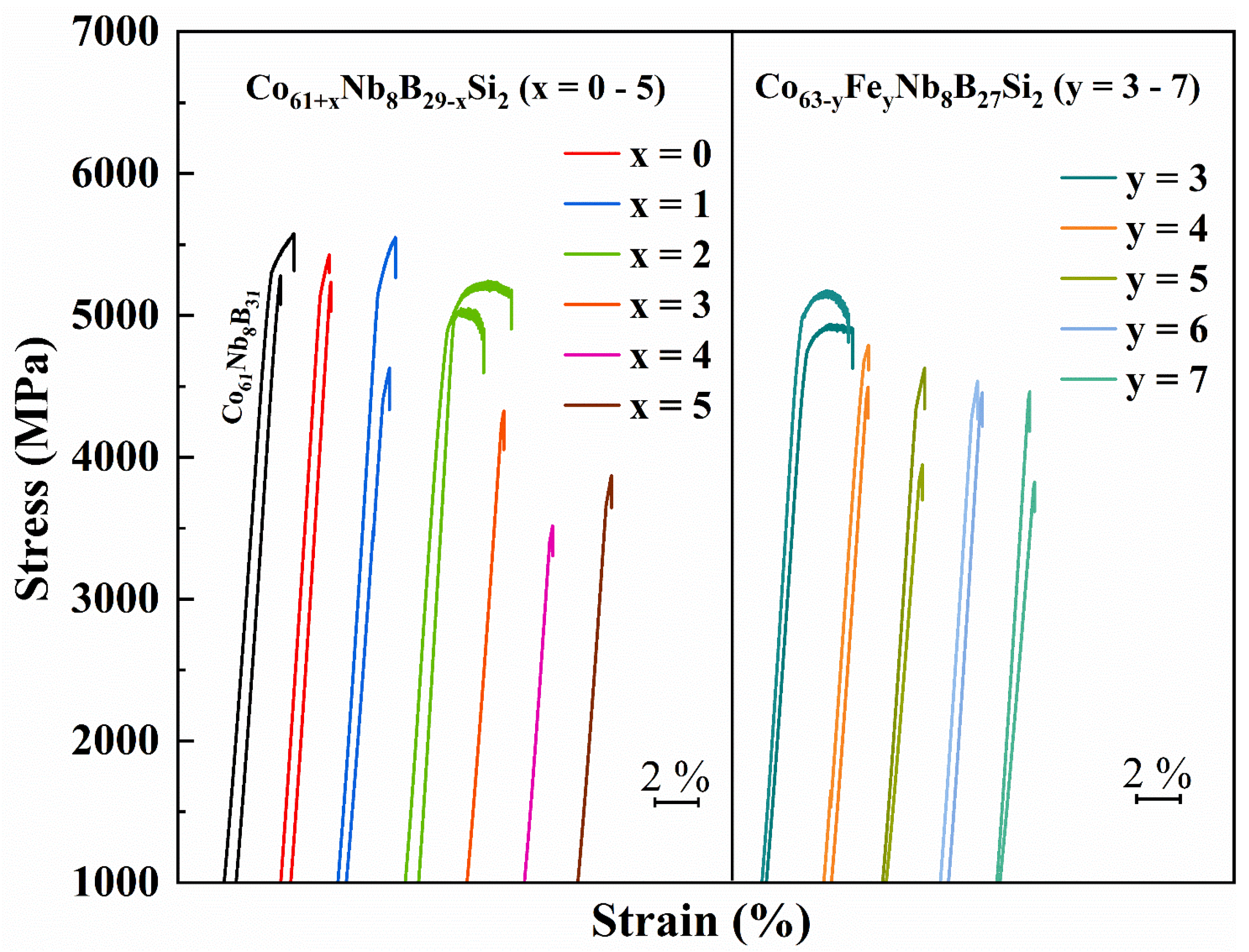


| Alloys | Dc (mm) | Tg (K) | Tx (K) | Tm (K) | Tl (K) | ΔTx (K) | σf (MPa) | εp (%) | E (GPa) |
|---|---|---|---|---|---|---|---|---|---|
| Co61Nb8B31 | 1 | 899.6 | 930.7 | 1432.5 | 1462.1 | 31.1 | 5576 | 1.85 | 225 |
| Co61Nb8B29Si2 | 1 | 899.3 | 943.8 | 1385.8 | 1431.3 | 44.5 | 5426 | 0.68 | 239 |
| Co62Nb8B28Si2 | 2 | 891.4 | 929.2 | 1384.4 | 1431.8 | 37.8 | 5549 | 1.04 | 232 |
| Co63Nb8B27Si2 | 3 | 881.0 | 919.6 | 1387.4 | 1446.8 | 38.6 | 5177 | 3.81 | 228 |
| Co64Nb8B26Si2 | 2 | 875.3 | 909.8 | 1410.0 | 1454.6 | 34.5 | 4326 | 0.29 | 226 |
| Co65Nb8B25Si2 | 1 | 872.9 | 906.6 | 1422.7 | 1465.5 | 33.7 | 3517 | 0.32 | 223 |
| Co66Nb8B24Si2 | <1 | - | 909.7 | 1436.5 | 1469.9 | - | 3871 | 0.20 | 222 |
| Co60Fe3Nb8B27Si2 | 1 | 881.9 | 927.1 | 1383.7 | 1440.3 | 45.2 | 5010 | 2.86 | 233 |
| Co59Fe4Nb8B27Si2 | 1 | 876.6 | 920.5 | 1391.2 | 1426.2 | 43.9 | 4789 | 1.11 | 226 |
| Co58Fe5Nb8B27Si2 | 3 | 868.5 | 912.7 | 1397.0 | 1428.8 | 44.2 | 4628 | 0.85 | 236 |
| Co57Fe6Nb8B27Si2 | 2 | 879.6 | 921.7 | 1393.2 | 1427.1 | 42.1 | 4537 | 0.88 | 236 |
| Co56Fe7Nb8B27Si2 | 1 | 872.1 | 919.8 | 1395.9 | 1425.8 | 47.7 | 4462 | 0.77 | 246 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Quan, X.; Xu, L.; Zhao, Y.; Tang, X.; Liu, Q.; Zhang, B.; Wang, W.-H. Simultaneous Improvement of Glass-Forming Ability and Ductility in Co-Based BMGs Through Si/Fe Microalloying. Metals 2025, 15, 943. https://doi.org/10.3390/met15090943
Quan X, Xu L, Zhao Y, Tang X, Liu Q, Zhang B, Wang W-H. Simultaneous Improvement of Glass-Forming Ability and Ductility in Co-Based BMGs Through Si/Fe Microalloying. Metals. 2025; 15(9):943. https://doi.org/10.3390/met15090943
Chicago/Turabian StyleQuan, Xinlong, Liming Xu, Yong Zhao, Xuecheng Tang, Qing Liu, Bo Zhang, and Wei-Hua Wang. 2025. "Simultaneous Improvement of Glass-Forming Ability and Ductility in Co-Based BMGs Through Si/Fe Microalloying" Metals 15, no. 9: 943. https://doi.org/10.3390/met15090943
APA StyleQuan, X., Xu, L., Zhao, Y., Tang, X., Liu, Q., Zhang, B., & Wang, W.-H. (2025). Simultaneous Improvement of Glass-Forming Ability and Ductility in Co-Based BMGs Through Si/Fe Microalloying. Metals, 15(9), 943. https://doi.org/10.3390/met15090943






