Abstract
Advanced high-temperature oxidation resistance is a crucial characteristic of metallic materials in porous burners. Extreme combustion conditions could lead to oxidation-affected erosion of porous media at a long-time period of burner operation. In this paper, we numerically simulated oxide scale growth at a porous radiant burner fabricated by Ni-Al intermetallic alloy using the combustion synthesis method, focusing on the structure degradation caused by periodic oxide scale spallation. A three-dimensional geometrical model of a porous intermetallic scaffold was obtained by scanning the porous burner using the X-ray CT technique. The surface erosion was modeled by the surface reconstruction based on calculated values of spalled oxide layer thickness. The simulation revealed that the submerged flame results in non-uniform distribution of the temperature at the solid surface. Such non-isothermal conditions lead to a two-times thicker oxide scale at the external surface of the burner. Thin struts of the intermetallic scaffold are prone to oxidation-affected erosion first, which forms discontinues and further fragmentation. The porous scaffold could lose about 50% of initial weight before fragmentation under conditions with intense oxide spallation. In such large structural degradation, the average flow velocity could reduce by a factor of 3, leading to changing of flame stabilization region.
1. Introduction
During the last decades, porous alloys have been considered an alternative to porous ceramic in solid oxide fuel cells, exhaust gas filters, flame arrestors, porous burners, etc., due to higher resistance to thermal shocks, large temperature gradients, and thermal cycling.
In particular, Ni-Al intermetallics are characterized by superior high-temperature properties, crucially crucial in porous energy conversion and combustion devices [1,2]. As an alternative to additive manufacturing of porous materials for energy conversion applications [3,4], there is a relatively simple and energy-effective method to fabricate open porous intermetallics, such as a combustion synthesis in a self-propagating mode in which the necessary heat input is self-released during the formation of intermetallic phases in the reacting mixture of alloy components [5,6]. This method appears to be very sensitive to the synthesis conditions, such as the chemical and particle-size compositions of starting powder mixture, the porosity of the reacting specimen, initial preheating, etc., that allows variation of structure and properties of fabricated porous alloys. Within the framework of combustion applications, the Ni-Al-based material is preferable due to its plasticity and sufficient strength of almost up to the melting-point temperature [1].
In contrast to common ceramics, any alloys are prone to high-temperature oxidation. It is well-known that Ni-Al intermetallics alloys follow a parabolic oxidation law and have excellent resistance towards oxidation below 1200 °C under isothermal conditions [7], thanks to the thermally grown -AlO oxide scale that exhibits better protection compared with any other oxides of alloys components because they are not able to create a gas-tight scale with high adherence to the alloy surface.
However, under thermal cycling conditions, the alloy surface is prone to intense oxidation due to the effect of oxide scale spallation, the mechanism of which is based on the stress and defects accumulation in the growing scale and a decrease in the adhesion strength at the scale/alloy interface [8,9]. Ni-Al alloys may lose 70% of the thermally grown oxide scale due to spallation during cyclic oxidation in dry air at 1150 °C that was demonstrated in [10]. Therefore, oxidation resistance during thermal cycling can be considered a factor restricting the development and application of metal-based porous materials in the energy utilization and conversion industry.
This study is devoted to the simulation of the degradation of porous Ni-Al intermetallic alloys due to high-temperature oxidation-affected erosion and its influence on flame stabilization in coarse-pored thin-layered cylindrical porous burners made by the combustion synthesis method. The approach is based on the heat and mass transfer problem simulation at the pore scale in combination with the pore surface reconstruction algorithm using the parabolic oxidation law. Presented data could be useful for lifetime engineering estimation of intermetallic porous materials under combustion-related conditions.
2. Materials and Methods
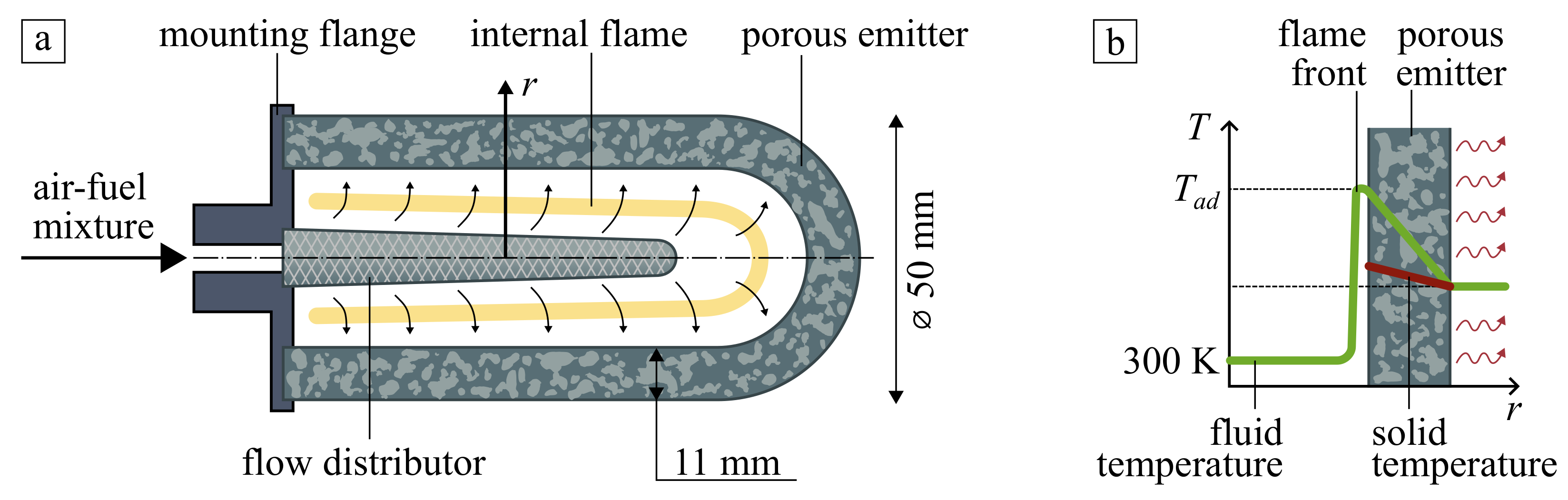
The considered radiant burner is schematically shown in Figure 1a. It consists of a porous emitter as a cylindrical shell with a semi-spherical head mounted to the flange. The fuel/air mixture passes through the conical flow distributor into the internal cavity of the emitter, where the flame is initiated and stabilized by flow rate control. Hot combustion products filtrate through the pores and heat the emitter. The heated burner emits infrared flux to the surrounding, which can be used for heating or thermal treatment of different products and materials. Typical simplified temperature profiles of gas and solid phases are shown in Figure 1b). A detailed description of the burner and other process parameters can be found in [11].
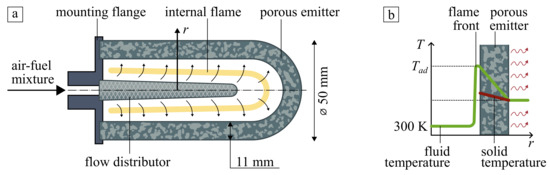
Figure 1.
(a) Schematic diagram of thin-layered cylindrical porous burner and (b) typical temperature profiles along with the radius of the burner.
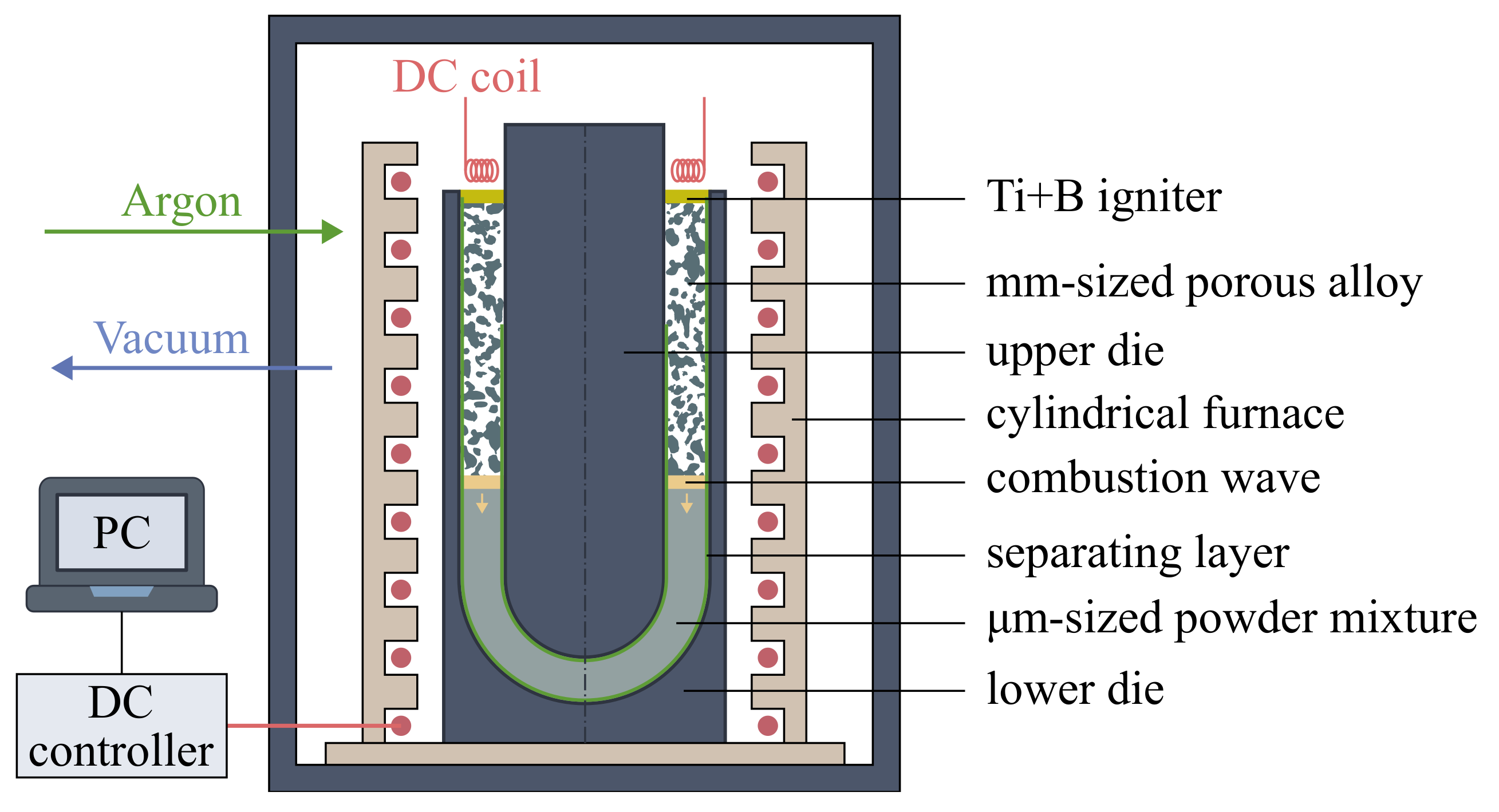
The porous burner was made by the combustion synthesis technology [12]. The scheme of the experimental setup used for the combustion synthesis is shown in Figure 2. The porous sample was synthesized in the argon atmosphere (1 bar) in a closed cylindrical reactor appended by an internal furnace with a DC controller. A stainless steel die consisted of the lower part in the form of a cup with a half-spherical floor and the upper part as a negative with a constant offset distance of 11 mm. A cellulose-based layer covered the inner surface of the template with a thickness of 200 µm. The layer separates the powder mixture from the steel wall of the die and makes it possible to control the combustion conditions near the boundary layer as described in [13]. The reaction mixture was filled in the die cavity and compacted by vibration to a porosity close to . The value of the porosity was calculated as one minus the relative density, which is the bulk density over the theoretical density :
where and are the mass and volume of the packed reaction mixture, respectively. The inverse rule of mixtures calculated the theoretical density:
where and are the mass fraction and the density of the i-th component of the reaction mixture, respectively. A highly exothermic Ti-B powder layer of 1-mm thickness was used to ignite the packed powder mixture. The chamber was evacuated to a residual pressure of 1 Pa followed by filling with high-purity argon to atmospheric pressure. Next, the die filled with the powder mixture was slowly and uniformly heated to a temperature range of 100 to 350 °C controlled using W-Re thermocouple located in the reaction mixture. A combustion synthesis in the preheated mixture was ignited by DC electric coil. After the process was completed and the setup cooled to the ambient temperature, the die with synthesized porous alloy was put off the sealed chamber. Porous alloys were purged by compressed air, washed in dilute hydrofluoric acid, flushed with pure water, dried, and annealed at 1200 °C for 3 h in the vacuum to achieve the equilibrium composition of the alloy.
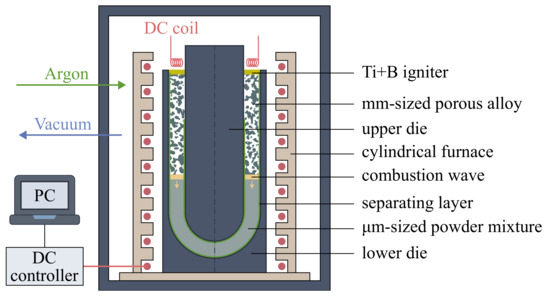
Figure 2.
Schematic diagram of the experimental setup used for combustion synthesis of alloys.
Synthesized emitter of the burner has a structure of highly permeable porous scaffold (resulting porosity of 0.55–0.60, the specific surface area below 10 m/g) comprised of tightly fused mm-sized spherical-like intermetallic struts forming a net of mm-sized interconnected pore channels. For capturing the geometrical features of the porous scaffold, we used X-ray Computed Tomography to reconstruct a sample of the porous emitter (see Figure 3a) approved for combustion applications [14], because there are no reliable algorithms for the virtual generation of porous structures made by combustion synthesis method in contrast with polymer or ceramic foams [15,16] due to large variability and structural irregularity. The CT analysis was performed using the YXLON Cheetah system (YXLON International GmbH, Hamburg, Germany). An accelerating voltage of 120 kV and a current of 400 µA were used to generate 1200 individual projections of the scaffold, with an exposure time of 0.3 s using a 0.2 mm thick Cu filter analogously with study in [17]. The image sequences were acquired at 32 µm resolution and further reconstructed in DragonFly 2021 computer software [18]. The resulting STL model is shown in Figure 3b in comparison with the initial sample. A wrapping procedure processed the resulting STL model to avoid local geometry defects such as small holes and overlapping surfaces following the recommendations in [19,20]. Due to the hardware limitations, only a 70°-sector of the whole cylindrical radiant burner was reconstructed and simulated to save computational cost. For numerical simulation, we used the region cut by side planes in the cylindrical part of the emitter and appended it to the upstream and downstream fluid regions. As a result, two (fluid and solid) volumetric zones were generated, as it is shown in Figure 3c.
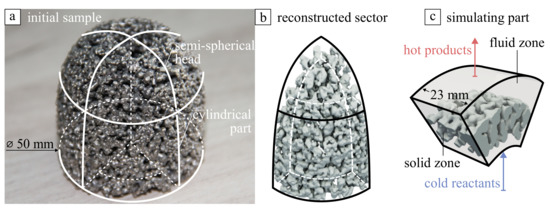
Figure 3.
(a) Photography of porous Ni-Al sample and (b) fragment reconstructed using X-ray CT (c) computational domain consisting of fluid and solid zones.
The pore-scale approach within the context of porous media combustion is based on the idea of simulating heat and mass transfer within a porous media with explicit consideration of its structure. In this study, we use the same problem statement as in [21], where a detailed description of the methodology, set of equations, boundary conditions, and other numerical setups in Ansys Fluent CFD software [22] are presented.
High-temperature oxidation is a slow process; therefore, it is aimless to simulate oxide scale growth with surface erosion due to spallation simultaneously with the combustion problem. In contrast with the timescale of oxidation, the characteristic time of thermal relaxation and flame stabilization is a fast process. It allows us to treat the problem as a decoupled one and implement a partitioned approach [23]: the equations governing the reacting flow within the porous media and the erosion of its surface are solved separately and sequentially. The main steps of the algorithm are as follows.
- Step 1
- Pore-scale simulation of combustion problem in the porous emitter.
- Step 2
- Extraction of the temperature field at the fluid-solid interface.
- Step 3
- Calculating the local oxide scale thickness increment based on the extracted temperature on each cell face by integrating the oxidation law using the 3-rd order explicit Runge-Kutta method over oxidation timestep (∼ 0.1 h), implemented as a subroutine in Fluent using UDF toolkit.
- Step 4
- Translation of the cell faces at the interface along with the direction opposite to the surface normal (reversed or negative extrusion in 3D modeling).
- Step 5
- Volumetric zones remeshing based on a pre-defined size function.
- Step 6
- Interpolation of the process parameters fields (temperature, species mass fractions, velocity components, and pressure) to the reconstructed geometry as initial conditions for simulation in the next cycle.
Calculating the scale thickness increment (Step 3) was performed based on a parabolic law of oxidation kinetic in the following form.
where is the parabolic oxidation rate constant as a function of the temperature. It is well established that the oxidation rate constant is a monotonically increasing function of the temperature. In this paper, the dependence was characterized by an approximation of the experimental data by Brumm and Grabke [24] obtained for AlO oxide scale isothermally grown over Ni-Al and Ni-Al-Cr alloys:
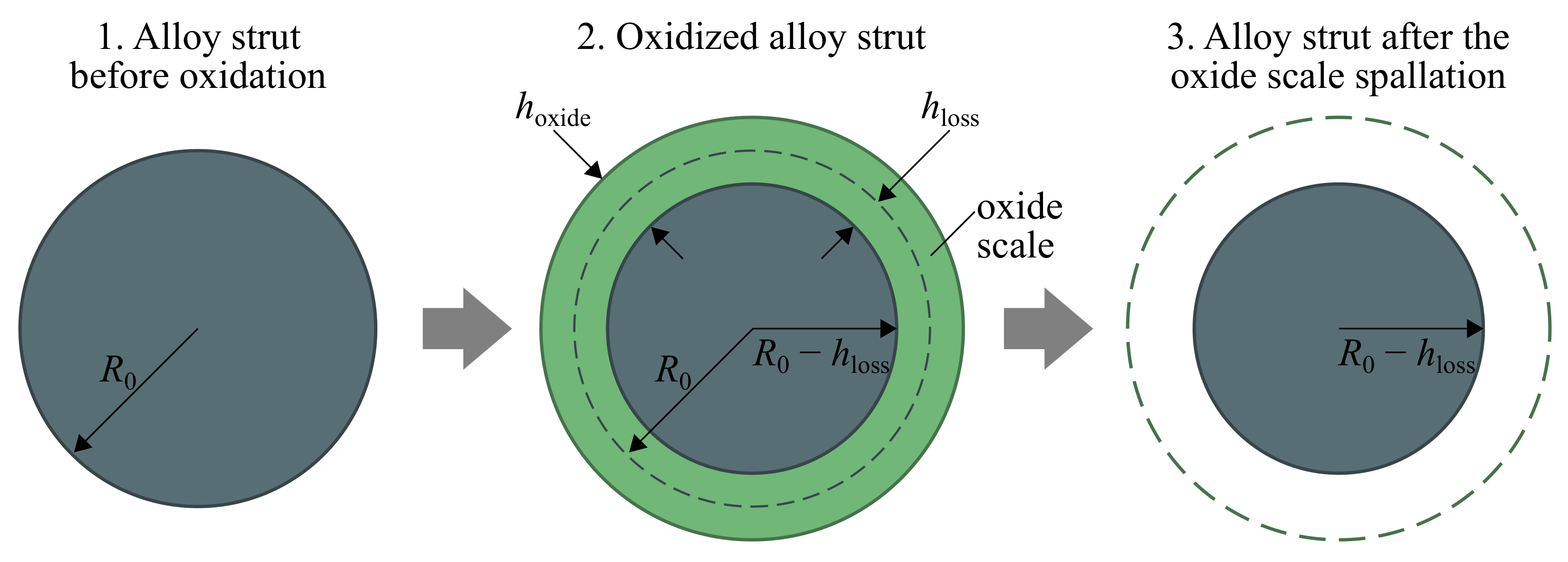
As the oxidation process is accompanied by capturing oxygen from the surrounding, the thickness of the growth oxide scale is larger than the thickness of the consumed metal layer , as illustrated in Figure 4. The resulting thickness of lost material was calculated by integration of the oxidation law at considered time interval:
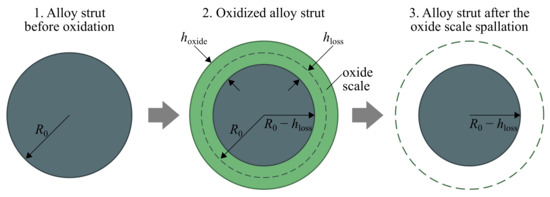
Figure 4.
Scheme of oxide scale formation with definition.
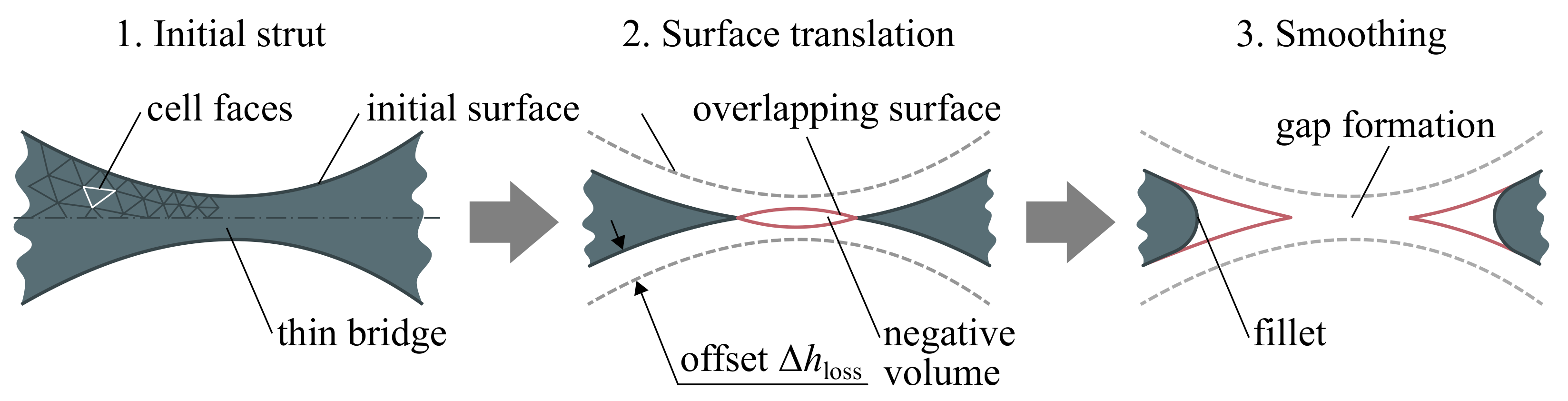
Spherical-like intermetallic struts of the porous scaffold are tightly fused by some sort of bridges that are eroded first, which can lead to gaps or discontinues formation. In the proposed algorithm (Step 4), this problem is treated by removing the overlapping surfaces nearby the bridges in a manner shown in Figure 5 that allows simulation of discontinues strut formation in the scaffold during oxidation-affected erosion.
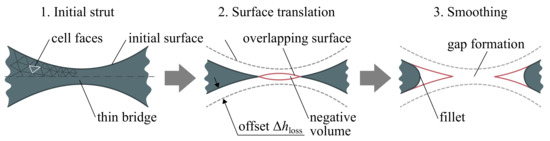
Figure 5.
Illustration of the surface translation algorithm nearby the thin bridge.
3. Results and Discussion
A primary goal in studying the applicability of porous alloys to combustion devices is to predict their service lifetime under oxidation conditions. The following expression can define the oxidation-limited lifetime of various porous and thin-walled alloy parts.
Here V and S are the volume and the surface area of the struts that constitute the permeable alloy part, respectively. The coefficient is the ratio of the molar masses of oxygen to the metals in the corresponding oxide scale ( for AlO), is the available reservoir of aluminum that provides selective growth of AlO oxide scale, is the alloy density, and is the dimensionless parameter empirically defining the rate of oxide scale spallation. There are three factors that affect the oxidation-limited lifetime:
- Dimension and shape factor (term I of Equation (6)). The lifetime is proportional to the square of the characteristic size of the alloy elements, so the bigger, the better. The characteristic size is defined as ; therefore, the shape of alloy elements is an important factor. The lifetime for a thin metal sheet with a thickness of d is twice higher than for a round wire with a diameter of d and three times higher than for a sphere with the same diameter. Hence, gas-permeable parts made of thin metal sheets, stacked wire, or expanded metal meshes are much more robust against oxidation than porous materials made by powder technologies. Porous materials made by the combustion synthesis method are characterized by developed surfaces with the complicated, irregular shape of struts that can be considered as a negative factor for oxidation resistance due to locally corrugated and curved scale shapes.
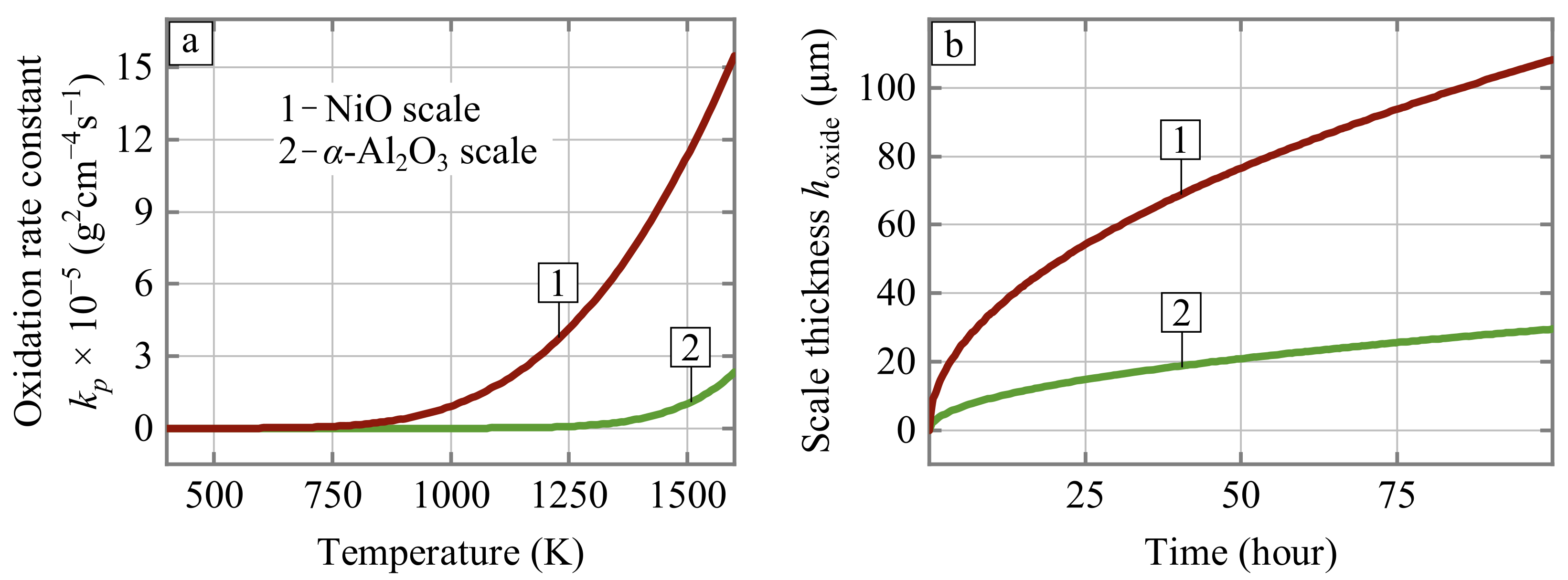
- Kinetic factor (term II of Equation (6)). The oxidation-limited lifetime of porous Ni-Al parts is directly proportional to the volume of the available aluminum reservoir and inversely proportional to the oxidation rate constant. It is widely accepted that the oxidation kinetic (i.e., the temperature dependence for oxidation rate constant k) remains unchanged as long as a selectively oxidized metal maintains the growth of the protective oxide scale. However, the oxidation of the base alloy component (nickel) accelerates not when the aluminum is fully consumed but much earlier. The aluminum can be selectively consumed during oxidation until its decreasing concentration in the intermetallic alloy remains above a threshold value , which is about 6.9 wt.% [25]. Therefore, the available reservoir of aluminum that provides selective growth of the AlO oxide scale can be defined as , where is the initial concentration of aluminum in the alloy. The moment when the aluminum concentration approaches can be considered the beginning of catastrophic breakaway oxidation of the alloy because the parabolic oxidation rate constant for nickel is an order of magnitude higher than those of Al [26]. The dependence for NiO scale growth can be described by:Figure 6 compares the oxidation rate constants defined by Equations (4) and (7) and the resulting scale thickness under constant temperature equal to 1300 K. Both these cases can be considered as limiting ones: the best (AlO) and worst (NiO) oxidation resistance of Ni-Al intermetallics. This comparison demonstrates how significantly the initial concentration of aluminum can influence oxidation-limited lifetime. Overall, the higher the initial concentration of aluminum in the alloy, the better.
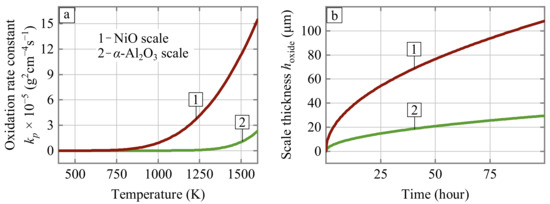 Figure 6. (a) Oxidation rate constants dependency on the temperature and (b) the scale thickness evolution for two (NiO, AlO) cases of oxide formation under 1300 K.
Figure 6. (a) Oxidation rate constants dependency on the temperature and (b) the scale thickness evolution for two (NiO, AlO) cases of oxide formation under 1300 K. - Spallation factor (term III of Equation (6)). The growth rate of the protective scale increases if the scale is prone to spallation. This increase corresponds to projecting back to the initial stages of the scale growth, where the slope of the dependence is higher (Figure 6b). The spallation resistance can be taken into account in a phenomenological manner by using dimensionless parameter ranging from 0 to 1, where means no spallation. The parameter is a function not only of the alloy chemical composition (the spallation resistance can be improved by minor doping of the Ni-Al alloy with REE or chromium) but also of the algorithms of burner operation. Let’s consider some heating boilers switching off once a day, designed using Ni-Al radiant burners. We can estimate that 70% of initial mass loss (this value close to the critical one in which physical fragmentation of the sample due to erosion of connecting struts occurs) will be achieved after ≈120 days of the boiler operation, assuming that the weight fraction of spalled AlO scale is 0.25. This period is only half of the heating season in regions with a continental climate. Obviously, other thermal cycling scenarios, oxidation constants, oxide spallation fraction, etc. would lead to another lifetime. However, such a rough estimation shows the importance of improvement of the intrinsic resistance of the alloy to spallation along with the development of the burner start and shut-down algorithms with slower heating and cooling of the porous emitter.
The surface erosion effect is illustrated in Figure 7 by a sequence of scaffold structures in two view projections with different eroded thicknesses obtained by the algorithm in Figure 5. The mass loss between the initial case and the sample with the lost thickness µm is about 60%. Initial porosity of 0.47 was reduced to 0.79. Further increasing of h results in the fragmentation of the sample into several parts due to gap formation in place of thin bridges.

Figure 7.
Porous matrix evolution during high-temperature oxidation-affected erosion. The cases are labeled by total average thickness of lost layer thickness .
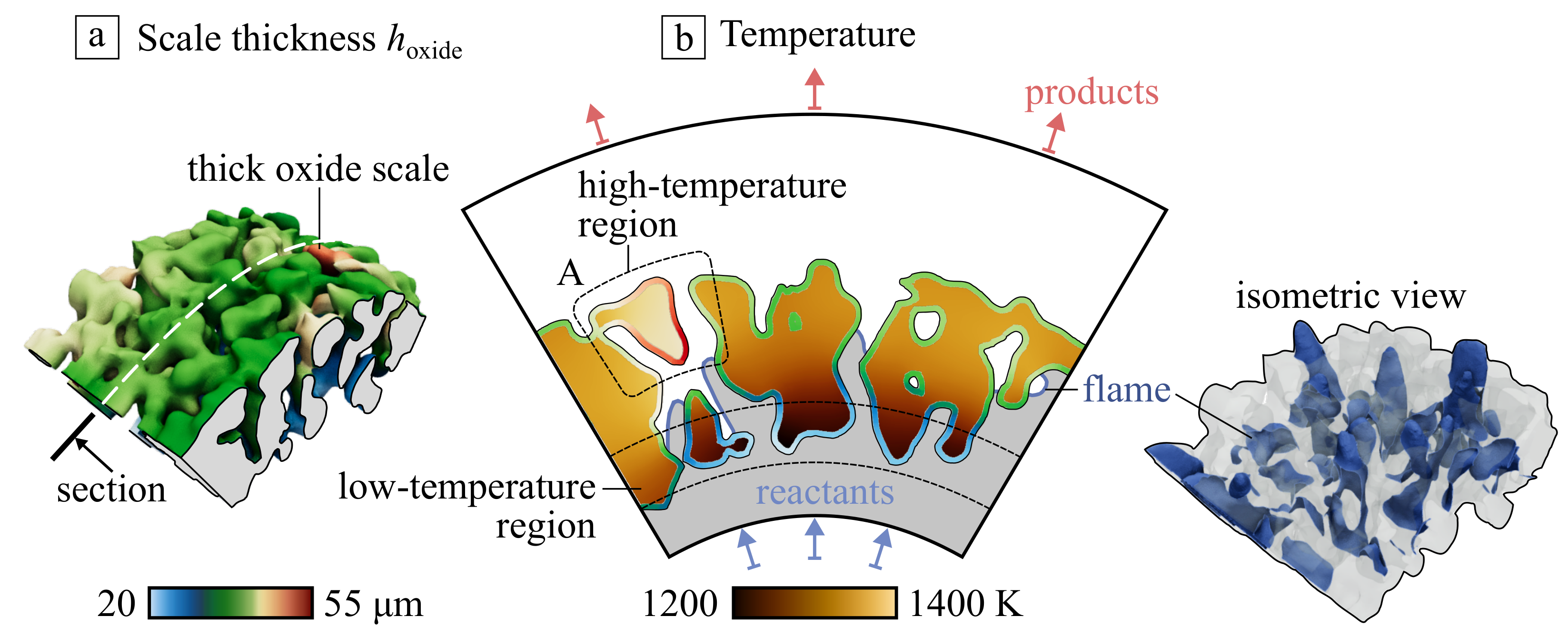
Figure 8a shows the scaffold with submerged flame. It can be noticed that the front is significantly distorted and stretched by the irregular flow passing through the media. A characteristic feature of this regime is intense heat recuperation from combustion products to the fresh mixture via the conductivity of the scaffold. It results in some temperature gradient: despite on large thermal conductivity of metal, the incoming cold flow cools the internal surface of the porous emitter. The resulting oxide scale thickness formed in these conditions is shown in Figure 8b in the central section of the model. The scale thickness is significantly non-uniform. The maximum difference in temperature of the solid phase is about 200 K; however, the scale thickness differs by more than two times due to the exponential character of dependency (see Equation (4)).
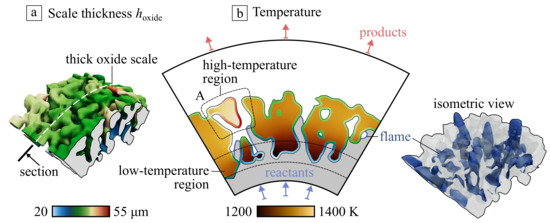
Figure 8.
(a) AlO scale thickness at the Ni-Al porous material in the case of submerged flame formed during 100 h and (b) the temperature distribution in central section appended by the scale thickness .
Important to mention the formation of the overheated scaffold fragments (see region A in Figure 8b). Such parts are connected with the entire material only by a thin bridge that could be considered a local resistance for conductive heat transfer. It leads to the separation of such elements first due to larger oxidation rate and low connectivity with other structural elements.
As expected, such extensive erosion of material influences the flame front structure significantly. As discussed in [21], the local flow field highly affects the flame. Fresh mixture carries away the flame front anchored nearby the internal emitter surface deep into the porous media. It results in highly stretched bell-shaped flamelets. The large and irregular flow velocity, the stretched flame front. During long-time oxidation with spallation, the eroded porous media’s porosity increases, leading to lower local velocity gradients and macroscopic flame thickness, as shown in Figure 9 for different thicknesses of the spalled oxide layers. The area of the flame sheet reduces by a factor of two, comparing the initial structure and the eroded one with µm. As discussed by Ellzey et al. [27], one of the significant practical aspects of porous burners relates to the problem of flame stabilization in a wide range of flow rates and mixture compositions. In the case of metallic porous media, the oxidation-affected erosion can result in undesirable flame flashback due to an increase in the porosity and decreasing in interstitial flow velocity as a result. This aspect is potentially crucial for two-layer or surface-stabilized burners based on metallic porous materials [28].

Figure 9.
Flame front localization within porous media under the different thicknesses of eroded material.
4. Conclusions
In this paper, we simulated oxide scale growth at porous Ni-Al intermetallic alloy made by the combustion synthesis method focusing on surface erosion due to scale spallation. A three-dimensional model was obtained by X-ray CT of a material sample. Oxidation-affected erosion was simulated by the surface reconstruction based on calculated values of scale thickness followed by the experimentally obtained oxidation constant as a function of the temperature.
- In a coarse-pored thin-layered porous burner, the temperature distribution over the surface is non-uniform due to flame anchoring and stretching effects. In this case, different thermal conditions of oxidation lead to different rates of oxide scale growth. The exponential character of dependency results in two times thicker oxide scale at the external surface of the emitter due to the temperature gradient when the average solid temperature of the emitter is about 1300 K.
- Porous materials fabricated by the combustion synthesis method could be represented as connected rounded strut elements connected by thin bridges, which are prone to oxidation-affected erosion first. The average diameter of the bridges should be taken into account for estimation of the oxidation resistance in the case of compositions and conditions under which the spallation phenomenon takes place because the formation of gaps or discontinues in the scaffold at long-time intervals leads to the scaffold fragmentation even if the total mass loss is far from a critical value.
- In the case of strong thermal and hydrodynamic coupling between the flame and porous media, shape degradation due to oxidation-affected erosion can significantly affect the flame front area. The simulation showed that the flame could change its anchoring location and is characterized by a lower stretching factor due to variation in the local flow field and average filtration velocity. From a practical point of view, it means that after a long period of high-temperature oxidation with spallation, the combustion regime could change, or even a flashback can occur. It should be considered when designing thin-layered porous burners with internal/submerged combustion regimes.
Author Contributions
Conceptualization, I.Y. and A.M.; methodology, I.Y.; formal analysis, N.P.; investigation, D.A. and S.Z.; data curation, N.P.; writing—original draft preparation, I.Y.; writing—review and editing, A.M.; visualization, S.Z.; project administration, A.M. All authors have read and agreed to the published version of the manuscript.
Funding
The research was funded by Russian Science Foundation (project No. 21-79-10445).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
The following abbreviations are used in this manuscript:
| CT | Computer tomography |
| REE | Rare-earth elements |
| PSS | Pore-scale simulation |
| STL | Stereolithography |
References
- Hemker, K.J.; Mills, M.J.; Nix, W.D. An Investigation of the Mechanisms That Control Intermediate Temperature Creep of Ni3Al. Acta Metall. Mater. 1991, 39, 1901–1913. [Google Scholar] [CrossRef]
- Solovyev, A.A.; Rabotkin, S.V.; Shipilova, A.V.; Kirdyashkin, A.I.; Ionov, I.V.; Kovalchuk, A.N.; Maznoy, A.S.; Kitler, V.D.; Borduleva, A.O. Solid Oxide Fuel Cell with Ni–Al Support. Int. J. Hydrog. Energy 2015, 40, 14077–14084. [Google Scholar] [CrossRef]
- Lervåg, M.; Sørensen, C.; Robertstad, A.; Brønstad, B.; Nyhus, B.; Eriksson, M.; Aune, R.; Ren, X.; Akselsen, O.; Bunaziv, I. Additive Manufacturing with Superduplex Stainless Steel Wire by Cmt Process. Metals 2020, 10, 272. [Google Scholar] [CrossRef]
- Jabbareh, M.; Assadi, H. Modelling of Microstructure Evolution during Laser Processing of Intermetallic Containing Ni-al Alloys. Metals 2021, 11, 1051. [Google Scholar] [CrossRef]
- Yeh, C.L.; Sung, W.Y. Combustion Synthesis of Ni3Al Intermetallic Compound in Self-Propagating Mode. J. Alloys Compd. 2004, 384, 181–191. [Google Scholar] [CrossRef]
- Yeh, C.L.; Ke, C.Y. Intermetallic/Ceramic Composites Synthesized from al–Ni–Ti Combustion with B4 c Addition. Metals 2020, 10, 873. [Google Scholar] [CrossRef]
- Pan, Y.; Chuang, T.; Yao, Y. Long-Term Oxidation Behaviour of Ni3Al Alloys with and without Chromium Additions. J. Mater. Sci. 1991, 26, 6097–6103. [Google Scholar] [CrossRef]
- Pérez, P.; González-Carrasco, J.; Adeva, P. Oxidation Behavior of a Ni 3 Al PM Alloy. Oxid. Met. 1997, 48, 143–170. [Google Scholar] [CrossRef]
- Fang, X.; Li, Y.; Yue, M.; Feng, X. Chemo-Mechanical Coupling Effect on High Temperature Oxidation: A Review. Sci. China Technol. Sci. 2019, 62, 1297–1321. [Google Scholar] [CrossRef]
- Maznoy, A.; Pichugin, N.; Kirdyashkin, A.; Yakovlev, E.; Yakovlev, I.; Zambalov, S.; Guschin, A. Predicting Oxidation-limited Lifetime of Ni-Al-Cr Porous Radiant Burners Made by Combustion Synthesis. J. Alloys Compd. 2023, 934, 167885. [Google Scholar] [CrossRef]
- Maznoy, A.; Yakovlev, I.; Pichugin, N.; Zambalov, S.; Tcoi, K. Performance Characteristics of a Thin-Layered Radial Porous Burner with Different Strategies for Fuel-Air Mixture Supply. Case Stud. Therm. Eng. 2022, 38. [Google Scholar] [CrossRef]
- Maznoy, A.; Kirdyashkin, A.; Kitler, V.; Pichugin, N.; Salamatov, V.; Tcoi, K. Self-Propagating High-Temperature Synthesis of Macroporous B2+L12 Ni-Al Intermetallics Used in Cylindrical Radiant Burners. J. Alloys Compd. 2019, 792, 561–573. [Google Scholar] [CrossRef]
- Pichugin, N.; Kirdyashkin, A.; Kitler, V.; Maznoy, A. The Effect of Separating Layer between Reacting Media and Molding Template on the Porous Structure of Combustion Synthesized Ni-Al Intermetallics. Mater. Lett. 2022, 314, 131854. [Google Scholar] [CrossRef]
- Maznoy, A.; Pichugin, N.; Yakovlev, I.; Fursenko, R.; Petrov, D.; Shy, S.S. Fuel Interchangeability for Lean Premixed Combustion in Cylindrical Radiant Burner Operated in the Internal Combustion Mode. Appl. Therm. Eng. 2021, 186, 115997. [Google Scholar] [CrossRef]
- Lautensack, C.; Giertzsch, M.; Godehardt, M.; Schladitz, K. Modelling a Ceramic Foam Using Locally Adaptable Morphology. J. Microsc. 2008, 230, 396–404. [Google Scholar] [CrossRef]
- Liebscher, A.; Redenbach, C. 3D Image Analysis and Stochastic Modelling of Open Foams. Int. J. Mater. Res. 2012, 103, 155–161. [Google Scholar] [CrossRef]
- Surmeneva, M.A.; Khrapov, D.; Prosolov, K.; Kozadayeva, M.; Koptyug, A.; Volkova, A.; Paveleva, A.; Surmenev, R.A. The Influence of Chemical Etching on Porous Structure and Mechanical Properties of the Ti6AL4V Functionally Graded Porous Scaffolds Fabricated by EBM. Mater. Chem. Phys. 2022, 275, 125217. [Google Scholar] [CrossRef]
- Dragonfly 2021.3 [Computer Software]. Object Research Systems; ORS Inc.: Montreal, QC, Canada. 2020. Available online: www.theobjects.com/dragonfly (accessed on 27 December 2022).
- Yakovlev, I.; Zambalov, S. Three-Dimensional Pore-Scale Numerical Simulation of Methane-Air Combustion in Inert Porous Media under the Conditions of Upstream and Downstream Combustion Wave Propagation through the Media. Combust. Flame 2019, 209, 74–98. [Google Scholar] [CrossRef]
- Partopour, B.; Dixon, A.G. An Integrated Workflow for Resolved-Particle Packed Bed Models with Complex Particle Shapes. Powder Technol. 2017, 322, 258–272. [Google Scholar] [CrossRef]
- Yakovlev, I.; Maznoy, A.; Zambalov, S. Pore-Scale Study of Complex Flame Stabilization Phenomena in Thin-Layered Radial Porous Burner. Combust. Flame 2021, 231, 111468. [Google Scholar] [CrossRef]
- ANSYS Fluent Theory Guide, Release 17.0; ANSYS Inc.: Canonsburg, PA, USA, 2016.
- Sigrist, J.F. Fluid-Structure Interaction: An Introduction to Finite Element Coupling; Wiley: Chichester, West Sussex, UK, 2015. [Google Scholar]
- Brumm, M.W.; Grabke, H.J. The Oxidation Behaviour of NiAl-I. Phase Transformations in the Alumina Scale during Oxidation of NiAl and NiAl-Cr Alloys. Corros. Sci. 1992, 33, 1677–1690. [Google Scholar] [CrossRef]
- Speck, P.; Young, D.J.; Zhang, J. Metal Dusting of Nickel–Aluminium Alloys. Oxid. Met. 2010, 73, 255–274. [Google Scholar] [CrossRef]
- Kuenzly, J.D.; Douglass, D.L. The Oxidation Mechanism of Ni3Al Containing Yttrium. Oxid. Met. 1974, 8, 139–178. [Google Scholar] [CrossRef]
- Ellzey, J.L.; Belmont, E.L.; Smith, C.H. Heat Recirculating Reactors: Fundamental Research and Applications. Prog. Energy Combust. Sci. 2019, 72, 32–58. [Google Scholar] [CrossRef]
- McDougald, N. Development and Demonstration of an Ultra Low NOx Combustor for Gas Turbines; Technical Report; Alzeta Corporation: San Jose, CA, USA, 2005. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).