The Effect of Vitamin D Supplementation on Skeletal Muscle in the mdx Mouse Model of Duchenne Muscular Dystrophy
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals and Ethics Approval
2.2. Contractility
2.3. Fatigue and Recovery
2.4. Histological Analysis
2.5. Statistical Analysis
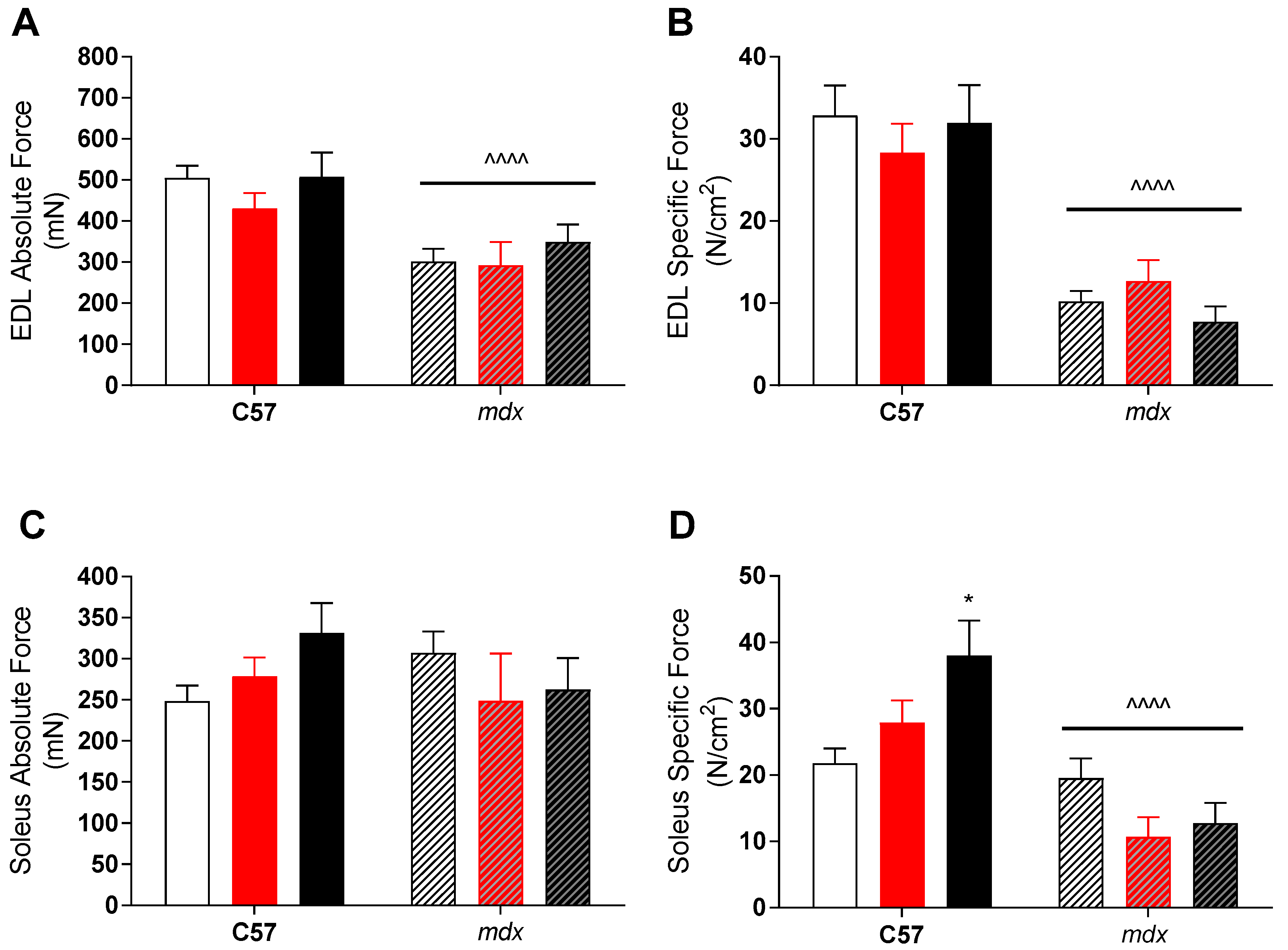
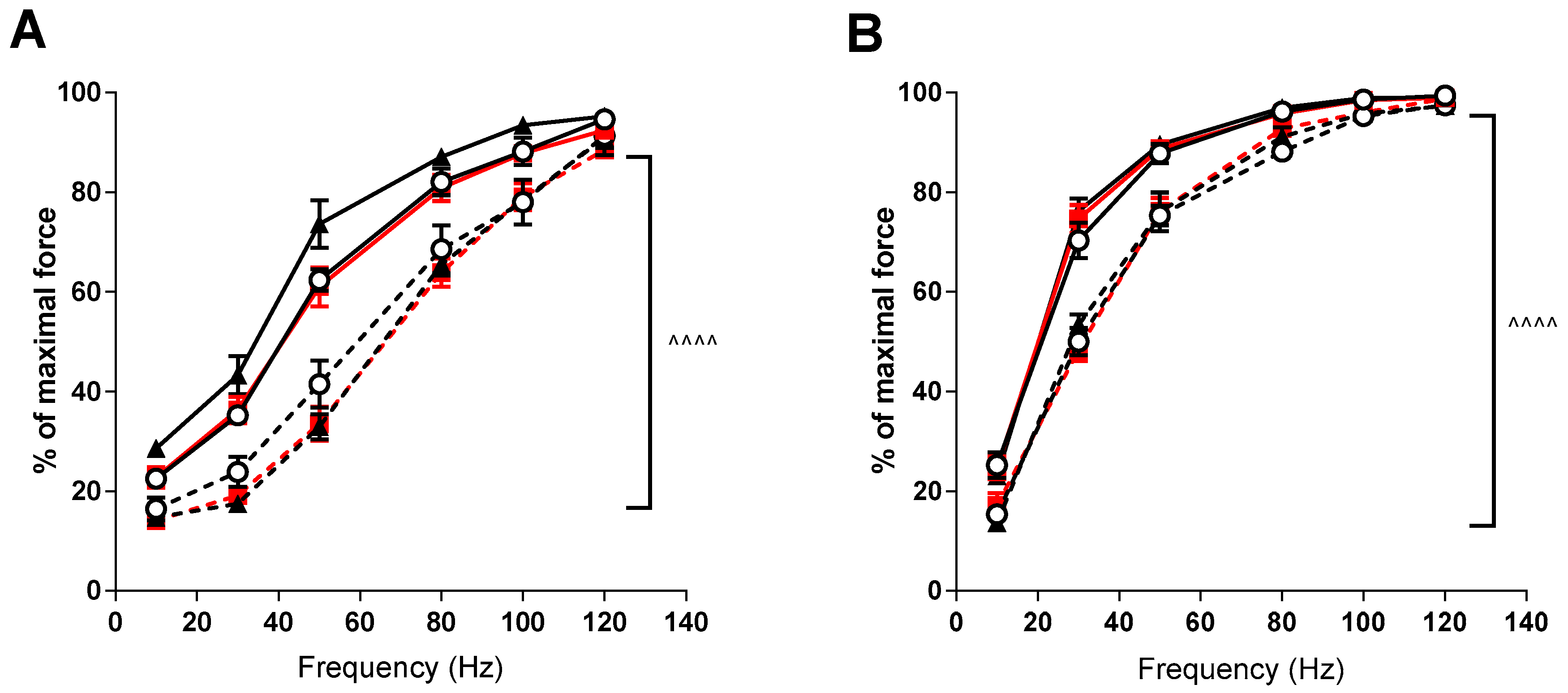
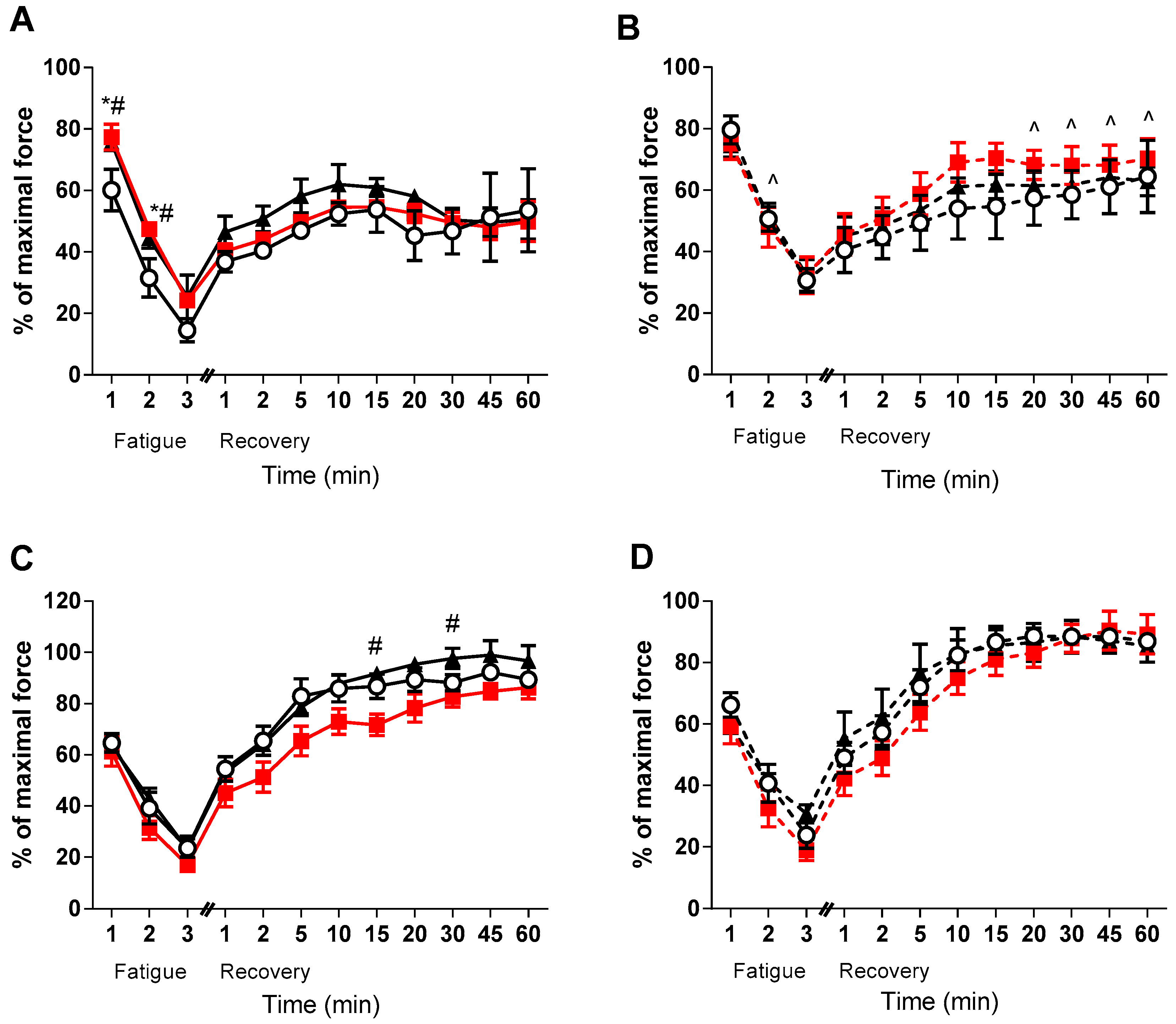
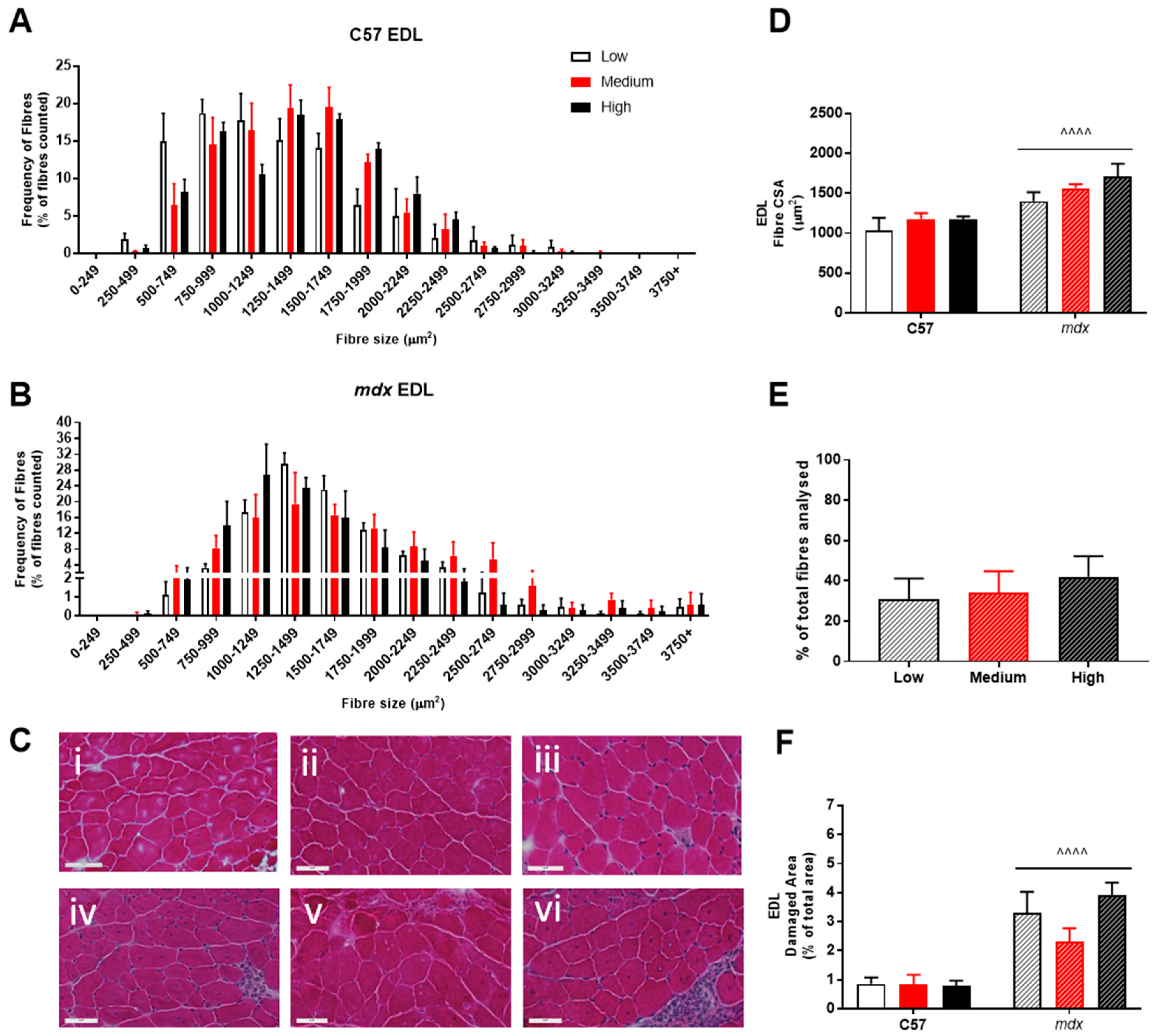
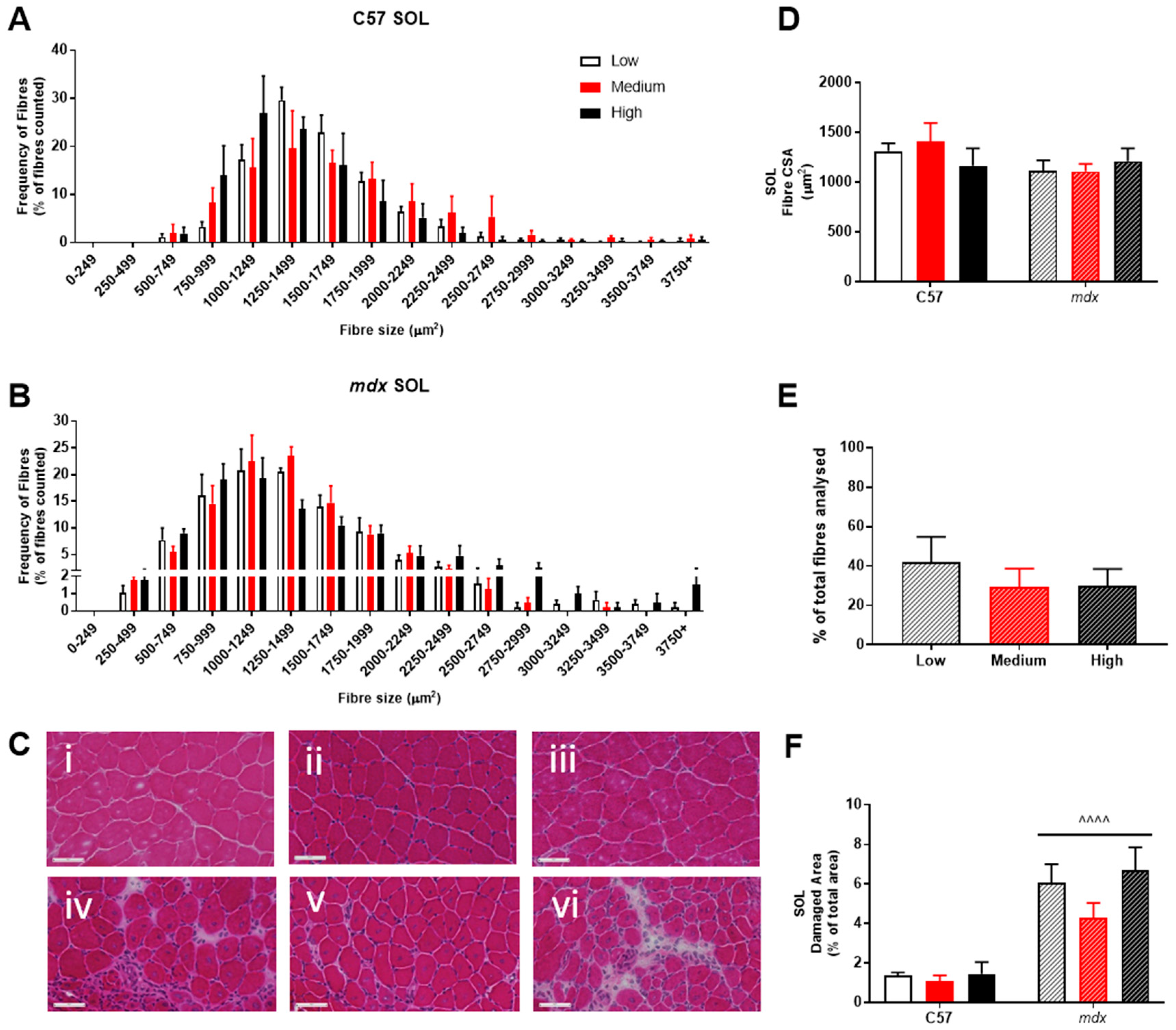
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Perez-Lopez, F.R. Vitamin D: The secosteroid hormone and human reproduction. Gynecol. Endocrinol. 2007, 23, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Parker-Autry, C.Y.; Burgio, K.L.; Richter, H.E. Vitamin D status: A review with implications for the pelvic floor. Int. Urogynecology J. 2012, 23, 1517–1526. [Google Scholar] [CrossRef]
- Christakos, S.; Dhawan, P.; Verstuyf, A.; Verlinden, L.; Carmeliet, G. Vitamin D: Metabolism, Molecular Mechanism of Action, and Pleiotropic Effects. Physiol. Rev. 2015, 96, 365–408. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, B. Vitamin D and human skeletal muscle. Scand. J. Med. & Sci. sports 2010, 20, 182–190. [Google Scholar] [CrossRef]
- Stratos, I.; Li, Z.; Herlyn, P.; Rotter, R.; Behrendt, A.-K.; Mittlmeier, T.; Vollmar, B. Vitamin D Increases Cellular Turnover and Functionally Restores the Skeletal Muscle after Crush Injury in Rats. Am. J. Pathol. 2013, 182, 895–904. [Google Scholar] [CrossRef]
- Owens, D.J.; Sharples, A.P.; Polydorou, I.; Alwan, N.; Donovan, T.; Tang, J.; Fraser, W.D.; Cooper, R.G.; Morton, J.P.; Stewart, C.; et al. A systems-based investigation into vitamin D and skeletal muscle repair, regeneration, and hypertrophy. Am. J. Physiol. Endocrinol. Metab. 2015, 309, E1019–E1031. [Google Scholar] [CrossRef] [PubMed]
- Girgis, C.M.; Cha, K.M.; Houweling, P.J.; Rao, R.; Mokbel, N.; Lin, M.; Clifton-Bligh, R.J.; Gunton, J.E. Vitamin D Receptor Ablation and Vitamin D Deficiency Result in Reduced Grip Strength, Altered Muscle Fibers, and Increased Myostatin in Mice. Calcif. Tissue Int. 2015, 97, 602–610. [Google Scholar] [CrossRef]
- Drittanti, L.; de Boland, A.R.; Boland, R. Stimulation of calmodulin synthesis in proliferating myoblasts by 1,25-dihydroxy-vitamin D3. Mol. Cell. Endocrinol. 1990, 74, 143–153. [Google Scholar] [CrossRef]
- Santillán, G.; Katz, S.; Vazquez, G.; Boland, R.L. TRPC3-like protein and Vitamin D receptor mediate 1α,25(OH)2D3-induced SOC influx in muscle cells. Int. J. Biochem. & Cell Biol. 2004, 36, 1910–1918. [Google Scholar] [CrossRef]
- Fairclough, R.J.; Bareja, A.; Davies, K.E. Progress in therapy for Duchenne muscular dystrophy. Exp. Physiol. 2011, 96, 1101–1113. [Google Scholar] [CrossRef]
- Bushby, K.M. Genetic and clinical correlations of Xp21 muscular dystrophy. J. Inherit. Metab. Dis. 1992, 15, 551–564. [Google Scholar] [CrossRef]
- Bianchi, M.L.; Morandi, L.; Andreucci, E.; Vai, S.; Frasunkiewicz, J.; Cottafava, R. Low bone density and bone metabolism alterations in Duchenne muscular dystrophy: response to calcium and vitamin D treatment. Osteoporos. Int. 2011, 22, 529–539. [Google Scholar] [CrossRef] [PubMed]
- Bellayou, H.; Hamzi, K.; Rafai, M.A.; Karkouri, M.; Slassi, I.; Azeddoug, H.; Nadifi, S. Duchenne and Becker Muscular Dystrophy: Contribution of a Molecular and Immunohistochemical Analysis in Diagnosis in Morocco. J. Biomed. Biotechnol. 2009, 2009, 5. [Google Scholar] [CrossRef] [PubMed]
- Yilmaz, O.; Karaduman, A.; Topaloglu, H. Prednisolone therapy in Duchenne muscular dystrophy prolongs ambulation and prevents scoliosis. Eur. J. Neurol. 2004, 11, 541–544. [Google Scholar] [CrossRef]
- Lynch, G.S.; Hinkle, R.T.; Chamberlain, J.S.; Brooks, S.V.; Faulkner, J.A. Force and power output of fast and slow skeletal muscles from mdx mice 6–28 months old. J. Physiol. 2001, 535, 591–600. [Google Scholar] [CrossRef] [PubMed]
- Sato, Y.; Iwamoto, J.; Kanoko, T.; Satoh, K. Low-dose vitamin D prevents muscular atrophy and reduces falls and hip fractures in women after stroke: a randomized controlled trial. Cerebrovasc. Dis. 2005, 20, 187–192. [Google Scholar] [CrossRef] [PubMed]
- Zhu, K.; Austin, N.; Devine, A.; Bruce, D.; Prince, R.L. A randomized controlled trial of the effects of vitamin D on muscle strength and mobility in older women with vitamin D insufficiency. J. Am. Geriatr. Soc. 2010, 58, 2063–2068. [Google Scholar] [CrossRef]
- Agostini, D.; Zeppa Donati, S.; Lucertini, F.; Annibalini, G.; Gervasi, M.; Ferri Marini, C.; Piccoli, G.; Stocchi, V.; Barbieri, E.; Sestili, P. Muscle and Bone Health in Postmenopausal Women: Role of Protein and Vitamin D Supplementation Combined with Exercise Training. Nutrients 2018, 10. [Google Scholar] [CrossRef]
- Van Veldhuizen, P.J.; Taylor, S.A.; Williamson, S.; Drees, B.M. Treatment of Vitamin D Deficiency in Patients with Metastatic Prostate Cancer May Improve Bone Pain and Muscle Strength. J. Urology 2000, 163, 187–190. [Google Scholar] [CrossRef]
- Dangain, J.; Vrbova, G. Muscle development in mdx mutant mice. Muscle & Nerve 1984, 7, 700–704. [Google Scholar] [CrossRef]
- Spurney, C.F.; Gordish-Dressman, H.; Guerron, A.D.; Sali, A.; Pandey, G.S.; Rawat, R.; Van Der Meulen, J.H.; Cha, H.J.; Pistilli, E.E.; Partridge, T.A.; et al. Preclinical drug trials in the mdx mouse: Assessment of reliable and sensitive outcome measures. Muscle & Nerve 2009, 39, 591–602. [Google Scholar] [CrossRef]
- Bulfield, G.; Siller, W.G.; Wight, P.A.; Moore, K.J. X chromosome-linked muscular dystrophy (mdx) in the mouse. Proc. Nat. Academy Sci. USA 1984, 81, 1189–1192. [Google Scholar] [CrossRef]
- Allen, D.G.; Gervasio, O.L.; Yeung, E.W.; Whitehead, N.P. Calcium and the damage pathways in muscular dystrophy. Can. J. Physiol. Pharmacol. 2010, 88, 83–91. [Google Scholar] [CrossRef] [PubMed]
- Pertille, A.; de Carvalho, C.L.T.; Matsumura, C.Y.; Neto, H.S.; Marques, M.J. Calcium-binding proteins in skeletal muscles of the mdx mice: potential role in the pathogenesis of Duchenne muscular dystrophy. Int. J. Exp. Pathol. 2010, 91, 63–71. [Google Scholar] [CrossRef]
- Hibaoui, Y.; Reutenauer-Patte, J.; Patthey-Vuadens, O.; Ruegg, U.T.; Dorchies, O.M. Melatonin improves muscle function of the dystrophic mdx5Cv mouse, a model for Duchenne muscular dystrophy. J. Pineal Res. 2011, 51, 163–171. [Google Scholar] [CrossRef]
- Harisseh, R.; Chatelier, A.; Magaud, C.; Déliot, N.; Constantin, B. Involvement of TRPV2 and SOCE in calcium influx disorder in DMD primary human myotubes with a specific contribution of α1-syntrophin and PLC/PKC in SOCE regulation. Am. J. Physiol. Cell Physiol. 2013, 304, C881–C894. [Google Scholar] [CrossRef]
- Radley-Crabb, H.G.; Marini, J.C.; Sosa, H.A.; Castillo, L.I.; Grounds, M.D.; Fiorotto, M.L. Dystropathology increases energy expenditure and protein turnover in the mdx mouse model of duchenne muscular dystrophy. PloS One 2014, 9, e89277. [Google Scholar] [CrossRef] [PubMed]
- Fontana, S.; Schillaci, O.; Frinchi, M.; Giallombardo, M.; Morici, G.; Di Liberto, V.; Alessandro, R.; De Leo, G.; Perciavalle, V.; Belluardo, N.; et al. Reduction in mdx mouse muscle degeneration by low-intensity endurance exercise: A proteomic analysis in quadriceps muscle of exercised compared with sedentary mdx mice. Biosci. Rep. 2015, 35, e00213. [Google Scholar] [CrossRef]
- Chan, S.; Head, S.I. The role of branched fibres in the pathogenesis of Duchenne muscular dystrophy. Exp. Physiol. 2011, 96, 564–571. [Google Scholar] [CrossRef] [PubMed]
- Brooks, S.V.; Faulkner, J.A. Contractile properties of skeletal muscles from young, adult and aged mice. J. Physiol. 1988, 404, 71–82. [Google Scholar] [CrossRef]
- Close, R.I. Dynamic properties of mammalian skeletal muscles. Physiol. Rev. 1972, 52, 129–197. [Google Scholar] [CrossRef]
- Petrof, B.J.; Shrager, J.B.; Stedman, H.H.; Kelly, A.M.; Sweeney, H.L. Dystrophin protects the sarcolemma from stresses developed during muscle contraction. P Proc. Nat. Academy Sci. USA 1993, 90, 3710–3714. [Google Scholar] [CrossRef]
- Call, J.A.; Warren, G.L.; Verma, M.; Lowe, D.A. Acute failure of action potential conduction in mdx muscle reveals new mechanism of contraction-induced force loss. J. Physiol. 2013, 591, 3765–3776. [Google Scholar] [CrossRef]
- Call, J.A.; Eckhoff, M.D.; Baltgalvis, K.A.; Warren, G.L.; Lowe, D.A. Adaptive strength gains in dystrophic muscle exposed to repeated bouts of eccentric contraction. J. Appl. Physiol. 2011, 111, 1768–1777. [Google Scholar] [CrossRef]
- Stapleton, D.I.; Lau, X.; Flores, M.; Trieu, J.; Gehrig, S.M.; Chee, A.; Naim, T.; Lynch, G.S.; Koopman, R. Dysfunctional muscle and liver glycogen metabolism in mdx dystrophic mice. PloS One 2014, 9, e91514. [Google Scholar] [CrossRef]
- Hayes, A.; Williams, D.A. Contractile function and low-intensity exercise effects of old dystrophic (mdx) mice. Am. J. Physiol. 1998, 274, C1138–C1144. [Google Scholar] [CrossRef]
- Hayes, A.; Williams, D.A. Beneficial effects of voluntary wheel running on the properties of dystrophic mouse muscle. J. appl. Physiol. 1996, 80, 670–679. [Google Scholar] [CrossRef]
- Webster, C.; Silberstein, L.; Hays, A.P.; Blau, H.M. Fast muscle fibers are preferentially affected in Duchenne muscular dystrophy. Cell 1988, 52, 503–513. [Google Scholar] [CrossRef]
- Pedemonte, M.; Sandri, C.; Schiaffino, S.; Minetti, C. Early Decrease of IIx Myosin Heavy Chain Transcripts in Duchenne Muscular Dystrophy. Biochem. Biophys. Res. Commun. 1999, 255, 466–469. [Google Scholar] [CrossRef]
- Fink, R.H.; Stephenson, D.G.; Williams, D.A. Calcium and strontium activation of single skinned muscle fibres of normal and dystrophic mice. J. Physiol. 1986, 373, 513–525. [Google Scholar] [CrossRef]
- Williams, D.A.; Head, S.I.; Bakker, A.J.; Stephenson, D.G. Resting calcium concentrations in isolated skeletal muscle fibres of dystrophic mice. J. Physiol. 1990, 428, 243–256. [Google Scholar] [CrossRef]
- Fink, R.H.; Stephenson, D.G.; Williams, D.A. Physiological properties of skinned fibres from normal and dystrophic (Duchenne) human muscle activated by Ca2+ and Sr2+. J. Physiol. 1990, 420, 337–353. [Google Scholar] [CrossRef]
- Tanihata, J.; Nagata, T.; Saito, T.; Ito, N.; Aoki, Y.; Nakamura, A.; Takeda, S. GP 85: Truncated dystrophin with exon 45–55 deletion induced muscle atrophy and fiber type change through the hyper-nitrosylation of the ryanodine receptor type-1 and constant release of Ca2+ to the cytosol. Neuromuscul. Disord. 2014, 24, 820. [Google Scholar] [CrossRef]
- Selsby, J.T.; Morine, K.J.; Pendrak, K.; Barton, E.R.; Sweeney, H.L. Rescue of Dystrophic Skeletal Muscle by PGC-1α Involves a Fast to Slow Fiber Type Shift in the mdx Mouse. PloS One 2012, 7, e30063. [Google Scholar] [CrossRef]
- Garcia, L.A.; Ferrini, M.G.; Norris, K.C.; Artaza, J.N. 1,25(OH)2vitamin D3 enhances myogenic differentiation by modulating the expression of key angiogenic growth factors and angiogenic inhibitors in C2C12 skeletal muscle cells. J. Steroid Biochem. Mol. Biol. 2013, 133, 1–11. [Google Scholar] [CrossRef]
- Dahlquist, D.T.; Dieter, B.P.; Koehle, M.S. Plausible ergogenic effects of vitamin D on athletic performance and recovery. J. Int. Soc. Sports Nutri. 2015, 12, 1–12. [Google Scholar] [CrossRef]
- Ward, K.A.; Das, G.; Berry, J.L.; Roberts, S.A.; Rawer, R.; Adams, J.E.; Mughal, Z. Vitamin D Status and Muscle Function in Post-Menarchal Adolescent Girls. J. Clin. Endocrinol. & Metab. 2009, 94, 559–563. [Google Scholar] [CrossRef]
- Wyon, M.A.; Koutedakis, Y.; Wolman, R.; Nevill, A.M.; Allen, N. The influence of winter vitamin D supplementation on muscle function and injury occurrence in elite ballet dancers: A controlled study. J. Sci. Med. Sport 2014, 17, 8–12. [Google Scholar] [CrossRef]
- Close, G.L.; Russell, J.; Cobley, J.N.; Owens, D.J.; Wilson, G.; Gregson, W.; Fraser, W.D.; Morton, J.P. Assessment of vitamin D concentration in non-supplemented professional athletes and healthy adults during the winter months in the UK: Implications for skeletal muscle function. J. Sports Sci. 2013, 31, 344–353. [Google Scholar] [CrossRef]
- Barker, T.; Martins, T.B.; Hill, H.R.; Kjeldsberg, C.R.; Dixon, B.M.; Schneider, E.D.; Henriksen, V.T.; Weaver, L.K. Circulating pro-inflammatory cytokines are elevated and peak power output correlates with 25-hydroxyvitamin D in vitamin D insufficient adults. Eur. J. Appl. Physiol. 2013, 113, 1523–1534. [Google Scholar] [CrossRef]
- Barker, T.; Henriksen, V.T.; Martins, T.B.; Hill, H.R.; Kjeldsberg, C.R.; Schneider, E.D.; Dixon, B.M.; Weaver, L.K. Higher serum 25-hydroxyvitamin D concentrations associate with a faster recovery of skeletal muscle strength after muscular injury. Nutrients 2013, 5, 1253–1275. [Google Scholar] [CrossRef] [PubMed]
- Mowry, D.A.; Costello, M.M.; Heelan, K.A. Association among cardiorespiratory fitness, body fat, and bone marker measurements in healthy young females. J. Am. Osteopath. Assoc. 2009, 109, 534–539. [Google Scholar] [PubMed]
- Ardestani, A.; Parker, B.; Mathur, S.; Clarkson, P.; Pescatello, L.S.; Hoffman, H.; Polk, D.; Thompson, P.D. Relation of Vitamin D Level to Maximal Oxygen Uptake in Adults. Am. J. Cardiol. 2011, 107, 1246–1249. [Google Scholar] [CrossRef] [PubMed]
- Weltman, A.; Snead, D.; Stein, P.; Seip, R.; Schurrer, R.; Rutt, R.; Weltman, J. Reliability and validity of a continuous incremental treadmill protocol for the determination of lactate threshold, fixed blood lactate concentrations, and VO2max. Int. J. Sports Med. 1990, 11, 26–32. [Google Scholar] [CrossRef]
| Measure | C57BL/10 | mdx | |||||
|---|---|---|---|---|---|---|---|
| 1000 IU n = 10 | 8000 IU n = 10 | 20,000 IU n = 9 | 1000 IU n = 7 | 8000 IU n = 8 | 20,000 IU n = 8 | ||
| Body Weight (g) | 22.1 ± 1.3 | 22.2 ± 1.6 | 23.9 ± 0.97 | 24.6 ± 0.5 ^ | 25.2 ± 1.0 ^ | 25.5 ± 1.1 ^ | |
| EDL | Mass (mg) | 9.5 ± 0.6 | 8.7 ± 0.4 | 8.8 ± 0.3 | 16.8 ± 2.2 ^ | 17.3 ± 3.4 ^^ | 23.1 ± 1.8 ^^^ |
| EDL:BM (mg/g) | 0.046 ± 0.002 | 0.041 ± 0.002 | 0.041 ± 0.002 | 0.068 ± 0.009 | 0.074 ± 0.015 ^ | 0.090 ± 0.007 ^^^ | |
| CSA (cm²) | 0.017 ± 0.001 | 0.016 ± 0.001 | 0.016 ± 0.001 | 0.035 ± 0.006 ^ | 0.038 ± 0.008 ^^ | 0.047 ± 0.004 ^^^ | |
| SOLEUS | Mass (mg) | 8.75 ± 0.47 | 7.42 ± 0.42 | 7.87 ± 0.43 | 14.9 ± 1.7 ^ | 14.8 ± 1.2 ^^ | 15.7 ± 2.4 ^^^ |
| Soleus:BM (mg/g) | 0.041 ± 0.002 | 0.035 ± 0.002 | 0.038 ± 0.003 | 0.061 ± 0.007 | 0.059 ± 0.005 | 0.072 ± 0.018 ^ | |
| CSA (cm²) | 0.010 ± 0.001 | 0.010 ± 0.001 | 0.009 ± 0.001 | 0.017 ± 0.002 ^ | 0.021 ± 0.002 ^^^ | 0.018 ± 0.002 ^^ | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Debruin, D.A.; Andreacchio, N.; Hanson, E.D.; Timpani, C.A.; Rybalka, E.; Hayes, A. The Effect of Vitamin D Supplementation on Skeletal Muscle in the mdx Mouse Model of Duchenne Muscular Dystrophy. Sports 2019, 7, 96. https://doi.org/10.3390/sports7050096
Debruin DA, Andreacchio N, Hanson ED, Timpani CA, Rybalka E, Hayes A. The Effect of Vitamin D Supplementation on Skeletal Muscle in the mdx Mouse Model of Duchenne Muscular Dystrophy. Sports. 2019; 7(5):96. https://doi.org/10.3390/sports7050096
Chicago/Turabian StyleDebruin, Danielle A., Nicola Andreacchio, Erik D. Hanson, Cara A. Timpani, Emma Rybalka, and Alan Hayes. 2019. "The Effect of Vitamin D Supplementation on Skeletal Muscle in the mdx Mouse Model of Duchenne Muscular Dystrophy" Sports 7, no. 5: 96. https://doi.org/10.3390/sports7050096
APA StyleDebruin, D. A., Andreacchio, N., Hanson, E. D., Timpani, C. A., Rybalka, E., & Hayes, A. (2019). The Effect of Vitamin D Supplementation on Skeletal Muscle in the mdx Mouse Model of Duchenne Muscular Dystrophy. Sports, 7(5), 96. https://doi.org/10.3390/sports7050096








