Friction vs. Area Scaling of Superlubric NaCl-Particles on Graphite
Abstract
1. Introduction
2. Materials and Methods
2.1. Nanoparticle Preparation
2.2. Nanoparticle Manipulation
3. Results
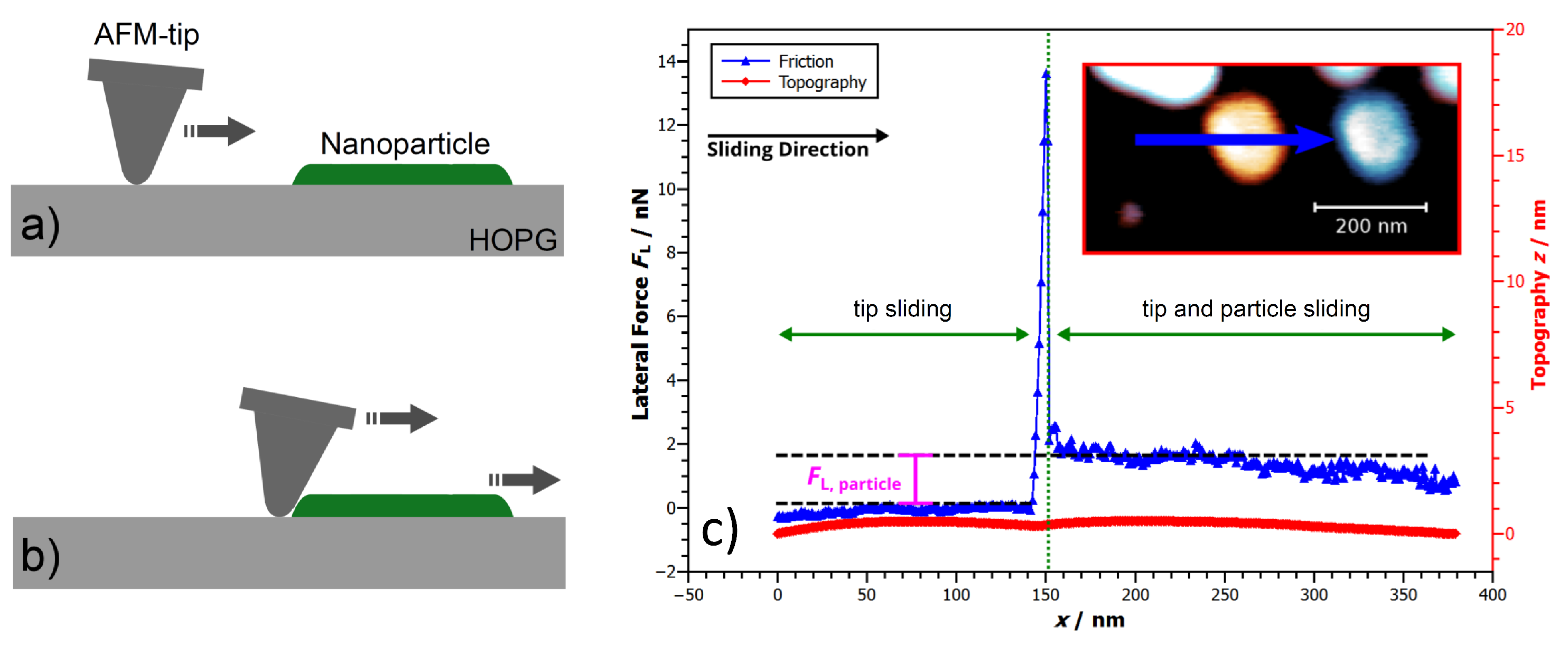
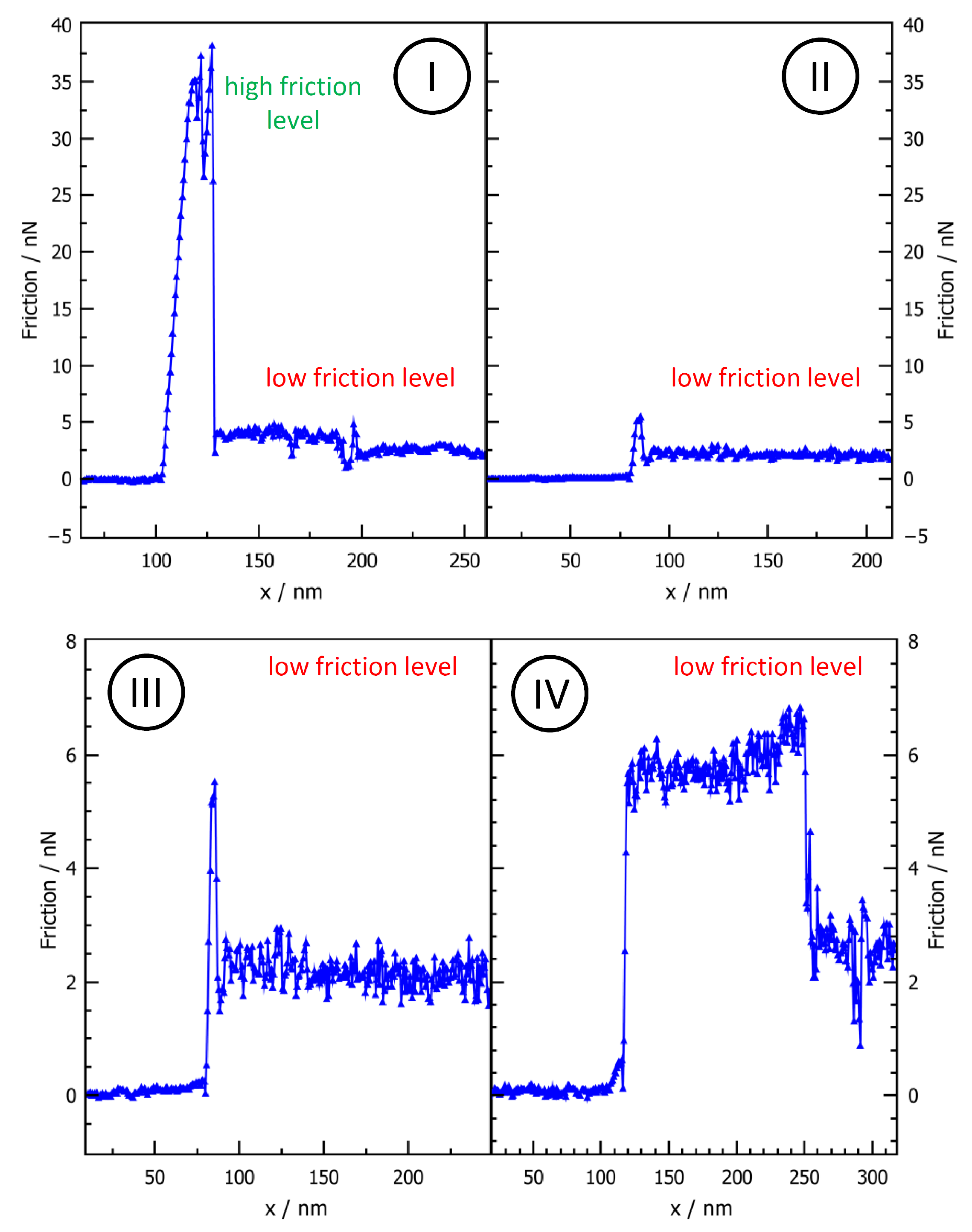
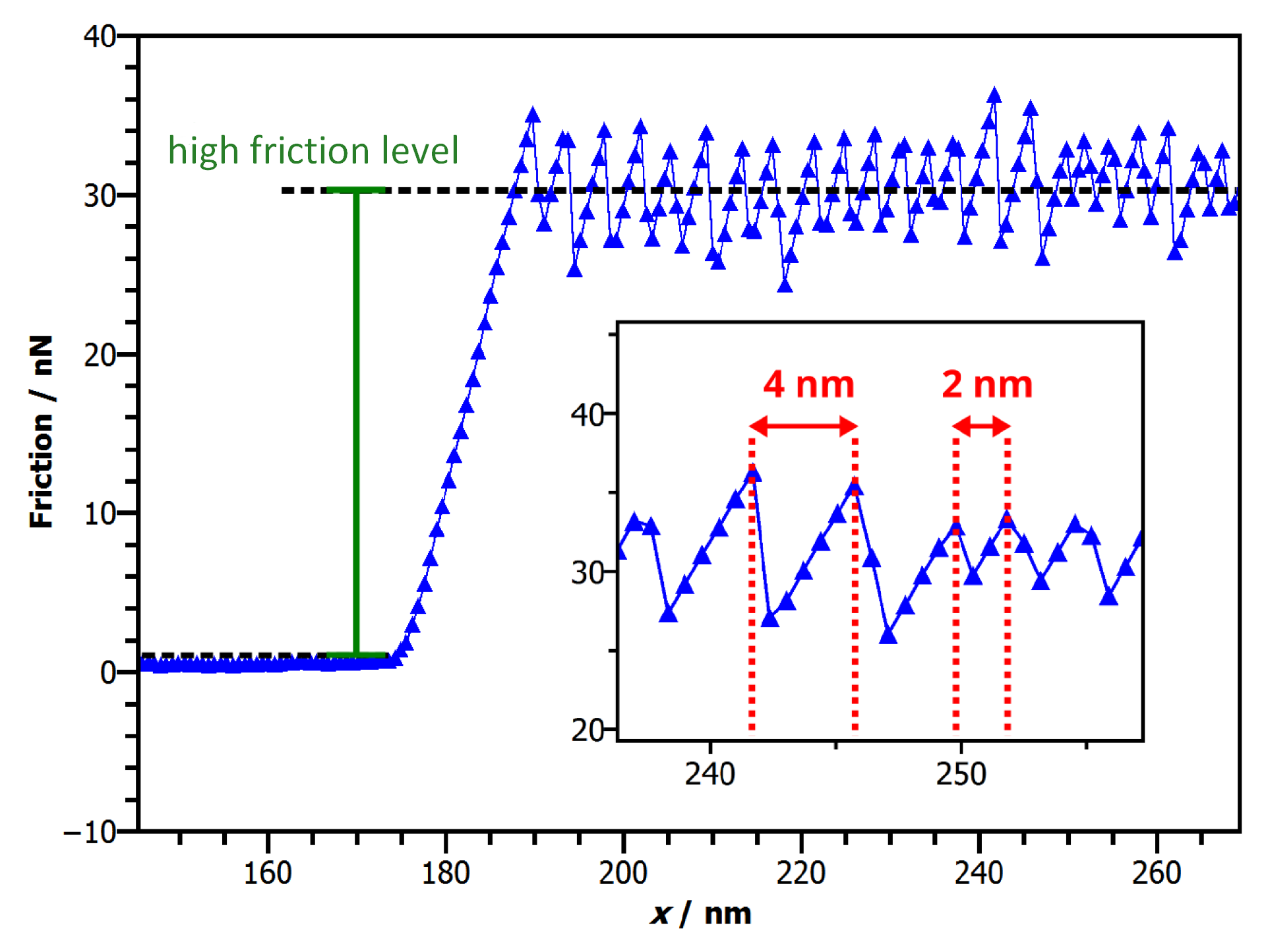
3.1. Friction Changes during Manipulation
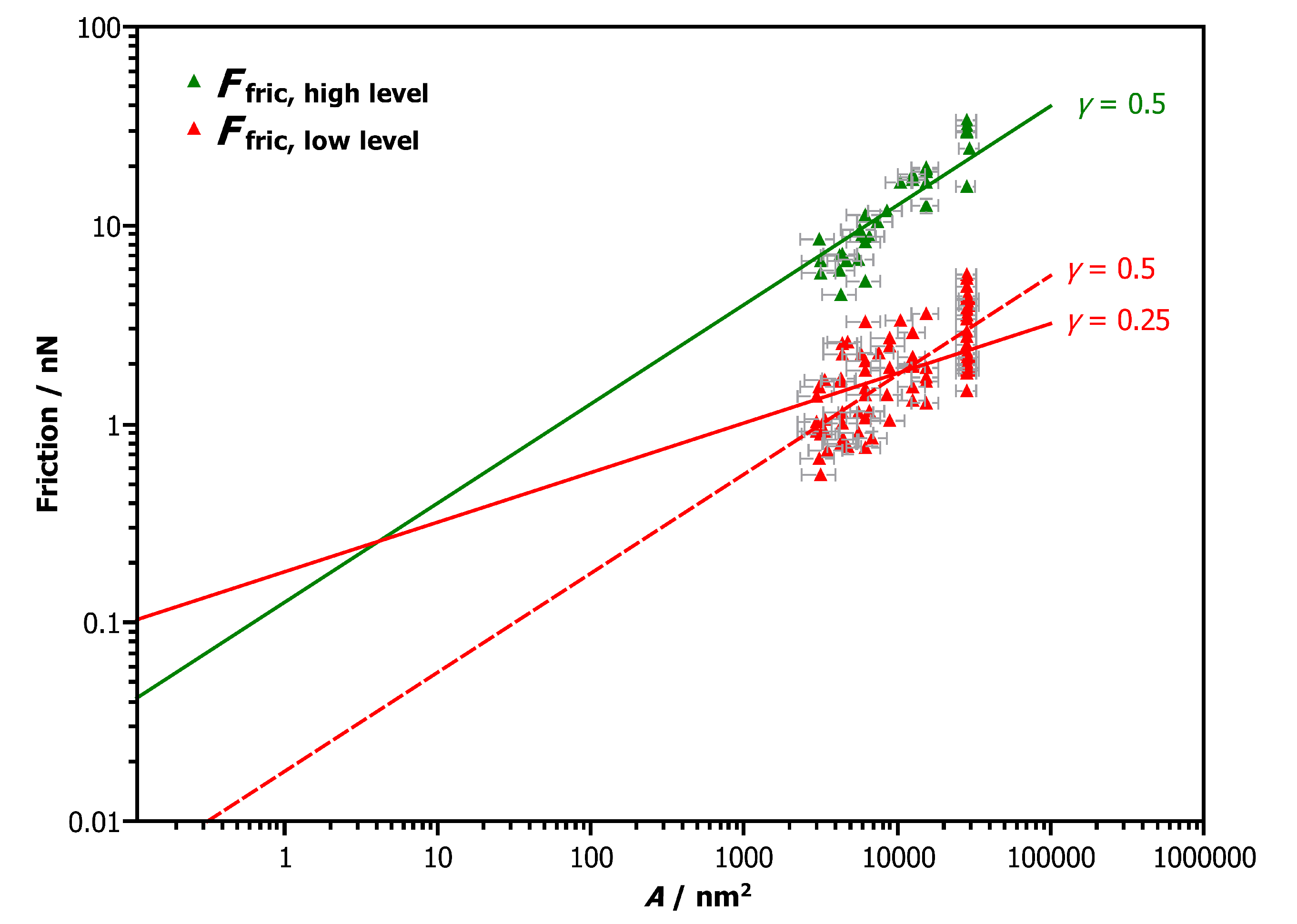
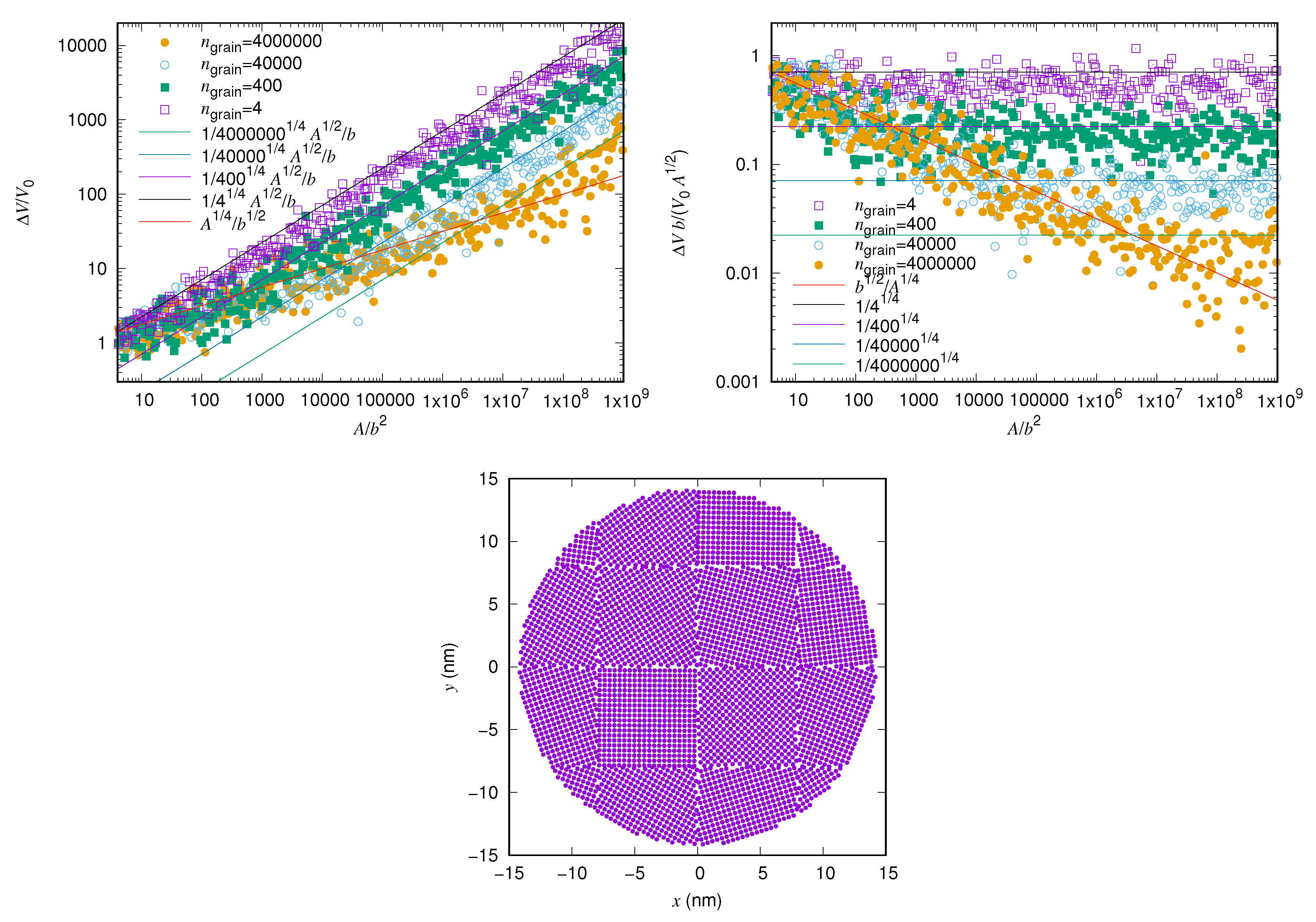
3.2. Friction versus Area Scaling
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| HOPG | Highly oriented pyrolythic grpahite |
| AFM | Atomic force microscope |
| NC-AFM | Non-contact atomic force microscope |
| VT | Variable temperature |
| UHV | Ultra high vacuum |
| SEM | Scanning electron microscope |
References
- Vanossi, A.; Dietzel, D.; Schirmeisen, A.; Meyer, E.; Pawlak, R.; Glatzel, T.; Kisiel, M.; Kawai, S.; Manini, N. Recent highlights in nanoscale and mesoscale friction. Beilstein J. Nanotechnol. 2018, 9, 1995–2014. [Google Scholar] [CrossRef] [PubMed]
- Baykara, M.Z.; Vazirisereshk, M.R.; Martini, A. Emerging superlubricity: A review of the state of the art and perspectives on future research. Appl. Phys. Rev. 2018, 5, 041102. [Google Scholar] [CrossRef]
- Dietzel, D.; Schwarz, U.D.; Schirmeisen, A. Nanotribological studies using nanoparticle manipulation: Principles and application to structural lubricity. Friction 2014, 2, 114–139. [Google Scholar] [CrossRef]
- Müser, M.; Wenning, L.; Robbins, M. Simple Microscopic Theory of Amontons’s Laws for Static Friction. Phys. Rev. Lett. 2001, 86, 1295–1298. [Google Scholar] [CrossRef] [PubMed]
- Dietzel, D.; Ritter, C.; Mönninghoff, T.; Fuchs, H.; Schirmeisen, A.; Schwarz, U. Frictional Duality Observed during Nanoparticle Sliding. Phys. Rev. Lett. 2008, 101, 125505. [Google Scholar] [CrossRef] [PubMed]
- Dietzel, D.; Feldmann, M.; Schwarz, U.D.; Fuchs, H.; Schirmeisen, A. Scaling Laws of Structural Lubricity. Phys. Rev. Lett. 2013, 111, 235502. [Google Scholar] [CrossRef] [PubMed]
- Feldmann, M.; Dietzel, D.; Fuchs, H.; Schirmeisen, A. Influence of Contact Aging on Nanoparticle Friction Kinetics. Phys. Rev. Lett. 2014, 112, 155503. [Google Scholar] [CrossRef] [PubMed]
- Feldmann, M.; Dietzel, D.; Tekiel, A.; Topple, J.; Grütter, P.; Schirmeisen, A. Universal Aging Mechanism for Static and Sliding Friction of Metallic Nanoparticles. Phys. Rev. Lett. 2016, 117. [Google Scholar] [CrossRef]
- Trillitzsch, F.; Guerra, R.; Janas, A.; Manini, N.; Krok, F.; Gnecco, E. Directional and angular locking in the driven motion of Au islands on MoS 2. Phys. Rev. B 2018, 98. [Google Scholar] [CrossRef]
- Dietzel, D.; Brndiar, J.; Stich, I.; Schirmeisen, A. Limitations of Structural Superlubricity: Chemical Bonds versus Contact Size. ACS Nano 2017, 11, 7642–7647. [Google Scholar] [CrossRef] [PubMed]
- Cihan, E.; Ipek, S.; Durgun, E.; Baykara, M.Z. Structural lubricity under ambient conditions. Nat. Commun. 2016, 7, 12055. [Google Scholar] [CrossRef] [PubMed]
- Özogul, A.; Ipek, S.; Durgun, E.; Baykara, M.Z. Structural superlubricity of platinum on graphite under ambient conditions: The effects of chemistry and geometry. Appl. Phys. Lett. 2017, 111, 211602. [Google Scholar] [CrossRef]
- Zheng, Q.; Jiang, B.; Liu, S.; Weng, Y.; Lu, L.; Xue, Q.; Zhu, J.; Jiang, Q.; Wang, S.; Peng, L. Self-Retracting Motion of Graphite Microflakes. Phys. Rev. Lett. 2008, 100, 067205. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Yang, J.; Grey, F.; Liu, J.Z.; Liu, Y.; Wang, Y.; Yang, Y.; Cheng, Y.; Zheng, Q. Observation of Microscale Superlubricity in Graphite. Phys. Rev. Lett. 2012, 108, 205503. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Grey, F.; Zheng, Q. The high-speed sliding friction of graphene and novel routes to persistent superlubricity. Sci. Rep. 2014, 4. [Google Scholar] [CrossRef] [PubMed]
- Vu, C.C.; Zhang, S.; Urbakh, M.; Li, Q.; He, Q.C.; Zheng, Q. Observation of normal-force-independent superlubricity in mesoscopic graphite contacts. Phys. Rev. B 2016, 94, 081405. [Google Scholar] [CrossRef]
- Ma, M.; Sokolov, I.M.; Wang, W.; Filippov, A.E.; Zheng, Q.; Urbakh, M. Diffusion through Bifurcations in Oscillating Nano- and Microscale Contacts: Fundamentals and Applications. Phys. Rev. X 2015, 5, 031020. [Google Scholar] [CrossRef]
- Deng, H.; Ma, M.; Song, Y.; He, Q.; Zheng, Q. Structural superlubricity in graphite flakes assembled under ambient conditions. Nanoscale 2018, 10, 14314–14320. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Mandelli, D.; Hod, O.; Urbakh, M.; Ma, M.; Zheng, Q. Robust microscale superlubricity in graphite/hexagonal boron nitride layered heterojunctions. Nat. Mater. 2018, 17, 894–899. [Google Scholar] [CrossRef] [PubMed]
- Müser, M.H. Theoretical Aspects of Superlubricity. In Fundamentals of Friction and Wear; Springer: Berlin, Germany, 2007; pp. 177–199. [Google Scholar]
- Sharp, T.A.; Pastewka, L.; Robbins, M.O. Elasticity limits structural superlubricity in large contacts. Phys. Rev. B 2016, 93, 121402. [Google Scholar] [CrossRef]
- Dienwiebel, M.; Verhoeven, G.; Pradeep, N.; Frenken, J.; Heimberg, J.; Zandbergen, H. Superlubricity of Graphite. Phys. Rev. Lett. 2004, 92, 126101. [Google Scholar] [CrossRef] [PubMed]
- de Wijn, A.S. (In)commensurability, scaling, and multiplicity of friction in nanocrystals and application to gold nanocrystals on graphite. Phys. Rev. B 2012, 86, 085429. [Google Scholar] [CrossRef]
- Dietzel, D.; de Wijn, A.S.; Vorholzer, M.; Schirmeisen, A. Friction fluctuations of gold nanoparticles in the superlubric regime. Nanotechnology 2018, 29, 155702. [Google Scholar] [CrossRef] [PubMed]
- Varini, N.; Vanossi, A.; Guerra, R.; Mandelli, D.; Capozza, R.; Tosatti, E. Static friction scaling of physisorbed islands: The key is in the edge. Nanoscale 2014, 7, 2093–2101. [Google Scholar] [CrossRef] [PubMed]
- Guerra, R.; Tosatti, E.; Vanossi, A. Slider thickness promotes lubricity: From 2D islands to 3D clusters. Nanoscale 2016, 8, 11108–11113. [Google Scholar] [CrossRef] [PubMed]
- Ma, M.; Benassi, A.; Vanossi, A.; Urbakh, M. Critical Length Limiting Superlow Friction. Phys. Rev. Lett. 2015, 114, 055501. [Google Scholar] [CrossRef] [PubMed]
- Zint, S.; Ebeling, D.; Ahles, S.; Wegner, H.A.; Schirmeisen, A. Subsurface-Controlled Angular Rotation: Triphenylene Molecules on Au(111) Substrates. J. Phys. Chem. C 2016, 120, 1615–1622. [Google Scholar] [CrossRef]
- Cao, P.; Xu, K.; Varghese, J.O.; Heath, J.R. The Microscopic Structure of Adsorbed Water on Hydrophobic Surfaces under Ambient Conditions. Nano Lett. 2011, 11, 5581–5586. [Google Scholar] [CrossRef]
- Luna, M.; Rieutord, F.; Melman, N.A.; Dai, Q.; Salmeron, M. Adsorption of Water on Alkali Halide Surfaces Studied by Scanning Polarization Force Microscopy. J. Phys. Chem. A 1998, 102, 6793–6800. [Google Scholar] [CrossRef]
- Krämer, L.; Pöschl, U.; Niessner, R. Microstructural rearrangement of sodium chloride condensation aerosol particles on interaction with water vapor. J. Aerosol Sci. 2000, 31, 673–685. [Google Scholar] [CrossRef]
- Mate, C.M.; Mcclelland, G.M.; Erlandsson, R.; Chiang, S. Atomic-Scale Friction of a Tungsten Tip on a Graphite Surface. Phys. Rev. Lett. 1987, 59, 1942–1946. [Google Scholar] [CrossRef] [PubMed]
- Varenberg, M.; Etsion, I.; Halperin, G. An improved wedge calibration method for lateral force in atomic force microscopy. Rev. Sci. Instrum. 2003, 74, 3362. [Google Scholar] [CrossRef]
- de Wijn, A.S.; Fusco, C.; Fasolino, A. Stability of superlubric sliding on graphite. Phys. Rev. E 2010, 81, 046105. [Google Scholar] [CrossRef] [PubMed]
- Matthews, J.W.; Grünbauma, E. The structure of gold films grown in ultra-high vacuum on sodium chloride substrates. Philos. Mag. A J. Theor. Exp. Appl. Phys. 1965, 11, 1233–1244. [Google Scholar] [CrossRef]
- Jinesh, K.B.; Frenken, J.W.M. Experimental Evidence for Ice Formation at Room Temperature. Phys. Rev. Lett. 2008, 101, 036101. [Google Scholar] [CrossRef] [PubMed]
- Kimmel, G.A.; Matthiesen, J.; Baer, M.; Mundy, C.J.; Petrik, N.G.; Dohnálek, Z.; Kay, B.D. No Confinement Needed: Observation of a Metastable Hydrophobic Wetting Two-Layer Ice on Graphene. J. Am. Chem. Soc. 2009, 131, 12838–12844. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: All samples and materials used in this work are readily available from commercial sources. |

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hartmuth, F.; Dietzel, D.; de Wijn, A.S.; Schirmeisen, A. Friction vs. Area Scaling of Superlubric NaCl-Particles on Graphite. Lubricants 2019, 7, 66. https://doi.org/10.3390/lubricants7080066
Hartmuth F, Dietzel D, de Wijn AS, Schirmeisen A. Friction vs. Area Scaling of Superlubric NaCl-Particles on Graphite. Lubricants. 2019; 7(8):66. https://doi.org/10.3390/lubricants7080066
Chicago/Turabian StyleHartmuth, Felix, Dirk Dietzel, Astrid S. de Wijn, and André Schirmeisen. 2019. "Friction vs. Area Scaling of Superlubric NaCl-Particles on Graphite" Lubricants 7, no. 8: 66. https://doi.org/10.3390/lubricants7080066
APA StyleHartmuth, F., Dietzel, D., de Wijn, A. S., & Schirmeisen, A. (2019). Friction vs. Area Scaling of Superlubric NaCl-Particles on Graphite. Lubricants, 7(8), 66. https://doi.org/10.3390/lubricants7080066



