Is Macular Telangiectasia Type 2 Associated with Hearing Loss and Cochlear Dysfunction? A Prospective Case–Control Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Participants
2.2. Ophthalmic Examination and Diagnostic Workup
2.3. Audiometric Assessments
2.3.1. Pure-Tone Audiometry and Tympanometry Evaluation
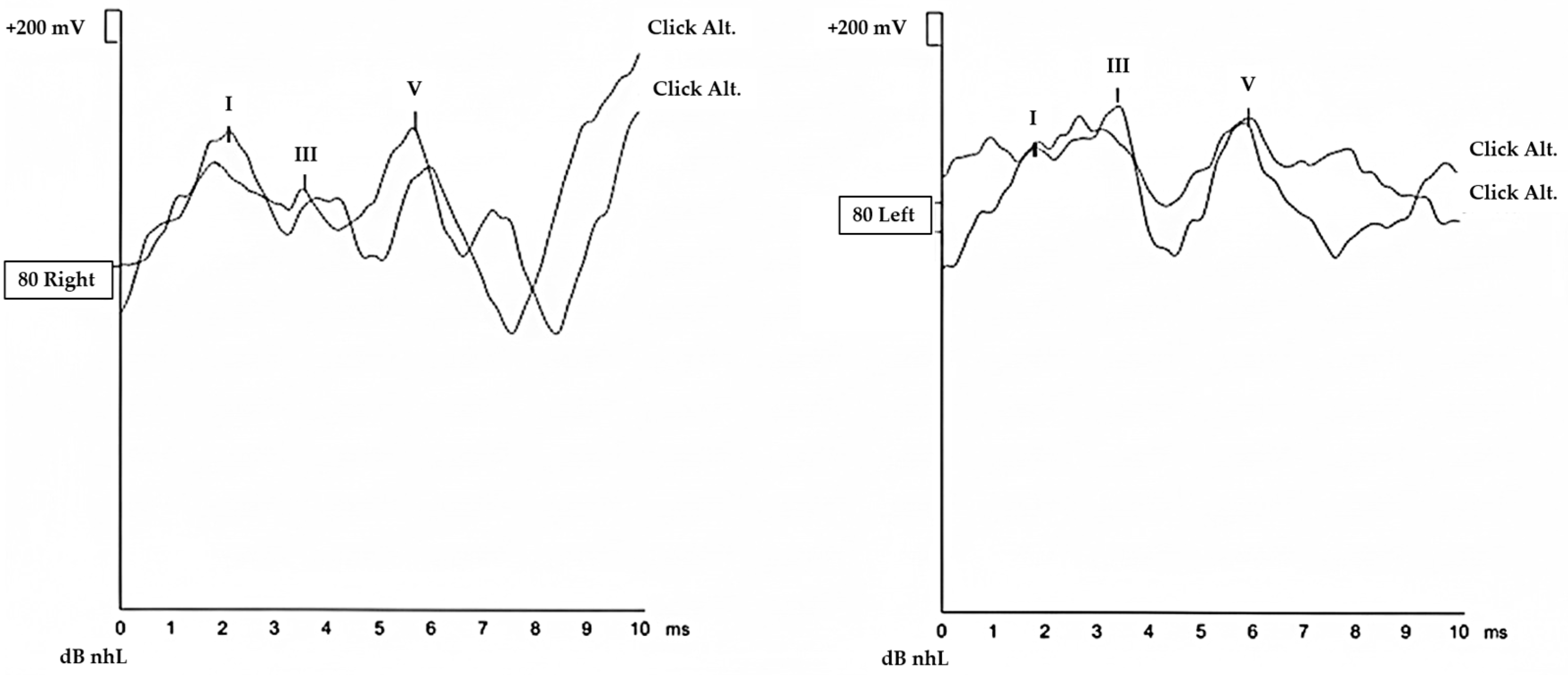
2.3.2. Auditory Brainstem Response (ABR) Testing
2.3.3. Distortion Product Otoacoustic Emissions (DPOAE)
2.4. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ABR | Auditory brainstem response |
| ANSI | American National Standards Institute |
| dB HL | Decibel hearing level |
| dB SPL | Decibel sound pressure level |
| DP-gram | Distortion product gram |
| DPOAE | Distortion product otoacoustic emissions |
| FA | Fluorescein angiography |
| f1, f2 | Primary-tone frequencies (DPOAE) |
| Hz | Hz: Hertz |
| IQR | Interquartile range |
| IPL | Interpeak latency |
| kHz | Kilohertz |
| L1, L2 | Primary-tone stimulus levels (DPOAE) |
| MacTel2 | Macular telangiectasia type 2 |
| OCT | Optical coherence tomography |
| PTA | Pure-tone audiometry |
| PTA4 | Four-frequency pure-tone average (0.5, 1, 2, and 4 kHz) |
| SNR | Signal-to-noise ratio |
| WHO | World Health Organization |
References
- He, M.; Yang, Z.; Chen, Y.; Zhao, X. Clinical characteristics, imaging features, and treatment outcomes of macular telangiectasia type 2: A comprehensive meta-analysis. Sci. Rep. 2026, 16, 2453. [Google Scholar] [CrossRef]
- Ntentakis, D.P.; Ntentaki, A.M.; Delavogia, E.; Sakuno, G.; Corrêa, V.S.M.C.; Efstathiou, N.E.; Aronow, M.E.; Chew, E.Y.; Miller, J.W.; Vavvas, D.G. Clinical histopathology and pathogenesis of macular telangiectasia type 2. Prog. Retin. Eye Res. 2026, 110, 101401. [Google Scholar] [CrossRef]
- Roisman, L.; Rosenfeld, P.J. Optical Coherence Tomography Angiography of Macular Telangiectasia Type 2. Dev. Ophthalmol. 2016, 56, 146–158. [Google Scholar] [CrossRef]
- Powner, M.B.; Gillies, M.C.; Zhu, M.; Vevis, K.; Hunyor, A.P.; Fruttiger, M. Loss of Müller’s cells and photoreceptors in macular telangiectasia type 2. Ophthalmology 2013, 120, 2344–2352. [Google Scholar] [CrossRef]
- Kedarisetti, K.C.; Narayanan, R.; Stewart, M.W.; Gurram, N.R.; Khanani, A.M. Macular Telangiectasia Type 2: A Comprehensive Review. Clin. Ophthalmol. 2022, 16, 3297–3309. [Google Scholar] [CrossRef]
- Eade, K.; Gantner, M.L.; Hostyk, J.A.; Nagasaki, T.; Giles, S.; Fallon, R.; Harkins-Perry, S.; Baldini, M.; Lim, E.W.; Scheppke, L.; et al. Serine biosynthesis defect due to haploinsufficiency of PHGDH causes retinal disease. Nat. Metab. 2021, 3, 366–377. [Google Scholar] [CrossRef]
- Bonelli, R.; Ansell, B.R.E.; Lotta, L.; Scerri, T.; Clemons, T.E.; Leung, I.; Consortium, M.; Peto, T.; Bird, A.C.; Sallo, F.B.; et al. Genetic disruption of serine biosynthesis is a key driver of macular telangiectasia type 2 aetiology and progression. Genome Med. 2021, 13, 39. [Google Scholar] [CrossRef]
- Bonelli, R.; Jackson, V.E.; Prasad, A.; Munro, J.E.; Farashi, S.; Heeren, T.F.C.; Pontikos, N.; Scheppke, L.; Friedlander, M.; Consortium, M.; et al. Identification of genetic factors influencing metabolic dysregulation and retinal support for MacTel, a retinal disorder. Commun. Biol. 2021, 4, 274, Erratum in Commun. Biol. 2021, 4, 473. https://doi.org/10.1038/s42003-021-01972-y. [Google Scholar] [CrossRef] [PubMed]
- Bonelli, R.; Woods, S.M.; Ansell, B.R.E.; Heeren, T.F.C.; Egan, C.A.; Khan, K.N.; Guymer, R.; Trombley, J.; Friedlander, M.; Bahlo, M.; et al. Systemic lipid dysregulation is a risk factor for macular neurodegenerative disease. Sci. Rep. 2020, 10, 12165. [Google Scholar] [CrossRef] [PubMed]
- Triplett, J.; Nicholson, G.; Sue, C.; Hornemann, T.; Yiannikas, C. Hereditary sensory and autonomic neuropathy type IC accompanied by upper motor neuron abnormalities and type II juxtafoveal retinal telangiectasias. J. Peripher. Nerv. Syst. 2019, 24, 224–229. [Google Scholar] [CrossRef] [PubMed]
- Wu, L. Unraveling the mysteries of macular telangiectasia 2: The intersection of philanthropy, multimodal imaging and molecular genetics. The 2022 founders lecture of the pan American vitreoretinal society. Int. J. Retin. Vitr. 2023, 9, 69. [Google Scholar] [CrossRef]
- Kunčič, A.; Urbančič, M.; Divjak, D.D.; Hudler, P.; Debeljak, N. Genetic Background of Macular Telangiectasia Type 2. Int. J. Mol. Sci. 2025, 26, 684. [Google Scholar] [CrossRef] [PubMed]
- Green, C.R.; Bonelli, R.; Ansell, B.R.E.; Tzaridis, S.; Handzlik, M.K.; McGregor, G.H.; Hart, B.; Trombley, J.; Reilly, M.M.; Bernstein, P.S.; et al. Divergent amino acid and sphingolipid metabolism in patients with inherited neuro-retinal disease. Mol. Metab. 2023, 72, 101716. [Google Scholar] [CrossRef]
- Seidman, M.D.; Quirk, W.S.; Shirwany, N.A. Mechanisms of alterations in the microcirculation of the cochlea. Ann. N. Y. Acad. Sci. 1999, 884, 226–232. [Google Scholar] [CrossRef]
- Teraoka, M.; Hato, N.; Inufusa, H.; You, F. Role of Oxidative Stress in Sensorineural Hearing Loss. Int. J. Mol. Sci. 2024, 25, 4146. [Google Scholar] [CrossRef]
- Kishimoto-Urata, M.; Urata, S.; Fujimoto, C.; Yamasoba, T. Role of Oxidative Stress and Antioxidants in Acquired Inner Ear Disorders. Antioxidants 2022, 11, 1469. [Google Scholar] [CrossRef]
- da Costa, K.V.T.; de Andrade, K.C.L.; di Cavalcanti, M.E.; Frizzo, A.C.F.; Carnaúba, A.T.L.; de Lemos Menezes, P. Hearing Loss at High Frequencies and Oxidative Stress: A New Paradigm for Different Etiologies. In An Excursus into Hearing Loss; Hatzopoulos, S., Ciorba, A., Eds.; IntechOpen: London, UK, 2018. [Google Scholar]
- Sulaiman, A.H.; Husain, R.; Seluakumaran, K. Evaluation of early hearing damage in personal listening device users using extended high-frequency audiometry and otoacoustic emissions. Eur. Arch. Otorhinolaryngol. 2014, 271, 1463–1470. [Google Scholar] [CrossRef] [PubMed]
- Toto, L.; Di Antonio, L.; Mastropasqua, R.; Mattei, P.A.; Carpineto, P.; Borrelli, E.; Rispoli, M.; Lumbroso, B.; Mastropasqua, L. Multimodal Imaging of Macular Telangiectasia Type 2: Focus on Vascular Changes Using Optical Coherence Tomography Angiography. Investig. Ophthalmol. Vis. Sci. 2016, 57, OCT268–OCT276. [Google Scholar] [CrossRef] [PubMed]
- Jerger, J. Clinical experience with impedance audiometry. Arch. Otolaryngol. 1970, 92, 311–324. [Google Scholar] [CrossRef]
- Chew, E.Y.; Peto, T.; Clemons, T.E.; Sallo, F.B.; Pauleikhoff, D.; Leung, I.; Jaffe, G.J.; Heeren, T.F.C.; Egan, C.A.; Issa, P.C.; et al. Macular Telangiectasia Type 2: A Classification System Using MultiModal Imaging MacTel Project Report Number 10. Ophthalmol. Sci. 2022, 3, 100261. [Google Scholar] [CrossRef]
- Issa, P.C.; Gillies, M.C.; Chew, E.Y.; Bird, A.C.; Heeren, T.F.; Peto, T.; Holz, F.G.; Scholl, H.P. Macular telangiectasia type 2. Prog. Retin. Eye Res. 2013, 34, 49–77. [Google Scholar] [CrossRef]
- Janssens-Bij de Vaate, M.; Rhebergen, K.S. Comparison of three calibration procedures for free-field reference speech audiometry. Int. J. Audiol. 2024, 63, 527–534. [Google Scholar] [CrossRef] [PubMed]
- ANSI/ASA S3.6-2018; Specification for Audiometers. American National Standards Institute (ANSI): New York, NY, USA, 2018.
- Humes, L.E. The World Health Organization’s hearing-impairment grading system: An evaluation for unaided communication in age-related hearing loss. Int. J. Audiol. 2019, 58, 12–20. [Google Scholar] [CrossRef]
- De Gruy, J.A.; Hopper, S.; Kelly, W.; Witcher, R.; Vu, T.H.; Spankovich, C. Defining Hearing Loss Severity Based on Pure-Tone Audiometry and Self-Reported Perceived Hearing Difficulty: National Health and Nutrition Examination Survey. J. Am. Acad. Audiol. 2023, 34, 100–113. [Google Scholar] [CrossRef]
- Bringmann, A.; Pannicke, T.; Grosche, J.; Francke, M.; Wiedemann, P.; Skatchkov, S.N.; Osborne, N.N.; Reichenbach, A. Müller cells in the healthy and diseased retina. Prog. Retin. Eye Res. 2006, 25, 397–424. [Google Scholar] [CrossRef]
- Leinonen, H.O.; Bull, E.; Fu, Z. Neural and Müller glial adaptation of the retina to photoreceptor degeneration. Neural Regen. Res. 2023, 18, 701–707. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Zheng, Z.; Xie, D.; Xia, L.; Chen, Y.; Dong, H.; Feng, Y. Serum lipid metabolism characteristics and potential biomarkers in patients with unilateral sudden sensorineural hearing loss. Lipids Health Dis. 2024, 23, 205. [Google Scholar] [CrossRef]
- Lim, E.W.; Fallon, R.J.; Bates, C.; Ideguchi, Y.; Nagasaki, T.; Handzlik, M.K.; Joulia, E.; Bonelli, R.; Green, C.R.; Ansell, B.R.E.; et al. Serine and glycine physiology reversibly modulate retinal and peripheral nerve function. Cell Metab. 2024, 36, 2315–2328.e6. [Google Scholar] [CrossRef]
- Gantner, M.L.; Eade, K.; Wallace, M.; Handzlik, M.K.; Fallon, R.; Trombley, J.; Bonelli, R.; Giles, S.; Harkins-Perry, S.; Heeren, T.F.C.; et al. Serine and Lipid Metabolism in Macular Disease and Peripheral Neuropathy. N. Engl. J. Med. 2019, 381, 1422–1433. [Google Scholar] [CrossRef] [PubMed]
- Janssen, T.; Niedermeyer, H.P.; Arnold, W. Diagnostics of the cochlear amplifier by means of distortion product otoacoustic emissions. ORL J. Otorhinolaryngol. Relat. Spec. 2006, 68, 334–339. [Google Scholar] [CrossRef]
- Go, N.A.; Stamper, G.C.; Johnson, T.A. Cochlear Mechanisms and Otoacoustic Emission Test Performance. Ear Hear. 2019, 40, 401–417. [Google Scholar] [CrossRef]
- Korres, G.S.; Balatsouras, D.G.; Tzagaroulakis, A.; Kandiloros, D.; Ferekidou, E.; Korres, S. Distortion product otoacoustic emissions in an industrial setting. Noise Health 2009, 11, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Dalhoff, E.; Zelle, D.; Bader, K. Latencies of Pulsed Distortion-Product Otoacoustic Emissions and Their Relation to Auditory Brainstem Responses. J. Assoc. Res. Otolaryngol. 2025. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Shen, W.; Zhu, M.; Coorey, N.J.; Nguyen, A.P.; Barthelmes, D.; Gillies, M.C. Anti-retinal antibodies in patients with macular telangiectasia type 2. Investig. Ophthalmol. Vis. Sci. 2013, 54, 5675–5683. [Google Scholar] [CrossRef][Green Version]
- Hepkarsi, S.; Aliyeva, A.; Kirazli, G.; Kirazli, T. Evaluating Inflammatory Markers in Sudden Sensorineural Hearing Loss: The Neutrophil-To Lymphocyte Ratio, The Systemic Immune Inflammation Index, and The Pan Immune Inflammation Value as Prognostic Tools. Osman. J. Med. 2025, 47, 386–394. [Google Scholar] [CrossRef]
- Aliyeva, A.; Sari, E. Evaluation of autoimmune and inflammatory markers in bilateral sudden hearing loss. Eur. Arch. Otorhinolaryngol. 2025, 282, 4637–4644. [Google Scholar] [CrossRef]
- Aliyeva, A.; Agayarov, O.Y.; Dogan, E.I. Assessing Auditory and Cochlear Function in Alopecia Areata Patients: Exploring the Link to Cochlear Melanocyte Damage. Cureus 2023, 15, e44882. [Google Scholar] [CrossRef] [PubMed]
- Iwasa, K.H. Effectiveness of outer hair cells as cochlear amplifier: In simple model systems. Biophys. J. 2025, 124, 3811, Erratum in Biophys. J. 2025, 124, 2383–2390. https://doi.org/10.1016/j.bpj.2025.05.029. [Google Scholar] [CrossRef]
- Bramhall, N.F. Use of the auditory brainstem response for assessment of cochlear synaptopathy in humans. J. Acoust. Soc. Am. 2021, 150, 4440. [Google Scholar] [CrossRef]
- Shin, S.A.; Lyu, A.R.; Jeong, S.H.; Kim, T.H.; Park, M.J.; Park, Y.H. Acoustic Trauma Modulates Cochlear Blood Flow and Vasoactive Factors in a Rodent Model of Noise-Induced Hearing Loss. Int. J. Mol. Sci. 2019, 20, 5316. [Google Scholar] [CrossRef]
- Gao, Y.; Wu, F.; He, W.; Cai, Z.; Pang, J.; Zheng, Y. Reactive Oxygen Species-Related Disruptions to Cochlear Hair Cell and Stria Vascularis Consequently Leading to Radiation-Induced Sensorineural Hearing Loss. Antioxid. Redox Signal. 2024, 40, 470–491. [Google Scholar] [CrossRef]
- Zhou, Y.; Fang, C.; Yuan, L.; Guo, M.; Xu, X.; Shao, A.; Zhang, A.; Zhou, D. Redox homeostasis dysregulation in noise-induced hearing loss: Oxidative stress and antioxidant treatment. J. Otolaryngol. Head Neck Surg. 2023, 52, 78. [Google Scholar] [CrossRef] [PubMed]
- Škerková, M.; Kovalová, M.; Mrázková, E. High-Frequency Audiometry for Early Detection of Hearing Loss: A Narrative Review. Int. J. Environ. Res. Public Health 2021, 18, 4702. [Google Scholar] [CrossRef] [PubMed]
- Troskie, N. Early Identification and Monitoring of Cochlear Damage Using Extended High-Frequency Audiometry: A Systematic Review. Ph.D. Dissertation, University of Pretoria, Pretoria, South Africa, 2021. [Google Scholar]
- Ueberfuhr, M.A.; Fehlberg, H.; Goodman, S.S.; Withnell, R.H. A DPOAE assessment of outer hair cell integrity in ears with age-related hearing loss. Hear. Res. 2016, 332, 137–150. [Google Scholar] [CrossRef] [PubMed]



| Variable | MacTel 2 Group (n:21) | Control Group (n:21) | p-Value |
|---|---|---|---|
| Gender | 1.000 1 | ||
| Female | 13 (61.9%) | 13 (61.9%) | |
| Male | 8 (38.1%) | 8 (38.1%) | |
| Age | |||
| Mean ± SD | 51.81 ± 8.00 | 52.38 ± 9.47 | 0.834 2 |
| Median (IQR) | 53 (45–58) | 52 (45–60) | 0.990 3 |
| (Min–Max) | 39–64 | 36–67 |
| Frequency | Side | MacTel 2 Group (n:21) | Control Group (n:21) | p-Value |
|---|---|---|---|---|
| 500 | Right | 12.38 ± 5.62 | 9.04 ± 3.74 | 0.037 |
| Left | 15.00 ± 9.21 | 9.76 ± 4.60 | 0.045 | |
| Both | 13.69 ± 7.65 | 9.40 ± 4.16 | 0.005 | |
| 1000 | Right | 13.80 ± 8.35 | 8.57 ± 4.78 | 0.028 |
| Left | 15.00 ± 10.00 | 8.10 ± 4.60 | 0.014 | |
| Both | 14.40 ± 9.11 | 8.33 ± 4.64 | <0.001 | |
| 2000 | Right | 21.67 ± 16.07 | 9.76 ± 6.97 | 0.010 |
| Left | 24.76 ± 16.54 | 10.23 ± 6.01 | 0.002 | |
| Both | 23.21 ± 16.18 | 10.00 ± 6.43 | <0.001 | |
| 4000 | Right | 31.67 ± 17.70 | 18.81 ± 14.04 | 0.008 |
| Left | 36.67 ± 19.96 | 16.90 ± 12.09 | <0.001 | |
| Both | 34.17 ± 18.80 | 17.86 ± 12.97 | <0.001 | |
| 6000 | Right | 39.29 ± 21.93 | 21.43 ± 17.62 | 0.003 |
| Left | 42.86 ± 22.17 | 17.86 ± 15.21 | <0.001 | |
| Both | 41.07 ± 21.85 | 19.64 ± 16.35 | <0.001 | |
| 8000 | Right | 35.48 ± 22.85 | 19.29 ± 16.90 | 0.013 |
| Left | 42.14 ± 22.34 | 17.38 ± 14.97 | 0.001 | |
| Both | 38.81 ± 22.57 | 18.33 ± 15.79 | <0.001 | |
| 10,000 | Right | 46.19 ± 25.19 | 25.00 ± 13.60 | 0.008 |
| Left | 53.33 ± 27.49 | 25.71 ± 12.97 | 0.003 | |
| Both | 49.76 ± 26.29 | 25.35 ± 12.13 | <0.001 | |
| 12,000 | Right | 57.62 ± 23.32 | 34.76 ± 17.28 | 0.002 |
| Left | 60.95 ± 26.77 | 36.43 ± 15.82 | 0.004 | |
| Both | 59.29 ± 24.85 | 35.60 ± 16.38 | <0.001 | |
| 14,000 | Right | 64.29 ± 15.91 | 42.62 ± 15.13 | <0.001 |
| Left | 67.14 ± 17.72 | 43.10 ± 15.45 | <0.001 | |
| Both | 65.71 ± 16.69 | 42.86 ± 15.10 | <0.001 | |
| 16,000 | Right | 59.29 ± 17.05 | 43.57 ± 13.80 | 0.003 |
| Left | 61.90 ± 14.53 | 42.86 ± 14.88 | <0.001 | |
| Both | 60.60 ± 15.70 | 43.21 ± 14.17 | <0.001 |
| Variables | MacTel 2 Group (n:21) | Control Group (n:21) | p-Value |
|---|---|---|---|
| PTA4 better ear (dB HL), mean ± SD | 19.52 ± 10.51 | 10.77 ± 5.90 | 0.003 |
| Hearing loss (better ear > 20 dB) n (%) | 8 (38.1%) | 2 (9.5%) | 0.067 |
| Hearing loss (better ear > 25 dB) n (%) | 7 (33.3%) | 0 (0.0%) | 0.009 |
| PTA4 (worse ear), mean ± SD (dB HL) | 23.21 ± 11.76 | 12.02 ± 6.07 | 0.001 |
| Hearing loss (worse ear > 20 dB), n (%) | 9 (42.9%) | 2 (9.5%) | 0.032 |
| Hearing loss (worse ear > 25 dB), n (%) | 9 (42.9%) | 1 (4.8%) | 0.009 |
| WHO grade (better ear PTA4) | 0.077 | ||
| Normal (≤20) | 13 (61.9%) | 19 (90.5%) | |
| Mild (21–34) | 6 (28.6%) | 2 (9.5%) | |
| Moderate (35–49) | 2 (9.5%) | 0 (0.0%) | |
| Moderately severe (50–64) | 0 (0.0%) | 0 (0.0%) | |
| Severe (65–79) | 0 (0.0%) | 0 (0.0%) | |
| Profound (≥80) | 0 (0.0%) | 0 (0.0%) | |
| WHO grade (worse ear PTA4) | 0.027 | ||
| Normal | 12 (57.2%) | 19 (90.5%) | |
| Mild | 4 (19.0%) | 2 (9.5%) | |
| Moderate | 5 (23.8%) | 0 (0.0%) | |
| Moderately severe | 0 (0.0%) | 0 (0.0%) | |
| Severe | 0 (0.0%) | 0 (0.0%) | |
| Profound | 0 (0.0%) | 0 (0.0%) | |
| Hearing loss pattern (PTA4 > 25 dB) | 0.009 | ||
| None | 12 (57.2%) | 20 (95.2%) | |
| Unilateral | 2 (9.5%) | 1 (4.8%) | |
| Bilateral | 7 (33.3%) | 0 (0.0%) |
| Frequency | Side | MacTel 2 Group (n:21) | Control Group (n:21) | p-Value |
|---|---|---|---|---|
| Wave I | Right | 1.65 ± 0.19 | 1.57 ± 0.21 | 0.287 |
| Left | 1.68 ± 0.19 | 1.63 ± 0.18 | 0.344 | |
| Both | 1.66 ± 0.18 | 1.60 ± 0.19 | 0.158 | |
| Wave V | Right | 5.84 ± 0.33 | 5.89 ± 0.27 | 1.000 |
| Left | 5.97 ± 0.24 | 5.91 ± 0.45 | 0.746 | |
| Both | 5.90 ± 0.28 | 5.90 ± 0.36 | 0.793 | |
| Interpeak I–V | Right | 4.20 ± 0.33 | 4.26 ± 0.29 | 0.704 |
| Left | 4.29 ± 0.25 | 4.31 ± 0.25 | 0.799 | |
| Both | 4.24 ± 0.29 | 4.28 ± 0.27 | 0.715 |
| Frequency | Side | MacTel 2 Group (n:21) | Control Group (n:21) | p-Value |
|---|---|---|---|---|
| 500 | Right | 1.30 ± 3.60 | 8.00 ± 2.05 | <0.001 |
| Left | 1.11 ± 3.67 | 9.68 ± 2.10 | <0.001 | |
| Both | 1.21 ± 3.48 | 8.84 ± 1.56 | <0.001 | |
| 1000 | Right | 0.18 ± 3.79 | 8.71 ± 2.28 | <0.001 |
| Left | 0.08 ± 3.52 | 10.26 ± 2.03 | <0.001 | |
| Both | 0.13 ± 3.49 | 9.49 ± 1.79 | <0.001 | |
| 2000 | Right | −0.70 ± 4.60 | 9.49 ± 2.28 | <0.001 |
| Left | −0.04 ± 3.91 | 10.83 ± 2.33 | <0.001 | |
| Both | −0.37 ± 4.09 | 10.16 ± 1.95 | <0.001 | |
| 4000 | Right | −0.43 ± 4.79 | 9.63 ± 2.09 | <0.001 |
| Left | −1.10 ± 4.82 | 11.34 ± 2.69 | <0.001 | |
| Both | −0.77 ± 4.74 | 10.49 ± 1.87 | <0.001 | |
| 6000 | Right | −1.07 ± 4.12 | 10.76 ± 2.06 | <0.001 |
| Left | −0.84 ± 4.37 | 11.98 ± 3.46 | <0.001 | |
| Both | −0.95 ± 4.11 | 11.37 ± 1.98 | <0.001 | |
| 8000 | Right | −0.18 ± 3.12 | 10.07 ± 2.61 | <0.001 |
| Left | −0.22 ± 3.64 | 11.96 ± 2.74 | <0.001 | |
| Both | −0.20 ± 3.00 | 11.01 ± 1.99 | <0.001 |
| Frequency | Side | MacTel2 Absent n (%) | Control Absent n (%) | p-Value |
|---|---|---|---|---|
| 500 | Right | 20 (95.2%) | 2 (9.5%) | <0.001 |
| Left | 20 (95.2%) | 0 (0.0%) | <0.001 | |
| 1000 | Right | 20 (95.2%) | 0 (0.0%) | <0.001 |
| Left | 20 (95.2%) | 0 (0.0%) | <0.001 | |
| 2000 | Right | 20 (95.2%) | 0 (0.0%) | <0.001 |
| Left | 20 (95.2%) | 0 (0.0%) | <0.001 | |
| 4000 | Right | 18 (85.7%) | 0 (0.0%) | <0.001 |
| Left | 20 (95.2%) | 0 (0.0%) | <0.001 | |
| 6000 | Right | 19 (90.5%) | 0 (0.0%) | <0.001 |
| Left | 20 (95.2%) | 0 (0.0%) | <0.001 | |
| 8000 | Right | 21 (100.0%) | 0 (0.0%) | <0.001 |
| Left | 21 (100.0%) | 0 (0.0%) | <0.001 |
| Grade, n (%) | Right Eye | Left Eye |
|---|---|---|
| I | 4 (19.0%) | 3 (14.3%) |
| II | 8 (38.1%) | 12 (57.1%) |
| III | 8 (38.1%) | 5 (23.8%) |
| IV | 1 (4.8%) | 1 (4.8%) |
| Total | 21 (100%) | 21 (100%) |
| Domain | Variable | Side | Spearman Rho | p-Value |
|---|---|---|---|---|
| ABR | Wave V Both | Bilateral | −0.246 | 0.281 |
| Wave I Both | Bilateral | −0.225 | 0.325 | |
| Interpeak I–V Both | Bilateral | −0.032 | 0.889 | |
| DPOAE | DPOAE_500_Both | Bilateral | 0.031 | 0.893 |
| DPOAE_1000_Both | Bilateral | 0.087 | 0.705 | |
| DPOAE_2000_Both | Bilateral | 0.097 | 0.674 | |
| DPOAE_4000_Both | Bilateral | −0.041 | 0.857 | |
| DPOAE_6000_Both | Bilateral | 0.073 | 0.752 | |
| DPOAE_8000_Both | Bilateral | 0.161 | 0.485 | |
| PTA | PTA_500_Both | Bilateral | 0.171 | 0.457 |
| PTA_1000_Both | Bilateral | 0.240 | 0.294 | |
| PTA_2000_Both | Bilateral | 0.290 | 0.201 | |
| PTA_4000_Both | Bilateral | −0.181 | 0.431 | |
| PTA_6000_Both | Bilateral | 0.066 | 0.774 | |
| PTA_8000_Both | Bilateral | 0.005 | 0.981 | |
| PTA_10000_Both | Bilateral | 0.027 | 0.906 | |
| PTA_12000_Both | Bilateral | −0.075 | 0.744 | |
| PTA_14000_Both | Bilateral | −0.0967 | 0.676 | |
| PTA_16000_Both | Bilateral | −0.147 | 0.523 | |
| PTA4 | PTA4_Both | Overall/Bilateral | 0.124 | 0.591 |
| PTA4_Worse | Overall/Bilateral | 0.117 | 0.612 | |
| PTA4_Better | Overall/Bilateral | 0.106 | 0.644 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Yüksel, Y.; Yıldız, M.; Erol, M.K.; Sonbay Yılmaz, N.D.; Toslak, Y.S.; Ercanlı, U.; Cengiz Ünal, A.; Çetinkaya, E.A. Is Macular Telangiectasia Type 2 Associated with Hearing Loss and Cochlear Dysfunction? A Prospective Case–Control Study. Diagnostics 2026, 16, 767. https://doi.org/10.3390/diagnostics16050767
Yüksel Y, Yıldız M, Erol MK, Sonbay Yılmaz ND, Toslak YS, Ercanlı U, Cengiz Ünal A, Çetinkaya EA. Is Macular Telangiectasia Type 2 Associated with Hearing Loss and Cochlear Dysfunction? A Prospective Case–Control Study. Diagnostics. 2026; 16(5):767. https://doi.org/10.3390/diagnostics16050767
Chicago/Turabian StyleYüksel, Yeşim, Muhammet Yıldız, Muhammet Kazım Erol, Nevreste Didem Sonbay Yılmaz, Yusuf Sühan Toslak, Ufuk Ercanlı, Ayse Cengiz Ünal, and Erdem Atalay Çetinkaya. 2026. "Is Macular Telangiectasia Type 2 Associated with Hearing Loss and Cochlear Dysfunction? A Prospective Case–Control Study" Diagnostics 16, no. 5: 767. https://doi.org/10.3390/diagnostics16050767
APA StyleYüksel, Y., Yıldız, M., Erol, M. K., Sonbay Yılmaz, N. D., Toslak, Y. S., Ercanlı, U., Cengiz Ünal, A., & Çetinkaya, E. A. (2026). Is Macular Telangiectasia Type 2 Associated with Hearing Loss and Cochlear Dysfunction? A Prospective Case–Control Study. Diagnostics, 16(5), 767. https://doi.org/10.3390/diagnostics16050767






