Gliflozins in Practice: Real-Life Use of Dapagliflozin and Empagliflozin in HFrEF Versus Clinical Trial Data
Abstract
1. Introduction
2. Materials and Methods
Statistical Analyses
3. Results
3.1. Comparison of Real-World Cohorts
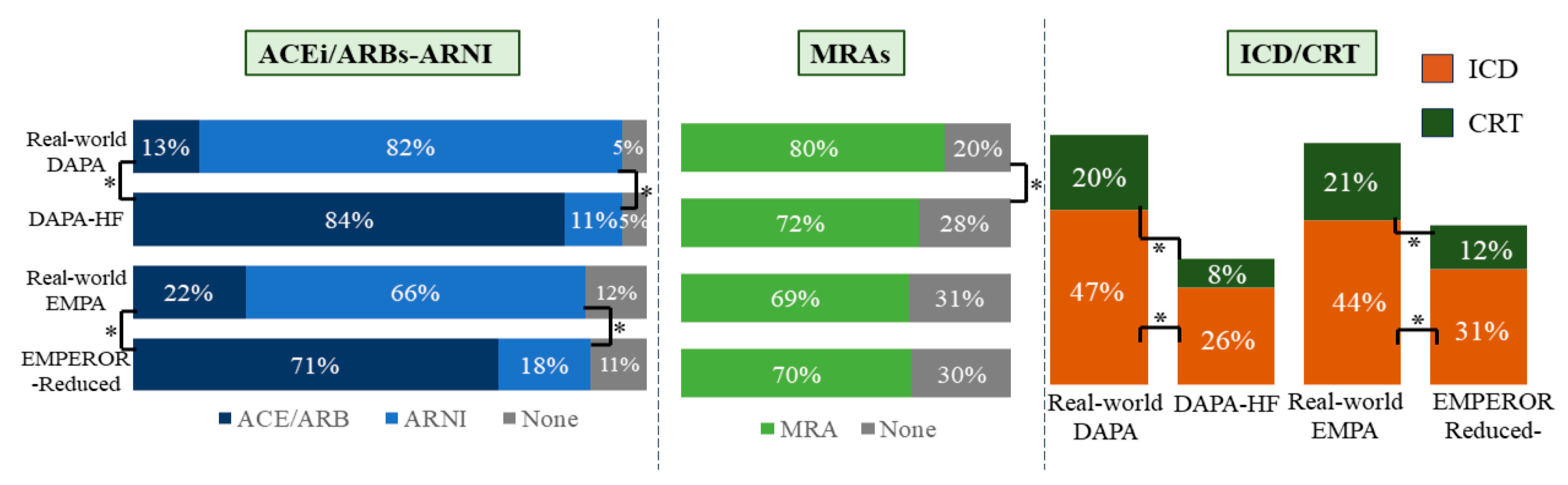
3.2. Real-World Cohorts Compared to Trials
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Preda, A.; Montecucco, F.; Carbone, F.; Camici, G.G.; Lüscher, T.F.; Kraler, S.; Liberale, L. SGLT2 inhibitors: From glucose-lowering to cardiovascular benefits. Cardiovasc. Res. 2024, 120, 443–460. [Google Scholar] [CrossRef]
- Mapelli, M.; Mattavelli, I.; Salvioni, E.; Capra, N.; Mantegazza, V.; Garlasche, A.; Campodonico, J.; Rubbo, F.M.; Paganin, C.; Capovilla, T.M.; et al. Dapagliflozin effects on exercise, cardiac remodeling, biomarkers, and renal and pulmonary function in heart failure patients: Not as good as expected? Front. Cardiovasc. Med. 2025, 12, 1542870. [Google Scholar] [CrossRef]
- McMurray, J.J.; Solomon, S.D.; Inzucchi, S.E.; Køber, L.; Kosiborod, M.N.; Martinez, F.A.; Ponikowski, P.; Sabatine, M.S.; Anand, I.S.; Bělohlávek, J.; et al. Dapagliflozin in Patients with Heart Failure and Reduced Ejection Fraction. N. Engl. J. Med. 2019, 381, 1995–2008. [Google Scholar] [CrossRef] [PubMed]
- Packer, M.; Anker, S.D.; Butler, J.; Filippatos, G.; Ferreira, J.P.; Pocock, S.J.; Carson, P.; Anand, I.; Doehner, W.; Haass, M.; et al. Effect of Empagliflozin on the Clinical Stability of Patients with Heart Failure and a Reduced Ejection Fraction: The EMPEROR-Reduced Trial. Circulation 2021, 143, 326–336. [Google Scholar] [CrossRef] [PubMed]
- Solomon, S.D.; McMurray, J.J.; Claggett, B.; de Boer, R.A.; DeMets, D.; Hernandez, A.F.; Inzucchi, S.E.; Kosiborod, M.N.; Lam, C.S.; Martinez, F.; et al. Dapagliflozin in Heart Failure with Mildly Reduced or Preserved Ejection Fraction. N. Engl. J. Med. 2022, 387, 1089–1098. [Google Scholar] [CrossRef] [PubMed]
- Anker, S.D.; Butler, J.; Filippatos, G.; Ferreira, J.P.; Bocchi, E.; Böhm, M.; Brunner–La Rocca, H.P.; Choi, D.J.; Chopra, V.; Chuquiure-Valenzuela, E.; et al. Empagliflozin in Heart Failure with a Preserved Ejection Fraction. N. Engl. J. Med. 2021, 385, 1451–1461. [Google Scholar] [CrossRef]
- McDonagh, T.A.; Metra, M.; Adamo, M.; Gardner, R.S.; Baumbach, A.; Böhm, M.; Burri, H.; Butler, J.; Čelutkienė, J.; Chioncel, O.; et al. 2023 Focused Update of the 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur. Heart J. 2023, 44, 3627–3639. [Google Scholar] [CrossRef]
- McDonagh, T.A. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur. Heart J. 2021, 42, 3599–3726. [Google Scholar] [CrossRef]
- Agostoni, P.; Corrà, U.; Cattadori, G.; Veglia, F.; La Gioia, R.; Scardovi, A.B.; Emdin, M.; Metra, M.; Sinagra, G.; Limongelli, G.; et al. Metabolic exercise test data combined with cardiac and kidney indexes, the MECKI score: A multiparametric approach to heart failure prognosis. Int. J. Cardiol. 2013, 167, 2710–2718. [Google Scholar] [CrossRef]
- Harris, P.A.; Taylor, R.; Thielke, R.; Payne, J.; Gonzalez, N.; Conde, J.G. Research electronic data capture (REDCap)—A metadata-driven methodology and workflow process for providing translational research informatics support. J. Biomed. Inform. 2009, 42, 377–381. [Google Scholar] [CrossRef]
- Harris, P.A.; Taylor, R.; Minor, B.L.; Elliott, V.; Fernandez, M.; O’Neal, L.; McLeod, L.; Delacqua, G.; Delacqua, F.; Kirby, J.; et al. The REDCap consortium: Building an international community of software platform partners. J. Biomed. Inform. 2019, 95, 103208. [Google Scholar] [CrossRef]
- Pabel, S.; Hamdani, N.; Luedde, M.; Sossalla, S. SGLT2 Inhibitors and Their Mode of Action in Heart Failure-Has the Mystery Been Unravelled? Curr. Heart Fail Rep. 2021, 18, 315–328. [Google Scholar] [CrossRef] [PubMed]
- Modzelewski, K.L.; Pipilas, A.; Bosch, N.A. Comparative Outcomes of Empagliflozin to Dapagliflozin in Patients with Heart Failure. JAMA Netw. Open 2024, 7, e249305. [Google Scholar] [CrossRef]
- Kocabas, U.; Ergin, I.; Yavuz, V.; Altın, C.; Kaplan, M.; Yılmaz Öztekin, G.M.; Doğduş, M.; Murat, S.; Murat, B.; Kıvrak, T.; et al. Real-world data on Empagliflozin and Dapagliflozin use in patients with HEART failure: The RED-HEART study. ESC Heart Fail. 2025, 12, 434–446. [Google Scholar] [CrossRef] [PubMed]
- Becher, P.M.; Schrage, B.; Ferrannini, G.; Benson, L.; Butler, J.; Carrero, J.J.; Cosentino, F.; Dahlström, U.; Mellbin, L.; Rosano, G.M.; et al. Use of sodium-glucose co-transporter 2 inhibitors in patients with heart failure and type 2 diabetes mellitus: Data from the Swedish Heart Failure Registry. Eur. J. Heart Fail. 2021, 23, 1012–1022. [Google Scholar] [CrossRef]
- Adhikari, R.; Jha, K.; Dardari, Z.; Heyward, J.; Blumenthal, R.S.; Eckel, R.H.; Alexander, G.C.; Blaha, M.J. National Trends in Use of Sodium-Glucose Cotransporter-2 Inhibitors and Glucagon-like Peptide-1 Receptor Agonists by Cardiologists and Other Specialties, 2015 to 2020. J. Am. Heart Assoc. 2022, 11, e023811. [Google Scholar] [CrossRef] [PubMed]
- Brax, H.; Fadlallah, R.; Al-Khaled, L.; Kahale, L.A.; Nas, H.; El-Jardali, F.; Akl, E.A. Association between physicians’ interaction with pharmaceutical companies and their clinical practices: A systematic review and meta-analysis. PLoS ONE 2017, 12, e0175493. [Google Scholar] [CrossRef]
- DeJong, C.; Inoue, K.; Durstenfeld, M.S.; Agarwal, A.; Chen, J.C.; Tseng, C.W.; Dudley, R.A.; Hsue, P.Y.; Kazi, D.S. Direct-to-Physician Marketing and Uptake of Optimal Medical Therapy for Heart Failure with Reduced Ejection Fraction. JACC Heart Fail. 2025, 13, 102380. [Google Scholar] [CrossRef]
- Johansson, I.; Dahlström, U.; Edner, M.; Näsman, P.; Ryden, L.; Norhammar, A. Prognostic Implications of Type 2 Diabetes Mellitus in Ischemic and Nonischemic Heart Failure. J. Am. Coll. Cardiol. 2016, 68, 1404–1416. [Google Scholar] [CrossRef]
- Paolillo, S.; Salvioni, E.; Filardi, P.P.; Bonomi, A.; Sinagra, G.; Gentile, P.; Gargiulo, P.; Scoccia, A.; Cosentino, N.; Gugliandolo, P.; et al. Long-term prognostic role of diabetes mellitus and glycemic control in heart failure patients with reduced ejection fraction: Insights from the MECKI Score database. Int. J. Cardiol. 2020, 317, 103–110. [Google Scholar] [CrossRef]
- Zinman, B.; Wanner, C.; Lachin, J.M.; Fitchett, D.; Bluhmki, E.; Hantel, S.; Mattheus, M.; Devins, T.; Johansen, O.E.; Woerle, H.J.; et al. Empagliflozin, Cardiovascular Outcomes, and Mortality in Type 2 Diabetes. N. Engl. J. Med. 2015, 373, 2117–2128. [Google Scholar] [CrossRef] [PubMed]
- Wiviott, S.D.; Raz, I.; Bonaca, M.P.; Mosenzon, O.; Kato, E.T.; Cahn, A.; Silverman, M.G.; Zelniker, T.A.; Kuder, J.F.; Murphy, S.A.; et al. Dapagliflozin and Cardiovascular Outcomes in Type 2 Diabetes. N. Engl. J. Med. 2019, 380, 347–357. [Google Scholar] [CrossRef] [PubMed]
- Zelniker, T.A.; Wiviott, S.D.; Raz, I.; Im, K.; Goodrich, E.L.; Bonaca, M.P.; Mosenzon, O.; Kato, E.T.; Cahn, A.; Furtado, R.H.; et al. SGLT2 inhibitors for primary and secondary prevention of cardiovascular and renal outcomes in type 2 diabetes: A systematic review and meta-analysis of cardiovascular outcome trials. Lancet 2019, 393, 31–39. [Google Scholar] [CrossRef]
- Engström, A.; Söderling, J.; Hviid, A.; Eliasson, B.; Gudbjörnsdottir, S.; Wintzell, V.; Hveem, K.; Jonasson, C.; Melbye, M.; Pasternak, B.; et al. Comparative cardiovascular and renal effectiveness of empagliflozin and dapagliflozin: Scandinavian cohort study. Eur. Heart J. Cardiovasc. Pharmacother. 2024, 10, 432–443. [Google Scholar] [CrossRef]
- Savarese, G.; Bodegard, J.; Norhammar, A.; Sartipy, P.; Thuresson, M.; Cowie, M.R.; Fonarow, G.C.; Vaduganathan, M.; Coats, A.J. Heart failure drug titration, discontinuation, mortality and heart failure hospitalization risk: A multinational observational study (US, UK and Sweden). Eur. J. Heart Fail. 2021, 23, 1499–1511. [Google Scholar] [CrossRef] [PubMed]



| Real-Life DAPA | Real-Life EMPA | p-Value | ||||
|---|---|---|---|---|---|---|
| n | media ± SD, n (%) | n | media ± SD, n (%) | |||
| Age (years) | 276 | 70 ± 12.6 | 94 | 73 ± 11 | 0.014 | |
| Sex | 276 | 94 | ||||
| Male | 200 (72.4%) | 70 (74%) | 0.705 | |||
| Female | 76 (27.6%) | 24 (26%) | ||||
| BMI (kg/m2) | 250 | 26.3 ± 4.2 | 82 | 27 ± 4 | 0.588 | |
| NYHA | 262 | 92 | 0.033 | |||
| I | 28 (11%) | 8 (9%) | ||||
| II | 187 (71%) | 55 (60%) | ||||
| III | 46 (17.6%) | 29 (31%) | ||||
| IV | 1 (0.4%) | |||||
| Etiology | 276 | 94 | 0.2824 | |||
| Ischemic | 133 (48%) | 52 (55%) | ||||
| Non Ischemic | 143 (52%) | 42 (44%) | ||||
| HFimpEF (n) | 31 (11%) | 21 (16%) | 0.1460 | |||
| LVEF (%) | 267 | 32 ± 8 | 85 | 34 ± 8 | 0.015 | |
| EDV (mL) | 225 | 183 (145–234) | 69 | 157 (118–202) | 0.003 | |
| ESV (mL) | 225 | 122 (90–168) | 68 | 105 (73–133) | 0.002 | |
| PASP (mmHg) | 210 | 32 (26–40) | 66 | 38 (29–47) | 0.003 | |
| SBP (mmHg) | 253 | 120 ± 17 | 76 | 121 ± 17 | 0.93 | |
| Heart rate (bpm) | 217 | 69 ± 14 | 74 | 69 ± 15 | 0.92 | |
| Hemoglobin (g/dL) | 243 | 13.7 (13–15) | 87 | 12.6 (11.1–15.5) | 0.004 | |
| Hematocrit (%) | 180 | 41 (37–44) | 62 | 38 (33–42) | 0.005 | |
| Creatinine (mg/dL) | 256 | 1.25 ± 0.7 | 89 | 1.4 ± 0.6 | 0.043 | |
| EGFR (mL/min) | 256 | 62 ± 22 | 89 | 59 ± 61 | 0.65 | |
| NT-proBNP (pg/mL) | 157 | 1047 (482–2559) | 41 | 1393 (507–3216) | 0.415 | |
| Diabetes | 273 | 42 (15%) | 94 | 27 (29%) | <0.001 | |
| Hba1c (%) | 93 | 6.6 ± 4.7 | 21 | 11 ± 14 | 0.207 | |
| PM | 273 | 43 (16%) | 94 | 11 (12%) | 0.339 | |
| ICD | 274 | 130 (47%) | 94 | 41 (44%) | 0.521 | |
| CRT | 275 | 54 (20%) | 94 | 20 (21%) | 0.732 | |
| ACE | 273 | 20 (7%) | 93 | 10 (11%) | 0.298 | |
| ARB | 272 | 16 (6%) | 93 | 10 (11%) | 0.115 | |
| Sacubitril/Valsartan | 274 | 224 (82%) | 93 | 61 (66%) | 0.001 | |
| Beta-blocker | 275 | 264 (96%) | 94 | 89 (95%) | 0.588 | |
| Loop diuretics | 274 | 193 (70.4%) | 94 | 73 (78%) | 0.177 | |
| Statin | 275 | 165 (60%) | 94 | 65 (69%) | 0.114 | |
| MRA | 274 | 217 (79%) | 94 | 65 (69%) | 0.047 | |
| Antiplatelet | 273 | 126 (46%) | 94 | 54 (57%) | 0.059 | |
| Anticoagulation therapy | 274 | 120 (44%) | 94 | 37 (39%) | 0.453 | |
| Digitalis | 273 | 21 (8%) | 94 | 5 (5%) | 0.439 | |
| Amiodarone | 274 | 105 (38%) | 94 | 32 (34%) | 0.459 | |
| Oral antidiabetic | 275 | 21 (8%) | 94 | 16 (17%) | 0.009 | |
| Insulin | 275 | 9 (3%) | 94 | 13 (14%) | <0.001 | |
| Creatinine at follow-up (mg/dL) | 190 | 1.3 ± 0.5 | 61 | 1.5 ± 0.7 | 0.015 | |
| EGFR at follow-up (mL/min) | 184 | 55 ± 21 | 59 | 50 ±24 | 0.1259 | |
| Composite outcome | 276 | 67 (24%) | 94 | 21 (22%) | 0.7039 | |
| Real-Life DAPA | DAPA HF | p-Value | Real-Life EMPA | EMPEROR | p-Value | ||
|---|---|---|---|---|---|---|---|
| n = 276 | n = 2373 | n = 94 | n = 1863 | ||||
| Age(years) | 70 ± 13 | 66 ± 11 | <0.0001 | 73 ± 11 | 67 ± 11 | <0.0001 | |
| Sex | Male | 200 (72.4%) | 1809 (76%) | 0.1662 | 70 (74%) | 1426(76.5%) | 0.6437 |
| Female | 76(27.6%) | 564 (23.8%) | 24 (26%) | 437 (23.5%) | |||
| BMI(kg/m2) | 26 ± 4 | 28 ± 6 | <0.0001 | 27 ± 4 | 28 ± 6 | 0.104 | |
| NYHA | I | 28 (11%) | 0 | <0.0001 | 8 (9%) | 0 | <0.0001 |
| II | 187 (71%) | 1606 (67.7%) | 55 (60%) | 1399 (75.1%) | |||
| III | 46 (17.6%) | 747 (31.5%) | 29 (31%) | 455 (24.4%) | |||
| IV | 1 (0.4%) | 20 (0.8%) | 9 (0.5%) | ||||
| Etiology | Ischemic | 133 (48%) | 1316 (55.5%) | 0.0252 | 52 (55%) | 983 (52.8) | 0.6724 |
| Non Ischemic | 143 (52%) | 1057 (46%) | 42 (44%) | 880 (47.2) | |||
| LVEF(%) | 32 ± 8 | 31.2 ± 6.7 | 0.070 | 34 ± 8 | 28 ± 6.0 | <0.0001 | |
| SBP (mmHg) | 120 ± 17 | 122.0 ± 16.3 | 0.064 | 121 ± 17 | 122.6 ± 15.9 | 0.391 | |
| Heart rate(bpm) | 69 ± 14 | 71.5 ± 11.6 | 0.003 | 69 ± 15 | 71.0 ± 11.7 | 0.154 | |
| EGFR (mL/min) | 62 ± 22 | 66.0 ± 19.6 | 0.0053 | 59 ± 61 | 61.8 ± 21.7 | 0.129 | |
| NT-proBNP (pg/mL) | 1047 (482-2559) | 1428 (857–2655) | <0.0001 | 1393 (507-3216) | 1887 (1077–3429) | 0.0062 | |
| ICD | 130 (47%) | 622 (26%) | <0.0001 | 41 (44%) | 578 (31%) | 0.0104 | |
| CRT | 54 (20%) | 190 (8%) | <0.0001 | 20 (21%) | 220 (12%) | 0.0063 | |
| ACE | 20 (7%) | 1332 (56.1%) | <0.0001 | 10 (11%) | |||
| ARB | 16 (6%) | 675 (28%) | <0.0001 | 10 (11%) | 1314 (71%) | <0.0001 | |
| Sacubitril/Valsartan | 224 (82%) | 250 (10.5%) | <0.0001 | 61 (66%) | 340 (18%) | <0.0001 | |
| Bblocker | 264 (96%) | 2278 (96%) | 0.9978 | 89 (95%) | 1765 (95%) | 1 | |
| Diuretics | 193 (70.4 %) | 2216 (93.4%) | <0.0001 | 73 (78%) | |||
| MRA | 217 (79%) | 1696 (71.5%) | 0.0068 | 65 (69%) | 1306 (70%) | 0.844 | |
| Insulin | 9 (3%) | 274/993 (27.6%) | <0.0001 | 13 (14%) | 664 (35.6) | <0.0001 | |
| Diabetes | 42 (15%) | 993 (41.8%) | <0.0001 | 27 (29%) | 577 (31.0) | 0.6452 | |
| History of HF hospitalisation | 165 (60%) | 1124 (47.4%) | <0.0001 | 84 (89%) | 664 (35.6) | <0.0001 | |
| Discontinuation | 15 (6%) | 249 (11%) | 0.0183 | 9 (11%) | |||
| Adverse event | Renal adverse event | 3 (21%) | 153 (97.5%) | <0.0001 | 1 (10%) | ||
| Major hypoglycemia | 1 (7%) | 4 (2.5%) | |||||
| Other | 10 (71%) | 0 | 8 (90%) |
| Real-Life DAPA | DAPA HF | p-Value | Real-Life EMPA | EMPEROR | p-Value | ||
|---|---|---|---|---|---|---|---|
| n = 276 | n = 2373 | n = 94 | n = 1863 | ||||
| HF hospitalization | 50 (19.6%) | 237 (11.5%) | <0.0001 | 16 (19%) | 246 (13.2) | 0.1376 | |
| Death | 22 (8%) | 503 (21%) | <0.0001 | 8 (9%) | 436 (23%) | 0.00013 | |
| Death cause | 0.6969 | 1 | |||||
| CV death | 9 (41%) | 227 (45%) | 3 (38%) | 187 (43%) | |||
| Other | 13 (59%) | 276 (55%) | 5 (62%) | 249 (57%) | |||
| Composite outcome | 67 (24%) | 386 (16%) | 0.0004 | 21 (22%) | 361 (19%) | 0.479 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Mapelli, M.; Caputo, R.; Valenti, M.; Rubbo, F.M.; Salvioni, E.; Mattavelli, I.; Galotta, A.; Piotti, A.; Puttini, F.; Manfrin, L.; et al. Gliflozins in Practice: Real-Life Use of Dapagliflozin and Empagliflozin in HFrEF Versus Clinical Trial Data. Diagnostics 2026, 16, 769. https://doi.org/10.3390/diagnostics16050769
Mapelli M, Caputo R, Valenti M, Rubbo FM, Salvioni E, Mattavelli I, Galotta A, Piotti A, Puttini F, Manfrin L, et al. Gliflozins in Practice: Real-Life Use of Dapagliflozin and Empagliflozin in HFrEF Versus Clinical Trial Data. Diagnostics. 2026; 16(5):769. https://doi.org/10.3390/diagnostics16050769
Chicago/Turabian StyleMapelli, Massimo, Rebecca Caputo, Massimo Valenti, Filippo Maria Rubbo, Elisabetta Salvioni, Irene Mattavelli, Arianna Galotta, Arianna Piotti, Fiorella Puttini, Laura Manfrin, and et al. 2026. "Gliflozins in Practice: Real-Life Use of Dapagliflozin and Empagliflozin in HFrEF Versus Clinical Trial Data" Diagnostics 16, no. 5: 769. https://doi.org/10.3390/diagnostics16050769
APA StyleMapelli, M., Caputo, R., Valenti, M., Rubbo, F. M., Salvioni, E., Mattavelli, I., Galotta, A., Piotti, A., Puttini, F., Manfrin, L., Vignati, C., Costantino, S., & Agostoni, P. (2026). Gliflozins in Practice: Real-Life Use of Dapagliflozin and Empagliflozin in HFrEF Versus Clinical Trial Data. Diagnostics, 16(5), 769. https://doi.org/10.3390/diagnostics16050769






