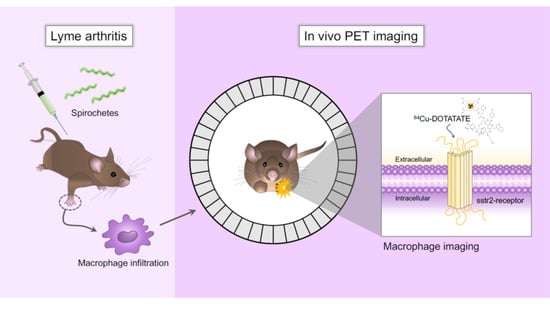
64Cu-DOTATATE Positron Emission Tomography (PET) of Borrelia Burgdorferi Infection: In Vivo Imaging of Macrophages in Experimental Model of Lyme Arthritis
Abstract
1. Introduction
2. Materials and Methods
2.1. Mice
2.2. Induction of Bb Infection
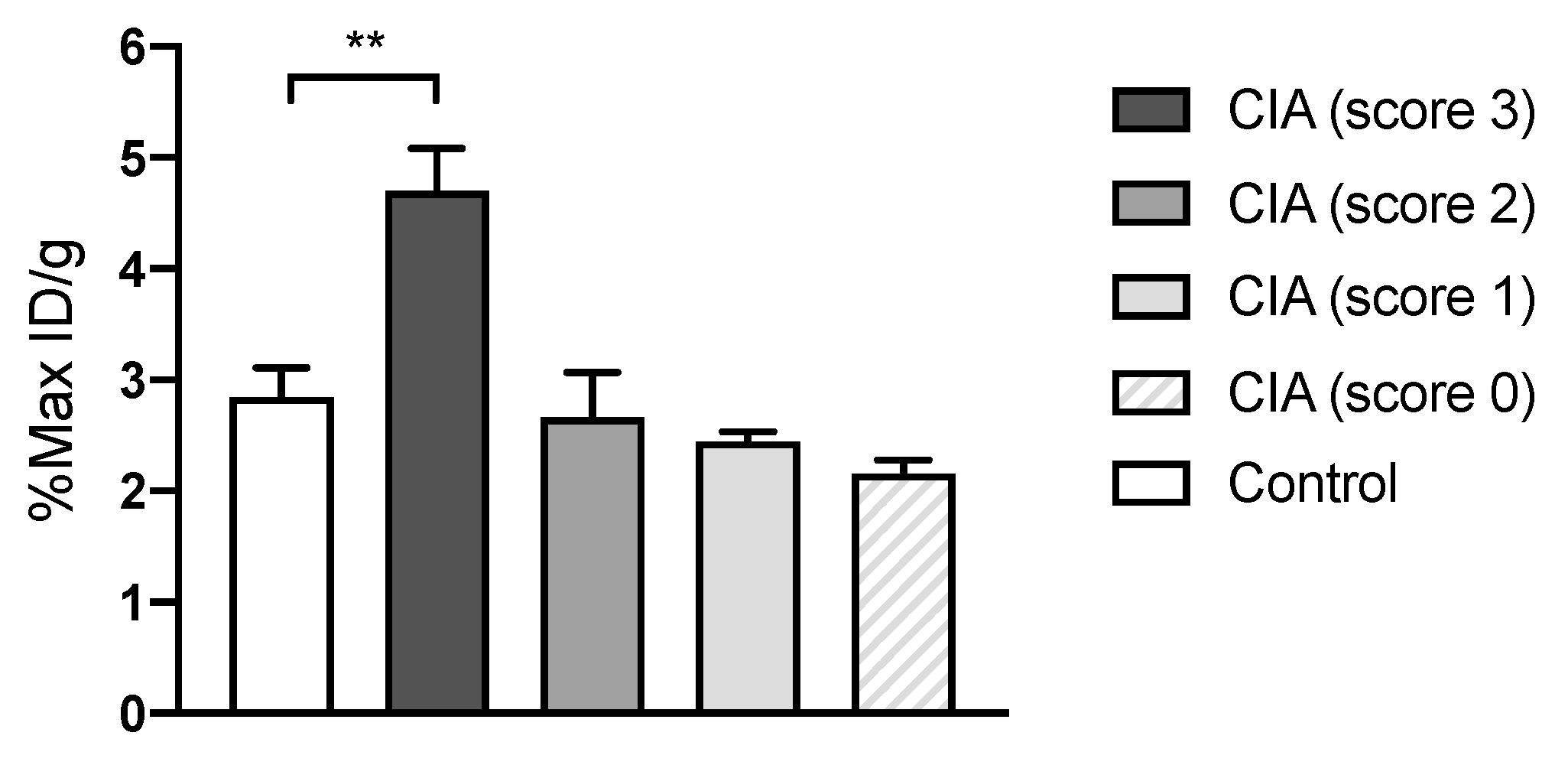
2.3. Collagen-Induced Arthritis
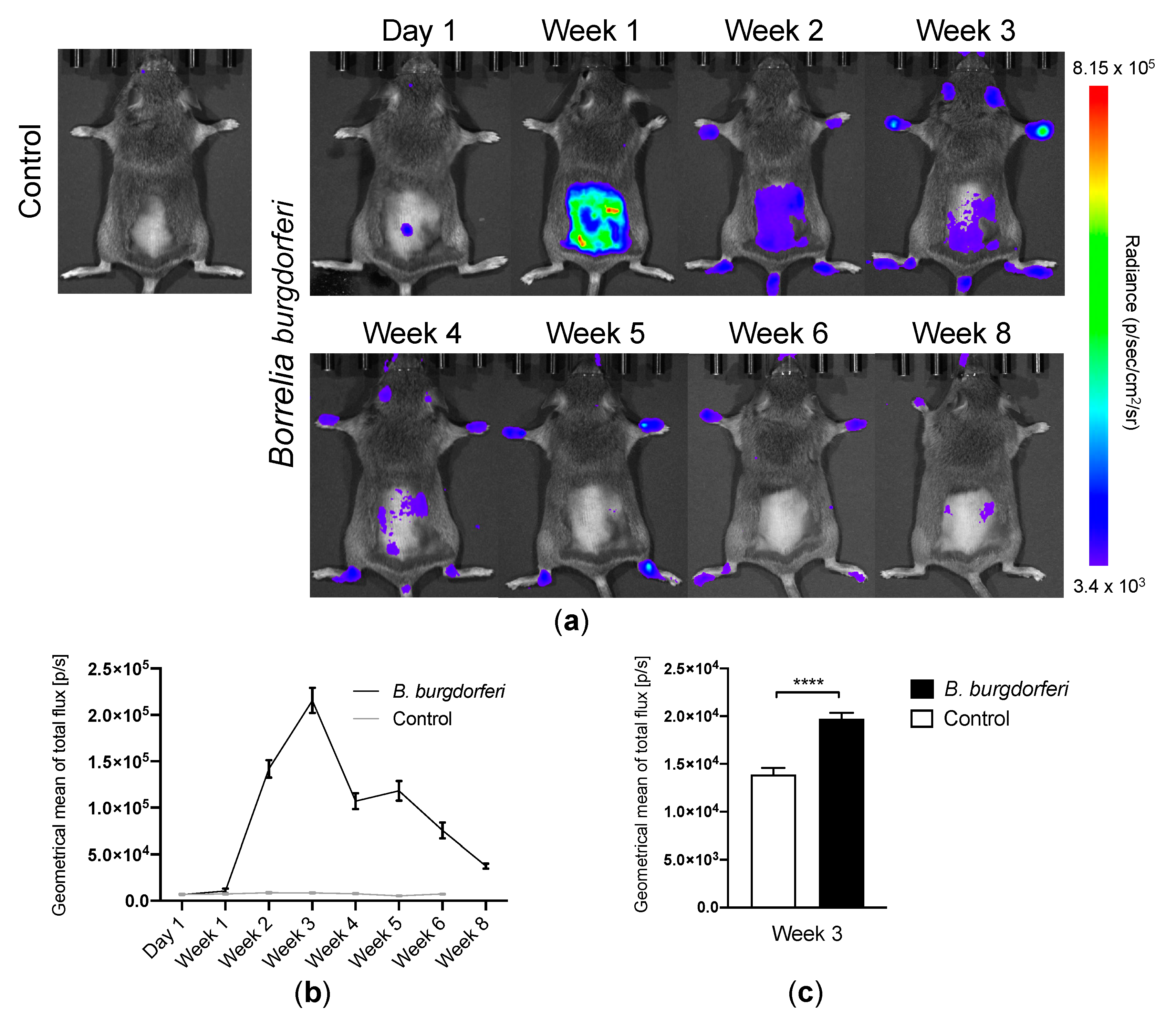
2.4. In Vivo Bb Bioluminescence Imaging
2.5. Synthesis and Radiolabeling of [64Cu]Cu-DOTATATE
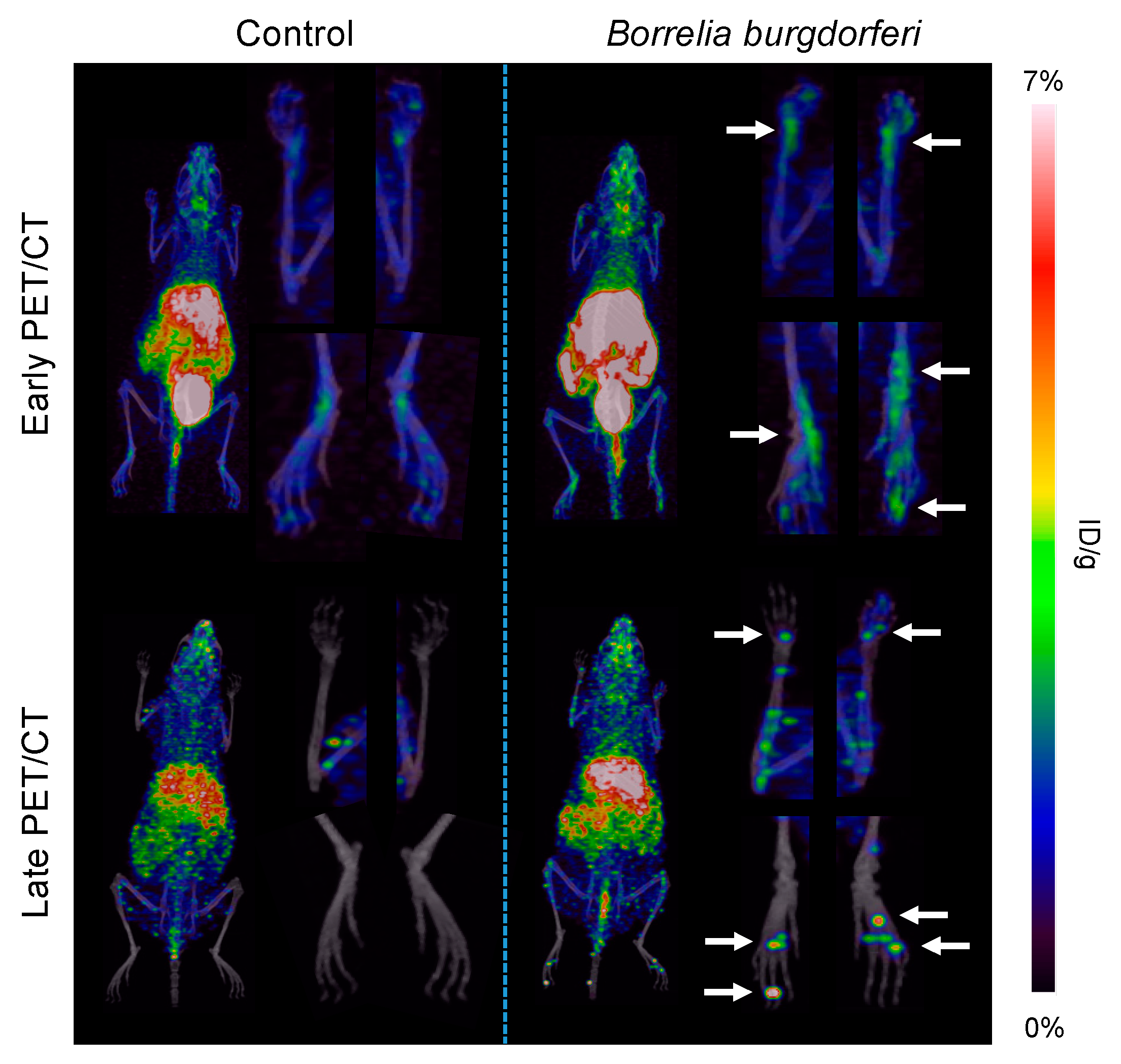
2.6. PET/CT Imaging Acquisition and Analysis
2.7. CD163 Enzyme-Linked Immunosorbent Assay
2.8. Statistical Analysis
3. Results
3.1. Bb Infection and Migration of Spirochetes to the Joints
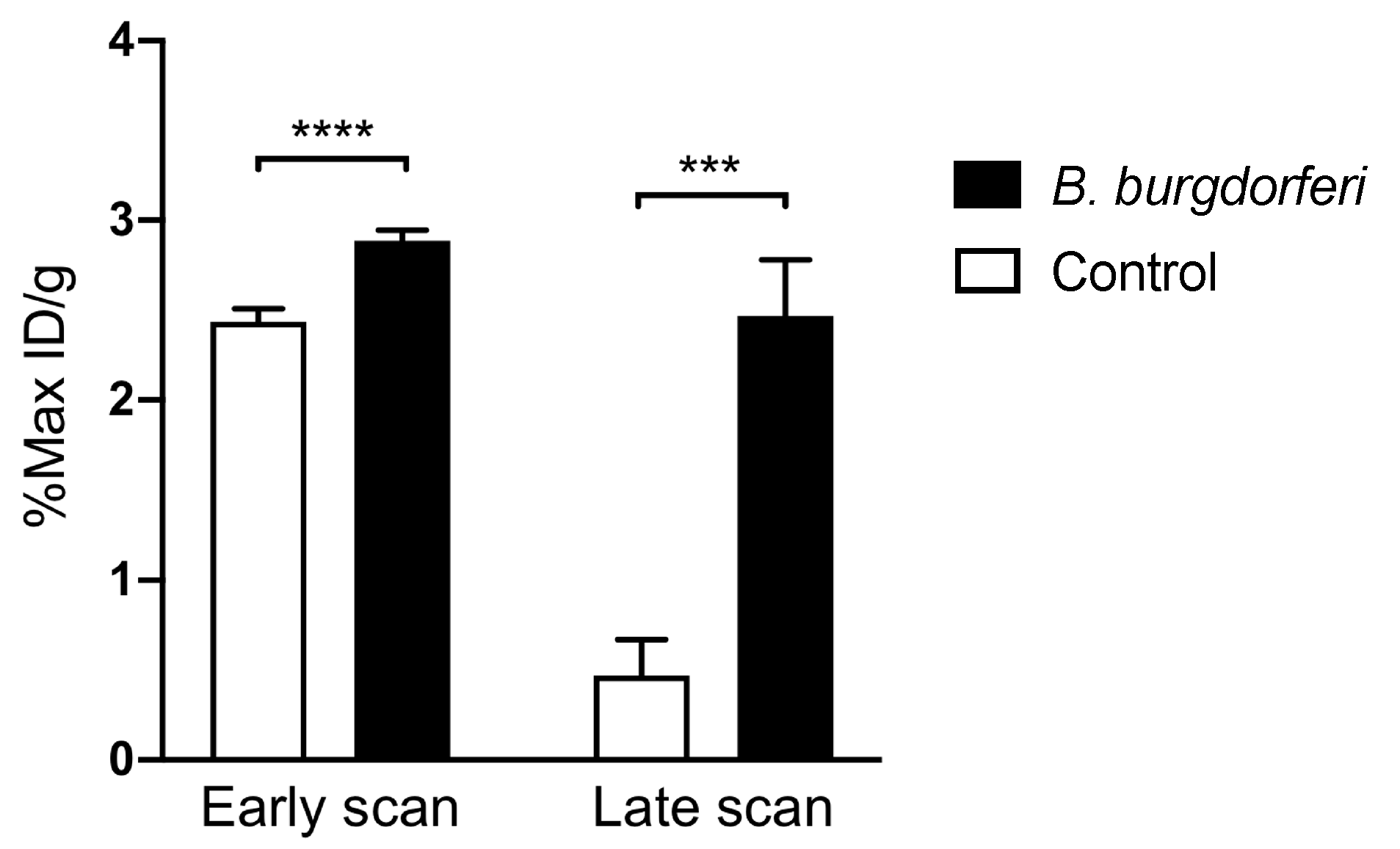
3.2. [64Cu]Cu-DOTATATE PET/CT Imaging of Macrophages in CIA and Lyme Arthritis
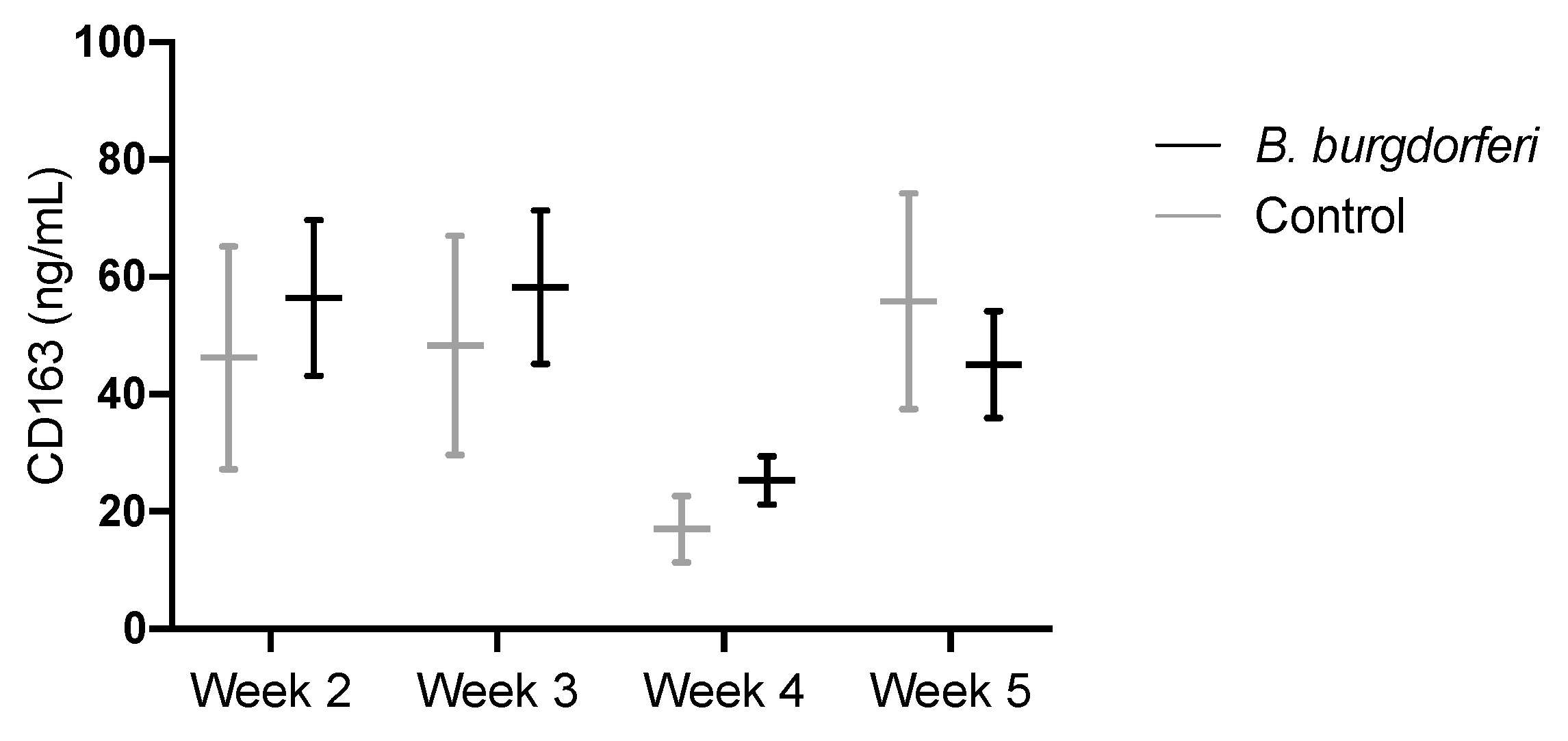
3.3. CD163 as a Circulating Marker for Macrophages
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Stanek, G.; Strle, F. Lyme borreliosis-from tick bite to diagnosis and treatment. FEMS Microbiol. Rev. 2018, 42, 233–258. [Google Scholar] [CrossRef] [PubMed]
- Steere, A.C.; Strle, F.; Wormser, G.P.; Hu, L.T.; Branda, J.A.; Hovius, J.W.; Li, X.; Mead, P.S. Lyme borreliosis. Nat. Rev. Dis. Primers 2016, 2, 16090. [Google Scholar] [CrossRef] [PubMed]
- Steere, A.C.; Glickstein, L. Elucidation of Lyme arthritis. Nat. Rev. Immunol. 2004, 4, 143–152. [Google Scholar] [CrossRef] [PubMed]
- Dessau, R.B.; van Dam, A.P.; Fingerle, V.; Gray, J.; Hovius, J.W.; Hunfeld, K.P.; Jaulhac, B.; Kahl, O.; Kristoferitsch, W.; Lindgren, P.E.; et al. To test or not to test? Laboratory support for the diagnosis of Lyme borreliosis: A position paper of ESGBOR, the ESCMID study group for Lyme borreliosis. Clin. Microbiol. Infect. 2018, 24, 118–124. [Google Scholar] [CrossRef]
- Obel, N.; Dessau, R.B.; Krogfelt, K.A.; Bodilsen, J.; Andersen, N.S.; Moller, J.K.; Roed, C.; Omland, L.H.; Christiansen, C.B.; Ellermann-Eriksen, S.; et al. Long term survival, health, social functioning, and education in patients with European Lyme neuroborreliosis: Nationwide population based cohort study. BMJ 2018, 361. [Google Scholar] [CrossRef]
- Raffetin, A.; Saunier, A.; Bouiller, K.; Caraux-Paz, P.; Eldin, C.; Gallien, S.; Jouenne, R.; Belkacem, A.; Salomon, J.; Patey, O.; et al. Unconventional diagnostic tests for Lyme borreliosis: A systematic review. Clin. Microbiol. Infect. 2020, 26, 51–59. [Google Scholar] [CrossRef]
- Shapiro, E.D.; Wormser, G.P. Lyme Disease in 2018: What Is New (and What Is Not). JAMA 2018, 320, 635–636. [Google Scholar] [CrossRef]
- Strle, K.; Drouin, E.E.; Shen, S.; El Khoury, J.; McHugh, G.; Ruzic-Sabljic, E.; Strle, F.; Steere, A.C. Borrelia burgdorferi stimulates macrophages to secrete higher levels of cytokines and chemokines than Borrelia afzelii or Borrelia garinii. J. Infect. Dis. 2009, 200, 1936–1943. [Google Scholar] [CrossRef]
- Lasky, C.E.; Olson, R.M.; Brown, C.R. Macrophage Polarization during Murine Lyme Borreliosis. Infect Immun. 2015, 83, 2627–2635. [Google Scholar] [CrossRef]
- Ramesh, G.; Didier, P.J.; England, J.D.; Santana-Gould, L.; Doyle-Meyers, L.A.; Martin, D.S.; Jacobs, M.B.; Philipp, M.T. Inflammation in the pathogenesis of lyme neuroborreliosis. Am. J. Pathol. 2015, 185, 1344–1360. [Google Scholar] [CrossRef]
- Eiffert, H.; Karsten, A.; Schlott, T.; Ohlenbusch, A.; Laskawi, R.; Hoppert, M.; Christen, H.J. Acute peripheral facial palsy in Lyme disease—A distal neuritis at the infection site. Neuropediatrics 2004, 35, 267–273. [Google Scholar] [CrossRef] [PubMed]
- Wooten, R.M.; Weis, J.J. Host-pathogen interactions promoting inflammatory Lyme arthritis: Use of mouse models for dissection of disease processes. Curr. Opin. Microbiol. 2001, 4, 274–279. [Google Scholar] [CrossRef]
- Xu, Q.; Seemanapalli, S.V.; Reif, K.E.; Brown, C.R.; Liang, F.T. Increasing the recruitment of neutrophils to the site of infection dramatically attenuates Borrelia burgdorferi infectivity. J. Immunol. 2007, 178, 5109–5115. [Google Scholar] [CrossRef] [PubMed]
- Dalm, V.A.; van Hagen, P.M.; van Koetsveld, P.M.; Achilefu, S.; Houtsmuller, A.B.; Pols, D.H.; van der Lely, A.J.; Lamberts, S.W.; Hofland, L.J. Expression of somatostatin, cortistatin, and somatostatin receptors in human monocytes, macrophages, and dendritic cells. Am. J. Physiol. Endocrinol. Metab. 2003, 285, E344–E353. [Google Scholar] [CrossRef] [PubMed]
- Perez, J.; Viollet, C.; Doublier, S.; Videau, C.; Epelbaum, J.; Baud, L. Somatostatin binds to murine macrophages through two distinct subsets of receptors. J. Neuroimmunol. 2003, 138, 38–44. [Google Scholar] [CrossRef]
- Courtenay, J.S.; Dallman, M.J.; Dayan, A.D.; Martin, A.; Mosedale, B. Immunisation against heterologous type II collagen induces arthritis in mice. Nature 1980, 283, 666–668. [Google Scholar] [CrossRef] [PubMed]
- Tseng, J.C.; Kung, A.L. In vivo imaging method to distinguish acute and chronic inflammation. J. Vis. Exp. 2013, e50690. [Google Scholar] [CrossRef]
- Johnbeck, C.B.; Knigge, U.; Loft, A.; Berthelsen, A.K.; Mortensen, J.; Oturai, P.; Langer, S.W.; Elema, D.R.; Kjaer, A. Head-to-Head Comparison of (64)Cu-DOTATATE and (68)Ga-DOTATOC PET/CT: A Prospective Study of 59 Patients with Neuroendocrine Tumors. J. Nucl. Med. 2017, 58, 451–457. [Google Scholar] [CrossRef]
- Pedersen, S.F.; Sandholt, B.V.; Keller, S.H.; Hansen, A.E.; Clemmensen, A.E.; Sillesen, H.; Hojgaard, L.; Ripa, R.S.; Kjaer, A. 64Cu-DOTATATE PET/MRI for Detection of Activated Macrophages in Carotid Atherosclerotic Plaques: Studies in Patients Undergoing Endarterectomy. Arter. Thromb. Vasc. Boil. 2015, 35, 1696–1703. [Google Scholar] [CrossRef]
- Svendsen, P.; Etzerodt, A.; Deleuran, B.W.; Moestrup, S.K. Mouse CD163 deficiency strongly enhances experimental collagen-induced arthritis. Sci. Rep. 2020, 10, 12447. [Google Scholar] [CrossRef]
- Hubalek, Z.; Halouzka, J.; Heroldova, M. Growth temperature ranges of Borrelia burgdorferi sensu lato strains. J. Med. Microbiol. 1998, 47, 929–932. [Google Scholar] [CrossRef] [PubMed]
- Hyde, J.A.; Weening, E.H.; Chang, M.; Trzeciakowski, J.P.; Hook, M.; Cirillo, J.D.; Skare, J.T. Bioluminescent imaging of Borrelia burgdorferi in vivo demonstrates that the fibronectin-binding protein BBK32 is required for optimal infectivity. Mol. Microbiol. 2011, 82, 99–113. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, M.; Azevedo, N.F.; Ivask, A. Propidium iodide staining underestimates viability of adherent bacterial cells. Sci. Rep. 2019, 9, 6483. [Google Scholar] [CrossRef] [PubMed]
- Golde, W.T.; Gollobin, P.; Rodriguez, L.L. A rapid, simple, and humane method for submandibular bleeding of mice using a lancet. Lab. Anim. (NY) 2005, 34, 39–43. [Google Scholar] [CrossRef] [PubMed]
- Pietrosimone, K.M.; Jin, M.; Poston, B.; Liu, P. Collagen-Induced Arthritis: A model for Murine Autoimmune Arthritis. Bio-Protocol 2015, 5. [Google Scholar] [CrossRef]
- Pfeifer, A.; Knigge, U.; Mortensen, J.; Oturai, P.; Berthelsen, A.K.; Loft, A.; Binderup, T.; Rasmussen, P.; Elema, D.; Klausen, T.L.; et al. Clinical PET of neuroendocrine tumors using 64Cu-DOTATATE: First-in-humans study. J. Nucl. Med. 2012, 53, 1207–1215. [Google Scholar] [CrossRef]
- Kjaer, A.; Binderup, T.; Johnbeck, C.; Carlsen, E.; Langer, S.; Federspiel, B.; Knigge, U. Cu-64-DOTATATE somatostatin receptor Imaging in neuroendocrine tumors: Experience from 500 patients at Copenhagen ENETS Center of Excellence. J. Nucl. Med. 2019, 60 (Suppl. 1), 504. [Google Scholar]
- Binderup, T.; Knigge, U.; Loft, A.; Mortensen, J.; Pfeifer, A.; Federspiel, B.; Hansen, C.P.; Hojgaard, L.; Kjaer, A. Functional imaging of neuroendocrine tumors: A head-to-head comparison of somatostatin receptor scintigraphy, 123I-MIBG scintigraphy, and 18F-FDG PET. J. Nucl. Med. 2010, 51, 704–712. [Google Scholar] [CrossRef]
- Pedersen, S.F.; Graebe, M.; Hag, A.M.; Hojgaard, L.; Sillesen, H.; Kjaer, A. (18)F-FDG imaging of human atherosclerotic carotid plaques reflects gene expression of the key hypoxia marker HIF-1alpha. Am. J. Nucl. Med. Mol. Imaging 2013, 3, 384–392. [Google Scholar]
- Jamar, F.; Buscombe, J.; Chiti, A.; Christian, P.E.; Delbeke, D.; Donohoe, K.J.; Israel, O.; Martin-Comin, J.; Signore, A. EANM/SNMMI guideline for 18F-FDG use in inflammation and infection. J. Nucl. Med. 2013, 54, 647–658. [Google Scholar] [CrossRef]
- Pietikainen, A.; Siitonen, R.; Liljenback, H.; Eskola, O.; Soderstrom, M.; Roivainen, A.; Hytonen, J. In vivo imaging of Lyme arthritis in mice by [(18)F]fluorodeoxyglucose positron emission tomography/computed tomography. Scand. J. Rheumatol. 2018, 47, 37–47. [Google Scholar] [CrossRef] [PubMed]
- Vaidyanathan, S.; Patel, C.N.; Scarsbrook, A.F.; Chowdhury, F.U. FDG PET/CT in infection and inflammation--current and emerging clinical applications. Clin. Radiol. 2015, 70, 787–800. [Google Scholar] [CrossRef] [PubMed]
- Glaudemans, A.W.; Slart, R.H.; van Dijl, J.M.; van Oosten, M.; van Dam, G.M. Molecular imaging of infectious and inflammatory diseases: A terra incognita. J. Nucl. Med. 2015, 56, 659–661. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Munson, E.; Nardelli, D.T.; Du Chateau, B.K.; Callister, S.M.; Schell, R.F. Hamster and murine models of severe destructive Lyme arthritis. Clin. Dev. Immunol. 2012, 2012, 504215. [Google Scholar] [CrossRef]
- Barthold, S.W.; Beck, D.S.; Hansen, G.M.; Terwilliger, G.A.; Moody, K.D. Lyme borreliosis in selected strains and ages of laboratory mice. J. Infect. Dis. 1990, 162, 133–138. [Google Scholar] [CrossRef]
- Du Chateau, B.K.; England, D.M.; Callister, S.M.; Lim, L.C.; Lovrich, S.D.; Schell, R.F. Macrophages exposed to Borrelia burgdorferi induce Lyme arthritis in hamsters. Infect. Immun. 1996, 64, 2540–2547. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Skovsbo Clausen, A.; Ørbæk, M.; Renee Pedersen, R.; Oestrup Jensen, P.; Lebech, A.-M.; Kjaer, A. 64Cu-DOTATATE Positron Emission Tomography (PET) of Borrelia Burgdorferi Infection: In Vivo Imaging of Macrophages in Experimental Model of Lyme Arthritis. Diagnostics 2020, 10, 790. https://doi.org/10.3390/diagnostics10100790
Skovsbo Clausen A, Ørbæk M, Renee Pedersen R, Oestrup Jensen P, Lebech A-M, Kjaer A. 64Cu-DOTATATE Positron Emission Tomography (PET) of Borrelia Burgdorferi Infection: In Vivo Imaging of Macrophages in Experimental Model of Lyme Arthritis. Diagnostics. 2020; 10(10):790. https://doi.org/10.3390/diagnostics10100790
Chicago/Turabian StyleSkovsbo Clausen, Anne, Mathilde Ørbæk, Regitze Renee Pedersen, Peter Oestrup Jensen, Anne-Mette Lebech, and Andreas Kjaer. 2020. "64Cu-DOTATATE Positron Emission Tomography (PET) of Borrelia Burgdorferi Infection: In Vivo Imaging of Macrophages in Experimental Model of Lyme Arthritis" Diagnostics 10, no. 10: 790. https://doi.org/10.3390/diagnostics10100790
APA StyleSkovsbo Clausen, A., Ørbæk, M., Renee Pedersen, R., Oestrup Jensen, P., Lebech, A.-M., & Kjaer, A. (2020). 64Cu-DOTATATE Positron Emission Tomography (PET) of Borrelia Burgdorferi Infection: In Vivo Imaging of Macrophages in Experimental Model of Lyme Arthritis. Diagnostics, 10(10), 790. https://doi.org/10.3390/diagnostics10100790