Hydropriming and Osmotic Priming Induce Resistance against Aspergillus niger in Wheat (Triticum aestivum L.) by Activating β-1, 3-glucanase, Chitinase, and Thaumatin-like Protein Genes
Abstract
1. Introduction
2. Materials and Methods
2.1. Seed Collection and Preparation
2.1.1. Seed Sterilization
2.1.2. Seed Priming
2.1.3. Seed Sowing and Germination
2.2. Fungus Inoculum Preparation
Fungus Inoculation
2.3. Disease Severity Analysis
2.4. Determination of Biochemical Contents
2.5. Analysis of Physiological Parameters
2.6. RNA Extraction, Quantification, and cDNA Synthesis
2.7. Primer Designing and RT-PCR
2.8. Real-Time PCR Analysis
2.9. Statistical Analysis
3. Results
3.1. Biochemical Content Analysis of Primed Plants in Response to Fungal Stress
3.1.1. Proline
3.1.2. Protein Content
3.1.3. Sugar Content
3.1.4. Chlorophyll Content
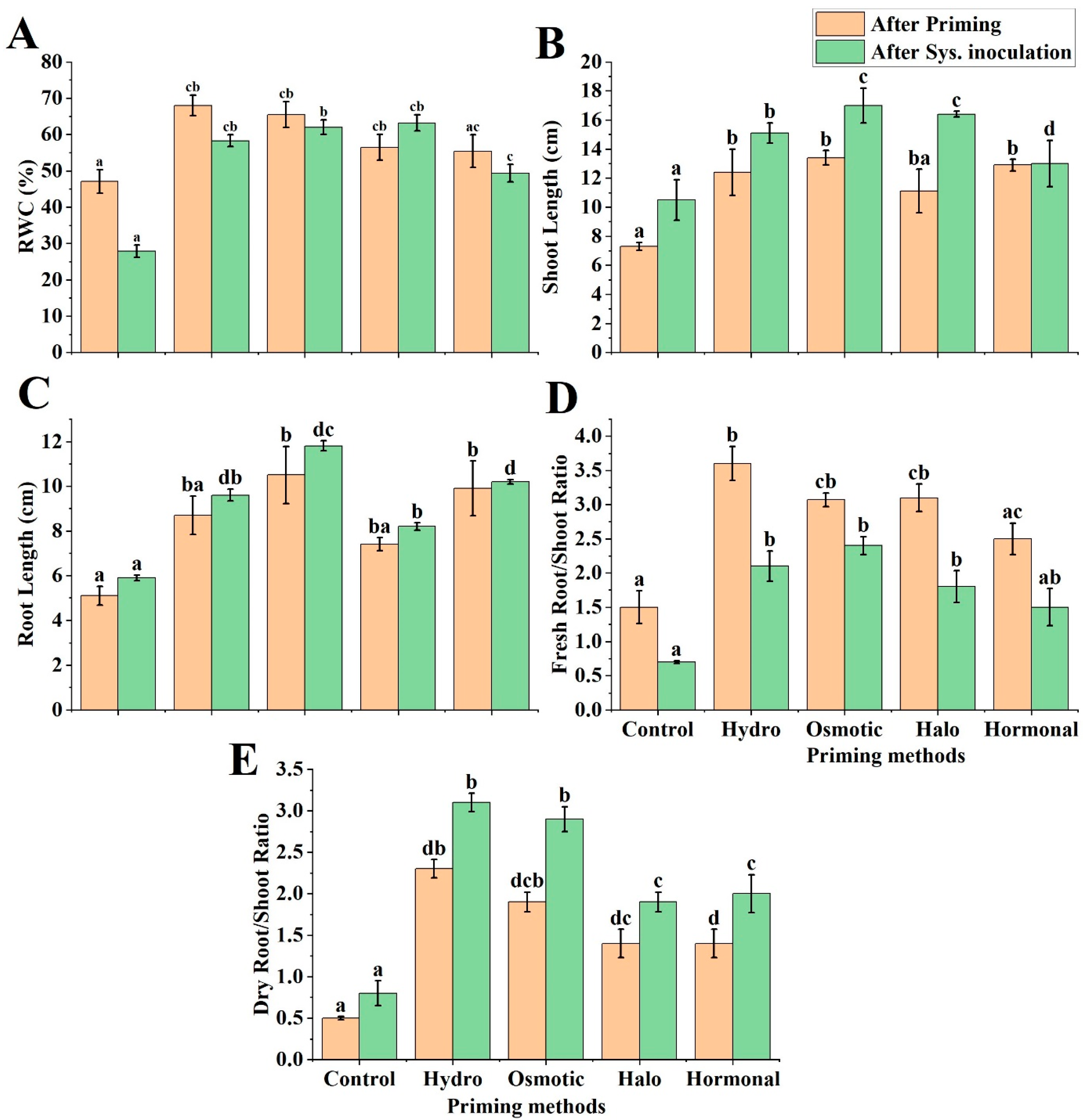
3.2. Response of Wheat Physiological Parameters to Different Priming Treatments
3.2.1. Relative Water Content (RWC)
3.2.2. Shoot Length
3.2.3. Root Length
3.2.4. Fresh and Dry Root/Shoot Ratio
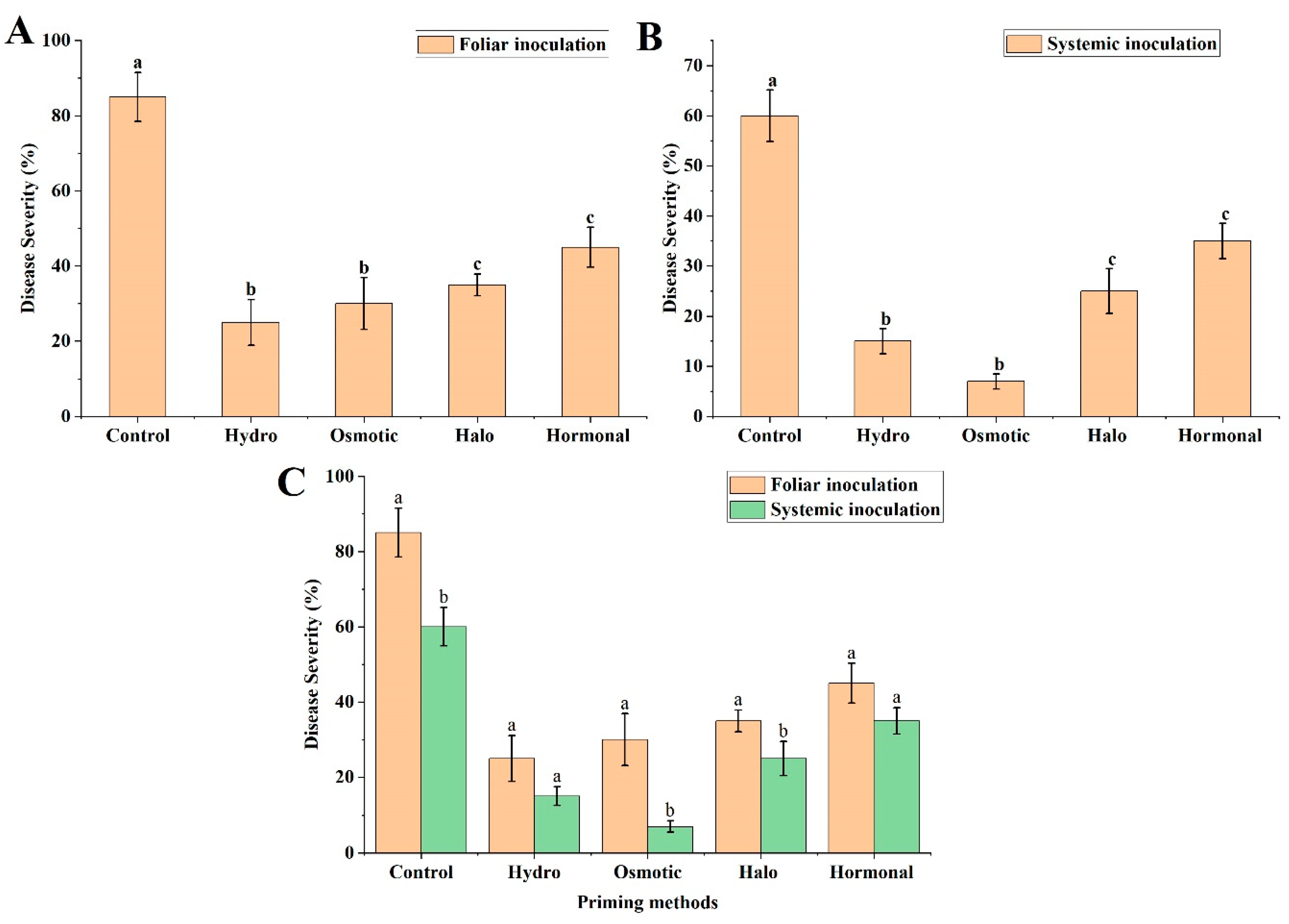
3.3. Disease Severity Analysis
3.3.1. Foliar Inoculation
3.3.2. Systemic Inoculation
3.3.3. Comparison of Foliar and Systemic Inoculation
3.3.4. Visual Assessment of Wilting
3.4. Expression Profiling of TLP, Chitinase, and β-1,3-glucanase Genes
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Khan, A.A. Preplant physiological seed conditioning. Hortic. Rev. 1992, 13, 131–181. [Google Scholar]
- Farooq, M.; Basra, S.; Rehman, H.; Saleem, B. Seed priming enhances the performance of late sown wheat (Triticum aestivum L.) by improving chilling tolerance. J. Agron. Crop. Sci. 2008, 194, 55–60. [Google Scholar] [CrossRef]
- Zhou, X.; Jia, X.; Zhang, Z.; Chen, K.; Wang, L.; Chen, H.; Yang, Z.; Li, C.; Zhao, L. AgNPs seed priming accelerated germination speed and altered nutritional profile of Chinese cabbage. Sci. Total Environ. 2022, 808, 151896. [Google Scholar] [CrossRef] [PubMed]
- Dessalew, F.; Ejeta, M.; Mola, T.; Haile, M. Effect of Halo, Hydro and Hormonal-priming on germination, seedling growth, seedling vigor and seed yield of carrot (Daucus carota) seed. Int. J. Nov. Res. Interdiscip. Stud. 2022, 9, 1–8. [Google Scholar]
- Ali, Q.; Daud, M.; Haider, M.Z.; Ali, S.; Rizwan, M.; Aslam, N.; Noman, A.; Iqbal, N.; Shahzad, F.; Deeba, F. Seed priming by sodium nitroprusside improves salt tolerance in wheat (Triticum aestivum L.) by enhancing physiological and biochemical parameters. Plant Physiol. Biochem. 2017, 119, 50–58. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Prasad, S.M. Management of chromium (VI) toxicity by calcium and sulfur in tomato and brinjal: Implication of nitric oxide. J. Hazard. Mater. 2019, 373, 212–223. [Google Scholar] [CrossRef]
- Ashraf, M.A.; Rasheed, R.; Hussain, I.; Hafeez, A.; Adrees, M.; Rehman, M.Z.U.; Rizwan, M.; Ali, S. Effect of different seed priming agents on chromium accumulation, oxidative defense, glyoxalase system and mineral nutrition in canola (Brassica napus L.) cultivars. Environ. Pollut. 2022, 309, 119769. [Google Scholar] [CrossRef]
- Kumari, N.; Rai, P.K.; Bara, B.M.; Singh, I. Effect of halo priming and hormonal priming on seed germination and seedling vigour in maize (Zea mays L.) seeds. J. Pharmacogn. Phytochem. 2017, 6, 27–30. [Google Scholar]
- Sheteiwy, M.S.; Shao, H.; Qi, W.; Daly, P.; Sharma, A.; Shaghaleh, H.; Hamoud, Y.A.; El-Esawi, M.A.; Pan, R.; Wan, Q.; et al. Seed priming and foliar application with jasmonic acid enhance salinity stress tolerance of soybean (Glycine max L.) seedlings. J. Sci. Food Agric. 2021, 101, 2027–2041. [Google Scholar] [CrossRef]
- Pawar, V.; Laware, S.L. Seed priming a critical review. Int. J. Sci. Res. Biol. Sci. 2018, 5, 94–101. [Google Scholar] [CrossRef]
- Khalid, M.F.; Hussain, S.; Anjum, M.A.; Ejaz, S.; Ahmad, M.; Jan, M.; Zafar, S.; Zakir, I.; Ali, M.A.; Ahmad, N. Hydropriming for plant growth and stress tolerance. In Priming and Pretreatment of Seeds and Seedlings; Hasanuzzaman, M., Fotopoulos, V., Eds.; Springer: Singapore, 2019; pp. 373–384. [Google Scholar]
- Ghassemi-Golezani, K.; Hassanpour-Bourkheili, S.; Bandeh-Hagh, A.; Abriz, S.F. Seed hydro-priming, a simple way for improving mungbean performance under water stress. Res. Crops 2014, 4, 12–18. [Google Scholar]
- Bhargaw, A.; Akanksha; Madane, D.A.; Chauhan, P. Effect of Halo Priming, Osmo Priming and Hydro Priming on Fresh and Accelerated Aged Seeds of Baby Corn (Zea mays L.) on Germination, Seedling Dry Weight, Seedling Length. Int. J. Curr. Microbiol. App. Sci. 2019, 8, 1474–1479. [Google Scholar] [CrossRef]
- Hosseinzadeh-Mahootchi, A.; Ghassemi-Golezani, K.; Zehtab-Salmasi, S.; Tourchi, M. Influence of seed invigoration and water supply on morphophysiological traits of chickpea. Int. J. Agron. Plant Produc. 2013, 4, 782–786. [Google Scholar]
- Sher, A.; Sarwar, T.; Nawaz, A.; Ijaz, M.; Sattar, A.; Ahmad, S. Methods of seed priming. In Priming and Pretreatment of Seeds and Seedlings; Hasanuzzaman, M., Fotopoulos, V., Eds.; Springer: Singapore, 2019; pp. 1–10. [Google Scholar]
- Sadeghi, H.; Khazaei, F.; Yari, L.; Sheidaei, S. Effect of seed osmopriming on seed germination behavior and vigor of soybean (Glycine max L.). Agric. Biol. Sci. 2011, 6, 39–43. [Google Scholar]
- Sunera, A.; Saqib, S.; Uddin, S.; Zaman, W.; Ullah, F.; Ayaz, A.; Asghar, M.; Rehman, S.; Munis, M.; Chaudhary, H. Characterization and phytostimulatory activity of bacteria isolated from tomato (Lycopersicon esculentum Mill.) rhizosphere. Microb. Pathog. 2020, 140, 103966. [Google Scholar] [CrossRef]
- Narayani, S.; Yashodhara, V.; Shukla, P.; Pragati, M. Effect of polyethylene glycol (PEG) 6000 on seed priming in drought tolerant and sensitive barley (Hordeum vulgare L.) seeds. Int. J. Plant Sci. 2016, 11, 75–78. [Google Scholar]
- Mushtaq, N.; Iqbal, S.; Hayat, F.; Raziq, A.; Ayaz, A.; Zaman, W. Melatonin in Micro-Tom Tomato: Improved Drought Tolerance via the Regulation of the Photosynthetic Apparatus, Membrane Stability, Osmoprotectants, and Root System. Life 2022, 12, 1922. [Google Scholar] [CrossRef]
- Reignault, P.; Cogan, A.; Muchembled, J.; Lounes-Hadj Sahraoui, A.; Durand, R.; Sancholle, M. Trehalose induces resistance to powdery mildew in wheat. New Phytol. 2001, 149, 519–529. [Google Scholar] [CrossRef]
- Sudisha, J.; Sharathchandra, R.; Amruthesh, K.; Kumar, A.; Shetty, H.S. Pathogenesis related proteins in plant defense response. In Plant Defence: Biological Control; Mérillon, J., Ramawat, K., Eds.; Springer: Dordrecht, The Netherlands, 2012; pp. 379–403. [Google Scholar]
- Sexton, A.C.; Howlett, B.J. Parallels in fungal pathogenesis on plant and animal hosts. Eukaryot. Cell 2006, 5, 1941–1949. [Google Scholar] [CrossRef] [PubMed]
- Misra, R.C.; Kamthan, M.; Kumar, S.; Ghosh, S.A. A thaumatin-like protein of Ocimum basilicum confers tolerance to fungal pathogen and abiotic stress in transgenic Arabidopsis. Sci. Rep. 2016, 6, 25340. [Google Scholar] [CrossRef]
- Ullah, A.; Hussain, A.; Shaban, M.; Khan, A.H.; Alariqi, M.; Gul, S.; Jun, Z.; Lin, S.; Li, J.; Jin, S. Osmotin: A plant defense tool against biotic and abiotic stresses. Plant Physiol. Biochem. 2018, 123, 149–159. [Google Scholar]
- Munis, M.F.H.; Tu, L.; Deng, F.; Tan, J.; Xu, L.; Xu, S.; Long, L.; Zhang, X. A thaumatin-like protein gene involved in cotton fiber secondary cell wall development enhances resistance against Verticillium dahliae and other stresses in transgenic tobacco. Biochem. Biophys. Res. Commun. 2010, 393, 38–44. [Google Scholar] [CrossRef]
- Muoki, R.C.; Paul, A.; Kumar, S.A. Shared response of thaumatin like protein, chitinase, and late embryogenesis abundant protein3 to environmental stresses in tea [Camellia sinensis (L.) O. Kuntze]. Funct. Integr. Genom. 2012, 12, 565–571. [Google Scholar] [CrossRef]
- Manghwar, H.; Hussain, A.; Ullah, A.; Gul, S.; Shaban, M.; Khan, A.H.; Ali, M.; Sani, S.G.A.S.; Chaudhary, H.J.; Munis, M.F.H. Expression analysis of defense related genes in wheat and maize against Bipolaris sorokiniana. Physiol. Mol. Plant Pathol. 2018, 103, 36–46. [Google Scholar] [CrossRef]
- Manghwar, H.; Hussain, A.; Ali, Q.; Saleem, M.H.; Abualreesh, M.H.; Alatawi, A.; Ali, S.; Munis, M.F.H. Disease severity, resistance analysis, and expression profiling of pathogenesis-related protein genes after the inoculation of Fusarium equiseti in wheat. Agronomy 2021, 11, 2124. [Google Scholar] [CrossRef]
- Grenier, J.; Potvin, C.; Asselin, A. Some fungi express β-1, 3-glucanases similar to thaumatin-like proteins. Mycologia 2000, 92, 841–848. [Google Scholar] [CrossRef]
- Rajam, M.; Chandola, N.; Goud, P.S.; Singh, D.; Kashyap, V.; Choudhary, M.; Sihachakr, D. Thaumatin gene confers resistance to fungal pathogens as well as tolerance to abiotic stresses in transgenic tobacco plants. Biol. Plant. 2007, 51, 135–141. [Google Scholar] [CrossRef]
- Yeh, S.; Moffatt, B.A.; Griffith, M.; Xiong, F.; Yang, D.S.; Wiseman, S.B.; Sarhan, F.; Danyluk, J.; Xue, Y.Q.; Hew, C.L. Chitinase genes responsive to cold encode antifreeze proteins in winter cereals. Plant Physiol. 2000, 124, 1251–1264. [Google Scholar] [CrossRef]
- Chu, K.T.; Ng, T.B. Purification and characterization of a chitinase-like antifungal protein from black turtle bean with stimulatory effect on nitric oxide production by macrophages. Biolo. Chem. 2005, 386, 19–24. [Google Scholar] [CrossRef]
- Saikia, R.; Singh, B.P.; Kumar, R.; Arora, D.K. Detection of pathogenesis related proteins—Chitinase and b-1, 3-glucanase in induced chickpea. Curr. Sci. 2005, 89, 659–663. [Google Scholar]
- Kirubakaran, S.I.; Sakthivel, N. Cloning and overexpression of antifungal barley chitinase gene in Escherichia coli. Protein Expr. Purif. 2007, 52, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Jach, G.; Görnhardt, B.; Mundy, J.; Logemann, J.; Pinsdorf, E.; Leah, R.; Schell, J.; Maas, C. Enhanced quantitative resistance against fungal disease by combinatorial expression of different barley antifungal proteins in transgenic tobacco. Plant J. 1995, 8, 97–109. [Google Scholar] [CrossRef]
- Borad, V.; Sriram, S. Pathogenesis-related proteins for the plant protection. Asian J. Exp. Sci. 2008, 22, 189–196. [Google Scholar]
- Rezzonico, E.; Flury, N.; Meins, F.; Beffa, R. Transcriptional down-regulation by abscisic acid of pathogenesis-related β-1, 3-glucanase genes in tobacco cell cultures. Plant Physiol. 1998, 117, 585–592. [Google Scholar] [CrossRef] [PubMed]
- Klarzynski, O.; Plesse, B.; Joubert, J.-M.; Yvin, J.-C.; Kopp, M.; Kloareg, B.; Fritig, B. Linear β-1, 3 glucans are elicitors of defense responses in tobacco. Plant Physiol. 2000, 124, 1027–1038. [Google Scholar] [CrossRef]
- Saboki Ebrahim, K.U.; Singh, B. Pathogenesis related (PR) proteins in plant defense mechanism. Sci. Against Microb. Pathog. 2011, 2, 1043–1054. [Google Scholar]
- Bennett and Waters, M.A.; Bennett, L.W. Seed hydration treatments for improved sweet corn germination and stand establishment. J. Am. Soc. Hortic. Sci. 1987, 112, 45–49. [Google Scholar] [CrossRef]
- Naik, B.S.; Shashikala, J.; Krishnamurthy, Y. Host growth characteristics influenced by seed inoculation with microorganisms. World J. Agric. Sci. 2008, 4, 891–895. [Google Scholar]
- Gashaw, G.; Alemu, T.; Tesfaye, K. Evaluation of disease incidence and severity and yield loss of finger millet varieties and mycelial growth inhibition of Pyricularia grisea isolates using biological antagonists and fungicides in vitro condition. J. Appl. Biosci. 2014, 73, 5883–5901. [Google Scholar]
- Tyree, M.T.; Vargas, G.; Engelbrecht, B.M.; Kursar, T.A. Drought until death do us part: A case study of the desiccation-tolerance of a tropical moist forest seedling-tree, Licania platypus (Hemsl.) Fritsch. J. Exp. Bot. 2002, 53, 2239–2247. [Google Scholar] [CrossRef]
- Tyree, M.T.; Engelbrecht, B.M.; Vargas, G.; Kursar, T.A. Desiccation tolerance of five tropical seedlings in Panama. Relationship to a field assessment of drought performance. Plant Physiol. 2003, 132, 1439–1447. [Google Scholar] [CrossRef]
- Engelbrecht, B.M.; Tyree, M.T.; Kursar, T.A. Visual assessment of wilting as a measure of leaf water potential and seedling drought survival. J. Trop. Ecol. 2007, 23, 497–500. [Google Scholar] [CrossRef]
- Johnson, R.R.; Balwani, T.L.; Johnson, L.; McClure, K.; Dehority, B. Corn Plant Maturity. II. Effect on Cellulose Digestibility and Soluble Carbohydrate Content. J. Anim. Sci. 1966, 25, 617–623. [Google Scholar] [CrossRef]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar] [CrossRef]
- Bates, L.; Waldren, R.; Teare, I. Rapid determination of free proline for water-stress studies. Plant Soil 1973, 39, 205–207. [Google Scholar] [CrossRef]
- Arnon, D.I. Copper enzymes in isolated chloroplasts. Polyphenoloxidase in Beta vulgaris. Plant Physiol. 1949, 24, 1. [Google Scholar] [CrossRef]
- Hussain, A.; Kamran, M.A.; Javed, M.T.; Hayat, K.; Farooq, M.A.; Ali, N.; Ali, M.; Manghwar, H.; Jan, F.; Chaudhary, H.J. Individual and combinatorial application of Kocuria rhizophila and citric acid on phytoextraction of multi-metal contaminated soils by Glycine max L. Environ. Exp. Bot. 2019, 159, 23–33. [Google Scholar] [CrossRef]
- Whetherley, P. Studies in the water relations of cotton plants. I. The field measurement of water deficit in leaves. New Phytol. 1950, 49, 81–87. [Google Scholar] [CrossRef]
- Kishor, K.; Polavarapu, B.; Hima Kumari, P.; Sunita, M.; Sreenivasulu, N. Role of proline in cell wall synthesis and plant development and its implications in plant ontogeny. Front. Plant Sci. 2015, 6, 544. [Google Scholar] [CrossRef]
- Jalal, A.; Ali, Q.; Manghwar, H.; Zhu, D. Identification, Phylogeny, Divergence, Structure, and Expression Analysis of A20/AN1 Zinc Finger Domain Containing Stress-Associated Proteins (SAPs) Genes in Jatropha curcas L. Genes 2022, 13, 1766. [Google Scholar] [CrossRef]
- Morkunas, I.; Ratajczak, L. The role of sugar signaling in plant defense responses against fungal pathogens. Acta Physiol. Plant. 2014, 36, 1607–1619. [Google Scholar] [CrossRef]
- Shah, S.H.; Houborg, R.; McCabe, M.F. Response of Chlorophyll, Carotenoid and SPAD-502 Measurement to Salinity and Nutrient Stress in Wheat (Triticum aestivum L.). Agronomy 2017, 7, 61. [Google Scholar] [CrossRef]
- Paparella, S.; Araújo, S.; Rossi, G.; Wijayasinghe, M.; Carbonera, D.; Balestrazzi, A. Seed priming: State of the art and new perspectives. Plant Cell Rep. 2015, 34, 1281–1293. [Google Scholar] [CrossRef] [PubMed]
- Hayat, K.; Menhas, S.; Bundschuh, J.; Zhou, P.; Niazi, N.K.; Amna; Hussain, A.; Hayat, S.; Ali, H.; Wang, J. Plant growth promotion and enhanced uptake of Cd by combinatorial application of Bacillus pumilus and EDTA on Zea mays L. Int. J. Phytoremediation 2020, 22, 1372–1384. [Google Scholar] [CrossRef]
- Manghwar, H.; Hussain, A. Mechanism of tobacco osmotin gene in plant responses to biotic and abiotic stress tolerance: A brief history. Biocell 2022, 46, 623. [Google Scholar] [CrossRef]
- Khoso, M.A.; Hussain, A.; Faujiah Nurhasanah Ritonga, Q.A.; Channa, M.M.; Alshegaihi, R.M.; Meng, Q.; Ali, M.; Zaman10, W.; Brohi, R.D.; Liu, F. WRKY transcription factors (TFs): Molecular switches to regulate drought, temperature, and salinity stresses in plants. Front. Plant Sci. 2022, 13, 1039329. [Google Scholar] [CrossRef]
- MA, J.-h.; GUO, S.-j.; WANG, Y.-g.; YANG, X.-h.; SHI, Q. Effects of seed priming on biomass allocation and osmotic substance contents of sorghum (Sorghum bicolor L.) seedlings under salt stress. Chin. J. Ecol. 2010, 29, 1950–1956. [Google Scholar]
- Farahmandfar, E.; Shirvan, M.B.; Sooran, S.A.; Hoseinzadeh, D. Effect of seed priming on morphological and physiological parameters of fenugreek seedlings under salt stress. Intern. J. Agric. Crop Sci. 2013, 5, 811–815. [Google Scholar]
- Kubala, S.; Wojtyla, Ł.; Quinet, M.; Lechowska, K.; Lutts, S.; Garnczarska, M. Enhanced expression of the proline synthesis gene P5CSA in relation to seed osmopriming improvement of Brassica napus germination under salinity stress. J. Plant Physiol. 2015, 183, 1–12. [Google Scholar] [CrossRef]
- Ali, Q.; Ayaz, M.; Yu, C.; Wang, Y.; Gu, Q.; Wu, H.; Gao, X. Cadmium tolerant microbial strains possess different mechanisms for cadmium biosorption and immobilization in rice seedlings. Chemosphere 2022, 303, 135206. [Google Scholar] [CrossRef]
- Sedghi, M.B.; BA, T.S. Desiccation tolerance in hydro-primed Calendula officinalis L. seeds as influence by slow and rapid drying back condition. Ann. Biol. Res. 2012, 3, 3563–3569. [Google Scholar]
- Shekoofeh, E.; Sepideh, H.; Roya, R. Role of mycorrhizal fungi and salicylic acid in salinity tolerance of Ocimum basilicum resistance to salinity. Afr. J. Biotechnol. 2012, 11, 2223–2235. [Google Scholar]
- Alasvandyari, F.; Mahdavi, B.; Hosseini, S.M. Glycine betaine affects the antioxidant system and ion accumulation and reduces salinity-induced damage in safflower seedlings. Arch. Biol. Sci. 2017, 69, 139–147. [Google Scholar] [CrossRef]
- Mohamed, A.K.S.; Qayyum, M.F.; Abdel-Hadi, A.M.; Rehman, R.A.; Ali, S.; Rizwan, M. Interactive effect of salinity and silver nanoparticles on photosynthetic and biochemical parameters of wheat. Arch. Agron. Soil Sci. 2017, 63, 1736–1747. [Google Scholar] [CrossRef]
- Rinez, I.; Ghezal, N.; Rinez, A.; Muhammad, F.; Dbara, S.; Saad, I.; Haouala, R. Improving salt tolerance in pepper by bio-priming with Padina pavonica and Jania rubens aqueous extracts. Int. J. Agric. Biol. 2018, 20, 513–523. [Google Scholar] [CrossRef]
- Anwar, S.; Shafi, M.; Bakht, J.; Tariq Jan, M.; Hayat, Y. Effect of salinity and seed priming on growth and biochemical parameters of different barely genotypes. Afr. J. Biotechnol. 2011, 10, 15278–15286. [Google Scholar] [CrossRef]
- SukSoon, L.; JaeHyeun, K. Total sugars, α-amylase activity, and germination after priming of normal and aged rice seeds. Korean J. Crop Sci. 2000, 45, 108–111. [Google Scholar]
- El-Tayeb, M. Response of barley grains to the interactive e. ect of salinity and salicylic acid. Plant Growth Regul. 2005, 45, 215–224. [Google Scholar]
- Liaquat, F.; Qunlu, L.; Arif, S.; Haroon, U.; Saqib, S.; Zaman, W.; Jianxin, S.; Shengquan, C.; Li, L.X.; Akbar, M. Isolation and characterization of pathogen causing brown rot in lemon and its control by using ecofriendly botanicals. Physiol. Mol. Plant Pathol. 2021, 114, 101639. [Google Scholar] [CrossRef]
- Klement, Z.; Goodman, R. The hypersensitive reaction to infection by bacterial plant pathogens. Annu. Rev. Phytopathol. 1967, 5, 17–44. [Google Scholar] [CrossRef]
- Jayapal, R.; Mahadevan, A. Biochemical changes in banana leaves in response to leaf spot pathogens. Indian Phytopathol. 1968, 21, 43–48. [Google Scholar]
- Bourioug, M.; Ezzaza, K.; Bouabid, R.; Alaoui-Mhamdi, M.; Bungau, S.; Bourgeade, P.; Alaoui-Sossé, L.; Alaoui-Sossé, B.; Aleya, L. Influence of hydro-and osmo-priming on sunflower seeds to break dormancy and improve crop performance under water stress. Environ. Sci. Pollut. Res. Int. 2020, 27, 13215–13226. [Google Scholar] [CrossRef]
- Bejandi, T.K.; Sedghi, M.; Sharifi, R.S.; Namvar, A.; Molaei, P. Seed priming and sulfur effects on soybean cell membrane stability and yield in saline soil. Pesqui. Agropecuária Bras. 2009, 44, 1114–1117. [Google Scholar] [CrossRef]
- Moulick, D.; Santra, S.; Ghosh, D. Seed priming with Se alleviate As induced phytotoxicity during germination and seedling growth by restricting As translocation in rice (Oryza sativa L. cv IET-4094). Ecotoxicol. Environ. Saf. 2017, 145, 449–456. [Google Scholar] [CrossRef]
- Meriem, B.F.; Kaouther, Z.; Chérif, H.; Tijani, M.; André, B. Effect of priming on growth, biochemical parameters and mineral composition of different cultivars of coriander (Coriandrum sativum L.) under salt stress. J. Stress Physiol. Biochem. 2014, 10, 84–109. [Google Scholar]
- Namdari, A.; Baghbani, A. Consequences of seed priming with salicylic acid and hydro priming on smooth vetch seedling growth under water deficiency. J. Agric. Sci. 2017, 9, 259. [Google Scholar] [CrossRef][Green Version]
- Mahboob, W.; Rehman, H.U.; Basra, S.M.A.; Afzal, I.; Abbas, M.A.; Naeem, M.; Sarwar, M. Seed priming improves the performance of late sown spring maize (Zea mays) through better crop stand and physiological attributes. Int. J. Agric. Biol. 2015, 17, 491–498. [Google Scholar] [CrossRef]
- Kumar, V.K.; Rajalekshmi, R. Effect of hydro-, halo-and osmopriming on seed germination and seedling performance of Psophocarpus tetragonolobus (L.) DC.(winged bean). J. Crop Sci. Biotechnol. 2021, 24, 411–428. [Google Scholar] [CrossRef]
- Sakhabutdinova, A.; Fatkhutdinova, D.; Bezrukova, M.; Shakirova, F. Salicylic acid prevents the damaging action of stress factors on wheat plants. Bulg. J. Plant Physiol. 2003, 21, 314–319. [Google Scholar]
- Wani, M.A.; Khan, F.U.; Din, A.; Nazki, I.T.; Iqbal, S.; Banday, N. Influence of priming treatments on germination, seedling growth and survival of China aster [Callistephus chinensis (L.)]. In Vegetos; Springer: Berlin/Heidelberg, Germany, 2022; pp. 1–10. [Google Scholar]
- Zhang, F.; Yu, J.; Johnston, C.R.; Wang, Y.; Zhu, K.; Lu, F.; Zhang, Z.; Zou, J. Seed priming with polyethylene glycol induces physiological changes in sorghum (Sorghum bicolor L. Moench) seedlings under suboptimal soil moisture environments. PLoS ONE 2015, 10, e0140620. [Google Scholar] [CrossRef]
- Lemmens, E.; Deleu, L.J.; De Brier, N.; De Man, W.L.; De Proft, M.; Prinsen, E.; Delcour, J.A. The impact of hydro-priming and osmo-priming on seedling characteristics, plant hormone concentrations, activity of selected hydrolytic enzymes, and cell wall and phytate hydrolysis in sprouted wheat (Triticum aestivum L.). ACS Omega 2019, 4, 22089–22100. [Google Scholar] [CrossRef] [PubMed]
- Al-Abdalall, A.H.A. Assessment of yield loss caused by root rots in wheat and barley. J. Food Agric. Environ. 2010, 8, 638–641. [Google Scholar]
- Zida, P.; Néya, B.; Stokholm, M.; Jensen, S.; Soalla, W.; Sérémé, P.; Lund, O. Increasing sorghum yields by seed treatment with an aqueous extract of the plant Eclipta alba may involve a dual mechanism of hydropriming and suppression of fungal pathogens. Crop Prot. 2018, 107, 48–55. [Google Scholar] [CrossRef]
- Rashid, A.; Harris, D.; Hollington, P.; Ali, S. On-farm seed priming reduces yield losses of mungbean (Vigna radiata) associated with mungbean yellow mosaic virus in the North West Frontier Province of Pakistan. Crop Prot. 2004, 23, 1119–1124. [Google Scholar] [CrossRef]
- Moosa, A.; Farzand, A.; Sahi, S.T.; Khan, S.A. Transgenic expression of antifungal pathogenesis-related proteins against phytopathogenic fungi–15 years of success. Isr. J. Plant Sci. 2018, 65, 38–54. [Google Scholar] [CrossRef]
- Cao, J.; Lv, Y.; Hou, Z.; Li, X.; Ding, L. Expansion and evolution of thaumatin-like protein (TLP) gene family in six plants. Plant Growth Regul. 2016, 79, 299–307. [Google Scholar] [CrossRef]
- Lindqvist-Kreuze, H.; Carbajulca, D.; Gonzalez-Escobedo, G.; Perez, W.; Bonierbale, M. Comparison of transcript profiles in late blight-challenged Solanum cajamarquense and B3C1 potato clones. Mol. Plant Pathol. 2010, 11, 513–530. [Google Scholar] [CrossRef]
- Van den Burg, H.A.; Westerink, N.; Francoijs, K.-J.; Roth, R.; Woestenenk, E.; Boeren, S.; de Wit, P.J.; Joosten, M.H.; Vervoort, J. Natural disulfide bond-disrupted mutants of AVR4 of the tomato pathogen Cladosporium fulvum are sensitive to proteolysis, circumvent Cf-4-mediated resistance, but retain their chitin binding ability. J. Biol. Chem. 2003, 278, 27340–27346. [Google Scholar] [CrossRef]
- Ferreira, R.B.; Monteiro, S.; Freitas, R.; Santos, C.N.; Chen, Z.; Batista, L.M.; Duarte, J.; Borges, A.; Teixeira, A.R. The role of plant defence proteins in fungal pathogenesis. Mol. Plant Pathol. 2007, 8, 677–700. [Google Scholar] [CrossRef]
- Acharya, K.; Pal, A.K.; Gulati, A.; Kumar, S.; Singh, A.K.; Ahuja, P.S. Overexpression of Camellia sinensis thaumatin-like protein, CsTLP in potato confers enhanced resistance to Macrophomina phaseolina and Phytophthora infestans infection. Mol. Biotechnol. 2013, 54, 609–622. [Google Scholar] [CrossRef]
- Mahdavi, F.; Sariah, M.; Maziah, M. Expression of rice thaumatin-like protein gene in transgenic banana plants enhances resistance to Fusarium wilt. Appl. Biochem. Biotechnol. 2012, 166, 1008–1019. [Google Scholar] [CrossRef] [PubMed]





| S. No. | Protein | Primers |
|---|---|---|
| 1 | Thaumatin-like protein (TLP) | Forward 5′ GCAGTCAAGGCAGTTGGTGGTA 3′, Reverse 5′ GCAGTCAAGGCAGTTGGTGGTA 3′ |
| 2 | Chitinase | Forward 5′ CGCAGTCACCTAAACCTTCG 3′ Reverse 5′ GCAGTAGCGCTTGTAGAACC 3′ |
| 3 | β 1,3-glucanase | Forward 5′ CTACAGGTCCAAGGGCATCA 3′ Reverse 5′ CCGGACATTGTTCTGAACCC 3′ |
| 4 | Actin | Forward 5′ CAAAGAGATCACGGCCCTTG 3′ Reverse 5′ ACTTCATGTGGACAATGCCG 3′ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gul, S.; Hussain, A.; Ali, Q.; Alam, I.; Alshegaihi, R.M.; Meng, Q.; Zaman, W.; Manghwar, H.; Munis, M.F.H. Hydropriming and Osmotic Priming Induce Resistance against Aspergillus niger in Wheat (Triticum aestivum L.) by Activating β-1, 3-glucanase, Chitinase, and Thaumatin-like Protein Genes. Life 2022, 12, 2061. https://doi.org/10.3390/life12122061
Gul S, Hussain A, Ali Q, Alam I, Alshegaihi RM, Meng Q, Zaman W, Manghwar H, Munis MFH. Hydropriming and Osmotic Priming Induce Resistance against Aspergillus niger in Wheat (Triticum aestivum L.) by Activating β-1, 3-glucanase, Chitinase, and Thaumatin-like Protein Genes. Life. 2022; 12(12):2061. https://doi.org/10.3390/life12122061
Chicago/Turabian StyleGul, Summia, Amjad Hussain, Qurban Ali, Intikhab Alam, Rana M. Alshegaihi, Qinglin Meng, Wajid Zaman, Hakim Manghwar, and Muhammad Farooq Hussain Munis. 2022. "Hydropriming and Osmotic Priming Induce Resistance against Aspergillus niger in Wheat (Triticum aestivum L.) by Activating β-1, 3-glucanase, Chitinase, and Thaumatin-like Protein Genes" Life 12, no. 12: 2061. https://doi.org/10.3390/life12122061
APA StyleGul, S., Hussain, A., Ali, Q., Alam, I., Alshegaihi, R. M., Meng, Q., Zaman, W., Manghwar, H., & Munis, M. F. H. (2022). Hydropriming and Osmotic Priming Induce Resistance against Aspergillus niger in Wheat (Triticum aestivum L.) by Activating β-1, 3-glucanase, Chitinase, and Thaumatin-like Protein Genes. Life, 12(12), 2061. https://doi.org/10.3390/life12122061










