Fatty Liver, and Not Visceral Fat, Is More Associated with Liver Fibrosis and Diabetes in Non-Obese Japanese Individuals: A Cross-Sectional Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Methods
2.3. Statistical Analysis
3. Results
3.1. Characteristics
3.2. Complications
3.3. Blood Biochemical Examination
3.4. FIB4 Index
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Younossi, Z.; Tacke, F.; Arrese, M.; Chander Sharma, B.; Mostafa, I.; Bugianesi, E.; Wong, V.W.; Yilmaz, Y.; George, J.; Fan, J.; et al. Global perspectives on nonalcoholic fatty liver disease and nonalcoholic steatohepatitis. Hepatology 2019, 69, 2672–2682. [Google Scholar] [CrossRef]
- Sung, K.C.; Wild, S.H.; Byrne, C.D. Development of new fatty liver, or resolution of existing fatty liver, over five years of follow-up, and risk of incident hypertension. J. Hepatol. 2014, 60, 1040–1045. [Google Scholar] [CrossRef] [PubMed]
- Tsuneto, A.; Hida, A.; Sera, N.; Imaizumi, M.; Ichimaru, S.; Nakashima, E.; Seto, S.; Maemura, K.; Alkahoshi, M. Fatty liver incidence and predictive variables. Hypertens. Res. 2010, 33, 638–643. [Google Scholar] [CrossRef]
- Chang, Y.; Jung, H.S.; Cho, J.; Zhang, Y.; Yun, K.E.; Lazo, M.; Pastor-Barriuso, R.; Ahn, J.; Kim, C.-W.; Rampal, S.; et al. Metabolically healthy obesity and the development of nonalcoholic fatty liver disease. Am. J. Gastroenterol. 2016, 111, 1133–1140. [Google Scholar] [CrossRef]
- Eslam, M.; Newsome, P.N.; Sarin, S.K.; Anstee, Q.M.; Targher, G.; Romero-Gomez, M.; Zelber-Sagi, S.; Wong, V.W.-S.; Dufour, J.-F.; Schattenberg, J.M.; et al. A new definition for metabolic dysfunction-associated fatty liver disease: An international expert consensus statement. J. Hepatol. 2020, 73, 202–209. [Google Scholar] [CrossRef]
- Everhart, J.E.; Bambha, K.M. Fatty liver: Think globally. Hepatology 2010, 51, 1491–1493. [Google Scholar] [CrossRef]
- Shah, A.G.; Lydecker, A.; Murray, K.; Tetri, B.N.; Contos, M.J.; Sanyal, A.J.; NASH Clinical Research Net work. Comparison of noninvasive markers of fibrosis in patients with nonalcoholic fatty liver disease. Clin. Gastroenterol. Hepatol. 2009, 7, 1104–1112. [Google Scholar] [CrossRef]
- Katsuki, A.; Sumida, Y.; Urakawa, H.; Gabazza, E.C.; Murashima, S.; Maruyama, N.; Morioka, K.; Nakatani, K.; Yano, Y.; Adachi, Y. Increased visceral fat and serum levels of triglyceride are associated with insulin resistance in Japanese metabolically obese, normal weight subjects with normal glucose tolerance. Diabetes Care 2003, 26, 2341–2344. [Google Scholar] [CrossRef]
- Takeno, K.; Tamura, Y.; Kawaguchi, M.; Kakehi, S.; Watanabe, T.; Funayama, T.; Furukawa, Y.; Kaga, H.; Yamamoto, R.; Kim, M.; et al. Relation Between Insulin Sensitivity and Metabolic Abnormalities in Japanese Men With BMI of 23–25 kg/m2. J. Clin. Endocrinol. Metab. 2016, 101, 3676–3684. [Google Scholar] [CrossRef]
- Chooi, Y.C.; Ding, C.; Chan, Z.; Choo, J.; Sadananthan, S.A.; Michael, N.; Lee, Y.; Velan, S.S.; Magkos, F. Moderate Weight Loss Improves Body Composition and Metabolic Function in Metabolically Unhealthy Lean Subjects. Obesity (Silver Spring, MD) 2018, 26, 1000–1007. [Google Scholar] [CrossRef]
- Ding, C.; Chan, Z.; Chooi, Y.C.; Choo, J.; Sadananthan, SA.; Chang, A.; Sasikala, S.; Michael, N.; Velan, S.S.; Magkos, F. Regulation of glucose metabolism in nondiabetic, metabolically obese normal-weight Asians. Am. J. Physiol. Endocrinol. Metab. 2018, 314, E494–E502. [Google Scholar] [CrossRef]
- Rattarasarn, C. Dysregulated lipid storage and its relationship with insulin resistance and cardiovascular risk factors in non-obese Asian patients with type 2 diabetes. Adipocyte 2018, 7, 71–80. [Google Scholar] [CrossRef] [PubMed]
- Furukawa, Y.; Tamura, Y.; Takeno, K.; Funayama, T.; Kaga, H.; Suzuki, R.; Watanabe, T.; Kakehi, S.; Kanazawa, A.; Kawamori, R.; et al. Impaired peripheral insulin sensitivity in non-obese Japanese patients with type 2 diabetes mellitus and fatty liver. J. Diabetes Investig. 2017, 9, 529–535. [Google Scholar] [CrossRef] [PubMed]
- Kadowaki, S.; Tamura, Y.; Someya, Y.; Takeno, K.; Kaga, H.; Sugimoto, D.; Kakehi, S.; Funayama, T.; Furukawa, Y.; Suzuki, R.; et al. Fatty liver has stronger association with insulin resistance than visceral fat accumulation in nonobese Japanese men. J. Endocr. Soc. 2019, 3, 1409–1416. [Google Scholar] [CrossRef]
- The Japan Society of Hepatology. Medical Treatment Guideline for NASH · NAFLD, 1st ed.; Bunkodo: Tokyo, Japan, 2015; pp. 2–7. ISBN 978-4-8306-1888-8. [Google Scholar]
- Examination Committee of Criteria for ‘Obesity Disease’ in Japan. New criteria for ‘obesity disease’ in Japan. Circ. J. 2002, 66, 987–992.
- Sterling, R.K.; Lissen, E.; Clumeck, N.; Sola, R.; Correa, M.C.; Montaner, J.; Sulkowski, M.S.; Torriani, F.J.; Dieterich, D.T.; Thomas, D.L.; et al. Development of a simple noninvasive index to predict significant fibrosis in patients with HIV/HCV coinfection. Hepatology 2006, 43, 1317–1325. [Google Scholar] [CrossRef]
- Sumida, Y.; Yoneda, M.; Hyogo, H.; Itoh, Y.; Ono, M.; Fujii, H.; Eguchi, Y.; Suzuki, Y.; Aoki, N.; Kanemasa, K.; et al. Validation of the FIB4 index in a Japanese nonalcoholic fatty liver disease population. BMC Gastroenterol. 2012, 12, 2. [Google Scholar] [CrossRef]
- Nazare, J.A.; Smith, J.D.; Borel, A.L.; Haffner, S.M.; Balkau, B.; Ross, R.; Massien, C.; Alméras, N.; Després, J.-P. Ethnic influences on the relations between abdominal subcutaneous and visceral adiposity, liver fat, and cardiometabolic risk profile: The international study of prediction of intra-abdominal adiposity and its relationship with cardiometabolic risk/intra-abdominal adiposity. Am. J. Clin. Nutr. 2012, 96, 714–726. [Google Scholar]
- Tobari, M.; Hashimoto, E.; Taniai, M.; Ikarashi, Y.; Kodama, K.; Kogiso, T.; Tokushige, K.; Takayoshi, N.; Hashimoto, N. Characteristics of non-alcoholic steatohepatitis among lean patients in Japan: Not uncommon and not always benign. J. Gastroenterol. Hepatol. 2019, 34, 1404–1410. [Google Scholar] [CrossRef]
- Tobari, M.; Hashimoto, E. Characteristic features of nonalcoholic fatty liver disease in Japan with a focus on the roles of age, sex and body mass index. Gut Liver 2020. [Google Scholar] [CrossRef]
- Eguchi, Y.; Hyogo, H.; Ono, M.; Mizuta, T.; Ono, N.; Fujimoto, K.; Chayama, K.; Saibara, T. Prevalence and associated metabolic factors of nonalcoholic fatty liver disease in the general population from 2009 to 2010 in Japan: A multicenter large retrospective study. J. Gastroenterol. 2012, 47, 586–595. [Google Scholar] [CrossRef] [PubMed]
- Ye, Q.; Zou, B.; Yeo, Y.H.; Li, J.; Huang, D.Q.; Wu, Y.; Yang, H.; Liu, C.; Kam, L.Y.; Tan, X.X.E.; et al. Global prevalence, incidence, and outcomes of non-obese or lean non-alcoholic fatty liver disease: A systematic review and meta-analysis. Lancet Gastroenterol. Hepatol. 2020. [Google Scholar] [CrossRef]
- Younes, R.; Bugianesi, E. NASH in lean individuals. Semin. Liver Dis. 2019, 39, 86–95. [Google Scholar] [CrossRef] [PubMed]
- Cusi, K. Nonalcoholic steatohepatitis in nonobese patients: Not so different after all. Hepatology 2017, 65, 4–7. [Google Scholar] [CrossRef] [PubMed]
- Albhaisi, S.; Chowdhury, A.; Sanyal, A.J. Non-alcoholic fatty liver disease in lean individuals. JHEP Rep. 2019, 1, 329–341. [Google Scholar] [CrossRef]
- Leung, J.C.; Loong, T.C.; Wei, J.L.; Wong, G.L.; Chan, A.W.; Choi, P.C.; Shu, S.S.-T.; Chim, A.M.; Chan, H.L.-Y.; Wong, V.W. Histological severity and clinical outcomes of nonalcoholic fatty liver disease in nonobese patients. Hepatology 2017, 65, 54–64. [Google Scholar] [CrossRef]
- Zou, B.; Yeo, Y.H.; Nguyen, V.H.; Cheung, R.; Ingelsson, E.; Nguyen, M.H. Prevalence, characteristics and mortality outcomes of obese, nonobese and lean NAFLD in the United States, 1999–2016. J. Intern. Med. 2020, 288, 139–151. [Google Scholar] [CrossRef]
- Angulo, P.; Kleiner, D.E.; Dam-Larsen, S.; Adams, L.A.; Bjornsson, E.S.; Charatcharoenwitthaya, P.; Mills, P.R.; Keach, J.C.; Lafferty, H.D.; Stahler, A.; et al. Liver fibrosis, but no other histologic features, is associated with long-term outcomes of patients with nonalcoholic fatty liver disease. Gastroenterology 2015, 149, 389–397.e10. [Google Scholar] [CrossRef]
- Younossi, Z.M.; Koenig, A.B.; Abdelatif, D.; Fazel, Y.; Henry, L.; Wymer, M. Global epidemiology of nonalcoholic fatty liver disease-Meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology 2016, 64, 73–84. [Google Scholar] [CrossRef]
- Dulai, P.S.; Singh, S.; Patel, J.; Soni, M.; Prokop, L.J.; Younossi, Z.; Sebastiani, G.; Ekstedt, M.; Hagstrom, H.; Nasr, P.; et al. Increased risk of mortality by fibrosis stage in nonalcoholic fatty liver disease: Systematic review and meta-analysis. Hepatology 2017, 65, 1557–1565. [Google Scholar] [CrossRef]
- Hagström, H.; Nasr, P.; Ekstedt, M.; Hammar, U.; Stål, P.; Hultcrantz, R.; Kechagias, S. Fibrosis stage but not NASH predicts mortality and time to development of severe liver disease in biopsy-proven NAFLD. J. Hepatol. 2017, 67, 1265–1273. [Google Scholar] [CrossRef] [PubMed]
- Yoshitaka, H.; Hamaguchi, M.; Kojima, T.; Fukuda, T.; Ohbora, A.; Fukui, M. Nonoverweight nonalcoholic fatty liver disease and incident cardiovascular disease: A post hoc analysis of a cohort study. Medicine (Baltimore) 2017, 96, e6712. [Google Scholar] [CrossRef] [PubMed]
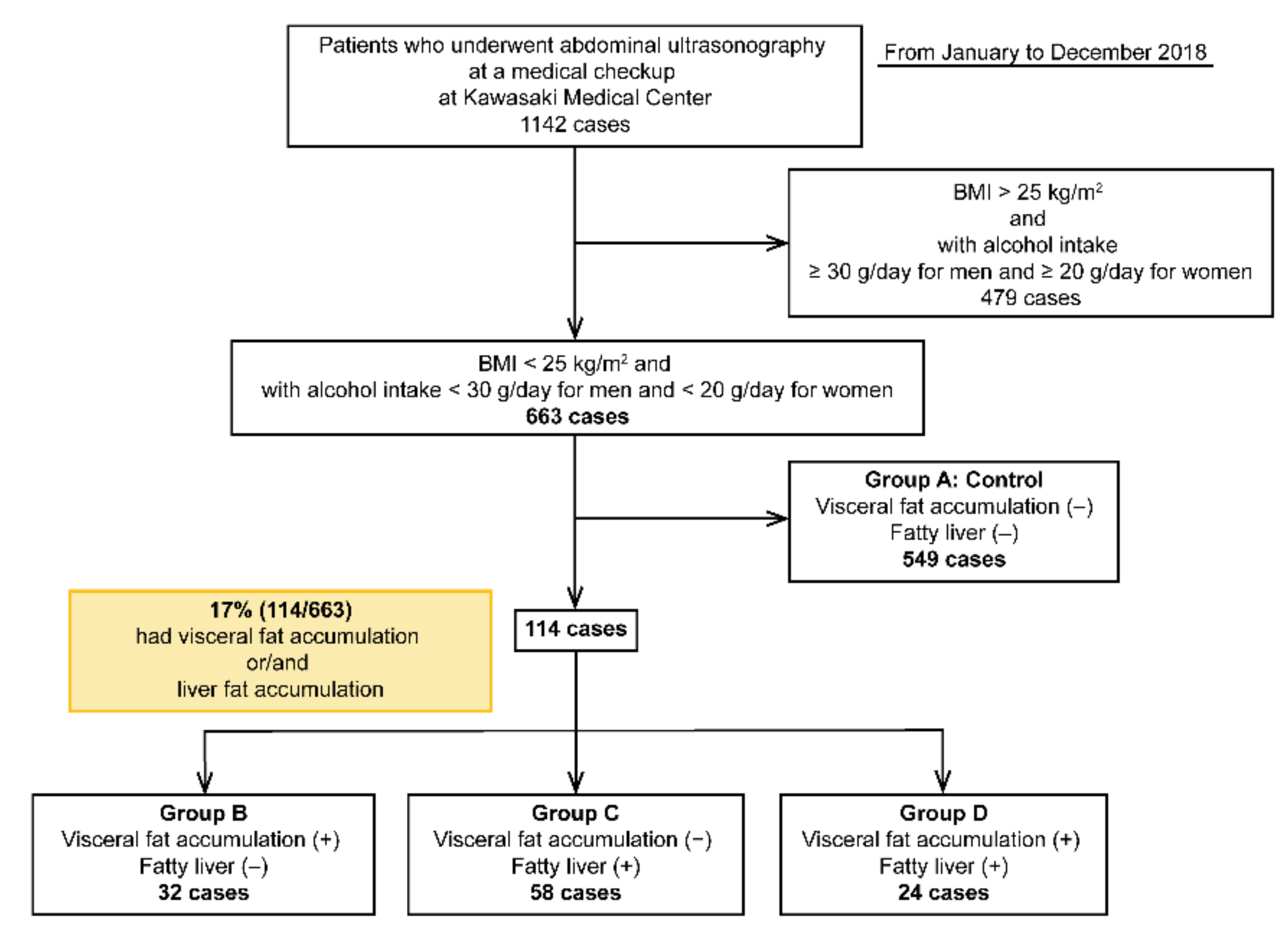
| Number of Patients | 663 |
|---|---|
| Age (years) | 50 (22–84) |
| Gender (M/F) | 299/364 |
| BMI (kg/m2) | 21.5 (13–24.9) |
| Body fat percentage (%) | 23.4 (6.6–39.8) |
| Abdominal circumference (cm) | |
| Men (n = 299) | 80.4 (63.8–96.8) |
| Women (n = 364) | 75.8 (58.2–93.5) |
| Diabetes (+/−) | 25/638 |
| Dyslipidemia (+/−) | 212/451 |
| Hypertension (+/−) | 88/575 |
| ALT (IU/L) | 16 (4–132) |
| AST (IU/L) | 20 (9–119) |
| Γ-GTP (IU/L) | 19 (6–338) |
| T-Bilirubin (mg/dL) | 0.9 (0.4–3.2) |
| Albumin (g/dL) | 4.5 (3.2–5.4) |
| Platelet count (104/µg) | 20.6 (9.4–40.1) |
| Cholinesterase (IU/L) | 327 (164–562) |
| Total cholesterol (ng/dL) | 207 (125–316) |
| Triglyceride (ng/dL) | 73 (21–958) |
| LDL-C (ng/dL) | 118 (32–207) |
| Uric acid | 5.0 (1.9–9.4) |
| Fasting blood sugar | 96 (71–296) |
| HbA1c (%) | 5.6 (4.9–12.2) |
| FIB4 index | 0.9 (0.17–5.56) |
| FIB4 index (<1.3/1.3–2.66/ ≥2.67) | 517/134/12 |
| Group A Control | Group B VFA (+) FL (−) | Group C VFA (−) FL (+) | Group D VFA (+) FL (+) | p-Value | |
|---|---|---|---|---|---|
| Number of patients | 549 | 32 | 58 | 24 | |
| Age (years) | 48.5 ± 10.9 | 48.8 ± 10.9 | 52.7 ± 11.1 b | 55.1 ± 12.4 c | 0.0026 |
| Gender, male (%) | 38.6 | 87.5 a | 62 b,d | 95.8 c,f | <0.0001 |
| BMI (kg/m2) | 20.9 ± 2.2 | 23.3 ± 1.0 a | 23.1 ± 1.3 b | 23.9 ± 0.8 c,e | <0.0001 |
| Body fat percentage (%) | 24.0 ± 6.7 | 25.4 ± 5 | 26.4 ± 6.7 | 23.4 ± 3.1 | 0.0543 |
| Abdominal circumference (cm) | |||||
| Men (n = 299) (212/28/36/23) | 78.0 ± 4.9 | 87.6 ± 1.9 a | 83.2 ± 3.9 b,d | 88.7 ± 3.2 c,f | <0.0001 |
| Women (n = 364) (337/4/22/1) | 74.8 ± 7.6 | 89.0 ± 2.7 a | 82.2 ± 5.0 b,d | 93.5 | <0.0001 |
| Group A Control | Group B VFA (+) FL (-) | Group C VFA (-) FL (+) | Group D VFA (+) FL (+) | p-Value | |
|---|---|---|---|---|---|
| Number of patients | 549 | 32 | 58 | 24 | |
| Diabetes (+/−) | 8/541 (1.5%) | 0/32 (0%) | 11/47 (19%) b,d | 6/18 (25%) c,e | <0.0001 |
| Dyslipidemia (+/−) | 152/397 (38%) | 17/15 (53%) a | 31/27 (53%) b | 12/12 (50%) c | <0.0001 |
| Hypertension (+/−) | 61/488 (12%) | 6/26 (18%) | 15/43 (25%) b | 6/18 (25%) c | 0.0074 |
| Group A Control | Group B VFA (+) FL (−) | Group C VFA (−) FL (+) | Group D VFA (+) FL (+) | p-Value | |
|---|---|---|---|---|---|
| Number of subjects | 549 | 32 | 58 | 24 | |
| ALT (IU/L) | 17 ± 10 | 22 ± 9 a | 28 ± 14 b,d | 36 ± 21 c,e | <0.0001 |
| AST (IU/L) | 20 ± 7 | 22 ± 9 a | 24 ± 6.2 b | 26 ± 11 c,f | <0.0001 |
| Γ-GTP (IU/L) | 25 ± 24 | 50 ± 43 a | 35 ± 20 b,d | 64 ± 79 c | <0.0001 |
| T-Bilirubin (mg/dL) | 1.0 ± 0.4 | 1.1 ± 0.5 | 0.9 ± 0.4 | 1.0 ± 0.3 | 0.1599 |
| Albumin (g/dL) | 4.5 ± 0.3 | 4.5 ± 0.3 | 4.6 ± 0.2 | 4.5 ± 0.3 | 0.0163 |
| Platelet count (104/µg) | 21 ± 4.8 | 21 ± 4.6 | 22 ± 4.8 | 21 ± 4.7 | 0.2361 |
| Cholinesterase (IU/L) | 327 ± 68 | 348 ± 59 | 394 ± 68 b,d | 380 ± 56 c,e | <0.0001 |
| Total cholesterol (ng/dL) | 208 ± 34 | 208 ± 32 | 212 ± 35 | 199 ± 32 | 0.6883 |
| Triglyceride (ng/dL) | 79 ± 44 | 114 ± 93 a | 145 ± 123 b,d | 138 ± 111 c,e | <0.0001 |
| LDL-C (ng/dL) | 119 ± 23 | 126 ± 31 | 125 ± 29 | 117 ± 28 | 0.1247 |
| Uric acid | 5.0 ± 1.3 | 6.0 ± 1.1 a | 5.7 ± 1.1 b | 6.1 ± 1.2 c | <0.0001 |
| Fasting blood sugar | 96 ± 13 | 99 ± 7.9 a | 106 ± 15 b | 105 ± 16 c | <0.0001 |
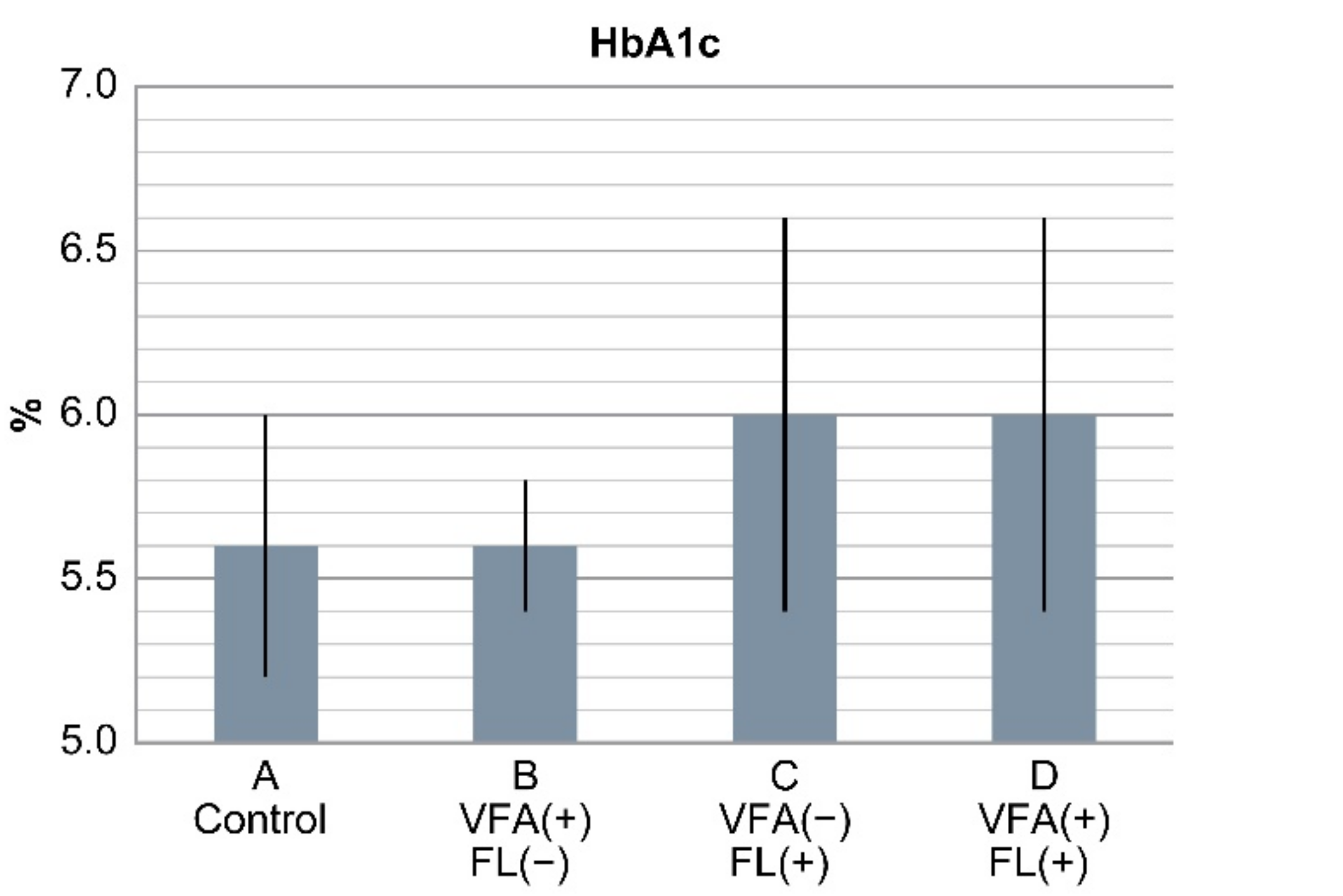
| HbA1c (%) | 5.6 ± 0.4 | 5.6 ± 0.2 | 6.0 ± 0.6 b,d | 6.0 ± 0.6 c,e | <0.0001 |
| Group A Control | Group B VFA (+) FL (−) | Group C VFA (−) FL (+) | Group D VFA (+) FL (+) | p-Value | |
|---|---|---|---|---|---|
| Number of patients | 549 | 32 | 58 | 24 | |
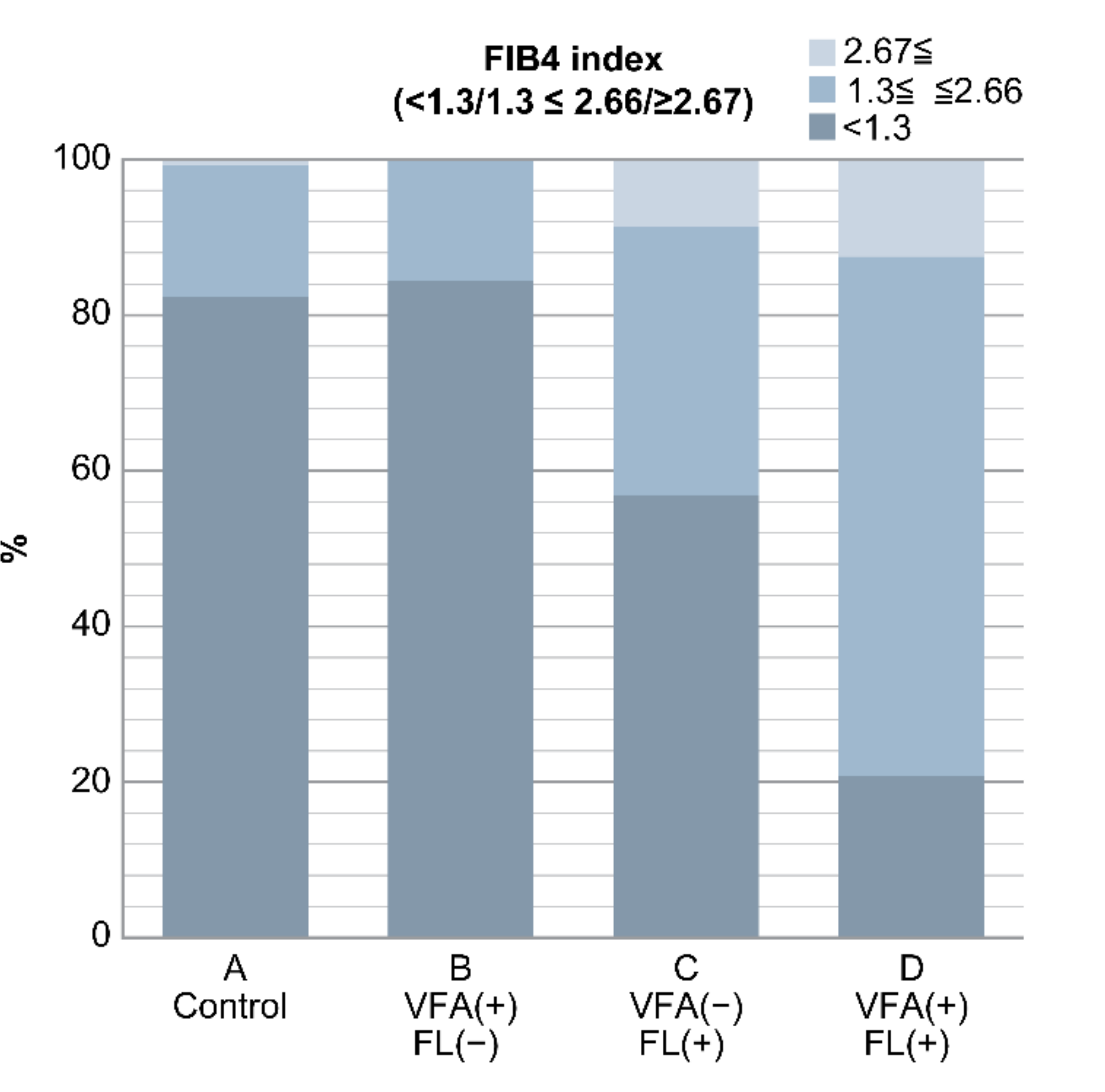
| FIB4 index | 0.9 ± 0.05 | 1.0 ± 0.3 | 1.4 ± 0.7 b,d | 1.8 ± 0.8 c,e,f | <0.0001 |
| FIB4 index (<1.3/1.3–2.66/ ≥ 2.67) | 452/93/4 | 27/5/0 | 33/20/5 b,d | 5/16/3 c,e,f | <0.0001 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Urata, N.; Kawanaka, M.; Nishino, K.; Ishii, K.; Tanikawa, T.; Suehiro, M.; Sasai, T.; Haruma, K.; Kawamoto, H.; Nakamura, J.; et al. Fatty Liver, and Not Visceral Fat, Is More Associated with Liver Fibrosis and Diabetes in Non-Obese Japanese Individuals: A Cross-Sectional Study. Life 2020, 10, 175. https://doi.org/10.3390/life10090175
Urata N, Kawanaka M, Nishino K, Ishii K, Tanikawa T, Suehiro M, Sasai T, Haruma K, Kawamoto H, Nakamura J, et al. Fatty Liver, and Not Visceral Fat, Is More Associated with Liver Fibrosis and Diabetes in Non-Obese Japanese Individuals: A Cross-Sectional Study. Life. 2020; 10(9):175. https://doi.org/10.3390/life10090175
Chicago/Turabian StyleUrata, Noriyo, Miwa Kawanaka, Ken Nishino, Katsunori Ishii, Tomohiro Tanikawa, Mitsuhiko Suehiro, Takako Sasai, Ken Haruma, Hirofumi Kawamoto, Jun Nakamura, and et al. 2020. "Fatty Liver, and Not Visceral Fat, Is More Associated with Liver Fibrosis and Diabetes in Non-Obese Japanese Individuals: A Cross-Sectional Study" Life 10, no. 9: 175. https://doi.org/10.3390/life10090175
APA StyleUrata, N., Kawanaka, M., Nishino, K., Ishii, K., Tanikawa, T., Suehiro, M., Sasai, T., Haruma, K., Kawamoto, H., Nakamura, J., Manabe, N., & Kamada, T. (2020). Fatty Liver, and Not Visceral Fat, Is More Associated with Liver Fibrosis and Diabetes in Non-Obese Japanese Individuals: A Cross-Sectional Study. Life, 10(9), 175. https://doi.org/10.3390/life10090175




