Preventive Moderate Continuous Running-Exercise Conditioning Improves the Healing of Non-Critical Size Bone Defects in Male Wistar Rats: A Pilot Study Using µCT
Abstract
1. Introduction
2. Materials and Methods
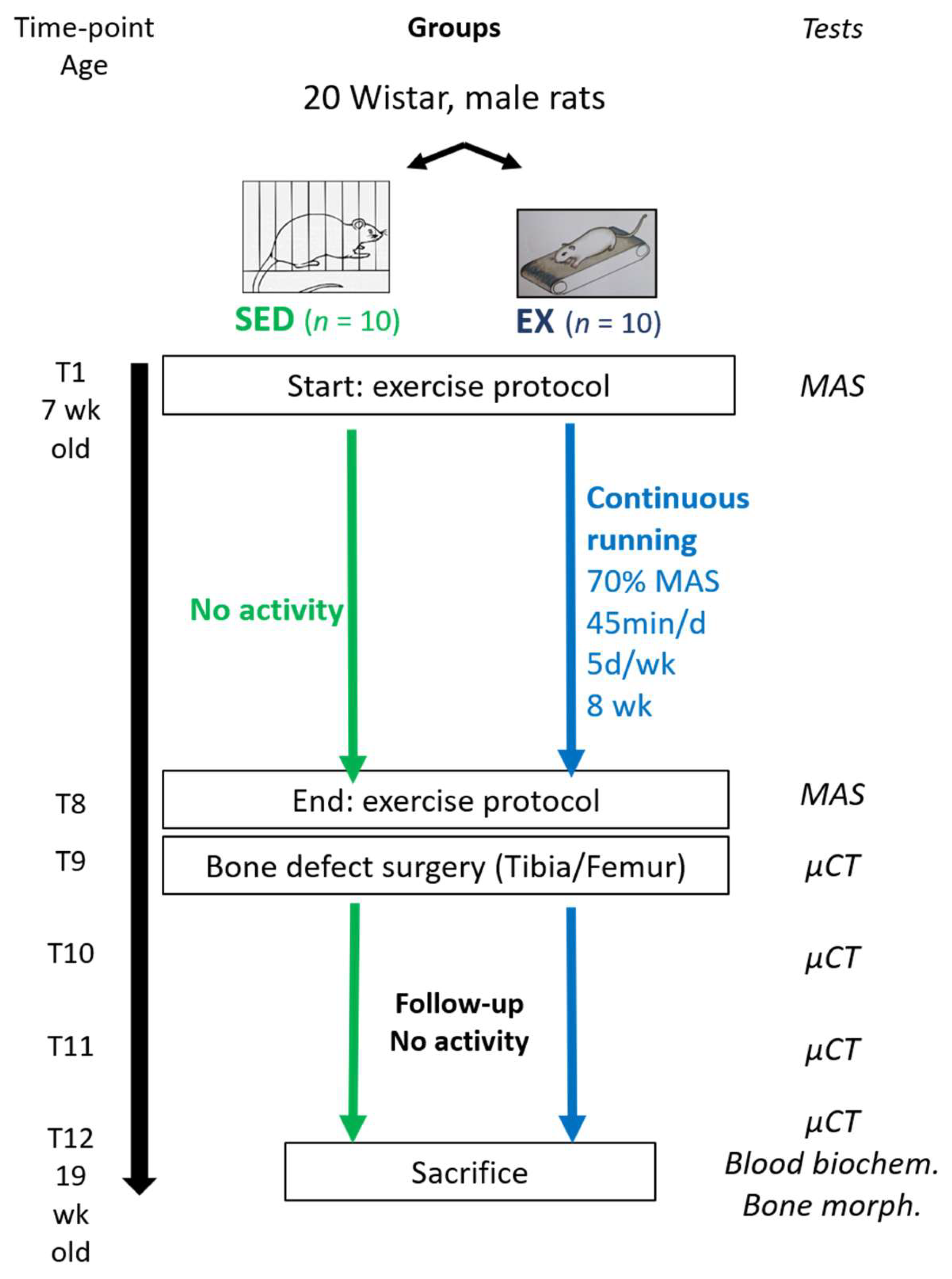
2.1. Animals
2.2. Experimental Design
2.3. Body Parameters, Food and Water Follow-Up
2.4. Exercise Training Protocols
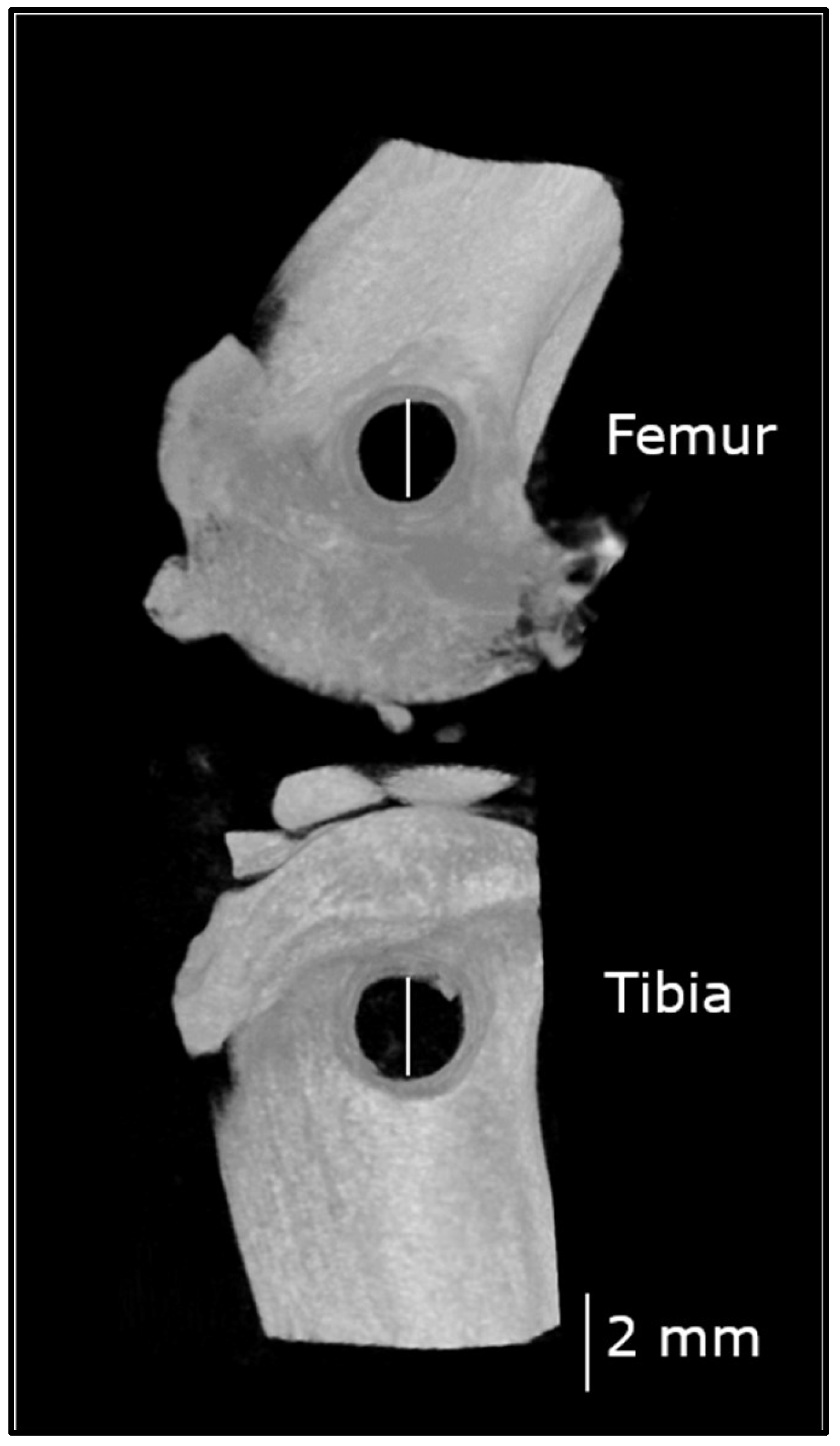
2.5. Non-Critical Sized Bone Defect Surgery
2.5.1. Anesthesia
2.5.2. Surgical Procedure
2.6. Postoperative Microcomputed Tomography (µCT) and Computation
2.6.1. µCT Settings
2.6.2. µCT Reconstruction and Data Computation
2.7. Blood Analyses
2.8. Data Analysis and Statistical Analysis
3. Results
3.1. Body Parameters, Food and Water Consumption
3.2. Exercise Training
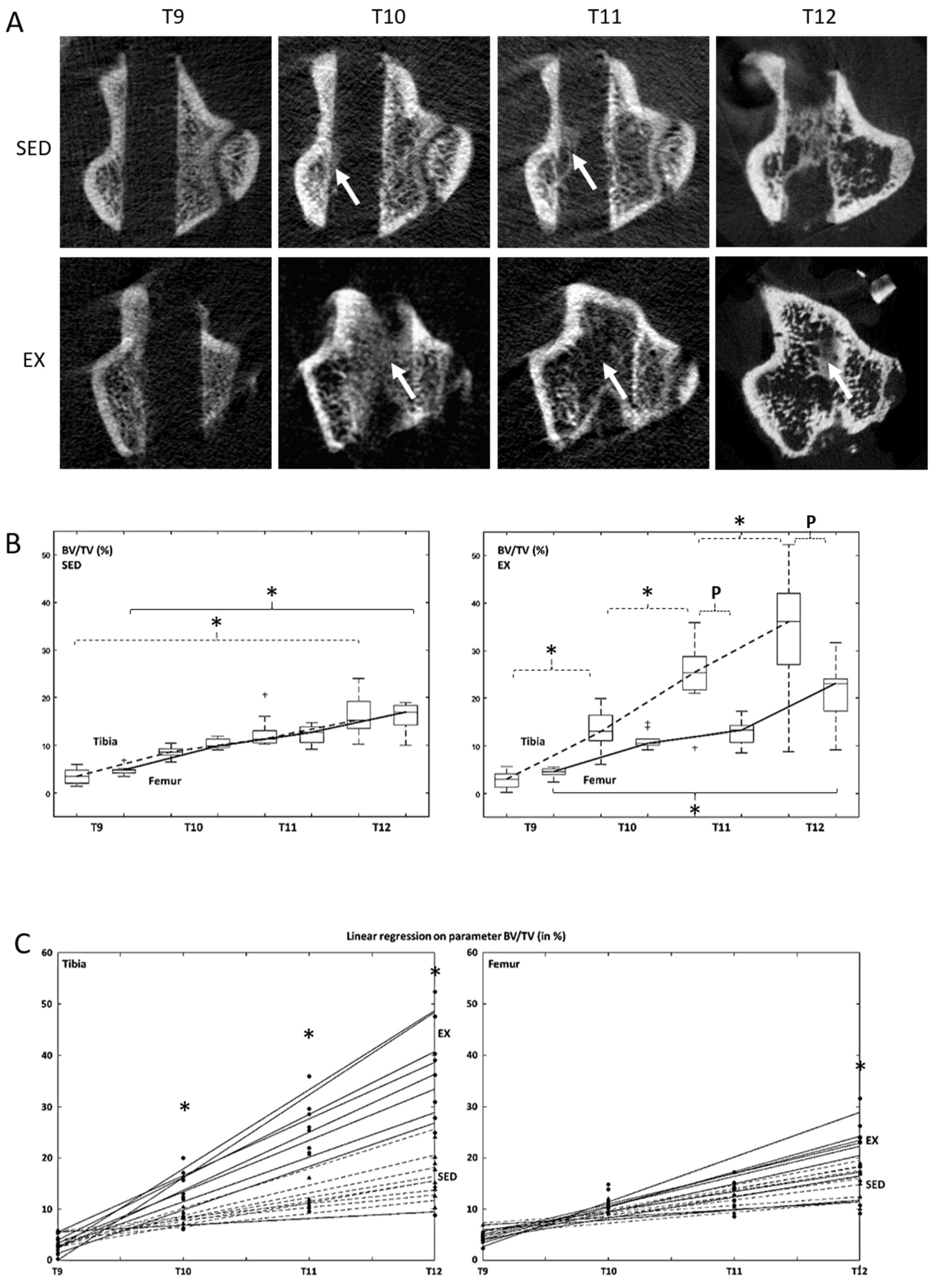
3.3. Microcomputed Tomography Bone Formation and Bone Mineral Density Assessment
3.4. Blood Analysis Parameters
4. Discussion
4.1. Preemptive MCR Improves Bone Healing Tissue Microarchitecture and Mineral Density
4.2. Effects of Preemptive MCR on Bone Healing Depend on the Bone Studied
4.3. Putative Explanations Pertaining to the Positive Effects of MCR on Bone Healing
4.4. Limitations
4.5. Perspectives in Humans
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cheung, A.M.; Giangregorio, L. Mechanical stimuli and bone health: What is the evidence? Curr. Opin. Rheumatol. 2012, 24, 561–566. [Google Scholar] [CrossRef] [PubMed]
- Maïmoun, L.; Sultan, C. Effects of physical activity on bone remodeling. Metabolism 2011, 60, 373–388. [Google Scholar] [CrossRef] [PubMed]
- Troy, K.; Mancuso, M.; Butler, T.; Johnson, J. Exercise Early and Often: Effects of Physical Activity and Exercise on Women’s Bone Health. Int. J. Environ. Res. Public Health 2018, 15, 878. [Google Scholar] [CrossRef] [PubMed]
- Hong, A.R.; Kim, S.W. Effects of Resistance Exercise on Bone Health. Endocrinol. Metab. 2018, 33, 435. [Google Scholar] [CrossRef]
- Boudenot, A.; Achiou, Z.; Portier, H. Does running strengthen bone? Appl. Physiol. Nutr. Metab. 2015, 40, 1309–1312. [Google Scholar] [CrossRef]
- Huang, T.H.; Lin, S.C.; Chang, F.L.; Hsieh, S.S.; Liu, S.H.; Yang, R.S. Effects of different exercise modes on mineralization, structure, and biomechanical properties of growing bone. J. Appl. Physiol. 2003, 95, 300–307. [Google Scholar] [CrossRef]
- Honda, A.; Sogo, N.; Nagasawa, S.; Shimizu, T.; Umemura, Y. High-impact exercise strengthens bone in osteopenic ovariectomized rats with the same outcome as Sham rats. J. Appl. Physiol. 2003, 95, 1032–1037. [Google Scholar] [CrossRef]
- Notomi, T.; Okimoto, N.; Okazaki, Y.; Tanaka, Y.; Nakamura, T.; Suzuki, M. Effects of tower climbing exercise on bone mass, strength, and turnover in growing rats. J. Bone Miner. Res. Off. J. Am. Soc. Bone Miner. Res. 2001, 16, 166–174. [Google Scholar] [CrossRef]
- Bourrin, S.; Palle, S.; Pupier, R.; Vico, L.; Alexandre, C. Effect of physical training on bone adaptation in three zones of the rat tibia. J. Bone Miner. Res. Off. J. Am. Soc. Bone Miner. Res. 1995, 10, 1745–1752. [Google Scholar] [CrossRef]
- Joo, Y.-I.; Sone, T.; Fukunaga, M.; Lim, S.-G.; Onodera, S. Effects of endurance exercise on three-dimensional trabecular bone microarchitecture in young growing rats. Bone 2003, 33, 485–493. [Google Scholar] [CrossRef]
- Iwamoto, J.; Takeda, T.; Sato, Y. Effect of treadmill exercise on bone mass in female rats. Exp. Anim. 2005, 54, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Iwamoto, J.; Yeh, J.K.; Aloia, J.F. Differential effect of treadmill exercise on three cancellous bone sites in the young growing rat. Bone 1999, 24, 163–169. [Google Scholar] [CrossRef]
- Hagihara, Y.; Nakajima, A.; Fukuda, S.; Goto, S.; Iida, H.; Yamazaki, M. Running exercise for short duration increases bone mineral density of loaded long bones in young growing rats. Tohoku J. Exp. Med. 2009, 219, 139–143. [Google Scholar] [CrossRef] [PubMed]
- Iwamoto, J.; Shimamura, C.; Takeda, T.; Abe, H.; Ichimura, S.; Sato, Y.; Toyama, Y. Effects of treadmill exercise on bone mass, bone metabolism, and calciotropic hormones in young growing rats. J. Bone Miner. Metab. 2004, 22, 26–31. [Google Scholar] [CrossRef]
- Horcajada-Molteni, M.N.; Davicco, M.J.; Collignon, H.; Lebecque, P.; Coxam, V.; Barlet, J.P. Does endurance running before orchidectomy prevent osteopenia in rats? Eur. J. Appl. Physiol. 1999, 80, 344–352. [Google Scholar] [CrossRef]
- Davicco, M.J.; Horcajada-Molteni, M.N.; Gaumet-Meunier, N.; Lebecque, P.; Coxam, V.; Barlet, J.P. Endurance training and bone metabolism in middle-aged rats. Mech. Ageing Dev. 1999, 109, 83–96. [Google Scholar] [CrossRef]
- Iijima, H.; Aoyama, T.; Ito, A.; Yamaguchi, S.; Nagai, M.; Tajino, J.; Zhang, X.; Kuroki, H. Effects of short-term gentle treadmill walking on subchondral bone in a rat model of instability-induced osteoarthritis. Osteoarthr. Cartil. 2015, 23, 1563–1574. [Google Scholar] [CrossRef]
- Lavorato, V.N.; Del Carlo, R.J.; da Cunha, D.N.Q.; Okano, B.S.; Belfort, F.G.; de Freitas, J.S.; de Fatima Alves da Mota, G.; Quintão-Júnior, J.F.; Silame-Gomes, L.H.L.; Drummond, F.R.; et al. Mesenchymal stem cell therapy associated with endurance exercise training: Effects on the structural and functional remodeling of infarcted rat hearts. J. Mol. Cell. Cardiol. 2016, 90, 111–119. [Google Scholar] [CrossRef]
- Yamaguchi, S.; Aoyama, T.; Ito, A.; Nagai, M.; Iijima, H.; Tajino, J.; Zhang, X.; Kiyan, W.; Kuroki, H. The Effect of Exercise on the Early Stages of Mesenchymal Stromal Cell-Induced Cartilage Repair in a Rat Osteochondral Defect Model. PLoS ONE 2016, 11, e0151580. [Google Scholar] [CrossRef]
- Guerino, M.R.; Luciano, E.; Goncalves, M.; Leivas, T.P. Effects of chronic physical activity and of ultrasound treatment on bone consolidation. Physiol. Chem. Phys. Med. NMR 1999, 31, 131–138. [Google Scholar]
- Rueff-Barroso, C.R.; Milagres, D.; do Valle, J.; Casimiro-Lopes, G.; Nogueira-Neto, J.F.; Zanier, J.F.C.; Porto, L.C. Bone healing in rats submitted to weight-bearing and non-weight-bearing exercises. Med. Sci. Monit. Int. Med. J. Exp. Clin. Res. 2008, 14, BR231–BR236. [Google Scholar]
- Zhang, J.; He, F.; Zhang, W.; Zhang, M.; Yang, H.; Luo, Z.-P. Mechanical force enhanced bony formation in defect implanted with calcium sulphate cement. Bone Res. 2015, 3, 14048. [Google Scholar] [CrossRef] [PubMed]
- Maurel, D.B.; Boisseau, N.; Pallu, S.; Rochefort, G.Y.; Benhamou, C.-L.; Jaffre, C. Regular exercise limits alcohol effects on trabecular, cortical thickness and porosity, and osteocyte apoptosis in the rat. Jt. Bone Spine Rev. Rhum. 2013, 80, 492–498. [Google Scholar] [CrossRef] [PubMed]
- Bonnet, N.; Beaupied, H.; Vico, L.; Dolleans, E.; Laroche, N.; Courteix, D.; Benhamou, C.L. Combined effects of exercise and propranolol on bone tissue in ovariectomized rats. J. Bone Miner. Res. Off. J. Am. Soc. Bone Miner. Res. 2007, 22, 578–588. [Google Scholar] [CrossRef]
- Heikkinen, E.; Vihersaari, T.; Penttinen, R. Effect of Previous Exercise on Fracture Healing: A Biochemical Study with Mice. Acta Orthop. Scand. 1974, 45, 481–489. [Google Scholar] [CrossRef]
- Nogueira, J.E.; Branco, L.G.S.; Issa, J.P.M. Bone repair: Effects of physical exercise and LPS systemic exposition. Injury 2016, 47, 1828–1834. [Google Scholar] [CrossRef]
- Novelli, E.L.B.; Diniz, Y.S.; Galhardi, C.M.; Ebaid, G.M.X.; Rodrigues, H.G.; Mani, F.; Fernandes, A.A.H.; Cicogna, A.C.; Novelli Filho, J.L.V.B. Anthropometrical parameters and markers of obesity in rats. Lab. Anim. 2007, 41, 111–119. [Google Scholar] [CrossRef]
- Chennaoui, M.; Drogou, C.; Gomez-Merino, D.; Grimaldi, B.; Fillion, G.; Guezennec, C.Y. Endurance training effects on 5-HT1B receptors mRNA expression in cerebellum, striatum, frontal cortex and hippocampus of rats. Neurosci. Lett. 2001, 307, 33–36. [Google Scholar] [CrossRef]
- Krzesiak, A.; Cognard, C.; Sebille, S.; Carré, G.; Bosquet, L.; Delpech, N. High-intensity intermittent training is as effective as moderate continuous training, and not deleterious, in cardiomyocyte remodeling of hypertensive rats. J. Appl. Physiol. 2019, 126, 903–915. [Google Scholar] [CrossRef]
- Pica, A.J.; Brooks, G.A. Effects of training and age on VO2max in laboratory rats. Med. Sci. Sports Exerc. 1982, 14, 249–252. [Google Scholar] [CrossRef]
- Hulsart-Billström, G.; Bergman, K.; Andersson, B.; Hilborn, J.; Larsson, S.; Jonsson, K.B. A uni-cortical femoral defect model in the rat: Evaluation using injectable hyaluronan hydrogel as a carrier for bone morphogenetic protein-2: Bone regeneration in a uni-cortical rat femoral defect model. J. Tissue Eng. Regen. Med. 2015, 9, 799–807. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, T.; Goto, D.; Sato, S. Subtraction micro-computed tomography of angiogenesis and osteogenesis during bone repair using synchrotron radiation with a novel contrast agent. Lab. Investig. 2013, 93, 1054–1063. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, T.; Goto, D. Effect of low-intensity whole-body vibration on bone defect repair and associated. vascularization in mice. Med. Biol. Eng. Comput 2017, 55, 2257–2266. [Google Scholar] [CrossRef] [PubMed]
- Parfitt, A.M.; Drezner, M.K.; Glorieux, F.H.; Kanis, J.A.; Malluche, H.; Meunier, P.J.; Ott, S.M.; Recker, R.R. Bone histomorphometry: Standardization of nomenclature, symbols, and units: Report of the asbmr histomorphometry nomenclature committee. J. Bone Miner. Res. 2009, 2, 595–610. [Google Scholar] [CrossRef] [PubMed]
- Bouxsein, M.L.; Boyd, S.K.; Christiansen, B.A.; Guldberg, R.E.; Jepsen, K.J.; Müller, R. Guidelines for assessment of bone microstructure in rodents using micro-computed tomography. J. Bone Miner. Res. 2010, 25, 1468–1486. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Dong, X.; Leung, P.-C.; Wong, M.-S. Differential mRNA expression profiles in proximal tibia of aged rats in response to ovariectomy and low-Ca diet. Bone 2009, 44, 46–52. [Google Scholar] [CrossRef]
- Schultz, V.L.; Boass, A.; Garner, S.C.; Toverud, S.U. Several anesthetics, but not diethyl ether, cause marked elevation of serum parathyroid hormone concentration in rats. J. Bone Miner. Res. Off. J. Am. Soc. Bone Miner. Res. 1995, 10, 1298–1302. [Google Scholar] [CrossRef]
- Milliken, G.A.; Johnson, D.E. Analysis of Messy Data. Volume 1: Designed Experiments; Chapman & Hall/CRC Press: Boca Raton, FL, USA, 1992. [Google Scholar]
- Warden, S.J.; Galley, M.R.; Hurd, A.L.; Richard, J.S.; George, L.A.; Guildenbecher, E.A.; Barker, R.G.; Fuchs, R.K. Cortical and Trabecular Bone Benefits of Mechanical Loading Are Maintained Long Term in Mice Independent of Ovariectomy. J. Bone Miner. Res. 2014, 29, 1131–1140. [Google Scholar] [CrossRef]
- Hestehave Pedersen, R.; Rasmussen, M.; Overgaard, S.; Ding, M. Effects of P-15 Peptide Coated Hydroxyapatite on Tibial Defect Repair In Vivo in Normal and Osteoporotic Rats. BioMed Res. Int. 2015, 2015, 253858. [Google Scholar] [CrossRef]
- Xu, X.; Zhou, J.; Yang, F.; Wei, S.; Dai, H. Using Micro-Computed Tomography to Evaluate the Dynamics of Orthodontically Induced Root Resorption Repair in a Rat Model. PLoS ONE 2016, 11, e0150135. [Google Scholar] [CrossRef]
- Hildebrand, T.; Rüegsegger, P. Quantification of Bone Microarchitecture with the Structure Model Index. Comput. Methods Biomech. Biomed. Engin. 1997, 1, 15–23. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, K.R.; Stutz, C.M.; Mignemi, N.A.; Burns, M.C.; Murry, M.R.; Nyman, J.S.; Schoenecker, J.G. Micro-computed tomography assessment of the progression of fracture healing in mice. Bone 2012, 50, 1357–1367. [Google Scholar] [CrossRef]
- Liu, Z.; Gao, J.; Gong, H. Effects of treadmill with different intensities on bone quality and muscle properties in adult rats. Biomed. Eng. Online 2019, 18, 107. [Google Scholar] [CrossRef] [PubMed]
- Song, S.J.; Hutmacher, D.; Nurcombe, V.; Cool, S.M. Temporal expression of proteoglycans in the rat limb during bone healing. Gene 2006, 379, 92–100. [Google Scholar] [CrossRef] [PubMed]
- Hagihara, Y.; Fukuda, S.; Goto, S.; Iida, H.; Yamazaki, M.; Moriya, H. How many days per week should rats undergo running exercise to increase BMD? J. Bone Miner. Metab. 2005, 23, 289–294. [Google Scholar] [CrossRef]
- Kohrt, W.M.; Wherry, S.J.; Wolfe, P.; Sherk, V.D.; Wellington, T.; Swanson, C.M.; Weaver, C.M.; Boxer, R.S. Maintenance of Serum Ionized Calcium During Exercise Attenuates Parathyroid Hormone and Bone Resorption Responses: Calcium clamp during exercice. J. Bone Miner. Res. 2018, 33, 1326–1334. [Google Scholar] [CrossRef]
- Gardinier, J.D.; Mohamed, F.; Kohn, D.H. PTH Signaling during Exercise Contributes to Bone Adaptation. J. Bone Miner. Res. 2015, 30, 1053–1063. [Google Scholar] [CrossRef]
- Bouassida, A.; Latiri, I.; Bouassida, S.; Zalleg, D.; Zaouali, M.; Feki, Y.; Gharbi, N.; Zbidi, A.; Tabka, Z. Parathyroid hormone and physical exercise: A brief review. J. Sports Sci. Med. 2006, 5, 367–374. [Google Scholar]
- Yamashita, J.; McCauley, L.K. Effects of Intermittent Administration of Parathyroid Hormone and Parathyroid Hormone-Related Protein on Fracture Healing: A Narrative Review of Animal and Human Studies. JBMR Plus 2019, 3, e10250. [Google Scholar] [CrossRef]
- Li, L.; Chen, X.; Lv, S.; Dong, M.; Zhang, L.; Tu, J.; Yang, J.; Zhang, L.; Song, Y.; Xu, L.; et al. Influence of exercise on bone remodeling-related hormones and cytokines in ovariectomized rats: A model of postmenopausal osteoporosis. PLoS ONE 2014, 9, e112845. [Google Scholar] [CrossRef]
- Henderson, S.A.; Graham, H.K.; Mollan, R.A.B.; Riddoch, C.; Sheridan, B.; Johnston, H. Calcium homeostasis and exercise. Int. Orthop. 1989, 13, 69–73. [Google Scholar] [CrossRef] [PubMed]
- Einhorn, T.A. The Cell and Molecular Biology of Fracture Healing. Clin. Orthop. 1998, 355S, S7–S21. [Google Scholar] [CrossRef] [PubMed]
- Wolff, J. The Law of Bone Remodelling; Springer: Berlin/Heidelberg, Germany, 1986; ISBN 978-3-642-71031-5. [Google Scholar]
- Marotti, G. The osteocyte as a wiring transmission system. J. Musculoskelet. Neuronal Interact. 2000, 1, 133–136. [Google Scholar] [PubMed]
- Skerry, T. Neurotransmitters in bone. Introduction. J. Musculoskelet. Neuronal Interact. 2002, 2, 401–403. [Google Scholar] [PubMed]
- Abou-Khalil, R.; Yang, F.; Lieu, S.; Julien, A.; Perry, J.; Pereira, C.; Relaix, F.; Miclau, T.; Marcucio, R.; Colnot, C. Role of Muscle Stem Cells during Skeletal Regeneration: Muscle Stem Cells in Bone Repair. Stem Cells 2015, 33, 1501–1511. [Google Scholar] [CrossRef] [PubMed]
- Bourzac, C.; Bensidhoum, M.; Pallu, S.; Portier, H. Use of adult mesenchymal stromal cells in tissue repair: Impact of physical exercise. Am. J. Physiol. Cell Physiol. 2019, 317, C642–C654. [Google Scholar] [CrossRef] [PubMed]
- Razi, H.; Birkhold, A.I.; Weinkamer, R.; Duda, G.N.; Willie, B.M.; Checa, S. Aging Leads to a Dysregulation in Mechanically Driven Bone Formation and Resorption: Mechanoregulation of (Re)Modeling. J. Bone Miner. Res. 2015, 30, 1864–1873. [Google Scholar] [CrossRef]
- Vicente, W.S.; dos Reis, L.M.; Graciolli, R.G.; Graciolli, F.G.; Dominguez, W.V.; Wang, C.C.; Fonseca, T.L.; Velosa, A.P.; Roschel, H.; Teodoro, W.R.; et al. Bone Plasticity in Response to Exercise Is Sex-Dependent in Rats. PLoS ONE 2013, 8, e64725. [Google Scholar] [CrossRef]
- Yuan, Y.; Chen, X.; Zhang, L.; Wu, J.; Guo, J.; Zou, D.; Chen, B.; Sun, Z.; Shen, C.; Zou, J. The roles of exercise in bone remodeling and in prevention and treatment of osteoporosis. Prog. Biophys. Mol. Biol. 2016, 122, 122–130. [Google Scholar] [CrossRef]
- Warner, S.E.; Shea, J.E.; Miller, S.C.; Shaw, J.M. Adaptations in cortical and trabecular bone in response to mechanical loading with and without weight bearing. Calcif. Tissue Int. 2006, 79, 395–403. [Google Scholar] [CrossRef]
- Liu, S.; Li, Z.; Xu, S.; Xu, L.; Yang, M.; Ni, G. Intensity‑dependent effect of treadmill running on differentiation of rat bone marrow stromal cells. Mol. Med. Rep. 2018, 17, 7746–7756. [Google Scholar] [CrossRef] [PubMed]
- Sampson, H.W. Alcohol and Other Factors Affecting Osteoporosis Risk in Women. Alcohol Res. Health 2002, 26, 292. [Google Scholar] [PubMed]
- Court-Brown, C.M.; Wood, A.M.; Aitken, S. The epidemiology of acute sports-related fractures in adults. Injury 2008, 39, 1365–1372. [Google Scholar] [CrossRef] [PubMed]
- Waterman, B.R.; Gun, B.; Bader, J.O.; Orr, J.D.; Belmont, P.J. Epidemiology of Lower Extremity Stress Fractures in the United States Military. Mil. Med. 2016, 181, 1308–1313. [Google Scholar] [CrossRef] [PubMed]
- Lane, J.M.; Serota, A.C.; Raphael, B. Osteoporosis: Differences and similarities in male and female patients. Orthop. Clin. N. Am. 2006, 37, 601–609. [Google Scholar] [CrossRef]
| SED | EX | p Values | ||
|---|---|---|---|---|
| Weight (g) | T0 | 195 ± 9 | 207 ± 8 | p = 0.001 |
| T8 | 469 ± 41 | 495 ± 43 | p = 0.14 | |
| T12 | 521 ± 50 | 557 ± 29 | p = 0.08 | |
| Length (cm) | T0 | 19.4 ± 0.6 | 20.6 ± 0.7 | p = 0.001 |
| T12 | 27.0 ± 1.0 | 27.5 ± 0.9 | p = 0.35 | |
| BMI (g/cm2) | T0 | 0.52 ± 0.04 | 0.49 ± 0.05 | p = 0.1 |
| T12 | 0.71 ± 0.05 | 0.74 ± 0.04 | p = 0.5 | |
| MAS (m/min) | T1 | 23.4 ± 11.3 | 29.2 ± 7.9 | p = 0.11 |
| Bones | Groups | Time-Points | ||||
|---|---|---|---|---|---|---|
| T9 | T10 | T11 | T12 | |||
| BV/TV (%) | Tibia | SED | 3.4 ± 1.5 | 8.6 ± 1.1 | 12.7 ± 3.3 | 16.3 ± 4.0 ¶ |
| EX | 2.9 ± 1.6 | 13.4 ± 4.1 ¥ | 24.8 ± 6.8 ¥ | 34.2 ± 12.3 ¥ ¶ | ||
| Femur | SED | 4.8 ± 0.9 | 10.4 ± 1.0 | 12.2 ± 1.8 | 15.9 ± 2.9 ¶ | |
| EX | 4.4 ± 0.9 | 11.1 ± 1.8 | 13.0 ± 2.4 | 22.0 ± 5.6 ¶ | ||
| BS/BV (1/mm) | Tibia | SED | 57.6 ± 11.4 | 47.6 ± 9.3 ¥ | 34.5 ± 6.1¥ | 21.7 ± 3.4 ¥ ¶ |
| EX | 70.6 ± 26.6 | 43.6 ± 3.9 ¥ | 31.9 ± 8.7 ¥ | 16.4 ± 5.0 ¥ ¶ | ||
| Femur | SED | 58.9 ± 10.7 | 43.0 ± 8.3 ¥ | 39.1 ± 6.8 | 22.8 ± 3.8 ¥ ¶ | |
| EX | 58.0 ± 7.4 | 44.3 ± 3.5 ¥ | 40.8 ± 5.2 | 20.3 ± 3.6 ¥ ¶ | ||
| SMI | Tibia | SED | 2.78 ± 0.47 | 2.38 ± 0.18 ¥ | 2.28 ± 0.24 | 2.01 ± 0.28 ¥ ¶ |
| EX | 2.83 ± 0.15 | 2.40 ± 0.37 ¥ | 2.35 ± 0.56 | 1.97 ± 0.63 ¥ ¶ | ||
| Femur | SED | 2.42 ± 0.21 | 2.43 ± 0.27 | 2.31 ± 0.19 | 2.06 ± 0.68 ¥ ¶ | |
| EX | 2.54 ± 0.14 | 2.41 ± 0.27 | 2.57 ± 0.17 | 1.76 ± 0.36 ¥ ¶ | ||
| Tb.Pf (1/mm) | Tibia | SED | 23.09 ± 9.02 | 12.75 ± 3.20 ¥ | 9.56 ± 2.30 | 5.66 ± 1.48 ¶ |
| EX | 25.16 ± 6.75 | 12.02 ± 4.89 | 5.33 ± 5.50 | 3.59 ± 2.04 ¶ | ||
| Femur | SED | 17.71 ± 5.55 | 12.46 ± 3.57 ¥ | 10.19 ± 3.37 ¥ | 4.57 ± 1.82 ¶ | |
| EX | 19.03 ± 3.03 | 12.26 ± 3.66 ¥ | 12.44 ± 1.74 ¥ | 2.98 ± 1.89 ¶ | ||
| BMD (mg HA/cm3) | Tibia | SED | 125.4 ± 28. | |||
| EX | 108.1 ± 11.6 | |||||
| Femur | SED | 107.8 ± 5.1 | ||||
| EX | 114.0 ± 9.2 * | |||||
| Parameters | SED | EX |
|---|---|---|
| ALP (IU/L) | 256 ± 113 | 244 ± 31 |
| NTx (ng/mL) | 52 ± 6 | 44 ± 6 * |
| PTH (pg/mL) | 94 ± 86 | 200 ± 158 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bourzac, C.; Bensidhoum, M.; Manassero, M.; Chappard, C.; Michoux, N.; Pallu, S.; Portier, H. Preventive Moderate Continuous Running-Exercise Conditioning Improves the Healing of Non-Critical Size Bone Defects in Male Wistar Rats: A Pilot Study Using µCT. Life 2020, 10, 308. https://doi.org/10.3390/life10120308
Bourzac C, Bensidhoum M, Manassero M, Chappard C, Michoux N, Pallu S, Portier H. Preventive Moderate Continuous Running-Exercise Conditioning Improves the Healing of Non-Critical Size Bone Defects in Male Wistar Rats: A Pilot Study Using µCT. Life. 2020; 10(12):308. https://doi.org/10.3390/life10120308
Chicago/Turabian StyleBourzac, Céline, Morad Bensidhoum, Mathieu Manassero, Christine Chappard, Nicolas Michoux, Stéphane Pallu, and Hugues Portier. 2020. "Preventive Moderate Continuous Running-Exercise Conditioning Improves the Healing of Non-Critical Size Bone Defects in Male Wistar Rats: A Pilot Study Using µCT" Life 10, no. 12: 308. https://doi.org/10.3390/life10120308
APA StyleBourzac, C., Bensidhoum, M., Manassero, M., Chappard, C., Michoux, N., Pallu, S., & Portier, H. (2020). Preventive Moderate Continuous Running-Exercise Conditioning Improves the Healing of Non-Critical Size Bone Defects in Male Wistar Rats: A Pilot Study Using µCT. Life, 10(12), 308. https://doi.org/10.3390/life10120308






