Age and Origin of Monazite Symplectite in an Iron Oxide-Apatite Deposit in the Adirondack Mountains, New York, USA: Implications for Tracking Fluid Conditions
Abstract
1. Introduction
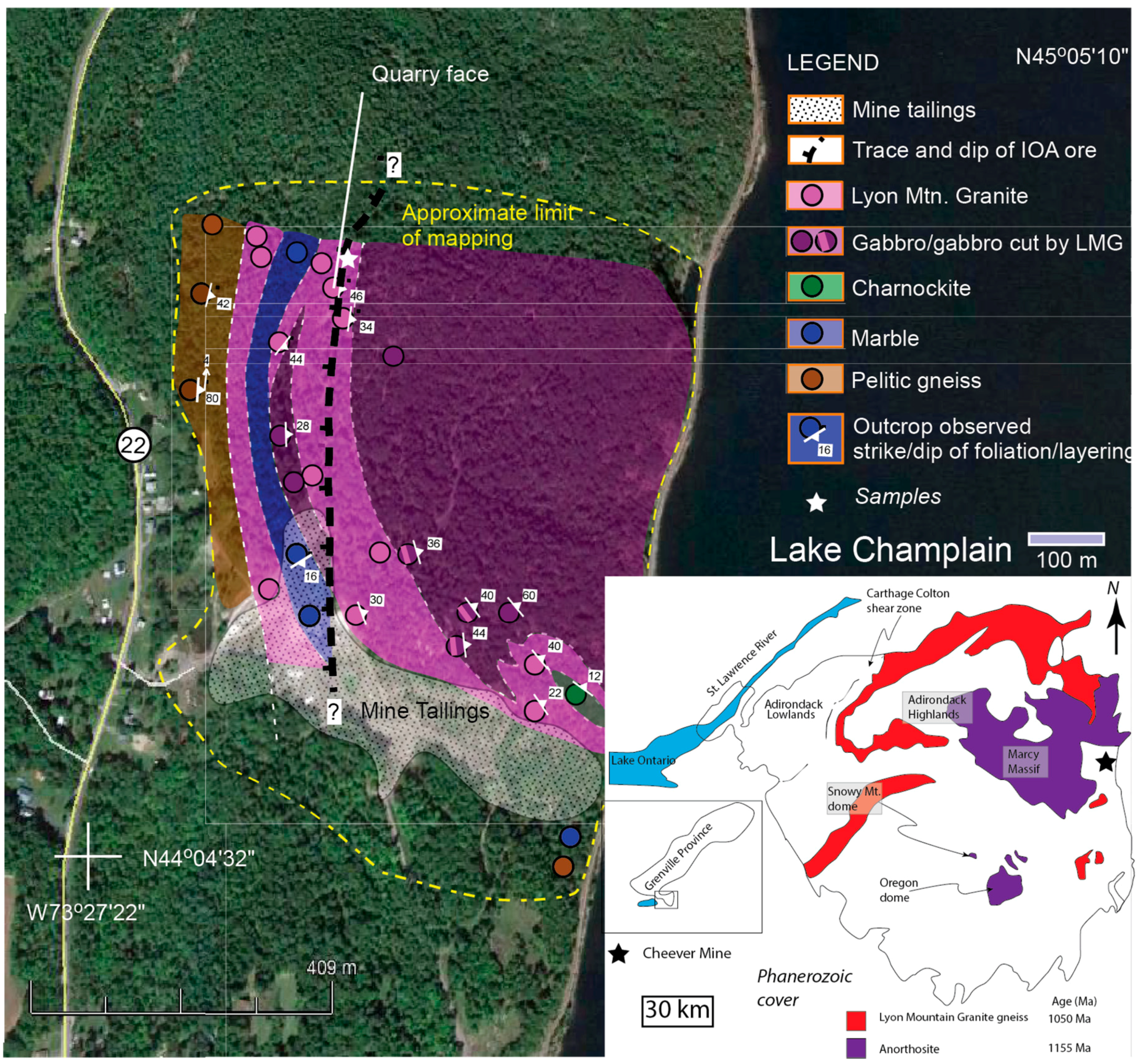
2. Geologic Setting
3. Sample Description
4. Analytical Methods
5. Results
5.1. Type-I Monazite (Sample VGA-14)
5.2. Type-II Monazite (Sample 4UR-015)
6. Discussion
6.1. Compositional Variability and Timing
6.2. Reaction Constraints
6.3. A Record of Evolving Fluid Conditions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Spear, F.S.; Pyle, J.M. Apatite, Monazite, and Xenotime in Metamorphic Rocks. Rev. Mineral. Geochem. 2002, 48, 293–335. [Google Scholar] [CrossRef]
- Williams, M.L.; Jercinovic, M.J.; Hetherington, C.J. Microprobe Monazite Geochronology: Understanding Geologic Processes by Integrating Composition and Chronology. Annu. Rev. Earth Planet. Sci. 2007, 35, 137–175. [Google Scholar] [CrossRef]
- Williams, M.; Jercinovic, M.; Harlov, D.; Budzyń, B.; Hetherington, C. Resetting monazite ages during fluid-related alteration. Chem. Geol. 2011, 283, 218–225. [Google Scholar] [CrossRef]
- Grand’Homme, A.; Janots, E.; Seydoux-Guillaume, A.-M.; Guillaume, D.; Bosse, V.; Magnin, V. Partial resetting of the U-Th-Pb systems in experimentally altered monazite: Nanoscale evidence of incomplete replacement. Geology 2016, 44, 431–434. [Google Scholar] [CrossRef]
- Zi, J.-W.; Rasmussen, B.; Muhling, J.R.; Fletcher, I.R.; Thorne, A.M.; Johnson, S.P.; Cutten, H.N.; Dunkley, D.J.; Korhonen, F.J. In situ U–Pb geochronology of xenotime and monazite from the Abra polymetallic deposit in the Capricorn Orogen, Australia: Dating hydrothermal mineralization and fluid flow in a long-lived crustal structure. Precambrian Res. 2015, 260, 91–112. [Google Scholar] [CrossRef]
- Harlov, D.E. Apatite: A fingerprint of metasomatic processes. Elements 2015, 11, 171–176. [Google Scholar] [CrossRef]
- Harlov, D.E.; Persson-Nilsson, K.; Jonsson, E.; Majka, J.; Högdahl, K. Fluorapatite-monazite-allanite relations in the Grängesberg apatite-iron oxide ore district, Bergslagen, Sweden. Am. Mineral. 2016, 101, 1769–1782. [Google Scholar]
- Bilenker, L.D.; Simon, A.C.; Reich, M.; Lundstrom, C.C.; Gajos, N.; Bindeman, I.; Barra, F.; Munizaga, R. Fe–O stable isotope pairs elucidate a high-temperature origin of Chilean iron oxide-apatite deposits. Geochim. Cosmochim. Acta 2016, 177, 94–104. [Google Scholar] [CrossRef]
- Valley, P.M.; Hanchar, J.M.; Whitehouse, M. New insights on the evolution of the Lyon Mountain Granite and associated Kiruna-type magnetite-apatite deposits, Adirondack Mountains, New York State. Geosphere 2011, 7, 357–389. [Google Scholar] [CrossRef]
- Chiarenzelli, J.; Selleck, B.; Lupulescu, M.; Regan, S.; Bickford, M.E.; Valley, P.; McLelland, J. Lyon Mountain Ferroan Leucogranite Suite: Response to Collision, Thickened Crust, and Extension in the Core of the Grenville Orogen. GSA Bull. 2017, 129, 1472–1488. [Google Scholar]
- Chiarenzelli, J.R.; Lupulescu, M.V.; Regan, S.P.; Singer, J.W. Age and Origin of the Mesoproterozoic Iron Oxide-Apatite Mineralization, Cheever Mine, Eastern Adirondacks, NY. Geosciences 2018, 8, 345. [Google Scholar] [CrossRef]
- Lupulescu, M.V.; Hughes, J.M.; Chiarenzelli, J.R.; Bailey, D.G. Texture, Crystal Structure, and Composition of Fluorapatites From Iron Oxide-Apatite (Ioa) Deposits, Eastern Adirondack Mountains, New York. Can. Mineral. 2017, 55, 399–417. [Google Scholar] [CrossRef]
- Chew, D.M.; Sylvester, P.J.; Tubrett, M.N. U–Pb and Th–Pb dating of apatite by LA-ICPMS. Chem. Geol. 2011, 280, 200–216. [Google Scholar] [CrossRef]
- Long, K.R.; Van Gosen, B.S.; Foley, N.K.; Cordier, D. The Principal Rare Earth Element Deposits of The United States—A Summary of Domestic Deposits and a Global Perspective; U.S. Geological Survey Scientific Investigations Report; USGS Western Mineral and Environmental Resources Science Center: North Gemini Drive Flagstaff, AZ, USA, 2010; 96p.
- McLelland, J.M.; Selleck, B.W.; Bickford, M.E. Review of the Proterozoic evolution of the Grenville Province, its Adirondack outlier, and the Mesoproterozoic inliers of the Appalachians. In From Rodinia to Pangea: The Lithotectonic Record of the Appalachian Region: Geological Society of America Memoir; Tollo, R.P., Bartholomew, M.J., Hibbard, J.P., Karabinos, P.M., Eds.; Geological Society of America: Boulder, CO, USA, 2010; Volume 206, pp. 1–29. [Google Scholar]
- Rivers, T. Assembly and preservation of lower, mid, and upper orogenic crust in the Grenville Province—Implications for the evolution of large hot long-duration orogens. Precambrian Res. 2008, 167, 237–259. [Google Scholar] [CrossRef]
- Selleck, B.W.; McLelland, J.M.; Bickford, M.E. Granite emplacement during tectonic exhumation: The Adirondack example. Geology 2005, 33, 781. [Google Scholar] [CrossRef]
- Wong, M.S.; Williams, M.L.; McLelland, J.M.; Jercinovik, M.J.; Kowalkoski, J. Late Ottawan extension in the eastern Adirondack Highlands: Evidence from structural studies and zircon and monazite geochronology. Geol. Soc. Am. Bull. 2011, 124, 857–869. [Google Scholar] [CrossRef]
- McLelland, J.; Hamilton, M.; Selleck, B.; McLelland, J.M.; Walker, D.; Orrell, S. Zircon U- Pb geochronology of the Ottawan orogeny, Adirondack Highlands, New York. Regional and tectonic implications. Precambrian Res. 2001, 109, 39–72. [Google Scholar] [CrossRef]
- Valley, P.M.; Hanchar, J.M.; Whitehouse, M.J. Direct dating of Fe oxide-(Cu-Au) mineralization by U/Pb zircon geochronology. Geology 2009, 37, 223–226. [Google Scholar] [CrossRef]
- Postel, A.W. Geology of the Clinton County Magnetite District, New York; US Geological Survey: Reston, VA, USA, 1952; Volume 237, p. 95.
- Geer, P.S.; Regan, S.P.; Walsh, G.J.; Williams, M.L.; Lupulescu, M.V.; Valley, P.M. Multiple stages of mineralization of the hammondville magnetite-apatite deposits. In Proceedings of the Northeast Geological Society of America Annual Meeting, Mount Washington, NH, USA, 21–23 March 2016. [Google Scholar]
- Knipping, J.L.; Bilenker, L.D.; Simon, A.C.; Reich, M.; Barra, F.; Deditius, A.P.; Lundstrom, C.; Bindeman, I.; Munizaga, R. Giant Kiruna-type deposits form by efficient flotation of magmatic magnetite suspensions. Geology 2015, 43, 591–594. [Google Scholar] [CrossRef]
- Lupulescu, M. Minerals from the Iron Deposits of New York State. Rocks Mineral. 2008, 83, 248–266. [Google Scholar] [CrossRef]
- Hill, B.M.; Clechenko, C.C.; Valley, J.W.; Hamilton, M.A.; McLelland, J.M.; Bickford, M.E. Direct dating of Adirondack massif anorthosite by U-Pb SHRIMP analysis of igneous zircon: Implications for AMCG complexes. Geol. Soc. Am. Bull. 2004, 116, 1299–1317. [Google Scholar]
- Regan, S.P.; Chiarenzelli, J.R.; McLelland, J.M.; Cousens, B.L. Evidence for an enriched asthenospheric source for coronitic metagabbros in the Adirondack Highlands. Geosphere 2011, 7, 694–709. [Google Scholar] [CrossRef]
- Lupulescu, M.V.; Chiarenzelli, J.R.; Bailey, D.G.; Regan, S.P. The magnetite-fluorapatite ores from the eastern Adirondacks, New York: Cheever Mine. In Proceedings of the New York State Geological Association Annual Meeting, Plattsburgh, NY, USA, 12–13 September 2015. [Google Scholar]
- Regan, S.P.; Walsh, G.J.; Williams, M.L.; Chiarenzelli, J.R.; Toft, M.; McAleer, R. Syn-collisional exhumation of hot middle crust in the Adirondack Mountains: Implications for extensional orogenesis in the southern Grenville Province. Geosphere 2019. under review. [Google Scholar]
- Budzyń, B.; Harlov, D.E.; Williams, M.L.; Jercinovic, M.J. Experimental determination of stability relations between monazite, fluorapatite, allanite, and REE-epidote as a function of pressure, temperature, and fluid composition. Am. Mineral. 2011, 96, 1547–1567. [Google Scholar]
- Budzyń, B.; Harlov, D.E.; Kozub-Budzyń, G.A.; Majka, J. Experimental constraints on the relative stabilities of the two systems monazite-(Ce)–allanite-(Ce)–Fluorapatite and xnotime-(Y)–(Y, HREE)-rich epidote–(Y,HREE)-rich fluorapatite, in high Ca and Na-Ca environments uner P-T conditions of 200–1000 MPa and 450–750 °C. Contrib. Mineral. Petrol. 2017, 111, 183–217. [Google Scholar]
- Jercinovic, M.J.; Williams, M.L.; Lane, E.D. In-situ trace element analysis of monazite and other fine-grained accessory minerals by EPMA. Chem. Geol. 2008, 254, 197–215. [Google Scholar] [CrossRef]
- Pouchou, J.L.; Pichoir, F. “PAP” phi-rho-Z procedure for improved quantitative microanalysis. In Microbeam Analysis; Armstrong, J.L., Ed.; San Francisco Press Inc.: San Francisco, CA, USA, 1985; pp. 104–106. [Google Scholar]
- Allaz, J.; Williams, M.L.; Jercinovic, M.J.; Donovan, J. A new technique for electron microprobe trace element analysis: The multipoint background method. In Proceedings of the EMAS 2011, Modern Developments and Applications in Microbeam Analysis, Angers, France, 15–19 May 2011; pp. 319–320. [Google Scholar]
- Dumond, F.; McLean, N.; Williams, M.L.; Jercinovic, M.J.; Bowring, S.A. High resolution dating of ranite petrogenesis and deformation in a lower crustal shear zone: Athabasca granulite terrane, western Canadian Shield. Chem. Geol. 2008, 16, 175–196. [Google Scholar] [CrossRef]
- Vernon, R.H. A Practical Guide to Rock Microstructure; Cambridge University Press (CUP): Cambridge, MA, USA, 2004. [Google Scholar]
- Harlov, D.E.; Forster, H.-J. Fluid-induced nucleation of (Y + REE) phosphate minerals within apatite: Nature and experiment. Part II. Fluorapatite. Am. Mineral. 2003, 88, 1209–1229. [Google Scholar] [CrossRef]
- Betkowski, W.B.; Harlov, D.E.; Rakovan, J.F. Hydrothermal mineral replacement reactions for an apatite-monazite assemblage in alkali-rich fluids at 300–600 °C and 100 MPa. Am. Mineral. 2016, 101, 2620–2637. [Google Scholar] [CrossRef]
- Harlov, D.E.; Wirth, R.; Förster, H.-J. An experimental study of dissolution–reprecipitation in fluorapatite: Fluid infiltration and the formation of monazite. Contrib. Mineral. Petrol. 2005, 150, 268–286. [Google Scholar] [CrossRef]
- Harlov, D.E.; Wirth, R.; Hetherington, C.J. Fluid-mediated partial alteration in monazite: The role of coupled dissolution–reprecipitation in element redistribution and mass transfer. Contrib. Mineral. Petrol. 2010, 162, 329–348. [Google Scholar] [CrossRef]
- Tropper, P.; Manning, C.E.; Harlov, D.E. Solubility of CePO4 monazite and YPO4 xenotime in H2O and H2O–NaCl at 800 °C and 1 GPa: Implications for REE and Y transport during high-grade metamorphism. Chem. Geol. 2011, 282, 58–66. [Google Scholar] [CrossRef]
- Tropper, P.; Manning, C.E.; Harlov, D.E. Experimental determination of CePO4 and YPO4 solubilities in H2O-NaF at 800 °C and 1 GPa: Implications for rare earth element transport in high-grade metamorphic fluids. Geofluids 2013, 13, 372–380. [Google Scholar] [CrossRef]
- Harlov, D.E.; Marschall, H.R.; Hanel, M. Fluorapatite-monazite relationships in granulite-facies metapelites, Schwarzwald, southwest Germany. Mineral. Mag. 2007, 71, 223–234. [Google Scholar] [CrossRef]
- Schermaier, A.; Finger, F.; Broska, I.; Roberts, M.P. Replacement of primary monazite by apatite-allanite-epidote coronas in an amphibolite facies granite gneiss from the Eastern Alps. Am. Mineral. 1998, 83, 248–258. [Google Scholar]
- Cherniak, D.; Watson, E.; Grove, M.; Harrison, T. Pb diffusion in monazite: A combined RBS/SIMS study. Geochim. Cosmochim. Acta 2004, 68, 829–840. [Google Scholar] [CrossRef]
- Cherniak, D.; Pyle, J. Th diffusion in monazite. Chem. Geol. 2008, 256, 52–61. [Google Scholar] [CrossRef]
- Seydoux-Guillaume, A.-M.; Paquette, J.-L.; Wiedenbeck, M.; Montel, J.-M.; Heinrich, W. Experimental resetting of the U–Th–Pb systems in monazite. Chem. Geol. 2002, 191, 165–181. [Google Scholar] [CrossRef]
- Seydoux-Guillaume, A.-M.; Montel, J.-M.; Bingen, B.; Bosse, V.; De Parseval, P.; Paquette, J.-L.; Janots, E.; Wirth, R. Low-temperature alteration of monazite: Fluid mediated coupled dissolution–precipitation, irradiation damage, and disturbance of the U–Pb and Th–Pb chronometers. Chem. Geol. 2012, 330, 140–158. [Google Scholar] [CrossRef]
- Allaz, J.; Selleck, B.; Williams, M.L.; Jercinovic, M.J. Microprobe analysis and dating of monazite from the Potsdam Formation, New York: A progressive record of chemical reaction and fluid interaction. Am. Mineral. 2013, 98, 1106–1119. [Google Scholar] [CrossRef]
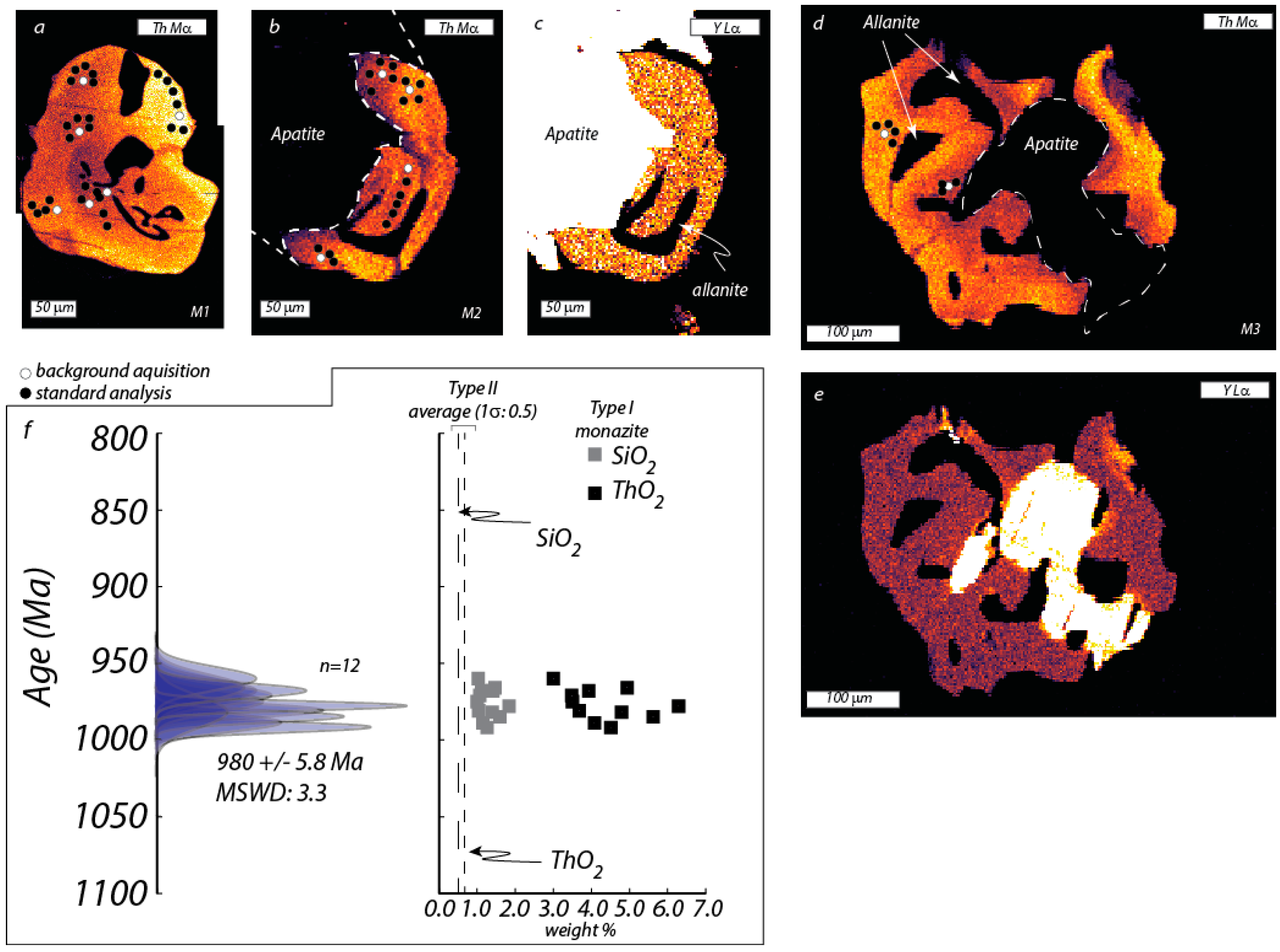
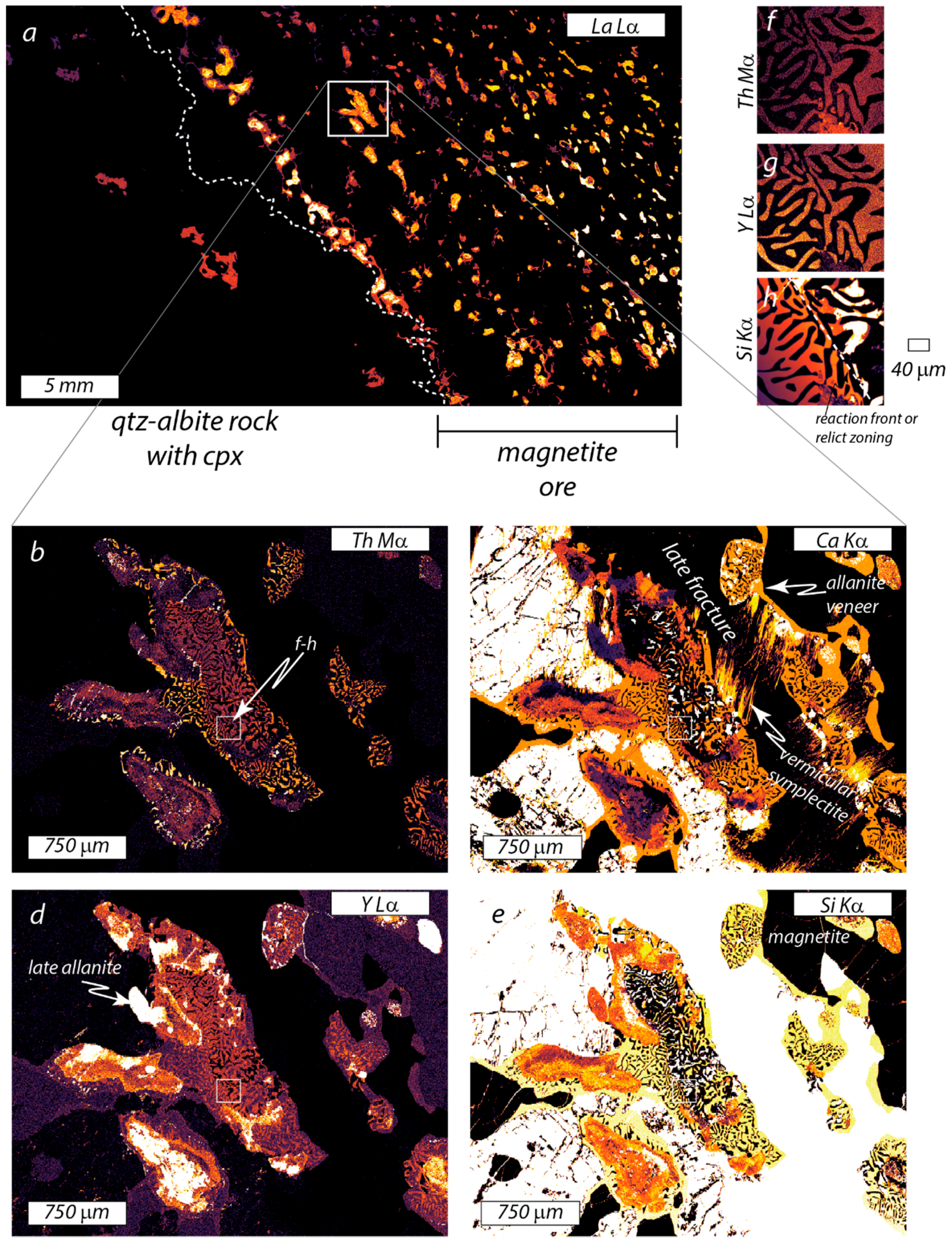
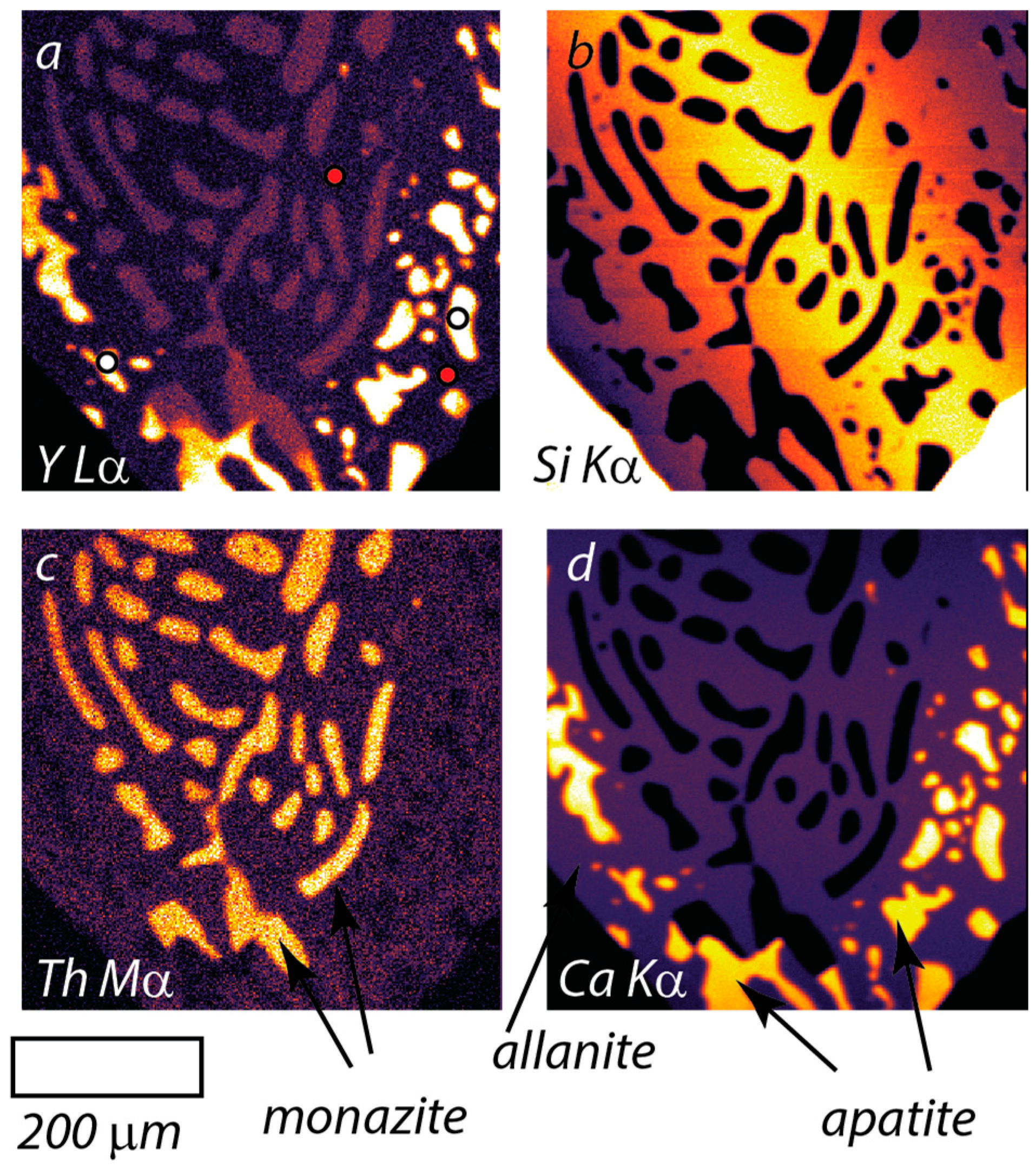
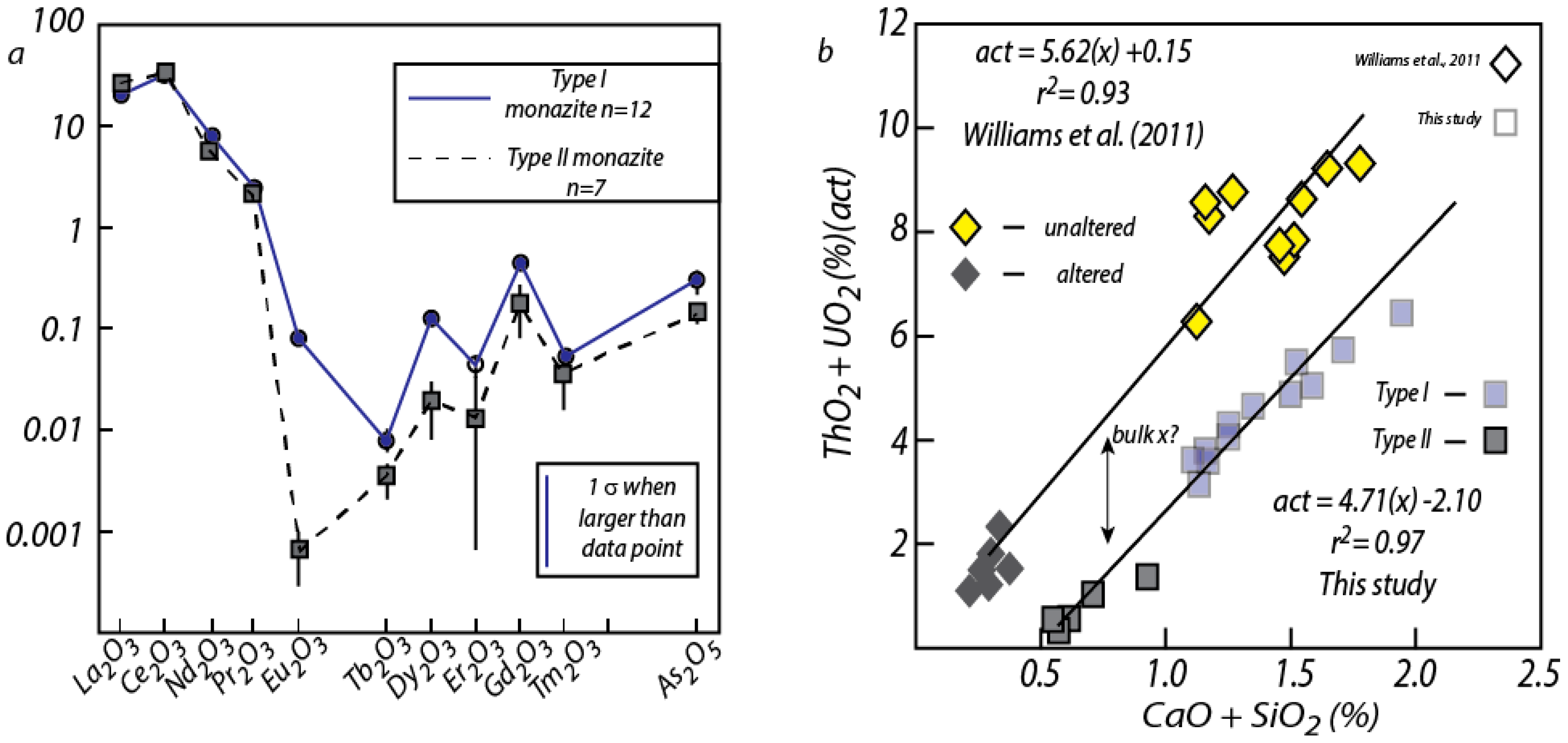
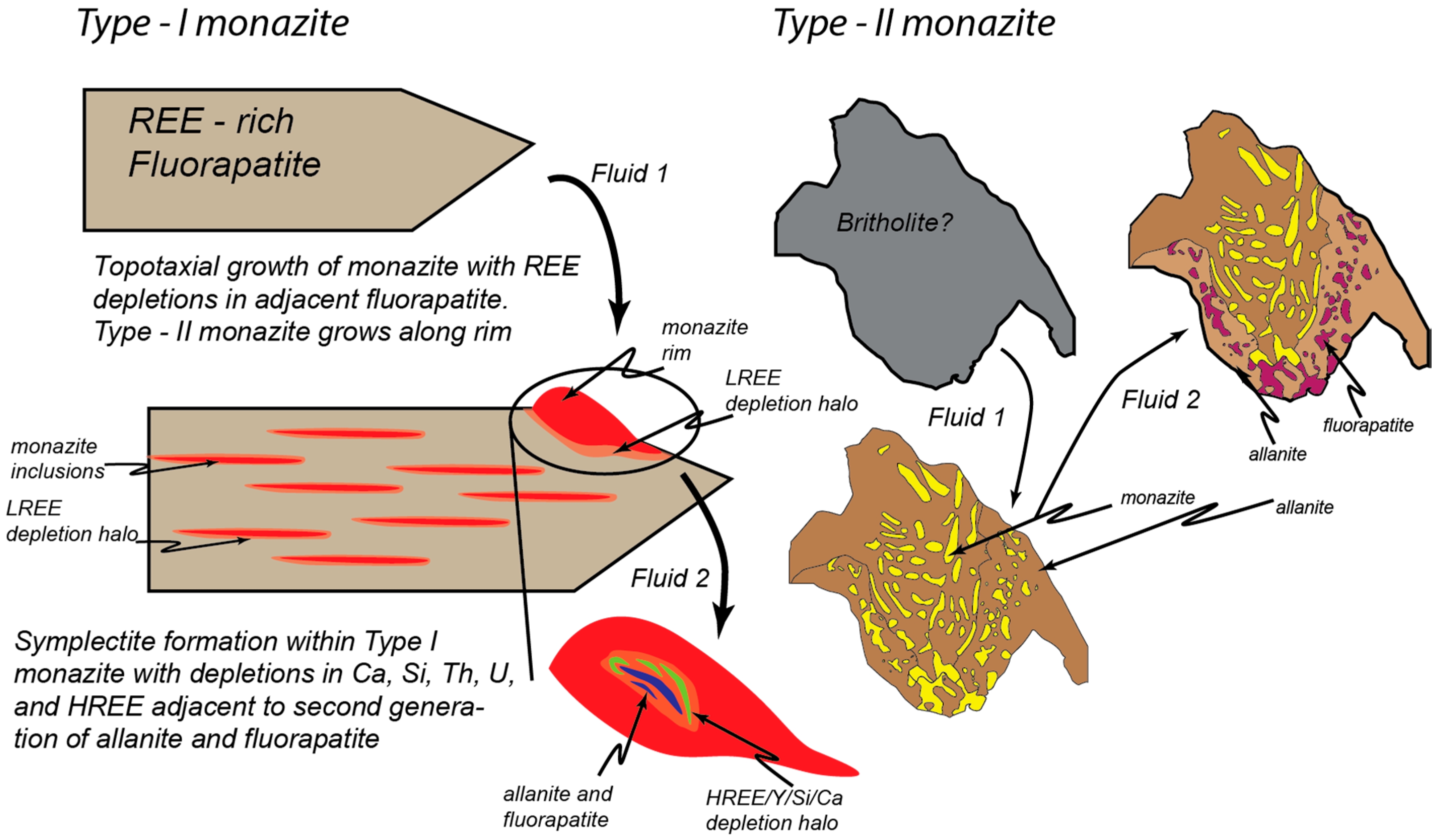
| VGA-14: Type I Monazite | 4UR-015: Type II Monazite | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sample | M1-1 | M2-1 | M2-2 | M2-3 | M2-4 | M1-3 | M1-4 | M1-5 | M1-2 | M1-6 | M3-1 | M3-2 | Core1 | Core2 | Core3 | Rim1 | Rim2 | Core4 | Core5 |
| P2O5 | 28.49 | 28.00 | 28.92 | 28.38 | 28.68 | 28.93 | 28.34 | 28.05 | 28.49 | 29.22 | 27.38 | 29.05 | 28.90 | 28.77 | 28.80 | 28.82 | 28.87 | 28.94 | 29.10 |
| SiO2 | 1.26 | 1.41 | 1.04 | 1.15 | 1.00 | 1.07 | 1.47 | 1.60 | 1.40 | 1.03 | 1.84 | 1.12 | 0.54 | 0.51 | 0.53 | 0.65 | 0.85 | 0.51 | 0.51 |
| SO3 | 0.03 | 0.03 | 0.01 | 0.01 | 0.02 | 0.02 | 0.03 | 0.02 | 0.01 | 0.00 | 0.02 | 0.01 | 0.09 | 0.05 | 0.06 | 0.05 | 0.09 | 0.06 | 0.06 |
| ThO2 | 4.51 | 5.33 | 3.68 | 4.08 | 3.50 | 3.48 | 4.94 | 5.62 | 4.79 | 3.00 | 6.29 | 3.93 | 0.41 | 0.51 | 0.46 | 1.03 | 1.47 | 0.46 | 0.45 |
| UO2 | 0.12 | 0.14 | 0.14 | 0.15 | 0.14 | 0.12 | 0.13 | 0.14 | 0.13 | 0.12 | 0.16 | 0.13 | 0.02 | 0.04 | 0.05 | 0.04 | 0.02 | 0.06 | 0.05 |
| Y2O3 | 0.66 | 0.61 | 0.67 | 0.62 | 0.67 | 0.70 | 0.67 | 0.65 | 0.65 | 0.74 | 0.65 | 0.76 | 0.11 | 0.17 | 0.17 | 0.14 | 0.08 | 0.16 | 0.15 |
| As2O3 | 0.16 | 0.09 | 0.18 | 0.14 | 0.13 | 0.14 | 0.12 | 0.11 | 0.18 | 0.13 | 0.13 | 0.13 | 0.26 | 0.21 | 0.36 | 0.23 | 0.26 | 0.42 | 0.30 |
| La2O3 | 20.90 | 20.90 | 20.52 | 21.52 | 20.61 | 20.87 | 21.22 | 21.22 | 21.54 | 20.69 | 21.04 | 20.11 | 28.59 | 23.82 | 25.28 | 25.32 | 30.58 | 25.78 | 26.32 |
| Ce2O3 | 31.54 | 31.52 | 32.81 | 32.27 | 32.79 | 32.79 | 31.68 | 31.38 | 31.85 | 33.23 | 31.04 | 32.55 | 33.21 | 34.87 | 34.78 | 34.33 | 31.31 | 34.81 | 34.56 |
| Pr2O3 | 2.51 | 2.46 | 2.66 | 2.53 | 2.61 | 2.65 | 2.50 | 2.48 | 2.47 | 2.74 | 2.46 | 2.69 | 1.94 | 2.45 | 2.36 | 2.28 | 1.71 | 2.35 | 2.25 |
| Nd2O3 | 8.34 | 8.17 | 8.84 | 8.37 | 8.82 | 8.96 | 8.40 | 8.20 | 8.28 | 9.21 | 8.10 | 9.26 | 5.11 | 6.91 | 6.52 | 6.34 | 4.27 | 6.38 | 6.06 |
| Sm2O3 | 0.17 | 0.18 | 0.21 | 0.18 | 0.21 | 0.25 | 0.27 | 0.20 | 0.20 | 0.27 | 0.21 | 0.30 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| Eu2O3 | 0.07 | 0.06 | 0.05 | 0.06 | 0.06 | 0.10 | 0.11 | 0.09 | 0.09 | 0.08 | 0.08 | 0.12 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| Gd2O3 | 0.45 | 0.46 | 0.50 | 0.45 | 0.49 | 0.51 | 0.50 | 0.41 | 0.34 | 0.52 | 0.42 | 0.53 | 0.10 | 0.27 | 0.28 | 0.24 | 0.04 | 0.28 | 0.11 |
| Tb2O3 | 0.01 | 0.01 | 0.00 | 0.01 | 0.01 | 0.00 | 0.02 | 0.00 | 0.01 | 0.00 | 0.01 | 0.00 | 0.00 | 0.00 | 0.02 | 0.00 | 0.00 | 0.00 | 0.00 |
| Dy2O3 | 0.14 | 0.12 | 0.15 | 0.14 | 0.13 | 0.14 | 0.14 | 0.14 | 0.12 | 0.13 | 0.13 | 0.17 | 0.01 | 0.03 | 0.02 | 0.03 | 0.00 | 0.03 | 0.02 |
| Er2O3 | 0.04 | 0.04 | 0.05 | 0.05 | 0.03 | 0.06 | 0.05 | 0.03 | 0.03 | 0.04 | 0.03 | 0.06 | 0.00 | 0.02 | 0.03 | 0.01 | 0.00 | 0.03 | 0.00 |
| Tm2O3 | 0.07 | 0.04 | 0.06 | 0.06 | 0.06 | 0.07 | 0.04 | 0.04 | 0.03 | 0.05 | 0.04 | 0.08 | 0.03 | 0.04 | 0.05 | 0.02 | 0.00 | 0.06 | 0.04 |
| Yb2O3 | 0.01 | 0.00 | 0.00 | 0.00 | 0.02 | 0.00 | 0.00 | 0.00 | 0.01 | 0.02 | 0.00 | 0.00 | 0.00 | 0.01 | 0.03 | 0.01 | 0.00 | 0.04 | 0.01 |
| CaO | 0.10 | 0.12 | 0.11 | 0.10 | 0.12 | 0.09 | 0.12 | 0.10 | 0.10 | 0.10 | 0.10 | 0.12 | 0.04 | 0.04 | 0.07 | 0.07 | 0.07 | 0.06 | 0.04 |
| PbO | 0.21 | 0.24 | 0.18 | 0.20 | 0.17 | 0.16 | 0.22 | 0.26 | 0.22 | 0.14 | 0.29 | 0.18 | 0.02 | 0.03 | 0.03 | 0.05 | 0.07 | 0.03 | 0.02 |
| Total | 99.85 | 99.99 | 100.76 | 100.41 | 100.26 | 101.09 | 100.97 | 100.72 | 100.93 | 101.43 | 100.37 | 101.33 | 99.41 | 98.75 | 99.97 | 99.69 | 99.72 | 100.53 | 100.07 |
| Age (Ma) | 992 | 968 | 981 | 989 | 975 | 971 | 966 | 985 | 982 | 960 | 978 | 968 | 947 | 994 | |||||
| 2σ | 7 | 15 | 9 | 12 | 14 | 13 | 17 | 8 | 21 | 15 | 7 | 10 | 18 | 18 | |||||
| Sample | FAp-1 | FAp-2 | FAp-3 | Aln-1 | Aln-2 | Aln-3 | Aln-4 |
|---|---|---|---|---|---|---|---|
| P | 16.4 | 13.81 | 13.39 | na | na | na | na |
| Si | 1.21 | 2.31 | 2.42 | 13.07 | 13.57 | 13.32 | 13.17 |
| Th | na | na | na | 0.06 | 0.01 | 0.04 | bd |
| Y | 1.49 | 2.74 | 0.89 | 0.13 | 0.11 | 0.10 | 0.09 |
| La | 0.29 | 0.58 | 4.04 | 9.07 | 8.00 | 10.25 | 9.18 |
| Ce | 1.23 | 2.55 | 6.65 | 11.14 | 11.80 | 10.83 | 11.40 |
| Pr | 0.27 | 0.61 | 1.03 | 1.81 | 1.89 | 2.01 | 1.91 |
| Nd | 1.42 | 2.68 | 2.27 | 1.79 | 2.38 | 1.50 | 1.76 |
| Sm | 0.19 | 0.36 | 0.16 | 0.03 | 0.02 | Bd | bd |
| Gd | 0.37 | 0.64 | 0.14 | bd | bd | bd | bd |
| Dy | 0.24 | 0.45 | 0.09 | bd | bd | bd | bd |
| Fe | 0.20 | 0.36 | 0.33 | 16.41 | 16.46 | 17.89 | 18.07 |
| Al | na | na | na | 4.70 | 4.79 | 3.81 | 3.77 |
| Na | 0.02 | 0.04 | 0.16 | na | na | na | na |
| Mg | na | na | na | 0.35 | 0.34 | 0.31 | 0.31 |
| Ca | 36.05 | 32.77 | 29.33 | 6.58 | 6.47 | 6.34 | 6.36 |
| Mn | bd | bd | 0.02 | 0.05 | 0.04 | 0.05 | 0.04 |
| Cl | 0.29 | 0.37 | 0.12 | na | na | na | na |
| F | 2.53 | 2.34 | 3.44 | na | na | na | na |
| Total | 100.30 | 98.36 | 100.76 | 99.29 | 99.26 | 98.93 | 98.54 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Regan, S.; Lupulescu, M.; Jercinovic, M.; Chiarenzelli, J.; Williams, M.; Singer, J.; Bailey, D. Age and Origin of Monazite Symplectite in an Iron Oxide-Apatite Deposit in the Adirondack Mountains, New York, USA: Implications for Tracking Fluid Conditions. Minerals 2019, 9, 65. https://doi.org/10.3390/min9010065
Regan S, Lupulescu M, Jercinovic M, Chiarenzelli J, Williams M, Singer J, Bailey D. Age and Origin of Monazite Symplectite in an Iron Oxide-Apatite Deposit in the Adirondack Mountains, New York, USA: Implications for Tracking Fluid Conditions. Minerals. 2019; 9(1):65. https://doi.org/10.3390/min9010065
Chicago/Turabian StyleRegan, Sean, Marian Lupulescu, Michael Jercinovic, Jeffrey Chiarenzelli, Michael Williams, Jared Singer, and David Bailey. 2019. "Age and Origin of Monazite Symplectite in an Iron Oxide-Apatite Deposit in the Adirondack Mountains, New York, USA: Implications for Tracking Fluid Conditions" Minerals 9, no. 1: 65. https://doi.org/10.3390/min9010065
APA StyleRegan, S., Lupulescu, M., Jercinovic, M., Chiarenzelli, J., Williams, M., Singer, J., & Bailey, D. (2019). Age and Origin of Monazite Symplectite in an Iron Oxide-Apatite Deposit in the Adirondack Mountains, New York, USA: Implications for Tracking Fluid Conditions. Minerals, 9(1), 65. https://doi.org/10.3390/min9010065





