Insights into Adsorption of Chlorobenzene in High Silica MFI and FAU Zeolites Gained from Chromatographic and Diffractometric Techniques
Abstract
:1. Introduction
2. Experimental Methods
2.1. Materials and Methods
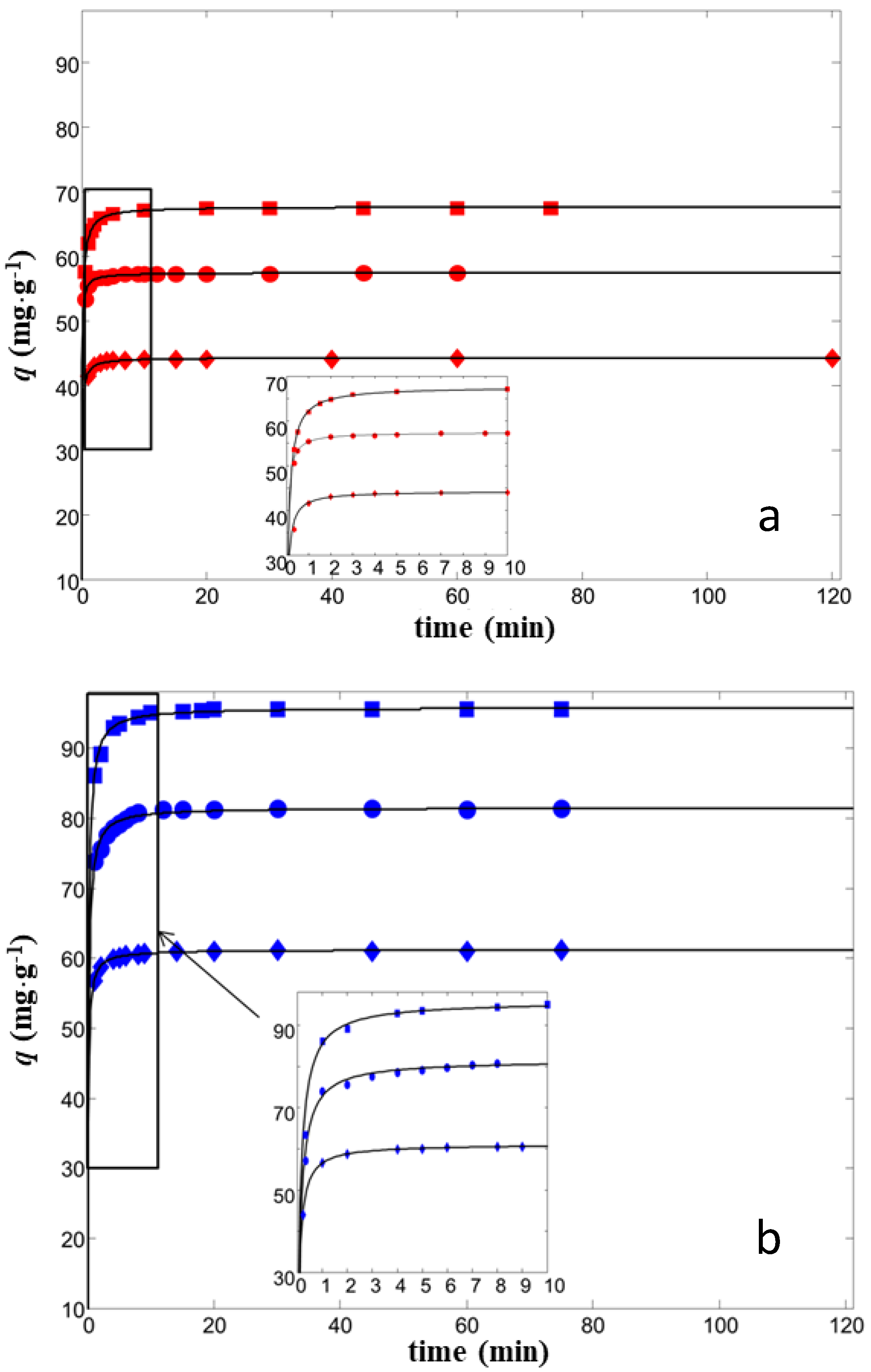
2.2. Batch Adsorption
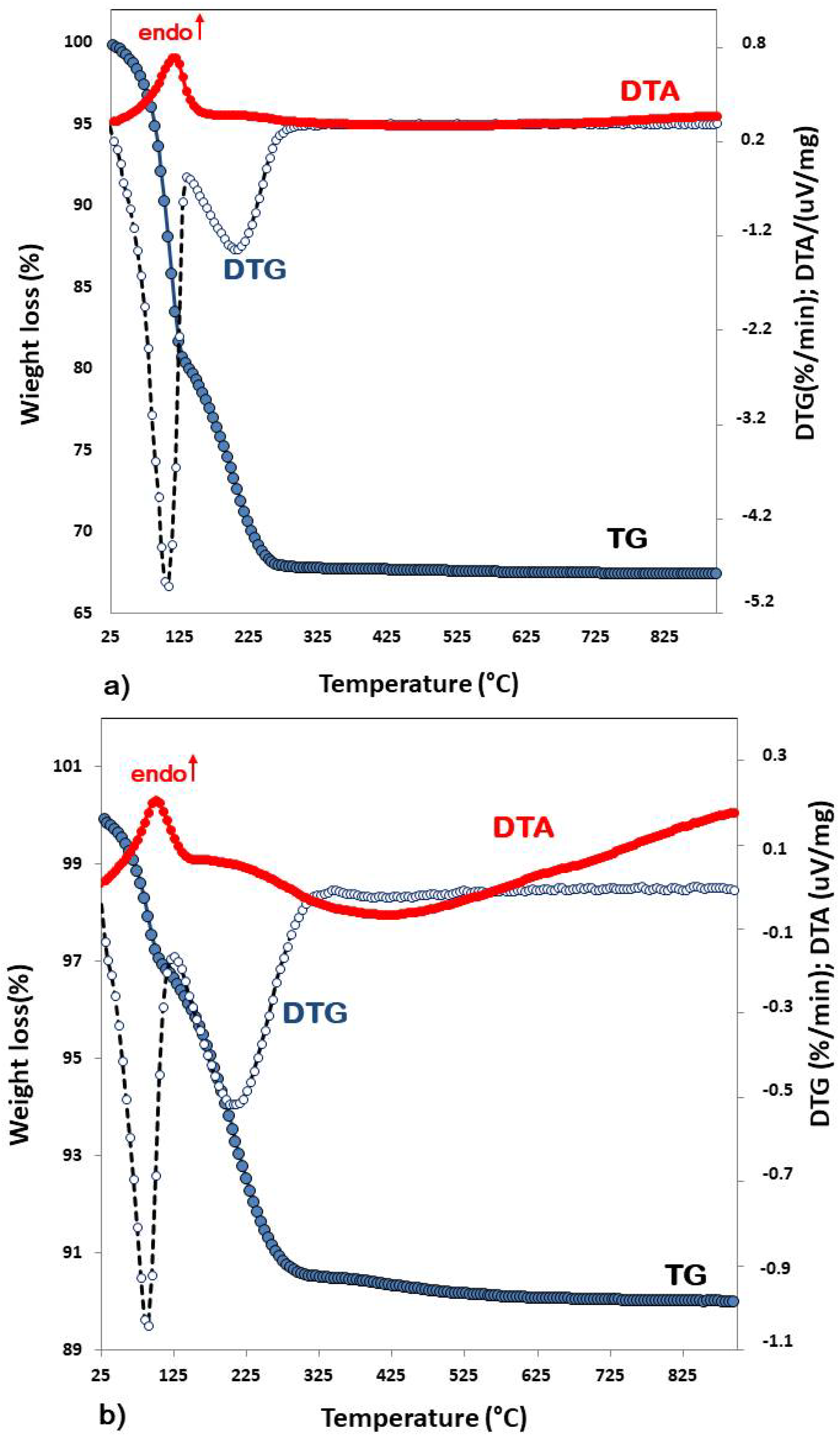
2.3. Thermal Analyses
2.4. X-ray Powder Diffraction Data Collection and Refinement Strategy
3. Results and Discussion
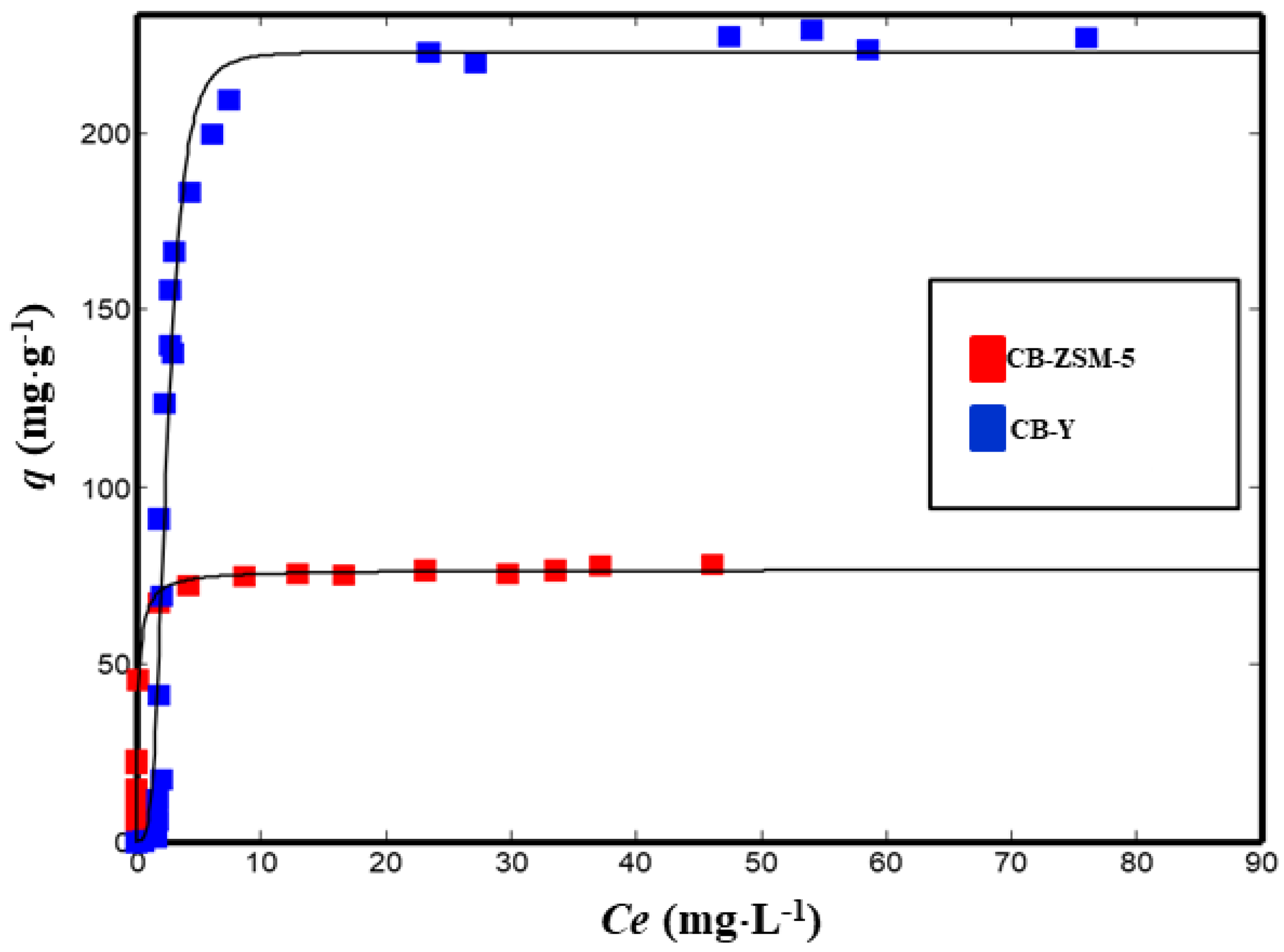
3.1. Adsorption Isotherms from Aqueous Solutions
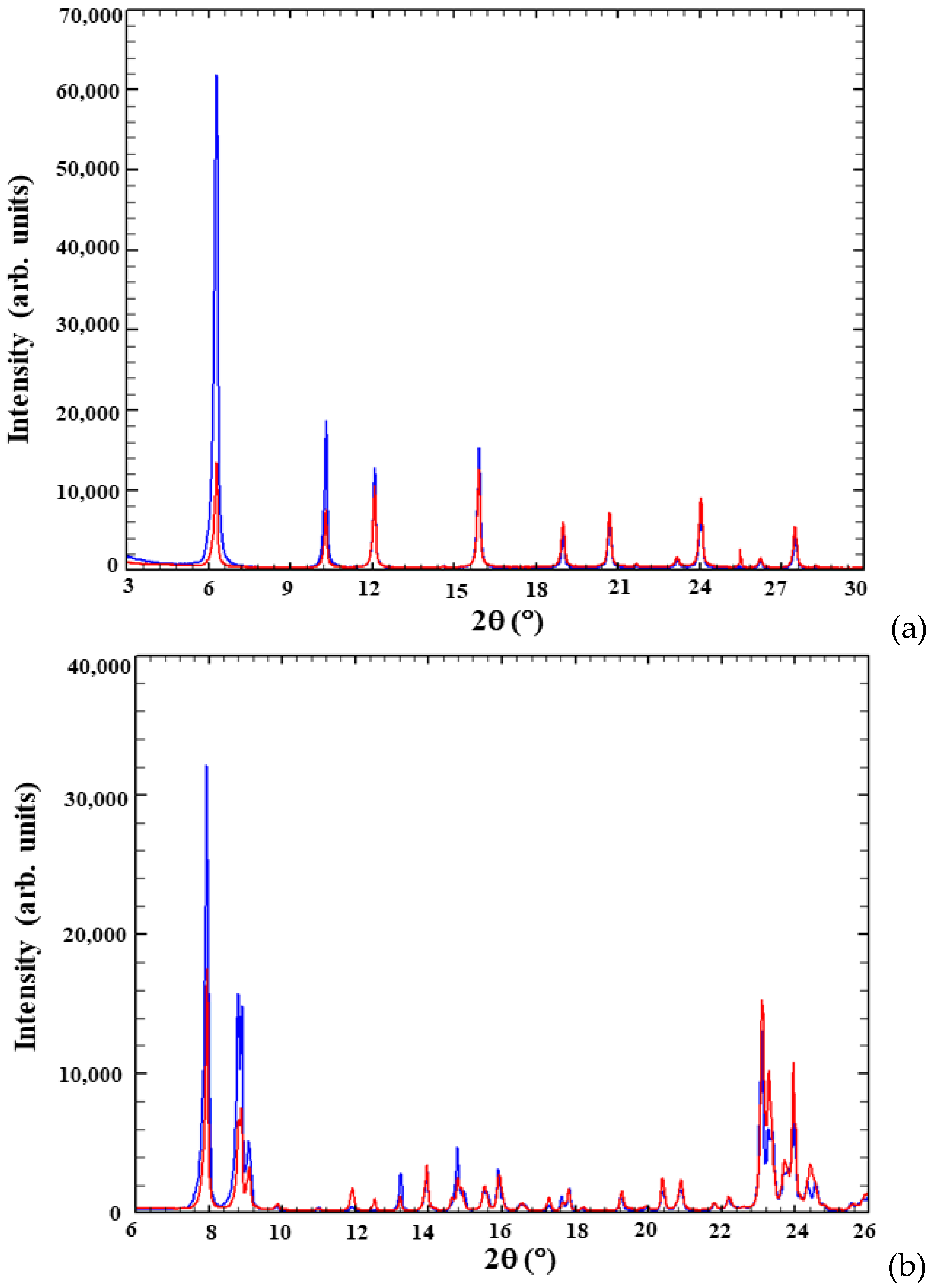
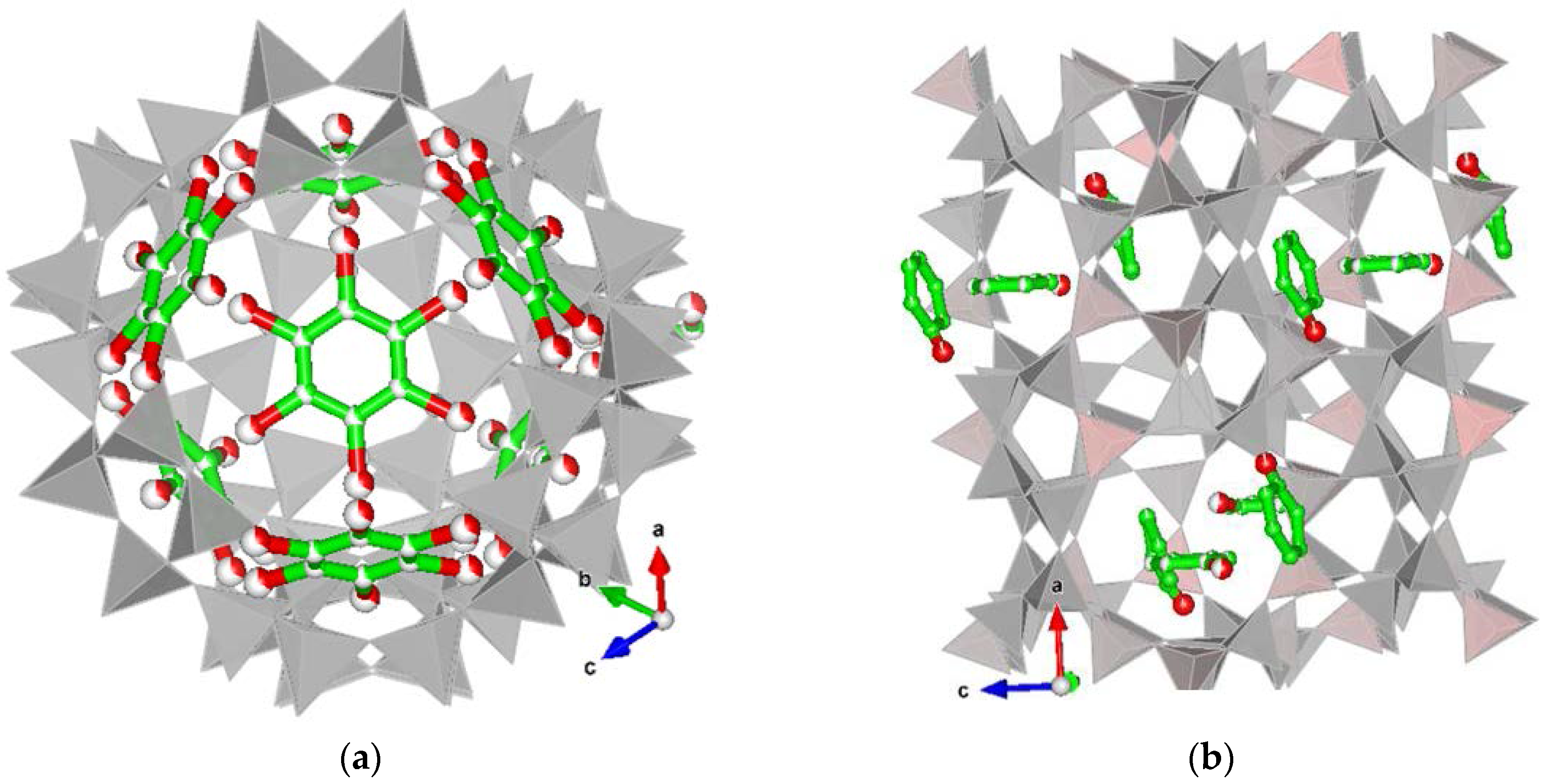
3.2. Structural Modifications upon Chlorobenzene Adsorption on High Silica Zeolites
3.2.1. Y–CB
3.2.2. ZSM-5–CB
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Sennour, R.; Mimane, G.; Benghalem, A.; Taleb, S. Removal of the persistent pollutant chlorobenzene by adsorption onto activated montmorillonite. Appl. Clay Sci. 2009, 43, 503–506. [Google Scholar] [CrossRef]
- McClenny, W.A.; Oliver, K.D.; Jacumin, H.H., Jr.; Daughtrey, E.H., Jr. Ambient level volatile organic compound (VOC) monitoring using solid adsorbents—Recent US EPA studies. J. Environ. Monitor. 2002, 4, 695–705. [Google Scholar] [CrossRef]
- Khan, F.I.; Ghoshal, A.K. Removal of Volatile Organic Compounds from polluted air. J. Prevent. Proc. 2000, 13, 527–545. [Google Scholar] [CrossRef]
- Sarti, E.; Chenet, T.; Pasti, L.; Cavazzini, A.; Rodeghero, E.; Martucci, A. Effect of silica alumina ratio and thermal treatment of beta zeolites on the adsorption of toluene from aqueous solutions. Minerals 2017, 7, 22. [Google Scholar] [CrossRef]
- Rodeghero, E.; Pasti, L.; Sarti, E.; Cruciani, G.; Bagatin, R.; Martucci, A. Temperature-induced desorption of methyl tert-butyl ether confined on ZSM-5: An in situ synchrotron XRD powder diffraction study. Minerals 2017, 7, 34. [Google Scholar] [CrossRef]
- Rodeghero, E.; Martucci, A.; Cruciani, G.; Bagatin, R.; Sarti, E.; Bosi, V.; Pasti, L. Kinetics and dynamic behaviour of toluene desorption from ZSM-5 using in situ high-temperature synchrotron powder X-ray diffraction and chromatographic techniques. Catal. Today 2016, 227, 118–125. [Google Scholar] [CrossRef]
- Arletti, R.; Martucci, A.; Alberti, A.; Pasti, L.; Nassi, M.; Bagatin, R. Location of MTBE and toluene in the channel system of the zeolite mordenite: Adsorption and host–guest interactions. J. Solid State Chem. 2012, 194, 135–142. [Google Scholar] [CrossRef]
- Pasti, L.; Rodeghero, E.; Sarti, E.; Bosi, V.; Cavazzini, A.; Bagatin, R.; Martucci, A. Competitive adsorption of VOCs from binary aqueous mixtures on zeolite ZSM-5. RSC Adv. 2016, 6, 54544–54552. [Google Scholar] [CrossRef]
- Pasti, L.; Sarti, E.; Cavazzini, A.; Marchetti, N.; Dondi, F.; Martucci, A. Factors affecting drug adsorption on beta zeolites. J. Sep. Sci. 2013, 36, 1604–1611. [Google Scholar] [CrossRef] [PubMed]
- Martucci, A.; Pasti, L.; Marchetti, N.; Cavazzini, A.; Dondi, F.; Alberti, A. Adsorption of pharmaceuticals from aqueous solutions on synthetic zeolites. Micropor. Mesopor. Mat. 2012, 148, 174–183. [Google Scholar] [CrossRef]
- Braschi, I.; Blasioli, S.; Gigli, L.; Gessa, C.E.; Alberti, A.; Martucci, A. Removal of sulfonamide antibiotics from water: Evidence of adsorption into an organophilic zeolite Y by its structural modifications. J. Hazard. Mater. 2010, 17, 218–225. [Google Scholar] [CrossRef] [PubMed]
- Blasioli, S.; Martucci, A.; Paul, G.; Gigli, L.; Cossi, M.; Johnston, C.T.; Marchese, L.; Braschi, I. Removal of sulfamethoxazole sulfonamide antibiotic from water by high silica zeolites: A study of the involved host–guest interactions by a combined structural, spectroscopic, and computational approach. J. Colloid. Interf. Sci. 2014, 419, 148–159. [Google Scholar] [CrossRef] [PubMed]
- Pasti, L.; Martucci, A.; Nassi, M.; Cavazzini, A.; Alberti, A.; Bagatin, R. The role of water in DCE adsorption from aqueous solutions onto hydrophobic zeolites. Micropor. Mesopor. Mat. 2012, 160, 182–193. [Google Scholar] [CrossRef]
- Guo, Y.; Li, Y.; Zhu, T.; Ye, M.; Wang, X. Adsorption of SO2 and chlorobenzene on activated carbon. Adsorpt. 2013, 19, 1109–1116. [Google Scholar] [CrossRef]
- Crisafully, R.; Milhome, M.A.L.; Cavalcante, R.M.; Silveira, E.R.; De Keukeleire, D.; Nascimento, R.F. Removal of some polycyclic aromatic hydrocarbons from petrochemical wastewater using low-cost adsorbents of natural origin. Bioresource Technol. 2008, 99, 4515–4519. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.K.; Ryu, S.K.; Kim, B.J.; Park, S.J. Roles of Acid-Base Interactions in Hydrogen Chloride Removal by Activated Carbon Fibers. J. Ind. Eng. Chem. 2006, 12, 121–126. [Google Scholar]
- Park, S.J.; Kim, B.J. Influence of oxygen plasma treatment on hydrogen chloride removal of activated carbon fibers. J. Colloid Interf. Sci. 2004, 275, 590–595. [Google Scholar] [CrossRef] [PubMed]
- Chin, C.J.M.; Shih, M.W.; Tsai, H.J. Adsorption of nonpolar benzene derivatives on single-walled carbon nanotubes. Appl. Surf. Sci. 2010, 256, 6035–6039. [Google Scholar] [CrossRef]
- Long, C.; Li, Q.; Li, Y.; Liu, Y.; Li, A.; Zhang, Q. Adsorption characteristics of benzene–chlorobenzene vapor on hypercrosslinked polystyrene adsorbent and a pilot-scale application study. Chem. Eng. J. 2010, 160, 723–728. [Google Scholar] [CrossRef]
- Chen, W.; Duan, L.; Zhu, D. Adsorption of polar and nonpolar organic chemicals to carbon nanotubes. Environ. Sci. Technol. 2007, 41, 8295–8300. [Google Scholar] [CrossRef] [PubMed]
- Balamurugan, K.; Subramanian, V. Adsorption of chlorobenzene onto (5,5) armchair single-walled Carbon nanotube and graphene sheet: Toxicity versus adsorption strength. J. Phys. Chem. C 2013, 117, 21217–21227. [Google Scholar] [CrossRef]
- Lee, J.J.; Choi, J.; Park, J.W. Simultaneous sorption of lead and chlorobenzene by organobentonite. Chemosphere 2002, 49, 1309–1315. [Google Scholar] [CrossRef]
- Witthuhn, B.; Klauth, P.; Pernyeszi, T.; Vereecken, H.; Klumpp, E. Organoclays for Aquifer Bioremediation: Adsorption of Chlorobenzene on Organoclays and its Degradation by RHODOCOCCUS B528. Water Air Soil Poll. Focus 2006, 6, 317–329. [Google Scholar] [CrossRef]
- Jarraya, I.; Fourmentin, S.; Benzina, M.; Bouaziz, S. VOC adsorption on raw and modified clay materials. Chem. Geol. 2010, 275, 1–8. [Google Scholar] [CrossRef]
- Altare, C.R.; Bowman, R.S.; Katz, L.E.; Kinney, K.A.; Sullivan, E.J. Regeneration and long-term stability of surfactant-modified zeolite for removal of volatile organic compounds from produced water. Micropor. Mesopor. Mat. 2007, 105, 305–316. [Google Scholar] [CrossRef]
- Anderson, M.A. Removal of MTBE and Other Organic Contaminants from Water by Sorption to High Silica Zeolites. Environ. Sci. Technol. 2000, 34, 725–727. [Google Scholar] [CrossRef]
- Damjanović, L.; Rakić, V.; Rac, V.; Stošić, D.; Auroux, A. The investigation of phenol removal from aqueous solutions by zeolites as solid adsorbents. J. Hazard. Mat. 2010, 184, 477–484. [Google Scholar] [CrossRef] [PubMed]
- Perego, C.; Bagatin, R.; Tagliabue, M.; Vignola, R. Zeolites and related mesoporous materials for multi-talented environmental solutions. Micropor. Mesopor. Mat. 2013, 166, 37–49. [Google Scholar] [CrossRef]
- Leardini, L.; Martucci, A.; Braschi, I.; Blasioli, S.; Quartieri, S. Regeneration of high-silica zeolites after sulfamethoxazole antibiotic adsorption: A combined in situ high-temperature synchrotron X-ray powder diffraction and thermal degradation study. Mineral. Mag. 2014, 78, 1141–1160. [Google Scholar] [CrossRef]
- Braschi, I.; Blasioli, S.; Buscaroli, E.; Montecchio, D.; Martucci, A. Physicochemical regeneration of high silica zeolite Y used to clean-up water polluted with sulfonamide antibiotics. J. Environ. Sci. 2016, 43, 302–312. [Google Scholar] [CrossRef] [PubMed]
- Vignola, R.; Cova, U.; Fabiani, F.; Sbardellati, T.; Sisto, R.; Vignola, R. Process for the regeneration of nonpolar adsorbing zeolites used for the treatment of contaminated water. Patent WO/2009/000429, 31 December 2008. [Google Scholar]
- Alberti, A.; Martucci, A. Phase transformations and structural modifications induced by heating in microporous materials. Stud. Surf. Sci. Catal. 2005, 155, 19–43. [Google Scholar] [CrossRef]
- Alberti, A.; Martucci, A. Reconstructive phase transitions in microporous materials: Rules and factors affecting them. Micropor. Mesopor. Mat. 2011, 141, 192–198. [Google Scholar] [CrossRef]
- Baerlocher, C.; McCusker, L.B.; Olson, D.H. Atlas of Zeolite Framework Types, 6th ed.; Elsevier: Amsterdam, The Netherland, 2007. [Google Scholar]
- Larson, A.C.; Von Dreele, R.B. GSAS General Structure Analysis System, LANSCE, MS-H805; Los Alamos National Laboratory: Los Alamos, NM, USA, 1994. [Google Scholar]
- Toby, B.H. EXPGUI, a graphical user interface for GSAS. J. Appl. Crystallogr. 2001, 34, 210–213. [Google Scholar] [CrossRef]
- Azizian, S.; Haerifar, M.; Bashiri, H. Adsorption of methyl violet onto granular activated carbon: Equilibrium, kinetics and modeling. Chem. Eng. J. 2009, 146, 36–41. [Google Scholar] [CrossRef]
- De Moor, B.A.; Reyniers, M.F.; Gobin, O.C.; Lercher, J.A.; Marin, G.B. Adsorption of C2−C8 n-Alkanes in Zeolites. J. Phys. Chem. C 2011, 115, 1204–1219. [Google Scholar] [CrossRef]
- Martucci, A.; Braschi, I.; Bisio, C.; Sarti, E.; Rodeghero, E.; Bagatin, R.; Pasti, L. Influence of water on the retention of methyl tertiary-butyl ether by high silica ZSM-5 and Y zeolites: A multidisciplinary study on the adsorption from liquid and gas phase. RSC Advances 2015, 5, 86997–87006. [Google Scholar] [CrossRef]
- Martucci, A.; Braschi, I.; Marchese, L.; Quartieri, S. Recent advances in clean-up strategies of waters polluted with sulfonamide antibiotics: A review of sorbents and related properties. Mineral. Mag. 2014, 78, 1115–1140. [Google Scholar] [CrossRef]
- Li, X.; Chen, G.H. Surface modified graphite nanosheets used as adsorbent to remove 1,2-dichlorobenzene from water. Mater. Lett. 2009, 63, 930–932. [Google Scholar] [CrossRef]
- Yu, F.; Ma, J.; Wu, Y. Adsorption of toluene, ethylbenzene and m-xylene on multi-walled carbon nanotubes with different oxygen contents from aqueous solutions. J. Hazard. Mater. 2011, 192, 1370–1379. [Google Scholar] [CrossRef] [PubMed]
- Krishna, R.; van Baten, J.M. Highlighting a Variety of Unusual Characteristics of Adsorption and Diffusion in Microporous Materials Induced by Clustering of Guest Molecules. Langmuir 2010, 26, 8450–8463. [Google Scholar] [CrossRef] [PubMed]
- Wragg, D.S.; Morris, R.E.; Burton, A.W. Pure silica zeolite-type frameworks: A structural analysis. Chem. Mater. 2008, 20, 1561–1570. [Google Scholar] [CrossRef]
- Leardini, L.; Martucci, A.; Alberti, A.; Cruciani, G. Template burning effects on stability and boron coordination in boron levyne studied by in situ time resolved synchrotron powder diffraction. Micropor. Mesopor. Mater. 2013, 167, 117–126. [Google Scholar] [CrossRef]
- Baur, W.H. Self-limiting distortion by antirotating hinges is the principle of flexible but noncollapsible frameworks. J. Solid State Chem. 1992, 97, 243–247. [Google Scholar] [CrossRef]
- Momma, K.; Izumi, F. VESTA 3 for three-dimensional visualization of crystal, volumetric and morphology data. J. Appl. Crystallogr. 2011, 44, 1272–1276. [Google Scholar] [CrossRef]
| Parameter | ZSM-5 | ZSM-5–CB | Y | Y–CB |
|---|---|---|---|---|
| [Si96O192] | [Si96O192]·6(C6H5Cl)·8(H2O) | [Si192O384] | [Si192O384]·32(C6H5Cl)·70(H2O) | |
| Space group | P21/n | P21/n | Fd-3m | Fd-3 |
| a (Å) | 19.899(5) | 19.919(1) | 24.259(1) | 24.263(1) |
| b (Å) | 20.117(6) | 20.107(1) | 24.259(1) | 24.263(1) |
| c (Å) | 13.389(4) | 13.3967(1) | 24.259(1) | 24.263(1) |
| α (°) | 90 | 90 | 90 | 90 |
| β (°) | 90.546(3) | 90.528(3) | 90 | 90 |
| γ (°) | 90 | 90 | 90 | 90 |
| V (Å3) | 5359.9(3) | 5365.7(5) | 14,277.1(1) | 14,284.4(6) |
| Wavelength (Å): Cu Kα1 | 1.540593 | 1.540593 | 1.540593 | 1.540593 |
| Cu Kα2 | 1.544427 | 1.544427 | 1.544427 | 1.544427 |
| Refined 2θ (°) range | 3°–110° | 3°–110° | 3°–100° | 3°–100° |
| Contributing reflections | 5861 | 6362 | 620 | 620 |
| Nobs | 5350 | 5350 | 4850 | 4850 |
| Nvar | 289 | 301 | 40 | 40 |
| Rwp (%) | 10.3 | 13.3 | 12.8 | 12.9 |
| Rp (%) | 9.4 | 10.4 | 12.5 | 12.6 |
| RF2 (%) | 7.00 | 7.25 | 9.95 | 10.0 |
| Rp = Σ[Yio − Yic]/ΣYio; Rwp = [Σwi(Yio − Yic)2/ΣwiYio2]0.5; RF2 = Σ|Fo2 − Fc2|/|Fo2| | ||||
| C0 (mg·L−1) | qt (mg·g−1) | k2 (g·L−1·mg−1) | R2 | |
|---|---|---|---|---|
| ZSM-5 | 15 | 23.4 (23.3, 23.5) | 0.44 (0.41, 0.47) | 0.9845 |
| 25 | 44.3 (44.2, 44.3) | 0.37 (0.33, 0.41) | 0.9803 | |
| 35 | 67.6 (67.5, 67.6) | 0.21 (0.19, 0.23) | 0.9985 | |
| Y | 25 | 39.3 (39.2, 39.4) | 0.14 (0.11, 0.17) | 0.9822 |
| 35 | 70.3 (70.2, 70.3) | 0.13 (0.11, 0.15) | 0.9969 | |
| 45 | 82.3 (82.4, 82.5) | 0.10 (0.085, 0.12) | 0.9935 |
| R2 | SSE | |||
|---|---|---|---|---|
| CB–ZSM-5 | Langmuir | |||
| qs | 76.6 (74.8, 79.5) | 0.9874 | 198 | |
| b | 5.0 (3.7, 6.6) | |||
| CB–Y | Hill | |||
| qs | 227 (218, 245) | 0.9654 | 2135 | |
| KH | 9.2 (6.6, 12.1) | |||
| n | 5.5 (4.3, 6.7) | |||
| Parameter | Y–CB | Y [11] |
|---|---|---|
| O4–O4 distance (Å) | 11.16 | 9.81 |
| O1–O1 distance (Å) | 9.68 | 9.70 |
| Free diameter O4–O4 (Å) | 8.46 | 7.11 |
| Free diameter O1–O1 (Å) | 6.98 | 7.00 |
| Mean diameter (Å) | 7.72 | 7.06 |
| Mean radius (Å) | 3.86 | 3.53 |
| C.F.A. (Å2) | 46.81 | 39.07 |
| ε | 1.21 | 1.01 |
| Straight Channel | ||||||
| O–O distance (Å) | O7–O1 | O8–O2 | O31–O37 | O44–O46 | O47–O48 | ε |
| ZSM-5–CB | 8.22 | 8.20 | 8.02 | 8.22 | 7.97 | 1.03 |
| ZSM-5 | 7.90 | 7.94 | 8.04 | 8.41 | 7.80 | 1.08 |
| Free diameter (Å) | O7–O1 | O8–O2 | O31–O37 | O44–O46 | O47–O48 | |
| ZSM-5–CB | 5.52 | 5.50 | 5.32 | 5.52 | 5.27 | - |
| ZSM-5 | 5.20 | 5.24 | 5.34 | 5.71 | 5.10 | - |
| Mean diameter (Å) | Mean radius (Å) | C.F.A. (Å2) | ||||
| ZSM-5–CB | 5.43 | 2.71 | 23.12 | - | ||
| ZSM-5 | 5.32 | 2.66 | 22.20 | - | ||
| O–O distance (Å) | O11–O5 | O20–O18 | O21–O22 | O27–O33 | O28–O34 | ε |
| ZSM-5–CB | 8.08 | 7.89 | 8.11 | 8.22 | 8.08 | 1.04 |
| ZSM-5 | 8.19 | 8.35 | 7.98 | 8.13 | 7.76 | 1.08 |
| Free diameter (Å) | O11–O5 | O20–O18 | O21–O22 | O27–O33 | O28–O34 | |
| ZSM-5–CB | 5.38 | 5.19 | 5.41 | 5.52 | 5.38 | |
| ZSM-5 | 5.49 | 5.65 | 5.28 | 5.43 | 5.06 | |
| Mean diameter (Å) | Mean radius (Å) | C.F.A. (Å2) | ||||
| ZSM-5–CB | 5.37 | 2.69 | 22.67 | |||
| ZSM-5 | 5.38 | 2.69 | 22.74 | |||
| Sinusoidal Channel | ||||||
| O–O distance (Å) | O20–O15 | O24–O26 | O27–O2 | O28–O1 | O41–O46 | ε |
| ZSM-5–CB | 8.08 | 7.87 | 7.76 | 8.16 | 8.69 | 1.12 |
| ZSM-5 | 8.15 | 8.07 | 7.97 | 7.76 | 8.51 | 1.10 |
| Free diameter (Å) | O20–O15 | O24–O26 | O27–O2 | O28–O1 | O41–O46 | |
| ZSM-5–CB | 5.38 | 5.17 | 5.06 | 5.46 | 5.99 | |
| ZSM-5 | 5.45 | 5.37 | 5.27 | 5.06 | 5.81 | |
| Mean diameter (Å) | Mean radius (Å) | C.F.A. (Å2) | ||||
| ZSM-5–CB | 5.41 | 2.71 | 23.01 | |||
| ZSM-5 | 5.39 | 2.70 | 22.82 | |||
| O–O distance (Å) | O17–O18 | O23–O25 | O30–O5 | O31–O4 | O44–O43 | ε |
| ZSM-5–CB | 7.50 | 8.58 | 8.20 | 7.89 | 7.75 | 1.14 |
| ZSM-5 | 7.30 | 8.31 | 8.32 | 8.05 | 7.95 | 1.14 |
| Free diameter (Å) | O17–O18 | O23–O25 | O30–O5 | O31–O4 | O44–O43 | |
| ZSM-5–CB | 4.80 | 5.88 | 5.50 | 5.19 | 5.05 | |
| ZSM-5 | 4.60 | 5.61 | 5.62 | 5.35 | 5.25 | |
| Mean diameter (Å) | Mean radius (Å) | C.F.A. (Å2) | ||||
| ZSM-5–CB | 5.28 | 2.64 | 21.92 | |||
| ZSM-5 | 5.29 | 2.64 | 21.93 | |||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pasti, L.; Rodeghero, E.; Beltrami, G.; Ardit, M.; Sarti, E.; Chenet, T.; Stevanin, C.; Martucci, A. Insights into Adsorption of Chlorobenzene in High Silica MFI and FAU Zeolites Gained from Chromatographic and Diffractometric Techniques. Minerals 2018, 8, 80. https://doi.org/10.3390/min8030080
Pasti L, Rodeghero E, Beltrami G, Ardit M, Sarti E, Chenet T, Stevanin C, Martucci A. Insights into Adsorption of Chlorobenzene in High Silica MFI and FAU Zeolites Gained from Chromatographic and Diffractometric Techniques. Minerals. 2018; 8(3):80. https://doi.org/10.3390/min8030080
Chicago/Turabian StylePasti, Luisa, Elisa Rodeghero, Giada Beltrami, Matteo Ardit, Elena Sarti, Tatiana Chenet, Claudia Stevanin, and Annalisa Martucci. 2018. "Insights into Adsorption of Chlorobenzene in High Silica MFI and FAU Zeolites Gained from Chromatographic and Diffractometric Techniques" Minerals 8, no. 3: 80. https://doi.org/10.3390/min8030080
APA StylePasti, L., Rodeghero, E., Beltrami, G., Ardit, M., Sarti, E., Chenet, T., Stevanin, C., & Martucci, A. (2018). Insights into Adsorption of Chlorobenzene in High Silica MFI and FAU Zeolites Gained from Chromatographic and Diffractometric Techniques. Minerals, 8(3), 80. https://doi.org/10.3390/min8030080