Thermal Stability of Woolly Erionite-K and Considerations about the Heat-Induced Behaviour of the Erionite Group
Abstract
1. Introduction
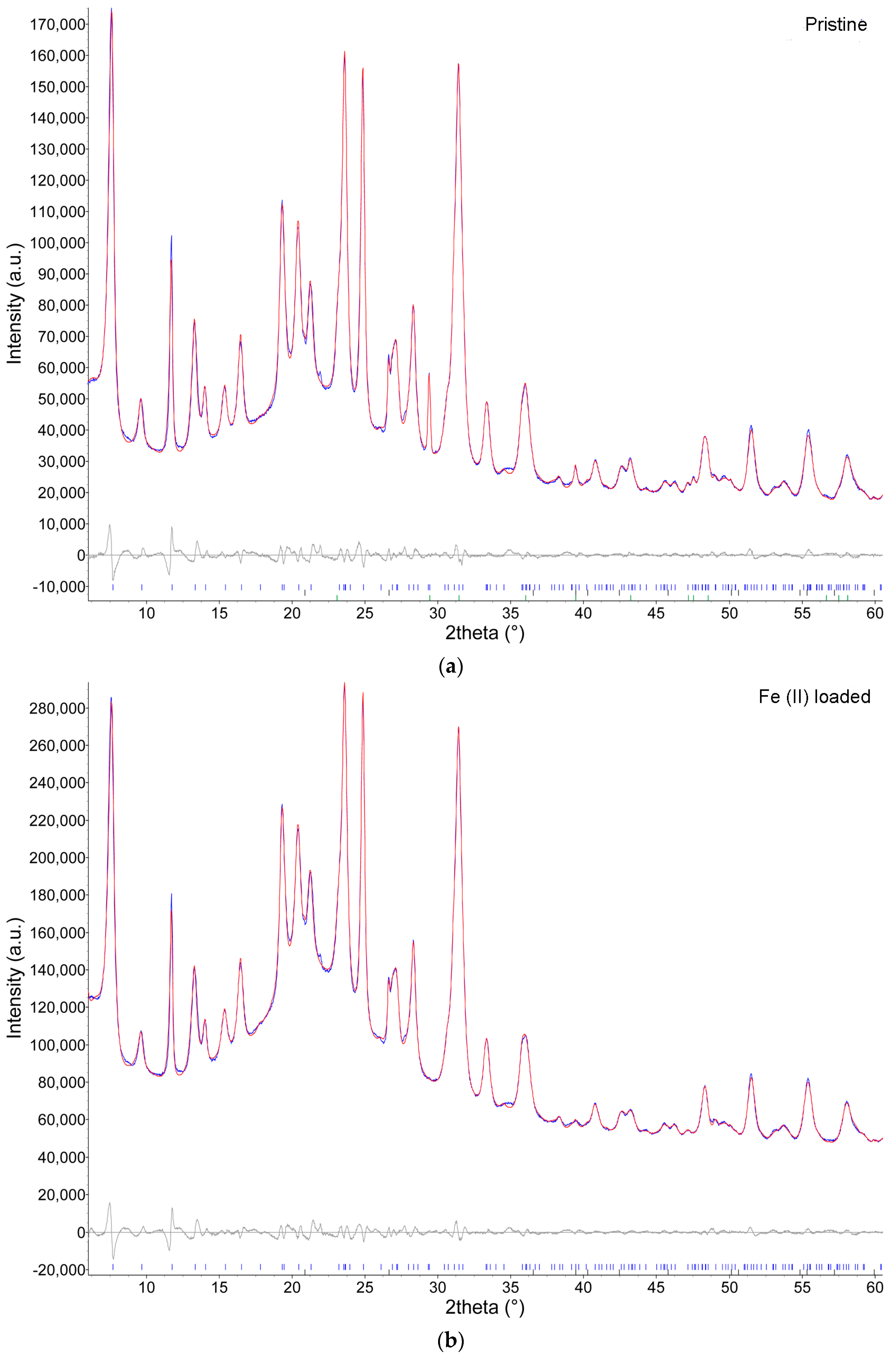
2. Materials and Methods
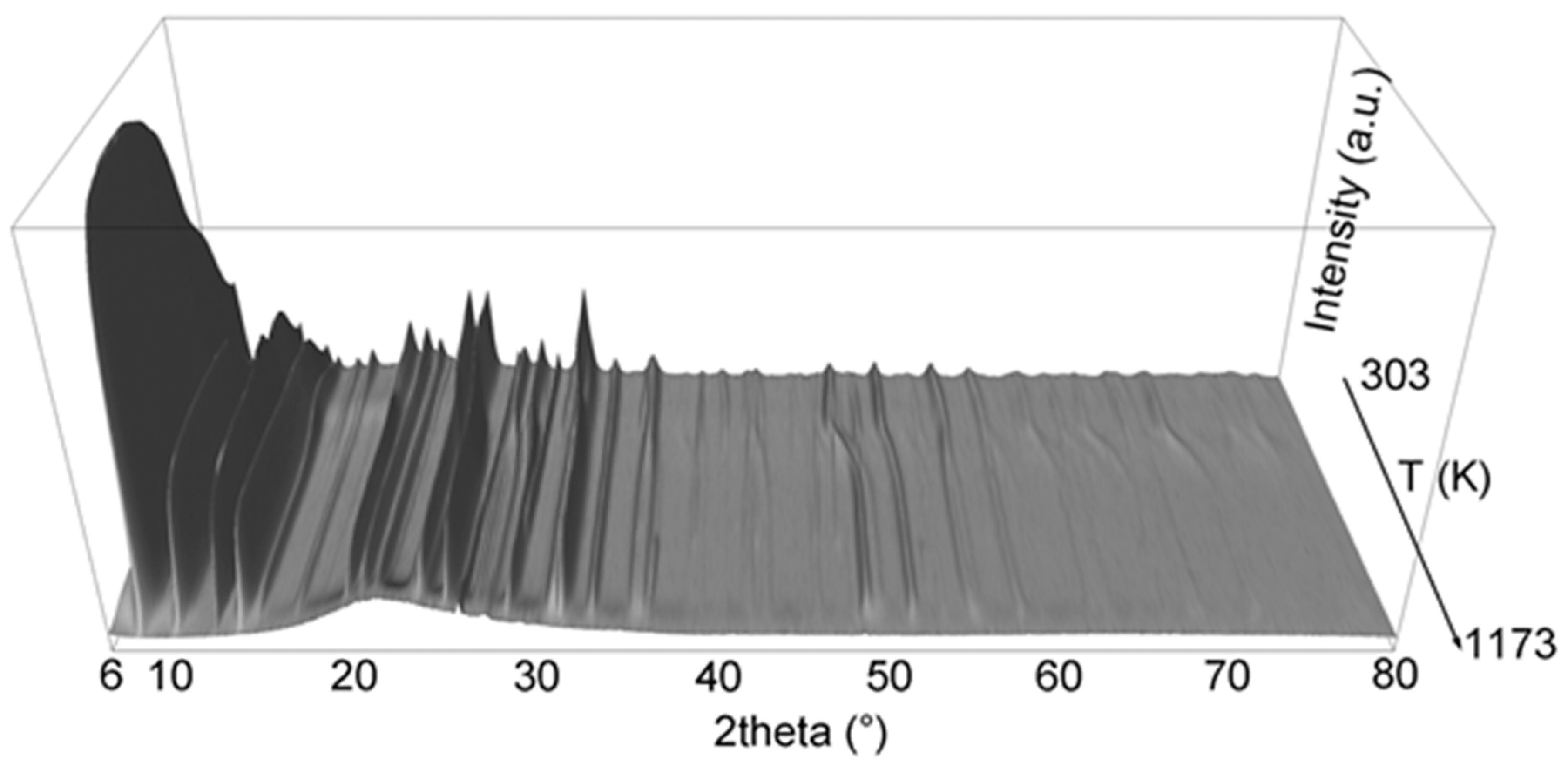
3. Results and Discussion
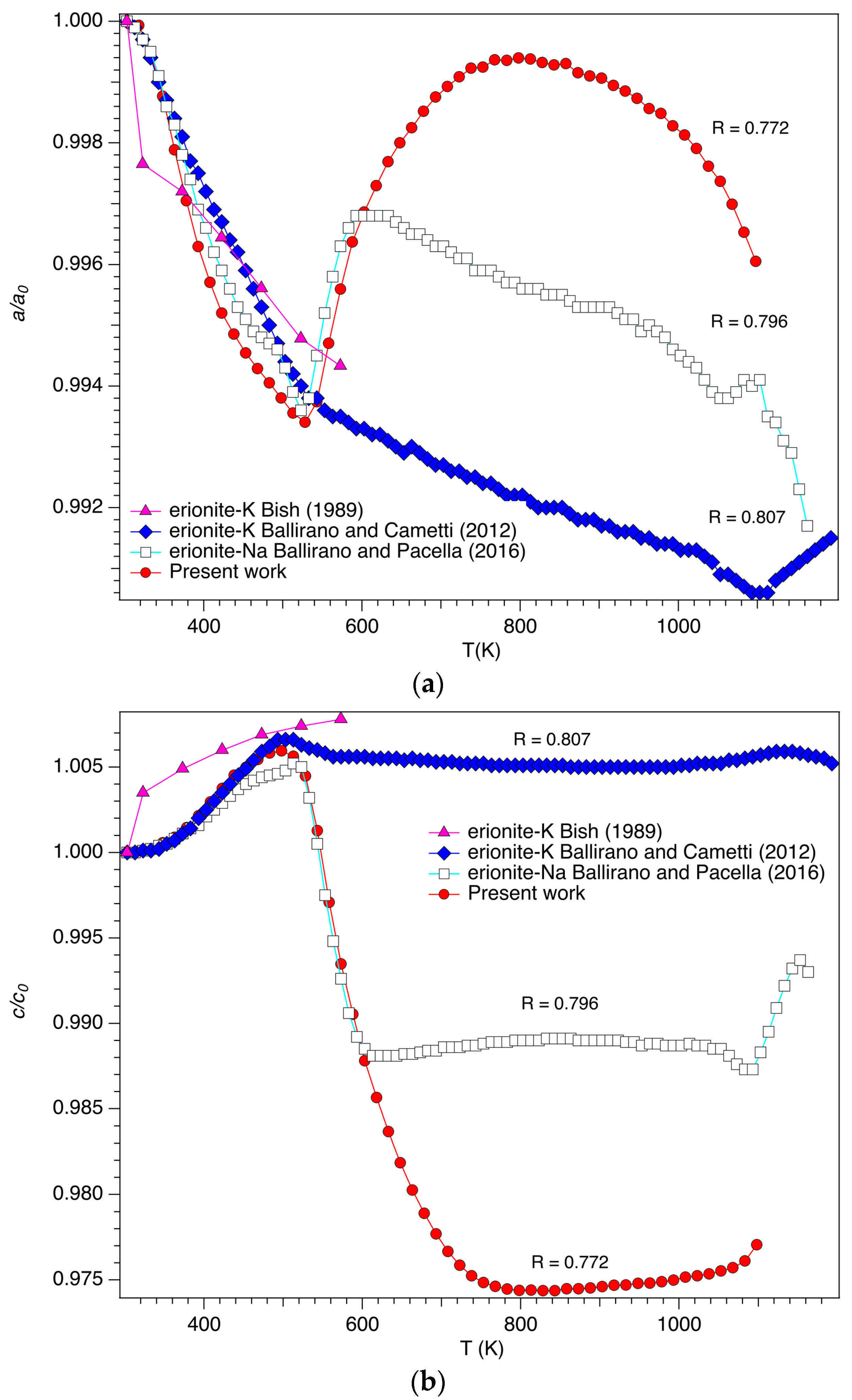
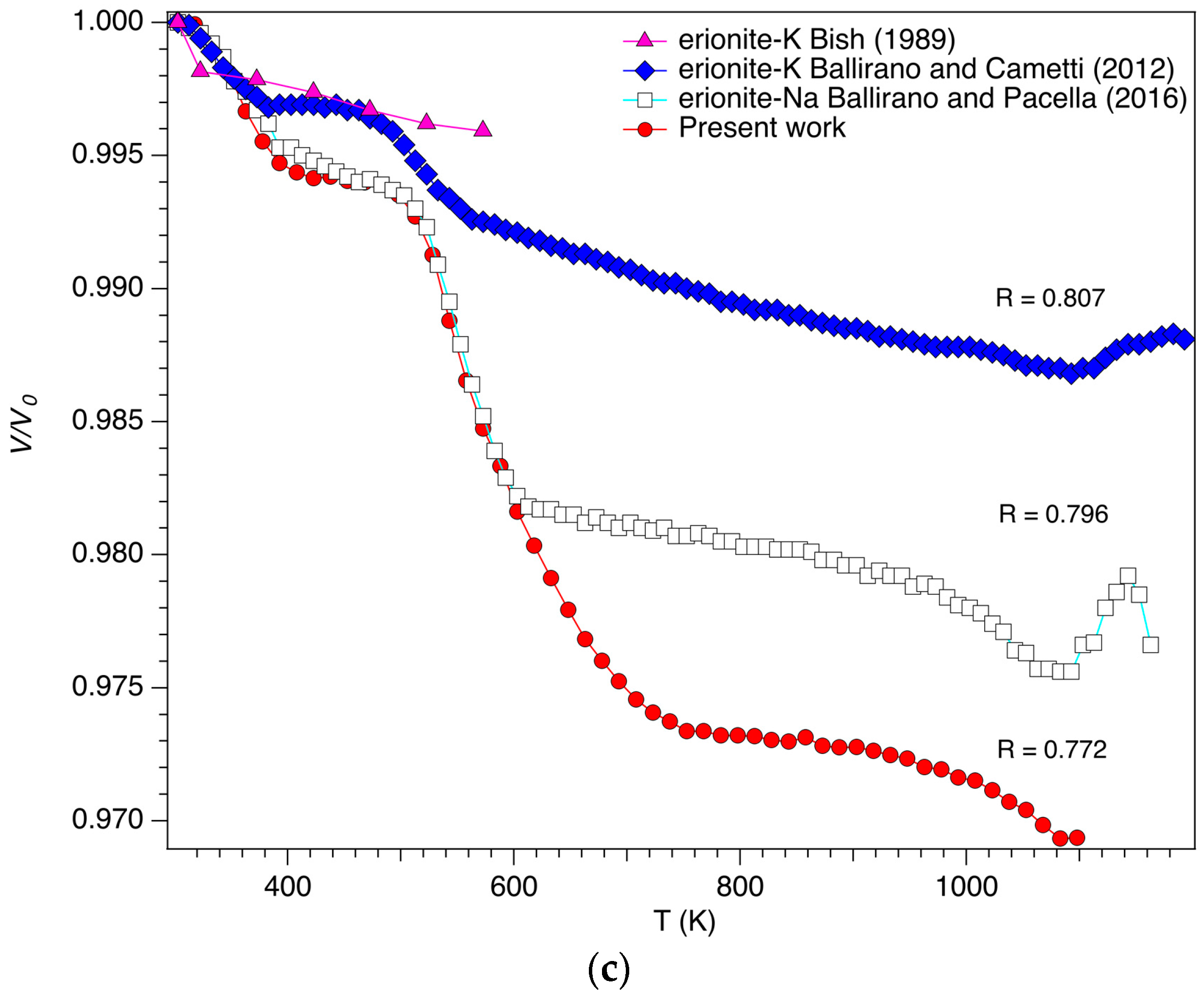
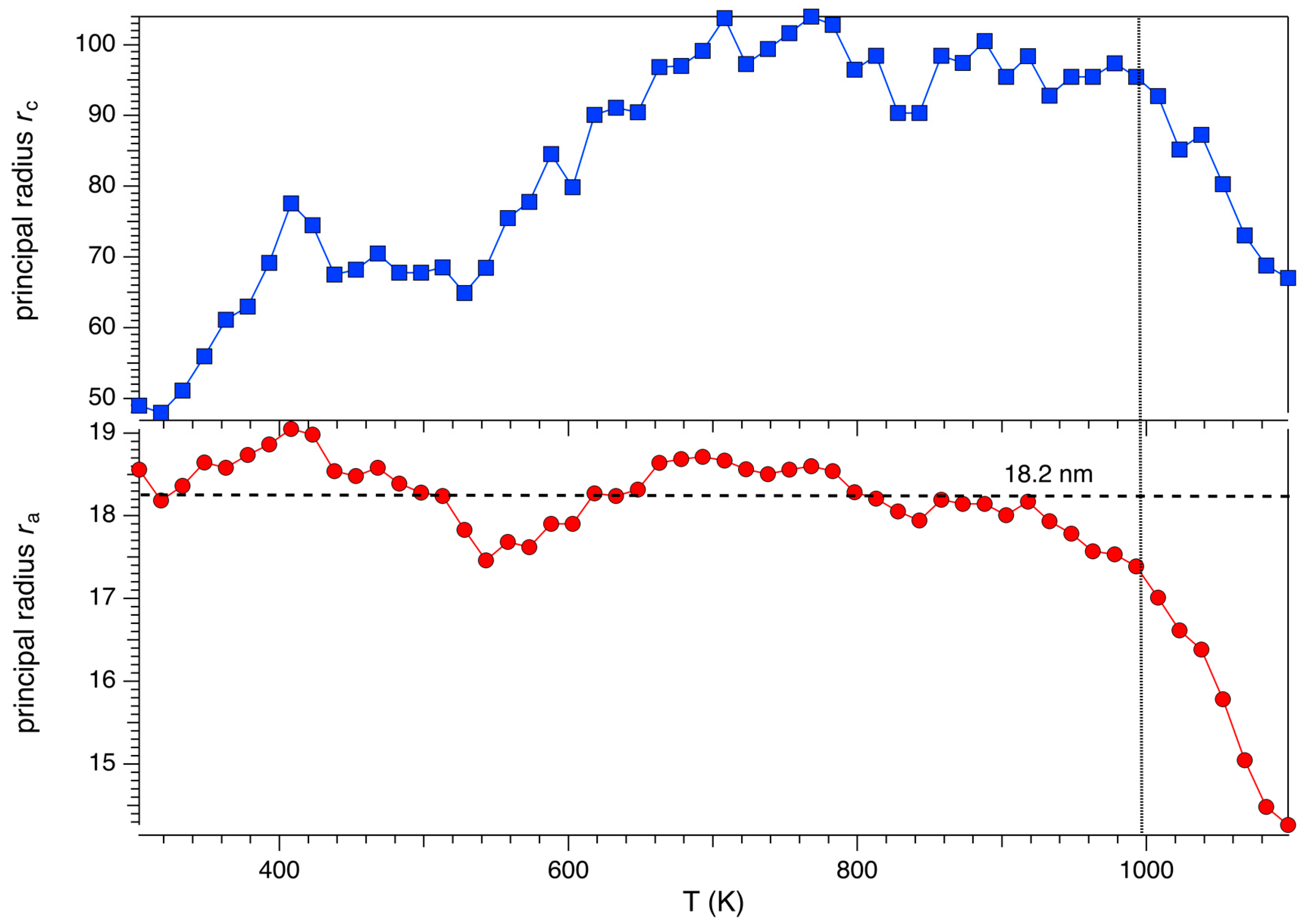
3.1. Cell Parameters and Volume Dependence from Temperature
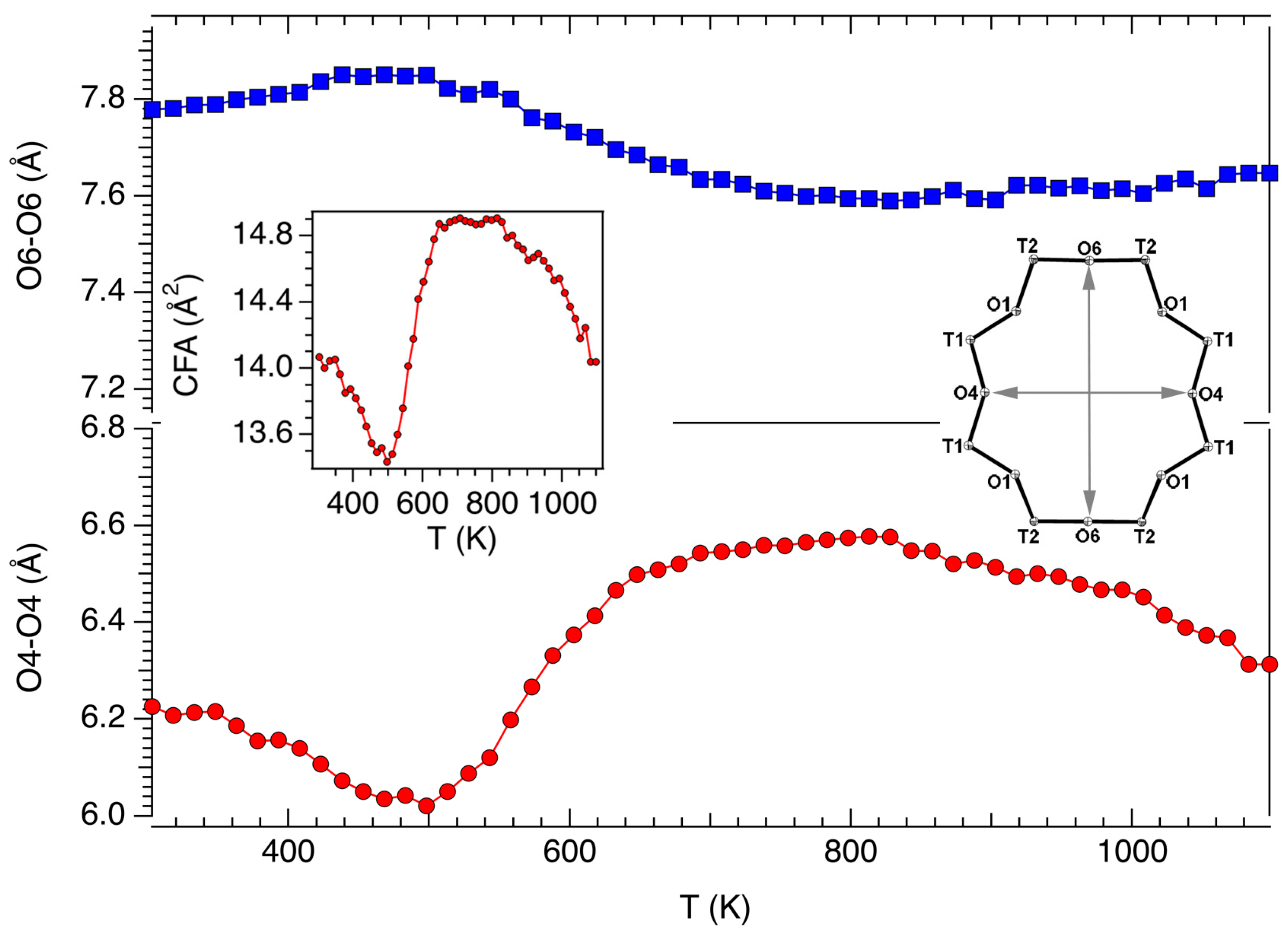
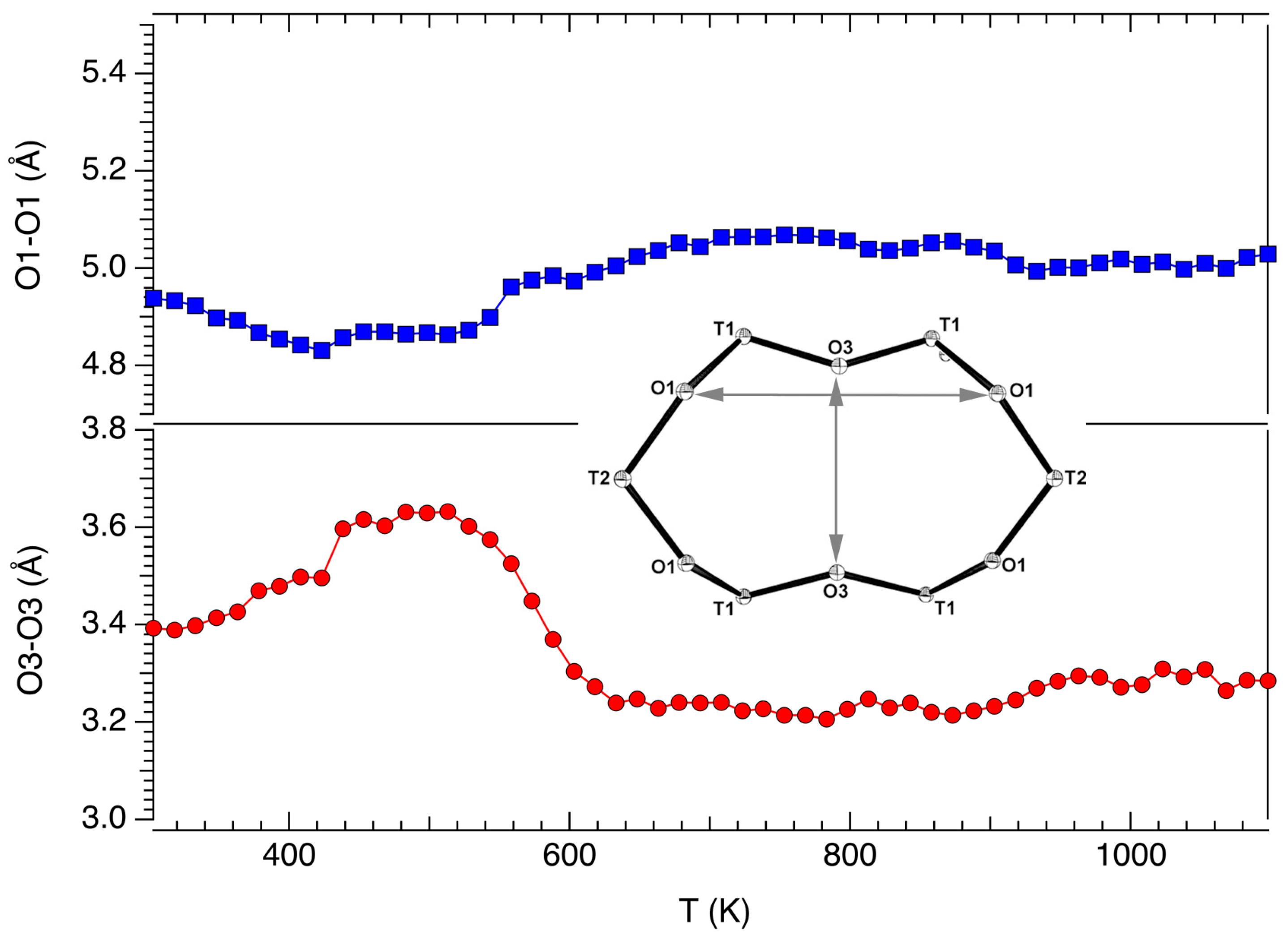
3.2. Structural Modifications: Framework
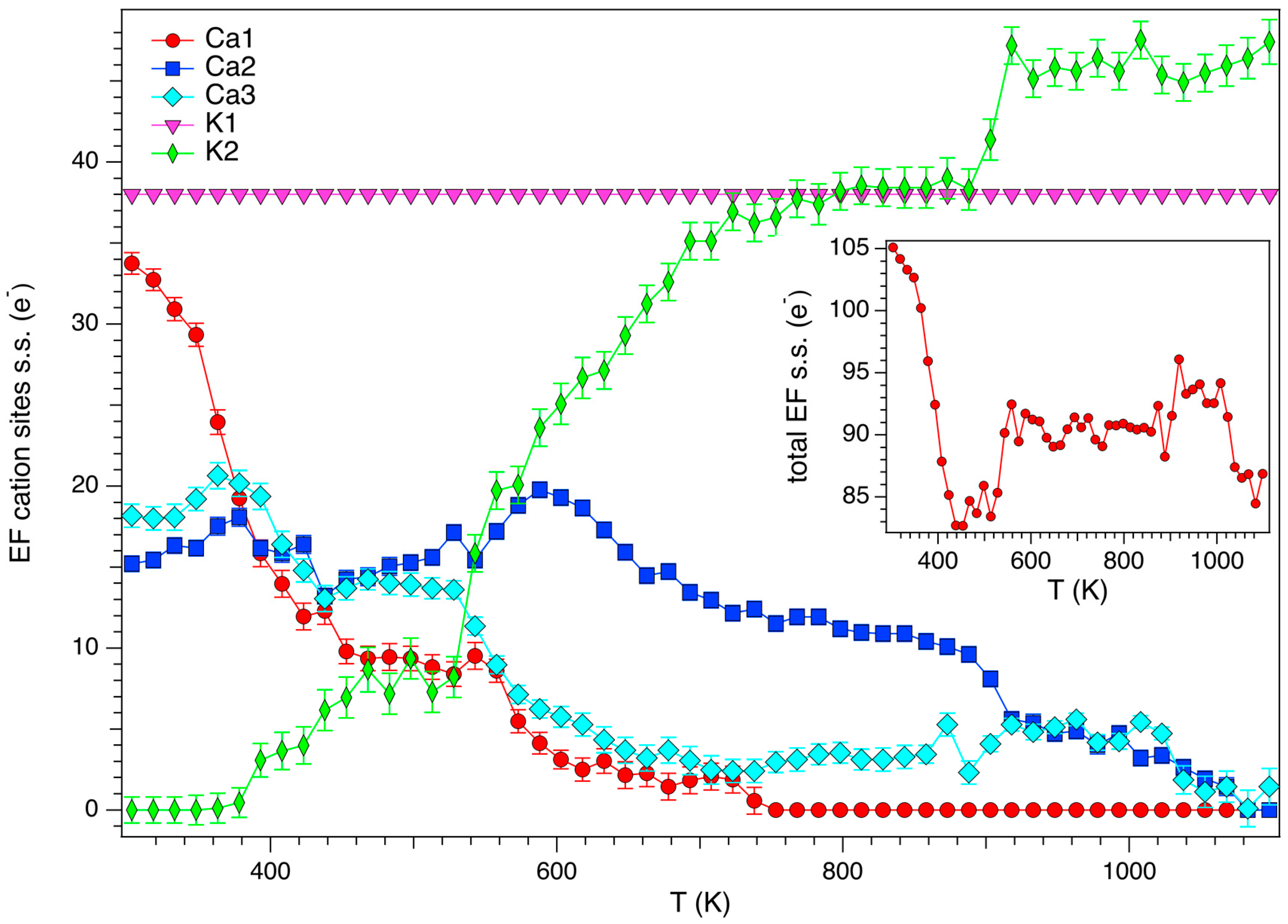
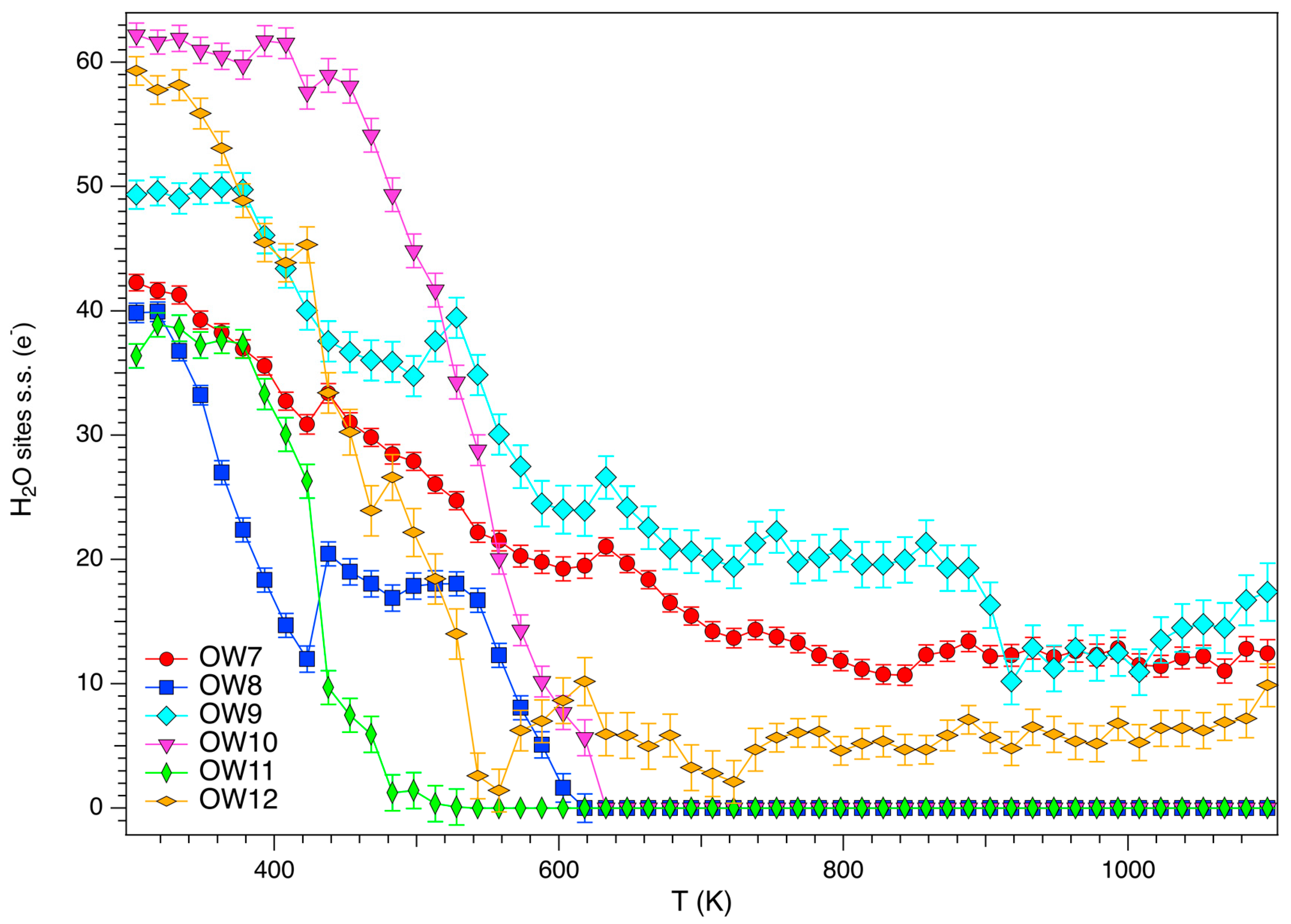
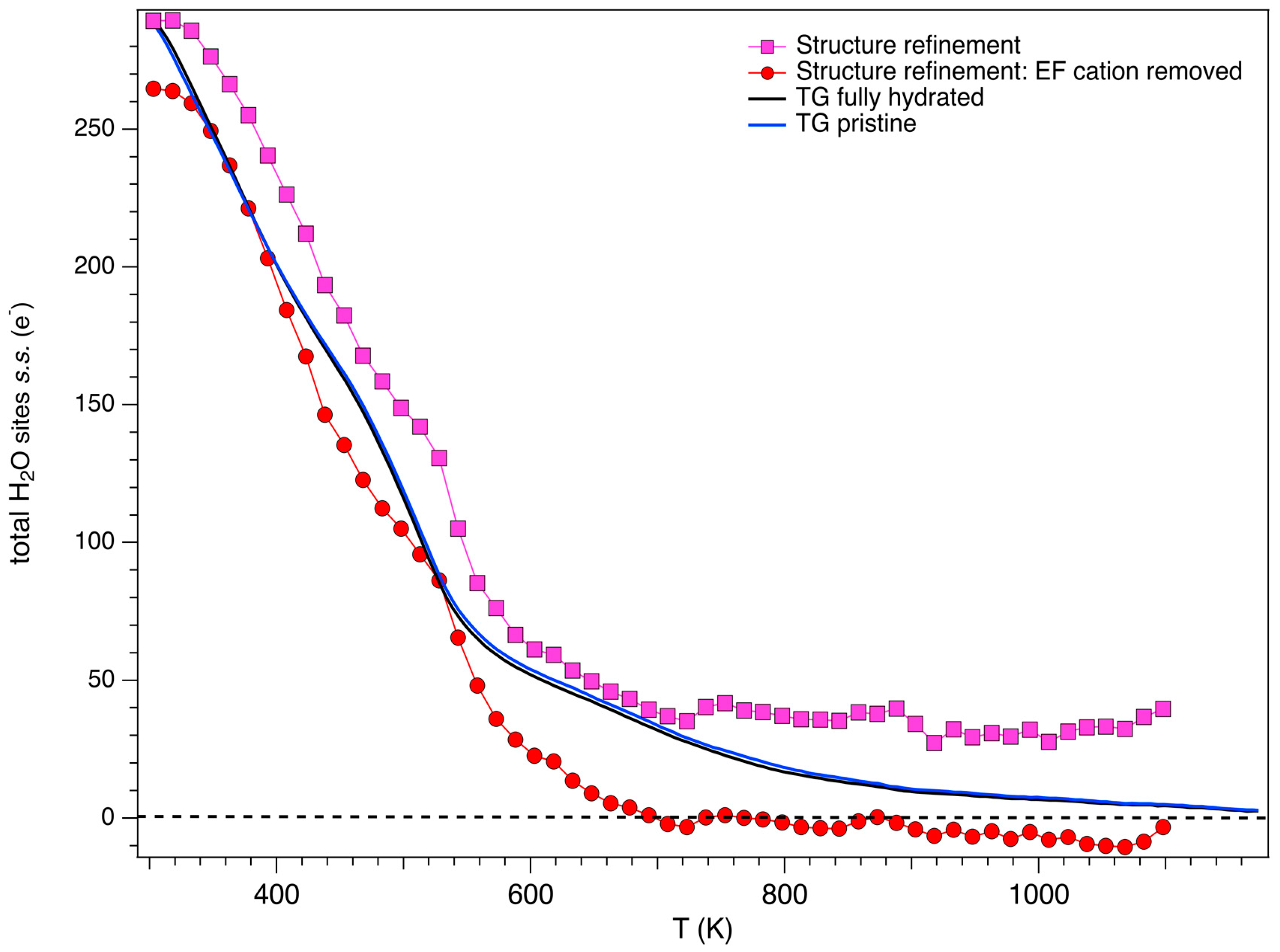
3.3. Structural Modifications: EF Cations and Water Molecules
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gottardi, G.; Galli, E. Natural Zeolites; Springer: Heidelberg, Germany, 1985; 409p. [Google Scholar]
- IARC. IARC Monographs on the evaluation of the carcinogenic risk of chemicals to humans. Silica Some Silic. 1997, 42, 225–239. [Google Scholar]
- Carbone, M.; Baris, Y.I.; Bertino, P.; Brass, B.; Comertpay, S.; Dogan, A.U.; Gaudino, G.; Jube, S.; Kanodia, S.; Partridge, C.R.; et al. Erionite exposure in North Dakota and Turkish villages with mesothelioma. Proc. Natl. Acad. Sci. USA 2011, 108, 13618–13623. [Google Scholar] [CrossRef] [PubMed]
- Kawahara, A.; Curien, H. La structure crystalline de l’érionite. B. Soc. Fr. Mineral. Cr. 1969, 92, 250–256. [Google Scholar]
- Coombs, D.S.; Alberti, A.; Armbruster, T.; Artioli, G.; Colella, C.; Galli, E.; Grice, J.D.; Liebau, F.; Mandarino, J.A.; Minato, H.; et al. Recommended nomenclature for zeolite minerals; report of the Subcommittee on Zeolites of the International Mineralogical Association, Commission on New Minerals and Mineral Names. Can. Mineral. 1997, 35, 1571–1606. [Google Scholar] [CrossRef]
- Wise, W.S.; Tschernich, R.W. The chemical compositions and origin of the zeolites offretite, erionite, and levyne. Am. Mineral. 1976, 61, 853–863. [Google Scholar]
- Passaglia, E.; Artioli, G.; Gualtieri, A. Crystal chemistry of the zeolites erionite and offretite. Am. Mineral. 1998, 83, 577–589. [Google Scholar] [CrossRef]
- Gualtieri, A.; Artioli, G.; Passaglia, E.; Bigi, S.; Viani, A.; Hanson, J.C. Crystal structure-crystal chemistry relationships in the zeolites erionite and offretite. Am. Mineral. 1998, 83, 590–600. [Google Scholar] [CrossRef]
- Staples, L.W.; Gard, J.A. The fibrous zeolite erionite: Its occurrence, unit cell, and structure. Mineral. Mag. 1959, 32, 261–281. [Google Scholar] [CrossRef]
- Gard, J.A.; Tait, J.M. Refinement of the crystal structure of erionite. In Proceedings of the Third International Conference on Molecular Sieves; Uytterhoeven, J.B., Ed.; Leuven University Press: Leuven, Belgium, 1973; p. 94. [Google Scholar]
- Meier, W.M.; Groner, M. Zeolite structure type EAB: Crystal structure and mechanism for the topotactic transformation of the Na, TMA form. J. Solid State Chem. 1981, 37, 204–218. [Google Scholar] [CrossRef]
- Smith, J.V.; Bennett, J.M. Enumeration of 4-connected 3-dimensional nets and classification of framework silicates: The infinite set of ABC-6 nets; the Archimedean and 6-related nets. Am. Mineral. 1981, 66, 777–788. [Google Scholar]
- Smith, J.V.; Rinaldi, F.; Dent Glasser, L.S. Crystal structures with a chabazite framework. II. Hydrated Ca-chabazite at room temperature. Acta Cryst. 1963, 16, 45–53. [Google Scholar] [CrossRef]
- Rüdinger, B.; Tillmanns, E.; Hentschel, G. Bellbergite—A new mineral with the structure type EAB. Mineral. Petrol. 1993, 48, 147–152. [Google Scholar]
- Ballirano, P.; Merlino, S.; Bonaccorsi, E.; Maras, A. The crystal structure of liottite, a six-layer member of the cancrinite group. Can. Mineral. 1996, 34, 1021–1030. [Google Scholar]
- Alberti, A.; Martucci, A.; Galli, E.; Vezzalini, G. A reexamination of the crystal structure of erionite. Zeolites 1997, 19, 349–352. [Google Scholar] [CrossRef]
- Ballirano, P.; Andreozzi, G.B.; Dogan, M.; Dogan, A.U. Crystal structure and iron topochemistry of erionite-K from Rome, Oregon, U.S.A. Am. Mineral. 2009, 94, 1262–1270. [Google Scholar] [CrossRef]
- Schlenker, J.L.; Pluth, J.J.; Smith, J.V. Dehydrated natural erionite with stacking faults of the offretite type. Acta Crystallogr. 1977, B33, 3265–3268. [Google Scholar] [CrossRef]
- Ballirano, P.; Cametti, G. Dehydration dynamics and thermal stability of erionite-K: Experimental evidence of the “internal ionic exchange” mechanism. Microporous Mesoporous Mater. 2012, 163, 160–168. [Google Scholar] [CrossRef]
- Ballirano, P.; Pacella, A. Erionite-Na upon heating: Dehydration dynamics and exchangeable cations mobility. Sci. Rep. 2016, 6, 22786. [Google Scholar] [CrossRef] [PubMed]
- Bish, D.L.; Carey, J.W. Thermal properties of natural zeolites. In Natural Zeolites: Occurrence, Properties, Applications; Bish, D.L., Ming, D.W., Eds.; Mineralogical Society of America: Chantilly, VA, USA, 2001; Volume 45, pp. 403–452. [Google Scholar]
- Cruciani, G. Zeolites upon heating: Factors governing their thermal stability and structural changes. J. Phys. Chem. Solids 2006, 67, 1973–1994. [Google Scholar] [CrossRef]
- Pacella, A.; Cremisini, C.; Nardi, E.; Montereali, M.R.; Pettiti, I.; Giordani, M.; Mattioli, M.; Ballirano, P. Different erionite series species are able to fix iron into the structure: Implication for fibrous erionite toxicity. Minerals. accepted.
- Mattioli, M.; Giordani, M.; Dogan, M.; Cangiotti, M.; Avella, G.; Giorgi, R.; Dogan, A.U.; Ottaviani, M.F. Morpho-chemical characterization and surface properties of carcinogenic zeolite fibers. J. Hazard Mater. 2016, 306, 140–148. [Google Scholar] [CrossRef] [PubMed]
- Ballirano, P.; Melis, E. Thermal behaviour of β-anhydrite CaSO4 to 1,263 K. Phys. Chem. Miner. 2007, 12, 289–295. [Google Scholar] [CrossRef]
- Bruker AXS. Topas V.4.2: General Profile and Structure Analysis Software for Powder Diffraction Data; Bruker AXS: Karlsruhe, Germany, 2009. [Google Scholar]
- Katerinopoulou, A.; Balic-Zunic, T.; Lundegaard, L.F. Application of the ellipsoid modeling of the average shape of nanosized crystallites in powder diffraction. J. Appl. Crystallogr. 2012, 45, 22–27. [Google Scholar] [CrossRef]
- Cametti, G.; Pacella, A.; Mura, F.; Rossi, M.; Ballirano, P. New morphological, chemical, and structural data of woolly erionite-Na from Durkee, Oregon, U.S.A. Am. Mineral. 2013, 98, 2155–2163. [Google Scholar] [CrossRef]
- Young, R.A. Introduction to the Rietveld method. In The Rietveld Method; Young, R.A., Ed.; Oxford University Press: Oxford, UK, 1993; pp. 1–38. [Google Scholar]
- Bish, D.L. Determination of dehydration behaviour of zeolites using Rietveld refinement and high-temperature X-ray diffraction data. Geol. Soc. Am. Annu. Meet. Abstr. Programs 1989, 21, A73. [Google Scholar]
- Sheppard, R.A.; Gude, A.J., III. Chemical composition and physical properties of the related zeolites offretite and erionite. Am. Mineral. 1969, 54, 875–886. [Google Scholar]
- Ballirano, P.; Sadun, C. Thermal behavior of trehalose dihydrate (Th) and β-anhydrous trehalose (Tβ) by in-situ laboratory parallel-beam X-ray powder diffraction. Struct. Chem. 2009, 20, 815–823. [Google Scholar] [CrossRef]
- Matassa, R.; Famigliari, G.; Relucenti, M.; Battaglione, E.; Downing, C.; Pacella, A.; Cametti, G.; Ballirano, P. A Deep Look into Erionite Fibres: An Electron Microscopy Investigation of their Self-Assembly. Sci. Rep. 2015, 5, 16757. [Google Scholar] [CrossRef] [PubMed]
- Baerlocher, C.; McCusker, L.B.; Olson, D.H. Atlas of Zeolite Framework Types, 5th ed.; Elsevier: London, UK, 2001; p. 398. [Google Scholar]
- Hammonds, K.D.; Deng, H.; Heine, V.; Dove, M.T. How floppy modes give rise to adsorption sites in zeolites. Phys. Rev. Lett. 1997, 78, 3701–3704. [Google Scholar] [CrossRef]
- Hammonds, K.D.; Heine, V.; Dove, M.T. Rigid-unit modes and the quantitative determination of the flexibility possessed by zeolite frameworks. J. Phys. Chem. 1998, B102, 1759–1767. [Google Scholar] [CrossRef]
- Mon, J.; Deng, Y.; Flury, M.; Harsh, J.B. Cesium incorporation and diffusion in cancrinite, sodalite, zeolite and allophane. Microporous Mesoporous Mater. 2005, 86, 277–286. [Google Scholar] [CrossRef]
- Bloise, A.; Catalano, M.; Barrese, E.; Gualtieri, A.F.; Bursi Gandolfi, N.; Capella, S.; Belluso, E. TG/DSC study of the thermal behavior of hazardous mineral fibres. J. Therm. Anal. Calorim. 2016, 123, 2225–2239. [Google Scholar] [CrossRef]
- Pacella, A.; Cremisini, C.; Nardi, E.; Montereali, M.R.; Pettiti, I.; Ballirano, P. The mechanism of iron binding processes in erionite fibres. Sci. Rep. 2017, 7, 1319. [Google Scholar] [CrossRef] [PubMed]
- Armbruster, T. Dehydration mechanism of clinoptilolite and heulandite: Single-crystal X-ray study of Na-poor, Ca-, K-, Mg-rich clinoptiloite at 100 K. Am. Mineral. 1993, 78, 260–264. [Google Scholar]
- Ballirano, P.; Pacella, A.; Cremisini, C.; Nardi, E.; Fantauzzi, M.; Atzei, D.; Rossi, A.; Cametti, G. Fe (II) segregation at a specific crystallographic site of fibrous erionite: A first step toward the understanding of the mechanisms inducing its carcinogenicity. Microporous Mesoporous Mater. 2015, 211, 49–63. [Google Scholar] [CrossRef]
- Eborn, S.K.; Aust, A.E. Effect of iron acquisition on induction of DNA singlestrand breaks by erionite, a carcinogenic mineral fiber. Arch. Biochem. Biophys. 1995, 316, 507–514. [Google Scholar] [CrossRef] [PubMed]
| Chemical Formula | (Ca2.03Na0.73K2.52Mg0.26)[Al8.22Si27.78O71.80]·29.71H2O |
|---|---|
| R = Si/(Si + Al) | 0.772 |
| Space group | P63/mmc |
| Cell parameters at 303 K (Å) | a = 13.2505(5) |
| c = 15.0497(6) | |
| Cell volume at 303 K (Å3) | 2288.36(18) |
| Instrument | D8 Advance (Bruker AXS, Karlsruhe, Germany) |
| X-ray tube | Cu operating at 40 kV and 40 mA |
| Incident beam optics | 60 mm multilayer (Göbel) X-ray mirror |
| Sample mount | Rotating capillary (60 r/min) |
| Soller slits | 2 (2.3° incident beam; radial diffracted beam) |
| Divergence slit | 0.6 mm |
| Detector | PSD VÅNTEC-1 |
| Data range (°2θ) | 6–145 |
| Step size (°2θ) | 0.022 |
| Counting time (s) | 2 |
| wRp (%) | 2.19–3.06 |
| Rp (%) | 1.70–2.35 |
| RBragg (%) | 0.25–0.58 |
| Sample | Reference | R | (Z/r)wt | Tdehyd (K) | Tbreak (K) | |
|---|---|---|---|---|---|---|
| End | Start | End | ||||
| Erionite-K | Ballirano and Cametti [19] | 0.807 | 0.096 | 573 | 1113 | 1193 |
| Erionite-Na | Ballirano and Pacella [20] | 0.796 | 0.090 | 600 | 1063 | 1173 |
| Erionite-K(Ca) | Present work | 0.772 | 0.090 | 658 | 1053 | 1158 |
| Erionite-Ca(K) | Cruciani [22] | 0.75 | 0.10 | <673 | <1023 | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ballirano, P.; Pacella, A.; Bloise, A.; Giordani, M.; Mattioli, M. Thermal Stability of Woolly Erionite-K and Considerations about the Heat-Induced Behaviour of the Erionite Group. Minerals 2018, 8, 28. https://doi.org/10.3390/min8010028
Ballirano P, Pacella A, Bloise A, Giordani M, Mattioli M. Thermal Stability of Woolly Erionite-K and Considerations about the Heat-Induced Behaviour of the Erionite Group. Minerals. 2018; 8(1):28. https://doi.org/10.3390/min8010028
Chicago/Turabian StyleBallirano, Paolo, Alessandro Pacella, Andrea Bloise, Matteo Giordani, and Michele Mattioli. 2018. "Thermal Stability of Woolly Erionite-K and Considerations about the Heat-Induced Behaviour of the Erionite Group" Minerals 8, no. 1: 28. https://doi.org/10.3390/min8010028
APA StyleBallirano, P., Pacella, A., Bloise, A., Giordani, M., & Mattioli, M. (2018). Thermal Stability of Woolly Erionite-K and Considerations about the Heat-Induced Behaviour of the Erionite Group. Minerals, 8(1), 28. https://doi.org/10.3390/min8010028








