Applications of Clays in Nanocomposites and Ceramics
Abstract
1. Introduction
2. Polymer/Clay Nanocomposites
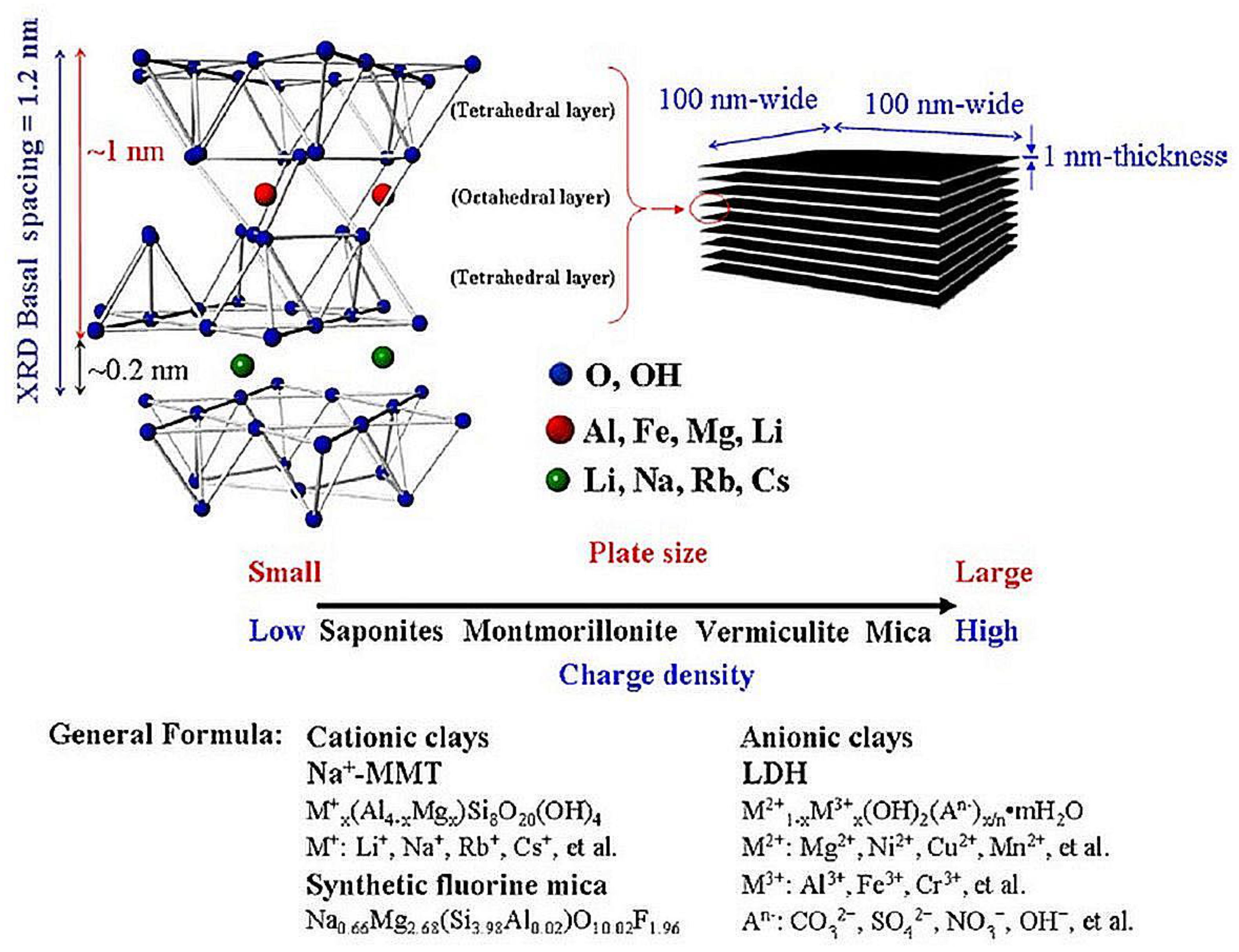
2.1. Nanofiller Sources
Montmorillonite
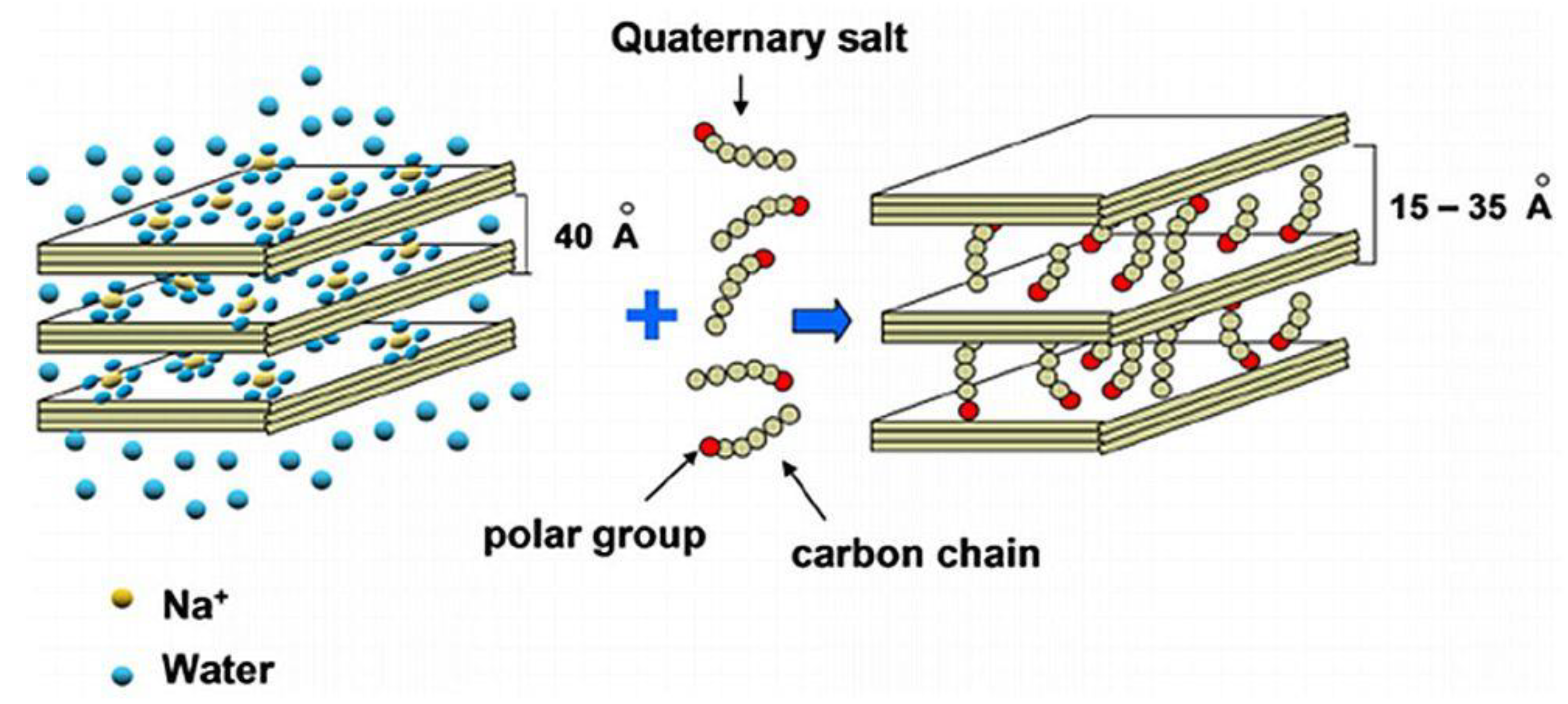
2.2. Clay Organophilization
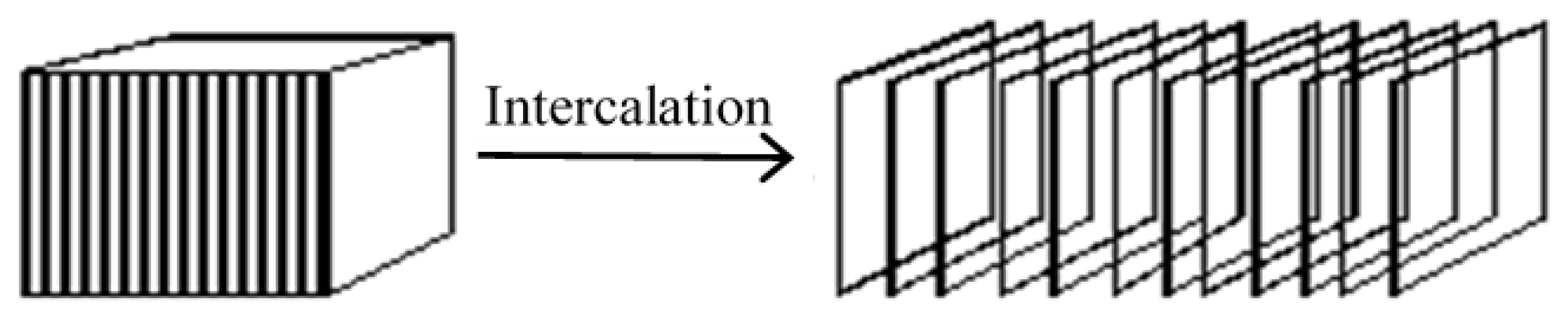
2.2.1. Ion-Exchange Intercalation
2.2.2. Ion-Dipole Intercalation
2.2.3. Grafting
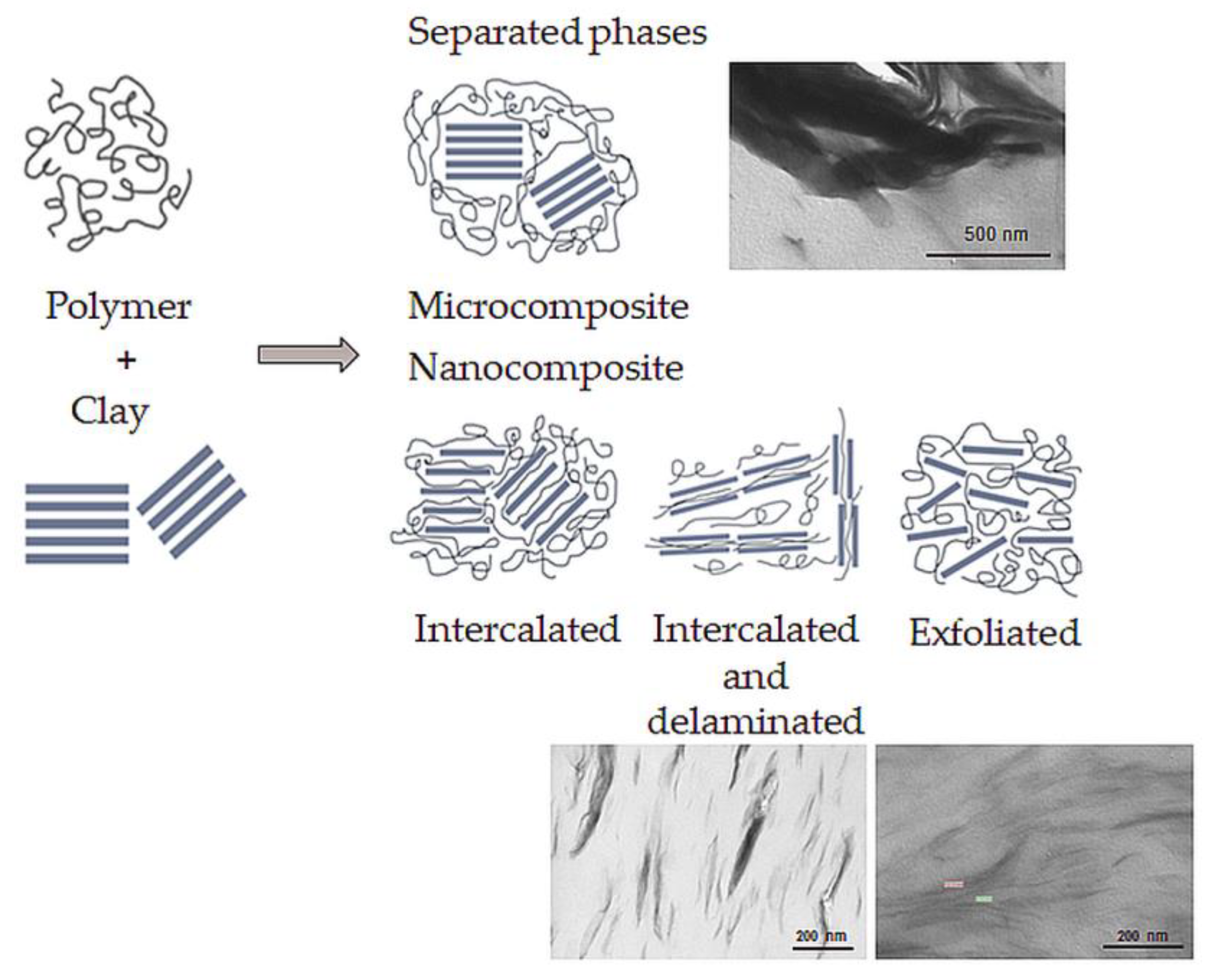
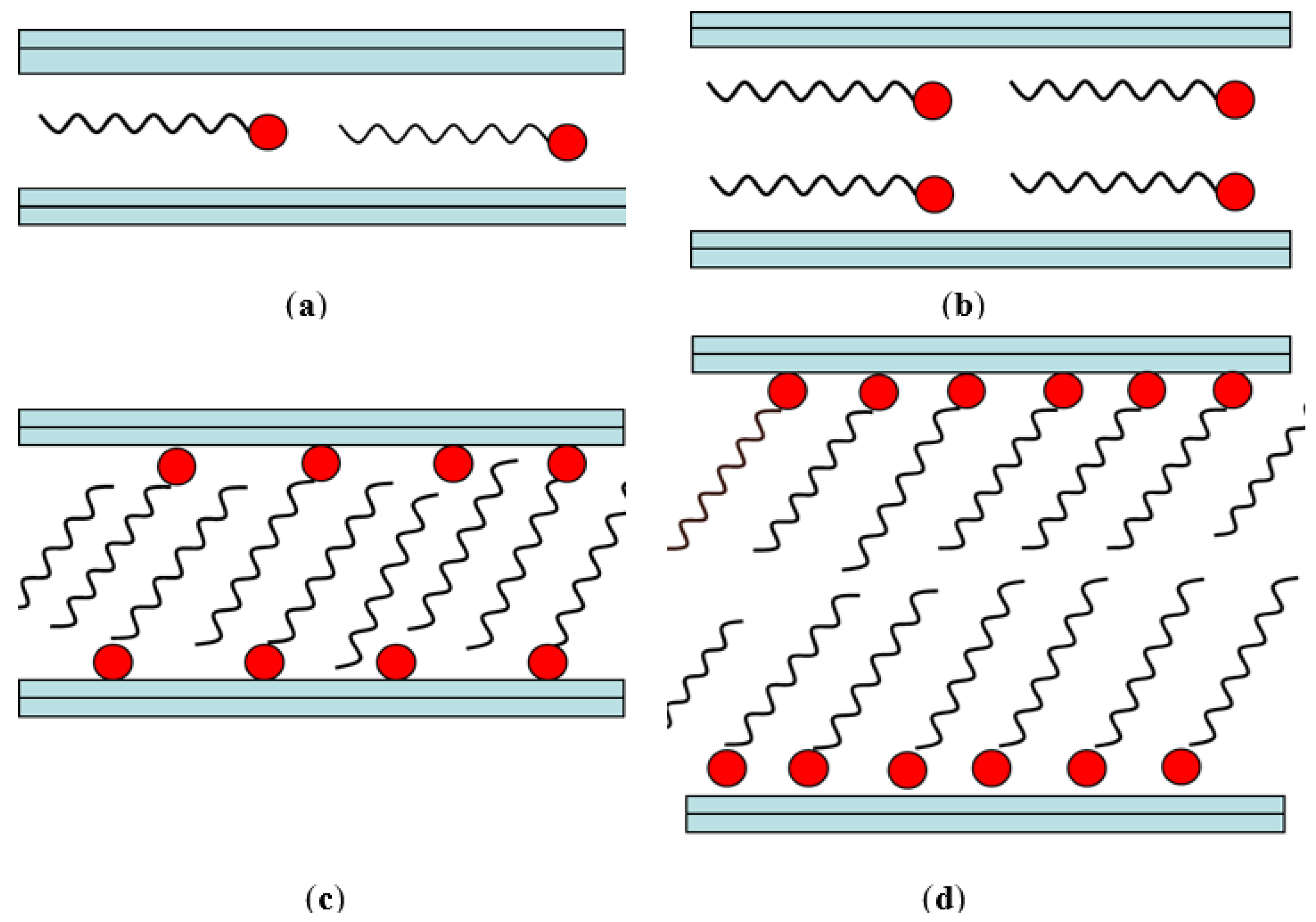
2.3. Clay Application in Nanocomposites
- Microcomposite, where the clay acts as a conventional filler. The final material belongs to traditional composite materials.
- Intercalated nanocomposite consists of a regular insertion of the polymer between the clay layers. The final material belongs to nanocomposites.
- Intercalated and partially delaminated nanocomposites, an intermediate step between intercalated and exfoliated structure. The final material belongs to nanocomposites.
- Exfoliated nanocomposite where the filler is delaminated to 1 nm-thick layers. The final material belongs to nanocomposites.
2.3.1. Polymer/Clay Nanocomposite Processing
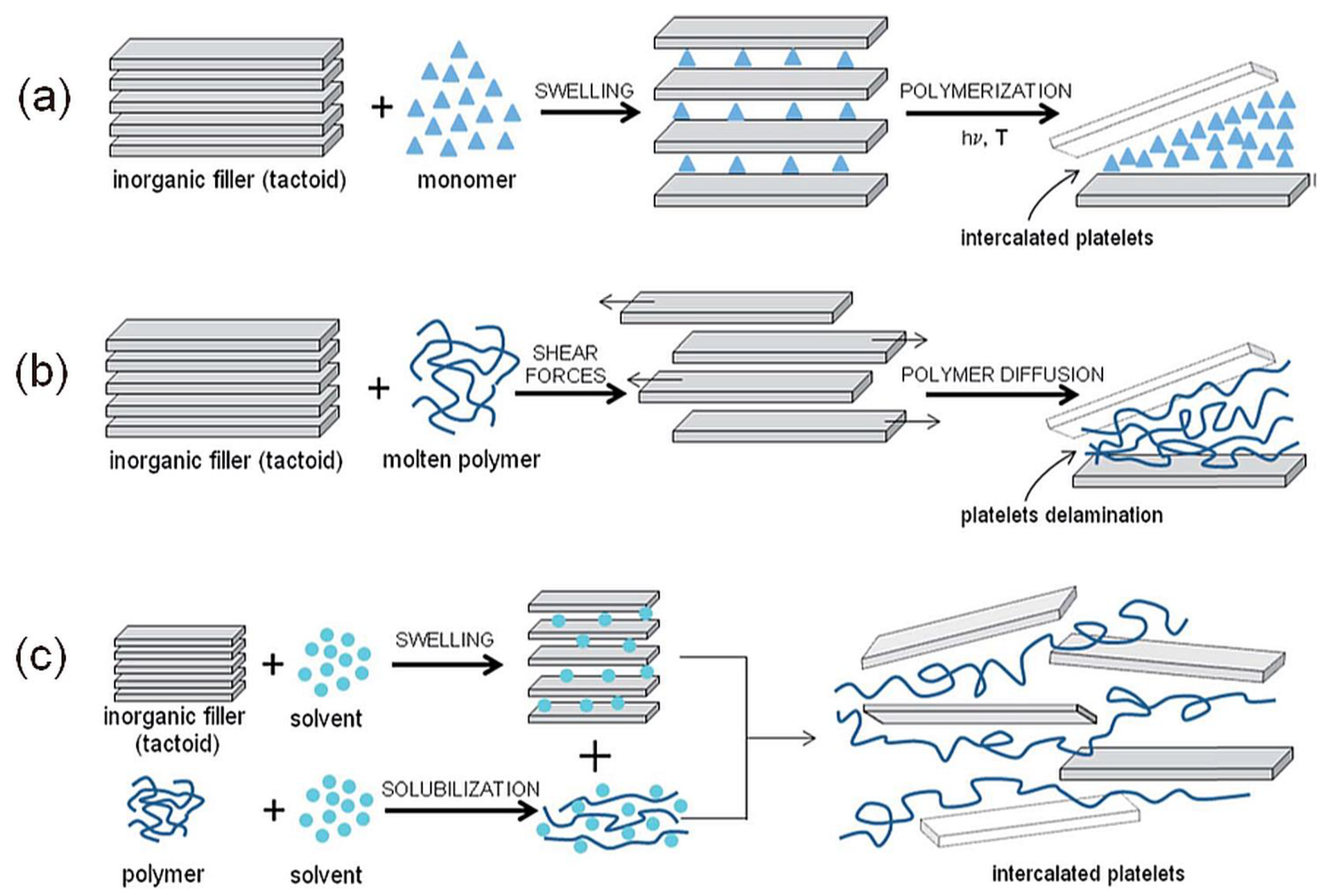
- In-situ polymerization method, intercalation of a suitable monomer followed by polymerization. The first method used to synthesize polymer/clay nanocomposites is based on polyamide 6.
- Solution method, intercalation of dissolved polymer from a solution. The drawback of this method is the requirement of a suitable solvent. It has been shown that intercalation only occurs for certain polymer/solvent or monomer/solvent pairs [77]. Nanocomposites based on high-density polyethylene [78], and polyimide [79] can be synthesized by this method.
- Melt intercalation method, mixing the clay (usually organoclay) with the polymer matrix above its softening point in either static or flow conditions. The polymer chains spread from the molten mass into the silicate galleries to form either intercalated or delaminated hybrids according to the degree of penetration [56]. This process was first reported by Vaia et al. [80] in 1993. This method is relatively easy and allows for the use of current processing equipment for nanocomposite technology. Traditional processing techniques could be used for melt intercalation, like a two-roll mill, twin-screw extruder (PA, PP, PE, and PVC), injection molding, blow molding, and thermal spraying [2].
2.3.2. Polymer Matrix

2.3.3. Polymer/Clay Nanocomposite Advantages
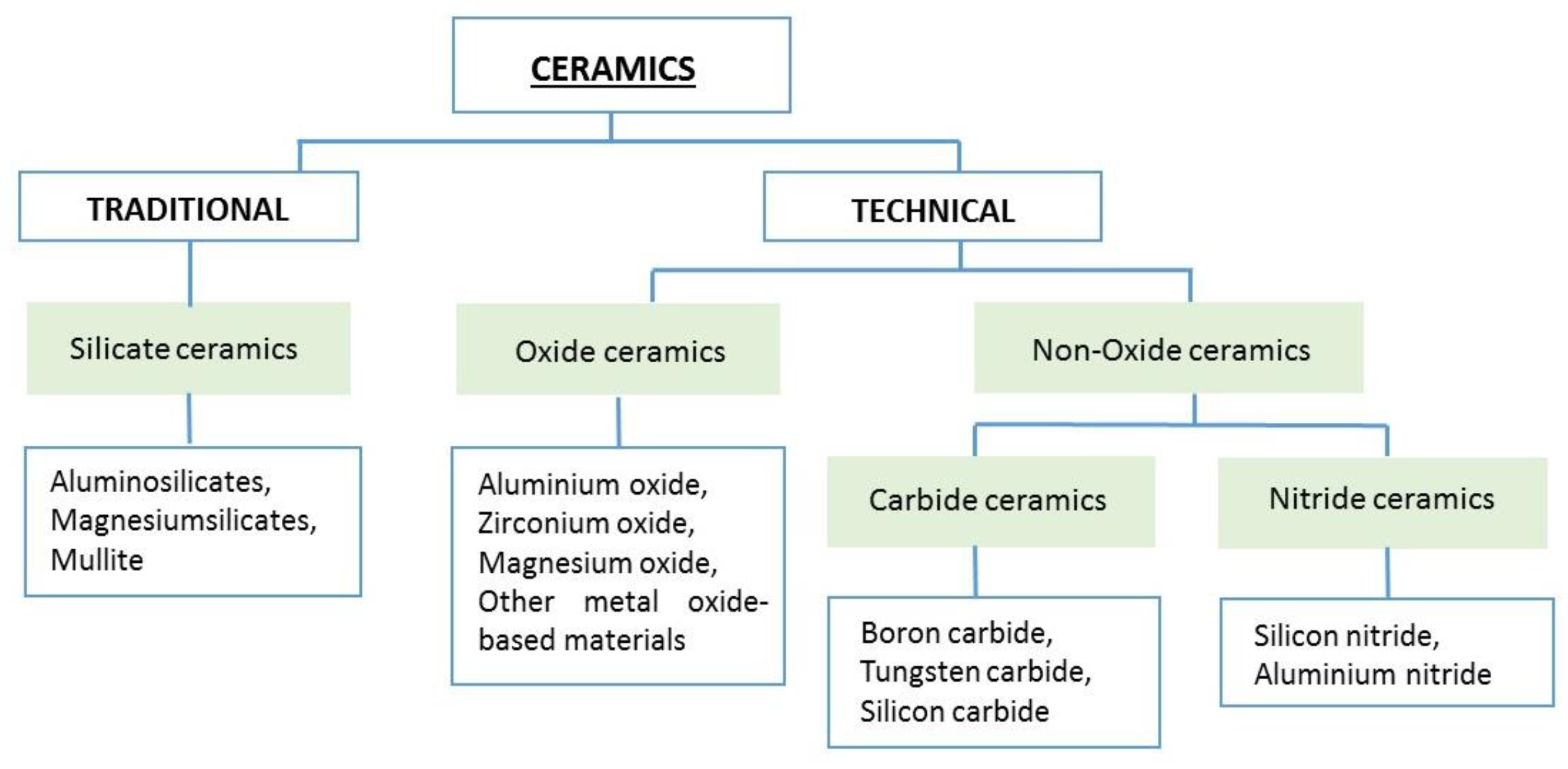
3. Ceramic Materials
3.1. Ceramics Based on Clays
Silicate Ceramics
| Pre-ceramic Mixtures | |||||
|---|---|---|---|---|---|
| Clay Minerals | Other | Type of Ceramics | Sintering Temperature (°C) | Applications | Ref. |
| Talc | - | Enstatite | 1275, 1350, 1375 | Machinable prosthesis | [171] |
| Talc, clay | BaCO3 | Steatite | 1240–1380 | - | [167] |
| Talc | BaCO3 | Steatite | 1380 | - | [170] |
| Talc, montmorillonite Talc, kaolinite Talc, montmorillonite Talc, kaolinite, Acid treated talc | Na2CO3 Na2CO3 | Steatite | 1300 | - | [165] |
| Talc, clay | BaCO3, boric acid | Steatite | 1000–1200 | High temperature electrical applications | [166] |
| Talc, kaolin | - | Steatite | 1200, 1250, 1300 | Material in electrical insulation | [163] |
| Talc, clay | Dolomite, polyacrylic acid | Steatite | 1275, 1300 | - | [172] |
| Talc | MgO | Forsterite | 1200, 1300, 1400, 1500 | Biomedical applications | [177] |
| Talc | MgCO3 | Forsterite | 1000 | - | [180] |
| Talc | MgCO3, NH4Cl | Forsterite | 1000 | Bioceramics (bone tissue engineering applications) | [181] |
| Talc | MgCO3, NH4Cl | Forsterite | 1000, 1200 | - | [173] |
| Talc | MgO | Forsterite | 1200 | - | [174] |
| Talc | MgO | Forsterite | 1200–1500 | - | [175] |
| Talc Talc Talc | MgO NH4F NH4Cl | Forsterite Forsterite Forsterite | 1000, 1200 | - | [176] |
| Talc Talc | MgCO3 MgO | Forsterite Forsterite | 1000, 1200 | - | [178] |
| Talc | calcined MgCO3 | Forsterite | 1400 | - | [182] |
| Talc Talc | MgCO3 MgO | Forsterite Forsterite | 1200, 1300 | Refractory ceramics | [179] |
| Talc | MgCO3, CaCO3 | Forsterite/diopside | 1200 | Tissue engineering | [183] |
| Talc, kaolin, Talc, kaolin Talc, kaolin, pyrophyllite Talc, pyrophyllite Talc, kaolin, Talc, pyrophyllite | Mg Al2O3 Al2O3 Al2O3 Mg, Al2O3 Mg, Al2O3 | Cordierite | 1000–1380 | - | [190] |
| Talc Talc, kaolinite | Al2O3, BaCO3 Al2O3 | Steatite Cordierite | 1320 1250 | - | [184] |
| Talc, kaolin waste | MgO | Cordierite | 950, 1050, 1150, 1250, 1350 | Refractory and insulating materials | [195] |
| Kaolinite | Mg(OH)2 | Cordierite | 1350 | Substrate material for circuit boards, thermal shock resistance ceramics | [193] |
| Talc | Diatomite, Al2O3 | Cordierite | 1300, 1350, 1400 | - | [185] |
| Talc | Fly ash, fused silica, Al2O3 | Cordierite | 1200, 1300, 1350, 1375 | - | [196] |
| Kaolin, attapulgite | MgO | Cordierite | 1200 | Thermal insulator | [202] |
| Kaolinite, talc, vermiculite | Cordierite/steatite | 1300 | - | [199] | |
| Kaolinite, talc, vermiculite, organo-vermiculite | Cordierite/steatite | 1300 | - | [200] | |
| Kaolinite, talc, vermiculites | Al2O3, Al(OH)3 | Cordierite | 1300 | - | [187] |
| Kaolinite, talc, vermiculite | Al2O3 | Cordierite | 1300 | - | [191] |
| Kaolinite, talc, vermiculite, organo-vermiculite | Cordierite/steatite | 1300 | - | [203] | |
| Talc, kaolinite, vermiculite | MgO, Al(OH)3 | Cordierite | 1300 | - | [192] |
| Talc, kaolinite, ball clay | Al2O3, silica sand | Cordierite | 1300 | Industrial manufacture of porous ceramic materials | [194] |
| Talc, kaolinite, CeO2/vermiculite | - | Cordierite/CeO2 | 1300 | Photocatalysts | [204,205] |
| Talc | Polysiloxane, Al2O3, expandable microspheres | Cordierite | 1300 | - | [198] |
| Talc, kaolinite | Al(OH)3 | Cordierite | 1260 | - | [189] |
| Kaolin, sepiolite Kaolin, talc | Quartz SiO2, feldspar | Cordierite Cordierite | 1250, 1300, 1350 | Application in foundry | [201] |
| Talc | Fly ash, Al2O3 | Cordierite | 1350 | Refractory application | [197] |
| Talc, kaolin | Feldspar, albite, andalusite, Al2O3 | Cordierite/mullite | 1340–1420 | Thermal storage materials | [207] |
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- History of Nanotechnology. Available online: https://trynano.org/about-nanotechnology/history-of-nanotechnology/ (accessed on 28 November 2023).
- Ajayan, P.M.; Schadler, L.S.; Braun, P.V. Nanocomposite Science and Technology; Wiley—WCH: Weinheim, Germany, 2003; ISBN 3-527-30359-6. [Google Scholar]
- What Is Nanotechnology? Available online: https://www.nanowerk.com/nanotechnology/introduction/introduction_to_nanotechnology_1.php (accessed on 19 November 2023).
- Nanocomposites Market, Global Indurstry Analysis, Size, Share, Growth & Forecast 2022–2031. Available online: https://growthmarketreports.com/report/nanocomposites-market-global-industryanalysis#:~:text=The%20global%20nanocomposites%20market%20size%20was%20USD%205.6,use%20of%20nanocomposites%20in%20biomedical%20applications%20and%20packaging (accessed on 29 November 2023).
- Bergaya, F.; Theng, B.K.G.; Lagaly, G. Chapter 1: General introduction: Clays, clay minerals and clay science. In Handbook of Clay Science, Developments in Clay Science; Elsevier: Amsterdam, The Netherlands, 2006. [Google Scholar]
- Harvey, C.C.; Lagaly, G. Chapter 10.1: Conventional Applications. In Handbook of Clay Science, Developments in Clay Science; Elsevier: Amsterdam, The Netherlands, 2006. [Google Scholar]
- Konta, J. Clay and man: Clay raw materialsin the service of man. Appl. Clay Sci. 1995, 10, 275–335. [Google Scholar] [CrossRef]
- Valášková, M. Clays, Clay minerals and cordierite ceramics—A review. Ceram. Silik. 2015, 59, 331–340. [Google Scholar]
- Bergaya, F.; Jaber, M.; Lambert, J.-F. Chapter 1: Clays and Clay Mineral. In Rubber-Clay Nanocomposites: Science, Technology, and Applications; Galimberti, M., Ed.; John Wiley & Sons: Hoboken, NJ, USA, 2011. [Google Scholar]
- Pospisil, M.; Capkova, P.; Merinska, D.; Malac, Z.; Simonik, J. Sructure analysis of montmorillonite intercalated with cetylpyridinium and cetyltrimethylammonium: Molecular simulations and XRD analysis. J. Colloid Interface Sci. 2001, 236, 127–131. [Google Scholar] [CrossRef]
- Murray, H.H. Overview, clay mineral applications. Appl. Clay Sci. 1991, 5, 379–395. [Google Scholar] [CrossRef]
- Glebova, A.A.; Skovorodnikova, M.S.; Pavlova, I.A.; Farafontova, E.P. Research on the Ceramicc Properties of Orenburg Oblast Clay. Glass Ceram. 2023, 79, 11–12. [Google Scholar] [CrossRef]
- Yang, X.; Yang, W.; Hu, J. Preparation of Low-Dielectric-Constant Kaolin Clay Ceramics by Chemical Cleaning Method. Front. Mater. 2021, 8, 692759. [Google Scholar] [CrossRef]
- Petronela, N. The Influence of Drying Conditions of Clay-Based Polymer Coatings on Coated Paper Properties. Coatings 2021, 11, 12. [Google Scholar] [CrossRef]
- Devisetti, S.; Lempsink, G.; Malla, P.B. Use of kaolin clay in aqueous barrier coating applications. Tappi J. 2023, 22, 685–697. [Google Scholar] [CrossRef]
- Granetto, M.; Serpella, L.; Fogliatto, S.; Re, L.; Bianco, C.; Vidotto, F.; Tosco, T. Natural clay and biopolymer-based nanopesticides to control the environ-mental spread of a soluble herbicide. Sci. Total Environ. 2022, 806 Pt 3, 151199. [Google Scholar] [CrossRef]
- Mahmoodi, A.; Dadras, A.; Jiryaei, Z.; Khorasani, M.; Shi, X.M. A Yellow Lead-Free Pavement Marking Paint Based on Hybrid Dye-Clay Nanopigment: Morphological, Thermome-chemical, and Photophysical Properties. ACS Sustain. Chem. Eng. 2021, 9, 16466–16473. [Google Scholar] [CrossRef]
- Vakhitova, L.; Kalafat, K.; Vakhitov, R.; Drizhd, V.; Taran, N.; Bessarabov, V. Nano-clays as rheology modifiers in intumescent coatings for steel building structures. Chem. Eng. J. Adv. 2023, 16, 100544. [Google Scholar] [CrossRef]
- Choi, G.; Piao, H.Y.; Eom, S.; Choy, J.H. Vectorized Clay Nanoparticles in Therapy and Diagnosis. Clay Clays Miner. 2019, 67, 25–43. [Google Scholar] [CrossRef]
- Nomicisio, C.; Ruggeri, M.; Bianchi, E.; Vigani, B.; Valentino, C.; Aguzzi, C.; Viseras, C.; Rossi, S.; Sandri, G. Natural and Synthetic Clay Minerals in the Pharmaceutical and Biomedical Fields. Pharmaceutics 2023, 15, 1368. [Google Scholar] [CrossRef] [PubMed]
- Torbert, H.A.; Harmel, R.D.; Potter, K.N.; Dozier, M. Evaluation of some phosphorus index criteria in cultivated agriculture in clay soils. J. Soil Water Conserv. 2005, 60, 21–29. [Google Scholar]
- Kubo, K.; Hirayama, T.; Fujimura, S.; Eguchi, T.; Nihei, N.; Hamamoto, S.; Takeuchi, M.; Saito, T.; Ota, T.; Shinano, T. Potassium behavior and clay mineral composition in the soil with low effectiveness of potassium application. Soil Sci. Plant Nutr. 2018, 64, 265–271. [Google Scholar] [CrossRef]
- Degrave-Lemeurs, M.; Glé, P.; de Menibus, A.H. Acoustical properties of hemp concretes for buildings thermal insulation: Application to clay and lime binders. Constr. Build. Mater. 2018, 160, 462–474. [Google Scholar] [CrossRef]
- Zhou, S.Q.; Niu, Y.Q.; Liu, J.H.; Chen, X.X.; Li, C.S.; Gates, W.P.; Zhou, C.H. Functional Montmorillonite/Polymer Coatings. Clays Clay Miner. 2022, 70, 209–232. [Google Scholar] [CrossRef]
- Moradihamedani, P. Recent development in polymer/montmorillonite clay mixed matrix membranes for gas separation: A short review. Polym. Bull. 2023, 80, 4663–4687. [Google Scholar] [CrossRef]
- Zhu, Y.; Iroh, J.O.; Rajagopolan, R.; Aykanat, A.; Vaia, R. Optimizing the Synthesis and Thermal Properties of Conducting Polymer-Montmorillonite Clay Nanocomposites. Energies 2022, 15, 1291. [Google Scholar] [CrossRef]
- Viseras, C.; Sánchez-Espejo, R.; Palumbo, R.; Liccardi, N.; García-Villén, F.; Borrego-Sánchez, A.; Massaro, M.; Riela, S.; López-Galindo, A. Clays in Cosmetics and Personal-Care Products. Clays Clay Miner. 2022, 69, 561–575. [Google Scholar] [CrossRef]
- Suzuki, A.; Shikinaka, K.; Ishii, R.; Yoshida, H.; Ebina, T.; Ishida, T.; Nge, T.T.; Yamada, T. Heat-resistant insulation film containing clay and wood components. Appl. Clay Sci. 2019, 180, 105189. [Google Scholar] [CrossRef]
- Singh, N.B. Clays and Clay Minerals in the Construction Industry. Minerals 2022, 12, 301. [Google Scholar] [CrossRef]
- Das, P.; Manna, S.; Behera, A.K.; Shee, M.; Basak, P.; Sharma, A.K. Current synthesis and characterization techniques for clay-based polymer nano-composites and its biomedical applications: A review. Environ. Res. 2022, 212, 113534. [Google Scholar] [CrossRef]
- Barry Carter, C.; Grant Norton, M. Ceramic Materials: Science and Engineering; Springer: Berlin/Heidelberg, Germany, 2007. [Google Scholar]
- Kagonbé, B.P.; Tsozué, D.; Nzeukou, A.N.; Ngos III, S. Mineralogical, physico-chemical and ceramic properties of clay materials from Sekandé and Gashiga (North, Cameroon) and their suitability in earthenware production. Heliyon 2021, 7, e07608. [Google Scholar] [CrossRef]
- Global Nanoceramics Market—Industry Trends and Forecast to 2030. Available online: https://www.databridgemarketresearch.com/reports/global-nanoceramics-market (accessed on 29 November 2023).
- Krishnamoorti, R.; Vaia, A.R. Polymer Nanocomposites: Synthesis, Characterization, and Modeling; American Chemical Society: Washington, DC, USA, 2002; ISBN 0-8412-3768-9. [Google Scholar]
- Lai, Y.-H.; Chiu, C.-W.; Chen, J.-G.; Wang, C.-C.; Ho, K.-C. Enhancing the performance of dye-sensitized solar cells by incorporating nanosilicate platelets in gel electrolyte. Sol. Energy Mater. Sol. Cells 2009, 93, 1860–1864. [Google Scholar] [CrossRef]
- Pethrick Richard, A. Polymer Science and Technology for Scientists and Engineers; Whittles Publishing: Caithnes, UK, 2010; p. 354. ISBN 978-1904445-40-1. [Google Scholar]
- Bee, S.-L.; Abdullah, M.A.A.; Lee Bee, S.-T.; Sin, T.; Rahmat, A.R. Polymer nanocomposites based on silylated-montmorillonite: A review. Prog. Polym. Sci. 2018, 85, 57–82. [Google Scholar] [CrossRef]
- Sapalidis, A.A.; Katsaros, F.K.; Kanellopoulos, N.K. PVA/montmorillonite nanocomposites: Development and properties. In Nanocomposites and Polymers with Analytical Methods; Cappoletti, J., Ed.; InTech: Rijeka, Croatia, 2011; pp. 29–50. [Google Scholar]
- Zulfiqar, S.; Kausar, A.; Rizwan, M.; Sarwar, M.I. Probing the role of surface treated montmorillonite on the properties of semiaromatic polyamide/clay nanocomposites. Appl. Surf. Sci. 2008, 255, 2080–2086. [Google Scholar] [CrossRef]
- Bertuoli, P.T.; Piazza, D.; Scienza, L.C.; Zattera, A.J. Preparation and characterization of montmorillonite modified with 3-aminopropyltriethoxysilane. Appl. Clay Sci. 2014, 87, 46–51. [Google Scholar] [CrossRef]
- Kádár, F.; Százdi, L.; Fekete, E.; Pukánszky, B. Surface characteristics of layered silicates: Influence on the properties of clay/polymer nanocomposites. Langmuir 2006, 22, 7848–7854. [Google Scholar] [CrossRef]
- Fudala, A.; Palinko, I.; Kiricsi, I. Preparation and characterization of hybrid organic-inorganic composite materials using the amphoteric property of amino acids: Amino acid intercalated layered double hydroxide and montmorillonite. Inorg. Chem. 1999, 38, 4653–4658. [Google Scholar] [CrossRef]
- Zhu, J.; Zhanga, P.; Qinga, Y.; Wena, K.; Sua, X.; Ma, L.; Jingming Wei, J.; Liu, H.; He, H.; Xi, Y. Novel intercalation mechanism of zwitterionic surfactant modified montmorillonites. Appl. Clay Sci. 2017, 141, 265–271. [Google Scholar] [CrossRef]
- Lagaly, G. Interaction of Alkylamines with different types of layered compounds. Solid State Ion. 1986, 22, 43–51. [Google Scholar] [CrossRef]
- Vaia, R.A.; Teukolsky, R.K.; Giannelis, E.P. Interlayer Structure and Molecular Environment of Alkylammonium Layered Silicates. Chem. Mater. 1994, 6, 1017–1022. [Google Scholar] [CrossRef]
- Rappé, A.K.; Casewit, C.J.; Colwell, K.S.; Goddard, W.A.; Skiff, W.M. UFF, a Full Periodic-Table Force-field for Molecular Mechanics and Molecular-Dynamics simulations. J. Am. Chem. Soc. 1992, 114, 10024–10035. [Google Scholar] [CrossRef]
- Berendsen, H.J.C.; Postma, J.P.M.; van Gunsteren, W.F.; DiNola, A.; Haak, J.R. Molecular-Dynamics with Coupling to an External Bath. J. Chem. Phys. 1984, 81, 3684–3690. [Google Scholar] [CrossRef]
- Kalendova, A.; Pospisil, M.; Kovarova, L.; Merinska, D.; Valaskova, M.; Vlkova, H.; Simonik, J.; Capkova, P. Influence of chain length on inter-calation process of polyvinylchloride/clay nanocomposites based on alkyl-amine. Plast. Rubber Compos. 2004, 33, 279–286. [Google Scholar] [CrossRef]
- Cavalcanti, J.V.F.L.; Abreu, C.; Carvalho, M.N.; Motta, M. Removal of Effluent from Petrochemical Wastewater by Adsorption Using Organoclay. In Petrochemicals; IntechOpen: London, United Kingdom, 2012; pp. 277–294. [Google Scholar] [CrossRef]
- Meier, L.P.; Nuesch, R. The Lower Cation Exchange Capacity Limit of Montmorillonite. J. Colloid Interface Sci. 1999, 217, 77–85. [Google Scholar] [CrossRef]
- Rappé, A.K.; Goddard, W.A. Charge Equilibration for Molecular-Dynamics Simulations. J. Phys. Chem. 1991, 95, 3358–3363. [Google Scholar] [CrossRef]
- Karasawa, A.; Goddard, W.A. Acceleration of convergence for Lattice Sums. J. Phys. Chem. 1989, 93, 7320–7327. [Google Scholar] [CrossRef]
- Dabbaghianamiria, D.M.; Beall, G.W. Self-assembling nanostructured intercalates via ion–dipole bonding. Dalton Trans. 2018, 47, 3178–3184. [Google Scholar] [CrossRef]
- Merinska, D.; Chmielova, M.; Kalendova, A.; Weiss, Z.; Capkova, P.; Simonik, J. Montmorillonite co-intercalated with octadecylamine and stearic acid by low temperature melting and its influence on PP nanocomposites. Int. Polym. Process. 2003, 18, 133–137. [Google Scholar] [CrossRef]
- Pospisil, M.; Kalendova, A.; Capkova, P.; Simonik, J.; Valaskova, M. Structure analysis of intercalated layer silicates: Combination of molecular simulations and experiment. J. Colloid Interface Sci. 2004, 277, 154–161. [Google Scholar] [CrossRef]
- Bergaya, F.; Jaber, M.; Lambert, J.F. Organophilic clay minerals. In Rubber-Clay Nanocomposites: Science, Technology, and Applications; Galimberti, M., Ed.; John Wiley & Sons: New York, NY, USA, 2011; pp. 45–86. [Google Scholar]
- Shen, W.; He, H.; Zhu, J.; Yuan, P.; Frost, R.L. Grafting of montmorillonite with different functional silanes via two different reaction systems. J. Colloid Interface Sci. 2007, 313, 268–273. [Google Scholar] [CrossRef]
- Theng, B.K.G. Formation and Properties of Clay-Polymer Complexes, 2nd ed.; Elsevier Ltd.: Amsterdam, The Netherlands, 2012; pp. 1–511. ISBN 9780444533548. [Google Scholar]
- Albdiry, M.T.; Yousif, B.F.; Ku, H.; Lau, K.T. A critical review on the manufacturing processes in relation to the properties of nanoclay/polymer composites. J. Compos. Mater. 2013, 47, 1093–1115. [Google Scholar] [CrossRef]
- Buruga, K.; Song, H.; Shang, J.; Bolan, N.; Kim, K.-H. A review on functional polymer-clay based nanocomposite membranes for treatment of water. J. Hazard. Mater. 2019, 379, 120584. [Google Scholar] [CrossRef]
- Zhu, T.T.; Zhou, C.H.; Kabwe, F.B.; Wu, Q.Q.; Li, C.S.; Zhang, J.R. Exfoliation of montmorillonite and related properties of clay/polymer nanocomposites. Appl. Clay Sci. 2019, 169, 48–66. [Google Scholar] [CrossRef]
- Young, R.J.; Kinloch, I.A.; Gong, L.; Novoselov, K.S. The mechanics of graphene nanocomposites: A review. Compos. Sci. Technol. 2012, 72, 1459–1476. [Google Scholar] [CrossRef]
- Du, J.H.; Cheng, H.M. The fabrication, properties, and uses of graphene/polymer composites. Macromol. Chem. Phys. 2012, 213, 1060–1077. [Google Scholar] [CrossRef]
- Li, X.Q.; Wang, C.Y.; Cao, Y.; Wang, G.X. Functional MXene materials: Progress of their applications. Chem. Asian J. 2018, 13, 2742–2757. [Google Scholar] [CrossRef]
- Tu, S.B.; Jiang, Q.; Zhang, X.X.; Alshareef, H.N. Large dielectric constant enhancement in MXene percolative polymer composites. ACS Nano 2018, 12, 3369–3377. [Google Scholar] [CrossRef]
- Calvert, P. Nanotube composites—A recipe for strength. Nature 1999, 399, 210–211. [Google Scholar] [CrossRef]
- Liu, M.X.; Jia, Z.X.; Jia, D.M.; Zhou, C.R. Recent advance in research on halloysite nanotubes-polymer nanocomposite. Prog. Polym. Sci. 2014, 39, 1498–1525. [Google Scholar] [CrossRef]
- Papoulis, D. Halloysite based nanocomposites and photocatalysis: A Review. Appl. Clay Sci. 2019, 168, 164–174. [Google Scholar] [CrossRef]
- Albdiry, M.T.; Yousif, B.F. Toughening of brittle polyester with functionalized halloysite nanocomposites. Compos. Part B Eng. 2019, 160, 94–109. [Google Scholar] [CrossRef]
- Gao, D.; Chang, R.; Lyu, B.; Ma, J. Growth from spherical to rod-like SiO2: Impact on microstructure and performance of nanocomposite. J. Alloys Compd. 2019, 810, 151814. [Google Scholar] [CrossRef]
- Mark, J.E. Ceramic-reinforced polymers and polymer-modified ceramics. Polym. Eng. Sci. 1996, 36, 2905–2920. [Google Scholar] [CrossRef]
- Herron, N.; Thorn, D.L. Nanoparticles: Uses and relationships to molecular cluster compounds. Adv. Mater. 1998, 10, 1173–1184. [Google Scholar] [CrossRef]
- Huang, P.; Shi, H.Q.; Fu, S.Y.; Xiao, H.M.; Hu, N.; Li, Y.Q. Greatly decreased redshift and largely enhanced refractive index of mono-dispersed ZnO-QD/silicone nanocomposites. J. Mater. Chem. 2016, 4, 8663–8669. [Google Scholar] [CrossRef]
- Fu, S.; Sun, Z.; Huang, P.; Li, Y.; Hu, N. Some basic aspects of polymer nanocomposites: A critical review. Nano Mater. Sci. 2019, 1, 2–30. [Google Scholar] [CrossRef]
- Information Resources Management Association. Materials Science and Engineering—Concepts, Methodologies, Tools, and Application; IGI Global: Hershey, PA, USA, 2017; Volume 1, ISBN 9781522517986. [Google Scholar]
- Unalan, I.U.; Cerri, G.; Marcuzzo, E.; Cozzolino, C.A.; Farris, S. Nanocomposite films and coatings using inorganic nanobuilding blocks (NBB): Current applications and future opportunities in the food packaging sector. RSC Adv. 2014, 4, 29393–29428. [Google Scholar] [CrossRef]
- Okamota, M. Recent Advances in Polymer/Layered Silicate Nanocomposites: An Overview from Science to Technology. Mater. Sci. Technol. 2006, 22, 756–779. [Google Scholar] [CrossRef]
- Dadfar, S.M.R.; Ramazani, S.A.A.; Dadfar, S.M.A. Investigation of Oxygen Barrier Properties of Organoclay/HDPE/EVA Nanocomposite Films Prepared Using a Two-Step Solution Method. Polym. Compos. 2009, 30, 812–819. [Google Scholar] [CrossRef]
- Yu, Y.-Y.; Chiu, C.-T.; Chueh, C.-C. Solution-Processable, Transparent Polyimide for High-Performance High-k Nanocomposite: Synthesis, Characterization, and Dielectric Applications in Transistors. Asian J. Org. Chem. 2018, 7, 2263–3370. [Google Scholar] [CrossRef]
- Vaia, R.A.; Ishii, H.; Giannelis, E.P. Synthesis and Properties of 2-Dimensional Nanostructures by Direct Intercalation of Polymer Melts in Layered Silicates. Chem. Mater. 1993, 5, 1694–1696. [Google Scholar] [CrossRef]
- Tiwari, S.K.; Sarang Pande, S.; Bobade, S.M.; Kumar, S. A Targeted Functional Value Based Nanoclay/PA12 Composite, Material Development for Selective Laser Sintering Process. Procedia Manuf. 2017, 21, 630–637. [Google Scholar] [CrossRef]
- Hasani, Z.; Youssefi, M.; Borhani, S.; Mallakpour, S. Structure and properties of nylon-6/amino acid modified nanoclay composite fibers. J. Text. Inst. 2019, 110, 1336–1342. [Google Scholar] [CrossRef]
- Liu, X.C.; Liu, Y.J.; Shi, P.; Lai, D.W.; Yi, Z.W.; Mao, L.; Yang, J.; Zheng, W. Thermal, Rheological and Mechanical Properties of PA6-66 Nanocomposites Co-Incorporated with Montmorillonite and Nanosilica. Nanosci. Nanotechnol. Lett. 2018, 10, 177–184. [Google Scholar] [CrossRef]
- Zhang, Q.; Li, D.; Lai, D.; You, Y.; Ou, B. Preparation, microstructure, mechanical, and thermal properties of in situ polymerized polyimide/organically modified sericite mica composites. Polym. Compos. 2016, 37, 2243–2251. [Google Scholar] [CrossRef]
- Kwon, K.; Chang, J.-H. Comparison of the properties of polyimide nanocomposites containing three different nanofillers: Organoclay, functionalized graphene, and organoclay/functionalized graphene complex. J. Compos. Mater. 2015, 49, 3031–3044. [Google Scholar] [CrossRef]
- Dabrowski, F.; Bourbigot, S.; Delobel, R.; Le Bras, M. Kinetic modelling of the thermal degradation of polyamide-6 nanocomposite. Eur. Polym. J. 2000, 36, 273–284. [Google Scholar] [CrossRef]
- Utracki, L.A.; Lyngaae-Jorgensen, J. Dynamic melt flow of nanocomposites based on poly-epsilon-caprolactam. Rheol. Acta 2002, 41, 394–407. [Google Scholar] [CrossRef]
- Bourbigot, S.; Devaux, E.; Flambard, X. Flammability of polyamide-6/clay hybrid nanocomposite textiles. Polym. Degrad. Stab. 2002, 75, 397–402. [Google Scholar] [CrossRef]
- Utracki, L.A. Equations of State for Polyamide-6 and Its Nanocomposites. II. Effects of Clay. J. Polym. Sci. Part B Polym. Phys. 2009, 47, 966–980. [Google Scholar] [CrossRef]
- Utracki, L.A. Equations of State for Polyamide-6 and Its Nanocomposites. 1. Fundamentals and the Matrix. J. Polym. Sci. Part B-Polym. Phys. 2009, 47, 299–313. [Google Scholar] [CrossRef]
- dos Santos Filho, E.A.; de Medeiros, K.M.; Araujo, E.M.; Ferreira, R.D.B.; Oliveira, S.S.L.; Medeiros, V.D. Membranes of polyamide 6/clay/salt for water/oil separation. Mater. Res. Express 2019, 6, 105313. [Google Scholar] [CrossRef]
- Kojima, Y.; Usuki, A.; Kawasumi, M.; Okada, A.; Kurauchi, T.; Kamigaito, O. Synthesis of Nylon-6-Clay Hybrid by Montmorillonite Intercalated with Epsilon-Caprolactam. J. Polym. Sci. Part A Polym. Chem. 1993, 31, 983–986. [Google Scholar] [CrossRef]
- Kojima, Y.; Usuki, A.; Kawasumi, M.; Okada, A.; Fukushima, Y.; Kurauchi, T.; Kamigaito, O. Mechanical-Properties of Nylon 6-Clay Hybrid. J. Mater. Res. 1993, 8, 1185–1189. [Google Scholar] [CrossRef]
- Kojima, Y.; Usuki, A.; Kawasumi, M.; Okada, A.; Kurauchi, T.; Kamigaito, O. One-Pot Synthesis of Nylon-6 Clay Hybrid. J. Polym. Sci. Part A Polym. Chem. 1993, 31, 1755–1758. [Google Scholar] [CrossRef]
- Usuki, A.; Kojima, Y.; Kawasumi, M.; Okada, A.; Fukushima, Y.; Kurauchi, T.; Kamigaito, O. Synthesis of Nylon 6-Clay Hybrid. J. Mater. Res. 1993, 8, 1179–1184. [Google Scholar] [CrossRef]
- Kojima, Y.; Usuki, A.; Kawasumi, M.; Okada, A.; Kurauchi, T.; Kamigaito, O.; Kaji, K. Fine-Structure of Nylon-6-Clay Hybrid. J. Polym. Sci. Part B Polym. Phys. 1994, 32, 625–630. [Google Scholar] [CrossRef]
- Azuma, H.; Takeichi, A.; Noda, S. Structure-Analysis of Nylon6-Clay Hybrid by Spectral Reflectance of Laser-Plasma Soft X Rays. Jpn. J. Appl. Phys. Part 1–Regul. Pap. Short Notes Rev. Pap. 1993, 32, 5558–5563. [Google Scholar] [CrossRef]
- Fukusima, Y. Development of clay minerals/organic polymer nano-composit materials. Nippon. Kagaku Kaishi 2000, 9, 605–611. [Google Scholar] [CrossRef]
- Wu, T.M.; Liao, C.S. Crystalline transitions in nylon/clay nanocomposites. In Proceedings of the 58th Annual Technical Conference of the Society-of-Plastics-Engineers ANTEC 2000—Proceedings, Orlando, FL, USA, 2–4 April 2000; Volume I–III, pp. 1514–1517. [Google Scholar]
- Devaux, E.; Bourbigot, S.; El Achari, A. Crystallization behavior of PA-6 clay nanocomposite hybrid. J. Appl. Polym. Sci. 2002, 86, 2416–2423. [Google Scholar] [CrossRef]
- Tanaka, G.; Goettler, L.A. Predicting the binding energy for nylon 6,6/clay nanocomposites by molecular modeling. Polymer 2002, 43, 541–553. [Google Scholar] [CrossRef]
- Silva, T.F.; Soares, B.G.; Ferreira, S.C.; Livi, S. Silylated montmorillonite as nanofillers for plasticized PVC nanocomposites: Effect of the plasticizer. Appl. Clay Sci. 2014, 99, 93–99. [Google Scholar] [CrossRef]
- Thabet, A.; Ebnalwaled, A.A. Improvement of surface energy properties of PVC nanocomposites for enhancing electrical applications. Measurement 2017, 110, 78–83. [Google Scholar] [CrossRef]
- Trlica, J.; Kalendova, A.; Malac, Z.; Simonik, J.; Pospisil, L. PVC/Clay Nanocomposites. In Proceedings of the 59th Annual Technical Conference ANTEC 2001—Proceeding, Dallas, TX, USA, 6–10 May 2001; pp. 2162–2165. [Google Scholar]
- Kalendova, A.; Kovarova, L.; Malac, J.; Vaculik, J.; Malac, Z.; Simonik, J.; Hrncirik, J. Modified Clay in Polyvinylchloride (PVC) Matrix. In Proceedings of the 60th Annual Technical Conference ANTEC 2002—Proceeding, San Francisco, CA, USA, 5–9 May 2002; pp. 2250–2254. [Google Scholar]
- Pospíšil, M.; Čapková, P.; Weiss, Z.; Maláč, Z.; Šimoník, J. Intercalation of Octadecylamine into Montmorillonite: Molecular Simulations and XRD Analysis. J. Colloid Interface Sci. 2002, 245, 126–132. [Google Scholar] [CrossRef]
- Lagaly, G.; Weiss, A. Arrangement and orientation of tensides on silicate surfaces II. Paraffin-like structure in alkylammonium layer silicate with a high layer charge (mica). Kolloid Z. Z. Fur Polym. 1970, 237, 364–368. [Google Scholar] [CrossRef]
- Lagaly, G.; Weiss, A. Arrangement and orientation of cationic tensides on silicate surfaces IV. Arrangement of alkylammonium ions in low-charged silicates of films. Kolloid Z. Z. Fur Polym. 1971, 243, 48. [Google Scholar] [CrossRef]
- Lagaly, G.; Weiss, A. Layer intercalation coumpouns as models for structure and structural conversions of monomolecular and bimolecular films of long-chain compounds II. Phase changes in alkylammonium layer silicate-alkanol complexes. Kolloid Z. Z. Fur Polym. 1971, 248, 979. [Google Scholar] [CrossRef]
- Arvanitoyannis, I.; Nikolaou, E.; Yamamoto, N. Novel biodegradable copolyamides based on adipic acid, bis (p-aminocyclohexyl) methane and several alpha-amino-acids—Synthesis, characterization and study od their degradability for food packaging applications. Polymer 1994, 35, 4678–4689. [Google Scholar] [CrossRef]
- Arjmandi, R.; Hassan, A.; Haafiz, M.K.M.; Zakaria, Z.; Islam, M.S. Effect of hydrolysed cellulose nanowhiskers on properties of montmorillonite/polylactic acid nanocomposites. Int. J. Biol. Macromol. 2016, 82, 998–1010. [Google Scholar] [CrossRef]
- Lima, E.M.B.; Lima, A.M.; Silva, M.; Minguita, A.P.S.; Paula, A.; dos Santos, N.R.R.; Pereira, I.C.S.; Neves, T.T.M.; Goncalves, L.F.D.; Moreira, A.P.D.; et al. Poly(lactic acid) biocomposites with mango waste and organo-montmorillonite for packaging. J. Appl. Polym. Sci. 2019, 136, 47512. [Google Scholar] [CrossRef]
- Kalendova, A.; Smotek, J.; Stloukal, P.; Kracalik, K.M.; Slouf, M.; Laske, S. Transport Properties of PLA/Clay Nanocomposites. Polym. Eng. Sci. 2019, 59, 2498–2501. [Google Scholar] [CrossRef]
- Fayyazbakhsh, A.; Koutny, M.; Kalendova, A.; Sasinkova, D.; Julinova, M.; Kadleckova, M. Selected Simple Natural Antimicrobial Terpenoids as Additives to Control Biodegradation of Polyhydroxy Butyrate. Int. J. Mol. Sci. 2022, 23, 14079. [Google Scholar] [CrossRef]
- Julinova, M.; Šasinkova, D.; Minarik, A.; Kaszonyiova, M.; Kalendova, A.; Kadleckova, M.; Fayyazbakhsh, A.; Koutny, M. Comprehensive Biodegradation Analysis of Chemically Modified Poly(3-hydroxybutyrate) Materials with Different Crystal Structures. Biomacromolecules 2023, 24, 4939–4957. [Google Scholar] [CrossRef]
- Therias, S.; Murariu, M.; Dubois, P. Bionanocomposites based on PLA and halloysite nanotubes: From key properties to photooxidative degradation. Polym. Degrad. Stab. 2017, 145, 60–69. [Google Scholar] [CrossRef]
- Ghorpade, V.M.; Gennadios, A.; Hanna, M.A. Laboratory composting of extruded poly(lactic acid) sheets. Bioresour. Technol. 2001, 76, 57–61. [Google Scholar] [CrossRef]
- Harris, A.M.; Lee, E.C. Heat and humidity performance of injection molded PLA for durable applications. J. Appl. Polym. Sci. 2010, 115, 1380–1389. [Google Scholar] [CrossRef]
- Prakalathan, K.; Mohanty, S.; Nayak, S.K. Polylactide/modified layered silicates nanocomposites: A critical analysis of morphological, mechanical and thermalproperties. J. Reinf. Plast. Compos. 2012, 31, 1300–1310. [Google Scholar] [CrossRef]
- Darie, R.N.; Paslaru, E.; Sdrobis, A.; Pricope, G.M.; Hitruc, G.E.; Poiatǎ, A.; Baklavaridis, A.; Vasile, C. Effect of nanoclay hydrophilicity on the poly(lactic acid)/clay nanocomposites properties. Ind. Eng. Chem. Res. 2014, 53, 7877–7890. [Google Scholar] [CrossRef]
- Murariu, M.; Dubois, P. PLA composites: From production to properties. Adv. Drug Deliv. Rev. 2016, 107, 17–46. [Google Scholar] [CrossRef]
- Connolly, M.; Zhang, Y.; Brown, D.M.; Ortuno, N.; Jorda-Beneyto, M.; Stone, V.; Fernandes, T.F.; Johnston, H.J. Novel polylactic acid (PLA)-organoclay nanocomposite bio-packaging for the cosmetic industry; migration studies and in vitro assessment of the dermal toxicity of migration extracts. Polym. Degrad. Stab. 2019, 168, 108938. [Google Scholar] [CrossRef]
- Moldovan, A.; Cuc, S.; Prodan, D.; Rusu, M.; Popa, D.; Taut, A.C.; Petean, I.; Bombos, D.D.R.; Nemes, O. Development and Characterization of Polylactic Acid (PLA)-Based Nanocomposites Used for Food Packaging. Polymers 2023, 15, 2855. [Google Scholar] [CrossRef]
- Choudalakis, G.; Gotsis, A. Permeability of polymer/clay nanocomposites: A review. Eur. Polym. J. 2009, 45, 967–984. [Google Scholar] [CrossRef]
- Lu, C.; Mai, Y.W. Influence of aspect ratio on barrier properties of polymer-clay nanocomposites. Phys. Rev. Lett. 2005, 95, 088303. [Google Scholar] [CrossRef]
- Lan, T.; Kaviratna, P.D.; Pinnavaia, T.J. Mechanism of clay tactorid exfoliation in epoxy-clay nanocomposites. Chem. Mater. 1995, 7, 2144–2150. [Google Scholar] [CrossRef]
- Chan, Y.-N.; Juang, T.-Y.; Liao, Y.-L.; Dai, S.A.; Lin, J.-J. Preparation of clay/epoxy nanocomposites by layered-double-hydroxide initiated self-polymerization. Polymer 2008, 49, 4796–4801. [Google Scholar] [CrossRef]
- Lan, T.; Kaviratna, P.D.; Pinnavaia, T.J. Epoxy self-polymerization in smectite clays. J. Phys. Chem. Solids 1996, 57, 1005–1010. [Google Scholar] [CrossRef]
- Wang, Z.; Lan, T.; Pinnavaia, T.J. Hybrid organic-inorganic nanocomposites formed from an epoxy polymer and a layered silicic acid (magadiite). Chem. Mater. 1996, 8, 2200. [Google Scholar] [CrossRef]
- Wang, Z.; Pinnavaia, T.J. Nanolayer reinforcement of elastomeric polyurethane. Chem. Mater. 1998, 10, 3769. [Google Scholar] [CrossRef]
- Fernández, M.; Landa, M.; Muñoz, M.E.; Santamaría, A. Electrical conductivity of PUR/MWCNT nanocomposites in the molten state, during crystallization and in the solid state. Eur. Polym. J. 2011, 47, 2078–2086. [Google Scholar] [CrossRef]
- Cai, D.Y.; Song, M. High mechanical performance polyurea/organoclay nanocomposites. Compos. Sci. Technol. 2014, 103, 44–48. [Google Scholar] [CrossRef]
- Peng, S.; Iroh, J.O. Dependence of the Dynamic Mechanical Properties and Structure of Polyurethane-Clay Nanocomposites on the Weight Fraction of Clay. J. Compos. Sci. 2022, 6, 173. [Google Scholar] [CrossRef]
- Mohammadi, R.S.; Tabatabaei, S.H.; Ajji, A. Peelable clay/PE nanocomposite seals with ultra-wide peelable heat seal temperature window. Appl. Clay Sci. 2018, 158, 132–142. [Google Scholar] [CrossRef]
- Santamaría, P.; Eguiazabal, J.I. Structure and mechanical properties of blown films of ionomer-compatibilized LDPE nanocomposites. Polym. Test. 2012, 31, 367–374. [Google Scholar] [CrossRef]
- Szustakiewicz, K.; Kiersnowski, A.; Gazińska, M.; Bujnowicz, K.; Pigłowski, J. Flammability, structure and mechanical properties of PP/OMMT nanocomposites. Polym. Degrad. Stab. 2011, 96, 291–294. [Google Scholar] [CrossRef]
- de Mesquita, P.; Jordania, P.; Alves; Soares, T.; Renata, B. Development and characterization of green polyethylene/clay/antimicrobial additive nanocomposites. Polim. Cienc. E Tecnol. 2023, 32, e2022022. [Google Scholar] [CrossRef]
- Zare, Y.; Rhee, K.Y. Estimation of Tensile Modulus for Cross-Linked Polyethylene/Clay Shape Memory Nanocomposites. Phys. Mesomech. 2021, 24, 211–218. [Google Scholar] [CrossRef]
- Mohan, C.; Kumari, N.; Dixit, S. Effect of various types of clay minerals on mechanical and thermal properties of PMMA polymer composite films. MRS Adv. 2022, 7, 933–938. [Google Scholar] [CrossRef]
- Prado, B.R.; Bartoli, J.R. Synthesis and characterization of PMMA and organic modified montmorillonites nanocomposites via in situ polymerization assisted by sonication. Appl. Clay Sci. 2018, 160, 132–143. [Google Scholar] [CrossRef]
- Pugazhenthi, G.; Suresh, K.R.; Kumar, V.; Kumar, M.; Surin, R.R. A Simple Sonication Assisted Solvent Blending Route for Fabrication of Exfoliated Polystyrene (PS)/Clay Nanocomposites: Role of Various Clay Modifiers. Mater. Today Proc. 2018, 5 Pt 2, 13191–13210. [Google Scholar] [CrossRef]
- Alangari, A.M.; Al Juhaiman, L.A.; Mekhamer, W.K. Enhanced Coating Protection of C-Steel Using Polystyrene Clay Nanocomposite Impregnated with Inhibitors. Polymers 2023, 15, 372. [Google Scholar] [CrossRef]
- Hashem, A.; Sheida, E. Organically modified clay as an enhancement filler in novel polyimide mixed matrix membranes for gas separation. Polym. Bull. 2023. early access. [Google Scholar] [CrossRef]
- Lysenkov, E.A.; Klepko, V.; Lazarenko, M.M. Structure-Properties Relationships of Nanocomposites Based on Polyethylene Oxide and Anisometric Nanoparticles. Nanomater. Nanocompos. Nanostruct. Surf. Their Appl. 2023, 279, 409–437. [Google Scholar] [CrossRef]
- Kracalik, M. Recycled clay/PET nanocomposites evaluated by novel rheological analysis approach. Appl. Clay Sci. 2018, 166, 181–184. [Google Scholar] [CrossRef]
- Chung, T.C. Synthesis of functional polyolefin copolymers with graft and block structures. Prog. Polym. Sci. 2002, 27, 39–85. [Google Scholar] [CrossRef]
- Merinska, D.; Tesarikova, A.; Kalendova, A. Polyethylene/Ethylene Vinyl Acetate and Ethylene Octene Copolymer/Clay Nanocomposite Films: Different Processing Conditions and Their Effect on Properties. Polym. Eng. Sci. 2019, 59, 514–2521. [Google Scholar]
- Cherifi, Z.; Boukoussa, B.; Zaoui, A.; Belbachir, M. Structural, morphological and thermal properties of nanocomposites poly (GMA)/clay prepared by ultrasound and in-situ polymerization. Ultrason. Sonochem. 2018, 48, 188–198. [Google Scholar] [CrossRef]
- Benneghmouche, Z.; Benachour, D. Effect of organophilic clay addition on properties of SAN/EPDM blends. Compos. Interfaces 2019, 26, 711–727. [Google Scholar] [CrossRef]
- Xie, W.M.; Chen, H.Y.; He, D.S.; Zhang, Y.; Fu, L.J.; Ouyang, J.; Yang, H.M. An emerging mineral-based composite flame retardant coating: Preparation and enhanced fireproof performance. Surf. Coat. Technol. 2019, 367, 118–126. [Google Scholar] [CrossRef]
- Liu, J.J.; Zhou, K.Q.; Wen, P.Y.; Wang, B.B.; Hu, Y.; Gui, Z. The influence of multiple modified MMT on the thermal and fire behavior of poly (lactic acid) nanocomposites. Polym. Adv. Technol. 2015, 26, 626–634. [Google Scholar] [CrossRef]
- Nanda, T.; Sharma, G.; Mehta, R.; Shelly, D.; Singh, K. Mechanisms for enhanced impact strength of epoxy based nanocomposites reinforced with silicate platelets. Mater. Res. Express 2019, 6, 065061. [Google Scholar] [CrossRef]
- Stloukal, P.; Kalendova, A.; Mattausch, H.; Laske, S.; Holzer, C.; Koutny, M. The influence of a hydrolysis-inhibiting additive on the degradation and biodegradation of PLA and its nanocomposites. Polym. Test. 2015, 41, 124–132. [Google Scholar] [CrossRef]
- Tabsan, N.; Suchiva, K.; Wirasate, S. Effect of montmorillonite on abrasion resistance of SiO2-filled polybutadiene. Compos. Itnerfaces 2012, 19, 1–13. [Google Scholar] [CrossRef]
- Thomas, S.; George, S.C.; Thomas, S. Rigid Amorphous Phase: Mechanical and Transport Properties of Nitrile Rubber/Clay Nanocomposites. Prog. Rubber Plast. Recycl. Technol. 2017, 33, 103–126. [Google Scholar] [CrossRef]
- Gopi, J.A.; Patel, S.K.; Chandra, A.K.; Tripathy, D.K. SBR-clay-carbon black hybrid nanocomposites for tire tread application. J. Polym. Res. 2011, 18, 1625–1634. [Google Scholar] [CrossRef]
- Utracki, L.A. Clay-Containing Polymeric Nanocomposites; Rapra Technology: Shawbury, UK, 2004; Volume 1, ISBN 1-85957-485-8. [Google Scholar]
- Quesada, D.E.; Villarejo, L.P.; Sánchez-Soto, P. Introductory Chapter. In Ceramic Materials—Synthesis, Characterization, Applications and Recycling; IntechOpen: London, UK, 2019. [Google Scholar] [CrossRef]
- Saleh, T.A. Chapter 2: Materials: Types and general classifications. In Polymer Hybrid Materials and Nanocomposites: Fundamentals and Applications; Applied Science Publishers, William Andrew: Cambridge, MA, USA, 2021; pp. 27–58. [Google Scholar]
- Hübner, G. Natural and synthetic raw materials for technical ceramics. Eur. J. Mineral. 1991, 3, 651–665. [Google Scholar] [CrossRef]
- Hlaváč, J. Základy Technologie Silikátů; Státní Nakladatelství Technické Literatury: Prag, Czech Republic, 1981; 508p. (In Czech) [Google Scholar]
- Aşkin, A.; Tatar, I.; Kilinc, Ş.; Tezel, Ö. The Utilization of Waste Magnesite in the Production of the Cordierite Ceramic. Energy Procedia 2017, 107, 137–143. [Google Scholar] [CrossRef]
- Araujo, E.D.; Silva, K.R.; Freitas Grilo, J.P.F.; Macedo, D.A.; Lima Santana, L.N.; Araújo Neves, G. Dielectric Properties of Steatite Ceramics Produced from Talc and Kaolin Wastes. Mater. Res. 2022, 25, e20210428. [Google Scholar] [CrossRef]
- Terzić, A.; Andrić, L.; Stojanović, J.; Obradović, N.; Kostović, M. Mechanical Activation as Sintering Pre-treatment of Talc for Steatite Ceramics. Sci. Sinter. 2014, 46, 247–258. [Google Scholar] [CrossRef]
- Kupková, J.; Valášková, M.; Študentová, S. Influence of acid-treated talc and Na2CO3 flux on mineralogical phase composition and porosity in steatite ceramics. Int. J. Appl. Ceram. Technol. 2017, 14, 803–809. [Google Scholar] [CrossRef]
- Soykan, H.S. Low-temperature fabrication of steatite ceramics with boron oxide addition. Ceram. Int. 2007, 33, 911–914. [Google Scholar] [CrossRef]
- Vela, E.; Peiteado, M.; García, F.; Caballero, A.C.; Fernández, J.F. Sintering behaviour of steatite materials with barium carbonate flux. Ceram. Int. 2007, 33, 1325–1329. [Google Scholar] [CrossRef]
- Valášková, M.; Simha Martynková, G.; Zdrálková, J.; Vlček, J.; Matějková, P. Cordierite composites reinforced with zircon arising from zirconium-vermiculite precursor. Mater. Lett. 2012, 80, 158–161. [Google Scholar] [CrossRef]
- Lee, W.E.; Heuer, A.H. On the polymorphism of Enstatite. J. Am. Ceram. Soc. 1987, 701, 349–360. [Google Scholar] [CrossRef]
- Mielcarek, W.; Nowak-Woźny, D.; Prociów, K. Correlation between MgSiO3 phases and mechanical durability of steatite ceramics. J. Eur. Ceram. Soc. 2004, 24, 3817–3821. [Google Scholar] [CrossRef]
- Goeuriot, D.; Dubois, J.C.; Merle, D.; Thevenot, F.; Exbrayat, P. Enstatite Based Ceramics for Machinable Prosthesis Applications. J. Eur. Ceram. Soc. 1998, 18, 2045–2056. [Google Scholar] [CrossRef]
- Makovše, K.; Ramšak, I.; Malič, B.; Bobna, V.; Kuščer, D. Processing of steatite ceramic with a low dielectric constant and low dielectric losses. Inf. MIDEM 2016, 46, 100–105. [Google Scholar]
- Tavangarian, F.; Emadi, R. Effects of mechanical activation and chlorine on nanoparticle forsterite formation. Mater. Lett. 2011, 65, 126–129. [Google Scholar] [CrossRef]
- Sara Lee, K.Y.; Christopher Chin, K.M.; Ramesh, S.; Tan, C.Y.; Hassan, M.A.; Purbolaksono, J.; Teng, W.D.; Sopyan, I. Effect of ultrasonication on synthesis of forsterite ceramics. Adv. Mat. Res. 2012, 576, 252–255. [Google Scholar] [CrossRef]
- Sara Lee, K.Y.; Christopher Chin, K.M.; Ramesh, S.; Purbolaksono, J.; Hassan, M.A.; Hamdi, M.; Teng, W.D. Characterization of forsterite ceramics. J. Ceram. Process. Res. 2013, 14, 131–133. [Google Scholar]
- Emadi, R.; Tavangarian, F.; Zamani Foroushani, R.; Gholamrezaie, A. The influences of fluorine and chlorine ions on the formation of nanostructure forsterite during mechanical activation of talc and periclase. Ceram. Process. Res. 2011, 12, 538–543. [Google Scholar]
- Ramesh, S.; Yaghoubi, A.; Sara Lee, K.Y.; Christopher Chin, K.M.; Purbolaksono, J.; Hamdi, M.; Hassa, M.A. Nanocrystalline forsterite for biomedical applications: Synthesis, microstructure and mechanical properties. J. Mech. Behav. Biomed. Mater. 2013, 25, 63–69. [Google Scholar] [CrossRef]
- Tavangarian, F.; Emadi, R. Synthesis of pure nanocrystalline magnesium silicate powder. Ceram. Silik. 2010, 54, 122–127. [Google Scholar]
- Nguyen, M.; Sokolar, R. The influence of the raw materials mixture on the properties of forsterite ceramics. IOP Conf. Ser. Mater. Sci. Eng. 2018, 385, 012039. [Google Scholar] [CrossRef]
- Tavangarian, F.; Emadi, R.; Shafyei, A. Influence of mechanical activation and thermal treatment time on nanoparticle forsterite formation mechanism. Powder Technol. 2010, 198, 412–416. [Google Scholar] [CrossRef]
- Tavangarian, F.; Emadi, R. Nanostructure effects on the bioactivity of forsterite bioceramic. Mater. Lett. 2011, 65, 740–743. [Google Scholar] [CrossRef]
- Mustafa, E.; Khalil, N.; Gamal, A. Sintering and microstructure of spinel-forsterite bodies. Ceram. Int. 2002, 28, 663–667. [Google Scholar] [CrossRef]
- Sadeghzade, S.; Emadi, R.; Ghomi, H. Mechanical alloying synthesis of forsterite-diopside nanocomposite powder for using in tissue engineering. Ceram. Silik. 2015, 59, 1–5. [Google Scholar]
- Gökce, H.; Agaogullari, D.; Lütfi Övecoglu, M.; Duman, I.; Boyraz, T. Characterization of microstructural and thermal properties of steatite/cordierite ceramics prepared by using natural raw materials. J. Eur. Ceram. Soc. 2011, 31, 2741–2747. [Google Scholar] [CrossRef]
- Goren, R.; Gocmez, H.; Ozgur, C. Synthesis of cordierite powder talc, diatomite and alumina. Ceram. Int. 2006, 32, 407–409. [Google Scholar] [CrossRef]
- Chowdhury, A.; Maitra, S.; Das, S.; Sen, A.; Samanta, G.K.; Datta, P. Synthesis, Properties and Applications of Cordierite Ceramics, Part 1. Interceram Int. Ceram. Rev. 2007, 56, 18–22. [Google Scholar]
- Valášková, M.; Simha Martynková, G.; Smetana, B.; Študentová, S. Influence of vermiculite on the formation of porous cordierites. Appl. Clay Sci. 2009, 46, 196–201. [Google Scholar] [CrossRef]
- Tamborenea, S.; Mazzoni, A.D.; Aglietti, E.F. Mechanochemical activation of minerals on the cordierite synthesis. Thermochim. Acta 2004, 411, 219–224. [Google Scholar] [CrossRef]
- Gusev, A.A.; Avvakumov, E.G.; Vinokurova, O.B.; Salostii, V.P. The Effect of transition metal oxides on the strength, phase composition and microstructure of cordierite ceramics. Glass Ceram. 2001, 58, 24–26. [Google Scholar] [CrossRef]
- Pavlikov, V.M.; Garmash, E.P.; Yurchenko, V.A.; Pleskach, I.V.; Oleinik, G.S.; Grigorév, O.M. Mechanochemical activation of kaolin, pyrophyllite, and talcum and its effect on the synthesis of cordierite ceramics. Powder Metall. Met. Ceram. 2011, 49, 564–574. [Google Scholar] [CrossRef]
- Valášková, M.; Simha Martynková, G. Microporous Cordierite Ceramics Prepared from Clay Mineral Mixtures Containing Vermiculite. J. Sci. Conf. Proc. 2010, 2, 49–52. [Google Scholar] [CrossRef]
- Valášková, M.; Simha Martynková, G. Preparation and characterization of porous cordierite for potential use in cellular ceramics. Chem. Pap. 2009, 63, 445–449. [Google Scholar] [CrossRef]
- Kobayashi, Y.; Sumi, K.; Kato, E. Preparation of dense cordierite ceramics from magnesium compounds and kaolinite without additives. Ceram. Int. 2000, 26, 739–743. [Google Scholar] [CrossRef]
- Alves, H.M.; Tarí, G.; Fonseca, A.T.; Ferreira, J.M.F. Processing of porous cordierite bodies by starch consolidation. Mater. Res. Bull. 1998, 33, 1439–1448. [Google Scholar] [CrossRef]
- Almeida, E.P.; Brito, I.P.; Ferreira, H.C.; Lira, H.L.; Santana, L.N.L.; Neves, G.A. Cordierite obtained from compositions containing kaolin waste, talc and magnesium oxide. Ceram. Int. 2018, 44, 1719–1725. [Google Scholar] [CrossRef]
- Goren, R.; Ozgur, C.; Gocmez, H. The preparation of cordierite from talc, fly ash, fused silica and alumina mixtures. Ceram. Int. 2006, 32, 53–56. [Google Scholar] [CrossRef]
- Kumar, S.; Singh, K.K.; Ramachandrarao, P. Synthesis of cordierite from fly ash and its refractory properties. J. Mater. Sci. Lett. 2000, 19, 1263–1265. [Google Scholar] [CrossRef]
- Song, I.H.; Kim, M.J.; Kim, H.D.; Kim, Y.W. Processing of microcellular cordierite ceramics from a preceramic polymer. Scr. Mater. 2006, 54, 1521–1525. [Google Scholar] [CrossRef]
- Valášková, M.; Zdrálková, J.; Simha Martynková, G.; Smetana, B.; Vlček, J.; Študentová, S. Structural variability of high purity cordierite/steatite ceramics sintered from mixtures with various vermiculites. Ceram. Int. 2014, 40, 8489–8498. [Google Scholar] [CrossRef]
- Valášková, M.; Zdrálková, J.; Tokarský, J.; Simha Martynková, G.; Ritz, M.; Študentová, S. Structural characteristic of cordierite/steatite ceramics sintered from mixtures containing pore-forming organovermiculite. Ceram. Int. 2014, 40, 15717–15725. [Google Scholar] [CrossRef]
- Acimovic, Z.; Pavlovic, L.; Trumbulovic, L.; Andric, L.; Stamatovic, M. Synthesis and characterization of the cordierite ceramics from nonstandard raw materials for application in foundry. Mater. Lett. 2003, 57, 2651–2656. [Google Scholar] [CrossRef]
- Li, Y.; Cao, W.; Feng, J.; Gong, L.; Cheng, X. Fabrication of cordierite foam ceramics using direct foaming and slip casting method with plaster moulds. Adv. Appl. Ceram. 2015, 114, 465–470. [Google Scholar] [CrossRef]
- Valášková, M.; Mikeska, M.; Študentová, S.; Simha Martynková, G. Cordierite/steatite ceramics sintered from talc, kaolin and vermiculites: Comparison of natural and organovermiculites effect. Mater. Today Proc. 2018, 5, S88–S95. [Google Scholar] [CrossRef]
- Valášková, M.; Kočí, K.; Kupková, J. Cordierite/steatite/CeO2 porous materials—preparation, structural characterization and their photocatalytic activity. Microporous Mesoporous Mater. 2015, 207, 120–125. [Google Scholar] [CrossRef]
- Valášková, M.; Hundáková, M.; Smetana, B.; Drozdová, L.; Klemm, V.; Rafaja, D. Cordierite/CeO2 ceramic nanocomposites from vermiculite with fixed CeO2 nanoparticles, talc and kaolin. Appl. Clay Sci. 2019, 179, 105150. [Google Scholar] [CrossRef]
- Valášková, M.; Tokarský, J.; Hundáková, M.; Zdrálková, J.; Smetana, B. Role of vermiculite and zirconium-vermiculite on the formation of zircon-cordierite nanocomposites. Appl. Clay Sci. 2013, 75–76, 100–108. [Google Scholar] [CrossRef]
- Cheng, H.; Ye, F.; Chang, J.; Wu, S. In-situ synthesis and thermal shock resistance of a cordierite-mullite composite for solar thermal storage. Int. J. Appl. Ceram. Tec. 2019, 16, 772–780. [Google Scholar] [CrossRef]
- Anadaão, P. Polymer/Clay Nanocomposites: Concepts, Researches, Applications and Trends for the Future; IntechOpen Limited: London, UK, 2012. [Google Scholar] [CrossRef]


| Composition | Permeability
[m2 Pa−1s−1] × 10−16 | % to Pure PLA [%] | Permeability Q(Air) [m2 Pa−1s−1] × 10−16 | %Q(Air) to Pure PLA [%] |
|---|---|---|---|---|
| PLA | 2.28 | 0 | 3.94 | 0 |
| PLA/10A | 0.71 | −69 | 1.57 | −60 |
| PLA/20A | 0.84 | −63 | 1.95 | −51 |
| PLA/30B | 0.80 | −65 | 1.35 | −66 |
| PLA/Na+ | 5.56 | 144 | 5.80 | 47 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kalendova, A.; Kupkova, J.; Urbaskova, M.; Merinska, D. Applications of Clays in Nanocomposites and Ceramics. Minerals 2024, 14, 93. https://doi.org/10.3390/min14010093
Kalendova A, Kupkova J, Urbaskova M, Merinska D. Applications of Clays in Nanocomposites and Ceramics. Minerals. 2024; 14(1):93. https://doi.org/10.3390/min14010093
Chicago/Turabian StyleKalendova, Alena, Jana Kupkova, Martina Urbaskova, and Dagmar Merinska. 2024. "Applications of Clays in Nanocomposites and Ceramics" Minerals 14, no. 1: 93. https://doi.org/10.3390/min14010093
APA StyleKalendova, A., Kupkova, J., Urbaskova, M., & Merinska, D. (2024). Applications of Clays in Nanocomposites and Ceramics. Minerals, 14(1), 93. https://doi.org/10.3390/min14010093








