Post-Serpentinization Formation of Theophrastite-Zaratite by Heazlewoodite Desulfurization: An Implication for Shallow Behavior of Sulfur in a Subduction Complex
Abstract
1. Introduction
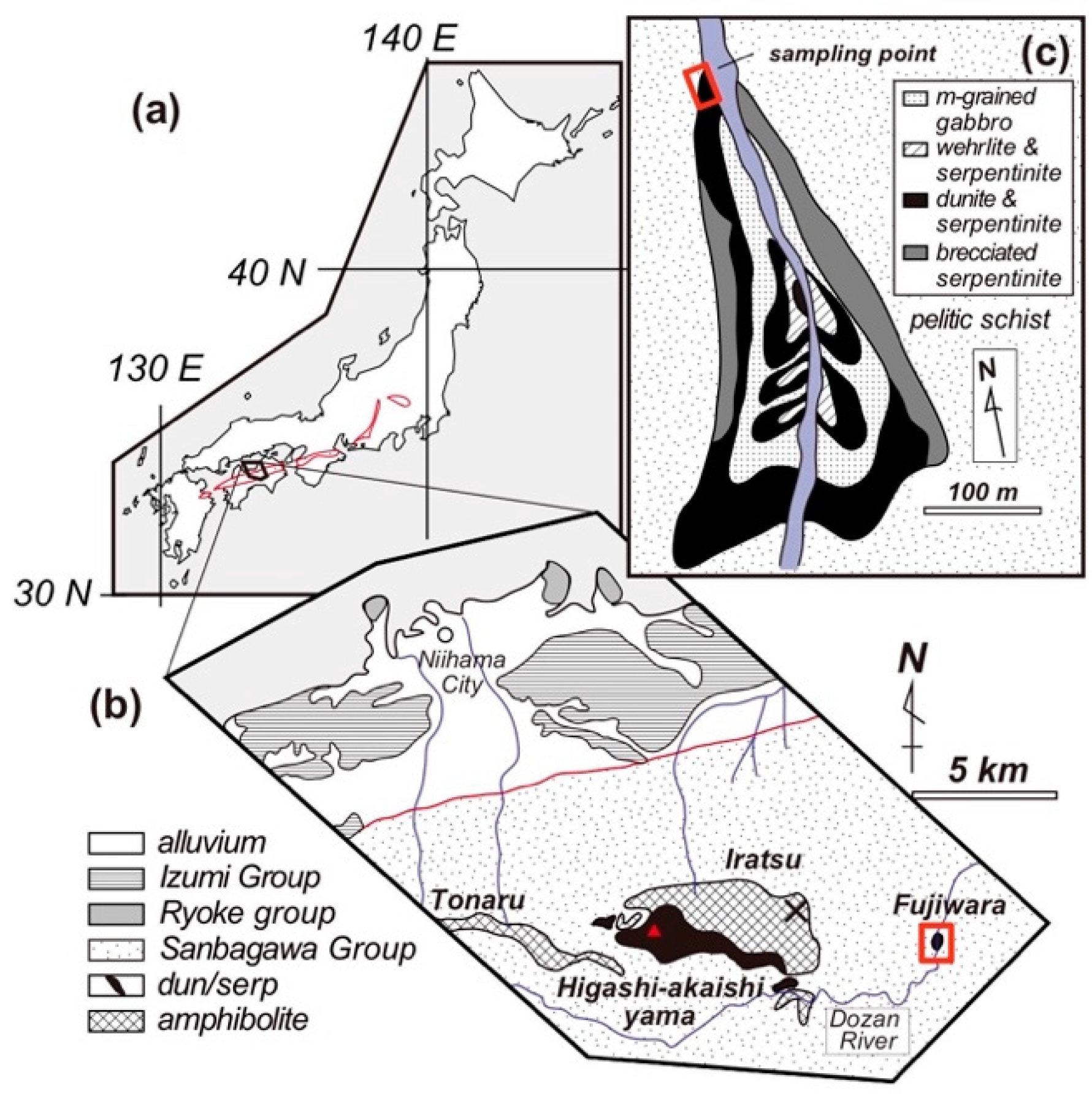
2. Geological Background and Petrography
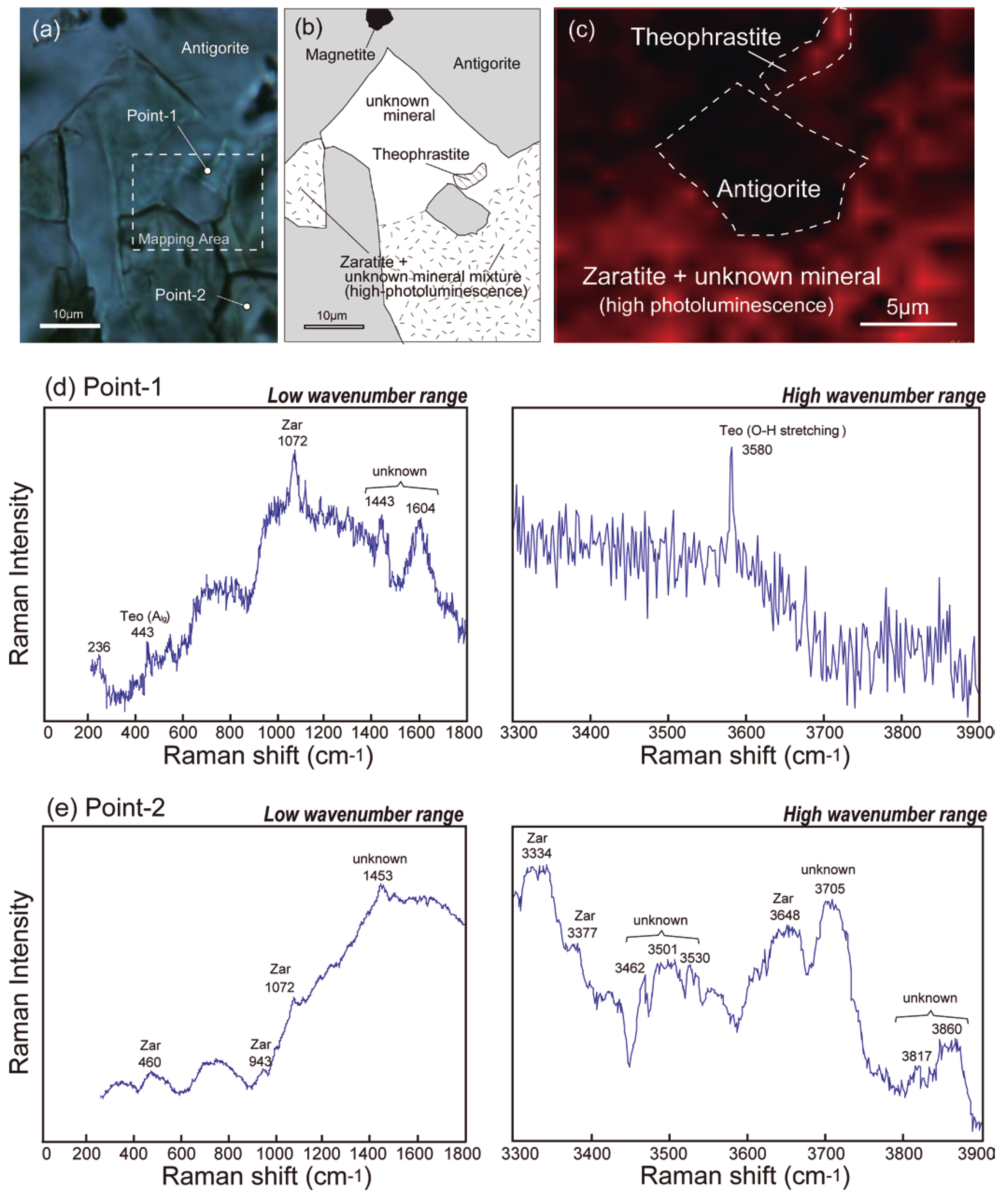
3. Raman Spectroscopy
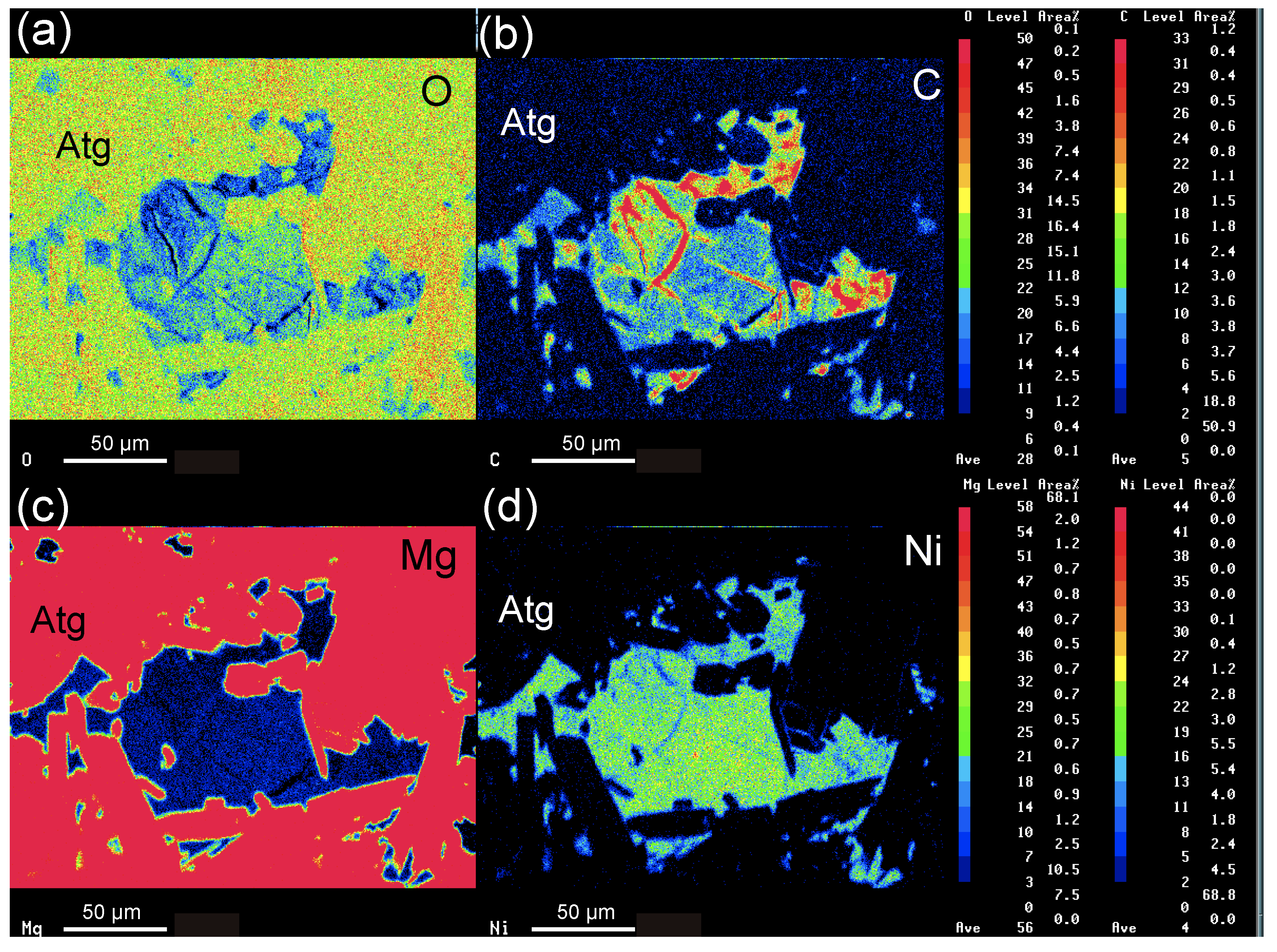
4. Mineral Chemistry
5. Discussion
5.1. Behavior of Nickel during Serpentinization of Olivine
5.2. Origin of Theophrastite-Zaratite in the Fujiwara Meta-Dunite
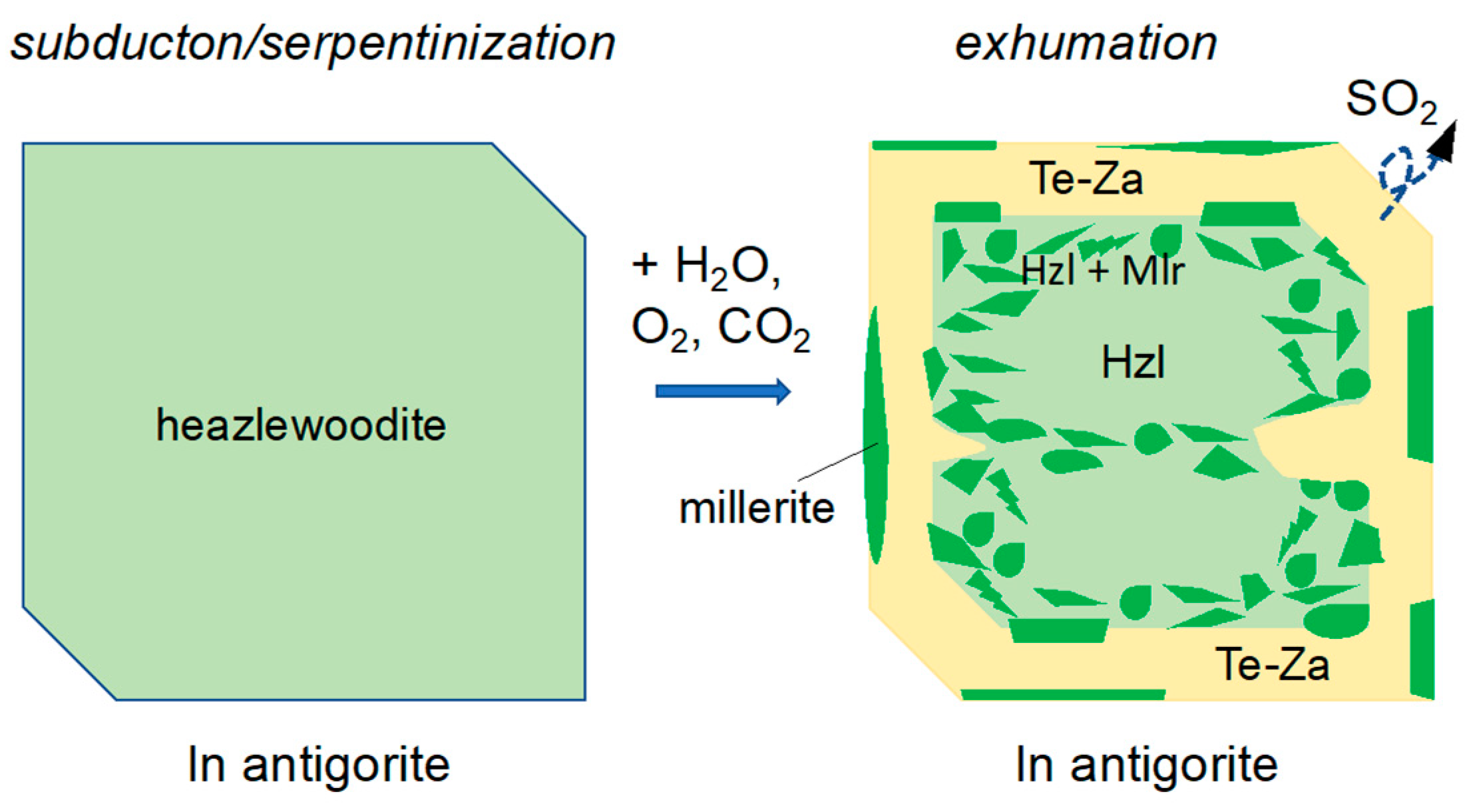
5.3. Desulfurization of Heazlewoodite and the Behavior of Sulfur
6. Conclusions and Implications
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Schwarzenbach, E.M.; Caddick, M.J.; Petroff, M.; Gill, B.C.; Cooperdock, E.H.G.; Barnes, J.D. Sulphur and carbon cycling in the subduction zone mélange. Sci. Rep. 2018, 8, 15517. [Google Scholar] [CrossRef]
- Piccoli, F.; Hermann, J.; Pettke, T.; Connolly, J.A.; Kempf, E.D.; Duarte, J.F.V. Subducting serpentinites release reduced, not oxidized, aqueous fluids. Sci. Rep. 2019, 9, 19573. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Schwarzenbach, E.M.; John, T.; Ague, J.J.; Huang, F.; Gao, J.; Klemd, R.; Whitehouse, M.J.; Wang, X.-S. Uncovering and quantifying the subduction zone sulfur cycle from the slab perspective. Nat. Commun. 2020, 11, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Alt, J.C.; Shanks, W.C. Microbial sulfate reduction and the sulfur budget for a complete section of altered oceanic basalts, IODP Hole 1256D (eastern Pacific). Earth Planet. Sci. Lett. 2011, 310, 73–83. [Google Scholar] [CrossRef]
- Alt, J.C.; Shanks, W.C. Sulfur in serpentinized oceanic peridotites: Serpentinization processes and microbial sulfate reduction. J. Geophys. Res. 1998, 103, 9917–9929. [Google Scholar] [CrossRef]
- Schwarzenbach, E.M.; Früh-Green, G.L.; Bernasconi, S.M.; Alt, J.C.; Shanks, W.C., III; Gaggero, L.; Crispini, L. Sulfur geochemistry of peridotite-hosted hydrothermal systems: Comparing the Ligurian ophiolites with oceanic serpentinites. Geochim. Cosmochim. Acta 2012, 91, 283–305. [Google Scholar] [CrossRef]
- Alt, J.C.; Schwarzenbach, E.M.; Früh-Green, G.L.; Shanks, W.C., III; Bernasconi, S.M.; Garrido, C.J.; Crispini, L.; Gaggero, L.; Pardón-Navarta, J.A.; Marchesi, C. The role of serpenitnites in cycling of carbon and sulfur: Seafloor serpentinization and subduction metamorphism. Lithos 2013, 178, 40–54. [Google Scholar] [CrossRef]
- Alt, J.C.; Garrido, C.J.; Shanks, W.; Turchyn, A.V.; Padrón-Navarta, J.A.; Sánchez-Vizcaíno, V.L.; Gómez-Pugnaire, M.T.; Marchesi, C. Recycling of water, carbon, and sulfur during subduction of serpentinites: A stable isotope study of Cerro del Almirez, Spain. Earth Planet. Sci. Lett. 2012, 327, 50–60. [Google Scholar] [CrossRef]
- Richards, J.P. The oxidation state, and sulfur and Cu contents of arc magmas: Implications for metallogeny. Lithos 2015, 233, 27–45. [Google Scholar] [CrossRef]
- Sato, H. Nickel content of basaltic magmas: Identification of primary magmas and a measure of the degree of olivine fractionation. Lithos 1977, 10, 113–120. [Google Scholar] [CrossRef]
- Williams, K.L. An association of awaruite with heazlewoodite. Am. Mineral. 1960, 45, 450–453. [Google Scholar]
- Frost, B.R. On the Stability of Sulfides, Oxides, and Native Metals in Serpentinite. J. Petrol. 1985, 26, 31–63. [Google Scholar] [CrossRef]
- Abrajano, T.A.; Pasteris, J.D. Zambales ophiolite, Philippines II. Sulfide petrology of the critical zone of the Acoje Massif. Contrib. Mineral. Petrol. 1989, 103, 64–77. [Google Scholar] [CrossRef]
- Klein, F.; Bach, W. Fe-Ni-Co-O-S Phase Relations in Peridotite-Seawater Interactions. J. Petrol. 2009, 50, 37–59. [Google Scholar] [CrossRef]
- Sleep, N.H.; Meibom, A.; Fridriksson, T.; Coleman, R.G.; Bird, D.K. H2-rich fluids from serpentinization: Geochemical and biotic implications. Proc. Natl. Acad. Sci. USA 2004, 101, 12818–12823. [Google Scholar] [CrossRef] [PubMed]
- Marcopoulos, T.; Economou, M. Theophrstite, Ni(OH)2, a new mineral from northern Greece. Am. Mineral. 1981, 66, 1020–1021. [Google Scholar]
- Economou, M.; Marcopoulos, T. Genesis of the new mineral theophrastite, Ni(OH)2. Chem. Erde 1983, 42, 53–56. [Google Scholar]
- Livingstone, A.; Bish, D.L. On the new mineral thoephrastite, a nickel hydroxide, from Unst, Shetland, Scotland. Mineral. Mag. 1982, 46, 1–5. [Google Scholar] [CrossRef]
- Henry, D.A.; Birch, W.D. Otwayite and theophrastite from the Lord Brassey Mine, Tasmania. Mineral. Mag. 1992, 56, 252–255. [Google Scholar] [CrossRef][Green Version]
- Williams, K.L.; Threadgold, I.M.; Hounslow, A.W. Hellyerite, a new nickel carbonate from Heazlewood, Tasmania. Am. Mineral. 1959, 44, 533–538. [Google Scholar]
- Isaacs, T. The mineralogy and chemistry of the nickel carbonates. Mineral. Mag. J. Mineral. Soc. 1963, 33, 663–678. [Google Scholar] [CrossRef]
- Nickel, E.H.; Hallberg, J.A.; Halligan, R. Unusual nickel mineralisation at Nullagine, Western Australia. J. Geol. Soc. Aust. 1979, 26, 61–71. [Google Scholar] [CrossRef]
- Nickel, E.H.; Robinson, B.W.; Davis, C.E.S.; MacDonald, R.D. Otwayite, a new nickel mineral from Western Australia. Am. Mineral. 1977, 62, 999–1002. [Google Scholar]
- Hudson, D.R.; Travis, G.A. A native nickel-heazlewoodite-ferroan trevorite assemblage from Mount Clifford, Western Australia. Econ. Geol. 1981, 76, 1686–1697. [Google Scholar] [CrossRef]
- Springer, G. Compositional and structural variations in garnierites. Can. Mineral. 1974, 12, 381–388. [Google Scholar]
- Arai, S.; Ishimaru, S.; Mizukami, T. Methane and propane micro-inclusions in olivine in titanoclinohumite-bearing dunites from the Sanbagawa high-P metamorphic belt, Japan: Hydrocarbon activity in a subduction zone and Ti mobility. Earth Planet. Sci. Lett. 2012, 353, 1–11. [Google Scholar] [CrossRef]
- Onuki, H.; Yoshida, T.; Suzuki, T. The Fujiwara mafic-ultramafic complex in the Sanbagawa metamorphic belt of central Shikoku 1. Petrochemistry and rock-forming mineralogy. J. Jpn. Assoc. Petrol. Mineral. Econ. Geol. 1978, 73, 311–322. (In Japanese) [Google Scholar] [CrossRef]
- Ishibashi, K.; Miyahisa, M.; Sasaki, M. Titanclinohumite from Fujiwara ultramafic complex in the Higashi-Akaishi mountainland, Shikoku, Japan. J. Jpn. Assoc. Petrol. Mineral. Econ. Geol. 1978, 73, 18–25. (In Japanese) [Google Scholar] [CrossRef]
- Kunugiza, K. Metamorphism and origin of ultramafic bodies of the Sanbagawa metamorphic belt in central Shikoku. J. Jpn. Assoc. Petrol. Mineral. Econ. Geol. 1984, 79, 20–32. (In Japanese) [Google Scholar] [CrossRef]
- Mizukami, T.; Wallis, S.R. Structural and petrological constraints on the tectonic evolution of the garnet-lherzolite facies Higashi-akaishi peridotite body, Sanbagawa belt, SW Japan. Tectonics 2005, 24, 6012. [Google Scholar] [CrossRef]
- Miyashiro, A. Metamorphism and Metamorphic Belts; George Allen & Unwin: London, UK, 1973; 492p. [Google Scholar]
- Banno, S.; Sakai, C. Geology and metamorphic evolution of the Sanbagawa metamorphic belt, Japan. Geol. Soc. Lond. Spec. Publ. 1989, 43, 519–532. [Google Scholar] [CrossRef]
- Enami, M. Petrology of the Fujiwara mass and the surrounding pelitic schists in the Sanbagawa metamorphic belt, central Shikoku. J. Geol. Soc. Jpn. 1980, 86, 461–473. (In Japanese) [Google Scholar] [CrossRef][Green Version]
- Garuti, G.; Zaccarini, F. In situ alteration of platinum-group minerals at low temperature: Evidence from serpentinized and weathered chromitite of the Vourinos complex, Greece. Can. Mineral. 1997, 35, 611–626. [Google Scholar]
- Garuti, G.; Zaccarini, F.; Cabella, R.; Fershtater, G. Occurrence of unknown Ru-Os-Ir-Fe oxides in the chromitites of the Nurali ultramafic complex, southern Urals, Russia. Can. Mineral. 1997, 35, 1431–1439. [Google Scholar]
- Ahmed, A.H.; Arai, S.; Kadoshima, K. Possible platinum-group element (PGE) oxides in the PGE-mineralized chromitite from the Northern Oman Ophiolite. J. Mineral. Petrol. Sci. 2002, 97, 190–198. [Google Scholar] [CrossRef]
- Lutz, H.; Möller, H.; Schmidt, M. Lattice vibration spectra. Part LXXII. Brucite-type hydroxides M(OH)2 (M = Ca, Mn, Co, Fe, Cd)—IR and Raman spectra, neutron diffractions of Fe(OH)2. J. Mol. Struct. 1994, 328, 121–132. [Google Scholar] [CrossRef]
- Murli, C.; Sharma, S.M.; Kulshreshtha, S.K.; Sikka, S.K. High-pressure behavior of ß-Ni(OH)2—A Raman scattering study. Phys. B 2001, 307, 111–116. [Google Scholar] [CrossRef]
- Frost, R.L.; Dickfos, M.J.; Reddy, B.J. Raman spectroscopy of hydroxyl nickel carbonate minerals nullaginite and zaratite. J. Raman Spectr. 2008, 39, 1250–1256. [Google Scholar] [CrossRef]
- Frost, R.L.; Weier, M.L.; Martens, W.N.; Mills, S.J. The hydroxylated nickel carbonates otwayite and paraotwayite—A SEM, EDX and vibrational spectroscopic study. Neues Jahrbush Mineral. Abh. 2006, 183, 107–116. [Google Scholar] [CrossRef]
- Auzende, A.-L.; Daniel, I.; Reynard, B.; Lemaire, C.; Guyot, R. High-pressure behavior of serpentine minerals: A Raman spectroscopic study. Phys. Chem. Mineral. 2004, 31, 269–277. [Google Scholar] [CrossRef]
- Lorand, J.P.; Pinet, M. L’orcelite des péridotites de Beni Bousera (Maroc), Ronda (Espagne), Table Mountain et Blow-Me-Down Mountain (Terre-Neuve) et Du Pinde Septentrional (Grèce). Can. Mineral. 1984, 22, 553–560. (In French) [Google Scholar]
- Gritsenko, Y.D.; Spiridonov, E.M. Maucherite from metamorphic-hydrothermal assemblages of the Noril’sk ore field. Geol. Ore Depos. 2008, 50, 590–598. [Google Scholar] [CrossRef]
- Bindi, L.; Tredoux, M.; Zaccarini, F.; Miller, D.E.; Garuti, G. Non-stoichiometric nickel arsenides in nature: The structure of orcelite, Ni5−xAs2 (x = 0.25), from the Bon Accord oxide body, South Africa. J. Alloys Compd. 2014, 601, 175–178. [Google Scholar] [CrossRef]
- Haxel, G.B.; Wittke, J.H.; Epstein, G.S.; Jacobson, C.E. Serpentinization-related nickel, iron, and cobalt sulfide, arsenide, and intermetallic minerals in an unusual inland tectonic setting, southern Arizona, USA. Geol. Soc. Am. Spec. Pap. 2018, 540, 65–87. [Google Scholar]
- Kadota, R.; Minagawa, T.; Ohgoshi, Y.; Hamane, D. Ni-As-Sb metalloid alloys from Higashiakaishi peridotite mass. In Proceedings of the 118th Annual Meeting of the Geological Society of Japan and the 2011 Annual Meeting of the Japan Association of Mineralogical Sciences (Mito Conference), Mito, Japan, 9–11 September 2011; pp. R1–P14. (In Japanese). [Google Scholar]
- Kamenetsky, V.; Lygin, A.V.; Foster, J.G.; Meffre, S.; Maas, R.; Kamenetsky, M.B.; Goemann, K.; Beresford, S.W. A story of olivine from the McIvor Hill complex (Tasmania, Australia): Clues to the origin of the Avebury metasomatic Ni sulfide deposit. Am. Mineral. 2016, 101, 1321–1331. [Google Scholar] [CrossRef]
- Filippidis, A. Formation of awaruite in the system Ni-Fe-Mg-Si-O-H-S and olivine hydration with NaOH solution, an experimental study. Econ. Geol. 1985, 80, 1974–1980. [Google Scholar] [CrossRef]
- Savov, I.; Ryan, J.; D’Antonio, M.; Kelley, K.; Mattie, P. Geochemistry of serpentinized peridotites from the Mariana Forearc Conical Seamount, ODP Leg 125: Implications for the elemental recycling at subduction zones. Geochem. Geophys. Geosystems 2005, 6, Q04J15. [Google Scholar] [CrossRef]
- Peretti, A.; Dubessy, J.; Mullis, J.; Frost, B.R.; Trommsdorff, V. Highly reducing conditions during Alpine metamorphism of the Malenco peridotite (Sondrio, northern Italy) indicated by mineral paragenesis and H2 in fluid inclusions. Contrib. Mineral. Petrol. 1992, 112, 329–340. [Google Scholar] [CrossRef]
- Barnes, S.J. Chromite in Komatiites, II. Modification during Greenschist to Mid-Amphibolite Facies Metamorphism. J. Petrol. 2000, 41, 387–409. [Google Scholar] [CrossRef]
- Gahlan, H.A.; Arai, S.; Ahmed, A.H.; Ishida, Y.; Abdel-Aziz, Y.M.; Rahimi, A. Origin of magnetite veins in serpentinite from the Late Proterozoic Bou-Azzer ophiolite, Anti-Atlas, Morocco: An implication for mobility of iron during serpentinization. J. Afr. Earth Sci. 2006, 46, 318–330. [Google Scholar] [CrossRef]
- Tzamos, E.; Filippidis, A.; Michailidis, K.; Koroneos, A.; Rassios, A.; Grieco, G.; Pedrotti, M.; Stamoulis, K. Mineral Chemistry and Formation of Awaruite and Heazlewoodite in the Xerolivado Chrome Mine, Vourinos, Greece. Bull. Geol. Soc. Greece 2016, 50, 2047–2056. [Google Scholar] [CrossRef][Green Version]
- Evans, B.W. Lizardite versus antigorite serpentinite: Magnetite, hydrogen, and life (?). Geology 2010, 38, 879–882. [Google Scholar] [CrossRef]
- Klein, F.; Bach, W.; Humbphris, S.E.; Kahl, W.-A.; Jöns, N.; Moskowitz, B.; Berquó, T.S. Magnetite in seafloor serpentinites—Some like it hot. Geology 2014, 42, 135–138. [Google Scholar] [CrossRef]
- Merlini, A.; Grieco, G.; Diella, V. Ferritchromite and chromian-chlorite formation in melange-hosted Kalkan chromitite (Southern Urals, Russia). Am. Mineral. 2009, 94, 1459–1467. [Google Scholar] [CrossRef]
- Khedr, M.Z.; Arai, S. Hydrous peridotites with Ti-rich chromian spinel as a low-temperature forearc mantle facies: Evidence from the Happo-O’ne metaperidotites (Japan). Contrib. Mineral. Petrol. 2010, 159, 137–157. [Google Scholar] [CrossRef]
- Nickel, E.H.; Berry, L.G. The new mineral nullaginite and additional data on the related minerals rosasite and glaukosphaerite. Can. Mineral. 1981, 19, 315–324. [Google Scholar]
- Nickel, E.H.; Graham, J. Paraotowayite, a new nickel hydroxide mineral from Western Australia. Can. Mineral. 1987, 25, 409–411. [Google Scholar]
- Ziemniak, S.; Goyette, M. Nickel (II) Oxide Solubility and Phase Stability in High Temperature Aqueous Solutions. J. Solut. Chem. 2004, 33, 1135–1159. [Google Scholar] [CrossRef]
- Evans, B.W.; Johannes, W.; Oterdoom, H.; Trommsdorff, V. Stability of chrysotile and antigorite in the serpentinite multisystem. Schweiz. Mineral. Petrogr. Mitt. 1976, 56, 79–93. [Google Scholar]
- Economou-Eliopoulos, M. Apatite and Mn, Zn, Co-enriched chromite in Ni-laterites of northern Greece and their genetic significance. J. Geochem. Explor. 2003, 80, 41–54. [Google Scholar] [CrossRef]
- Prichard, H.M.; Ixer, R.B.; Lord, R.A.; Maynard, J.; Williams, N. Assemblages of platinum-group minerals and sulfides in silicate lithologies and chromite-rich rocks within the Shetland ophiolite. Can. Mineral. 1994, 32, 271–294. [Google Scholar]
- Buckley, A.N.; Woods, R. Electrochemical and XPS studies of the surface oxidation of synthetic heazlewoodite (Ni3S2). J. Appl. Electrochem. 1991, 21, 575–582. [Google Scholar] [CrossRef]
- Naldrett, A.J. Magmatic Sulfide Deposits: Geology, Geochemistry and Exploration; Clarendon Press/Oxford University Press: New York, NY, USA, 1989; 186p. [Google Scholar]
- Guillot, S.; Hattori, K.; Agard, P.; Schwartz, S.; Vidal, O. Exhumation processes in oceanic and continental subduction contexts: A review. In Subduction Zone Geodynamics; Lallemand, S., Funiciello, F., Eds.; Springer: Berlin, Germany, 2009; pp. 175–205. [Google Scholar]
- Shibata, T. On the Occurrence of Anhydrite in the Iratsu Ultramafic Complex, Central Shikoku. J. Jpn. Assoc. Miner. Petrol. Econ. Geol. 1972, 67, 76–83. [Google Scholar] [CrossRef]
- Yamamoto, M.; Shibata, T.; Kusakabe, M. Sulfur and oxygen isotope ratios of anhydrite from the Iratsu peridotite-hornblendite-epidote amphibolite complex, central Shikoku, Japan. Geochem. J. 1987, 21, 247–251. [Google Scholar] [CrossRef]
- Ota, T.; Terabayashi, M.; Katayama, I. Thermobaric structure and metamorphic evolution of the Iratsu eclogite body in the Sanbagawa belt, central Shikoku, Japan. Lithos 2004, 73, 95–126. [Google Scholar] [CrossRef]
- Endo, S.; Mizukami, T.; Wallis, S.R.; Tamura, A.; Arai, S. Orthopyroxene-rich Rocks from the Sanbagawa Belt (SW Japan): Fluid–Rock Interaction in the Forearc Slab–Mantle Wedge Interface. J. Petrol. 2015, 56, 1113–1137. [Google Scholar] [CrossRef]
- Ahmed, Z.; Hall, A. Nickeliferous opaque minerals associated with chromite alteration in the Sakhakot-Qila complex, Pakistan, and their compositional variation. Lithos 1982, 15, 39–47. [Google Scholar] [CrossRef]




| Phase | Ol | Ti-Chu | Chl | Atg | Chr | Fch | Mag | Te-Za | |||
|---|---|---|---|---|---|---|---|---|---|---|---|
| SiO2 | 40.47 | 40.90 | 37.02 | 34.75 | 44.09 | nd | 0.44 | 0.01 | 0.17 | 0.22 | 0.20 |
| TiO2 | 0.03 | 0.40 | 3.31 | nd | 0.03 | 0.91 | 0.73 | 016 | nd | nd | 0.03 |
| Al2O3 | nd | nd | nd | 11.76 | 0.40 | 17.51 | 3.16 | 0.03 | 0.01 | nd | 0.01 |
| Cr2O3 | nd | nd | 0.02 | 0.51 | nd | 35.39 | 31.11 | 0.18 | nd | nd | nd |
| FeO* | 10.95 | 4.95 | 6.63 | 3.20 | 1.16 | 35.66 | 53.49 | 91.81 | 0.15 | 0.28 | 0.81 |
| MnO | 0.79 | 0.21 | 0.35 | 0.01 | 0.04 | 1.18 | 2.50 | 0.17 | nd | nd | nd |
| MgO | 48.36 | 53.14 | 50.78 | 35.52 | 40.35 | 8.33 | 3.73 | 0.98 | 4,17 | 5.05 | 4.49 |
| CaO | 0.01 | 0.01 | 0.01 | 0.01 | nd | 0.02 | nd | nd | 0.37 | 0.31 | 0.26 |
| Na2O | nd | nd | 0.01 | 0.01 | nd | 0.02 | 0.05 | 0.04 | 0.03 | 0.03 | 0.05 |
| K2O | nd | nd | nd | 0.01 | 0.01 | nd | nd | nd | nd | nd | nd |
| NiO | 0.41 | 0.27 | 0.13 | 0.10 | 0.11 | 0.22 | 0.53 | 0.66 | 60.45 | 57.22 | 58.97 |
| Total | 101.04 | 99.88 | 98.25 | 85.87 | 86.18 | 99.24 | 95.72 | 94.02 | 65.38 | 63.11 | 64.81 |
| O | 4 | 4 | 13 | 14 | 7 | 4 | 4 | 4 | 10 | 10 | 10 |
| Si | 0.992 | 0.985 | 2.996 | 3.319 | 2.056 | 0.000 | 0.018 | 0.000 | 0.030 | 0.041 | 0.036 |
| Ti | 0.001 | 0.007 | 0.201 | 0.000 | 0.001 | 0.023 | 0.022 | 0.006 | 0.000 | 0.000 | 0.003 |
| Al | 0.000 | 0.000 | 0.000 | 1.323 | 0.022 | 0.701 | 0.153 | 0.002 | 0.001 | 0.000 | 0.003 |
| Cr | 0.000 | 0.000 | 0.001 | 0.038 | 0.000 | 0.950 | 1.011 | 0.007 | 0.000 | 0.000 | 0.000 |
| Fe* | 0.224 | 0.100 | 0.448 | 0.255 | 0.045 | 1.013 | 1.839 | 3.865 | 0.023 | 0.042 | 0.122 |
| Mn | 0.016 | 0.004 | 0.024 | 0.000 | 0.002 | 0.034 | 0.087 | 0.007 | 0.000 | 0.000 | 0.000 |
| Mg | 1.766 | 1.905 | 6.122 | 5.054 | 2.802 | 0.422 | 0.229 | 3.865 | 1.115 | 1.378 | 1.024 |
| Ca | 0.000 | 0.000 | 0.001 | 0.001 | 0.000 | 0.001 | 0.000 | 0.007 | 0.071 | 0.062 | 0.049 |
| Na | 0.000 | 0.000 | 0.001 | 0.002 | 0.000 | 0.002 | 0.004 | 0.000 | 0.010 | 0.009 | 0.018 |
| K | 0.000 | 0.000 | 0.000 | 0.001 | 0.000 | 0.000 | 0.000 | 0.004 | 0.000 | 0.000 | 0.000 |
| Ni | 0.008 | 0.005 | 0.009 | 0.008 | 0.004 | 0.006 | 0.018 | 0.000 | 8.721 | 8.432 | 8.533 |
| Total | 3.008 | 3.008 | 9.803 | 10.002 | 4.932 | 3.152 | 3.338 | 3.991 | 9.973 | 9.964 | 9.968 |
| Mg# | 0.887 | 0.950 | 0.932 | 0.952 | 0.984 | 0.405 | 0.211 | 0.056 | 0.980 | 0.970 | 0.908 |
| Cr# | 0.576 | 0.869 | 0.812 | ||||||||
| YCr | 0.465 | 0.471 | 0.003 | ||||||||
| YAl | 0.343 | 0.071 | 0.001 | ||||||||
| YFe | 0.192 | 0.458 | 0.997 | ||||||||
| Minerals | Hzl | Mlr | Hzl* | Mlr* | Orc | Mau | Awr |
|---|---|---|---|---|---|---|---|
| Fe | 1.66 | 0.66 | 0.31 | 22.01 | |||
| Co | 0.69 | 0.64 | 1.48 | ||||
| Ni | 71.45 | 64.27 | 71.46 | 63.49 | 61.90 | 52.16 | 33.02 |
| Sb | 3.56 | 3.16 | |||||
| S | 27.22 | 33.02 | 26.13 | 35.00 | 0.15 | ||
| As | 34.48 | 45.99 | |||||
| Total | 99.12 | 97.29 | 99.89 | 99.15 | 100.25 | 101.31 | 97.29 |
| Fe | 0.014 | 0.005 | 0.004 | 0.235 | |||
| Co | 0.004 | 0.005 | 0.015 | ||||
| Ni | 0.587 | 0.515 | 0.587 | 0.495 | 0.681 | 0.581 | 0.747 |
| Sb | 0.019 | 0.017 | |||||
| S | 0.409 | 0.485 | 0.393 | 0.499 | 0.003 | ||
| As | 0.297 | 0.402 | |||||
| Total | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 | 1.000 |
| Ni/(Ni + Fe) | 1.00 | 1.00 | 0.98 | 0.99 | 0.99 | 1.00 | 0.76 |
| Ni/As | 2.29 | 1.45 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arai, S.; Ishimaru, S.; Miura, M.; Akizawa, N.; Mizukami, T. Post-Serpentinization Formation of Theophrastite-Zaratite by Heazlewoodite Desulfurization: An Implication for Shallow Behavior of Sulfur in a Subduction Complex. Minerals 2020, 10, 806. https://doi.org/10.3390/min10090806
Arai S, Ishimaru S, Miura M, Akizawa N, Mizukami T. Post-Serpentinization Formation of Theophrastite-Zaratite by Heazlewoodite Desulfurization: An Implication for Shallow Behavior of Sulfur in a Subduction Complex. Minerals. 2020; 10(9):806. https://doi.org/10.3390/min10090806
Chicago/Turabian StyleArai, Shoji, Satoko Ishimaru, Makoto Miura, Norikatsu Akizawa, and Tomoyuki Mizukami. 2020. "Post-Serpentinization Formation of Theophrastite-Zaratite by Heazlewoodite Desulfurization: An Implication for Shallow Behavior of Sulfur in a Subduction Complex" Minerals 10, no. 9: 806. https://doi.org/10.3390/min10090806
APA StyleArai, S., Ishimaru, S., Miura, M., Akizawa, N., & Mizukami, T. (2020). Post-Serpentinization Formation of Theophrastite-Zaratite by Heazlewoodite Desulfurization: An Implication for Shallow Behavior of Sulfur in a Subduction Complex. Minerals, 10(9), 806. https://doi.org/10.3390/min10090806






