Abstract
Mismanagement of mine waste rock can mobilize acidity, metal (loid)s, and other contaminants, and thereby negatively affect downstream environments. Hence, strategic long-term planning is required to prevent and mitigate deleterious environmental impacts. Technical frameworks to support waste-rock management have existed for decades and typically combine static and kinetic testing, field-scale experiments, and sometimes reactive-transport models. Yet, the design and implementation of robust long-term solutions remains challenging to date, due to site-specificity in the generated waste rock and local weathering conditions, physicochemical heterogeneity in large-scale systems, and the intricate coupling between chemical kinetics and mass- and heat-transfer processes. This work reviews recent advances in our understanding of the hydrogeochemical behavior of mine waste rock, including improved laboratory testing procedures, innovative analytical techniques, multi-scale field investigations, and reactive-transport modeling. Remaining knowledge-gaps pertaining to the processes involved in mine waste weathering and their parameterization are identified. Practical and sustainable waste-rock management decisions can to a large extent be informed by evidence-based simplification of complex waste-rock systems and through targeted quantification of a limited number of physicochemical parameters. Future research on the key (bio)geochemical processes and transport dynamics in waste-rock piles is essential to further optimize management and minimize potential negative environmental impacts.
1. Introduction
1.1. A global Environmental Perspective on Mine Wastes
Mineral resource extraction and ore processing operations around the world produce significant amounts of waste, predominantly wastewater and non-profitable solid residues, i.e., waste rock and tailings. While technical innovations in mining and processing techniques have increased the overall efficiency of extraction, the great majority of raw material moved to access mineral ores is still discarded as waste (e.g., up to 99% for precious metals). As a result, several gigatons of mine waste are produced around the world each year [1,2], and this number is expected to grow as increasingly lower-grade and larger-scale deposits are being mined to keep up with exponentially growing global demand for mineral resources.
In the absence of economic incentive or technical ability to re-utilize and re-valorize mine wastes [3,4,5,6], they are often stored on-site indefinitely. The impacts of these mine wastes, even when properly managed in engineered storage facilities, can be wide-ranging and include ecological, hydrological, geotechnical, climatic, and environmental aspects pertaining to the quality of natural habitat, i.e., the atmosphere, ground- and surface-water, and soils. High-profile mine waste catastrophes are well-known by the public (e.g., tailings dam failures in Canada [7], Hungary or Brazil [8]), but deterioration of environmental quality from mining waste more often occurs gradually [9,10] and can even linger unnoticed for years or become apparent until decades after mine closure. An example thereof is acid rock drainage (ARD); the weathering of sulfidic mine waste that leads to acidic drainage with high metal concentrations [1,11] (the term acid mine drainage is increasingly substituted by acid rock drainage to indicate that acidic drainage can originate from sources other than mines). Acid rock drainage is an environmental problem of global scale and deterioration of water quality from acid rock drainage which may persist for decades to millennia.
To minimize detrimental environmental impacts from mine wastes, mine sites are required to abide by legislative environmental quality standards during the entire mining cycle, i.e., from exploration and development to decades past closure [12,13]. To this end, effective long-term waste management strategies need to be developed [14,15]. Wastewater quantity and quality predictions are a critical component of these strategies, as some form of treatment is usually required before wastewater can be released to receiving downstream environments, and most wastewater treatment requirements are long-term. Drainage from on-site mine waste critically contributes to wastewater quantity and quality, and the processes underlying mine waste weathering and drainage in practical, industry-relevant settings thus need to be quantitatively understood.
Prediction of drainage quantity and quality from mine wastes requires knowledge on the local geology and weathering climate, as well as the mine waste’s mineralogy, geochemistry, hydrogeology, et cetera. While local weathering conditions and the geology at a mining site are typically well-known, the properties of waste materials relevant to environmental management are usually unknown at early mine development stages and can hardly be determined a priori from theoretical calculations or by extrapolation of laboratory test results [13]. There can be a significant lag time between waste placement and onset of ARD, the composition of waste varies from mine site to mine site and can be highly heterogeneous even within sites, especially in complex geological settings. Therefore, the prediction of mine wastewater quality and quantity poses a major challenge for scientists and practitioners at sites around the world.
1.2. Waste Rock as Unique Class of Mine Waste
Mine waste rock and tailings are typically the two major waste types at mine sites, regardless of the mineral commodity (e.g., coal or base/precious metals), deposit type or extraction method (surface or open-pit mining versus in-situ or underground mining) [14]. Waste rock—distinct from other overburden spoil—consists of excavated low-grade bedrock that has been transported away to access profitable ore and is typically composed of relatively coarse, granular broken rock in the size range of sands to boulders. In turn, tailings are a composite slurry of (process) water and finely ground residuals that remain after ore comminution and beneficiation, which can contain secondary precipitates and processing reagents such as blasting agents or extraction chemicals [16]. For many types of ores, waste rock can behave in a geochemically contrasting way when compared to tailings, due to the following critical differences between tailings and waste rock:
- The finer-grained nature of tailings materials compared to coarser-grained waste rock may yield elevated exposed mineral surface area (which can, depending on the mineralogy, increase geochemical reaction rates), whereas the wider particle size range and textural properties of waste rock give rise to quite unique (non-uniform) hydrodynamic behavior, and,
- Storage practices for waste rock and tailings materials create distinct conditions that alter the controls of certain geochemical processes and physical transport mechanisms. Namely, waste rock is mostly placed in tall stockpiles that are porous, hydraulically unsaturated, and therefore relatively exposed to atmospheric conditions (i.e., mostly oxic environments) [16]. In contrast, tailings slurries are often pumped into tailings ponds, where particulates settle under limited ambient exposure (i.e., fully saturated, inundated tailings that can exhibit sub-oxic, reducing conditions [17], although tailings may also be stored as backfills or dry stacks).
At the same time, similar minerals, geochemical reactions, and physical transport processes can occur in waste rock and tailings: the conceptual hydrogeochemical model is often comparable for waste rock and tailings [2]. The dimensions of industrial-scale waste-rock piles and tailings facilities are also comparable (i.e., hundreds of tons of material): once in place, both materials are prohibitively expensive to move and therefore are practically stored indefinitely. In this review, we will discuss a conceptual hydrogeochemical framework that largely applies to both types of mine waste, but, in our discussion, place emphasis on waste rock.
1.3. Scope of This Review
In recent years, the understanding of processes controlling mine waste dynamics has critically increased due to, e.g., breakthroughs in mineral characterization at nanoscales and in-situ geophysical characterization to long-term field studies and applications of big data and mechanistic numerical models. This work presents an overview of the key hydrogeochemical and physical processes relevant to water quantity and quality from mine waste rock and ultimately sustainable wastewater management. Novel insights into the geochemical and mineralogical characteristics of waste rock, the in-situ assessment of mass- and heat transfer processes in large waste-rock piles and emerging applications of advanced reactive-transport modeling are presented. Previous reviews on select aspects of mine wastes are listed in Table 1. Certain processes discussed in this work also receive attention in the context of hydrometallurgy and bioleaching/biomining, where often the opposite objective is targeted, i.e., acceleration of mineral dissolution to mobilize and recover valuable metals (e.g., in heap leaching [4,18,19,20]) rather than stabilizing wastes to prevent mobilization (e.g., through covers and barriers [4,21,22,23]). This review is structured as follows: major geochemical mobilization reactions (i.e., oxidation and dissolution) and relevant attenuation processes (i.e., sorption and secondary mineral formation) are introduced in Section 2. Section 3 outlines relevant physical transport processes in waste-rock piles and their parameterization in heterogeneous systems. In Section 4, we discuss the couplings between physical and chemical processes and upscaling phenomena, as well as the role of numerical modelling in resolving such couplings for practical long-term predictions.

Table 1.
Reviews on various aspects of mine waste rock: weathering mechanisms, microbial interactions, characterization of physicochemical (bulk) properties, management, and reclamation.
2. Geochemical Processes in Mine Waste Rock
The exposure of previously buried geological material to atmospheric conditions triggers oxidation reactions and allows for water to infiltrate and percolate the waste rock (i.e., drainage) which can transfer dissolved solutes into the environment. In this section, major geochemical processes that affect drainage chemistry are discussed. On the broadest level, mine waste drainage pH is determined by the balance between acid-producing and acid-neutralizing (buffering) reactions, whereas solute concentrations are controlled by their waste-rock grade and solubility [16,51,54,57]. In addition, attenuation processes such as adsorption and secondary mineral formation [2] may reduce the mobility of solutes through (temporary) internal retention in the waste rock, as discussed below.
2.1. Acid-Producing Reactions
2.1.1. Metal-Sulfide Mine Waste
The majority of exploited chalcophile-metal ore deposits (e.g., Cu-, Zn-, Pb-bearing ores) are sulfide-type. While many mined chalcophile-metal deposits have undergone significant oxidative alteration of the ore zones prior to mining, sulfide minerals can remain abundant in the generated waste. Of the sulfide minerals in waste rock, pyrite (FeS2) is typically the most abundant. Other major phases include chalcopyrite (FeCuS2), covellite (CuS), sphalerite (ZnS), pyrrhotite (FeS), arsenopyrite (FeAsS), and galena (PbS). Because of their economic and environmental importance, the crystal structures, chemical compositions, physical properties, and phase relations of major sulfides have been well-established and previously reviewed [58]. The oxidative dissolution of a generalized metal sulfide (MexSy) is illustrated in Figure 1a: this reaction consumes water and oxygen and produces protons, metal and sulfate ions, and heat.
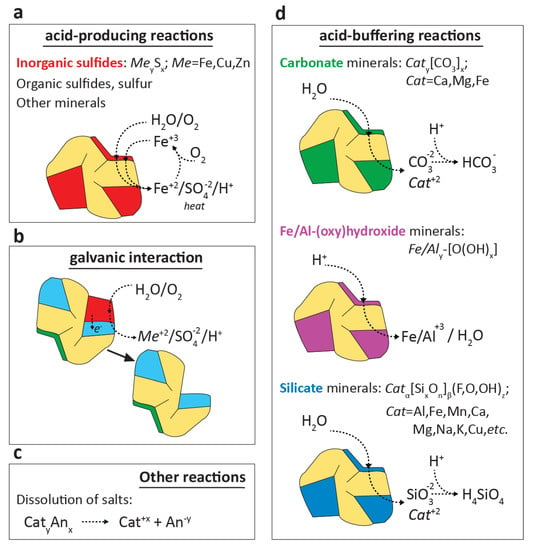
Figure 1.
Schematic grouping of key geochemical reactions (not balanced) that control mine waste-rock drainage quality. The oxidative dissolution of sulfide minerals is the major acid-producing reaction (a), which has been extensively studied for pyrite and other major sulfides. Galvanic interactions (b) can promote the preferential dissolution of sulfide minerals (Section 2.5). Additional reactions (c) can introduce metals and other solutes into the waste rock drainage without directly affecting drainage pH. The dissolution of carbonates, Fe/Al-(oxy)hydroxides, and silicates (simplified, generalized stoichiometries; (d) consumes protons and thereby perform a net-buffering action.
The reaction pathway for FeS2 oxidation has been studied extensively [26,59,60,61] and occurs via an electrochemical mechanism wherein electron exchange takes place on specific surface sites of the mineral [62]. Its overall congruent dissolution can be simplified by the equation:
Under oxic conditions, dissolved ferrous Fe2+ rapidly oxidizes into ferric Fe3+ which can act as an additional oxidant:
Due to the capability of Fe3+ ions to break metal sulfide bonds more effectively than protons [59], the Fe3+ reaction speeds up FeS2 oxidation by orders of magnitude and forms an important (auto)catalytic feedback (Equation (3) versus Equation (1); Figure 1a). Because FeS2 is naturally abundant in many chalcophile metal ores, Fe3+ also serves as an oxidant to other metal sulfides, e.g.,:
The kinetics of abiotic sulfide oxidation by Fe3+ allow for much faster sulfide oxidation than by oxygen alone, so that the above equations for sulfide oxidation with Fe3+ are more commonly used than those with oxygen, e.g.,:
Finally, ferrous Fe3+ is very insoluble under oxic and near-neutral conditions and readily precipitates as Fe3+-(oxy)hydroxide to release additional acidity:
The overall reaction equation for pyrite oxidation (combining Equations (2), (3), and (10)) is therefore often summarized as:
Despite the catalytic effect of Fe3+ on sulfide oxidation, the rates of the abiotic reactions described above are relatively slow, especially at low pH [16,63]. Sulfide oxidation reactions are catalyzed by a variety of Fe- and S-oxidizing bacteria that metabolically tap into the energy released during Fe and S oxidation [64]. Major chemolithotrophic bacteria involved in sulfide oxidation reactions include both acidophilic and neutrophilic iron and sulfide oxidizing species, e.g., ferrooxidans and thiooxidans species in the Thiobacillus, Leptospirillum, and Ferrobacillus genera [33,65,66]. The role of microbial sulfide mineral oxidation in controlling mine waste weathering rates has been well-established and the geomicrobiology of mine wastes previously reviewed [30,33,67,68] (Table 1).
Because oxidation of sulfidic minerals involves the transfer of many electrons from each sulfur atom to an aqueous oxidant, various intermediate sulfur species can exist (i.e., polysulfides, thiosulfate, and elemental sulfur) [59,69,70,71]. For major sulfides, (a)biotic oxidation pathways, intermediate reaction products, and corresponding kinetics have been characterized in a variety of settings, i.e., under ambient conditions versus at elevated temperatures to optimize leaching [27,72,73,74,75,76,77,78,79]. Various kinetic models exist for pyrite oxidation [2], but the oxidation mechanisms and kinetics for less-abundant sulfides remain comparatively underexplored (e.g., molybdenite (MoS2) [72,80]). The exact reaction mechanisms and kinetics of sulfide oxidation vary with weathering conditions (e.g., as shown for pyrite [75,81,82,83]). Yet, (hydro)metallurgical and geochemical studies often reveal similar overall rate dependencies (e.g., on dissolved oxygen and Fe concentrations or mineral surface area) and may deploy comparable kinetic models [84,85,86].
Metal sulfide minerals are rarely pure, especially in waste rock: e.g., Fe, Cu, or As, may be readily substituted by a range of (trace) metals in solid-solutions: As, Co, and Ni can be present in pyrite with up to several weight percent and sulfosalts typically contain a range of metalloids including As, Sb, Bi, and Se. Mineralogical impurities have a demonstrated effect on abiotic weathering kinetics [87], and even though microorganisms in mine waste appear quite tolerant to otherwise harmful metals [88], impurities may also affect biotic oxidation rates through inhibitive effects or shifts in community structure and functional diversity [89]. Finally, select other non-sulfur minerals may also oxidize to produce acidity, e.g., selenide, arsenide, telluride, and antimonide minerals. Due to their natural scarcity (typical abundances are <‰ of that of S), these metalloids tend to occur at trace levels in sulfides rather than as distinct phases [90] and are usually not relevant to the overall acid-producing capacity.
2.1.2. Coal Mine Waste
In addition to metal-sulfides waste rock, coal mine wastes can also be a major source of ARD [91,92,93,94,95,96,97]. Sulfur can occur in coal in three main forms: (i) inorganic sulfides, including authigenic minerals such as detrital pyrite, (ii) organically bound sulfur, including mercaptan, (di)sulfide and heterocyclic compounds, and (iii) inorganic sulfates, particularly in weathered coals [98,99]. Because of the relative abundance of inorganic sulfide compared to other forms of sulfur in coal waste rock, acidity production is typically attributed to pyritic material, even though pyrite concentrations in coal vary regionally [100,101,102]. While undesirable for the prevention of coal waste drainage acidification, oxidative de-pyritization (or desulfurization) as a major coal ore beneficiation objective has been extensively studied [103,104], including pathways [105] and kinetics [106,107,108].
An important difference between metal ore wastes versus coal wastes is that metal ore waste typically contains negligible organic carbon, whereas coal wastes can have several wt% organic carbon [93,98]. This organic material can, in the aqueous phase, subsequently ligate aqueous solutes and mineral surfaces and thereby alter the mobility of metals in coal waste-rock drainage, depending on the nature of the ligands and minerals (see Section 2.4.1). Organic matter may oxidize to induce oxygen depletion and alter the Fe2+/Fe3+ equilibrium (Equation (2)) as well as those of other redox-sensitive elements (e.g., Mn, As, S) [109]. Subsequent shifts in the microbial community structure to favor Fe and S reducers [110,111] may lead to net alkali generation (i.e., acid-neutralization) and in fact, certain acid–rock drainage prevention or remediation strategies rely on the purposed addition of organic matter to induce oxygen depletion [112,113,114].
2.2. Acid-Buffering Reactions
When present and abundant in waste rock, the dissolution of carbonate minerals (e.g., calcite, dolomite, ankerite), Al- and Fe-(oxy)hydroxides (e.g., gibbsite, ferrihydrite, respectively) and silicate minerals (e.g., feldspars, chlorites, smectites, micas, and amphiboles) consumes protons and introduces alkalinity that offsets acidity produced by sulfide oxidation [2,16]. The following (simplified) equations illustrate these reactions:
The acid-neutralizing dissolution reactions of carbonates, oxides and silicates accelerate at decreasing pH, in accordance with their solubility, creating a so-called buffering sequence for acid–rock drainage [16,115]: carbonates buffer acidity until they are typically depleted at pH < 6, Al- and Fe-(oxy)hydroxides dissolve to buffer acidity at pH < 4–5 and pH < 3–4, respectively, and the dissolution of aluminosilicates only significantly contributes to acid-buffering capacity at pH < 3. Carbonate dissolution neutralizes protons through the carbonate equilibrium, but dissolution of Fe-rich carbonate (e.g., siderite (FeCO3)) also introduces dissolved ferrous Fe that upon oxidation and hydrolysis (Equation (10)) releases surplus protons, reducing the net-neutralizing action [2,46]. Acid-buffering minerals dissolve simultaneously alongside progressing sulfide oxidation when they are naturally co-located in the waste-rock matrix [115]. In addition, they may also be intentionally mixed, blended, or added to sulfide-rich waste material as an active or passive acid–rock drainage remediation strategy [116,117,118].
A wealth of information is available on the molecular mechanisms, kinetics, and pH-dependencies of dissolution reactions of carbonate minerals [119,120,121], Fe-(oxy)hydroxides [122,123], and silicate minerals commonly encountered in waste rock [124,125,126]. The kinetics of carbonate dissolution are typically rapid compared to sulfide oxidation (i.e., far from equilibrium [127]), whereas the dissolution of silicates is orders-of-magnitude slower [2]. This introduces important ramifications as to the timescales required for effective acid-neutralization, for instsance when transport times are fast or experimental durations short (e.g., static tests; Section 2.6.1). In addition to consuming protons, dissolution of carbonate, oxide, or aluminosilicate minerals introduces additional solutes to a leachate, including Ca, Mg, Mn, Al, and Fe, and possibly metal impurities if present at considerable levels [128,129]. Mobilization of such solutes and its effect on drainage quality must be considered if these neutralizing materials are to be employed to prevent or remediate acid drainage [13]. Similar to sulfide oxidation, mineralogical heterogeneities (Section 2.5) strongly affect the efficacy of acid-neutralizing reactions. In contrast to sulfide-dominated acid-production, acid-neutralization can be governed by many minerals: prediction of the extent and timing of neutralization therefore requires characterization of the complete mineral assemblage.
2.3. The Geochemistry of Neutral Drainage
While the environmental impacts of ARD have received extensive attention, neutral to alkaline drainage can also compromise water quality: neutral rock drainage (NRD), also referred to as contaminated neutral drainage (CND; [129,130]) or metal leaching (ML; [131,132]), is the mobilization of hardness (mostly Ca and Mg), major ions such as sulfate and chloride, metal(oid)s, and other contaminants that are mobile under near-neutral, non-acidic conditions. Most metallic cations show higher solubility in acidic conditions, such as Cu, Ni, Zn, Co, and Mn [133], but some can remain in elevated concentrations at near-neutral values, mostly because their oxyhydroxides precipitate at pH between 6 and 10. Oxyanionic elements such as As, Se, and Mo exhibit elevated mobility at near-neutral pH, which has been demonstrated in a variety of hornfels, carbonate [126], and sedimentary [134] waste-rock types. Cations and oxyanions generally show inverse behavior in mine drainage in the sense that cations tend to adsorb or precipitate in near neutral and alkaline conditions, while oxyanions tend to do more so under (slightly) acidic conditions (see Section 2.4.1). Despite a growing awareness of its potential environmental impacts, there is currently no standardized definition of neutral mine drainage [132]. Table 2 lists examples of reported NRD cases, illustrating how neutral drainage composition can be widely different across sites (slightly acidic versus alkaline, and negligible versus elevated concentrations of sulfate, metal(loid)s, and other (non-metallic) contaminants). NRD from waste rock can arise in different situations:
- (i)
- Sulfide oxidation in the presence of sufficient acid-buffering or weathering of non-acid generating minerals (i.e., low-sulfide waste rock such as carbonates and silicates [135]), dissolution of salts [136];
- (ii)
- Insufficient treatment of ARD (e.g., abandoned mine sites using passive ARD treatment), where the pH is successfully increased to near-neutral but certain contaminants remain present at elevated concentrations;
- (iii)
- Within reclaimed ARD-generating mine wastes, where the rate of acid generation is decreased to levels that can be buffered by neutralizing minerals, but still allows for the leaching of metals.
Contaminant loads in neutral drainage can be controlled by their occurrence in sulfidic minerals as well as carbonate or silicate phases, or through the dissolution of (secondary) salts and oxides (Figure 1). For instance, elevated As levels in high-alkalinity drainage have been mostly attributed to the oxidation of arsenopyrite or As-bearing pyrite that had been neutralized [137,138] but may also originate from natively As-rich carbonate phases in the absence of Fe-oxides [128]. With the increased interest in the development of rare earth element (REE) mines, there is an increasing interest in REE geochemistry and ecotoxicity [139,140,141]. REE-bearing minerals can be found in many different geological settings (although not necessarily at economic concentrations), and REEs can be found as trace contaminants in other minerals. Therefore, they might represent an accessory contaminant in mine drainage from non-REE operations. Indeed, they are found in many ARD cases [142,143] but most REE deposits are within geological settings that are not prone to ARD generation, such as pegmatites, carbonatites, and peralkaline igneous deposits, as well as within placers and clays ([144] and references therein). Upon their release in near-neutral mine waters, REE concentrations are mostly controlled by secondary precipitation and sorption phenomena (Section 2.4) [145,146,147,148]. However, significant knowledge gaps remain as to the fate of REE released from mine wastes.

Table 2.
Examples of neutral rock drainage (NRD) chemistries reported at different mine sites around the world.
Table 2.
Examples of neutral rock drainage (NRD) chemistries reported at different mine sites around the world.
| Antamina, Peru [128] | Hitura, Finland [149,150] | Lac Tio, Canada [130] | Beaver Brook, Canada [151] | Greens Creek, United States [152] | Giant Mine, Canada [153] | |
|---|---|---|---|---|---|---|
| pH | 6.5–8.5 | 6.1–7.0 | 6.5–7.5 | 5.7–8.6 | 6.5–8.5 | 6.7 |
| Ni (mg/L) | N/R | 0.2–14.3 | 0.1–8.8 | N/R | 0–1 | 0.029 |
| Zn (mg/L) | 0.1–80 | 25–660 | N/R | N/R | 0–150 | 0.027 |
| Mn (mg/L) | 0.001–0.2 | 4.7–8.9 | N/R | N/R | 0–35 | 0.446 |
| Co (mg/L) | N/R | 0.05–7.2 | N/R | N/R | N/R | <0.007 |
| As (mg/L) | 0.001–1.0 | N/R | N/R | 0–2.3 | 0–0.03 | 4060 |
| Se (mg/L) | 0.001–0.2 | N/R | N/R | N/R | 0–0.02 | <0.03 |
| Sb (mg/L) | 0.001–0.2 | N/R | N/R | 0–26 | 0–0.06 | 11.9 |
| Mo (mg/L) | 0.0–1.0 | N/R | N/R | N/R | 0–0.02 | 0.07 |
| SO4 (g/L) | 0.1–2 | 2.1–5.2 | 0.1–3.5 | 0.075–0.9 | 2–8 | 0.5 |
| Ca (mg/L) | 50–600 | 200–450 | 10–70 | 9–231 | 400–800 | 313 |
N/R: not reported.
2.4. Attenuation Processes
Attenuation processes can retain solutes in the waste-rock matrix and thereby influence drainage quality [51,115,154]. Solute-specific attenuation through adsorption or secondary mineral formation can lead to apparent discrepancies between the elemental composition of bulk waste rock and observed drainage loads. A quantitative assessment of relevant attenuation mechanisms, informed by robust mineralogical analyses and/or geochemical equilibrium modeling, can help predict such discrepancies and optimize waste rock management.
2.4.1. Adsorption
Geochemical adsorption refers to the reversible attachment of aqueous solutes to mineral surfaces (schematic in Figure 2a). While adsorption is highly surface- and solute-specific, the general relevance of adsorption for mine waste drainage quality has been widely acknowledged [2]. Field and laboratory waste-rock studies have demonstrated that sorption can be a dominant attenuation mechanism for various mine waste-relevant solutes, from metal cations [155] to metal(oid) oxyanions [128].
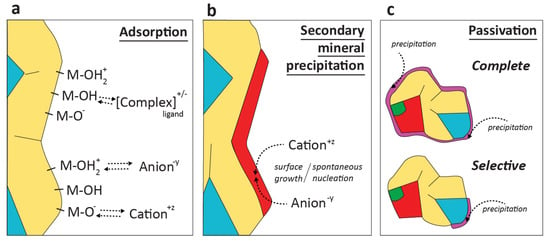
Figure 2.
Schematic of key geochemical attenuation processes in mine waste rock: adsorption (a) and secondary mineral formation (b). Through adsorption, (hydrated) ionic or ligated solutes and complexes adsorb to mineral surface groups (-M-(x)) through covalent bonding or electrostatic interaction. Secondary minerals form on mineral surfaces or spontaneously in solution through (co-)precipitation of cationic and anionic aqueous solutes. The occlusion of mineral surfaces by secondary mineral precipitation is referred to as passivation or armoring (c), discussed in Section 2.5.
Adsorption can involve relatively strong covalent binding (inner-sphere) or weaker electrostatic attraction between aqueous ions and oppositely charged mineral surfaces (outer-sphere). In both cases, adsorption is governed by the solute’s aqueous speciation as well as characteristics of the mineral surface (e.g., pH-dependent surface charge). The aqueous speciation chemistry for major inorganic ions in mine waste drainage has been previously reviewed (e.g., for Cu [156] and Zn [157]). Because most solutes present in mine drainage have an aqueous speciation that is strongly dependent on the solution chemistry, and because the surface characteristics of minerals present at a site may equally vary with the drainage type (i.e., acidic versus neutral versus alkaline), the many geochemical reactions underlying adsorption typically require modeling to be resolved and quantified. Geochemical equilibrium models, informed by thermodynamic and kinetic parameters for these reactions, are frequently used to study and predict net-attenuating effects under practice-relevant conditions [132]. However, even with thermodynamic databases supplying equilibrium or stability constants for aqueous complexation and hydrolysis reactions, the complete aqueous speciation of mine drainage may be challenging to predict, in particular for complex solutions that seasonally vary [115]. Evolving solution chemistries can induce desorption reactions, whereby previously adsorbed solutes may be re-introduced into the drainage [2]. Even though Lewis acid–base interactions and aqueous complexation reactions for major solutes are part of established geochemical equilibrium models, relevant aqueous ligands and potential competitive solutes need to be experimentally determined to inform such models. This may be especially challenging for e.g., coal waste-rock drainage [158,159] where a variety of organic acids can occur. Furthermore, significant temperature variations can occur within waste-rock piles (Section 3.3) and the ionic strengths of waste rock leachates can be unusually high, so that these aspects must be considered in aqueous speciation modeling.
The modes of surface binding are documented for major waste-rock solutes and detailed adsorption models exist for various mineral surfaces (e.g., multi-layer models, charge-distribution multi-site complexation model [160]). Yet, the application of quantitative sorption models in drainage predictions is scarce. This may be because the sorption properties of less-abundant solutes remain poorly studied (e.g., Mo [161]) and stability constants deduced in laboratory studies may not be representative for variable field conditions. Furthermore, the data required to inform sorption models, i.e., mineral surface characteristics such as sorption-site density or solution chemistry, is typically unavailable (or semi-quantitative at best) for large, heterogeneous waste-rock piles. Finally, the surface properties of waste rock may evolve as weathering progresses, e.g., through precipitation of amorphous secondary Fe-oxide coatings with elevated sorption capacity. As a result, adsorption modeling for waste rock often relies on estimated parameters, adoption of synthetic idealized phases (e.g., hydrous ferric oxide [128]), or extrapolation of generalized behavior (the Irving-Williams series) in stable drainage types. Adsorption is often considered a precursor for secondary mineral formation (Section 2.4.2) but the contribution of adsorption versus that of secondary minerals to overall attenuation remains to be quantitatively resolved.
2.4.2. Secondary Mineral Formation
Secondary minerals in waste rock are considered those minerals that form after the waste material is disposed (Figure 2b), i.e., distinct from primary minerals and oxidation products native to the excavated (ore) material. Major secondary mineral phases in mine waste have been reviewed previously [2] and include (oxy)hydroxides, (hydroxy)sulfates, (hydroxy)carbonates, and phosphates, or arsenates, depending on the waste-rock primary mineralogy. Specific secondary phases encountered at many mine sites include Fe-oxides (e.g., ferrihydrite, goethite, and lepidocrocite), the Fe-sulfates jarosite and melanterite, as well as gypsum (CaSO4) [2,54,115].
Secondary mineral precipitates are typically composed of different polymorphs of variable hydration: water-soluble salts may initially precipitate as hydrated or hydroxy-phases but recrystallize into more stable forms as the precipitates age, accelerated by evaporation and drying of the waste rock. The physical and hydrological conditions of waste-rock piles (size, particle size, hydraulic retention time; discussed in Section 3) have a strong impact on the geochemical water-rock interaction and therefore occurrence of secondary minerals. Secondary minerals often occur as distinct rims or coatings on weathered host particles, but their occurrence is not necessarily related to the waste-rock composition in their direct vicinity: mixing of different drainage types within heterogeneous waste-rock piles [162] (Section 3.1) or generally rapid infiltration rates compared to slow precipitation rates may facilitate secondary mineral precipitation seemingly unrelated to the local waste-rock composition [163]. While certain secondary phases readily precipitate (e.g., Fe-(hydroxy)oxides [2,16]), others may take decades to precipitate and crystalize to detectable levels [115]. The bulk mineralogy and drainage pH of waste-rock piles can indicate whether certain secondary mineral classes are likely to occur (e.g., secondary phosphates and carbonates are unstable at drainage pH< 4 [164]; secondary phases such as scorodite may be stable even under strongly acidic conditions [165]. Thus, secondary minerals typically exhibit a widely different stability under oxic versus anoxic or acidic versus neutral drainage conditions [2]. Their controls on long-term drainage quality may thus be difficult to quantify for heterogeneous waste-rock piles with seasonally and spatially variable drainage signatures. For instance, re-dissolution of secondary minerals under gradually acidifying drainage conditions or due to the reductive dissolution of Fe-oxides, have both been invoked to explain spikes in loading rates on the timescales of years [115].
Secondary minerals are often qualitatively inferred from chemical equilibrium modeling (i.e., mineral saturation indices) rather than unequivocally identified analytically [115,165]. Yet, mineral stability constants obtained in controlled laboratory experiments may not apply under field conditions, and secondary mineral occurrence is dictated by their precipitation kinetics more than by geochemical saturation (e.g., slower crystal growth rates occur under low degrees of saturation whereas high precipitation rates may be sustained by spontaneous nucleation at high oversaturation). The identification, let alone quantification, of secondary minerals in waste rock, e.g., with X-Ray diffraction (XRD), remains challenging for dispersed and poorly crystalline phases. Yet, even secondary phases with low overall bulk abundance can be substantial attenuating sinks: e.g., the scarce but rapidly precipitating wulfenite (PbMoO4) has been shown to present an important control on Pb and Mo drainage levels [161,166,167]. For successful drainage management, it is thus critical that the secondary mineral assemblage is quantified and monitored over time.
2.5. Mineral Reactivity
The generation of acidic and metal-bearing drainage from waste rock is the result of mineral-water interaction, and thus the nature of the fluid and the minerals themselves. The relative resistance to oxidation of common sulfide minerals in mine waste has been discussed: based mostly on field observations, pyrrhotite is considered the most reactive, followed by galena, sphalerite, bornite, pentlandite, arsenopyrite, marcasite, pyrite, and chalcopyrite [45,168,169]. The reactivity of potentially neutralizing minerals also influences mine drainage quality, e.g., calcite is considered to be 180 times more reactive than the most reactive silicate (wollastonite). In addition to its mineral composition (or overall sulfide grade), overall waste-rock reactivity is determined by additional petrographic and mineralogical factors that include crystallinity and morphology [24,170,171], surface defects and heterogeneities, and reduced liberation (i.e., reduced exposure to oxygen and fluids) caused by inclusion in primary minerals (occlusion) or secondary mineral precipitation (passivation or armoring; Figure 2c) [163,172,173,174,175].
Small-scale mineral features of waste rock are typically unknown in practice, since determining them requires more advanced analytical techniques that can be costly to apply to sufficient samples for a realistic assessment [45]. These factors, however, may explain why predictive laboratory testing does not always match field results (Section 2.6.1) [129,176]. For instance, occluded (unliberated) sulfides in silicates are less likely to oxidize, and both chemical and mineralogical acid–base accounting tests may thus overestimate potential acid generation, especially waste that has been exposed over times such that the exposed sulfides have oxidized. Further, sulfide oxidation may be slowed by the formation of secondary mineral rims of Fe-(oxyhydr)oxides, sulfates, or even secondary carbonates such as smithsonite, whereas sulfides that do not contain iron, such as stibnite, may oxidize more rapidly because they do not develop Fe-oxide rims [151]. Such passivation may thus occur selectively on specific minerals but also throughout the waste-rock matrix [177], and extensive precipitation can even reduce pore space and thereby alter the hydraulic properties of waste rock (Section 4). Deliberate sulfide passivation has also been proposed as an active acid drainage prevention technique [178,179], e.g., using metal-organic complex formation and passivation of sulfides [180,181]. Finally, the contact of two sulfidic phases in waste rock can result in electron exchange (galvanic interaction) that causes one phase to corrode more rapidly than the other (Figure 1b). For instance, the leaching of chalcopyrite can be enhanced through association with pyrite [182]. Galvanic interactions can substantially increase the leaching of one or both of the minerals that constitute the galvanic cell: galvanically promoted dissolution has been reported in laboratory and field studies with mine waste rock [163,183,184,185]. Even though mineral occlusion, passivation, and association are known to critically determine waste-rock reactivity [115,186], and techniques such as electron microscopy and automated mineralogy (e.g., MLA, QEMSCAN) now allow for such parameters to be quantified with increasing ease, few waste-rock studies and drainage prediction models have given consideration to mineralogical and petrographic aspects (e.g., through refinement of kinetic rates) [187,188,189,190], given the cost of a representative assessment at full scale (Section 2.6 and Section 3.4).
In general, the grain size of primary minerals is inversely correlated with reactivity, and thus finely-crushed tailings are expected to be more reactive in terms of sulfide oxidation and carbonate dissolution than coarse waste rock fragments. On the other hand, larger pore spaces and unsaturation of waste rock may result in a higher ingress of oxygen and periodic wetting and drying of mineral surfaces, which may increase reactivity (Section 3.2). The nature of the water interacting with the waste rock also affects reactivity. Oxygen-rich, slightly acidic meteoric water interacting with recently blasted waste rock will encounter freshly broken minerals surface, and possibly highly reactive mineral dust from blasting. Porewater reaching the lower portions of a waste-rock pile may be oxygen depleted, and Fe3+ may act as an oxidant under acid conditions in the absence of dissolved oxygen (Equation (3)). Under anoxic conditions at the bottom of a pile, or if the waste rock is submerged, Fe-(oxyhydr)oxides are susceptible to reductive dissolution, resulting in release of elements of concern that were attenuated by adsorption or co-precipitation. Waste rock submerged in a marine environment is unlikely to generate acidic drainage, given the strong buffering capacity of seawater, although the availability of ligands may increase the solubility of some metals [191].
2.6. Characterization of Bulk Waste-Rock Reactivity
While the geochemical reactions in weathering waste rock are universal on a molecular level, there is typically large variability in the weathering conditions and waste-rock composition and grain sizes across mine sites. As a result, sulfide oxidation and bulk weathering rates reported from laboratory and field experiments vary by orders-of-magnitude across sites (Table 3). Because it is virtually impossible to resolve all molecular-scale mineralogical heterogeneity, waste-rock reactivity or weathering rates are typically presented through representative bulk parameters such as drainage loads (i.e., net sulfate leaching) or oxygen consumption or heat production rates [192,193,194]. Similarly, the estimation of the acid-producing versus acid-neutralizing nature of waste-rock material is often based on bulk laboratory tests (static testing or acid–base accounting (ABA)) rather than (or complementary to) microscale mineralogical analyses.

Table 3.
Selection of waste-rock weathering rates reported for mine sites around the world, including field studies, laboratory experiments, and numerical modeling.
2.6.1. Static and Kinetic Testing
Static tests aim to quantify the acid-generating and acid-neutralizing capabilities of bulk mine waste materials [45]: paste-pH, acid-base accounting (ABA), and net acid generation (NAG) tests are common waste-rock classification methods. The detailed procedures, advantages, and shortcomings of static tests have been reviewed [45,46,132,208]. In brief, ABA is based on a determination of the total sulfur, sulfide, and carbon content of a waste-rock sample. The acid-producing potential (AP) is calculated by multiplying the sulfur or sulfide content by a stoichiometric factor [132], typically differing with sulfide reactivity (Section 2.5) [209,210]. Acid-neutralization potential (NP) is typically determined by titration with HCl (Sobek method [132,211,212]), although such NP determination has been repeatedly revised [213,214,215] and equally deploys correction factors [45,216,217,218]. Both AP and NP typically have units of kg CaCO3 per ton waste rock and ultimately aggregate unresolved mineralogical heterogeneity into bulk waste-rock properties. From AP and NP, a net-neutralizing potential (NNP = AP–NP; kg CaCO3/ton waste rock) or neutralizing potential ratio (NPR = NP/AP; unitless) can be calculated. Although different, site-specific cut-off values to assess acid-production risks are used, it is often assumed that NNP < −20 kg CaCO3/ton or NPR < 1 indicate potential net acid-generating material, whereas material with NNP > 20 kg CaCO3/ton or NPR > 3 is non-acid generating [132]. Considerable uncertainty remains associated with these NNP or NPR classification criteria and unexpected drainage acidification in high NPR materials, as well as non-acidic drainage from high-sulfide materials [219], have been reported. Being practical and cost-efficient, ABA analyses continue to be widely used and optimized, e.g., through (i) corrections for oxidation steps that dissolve organic material, (ii) corrections for samples with abundant forms of S other than sulfide [220], and (iii) improved determination of silicate NP [221,222].
Static testing is often complemented by kinetic testing to overcome the discrepancy between conditions for (short-term) laboratory testing on crushed, sieved samples and the long-term behavior of coarser waste rock observed in the field. Examples of kinetic tests include laboratory humidity cell tests with artificial wetting- and drying cycles [129,132], column tests [68], as well as field cell tests of a variety of sizes [128,177]. The benefits and limitations of humidity cell tests have been previously reviewed [176,223]. Kinetic tests can be used to predict long-term weathering dynamics, investigate specific geochemical processes, and test possible reclamation scenarios [175]. While long-term kinetic testing (i.e., months to years) is comparably expensive, kinetic tests are generally performed after static testwork has identified critical or uncertain samples. Field-based kinetic tests range from small (<1 m3) to large-scale (thousands of m3) experiments with waste-rock barrels, pads, or test piles, and although field tests can account for site-specific climatic conditions, they usually cannot be established until advanced stages of mine operation.
Several multi-scale field research programs with a combination of static and long-term kinetic tests have been conducted/completed recently, e.g., at the Aitik [195,200,224], Diavik [225,226,227,228], and Antamina [115,166,177,199,229,230] mines. These programs have investigated waste-rock weathering dynamics and drainage quality evolution in small-scale laboratory tests, meso-scale columns, and up to full-scale field experiments. A key advantage of such programs is the opportunity to investigate geochemical reactions and transport processes under quasi-controlled conditions across a range of scales, thereby providing a means to verify the reliability and accuracy of static tests for model parameterization (Section 4.2) and practical industrial-scale predictions. Additional multiscale investigations will be crucial to address remaining knowledge gaps, improve our conceptual understanding of waste rock weathering processes, and inform practical drainage quality prediction models.
2.6.2. Macroscale Geochemical Heterogeneity
Full-scale waste-rock piles, especially in complex geological settings [231], may exhibit a high degree of geochemical heterogeneity, arising from the mixing of different waste-rock materials and depositional practices (i.e., push- versus end-dumping [232]). On the one hand, such heterogeneities can complicate the development of predictive water quality models when the location and nature of more reactive waste materials is unknown. However, strategic placement of different materials (e.g., co-location or blending of acid-producing and acid-neutralizing materials) can also be an effective strategy to mitigate acid drainage risks [233]. Geochemical material heterogeneity can cause unpredictable weathering dynamics [115,234,235,236] and representative sampling is critical to characterize the spatial distribution of mineral reactivity that can significantly affect the overall drainage signature of composite systems. Relevant geological, lithological, and alteration units must be sampled relative to the amounts and particle size of each material [45,132]: inadequate sampling can contribute to substantial variability or incorrect assessment of waste-rock reactivity. Sampling and analysis of waste-rock properties or material heterogeneity must be fit-for-purpose to inform the deployed prediction model: generalization and extrapolation from static tests may be insufficient when done in lieu of appropriate kinetic testing, as illustrated by the wide range of weathering rates observed in the field (Table 3).
Thus, bulk waste-rock parameters can be used to aggregate variability in mineralogical composition of waste rock but static test results or bulk weathering indicators may not reflect in-situ sulfide oxidation rates and acid-generation in spatially heterogeneous systems, e.g., when retention by secondary minerals is insufficiently quantified. More accurate predictions of waste-rock drainage dynamics in full-scale systems may be achieved when static tests, detailed mineralogical analyses and targeted kinetic tests are effectively combined.
3. Physical Transport Processes
In addition to the geochemical weathering and attenuation processes discussed above, waste-rock drainage quality and quantities are controlled by the transport of gas, water, and heat through porous waste-rock piles: aqueous transport controls the ultimate export of solubilized solutes from the waste-rock pile, and gas transport can affect weathering rates through controlling oxygen supply required to sustain sulfide oxidation.
3.1. Aqueous Transport
Water flow within waste-rock piles is highly site-specific and varies with climate, (internal) pile structure, and hydrogeological waste-rock properties. At the macroscale (Figure 3), drainage of a waste-rock pile can be described by a water balance [16]:
in which P is precipitation, E is evapo(transpi)ration, R is runoff, G is groundwater exchange, R is reaction, and ∆S is change in internal storage (i.e., infiltration or drainage). Modern waste-rock piles are preferably placed onto relatively impermeable or lined surfaces to minimize interaction with groundwater if present at the site. Further, the highest oxidation rates listed in Table 3 indicate that water consumption by weathering reactions is maximally on the order of 0.01–0.1 kg H2O m−3 year−1, and thus only relevant to the water budget for tall piles stored at very dry conditions (<200 mm year−1).
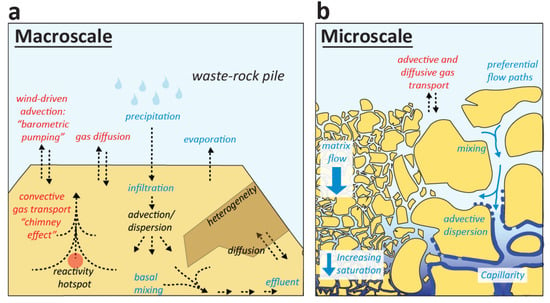
Figure 3.
Schematic illustration of relevant aqueous (blue) and gaseous (red) mass transport processes in waste-rock piles, distinguishing between macroscale (a) and porescale or microscale (b) processes.
Evaporation has been investigated for engineered waste-rock covers [237,238] but less for bare waste-rock piles [239]. Near-surface water and energy balances have been used to estimate evaporation rates for waste-rock piles by combining rainfall gauging, soil-water characteristic curves and suction measurements, in-situ moisture sensors, scintillometry, and eddy covariance measurements or surface heat mapping [240]. Evapo(transpi)ration rates inferred from local meteorological measurements are poorly representative for the quite unique, coarse-textured nature of waste rock and empirical relationships to estimate evaporation (e.g., Penman-Monteith) must be calibrated for such surface properties [241]. Water and energy balancing at various mine sites have indicated that evaporation may be the dominant component (i.e., >70%) of the water balance of waste-rock piles [241,242]. A comparison of twelve waste-rock piles [240] indicated that effective evaporation was lower for bare waste-rock piles compared to covered or vegetated piles. However, evaporation rates from waste-rock piles strongly differ from variability in local climatic conditions (wind speed, insolation [243]), slope aspects, as well as surface waste-rock properties (particle size, roughness, albedo). Further study is required to resolve the controls on evaporative fluxes posed by thermal gradients [239,244] (e.g., quantifying surface heating using infrared cameras) versus those of vapor pressure versus diffusive gradients [245] (e.g., mapping of saturation and the evaporative ‘drying front’ [246]).
Surface runoff from waste-rock piles may be deliberately minimized to avoid erosion on the pile batters, in which case it is typically insignificant for the water balance. Yet, certain reclamation strategies rely on maximizing runoff from compacted (subsurface) cover layers to limit percolation into underlying reactive material [236,247]. Material compaction (e.g., in traffic surfaces) can facilitate surface runoff and ponding, especially under flashy precipitation patterns. Eventually, the non-runoff or evaporated fraction of precipitation will infiltrate the unsaturated waste-rock pile. Downward propagation of precipitation forms a wetting front that becomes drainage constituting the base seepage. Changes in internal water storage can be significant when drainage fronts successively migrate through unsaturated waste rock as a result of seasonal precipitation patterns or during so-called wetting-up phases in newly deposited piles [225,229,248].
At the porescale (Figure 3), the drainage flow regime from a waste-rock pile is the result of infiltration through a typically large range of grain and pore sizes. Particle sizes in waste rock can vary from greater than 1-m boulders to sub-millimeter clay-sized fractions (i.e., wide particle size distributions and porosity ranges) [16]. Such variability must be accounted for conceptual hydrological models aimed at capturing waste-rock flow dynamics and parameters such as hydraulic conductivity can be highly non-uniform across poorly-sorted heterogeneous waste-rock piles [229,249]. Infiltration is often (conceptually) separated into a slower matric flow component, in which water flows under capillary and gravity forces as described by Richards’ equation, and a faster macropore (or non-capillary, preferential) flow component, which is more rapid and channelized [250]. Wetting fronts move according to kinematic velocity under dominantly matrix flow conditions but can travel hundreds of times faster than the average velocity as a result of preferential flow [251]. Preferential flow phenomena have been extensively studied and reviewed [252,253], including in mine waste rock [249,254,255,256].
Tracer tests with internal or externally applied conservative solutes can be used to identify and quantify preferential flow phenomena. Calibration of hydraulic parameters from tracer tests is often preferred over conventional field tests (e.g., infiltrometer, permeameter), which are challenging for large-scale systems and boulder-sized particles. Approaches to describe non-uniform flow in unsaturated porous media exist include advanced dual-domain (dual-porosity, dual-permeability) models that have meanwhile been applied to waste rock [229,230]. While preferential flow will be critical for the prediction of water quantity hydrographs and flushing dynamics, water quality may be dominantly controlled by matrix flow that facilitates longer contact times with finer-grainer particles with elevated reactive surface areas and thus solute mobilization [16]. Finally, freeze-and-thaw cycles may completely alter the hydrological regime in waste-rock piles and can thereby critically affect drainage dynamics [257,258]. The cryohydrogeology of waste-rock piles is strongly coupled to the thermal regime through phase transitions (Section 3.3) and has been described by model approaches that include the Clapeyron equation or Soil-Freezing Characteristic Curves [259]. Thus, waste-rock flow dynamics are highly site-specific and further research is required to optimize and parameterize hydrological models for full-scale waste-rock systems.
3.2. Gas Transport
Oxygen consumption from sulfide oxidation and CO2 production from carbonate dissolution may induce poregas pressures and compositions that differ from atmospheric conditions, particularly in large waste-rock piles with reactive material [194,198,199,260]. In smaller-scale (laboratory) experiments with less-reactive waste rock, poregas variations are typically neglected yet rarely experimentally determined. Gradients in poregas composition and pressure trigger gas transfer through diffusion and advection, respectively (Figure 3), which affect internal poregas distributions and the generally outward transport of CO2 versus inward replenishment of O2. The contribution of dissolved oxygen transport in percolating water is usually negligible (low solubility) [16] and convection as a combined heat and gas transfer mechanism is discussed below.
Molecular diffusion typically dominates gas transport in low permeability waste-rock systems: the controls of oxygen diffusion on sulfide oxidation rates have been estimated from mass-balance calculations (Fick’s laws) [192,194,261], and more advanced modeling of combined gas advection-diffusion with spatially-discretized diffusion parameters [262]. In addition to diffusion, pressure gradients can result in advective gas transport in porous media [263]. Estimates of advective gas transport using e.g., pneumatic heads and Darcy’s law for gas, have shown that barometric fluctuations can be relevant for gas transport particularly through more permeable coarse rubble zones at the bases of waste-rock piles [194,243,264,265].
Both diffusive and advective gas transfer are strongly controlled by the properties of the waste rock, including its permeability, degree of porewater saturation, and spatial heterogeneity therein [194,266]. Various empirical and semi-empirical formulae to estimate diffusivity and permeability from particle size distributions or porosity ranges exist [267,268,269] but these often invoke tortuosity or constrictivity parameters that are poorly defined [266]. Typical effective permeability and gas-diffusivity ranges for waste rock have been reviewed [16] and vary orders of magnitude between and within sites [194]. As a result, gas transfer is usually only quantitatively constrained for waste-rock piles, even though oxygen supply may be rate-limiting in full-scale prediction models. Site-specific field determination of gas transport properties, especially in dual-porosity media, is particularly challenging for full-sized waste-rock systems across large areal extents. Further research is therefore required to assess the applicability and practicality of in-situ sensing or gas injection tracer tests for such parameterization, considering that a quantitative understanding of gas transport limitations can be used to better assess weathering rates and optimize drainage quality predictions.
3.3. Heat Transport
Exothermic sulfide oxidation (e.g., 1000–1500 kJ·mol−1 for pyrite [270]) can cause internal temperatures in waste-rock piles to rise to tens of degrees above average ambient temperatures, e.g., up to 65 °C at some sites [204]. Thermal profiles have been measured in a large number of waste-rock piles [204,226,271,272], often by means of instrumented boreholes [194]. Heat transfer from reactive ‘hotspots’ in waste-rock piles is dominated by convective heat transfer, the basis of the so-called ‘chimney effect’ [206,273] (Figure 3). Convection is the combination of conduction (heat diffusion) and advection (bulk fluid flow); radiative transfer and viscous dissipation are typically assumed negligible in waste rock. Natural convection arises from buoyancy forces in waste-rock piles generated by oxidation reactions that produce heat and alter the density of the poregas (e.g., through water vapor [206]). Temperature gradients caused by internal heating trigger upward convection against the downward diffusive transport of oxygen into a waste-rock pile. Mass transfer estimates and modeling studies have shown that convective heat and gas transfer can sustain high waste-rock weathering rates [204,274]. The onset and strength of convective transport is determined by the temperature gradient (local atmospheric temperature versus the spatial distribution of reactive waste rock [236,273]) and waste-rock permeability. As a result, parameterization of heat transfer properties is not straightforward [16] and effective heat transfer properties vary widely as a function of porewater saturation and porosity [204,236,266,272]. Further work is required to relate heat transfer mechanisms to waste-rock properties and pile construction methods, and thereby help quantify the contribution of convection to overall weathering rates and ultimately drainage quality.
3.4. Physical Heterogeneity
Physical heterogeneity within waste-rock piles induces variations in their hydrogeological properties (water, air, and heat transport). In-situ investigation of temperature, poregas composition, moisture distribution, infiltration, and solute transport can help assess these heterogeneities on practical scales [275,276,277], e.g., by mapping flow channeling or diversion along structural features such as intermittent traffic surfaces or tipping benches. Recent improvements in our ability to measure in-situ properties at increasing resolution have helped characterize heterogeneity for improved drainage quality predictions and numerical models (Section 4.2).
Soil moisture sensing techniques have been recently reviewed [278]. Point measurement techniques provide precise measurements of hydrogeological properties but are challenging to extrapolate to larger areas because of the waste-rock heterogeneities. Previous work on the hydrogeological properties of waste-rock piles have used point measurements of water content, for instance using time-domain reflectometry (TDR) sensors [16,279,280], frequency-domain (FDR) sensors [281], and soil water potential sensors [282,283], or by oven-drying discrete waste-rock samples taken at several depths in boreholes [162,163]. Remote sensing techniques offer larger scales of investigation, but often at the expense of lower resolutions that might be inadequate to discriminate the internal fine structure of waste-rock piles. Nevertheless, electrical resistivity techniques (ERT; [275,281]), ground penetrating radar (GPR; [284,285,286]), electromagnetic induction (EMI; [284,287]), fibre optic distributed temperature sensing (FO-DTS; [288]), or oxygen/hydrogen isotopic signatures [162] have all been applied successfully to investigate hydrogeological dynamics in waste-rock piles. In addition, basal lysimeters are often used to record internal mixing and drainage at the bottom of waste-rock piles [115,280,282]. In addition to direct sensing and measurement techniques, physical and geochemical heterogeneity in waste-rock piles may be reverse-engineered from truck movement and placement records [289]. Ideally, multiple of the aforementioned methods are combined to gain a more comprehensive understanding of the overall hydrogeological behavior of heterogeneous waste-rock piles.
3.5. Coupling Between Geochemical Reactions and Physical Transport
The overall waste-rock drainage from a composite, heterogeneous waste-rock pile often reflects the mixed aggregate signal of individual reaction-infiltration pathways, each resulting from a sequence of interacting processes. The drainage composition arising at each individual pathway results from exposure to a usually wide range of physicochemical properties and the transport time of the infiltrating fluid. Even with a perfect description of the spatial heterogeneity in relevant properties (Section 3.4), prediction of such a fluid composition constitutes a challenging task, due to the coupled nature of the physicochemical processes described above [16,290]. Figure 4 schematically illustrates the different physical transport processes and geochemical reactions controlling waste-rock drainage quality and quantity and how they interact.
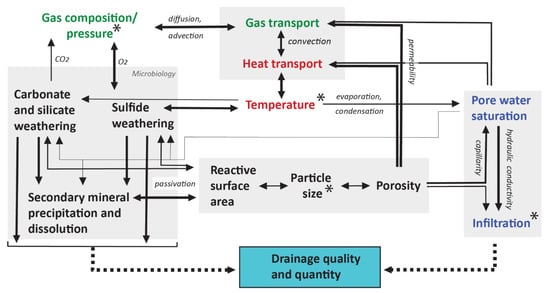
Figure 4.
Schematic illustration of major couplings and feedbacks between geochemical reactions and physical transport processes. Solid arrow lines indicate interactions or a control-hierarchy between different processes, with the width of the arrow being (qualitatively) proportional to the magnitude or strength of the feedback. External controls include that of site meteorology on infiltration, temperature, and gas pressure and that of site geology and communition method on waste-rock particle sizes, as indicated by the asterisks.
A selection of these couplings has been previously investigated [16,205,266]. Many processes and associated feedbacks operate virtually simultaneously. For instance, sulfide oxidation produces acidity that induces carbonate and silicate weathering and secondary mineral precipitation, all of which affect pH and the subsequent progress of oxidation [172]. Sulfide oxidation may undergo orders-of-magnitude acceleration under acidifying conditions, due to a fractional-order rate dependence on proton activity and due to increased oxidation by ferric ion. At the same time, sulfide oxidation is an exothermic reaction: the reaction rates of sulfide oxidation may increase by several factors between every 10 °C increase in temperature, as per Arrhenius’ relation [194]. Temperature also impacts geochemical reactions rates through minor changes in equilibrium and gas solubility constants, and fluid properties such as density and viscosity that can affect transport rates [266]. While some couplings thus enforce controls on magnitudes of percentages or fractions at most, others impose orders-of-magnitude influences. The task at hand is to assess the strength of the coupling, and many feedback mechanisms remain poorly quantified. One example is the cryo-hydrogeological feedback between latent heat, gas, and fluid flow behavior and reaction rates: ice is approximately 3.8 times more heat conductive than water at 0 °C and in cold climates, gas migration through partially frozen waste-rock piles can maintain oxidation reactions and control freeze–thaw dynamics [257,258].
The intricately coupled processes in waste rock can exert both positive and negative feedbacks towards poorer drainage quality (Figure 4). As detailed above, sulfide oxidation releases heat and acidity, both of which accelerate oxidation (Equation (1)) and induce a convective supply of oxygen that can accelerate oxidation (‘chimney effect’, Section 3.3). For such positive feedback loops, small parameter variability can yield significant reinforcing perturbations in long-term predictions. In contrast, drainage quality deterioration may also be lessened by negative feedbacks that decelerate the oxidation process. For instance, waste-rock weathering rates can be lowered by mineral surface passivation and pore clogging, a decreased microbial activity at elevated temperatures, or reduction of oxygen replenishment by mass transport limitations [194]. Finally, while certain feedbacks operate directly or on comparatively short time-scales (e.g., flow channeling and preferential flow), others can require years to become relevant (e.g., progressive dissolution of reactive grain sizes [291,292] or secondary mineral precipitation that affect reactive surface area [290,293] and porosity/permeability [266]).
Processes and their feedbacks can be accounted for in different layers of complexity. Firstly, they are dependent on the molecular-scale to mineral-scale physical chemistry, i.e., passivation, porosity, and grain size evolution. Secondly, large heterogeneous systems can display feedbacks that are controlled by the large-scale structural make-up of the waste-rock pile (e.g., local oxygen content and oxidation rates that vary with infiltrating precipitation fronts through compacted layers). The scale of the studied system and according prediction model thus will determine which processes and feedbacks need to be considered and parameterized.
4. Practical Waste-Rock Drainage Predictions
Prediction of drainage type, amount, and timing is required to reduce or prevent the potential detrimental environmental impacts of waste-rock weathering [132]. Such prediction is typically required already during mine planning/permitting stages, in which full-scale waste-rock storage facilities do not yet exist. Drainage quality and quantity predictions for long-term planning need to integrate a combination of geochemical and physical transport processes, while geochemistry and mineralogy determine the occurrence, abundance, and reactivity of potential solutes and hydrogeology determines their transport and mobility [51], and both are highly interconnected as discussed above.
The scope of investigation required to inform drainage predictions differs based on the cost-benefit context or engineering problem at hand. For instance, static test results and mass-balance estimates may suffice for shorter-term, smaller-scope problems (‘black box’ models), but long-term and full-size water management strategies for entire mine sites require comprehensive data sets and/or coupled reactive-transport models [13,294]. Experience is required to identify what data is needed for the practical problem at hand and how prediction model outcomes can be interpreted or not (“all models are wrong, some are useful” [295]). The development of mine waste-rock drainage models typically includes baseline scenarios and multiple (conceptual) prediction models that are iteratively updated as data becomes available during operation [296].
4.1. Scaling Phenomena
Direct extrapolation of waste-rock properties from small-scale and short-duration experiments into long-term prediction models for full-size systems can lead to erroneous drainage quantity and quality estimates because smaller-scale static tests of kinetic experiments do not reflect the complex or variable weathering conditions in the field and extent of material heterogeneity encountered in full-scale piles. In addition, many physicochemical processes have undetectable impacts below certain spatiotemporal dimensions [297]: e.g., convection does not exist in humidity cell tests. So-called scaling factors have been proposed to facilitate the use of laboratory parameters onto practice-relevant waste-rock systems [130,298], but these are often semi-empirical correction factors based on limited mechanistic evidence and remain poorly verified across different mine sites.
Upscaling phenomena arise when small-scale processes are extrapolated onto larger scales over orders-of-magnitude, without accounting for changes in continuity or heterogeneity in the studied system. Upscaling of drainage predictions must therefore address the difference between micro- versus macroscale parameters for kinetic or transport processes [299]: the task at hand is to identify which processes undergo relevant scale transitions that are not captured by model parameters (e.g., the onset of thermal convection or transitioning out of a uniform Darcian flow domain).
As discussed in Section 3.4, quantification of spatial heterogeneity in waste-rock piles is pertinent for successful drainage predictions. However, the degree of system heterogeneity may increase with its spatial dimensions [235], and further study (e.g., using variogram models) is required to assess for which parameters and to which extent increasing heterogeneity causes scaling phenomena. An opportunity to investigate scale transitions in waste-rock piles may be provided by dimensionless numbers: quantities relating the spatiotemporal scales of physicochemical processes. Examples include the Damköhler number (transport versus reaction timescales) and Rayleigh number (buoyancy-driven flow; convection), which has been used to assess waste-rock weathering dynamics in piles of various dimensions [206]. Future work on upscaling phenomena is required to improve drainage prediction models.
4.2. Reactive-Transport Models (RTMs)
Reactive transport modelling can be used to quantify the physicochemical processes and interactions between reactive fluids and engineered or geological porous media and has been applied for coupled geochemical and transport phenomena from the micrometer scale to the watershed scale [290,300]. In contrast to the geochemical (aqueous-phase) equilibrium models described above (Section 2.4.1), reactive-transport models (RTMs) are based on mass and energy conservation relations and process-based equations that describe transport (hydraulic advection and dispersion, diffusion, convection) as well as chemical reactions across solids, liquids, and gases [135,231,266,301]. As such, RTMs can account for time-discretized mass and heat transport as well as surface complexation, liquid-gas partitioning, mineral dissolution and precipitation reactions and various biogeochemical reactions [290,292]. Reactive-transport models have been widely deployed for the investigation of waste-rock weathering processes [135,203,296,302] and environmental impact assessment of (abandoned) mines [303], using codes like MIN3P [304], PHREEQC [305], Hytec [306], Geochemist’s Workbench [136], Toughreact [307], Crunchflow, or PFlotran [308].
Due to a typically modular approach, RTMs allow for the incremental inclusion of processes and their interactions [290] and constitute powerful investigative tools. They offer the possibility to quantitatively assess the relevance of certain processes through sensitivity analyses and can be used to investigate the effects of potential engineering solutions to mitigate risks associated to poor drainage quality, such as co-disposal techniques [235] and fluid control layers or covers [135,231,309]. While the versatility of RTMs may be their prime advantage, it also constitutes a limitation: the choice and parametrization of the considered processes. An immediate drawback is that multiple descriptions or solutions of a problem can yield the same drainage composition, which is referred to as non-uniqueness [310]. Setting-up an RTM requires modelling choices, ranging from the selection of relevant processes (heat generation, phase-changes, permeability evolution), solid phases, and their reactive properties (kinetic reactions) to the physical description (flow behavior, physical heterogeneity). While the challenge of such selections increases with the scale of the studied problem, the description of individual subcomponents alone can be difficult (e.g., the initial waste-rock mineralogy can consist of multiple (amorphous) phases present at levels below analytical detection limits for which thermodynamic or kinetic data is unavailable). In addition, RTMs often rely on mathematical equations for physical or chemical processes that simplify potential feedbacks or aggregate unresolved heterogeneity (e.g., through adoption of bulk-averaged parameters) [297]. For instance, the dependence of modelled waste-rock hydraulics on measurable parameters (e.g., matric suction) is often modeled with Van Genuchten relations [250,311,312], which are dependent on particle size [313] and therefore prone to change over time.
With increasing computational power and availability of big data, it has become possible to represent large-scale pile dynamics with stochastic [234,235] or (fully-) coupled process-based RTMs [227,301]. Emerging applications for mine waste-rock systems include dual-continuum approaches [230]. In contrast, relatively few applications of reactive-transport models with fully-coupled thermo-hydro-chemical descriptions (e.g., thermally-driven gas migration) have been performed on large heterogeneous 3D waste-rock systems. A primary challenge in reactive-transport drainage prediction models is not only to identify the processes that are relevant in the studied waste-rock domain, but also to accurately parametrize them: more advanced models for large heterogeneous piles are inherently more difficult to experimentally parametrize. Mechanistic reactive-transport models can be parameterized practically through synoptic sampling (e.g., tracer-tests [51]), and they have the potential to inform practical optimization of the design and management or waste-rock piles, or the mitigation of poor drainage quality. While RTMs have proven crucial to assess process-relevance and waste-rock weathering dynamics under various conditions [231], their development and verification is also laborious. In order to justify and establish their use for practical predictive purposes and management decision-making, it is critical that the relative sensitivities of the model inputs and outputs are translated into actual design parameters.
5. Concluding Remarks
In this review, we discussed critical hydrogeochemical processes affecting the weathering of mine waste rock and its potential environmental impacts. Geochemical and mineralogical reactions, in addition to water, air, and heat transport, are all of key importance in determining waste-rock weathering rates and thus drainage quality as well as quantity. The research reviewed here has been instrumental in providing an understanding of the controls on these individual physicochemical processes, as well as their coupling. Studying the mobility of mine waste pollutants and the underlying mechanisms from the micro- to the macroscale remains important, as expanding mining operations around the world pose increasing potential environmental risks: forward-thinking in the design of long-term dumps that could pose potential leachate quality problems is critical. An improved and quantitative understanding of the factors controlling mine waste-rock drainage dynamics, paired with a growing ability to map in-situ heterogeneity in full-scale systems and the power to harness large-scale, high-resolution data in practical models, will allow engineers and practitioners to develop more robust prediction models and sustainable management decisions. Continuing research can facilitate optimized waste management and thereby prevent waste-rock related environmental deterioration in the future.
Author Contributions
Conceptualization, B.V.; Investigation, B.V., N.S., B.P., H.J.; Writing-Review & Editing, B.V., N.S., B.P., H.J. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Hudson-Edwards, K.A.; Jamieson, H.E.; Lottermoser, B.G. Mine Wastes: Past, Present, Future. Elements 2011, 7, 375–380. [Google Scholar] [CrossRef]
- Blowes, D.W.; Ptacek, C.J.; Jambor, J.L.; Weisener, C.G. The geochemistry of acid mine drainage. Environ. Geochem. 2003, 9, 149–204. [Google Scholar] [CrossRef]
- Dino, G.A.; Cavallo, A.; Rossetti, P.; Garamvölgyi, E.; Sándor, R.; Coulon, F. Towards Sustainable Mining: Exploiting Raw Materials from Extractive Waste Facilities. Sustainability 2020, 12, 2383. [Google Scholar] [CrossRef]
- Lottermoser, B.G. Recycling, Reuse and Rehabilitation of Mine Wastes. Elements 2011, 7, 405–410. [Google Scholar] [CrossRef]
- Lèbre, É.; Corder, G.D.; Golev, A. Sustainable practices in the management of mining waste: A focus on the mineral resource. Miner. Eng. 2017, 107, 34–42. [Google Scholar] [CrossRef]
- Bian, Z.; Miao, X.; Lei, S.; Chen, S.; Wang, W.; Struthers, S. The challenges of reusing mining and mineral-processing wastes. Science 2012, 337, 702–703. [Google Scholar] [CrossRef]
- Byrne, P.; Hudson-Edwards, K.A.; Bird, G.; Macklin, M.G.; Brewer, P.A.; Williams, R.D.; Jamieson, H.E. Water quality impacts and river system recovery following the 2014 Mount Polley mine tailings dam spill, British Columbia, Canada. Appl. Geochem. 2018, 91, 64–74. [Google Scholar] [CrossRef]
- Santamarina, J.C.; Torres-Cruz, L.A.; Bachus, R.C. Why coal ash and tailings dam disasters occur. Science 2019, 364, 526–528. [Google Scholar] [CrossRef]
- Fischer, S.; Rosqvist, G.; Chalov, S.; Jarsjö, J. Disproportionate water quality impacts from the century-old nautanen copper mines, Northern Sweden. Sustainability 2020, 12, 1394. [Google Scholar] [CrossRef]
- Demchak, J.; Skousen, J.; McDonald, L.M. Longevity of acid discharges from underground mines located above the regional water table. J. Environ. Qual. 2004, 33, 656–668. [Google Scholar] [CrossRef]
- Lottermoser, B.G. Mine Wastes: Characterization, Treatment and Environmental Impacts, 3rd ed.; Springer: Berlin/Heidelberg, Germany, 2010; ISBN 3-642-12419-4. [Google Scholar] [CrossRef]
- Mudd, G.M. The Environmental sustainability of mining in Australia: Key mega-trends and looming constraints. Resour. Policy 2010, 35, 98–115. [Google Scholar] [CrossRef]
- Wolkersdorfer, C.; Nordstrom, D.K.; Beckie, R.D.; Cicerone, D.S.; Elliot, T.; Edraki, M.; Valente, T.; França, S.C.A.; Kumar, P.; Oyarzún, R.; et al. Guidance for the Integrated Use of Hydrological, Geochemical, and Isotopic Tools in Mining Operations. Mine Water Environ. 2020, 39, 204–228. [Google Scholar] [CrossRef]
- Hudson-Edwards, A.K.; Dold, B. Mine waste characterization, management and remediation. Minerals 2015, 5, 82–85. [Google Scholar] [CrossRef]
- Jamieson, H.E. Geochemistry and mineralogy of solid mine waste: Essential knowledge for predicting environmental impact. Elements 2011, 7, 381–386. [Google Scholar] [CrossRef]
- Amos, R.T.; Blowes, D.W.; Bailey, B.L.; Sego, D.C.; Smith, L.; Ritchie, A.I.M. Waste-rock hydrogeology and geochemistry. Appl. Geochem. 2015, 57, 140–156. [Google Scholar] [CrossRef]
- Lindsay, M.B.J.; Moncur, M.C.; Bain, J.G.; Jambor, J.L.; Ptacek, C.J.; Blowes, D.W. Geochemical and mineralogical aspects of sulfide mine tailings. Appl. Geochem. 2015, 57, 157–177. [Google Scholar] [CrossRef]
- Ghorbani, Y.; Franzidis, J.-P.; Petersen, J. Heap leaching technology—current state, innovations, and future directions: A review. Miner. Process. Extr. Metall. Rev. 2016, 37, 73–119. [Google Scholar] [CrossRef]
- Marsden, J.O.; Botz, M.M. Heap leach modeling—A review of approaches to metal production forecasting. Miner. Metall. Process. 2017, 34, 53–64. [Google Scholar] [CrossRef]
- Pradhan, N.; Nathsarma, K.C.; Srinivasa Rao, K.; Sukla, L.B.; Mishra, B.K. Heap bioleaching of chalcopyrite: A review. Miner. Eng. 2008, 21, 355–365. [Google Scholar] [CrossRef]
- Dold, B. Sustainability in metal mining: From exploration, over processing to mine waste management. Rev. Environ. Sci. Bio/Technol. 2008, 7, 275. [Google Scholar] [CrossRef]
- Johnson, D.B.; Hallberg, K.B. Acid mine drainage remediation options: A review. Sci. Total Environ. 2005, 338, 3–14. [Google Scholar] [CrossRef] [PubMed]
- Park, I.; Tabelin, C.B.; Jeon, S.; Li, X.; Seno, K.; Ito, M.; Hiroyoshi, N. A review of recent strategies for acid mine drainage prevention and mine tailings recycling. Chemosphere 2019, 219, 588–606. [Google Scholar] [CrossRef]
- Evangelou, V.P.; Zhang, Y.L. A review: Pyrite oxidation mechanisms and acid mine drainage prevention. Crit. Rev. Environ. Sci. Technol. 1995, 25, 141–199. [Google Scholar] [CrossRef]
- Cornelis, G.; Johnson, C.A.; Van Gerven, T.; Vandecasteele, C. Leaching mechanisms of oxyanionic metalloid and metal species in alkaline solid wastes: A review. Appl. Geochem. 2008, 23, 955–976. [Google Scholar] [CrossRef]
- Chandra, A.P.; Gerson, A.R. The mechanisms of pyrite oxidation and leaching: A fundamental perspective. Surf. Sci. Rep. 2010, 65, 293–315. [Google Scholar] [CrossRef]
- Li, Y.; Kawashima, N.; Li, J.; Chandra, A.P.; Gerson, A.R. A review of the structure, and fundamental mechanisms and kinetics of the leaching of chalcopyrite. Adv. Colloid Interface Sci. 2013, 197–198, 1–32. [Google Scholar] [CrossRef]
- Parbhakar-Fox, A.; Lottermoser, B. Principles of Sulfide Oxidation and Acid Rock Drainage. In Environmental Indicators in Metal Mining; Springer: Berlin/Heidelberg, Germany, 2016; pp. 15–34. ISBN 978-3-319-42729-4. [Google Scholar]
- Bosecker, K. Bioleaching: Metal solubilization by microorganisms. FEMS Microbiol. Rev. 1997, 20, 591–604. [Google Scholar] [CrossRef]
- Nordstrom, D.K.; Southam, G. Geomicrobiology of sulfide mineral oxidation. In Reviews in Mineralogy; Banfield, J.F., Nealson, K.H., Eds.; Mineralogical Society of America: Chantilly, VA, USA, 1997; Volume 35, pp. 361–390. [Google Scholar]
- Rawlings, D.E. Heavy metal mining using microbes. Annu. Rev. Microbiol. 2002, 56, 65–91. [Google Scholar] [CrossRef]
- Baker, B.J.; Banfield, J.F. Microbial communities in acid mine drainage. FEMS Microbiol. Ecol. 2003, 44, 139–152. [Google Scholar] [CrossRef]
- Johnson, D.B.; Hallberg, K.B. The microbiology of acidic mine waters. Res. Microbiol. 2003, 154, 466–473. [Google Scholar] [CrossRef]
- Watling, H.R. The bioleaching of sulphide minerals with emphasis on copper sulphides—A review. Hydrometallurgy 2006, 84, 81–108. [Google Scholar] [CrossRef]
- Rawlings, D.E.; Johnson, D.B. The microbiology of biomining: Development and optimization of mineral-oxidizing microbial consortia. Microbiology 2007, 153, 315–324. [Google Scholar] [CrossRef] [PubMed]
- Johnson, D.B. Biomining-biotechnologies for extracting and recovering metals from ores and waste materials. Curr. Opin. Biotechnol. 2014, 30, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.-N.; Kuang, J.-L.; Shu, W.-S. Microbial ecology and evolution in the acid mine drainage model system. Trends Microbiol. 2016, 24, 581–593. [Google Scholar] [CrossRef]
- Kaksonen, A.H.; Boxall, N.J.; Gumulya, Y.; Khaleque, H.N.; Morris, C.; Bohu, T.; Cheng, K.Y.; Usher, K.M.; Lakaniemi, A.M. Recent progress in biohydrometallurgy and microbial characterisation. Hydrometallurgy 2018, 180, 7–25. [Google Scholar] [CrossRef]
- Ledin, M.; Pedersen, K. The environmental impact of mine wastes—roles of microorganisms and their significance in treatment of mine wastes. Earth-Sci. Rev. 1996, 41, 67–108. [Google Scholar] [CrossRef]
- Akcil, A.; Koldas, S. Acid Mine Drainage (AMD): Causes, treatment and case studies. J. Clean. Prod. 2006, 14, 1139–1145. [Google Scholar] [CrossRef]
- Neculita, C.M.; Zagury, G.J.; Bussière, B. Passive treatment of acid mine drainage in bioreactors using sulfate-reducing bacteria: Critical review and research needs. J. Environ. Qual. 2007, 36, 1–16. [Google Scholar] [CrossRef]
- RoyChowdhury, A.; Sarkar, D.; Datta, R. Remediation of acid mine drainage-impacted water. Curr. Pollut. Rep. 2015, 1, 131–141. [Google Scholar] [CrossRef]
- Jamieson, H.E.; Walker, S.R.; Parsons, M.B. Mineralogical characterization of mine waste. Appl. Geochem. 2015, 57, 85–105. [Google Scholar] [CrossRef]
- Piatak, N.M.; Parsons, M.B.; Seal, R.R. Characteristics and environmental aspects of slag: A review. Appl. Geochem. 2015, 57, 236–266. [Google Scholar] [CrossRef]
- Parbhakar-Fox, A.; Lottermoser, B.G. A critical review of acid rock drainage prediction methods and practices. Miner. Eng. 2015, 82, 107–124. [Google Scholar] [CrossRef]
- Dold, B. Acid rock drainage prediction: A critical review. J. Geochem. Explor. 2017, 172, 120–132. [Google Scholar] [CrossRef]
- Kefeni, K.K.; Msagati, T.A.M.; Mamba, B.B. Acid mine drainage: Prevention, treatment options, and resource recovery: A review. J. Clean. Prod. 2017, 151, 475–493. [Google Scholar] [CrossRef]
- Lottermoser, B.G. Environmental Indicators in Metal. Mining; Springer: Berlin/Heidelberg, Germany, 2017; ISBN 978-3-319-42729-4. [Google Scholar] [CrossRef]
- Moodley, I.; Sheridan, C.M.; Kappelmeyer, U.; Akcil, A. Environmentally sustainable acid mine drainage remediation: Research developments with a focus on waste/by-products. Miner. Eng. 2018, 126, 207–220. [Google Scholar] [CrossRef]
- Rajaram, V.; Dutta, S.; Parameswaran, K. Sustainable Mining Practices: A Global Perspective; A.A. Balkema: Leiden, The Netherlands, 2005; ISBN 9058096890. [Google Scholar]
- Nordstrom, D.K. Hydrogeochemical processes governing the origin, transport and fate of major and trace elements from mine wastes and mineralized rock to surface waters. Appl. Geochem. 2011, 26, 1777–1791. [Google Scholar] [CrossRef]
- Öhlander, B.; Chatwin, T.; Alakangas, L. Management of sulfide-bearing waste, a challenge for the mining industry. Minerals 2012, 2, 1–10. [Google Scholar] [CrossRef]
- Simate, G.S.; Ndlovu, S. Acid mine drainage: Challenges and opportunities. J. Environ. Chem. Eng. 2014, 2. [Google Scholar] [CrossRef]
- Nordstrom, D.K.; Blowes, D.W.; Ptacek, C.J. Hydrogeochemistry and microbiology of mine drainage: An update. Appl. Geochem. 2015, 57, 3–16. [Google Scholar] [CrossRef]
- Batterham, R.J. The mine of the future—Even more sustainable. Miner. Eng. 2017, 107, 2–7. [Google Scholar] [CrossRef]
- Aznar-Sánchez, J.A.; García-Gómez, J.J.; Velasco-Muñoz, J.F.; Carretero-Gómez, A. Mining waste and its sustainable management: Advances in worldwide research. Minerals 2018, 8, 284. [Google Scholar] [CrossRef]
- Li, J.; Kawashima, N.; Fan, R.; Schumann, R.C.; Gerson, A.R.; Smart, R.S.C. Method for distinctive estimation of stored acidity forms in acid mine wastes. Environ. Sci. Technol. 2014, 48, 11445–11452. [Google Scholar] [CrossRef] [PubMed]
- Vaughan, D.J. Minerals Sulphides. In Reference Module in Earth Systems and Environmental Sciences; Elsevier: Amsterdam, The Netherlands, 2013; ISBN 978-0-12-409548-9. [Google Scholar]
- Elberling, B.; Schippers, A.; Sand, W. Bacterial and chemical oxidation of pyritic mine tailings at low temperatures. J. Contam. Hydrol. 2000, 41, 225–238. [Google Scholar] [CrossRef]
- Singer, P.C.; Stumm, W. Acidic mine drainage: The rate-determining step. Science 1970, 167, 1121–1123. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, E.C.; de Mendonça Silva, J.C.; Duarte, H.A. Pyrite oxidation mechanism by oxygen in aqueous medium. J. Phys. Chem. C 2016, 120, 2760–2768. [Google Scholar] [CrossRef]
- Tabelin, C.B.; Suchol, V.; Mayumi, I.; Naoki, H.; Toshifumi, I. Pyrite oxidation in the presence of hematite and alumina: II. Effects on the cathodic and anodic half-cell reactions. Sci. Total Environ. 2017, 581–582, 126–135. [Google Scholar] [CrossRef]
- Schippers, A.; Breuker, A.; Blazejak, A.; Bosecker, K.; Kock, D.; Wright, T.L. The biogeochemistry and microbiology of sulfidic mine waste and bioleaching dumps and heaps, and novel Fe(II)-oxidizing bacteria. Hydrometallurgy 2010, 104, 342–350. [Google Scholar] [CrossRef]
- Luther, G.W.; Findlay, A.; MacDonald, D.; Owings, S.; Hanson, T.; Beinart, R.; Girguis, P. Thermodynamics and kinetics of sulfide oxidation by oxygen: A look at inorganically controlled reactions and biologically mediated processes in the environment. Front. Microbiol. 2011, 2, 62. [Google Scholar] [CrossRef]
- Schrenk, M.O.; Edwards, K.J.; Goodman, R.M.; Hamers, R.J.; Banfield, J.F. Distribution of thiobacillus ferrooxidans and Leptospirillum ferrooxidans: Implications for generation of acid mine drainage. Science 1998, 279, 1519–1522. [Google Scholar] [CrossRef]
- Korehi, H.; Blöthe, M.; Schippers, A. Microbial diversity at the moderate acidic stage in three different sulfidic mine tailings dumps generating acid mine drainage. Res. Microbiol. 2014, 165, 713–718. [Google Scholar] [CrossRef]
- Hallberg, K.B. New perspectives in acid mine drainage microbiology. Hydrometall. 2010, 104, 448–453. [Google Scholar] [CrossRef]
- Blackmore, S.; Vriens, B.; Sorensen, M.; Power, I.M.; Smith, L.; Hallam, S.J.; Mayer, U.K.; Beckie, R.D. Microbial and geochemical controls on waste rock weathering and drainage quality. Sci. Total Environ. 2018, 640–641, 1004–1014. [Google Scholar] [CrossRef] [PubMed]
- Rimstidt, J.D.; Vaughan, D.J. Pyrite oxidation: A state-of-the-art assessment of the reaction mechanism. Geochim. Cosmochim. Acta 2003, 67, 873–880. [Google Scholar] [CrossRef]
- Schippers, A.; Sand, W. Bacterial leaching of metal sulfides proceeds by two indirect mechanisms via thiosulfate or via polysulfides and sulfur. Appl. Environ. Microbiol. 1999, 65, 319–321. [Google Scholar] [CrossRef] [PubMed]
- Borilova, S.; Mandl, M.; Zeman, J.; Kucera, J.; Pakostova, E.; Janiczek, O.; Tuovinen, O.H. Can sulfate be the first dominant aqueous sulfur species formed in the oxidation of pyrite by acidithiobacillus ferrooxidans? Front. Microbiol. 2018, 9, 3134. [Google Scholar] [CrossRef]
- Johnson, A.C.; Romaniello, S.J.; Reinhard, C.T.; Gregory, D.D.; Garcia-Robledo, E.; Revsbech, N.P.; Canfield, D.E.; Lyons, T.W.; Anbar, A.D. Experimental determination of pyrite and molybdenite oxidation kinetics at nanomolar oxygen concentrations. Geochim. Cosmochim. Acta 2019, 249, 160–172. [Google Scholar] [CrossRef]
- Kameia, G.; Ohmotob, H. The kinetics of reactions between pyrite and O2-bearing water revealed from in situ monitoring of DO, Eh and pH in a closed system. Geochim. Cosmochim. Acta 2000, 64, 2585–2601. [Google Scholar] [CrossRef]
- Janzen, M.P.; Nicholson, R.V.; Scharer, J.M. Pyrrhotite reaction kinetics: Reaction rates for oxidation by oxygen, ferric iron, and for nonoxidative dissolution. Geochim. Cosmochim. Acta 2000, 64, 1511–1522. [Google Scholar] [CrossRef]
- Nicholson, R.V.; Gillham, R.W.; Reardon, E.J. Pyrite oxidation in carbonate-buffered solution: 1. Experimental kinetics. Geochim. Cosmochim. Acta 1988, 52, 1077–1085. [Google Scholar] [CrossRef]
- Williamson, M.A.; Rimstidt, J.D. The kinetics and electrochemical rate-determining step of aqueous pyrite oxidation. Geochim. Cosmochim. Acta 1994, 58, 5443–5454. [Google Scholar] [CrossRef]
- Rimstidt, J.D.; Chermak, J.A.; Gagen, P.M. Rates of Reaction of Galena, Sphalerite, Chalcopyrite, and Arsenopyrite with Fe(III) in Acidic Solutions. In Environmental Geochemistry of Sulfide Oxid; American Chemical Society: Washington, DC, USA, 1993; Volume 550, pp. 1–2. ISBN 9780841227729. [Google Scholar]
- Walker, F.P.; Schreiber, M.E.; Rimstidt, J.D. Kinetics of arsenopyrite oxidative dissolution by oxygen. Geochim. Cosmochim. Acta 2006, 70, 1668–1676. [Google Scholar] [CrossRef]
- Heidel, C.; Tichomirowa, M.; Breitkopf, C. Sphalerite oxidation pathways detected by oxygen and sulfur isotope studies. Appl. Geochem. 2011, 26, 2247–2259. [Google Scholar] [CrossRef]
- Olson, G.J.; Clark, T.R. Bioleaching of molybdenite. Hydrometallurgy 2008, 93, 10–15. [Google Scholar] [CrossRef]
- Jerz, J.K.; Rimstidt, J.D. Pyrite oxidation in moist air Associate editor: M. A. McKibben. Geochim. Cosmochim. Acta 2004, 68, 701–714. [Google Scholar] [CrossRef]
- Moses, C.O.; Herman, J.S. Pyrite oxidation at circumneutral pH. Geochim. Cosmochim. Acta 1991, 55, 471–482. [Google Scholar] [CrossRef]
- Sun, H.; Chen, M.; Zou, L.; Shu, R.; Ruan, R. Study of the kinetics of pyrite oxidation under controlled redox potential. Hydrometallurgy 2015, 155, 13–19. [Google Scholar] [CrossRef]
- Long, H.; Dixon, D.G. Pressure oxidation of pyrite in sulfuric acid media: A kinetic study. Hydrometallurgy 2004, 73, 335–349. [Google Scholar] [CrossRef]
- Boon, M.; Heijnen, J.J. Chemical oxidation kinetics of pyrite in bioleaching processes. Hydrometallurgy 1998, 48, 27–41. [Google Scholar] [CrossRef]
- McKibben, M.A.; Barnes, H.L. Oxidation of pyrite in low temperature acidic solutions: Rate laws and surface textures. Geochim. Cosmochim. Acta 1986, 50, 1509–1520. [Google Scholar] [CrossRef]
- Lehner, S.; Savage, K. The effect of As, Co, and Ni impurities on pyrite oxidation kinetics: Batch and flow-through reactor experiments with synthetic pyrite. Geochim. Cosmochim. Acta 2008, 72, 1788–1800. [Google Scholar] [CrossRef]
- Navarro, C.A.; von Bernath, D.; Jerez, C.A. Heavy Metal Resistance Strategies of Acidophilic Bacteria and Their Acquisition: Importance for Biomining and Bioremediation. Biol. Res. 2013, 46, 363–371. [Google Scholar] [CrossRef]
- Chen, J.; He, F.; Zhang, X.; Sun, X.; Zheng, J.; Zheng, J. Heavy metal pollution decreases microbial abundance, diversity and activity within particle-size fractions of a paddy soil. FEMS Microbiol. Ecol. 2014, 87, 164–181. [Google Scholar] [CrossRef] [PubMed]
- Hendry, M.J.; Biswas, A.; Essilfie-Dughan, J.; Chen, N.; Day, S.J.; Barbour, S.L. Reservoirs of Selenium in Coal Waste Rock: Elk Valley, British Columbia, Canada. Environ. Sci. Technol. 2015, 49, 8228–8236. [Google Scholar] [CrossRef] [PubMed]
- Chon, H.-T.; Hwang, J.-H. Geochemical Characteristics of the Acid Mine Drainage in the Water System in the Vicinity of the Dogye Coal Mine in Korea. Environ. Geochem. Health 2000, 22, 155–172. [Google Scholar] [CrossRef]
- Black, A.; Craw, D. Arsenic, copper and zinc occurrence at the Wangaloa coal mine, southeast Otago, New Zealand. Int. J. Coal Geol. 2001, 45, 181–193. [Google Scholar] [CrossRef]
- Qureshi, A.; Maurice, C.; Öhlander, B. Potential of coal mine waste rock for generating acid mine drainage. J. Geochem. Explor. 2016, 160, 44–54. [Google Scholar] [CrossRef]
- Equeenuddin, S.M.; Tripathy, S.; Sahoo, P.K.; Panigrahi, M.K. Hydrogeochemical characteristics of acid mine drainage and water pollution at Makum Coalfield, India. J. Geochem. Explor. 2010, 105, 75–82. [Google Scholar] [CrossRef]
- Sahoo, P.K.; Tripathy, S.; Equeenuddin, S.M.; Panigrahi, M.K. Geochemical characteristics of coal mine discharge vis-à-vis behavior of rare earth elements at Jaintia Hills coalfield, northeastern India. J. Geochem. Explor. 2012, 112, 235–243. [Google Scholar] [CrossRef]
- Szczepanska, J.; Twardowska, I. Distribution and environmental impact of coal-mining wastes in Upper Silesia, Poland. Environ. Geol. 1999, 38, 249–258. [Google Scholar] [CrossRef]
- Banks, S.B.; Banks, D. Abandoned mines drainage: Impact assessment and mitigation of discharges from coal mines in the UK. Eng. Geol. 2001, 60, 31–37. [Google Scholar] [CrossRef]
- Hughes, J.; Craw, D.; Peake, B.; Lindsay, P.; Weber, P. Environmental characterisation of coal mine waste rock in the field: An example from New Zealand. Environ. Geol. 2007, 52, 1501–1509. [Google Scholar] [CrossRef]
- Calkins, W.H. The chemical forms of sulfur in coal: A review. Fuel 1994, 73, 475–484. [Google Scholar] [CrossRef]
- Kazadi Mbamba, C.; Harrison, S.T.L.; Franzidis, J.-P.; Broadhurst, J.L. Mitigating acid rock drainage risks while recovering low-sulfur coal from ultrafine colliery wastes using froth flotation. Miner. Eng. 2012, 29, 13–21. [Google Scholar] [CrossRef]
- Banerjee, D. Acid drainage potential from coal mine wastes: Environmental assessment through static and kinetic tests. Int. J. Environ. Sci. Technol. 2014, 11, 1365–1378. [Google Scholar] [CrossRef]
- Dutta, M.; Saikia, J.; Taffarel, S.R.; Waanders, F.B.; de Medeiros, D.; Cutruneo, C.M.N.L.; Silva, L.F.O.; Saikia, B.K. Environmental assessment and nano-mineralogical characterization of coal, overburden and sediment from Indian coal mining acid drainage. Geosci. Front. 2017, 8, 1285–1297. [Google Scholar] [CrossRef]
- Ghosh, W.; Dam, B. Biochemistry and molecular biology of lithotrophic sulfur oxidation by taxonomically and ecologically diverse bacteria and archaea. FEMS Microbiol. Rev. 2009, 33, 999–1043. [Google Scholar] [CrossRef]
- Acharya, C.; Sukla, L.B.; Misra, V.N. Biodepyritisation of coal. J. Chem. Technol. Biotechnol. 2004, 79, 1–12. [Google Scholar] [CrossRef]
- Schippers, A.; Rohwerder, T.; Sand, W. Intermediary sulfur compounds in pyrite oxidation: Implications for bioleaching and biodepyritization of coal. Appl. Microbiol. Biotechnol. 1999, 52, 104–110. [Google Scholar] [CrossRef]
- Davalos, A.; Pecina, E.T.; Soria, M.; Carrillo, F.R. Kinetics of Coal Desulfurization in An Oxidative Acid Media. Int. J. Coal Prep. Util. 2009, 29, 152–172. [Google Scholar] [CrossRef]
- Juszczak, A.; Domka, F.; Kozłowski, M.; Wachowska, H. Microbial desulfurization of coal with Thiobacillus ferrooxidans bacteria. Fuel 1995, 74, 725–728. [Google Scholar] [CrossRef]
- Hoffmann, M.R.; Faust, B.C.; Panda, F.A.; Koo, H.H.; Tsuchiya, H.M. Kinetics of the Removal of Iron Pyrite from Coal by Microbial Catalysis. Appl. Environ. Microbiol. 1981, 42, 259–271. [Google Scholar] [CrossRef] [PubMed]
- Liang, L.; McNabb, J.A.; Paulk, J.M.; Gu, B.; McCarthy, J.F. Kinetics of iron(II) oxygenation at low partial pressure of oxygen in the presence of natural organic matter. Environ. Sci. Technol. 1993, 27, 1864–1870. [Google Scholar] [CrossRef]
- Kalin, M.; Cairns, J.; McCready, R. Ecological engineering methods for acid mine drainage treatment of coal wastes. Resour. Conserv. Recycl. 1991, 5, 265–275. [Google Scholar] [CrossRef]
- Kaksonen, A.H.; Puhakka, J.A. Sulfate reduction based bioprocesses for the treatment of acid mine drainage and the recovery of metals. Eng. Life Sci. 2007, 7, 541–564. [Google Scholar] [CrossRef]
- Lefticariu, L.; Walters, E.R.; Pugh, C.W.; Bender, K.S. Sulfate reducing bioreactor dependence on organic substrates for remediation of coal-generated acid mine drainage: Field experiments. Appl. Geochem. 2015, 63, 70–82. [Google Scholar] [CrossRef]
- Küsel, K. Microbial cycling of iron and sulfur in acidic coal mining lake sediments. Water Air Soil Pollut. Focus 2003, 3, 67–90. [Google Scholar] [CrossRef]
- Ayora, C.; Caraballo, M.A.; Macias, F.; Rötting, T.S.; Carrera, J.; Nieto, J.-M. Acid mine drainage in the Iberian Pyrite Belt: 2. Lessons learned from recent passive remediation experiences. Environ. Sci. Pollut. Res. 2013, 20, 7837–7853. [Google Scholar] [CrossRef]
- Vriens, B.; Peterson, H.E.; Laurenzi, L.; Smith, L.; Aranda, C.; Mayer, K.U.; Beckie, R.D. Long-term monitoring of waste-rock weathering at Antamina, Peru. Chemosphere 2019, 215, 858–869. [Google Scholar] [CrossRef]
- Maree, J.P.; de Beer, M.; Strydom, W.F.; Christie, A.D.M.; Waanders, F.B. Neutralizing Coal Mine Effluent with Limestone to Decrease Metals and Sulphate Concentrations. Mine Water Environ. 2004, 23, 81–86. [Google Scholar] [CrossRef]
- Alakangas, L.; Andersson, E.; Mueller, S. Neutralization/prevention of acid rock drainage using mixtures of alkaline by-products and sulfidic mine wastes. Environ. Sci. Pollut. Res. 2013, 20, 7907–7916. [Google Scholar] [CrossRef]
- Davies, H.; Weber, P.; Lindsay, P.; Craw, D.; Peake, B.; Pope, J. Geochemical changes during neutralisation of acid mine drainage in a dynamic mountain stream, New Zealand. Appl. Geochem. 2011, 26, 2121–2133. [Google Scholar] [CrossRef]
- Chou, L.; Garrels, R.M.; Wollast, R. Comparative study of the kinetics and mechanisms of dissolution of carbonate minerals. Chem. Geol. 1989, 78, 269–282. [Google Scholar] [CrossRef]
- Morse, J.W.; Arvidson, R.S. The dissolution kinetics of major sedimentary carbonate minerals. Earth-Sci. Rev. 2002, 58, 51–84. [Google Scholar] [CrossRef]
- Kaufmann, G.; Dreybrodt, W. Calcite dissolution kinetics in the system CaCO3–H2O–CO2 at high undersaturation. Geochim. Cosmochim. Acta 2007, 71, 1398–1410. [Google Scholar] [CrossRef]
- Stumm, W.; Wollast, R. Coordination chemistry of weathering: Kinetics of the surface-controlled dissolution of oxide minerals. Rev. Geophys. 1990, 28, 53–69. [Google Scholar] [CrossRef]
- Schwertmann, U. Solubility and dissolution of iron oxides. Plant. Soil 1991, 130, 1–25. [Google Scholar] [CrossRef]
- White, A.F.; Brantley, S.L. Chemical weathering rates of silicate minerals; an overview. Rev. Mineral. Geochem. 1995, 31, 1–22. [Google Scholar]
- Gruber, C.; Kutuzov, I.; Ganor, J. The combined effect of temperature and pH on albite dissolution rate under far-from-equilibrium conditions. Geochim. Cosmochim. Acta 2016, 186, 154–167. [Google Scholar] [CrossRef]
- Rimstidt, J.D.; Dove, P.M. Mineral/solution reaction rates in a mixed flow reactor: Wollastonite hydrolysis. Geochim. Cosmochim. Acta 1986, 50, 2509–2516. [Google Scholar] [CrossRef]
- Kirste, D.; Pearce, J.; Golding, S. Parameterizing Geochemical Models: Do Kinetics of Calcite Matter? Procedia Earth Planet. Sci. 2017, 17, 606–609. [Google Scholar] [CrossRef]
- Vriens, B.; Skierszkan, E.K.; St-Arnault, M.; Salzsauler, K.A.; Aranda, C.; Mayer, K.U.; Beckie, R.D. Mobilization of metal(loid) oxyanions through circumneutral waste-rock drainage. ACS Omega 2019, 4, 10205–10215. [Google Scholar] [CrossRef]
- Plante, B.; Benzaazoua, M.; Bussière, B. Predicting Geochemical Behaviour of Waste Rock with Low Acid Generating Potential Using Laboratory Kinetic Tests. Mine Water Environ. 2011, 30, 2–21. [Google Scholar] [CrossRef]
- Plante, B.; Bussière, B.; Benzaazoua, M. Lab to field scale effects on contaminated neutral drainage prediction from the Tio mine waste rocks. J. Geochem. Explor. 2014, 137, 37–47. [Google Scholar] [CrossRef]
- Nicholson, R.V.; Rinker, M.J. Metal leaching from sulphide mine waste under neutral pH conditions. In Proceedings of the 5th International Conference on Acid Rock Drainage (ICARD), Denver, CO, USA, 21–24 May 2000; pp. 951–958. [Google Scholar]
- Price, W.A. Prediction Manual for Drainage Chemistry from Sulphidic Geologic Materials. MEND Report 1.20.1. Available online: https://www.fs.usda.gov/Internet/FSE_DOCUMENTS/stelprdb5336546.pdf (accessed on 1 July 2020).
- Tabelin, C.B.; Silwamba, M.; Paglinawan, F.C.; Mondejar, A.J.S.; Duc, H.G.; Resabal, V.J.; Opiso, E.M.; Igarashi, T.; Tomiyama, S.; Ito, M.; et al. Solid-phase partitioning and release-retention mechanisms of copper, lead, zinc and arsenic in soils impacted by artisanal and small-scale gold mining (ASGM) activities. Chemosphere 2020, 260, 127574. [Google Scholar] [CrossRef]
- Tamoto, S.; Tabelin, C.B.; Igarashi, T.; Ito, M.; Hirojohsi, N. Short and long term release mechanisms of arsenic, selenium and boron from a tunnel-excavated sedimentary rock under in situ conditions. J. Cont. Hydr. 2015, 175–176, 60–71. [Google Scholar] [CrossRef]
- Demers, I.; Molson, J.; Bussière, B.; Laflamme, D. Numerical modeling of contaminated neutral drainage from a waste-rock field test cell. Appl. Geochem. 2013, 33, 346–356. [Google Scholar] [CrossRef]
- Tabelin, C.B.; Sasaki, R.; Igarashi, T.; Park, I.; Tamoto, S.; Arima, T.; Ito, M.; Hiroyoshi, N. Simultaneous leaching of arsenite, arsenate, selenite and selenate, and their migration in tunnel-excavated sedimentary rocks: II. Kinetic and reactive transport modeling. Chemosphere 2017, 188, 444–454. [Google Scholar] [CrossRef]
- Nordstrom, D.K.; Archer, D.G. Arsenic Thermodynamic Data and Environmental Geochemistry—Arsenic in Ground Water: Geochemistry and Occurrence; Welch, A.H., Stollenwerk, K.G., Eds.; Springer: Boston, MA, USA, 2003; pp. 1–25. ISBN 978-0-306-47956-4. [Google Scholar]
- Kocourková, E.; Sracek, O.; Houzar, S.; Cempírek, J.; Losos, Z.; Filip, J.; Hršelová, P. Geochemical and mineralogical control on the mobility of arsenic in a waste rock pile at Dlouhá Ves, Czech Republic. J. Geochem. Explor. 2011, 110, 61–73. [Google Scholar] [CrossRef]
- Wei, X.; Zhang, S.; Shimko, J.; Dengler, R.W., II. Mine drainage: Treatment technologies and rare earth elements. Water Environ. Res. 2019, 91, 1061–1068. [Google Scholar] [CrossRef]
- González, V.; Vignati, D.; Leyval, C.; Giamberini, L. Environmental fate and ecotoxicity of lanthanides: Are they a uniform group beyond chemistry? Environ. Int. 2014, 71, 148–157. [Google Scholar] [CrossRef] [PubMed]
- Romero-Freire, A.; Turlin, F.; André-Mayer, A.-S.; Pelletier, M.; Cayer, A.; Giamberini, L. Giamberini Biogeochemical Cycle of Lanthanides in a Light Rare Earth Element-Enriched Geological Area (Quebec, Canada). Minerals 2019, 9, 573. [Google Scholar] [CrossRef]
- Ayora, C.; Macías, F.; Torres, E.; Lozano, A.; Carrero, S.; Nieto, J.-M.; Pérez-López, R.; Fernández-Martínez, A.; Castillo-Michel, H. Recovery of rare earth elements and yttrium from passive-remediation systems of acid mine drainage. Environ. Sci. Technol. 2016, 50, 8255–8262. [Google Scholar] [CrossRef]
- Royer-Lavallée, A.; Neculita, C.M.; Coudert, L. Removal and potential recovery of rare earth elements from mine water. J. Ind. Eng. Chem. 2020, 89, 47–57. [Google Scholar] [CrossRef]
- Edahbi, M.; Plante, B.; Benzaazoua, M. Environmental challenges and identification of the knowledge gaps associated with REE mine wastes management. J. Clean. Prod. 2019, 212, 1232–1241. [Google Scholar] [CrossRef]
- Jamieson, H.; Laidlow, A.; Parsons, M. Characterization of U and REE Mobility Downstream of U Tailings near Bancroft, Ontario. In Proceedings of the International Conference on Acid Rock Drainage (ICARD), Santiago, Chile, 21–24 April 2015. [Google Scholar]
- Edahbi, M.; Plante, B.; Benzaazoua, M.; Pelletier, M. Geochemistry of rare earth elements within waste rocks from the Montviel carbonatite deposit, Québec, Canada. Environ. Sci. Pollut. Res. 2018, 25, 10997–11010. [Google Scholar] [CrossRef]
- Edahbi, M.; Plante, B.; Benzaazoua, M.; Kormos, L.; Pelletier, M. Rare earth elements (La, Ce, Pr, Nd, and Sm) from a carbonatite deposit: Mineralogical characterization and geochemical behavior. Minerals 2018, 8, 55. [Google Scholar] [CrossRef]
- Edahbi, M.; Plante, B.; Benzaazoua, M.; Ward, M.; Pelletier, M. Mobility of rare earth elements in mine drainage: Influence of iron oxides, carbonates, and phosphates. Chemosphere 2018, 199, 647–654. [Google Scholar] [CrossRef]
- Kauppila, P.; Räisänen, M. Mineralogical and geochemical alteration of Hitura sulphide mine tailings with emphasis on nickel mobility and retention. J. Geochem. Explor. 2008, 97, 1–20. [Google Scholar] [CrossRef]
- Kauppila, P.; Räisänen, M.; Johnson, R. Geochemical Characterisation of Seepage and Drainage Water Quality from Two Sulphide Mine Tailings Impoundments: Acid Mine Drainage versus Neutral Mine Drainage. Mine Water Environ. 2008, 28, 30–49. [Google Scholar] [CrossRef]
- Radkova, A.; Jamieson, H.; Campbell, K. Antimony mobility during the early stages of stibnite weathering in tailings at the Beaver Brook Sb deposit, Newfoundland. Appl. Geochem. 2020, 115, 104528. [Google Scholar] [CrossRef]
- Lindsay, M.B.J.; Condon, P.D.; Jambor, J.L.; Lear, K.G.; Blowes, D.W.; Ptacek, C.J. Mineralogical, geochemical, and microbial investigation of a sulfide-rich tailings deposit characterized by neutral drainage. Appl. Geochem. 2009, 24, 2212–2221. [Google Scholar] [CrossRef]
- Jamieson, H.E.; Bromstad, M.; Nordstrom, D.K. Extremely arsenic-rich, pH-neutral waters from the Giant mine, Canada. In Proceedings of the First International Conference on Mine Water Solutions in Extreme Environments, Lima, Peru, 15–17 April 2013; pp. 82–94. [Google Scholar]
- Kwong, Y.T.J.; Percival, J.B.; Soprovich, E.A. Arsenic Mobilization and Attenuation in Near-Neutral Drainage—Implications for Tailings and Waste Rock Management for Saskatchewan Uranium Mines; Canadian Institute of Mining, Metallurgy and Petroleum: Montreal, QC, Canada, 2000; ISBN 1-894475-05-4. [Google Scholar]
- Plante, B.; Benzaazoua, M.; Bussière, B.; Biesinger, M.C.; Pratt, A.R. Study of Ni sorption onto Tio mine waste rock surfaces. Appl. Geochem. 2010, 25, 1830–1844. [Google Scholar] [CrossRef]
- Powell, K.J.; Brown, P.L.; Byrne, R.H.; Gajda, T.; Hefter, G.; Sjöberg, S.; Wanner, H. Chemical speciation of environmentally significant metals with inorganic ligands Part 2: The Cu2+ + OH-, Cl-, CO32-, SO42-, and PO43- systems (IUPAC Technical Report). Pure Appl. Chem. 2007, 79, 895–950. [Google Scholar] [CrossRef]
- Powell, K.J.; Brown, P.L.; Byrne, R.H.; Gajda, T.; Hefter, G.; Leuz, A.-K.; Sjöberg, S.; Wanner, H. Chemical speciation of environmentally significant metals with inorganic ligands. Part 5: The Zn2+ + OH-, Cl-, CO32-, SO42-, and PO43- systems (IUPAC Technical Report). Pure Appl. Chem. 2013, 85, 2249–2311. [Google Scholar] [CrossRef]
- Grafe, M.; Eick, M.J.; Grossl, P.R.; Saunders, A.M. Adsorption of Arsenate and Arsenite on Ferrihydrite in the Presence and Absence of Dissolved Organic Carbon. J. Environ. Qual. 2002, 31, 1115–1123. [Google Scholar] [CrossRef]
- Zhou, Y.-F.; Haynes, R.J. Sorption of Heavy Metals by Inorganic and Organic Components of Solid Wastes: Significance to Use of Wastes as Low-Cost Adsorbents and Immobilizing Agents. Crit. Rev. Environ. Sci. Technol. 2010, 40, 909–977. [Google Scholar] [CrossRef]
- Stumm, W.; Morgan, J.J. Aquatic Chemistry—Chemical Equilibria and Rates in Natural Waters, 3rd ed.; Wiley-Interscience: Hoboken, NJ, USA, 1995; ISBN 978-0471511854. [Google Scholar]
- Skierszkan, E.K.; Stockwell, J.S.; Dockrey, J.W.; Weis, D.; Beckie, R.D.; Mayer, K.U. Molybdenum (Mo) stable isotopic variations as indicators of Mo attenuation in mine waste-rock drainage. Appl. Geochem. 2017, 87, 71–83. [Google Scholar] [CrossRef]
- Bao, Z.; Blowes, D.W.; Ptacek, C.J.; Bain, J.; Holland, S.P.; Wilson, D.; Wilson, W.; MacKenzie, P. Faro waste rock project: Characterizing variably saturated flow behavior through full-scale waste-rock dumps in the continental subarctic region of northern Canada using field measurements and stable isotopes of water. Water Resour. Res. 2020, 56. [Google Scholar] [CrossRef]
- St-Arnault, M.; Vriens, B.; Klein, B.; Blaskovich, R.; Aranda, C.; Mayer, K.U.; Beckie, R.D. Geochemical and mineralogical assessment of localized reactive zones through a full scale heterogeneous waste rock pile. Miner. Eng. 2020, 145, 106089. [Google Scholar] [CrossRef]
- Al, T.A.; Martin, C.J.; Blowes, D.W. Carbonate-mineral/water interactions in sulfide-rich mine tailings. Geochim. Cosmochim. Acta 2000, 64, 3933–3948. [Google Scholar] [CrossRef]
- Tabelin, C.B.; Corpuz, R.D.; Igarashi, T.; Villacorte-Tabelin, M.; Diaz Alorro, R.; Yoo, K.; Raval, S.; Ito, M.; Hiroyoshi, N. Acid mine drainage formation and arsenic mobility under strongly acidic conditions: Importance of soluble phases, iron oxyhydroxides/oxides and nature of oxidation layer on pyrite. J. Hazard. Mat. 2020, 399, 122844. [Google Scholar] [CrossRef]
- Hirsche, D.T.; Blaskovich, R.; Mayer, K.U.; Beckie, R.D. A study of Zn and Mo attenuation by waste-rock mixing in neutral mine drainage using mixed-material field barrels and humidity cells. Appl. Geochem. 2017, 84, 114–125. [Google Scholar] [CrossRef]
- Conlan, M.J.W.; Mayer, K.U.; Blaskovich, R.; Beckie, R.D. Solubility controls for molybdenum in neutral rock drainage. Geochem. Explor. Environ. Anal. 2012, 12, 21–32. [Google Scholar] [CrossRef]
- Moncur, M.C.; Jambor, J.L.; Ptacek, C.J.; Blowes, D.W. Mine drainage from the weathering of sulfide minerals and magnetite. Appl. Geochem. 2009, 24, 2362–2373. [Google Scholar] [CrossRef]
- Plumlee, G.S. The environmental geology of mineral deposis. In The Environmental Geochemistry of Mineral Deposits. Part A: Processes, Techniques and Health Issues; Plumlee, G.S., Longsdon, M.S., Eds.; Society of Economic Geologists: Littleton, CO, USA, 1999; pp. 71–116. [Google Scholar]
- Weisener, C.G.; Weber, P.A. Preferential oxidation of pyrite as a function of morphology and relict texture. N. Z. J. Geol. Geophys. 2010, 53, 167–176. [Google Scholar] [CrossRef]
- Dold, B. Dissolution kinetics of schwertmannite and ferrihydrite in oxidized mine samples and their detection by differential X-ray diffraction (DXRD). Appl. Geochem. 2003, 18, 1531–1540. [Google Scholar] [CrossRef]
- Huminicki, D.M.C.; Rimstidt, J.D. Iron oxyhydroxide coating of pyrite for acid mine drainage control. Appl. Geochem. 2009, 24, 1626–1634. [Google Scholar] [CrossRef]
- V. Nicholson, R.; Gillham, R.W.; Reardon, E.J. Pyrite oxidation in carbonate-buffered solution: 2. Rate control by oxide coatings. Geochim. Cosmochim. Acta 1990, 54, 395–402. [Google Scholar] [CrossRef]
- Stott, M.B.; Watling, H.R.; Franzmann, P.D.; Sutton, D. The role of iron-hydroxy precipitates in the passivation of chalcopyrite during bioleaching. Miner. Eng. 2000, 13, 1117–1127. [Google Scholar] [CrossRef]
- Roy, V.; Demers, I.; Plante, B.; Thériault, M. Kinetic Testing for Oxidation Acceleration and Passivation of Sulfides in Waste Rock Piles to Reduce Contaminated Neutral Drainage Generation Potential. Mine Water Environ. 2020, 39, 242–255. [Google Scholar] [CrossRef]
- Maest, A.S.; Nordstrom, D.K. A geochemical examination of humidity cell tests. Appl. Geochem. 2017, 81, 109–131. [Google Scholar] [CrossRef]
- St-Arnault, M.; Vriens, B.; Klein, B.; Mayer, K.U.; Beckie, R.D. Mineralogical controls on drainage quality during the weathering of waste rock. Appl. Geochem. 2019, 108, 104376. [Google Scholar] [CrossRef]
- Fan, R.; Short, M.D.; Zeng, S.-J.; Qian, G.; Li, J.; Schumann, R.C.; Kawashima, N.; Smart, R.S.C.; Gerson, A.R. The Formation of Silicate-Stabilized Passivating Layers on Pyrite for Reduced Acid Rock Drainage. Environ. Sci. Technol. 2017, 51, 11317–11325. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Fan, R.; Short, M.D.; Li, J.; Schumann, R.C.; Xu, H.; Smart, R.S.C.; Gerson, A.R.; Qian, G. Formation of aluminum hydroxide-doped surface passivating layers on pyrite for acid rock drainage control. Environ. Sci. Technol. 2018, 52, 11786–11795. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Hiroyoshi, N.; Tabelin, C.B.; Naruwa, K.; Harada, C.; Ito, M. Suppressive effects of ferric-catecholate complexes on pyrite oxidation. Chemosphere 2019, 214, 70–78. [Google Scholar] [CrossRef] [PubMed]
- Park, I.; Tabelin, C.B.; Seno, K.; Jeon, S.; Ito, M.; Hiroyoshi, N. Simultaneous suppression of acid mine drainage formation and arsenic release by Carrier-microencapsulation using aluminum-catecholate complexes. Chemosphere 2018, 205, 414–425. [Google Scholar] [CrossRef] [PubMed]
- Berry, V.K.; Murr, L.E.; Hiskey, J.B. Galvanic interaction between chalcopyrite and pyrite during bacterial leaching of low-grade waste. Hydrometall. 1978, 3, 309–326. [Google Scholar] [CrossRef]
- Kwong, Y.T.J.; Swerhone, G.W.; Lawrence, J.R. Galvanic sulphide oxidation as a metal-leaching mechanism and its environmental implications. Geochem. Explor. Environ. Anal. 2003, 3, 337–343. [Google Scholar] [CrossRef]
- Qian, G.; Fan, R.; Short, M.D.; Schumann, R.C.; Li, J.; St.C. Smart, R.; Gerson, A.R. The effects of galvanic interactions with pyrite on the generation of acid and metalliferous drainage. Environ. Sci. Technol. 2018, 52, 5349–5357. [Google Scholar] [CrossRef]
- Chopard, A.; Plante, B.; Benzaazoua, M.; Bouzahzah, H.; Marion, P. Geochemical investigation of the galvanic effects during oxidation of pyrite and base-metals sulfides. Chemosphere 2017, 166, 281–291. [Google Scholar] [CrossRef]
- Hudson-Edwards, K.A.; Schell, C.; Macklin, M.G. Mineralogy and geochemistry of alluvium contaminated by metal mining in the Rio Tinto area, southwest Spain. Appl. Geochem. 1999, 14, 1015–1030. [Google Scholar] [CrossRef]
- Parbhakar-Fox, A.; Lottermoser, B.; Bradshaw, D. Evaluating waste rock mineralogy and microtexture during kinetic testing for improved acid rock drainage prediction. Miner. Eng. 2013, 52, 111–124. [Google Scholar] [CrossRef]
- Parbhakar-Fox, A.K.; Edraki, M.; Walters, S.; Bradshaw, D. Development of a textural index for the prediction of acid rock drainage. Miner. Eng. 2011, 24, 1277–1287. [Google Scholar] [CrossRef]
- Brough, C.P.; Warrender, R.; Bowell, R.J.; Barnes, A.; Parbhakar-Fox, A. The process mineralogy of mine wastes. Miner. Eng. 2013, 52, 125–135. [Google Scholar] [CrossRef]
- Wang, H.; Dowd, P.A.; Xu, C. A reaction rate model for pyrite oxidation considering the influence of water content and temperature. Miner. Eng. 2019, 134, 345–355. [Google Scholar] [CrossRef]
- Dold, B. Evolution of Acid Mine Drainage Formation in Sulphidic Mine Tailings. Minerals 2014, 4, 621–641. [Google Scholar] [CrossRef]
- Sracek, O.; Gélinas, P.; Lefebvre, R.; Nicholson, R. V Comparison of methods for the estimation of pyrite oxidation rate in a waste rock pile at Mine Doyon site, Quebec, Canada. J. Geochem. Explor. 2006, 91, 99–109. [Google Scholar] [CrossRef]
- Hollings, P.; Hendry, M.J.; Nicholson, R.V.; Kirkland, R.A. Quantification of oxygen consumption and sulphate release rates for waste rock piles using kinetic cells: Cluff lake uranium mine, northern Saskatchewan, Canada. Appl. Geochem. 2001, 16, 1215–1230. [Google Scholar] [CrossRef]
- Vriens, B.; St.Arnault, M.; Laurenzi, L.; Smith, L.; Mayer, K.U.; Beckie, R.D. Localized Sulfide oxidation limited by oxygen availability in a full-scale waste-rock pile. Vadose Zone J. 2018, 17, 1–68. [Google Scholar] [CrossRef]
- Eriksson, N.; Destouni, G. Combined effects of dissolution kinetics, secondary mineral precipitation, and preferential flow on copper leaching from mining waste rock. Water Resour. Res. 1997, 33, 471–483. [Google Scholar] [CrossRef]
- Strömberg, B.; Banwart, S. Weathering kinetics of waste rock from the Aitik copper mine, Sweden: Scale dependent rate factors and pH controls in large column experiments. J. Contam. Hydrol. 1999, 39, 59–89. [Google Scholar] [CrossRef]
- Lapakko, K. Comparison of Duluth Complex rock dissolution in the laboratory and field. In Proceedings of the Proceedings American Society of Mining and Reclamation, Pittsburgh, PA, USA, 24–29 April 1994; pp. 419–428. [Google Scholar]
- Lorca, M.E.; Mayer, K.U.; Pedretti, D.; Smith, L.; Beckie, R.D. Spatial and temporal fluctuations of pore-gas composition in sulfidic mine waste rock. Vadose Zone J. 2016, 15. [Google Scholar] [CrossRef]
- Vriens, B.; Smith, L.; Mayer, K.U.; Beckie, R.D. Poregas distributions in waste-rock piles affected by climate seasonality and physicochemical heterogeneity. Appl. Geochem. 2019, 100, 305–315. [Google Scholar] [CrossRef]
- Linklater, C.M.; Sinclair, D.J.; Brown, P.L. Coupled chemistry and transport modelling of sulphidic waste rock dumps at the Aitik mine site, Sweden. Appl. Geochem. 2005, 20, 275–293. [Google Scholar] [CrossRef]
- Strömberg, B.; Banwart, S. Kinetic modelling of geochemical processes at the Aitik mining waste rock site in northern Sweden. Appl. Geochem. 1994, 9, 583–595. [Google Scholar] [CrossRef]
- Harries, J.R.; Ritchie, A.I.M. The use of temperature profiles to estimate the pyritic oxidation rate in a waste rock dump from an opencut mine. Water. Air. Soil Pollut. 1981, 15, 405–423. [Google Scholar] [CrossRef]
- Molson, J.W.; Fala, O.; Aubertin, M.; Bussière, B. Numerical simulations of pyrite oxidation and acid mine drainage in unsaturated waste rock piles. J. Contam. Hydrol. 2005, 78, 343–371. [Google Scholar] [CrossRef]
- Lefebvre, R.; Hockley, D.; Smolensky, J.; Geélinas, P. Multiphase transfer processes in waste rock piles producing acid mine drainage. 1: Conceptual model and system characterization. J. Contam. Hydrol. 2001, 52, 137–164. [Google Scholar] [CrossRef]
- Lefebvre, R.; Hockley, D.; Smolensky, J.; Lamontagne, A. Multiphase transfer processes in waste rock piles producing acid mine drainage: 2. Applications of numerical simulation. J. Contam. Hydrol. 2001, 52, 165–186. [Google Scholar] [CrossRef]
- Kuo, E.Y.; Ritchie, A.I.M. The impact of convection on the overall oxidation rate in sulfidic waste rock dumps. In Proceedings Mining and the Environment II; Goldsack, D., Belzile, N., Yerwood, P., Hall, G., Eds.; Laurentian University: Sudbury, ON, Canada, 1999; pp. 211–220. [Google Scholar]
- Lefebvre, R.; Lamontagne, A.; Wels, C. Numerical simulations of acid drainage in the Sugar Shack South rock pile, Questa Mine, New Mexico, USA. In Proceedings of the Proceedings 2nd Joint IAH-CNC and CGS Groundwater Specialty Conference, 54th Canadian Geotechnical Conference, Calgary, AB, Canada, 16–19 September 2001. [Google Scholar]
- Karlsson, T.; Räisänen, M.L.; Lehtonen, M.; Alakangas, L. Comparison of static and mineralogical ARD prediction methods in the Nordic environment. Environ. Monit. Assess. 2018, 190, 719. [Google Scholar] [CrossRef]
- Chopard, A.; Benzaazoua, M.; Bouzahzah, H.; Plante, B.; Marion, P. A contribution to improve the calculation of the acid generating potential of mining wastes. Chemosphere 2017, 175, 97–107. [Google Scholar] [CrossRef] [PubMed]
- Schumann, R.; Stewart, W.; Miller, S.; Kawashima, N.; Li, J.; Smart, R. Acid–base accounting assessment of mine wastes using the chromium reducible sulfur method. Sci. Total Environ. 2012, 424, 289–296. [Google Scholar] [CrossRef] [PubMed]
- Weber, P.A.; Thomas, J.E.; Skinner, W.M.; Smart, R.S.C. A methodology to determine the acid-neutralization capacity of rock samples. Can. Mineral. 2005, 43, 1183–1192. [Google Scholar] [CrossRef]
- Paktunc, A.D. Mineralogical constraints on the determination of neutralization potential and prediction of acid mine drainage. Environ. Geol. 1999, 39, 103–112. [Google Scholar] [CrossRef]
- Skousen, J.; Renton, J.; Brown, H.; Evans, P.; Leavitt, B.; Brady, K.; Cohen, L.; Ziemkiewicz, P. Neutralization potential of overburden samples containing siderite. J. Environ. Qual. 1997, 26, 673–681. [Google Scholar] [CrossRef]
- Weber, P.A.; Thomas, J.E.; Skinner, W.M.; Smart, R.S.C. Improved acid neutralisation capacity assessment of iron carbonates by titration and theoretical calculation. Appl. Geochem. 2004, 19, 687–694. [Google Scholar] [CrossRef]
- Skousen, J.; Simmons, J.; McDonald, L.M.; Ziemkiewicz, P. Acid–base accounting to predict post-mining drainage quality on surface mines. J. Environ. Qual. 2002, 31, 2034–2044. [Google Scholar] [CrossRef] [PubMed]
- Morin, K.A.; Hutt, N.M. On the Nonsense of Arguing the Superiority of an Analytical Method for Neutralization Potential. Minesite Drainage Assessment Group, case study# 32. Available online: www.mdag.com/case_studies/cs32.html (accessed on 1 July 2020).
- Bouzahzah, H.; Benzaazoua, M.; Plante, B.; Bussiere, B. A quantitative approach for the estimation of the “fizz rating” parameter in the acid-base accounting tests: A new adaptations of the Sobek test. J. Geochem. Explor. 2015, 153, 53–65. [Google Scholar] [CrossRef]
- Bouzahzah, H.; Benzaazoua, M.; Bussiere, B.; Plante, B. Prediction of acid mine drainage: Importance of mineralogy and the test protocols for static and kinetic tests. Mine Water Environ. 2014, 33, 54–65. [Google Scholar] [CrossRef]
- Gerson, A.R.; Rolley, P.J.; Davis, C.; Feig, S.T.; Doyle, S.; Smart, R.S.C. Unexpected non-acid drainage from sulfidic rock waste. Sci. Rep. 2019, 9, 4357. [Google Scholar] [CrossRef]
- Pope, J.; Weber, P.; Mackenzie, A.; Newman, N.; Rait, R. Correlation of acid base accounting characteristics with the Geology of commonly mined coal measures, West Coast and Southland, New Zealand. N. Z. J. Geol. Geophys. 2010, 53, 153–166. [Google Scholar] [CrossRef]
- Miller, S.D.; Stewart, W.S.; Rusdinar, Y.; Schumann, R.E.; Ciccarelli, J.M.; Li, J.; Smart, R.S.C. Methods for estimation of long-term non-carbonate neutralisation of acid rock drainage. Sci. Total Environ. 2010, 408, 2129–2135. [Google Scholar] [CrossRef] [PubMed]
- Jambor, J.L.; Dutrizac, J.E.; Raudsepp, M. Measured and computed neutralization potentials from static tests of diverse rock types. Environ. Geol. 2007, 52, 1173–1185. [Google Scholar] [CrossRef]
- Sapsford, D.J.; Bowell, R.J.; Dey, M.; Williams, K.P. Humidity cell tests for the prediction of acid rock drainage. Miner. Eng. 2009, 22, 25–36. [Google Scholar] [CrossRef]
- Strömberg, B.; Banwart, S.A. Experimental study of acidity-consuming processes in mining waste rock: Some influences of mineralogy and particle size. Appl. Geochem. 1999, 14, 1–16. [Google Scholar] [CrossRef]
- Smith, L.J.D.; Bailey, B.L.; Blowes, D.W.; Jambor, J.L.; Smith, L.; Sego, D.C. The Diavik waste rock project: Initial geochemical response from a low sulfide waste rock pile. Appl. Geochem. 2013, 36, 210–221. [Google Scholar] [CrossRef]
- Pham, N.H.; Sego, D.C.; Arenson, L.U.; Blowes, D.W.; Amos, R.T.; Smith, L. The Diavik Waste Rock Project: Measurement of the thermal regime of a waste-rock test pile in a permafrost environment. Appl. Geochem. 2013, 36, 234–245. [Google Scholar] [CrossRef]
- Wilson, D.; Amos, R.; W. Blowes, D.; B. Langman, J.; Smith, L.; C. Sego, D. Diavik Waste Rock Project: Scale-up of a reactive transport model for temperature and sulfide-content dependent geochemical evolution of waste rock. Appl. Geochem. 2018, 96, 177–190. [Google Scholar] [CrossRef]
- Langman, B.J.; Moore, L.M.; Ptacek, J.C.; Smith, L.; Sego, D.; Blowes, W.D. Diavik Waste Rock Project: Evolution of mineral weathering, element release, and acid generation and neutralization during a five-year humidity cell experiment. Minerals 2014, 4, 257–278. [Google Scholar] [CrossRef]
- Blackmore, S.; Smith, L.; Ulrich Mayer, K.; Beckie, R.D. Comparison of unsaturated flow and solute transport through waste rock at two experimental scales using temporal moments and numerical modeling. J. Contam. Hydrol. 2014, 171, 49–65. [Google Scholar] [CrossRef]
- Blackmore, S.; Pedretti, D.; Mayer, K.U.; Smith, L.; Beckie, R.D. Evaluation of single- and dual-porosity models for reproducing the release of external and internal tracers from heterogeneous waste-rock piles. J. Contam. Hydrol. 2018, 214, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Lahmira, B.; Lefebvre, R.; Aubertin, M.; Bussière, B. Effect of heterogeneity and anisotropy related to the construction method on transfer processes in waste rock piles. J. Contam. Hydrol. 2016, 184, 35–49. [Google Scholar] [CrossRef] [PubMed]
- Pearce, S.; Birkham, T.; O’Kane, M.; Dobchuk, D. Linking Waste Rock Dump Construction and Design with AMD Risk: A Quantitative Approach; British Columbia Mine Reclamation Symposium, The University of British Columbia: Vancouver, BC, Canada, 2016. [Google Scholar] [CrossRef]
- Parbhakar-Fox, A.; Fox, N.; Jackson, L.; Cornelius, R. Forecasting geoenvironmental risks: Integrated applications of mineralogical and chemical data. Minerals 2018, 8, 541. [Google Scholar] [CrossRef]
- Pedretti, D.; Mayer, K.U.; Beckie, R.D. Stochastic multicomponent reactive transport analysis of low quality drainage release from waste rock piles: Controls of the spatial distribution of acid generating and neutralizing minerals. J. Contam. Hydrol. 2017, 201, 30–38. [Google Scholar] [CrossRef] [PubMed]
- Pedretti, D.; Mayer, K.U.; Beckie, R.D. Controls of uncertainty in acid rock drainage predictions from waste rock piles examined through Monte-Carlo multicomponent reactive transport. Stoch. Environ. Res. Risk Assess. 2020, 34, 219–233. [Google Scholar] [CrossRef]
- Lahmira, B.; Lefebvre, R.; Aubertin, M.; Bussière, B. Effect of material variability and compacted layers on transfer processes in heterogeneous waste rock piles. J. Contam. Hydrol. 2017, 204, 66–78. [Google Scholar] [CrossRef] [PubMed]
- Swanson, D.A.; Barbour, S.L.; Wilson, G.W.; O’Kane, M. Soil-atmosphere modelling of an engineered soil cover for acid generating mine waste in a humid, alpine climate. Can. Geotech. J. 2003, 40, 276–292. [Google Scholar] [CrossRef]
- Nicholls, E.M.; Drewitt, G.B.; Fraser, S.; Carey, S.K. The influence of vegetation cover on evapotranspiration atop waste rock piles, Elk Valley, British Columbia. Hydrol. Process. 2019, 33, 2594–2606. [Google Scholar] [CrossRef]
- Carey, S.K.; Barbour, S.L.; Hendry, M.J. Evaporation from a waste-rock surface, Key Lake, Saskatchewan. Can. Geotech. J. 2005, 42, 1189–1199. [Google Scholar] [CrossRef]
- Birkham, T.; O’Kane, M.; Goodbrand, A.; Barbour, S.L.; Carey, S.K.; Straker, J.; Baker, T.; Klein, R. Near-surface water balances of waste rock dumps. In Proceedings of the British Columbia Mine Reclamation Symposium, Prince George, BC, Canada, 22–25 September 2014. [Google Scholar]
- Peterson, H.E. Unsaturated Hydrology, Evaporation, and Geochemistry of Neutral and Acid Rock Drainage in Highly Heterogeneous Mine Waste Rock at the Antamina Mine, Peru. Ph.D. Thesis, The University of British Columbia, Vancouver, BC, Canada, 2014. [Google Scholar]
- Rohde, T.K.; Defferrard, P.L.; Lord, M. Store and release cover water balance for the south waste rock dump at Century mine. In Proceedings of the 11th International Conference on Mine Closure 2016, Perth, Australia, 15–17 March 2016; Fourie, A.B., Fourie, A.B., Tibbett, M., Tibbett, M., Eds.; Australian Centre for Geomechanics PP—Perth: Perth, Australia, 2016; pp. 47–59. [Google Scholar]
- Chi, X.; Amos, R.T.; Stastna, M.; Blowes, D.W.; Sego, D.C.; Smith, L. The Diavik Waste Rock Project: Implications of wind-induced gas transport. Appl. Geochem. 2013, 36, 246–255. [Google Scholar] [CrossRef]
- Sjoberg, D.B.; Lee, B.S.; Jian, Z. Prediction of water vapor movement through waste rock. J. Geotech. Geoenvironmental Eng. 2004, 130, 293–302. [Google Scholar] [CrossRef]
- Haghighi, E.; Or, D. Evaporation from porous surfaces into turbulent airflows: Coupling eddy characteristics with pore scale vapor diffusion. Water Resour. Res. 2013, 49, 8432–8442. [Google Scholar] [CrossRef]
- Wilson, G.W.; Fredlund, D.G.; Barbour, S.L. The effect of soil suction on evaporative fluxes from soil surfaces. Can. Geotech. J. 1997, 34, 145–155. [Google Scholar] [CrossRef]
- Ramasamy, M.; Power, C. Evolution of acid mine drainage from a coal waste rock pile reclaimed with a simple soil cover. Hydrology 2019, 6, 83. [Google Scholar] [CrossRef]
- Neuner, M.; Smith, L.; Blowes, D.W.; Sego, D.C.; Smith, L.J.D.; Fretz, N.; Gupton, M. The Diavik waste rock project: Water flow through mine waste rock in a permafrost terrain. Appl. Geochem. 2013, 36, 222–233. [Google Scholar] [CrossRef]
- Trinchero, P.; Beckie, R.; Sanchez-Vila, X.; Nichol, C. Assessing preferential flow through an unsaturated waste rock pile using spectral analysis. Water Resour. Res. 2011, 47, W07532. [Google Scholar] [CrossRef]
- Šimůnek, J.; Jarvis, N.J.; van Genuchten, M.T.; Gärdenäs, A. Review and comparison of models for describing non-equilibrium and preferential flow and transport in the vadose zone. J. Hydrol. 2003, 272, 14–35. [Google Scholar] [CrossRef]
- Nimmo, J.R. Simple predictions of maximum transport rate in unsaturated soil and rock. Water Resour. Res. 2007, 43. [Google Scholar] [CrossRef]
- Gerke, H.H. Preferential flow descriptions for structured soils. J. Plant. Nutr. Soil Sci. 2006, 169, 382–400. [Google Scholar] [CrossRef]
- Jarvis, N.J. A review of non-equilibrium water flow and solute transport in soil macropores: Principles, controlling factors and consequences for water quality. Eur. J. Soil Sci. 2007, 58, 523–546. [Google Scholar] [CrossRef]
- Shahhosseini, M.; Doulati Ardejani, F.; Amini, M.; Ebrahimi, L. The spatial assessment of acid mine drainage potential within a low-grade ore dump: The role of preferential flow paths. Environ. Earth Sci. 2019, 79, 28. [Google Scholar] [CrossRef]
- Nichol, C.; Smith, L.; Beckie, R. Field-scale experiments of unsaturated flow and solute transport in a heterogeneous porous medium. Water Resour. Res. 2005, 41, 1–11. [Google Scholar] [CrossRef]
- Eriksson, N.; Gupta, A.; Destouni, G. Comparative analysis of laboratory and field tracer tests for investigating preferential flow and transport in mining waste rock. J. Hydrol. 1997, 194, 143–163. [Google Scholar] [CrossRef]
- Sinclair, S.A.; Pham, N.; Amos, R.T.; Sego, D.C.; Smith, L.; Blowes, D.W. Influence of freeze–thaw dynamics on internal geochemical evolution of low sulfide waste rock. Appl. Geochem. 2015, 61, 160–174. [Google Scholar] [CrossRef]
- Langman, J.B.; Blowes, D.W.; Amos, R.T.; Atherton, C.; Wilson, D.; Smith, L.; Sego, D.C.; Sinclair, S.A. Influence of a tundra freeze-thaw cycle on sulfide oxidation and metal leaching in a low sulfur, granitic waste rock. Appl. Geochem. 2017, 76, 9–21. [Google Scholar] [CrossRef]
- Kurylyk, B.L.; Watanabe, K. The mathematical representation of freezing and thawing processes in variably-saturated, non-deformable soils. Adv. Water Resour. 2013, 60, 160–177. [Google Scholar] [CrossRef]
- Birkham, T.K.; Hendry, M.J.; Wassenaar, L.I.; Mendoza, C.A.; Lee, E.S. Characterizing Geochemical Reactions in Unsaturated Mine Waste-Rock Piles Using Gaseous O2, CO2, 12CO2, and 13CO2. Environ. Sci. Technol. 2003, 37, 496–501. [Google Scholar] [CrossRef]
- Elberling, B.; Nicholson, R.V.; Scharer, J.M. A combined kinetic and diffusion model for pyrite oxidation in tailings: A change in controls with time. J. Hydrol. 1994, 157, 47–60. [Google Scholar] [CrossRef]
- Pantelis, G.; Ritchie, A.I.M. Rate-limiting factors in dump leaching of pyritic ores. Appl. Math. Model. 1992, 16, 553–560. [Google Scholar] [CrossRef]
- Massmann, J.; Farrier, D. Effects of atmospheric pressures on gas transport in the vadose zone. Water Resour. Res. 1992, 28, 777–791. [Google Scholar] [CrossRef]
- Amos, R.T.; Blowes, D.W.; Smith, L.; Sego, D.C. Measurement of Wind-Induced Pressure Gradients in a Waste Rock Pile. Vadose Zone J. 2009, 8, 953–962. [Google Scholar] [CrossRef]
- Lahmira, B.; Lefebvre, R.; Hockley, D.; Phillip, M. Atmospheric Controls on Gas Flow Directions in a Waste Rock Dump. Vadose Zone J. 2014, 13. [Google Scholar] [CrossRef]
- Pantelis, G.; Ritchie, A.I.M.; Stepanyants, Y.A. A conceptual model for the description of oxidation and transport processes in sulphidic waste rock dumps. Appl. Math. Model. 2002, 26, 751–770. [Google Scholar] [CrossRef]
- Collin, M.; Rasmuson, A. A comparison of gas diffusivity models for unsaturated porous media. Soil Sci. Soc. Am. J. 1988, 52, 1559–1565. [Google Scholar] [CrossRef]
- Millington, R.J.; Quirk, J.P. Permeability of porous solids. Trans. Faraday Soc. 1961, 57, 1200–1207. [Google Scholar] [CrossRef]
- Wang, T.; Huang, Y.; Chen, X.; Chen, X. Using grain-size distribution methods for estimation of air permeability. Groundwater 2016, 54, 131–142. [Google Scholar] [CrossRef]
- Rohwerder, T.; Schippers, A.; Sand, W. Determination of reaction energy values for biological pyrite oxidation by calorimetry. Thermochim. Acta 1998, 309, 79–85. [Google Scholar] [CrossRef]
- Sracek, O.; Choquette, M.; Gélinas, P.; Lefebvre, R.; Nicholson, R. V Geochemical characterization of acid mine drainage from a waste rock pile, Mine Doyon, Québec, Canada. J. Contam. Hydrol. 2004, 69, 45–71. [Google Scholar] [CrossRef]
- Tan, Y.; Ritchie, A.I.M. In situ determination of thermal conductivity of waste rock dump material. Water. Air. Soil Pollut. 1997, 98, 345–359. [Google Scholar] [CrossRef]
- Ning, L.; Zhang, Y. Onset of thermally induced gas convection in mine wastes. Int. J. Heat Mass Transf. 1997, 40, 2621–2636. [Google Scholar] [CrossRef]
- Gou, W.; Parizek, R.R.; Rose, A.W. The role of thermal convection in resupplying O2 to strip coal-mine spoil. Soil Sci. 1994, 158, 47–55. [Google Scholar] [CrossRef]
- Anterrieu, O.; Chouteau, M.; Aubertin, M. Geophysical characterization of the large-scale internal structure of a waste rock pile from a hard rock mine. Bull. Eng. Geol. Environ. 2010, 69, 533–548. [Google Scholar] [CrossRef]
- Dobriyal, P.; Qureshi, A.; Badola, R.; Hussain, S.A. A review of the methods available for estimating soil moisture and its implications for water resource management. J. Hydrol. 2012, 458, 110–117. [Google Scholar] [CrossRef]
- Appels, W.M.; Ireson, A.M.; Barbour, S.L. Impact of bimodal textural heterogeneity and connectivity on flow and transport through unsaturated mine waste rock. Adv. Water Resour. 2018, 112, 254–265. [Google Scholar] [CrossRef]
- Babaeian, E.; Sadeghi, M.; Jones, S.B.; Montzka, C.; Vereecken, H.; Tuller, M. Ground, proximal, and satellite remote sensing of soil moisture. Rev. Geophys. 2019, 57, 530–616. [Google Scholar] [CrossRef]
- Nichol, C.; Smith, L.; Beckie, R. Time domain reflectrometry measurements of water content in coarse waste rock. Can. Geotech. J. 2003, 40, 137–148. [Google Scholar] [CrossRef]
- Smith, L.J.D.; Moncur, M.C.; Neuner, M.; Gupton, M.; Blowes, D.W.; Smith, L.; Sego, D.C. The Diavik waste rock project: Design, construction, and instrumentation of field-scale experimental waste-rock piles. Appl. Geochem. 2013, 36, 187–199. [Google Scholar] [CrossRef]
- Dimech, A.; Chouteau, M.; Aubertin, M.; Bussière, B.; Martin, V.; Plante, B. Three-dimensional time-lapse geoelectrical monitoring of water infiltration in an experimental mine waste rock pile. Vadose Zone J. 2019, 18. [Google Scholar] [CrossRef]
- Dubuc, J.; Pabst, T.; Aubertin, M. An assessment of the hydrogeological response of the flow control layer installed on the experimental waste rock pile at the lac Tio mine. In Proceedings of the GeoOttawa 2017—70th Canadian Geotechnical Conference, Ottawa, ON, Canada, 1–4 October 2017. [Google Scholar]
- Keller, J.; Busker, L.; Milczarek, M.; Rice, R.; Williamson, M. Monitoring of the geochemical evolution of waste rock facilities at Newmont’s Phoenix Mine. In Proceedings of the VI International Seminar on Mine Closure, Lake Louise, AB, Canada, 18–21 September 2011. [Google Scholar]
- Poisson, J.; Chouteau, M.; Aubertin, M.; Campos, D. Geophysical experiments to image the shallow internal structure and the moisture distribution of a mine waste rock pile. J. Appl. Geophys. 2009, 67, 179–192. [Google Scholar] [CrossRef]
- Van Dam, R.; Gutierrez, L.; Mclemore, V.; Wilson, G.; Hendrickx, J.; Walker, B. Near Surface Geophysics for the Structural Analysis of a Mine Rock Pile, Northern New Mexico. J. Am. Soc. Min. Reclam. 2005, 2005, 1178–1201. [Google Scholar] [CrossRef]
- Liu, X.; Chen, J.; Cui, X.; Liu, Q.; Cao, X.; Chen, X. Measurement of soil water content using ground-penetrating radar: A review of current methods. Int. J. Digit. Earth 2019, 12, 95–118. [Google Scholar] [CrossRef]
- Power, C.; Tsourlos, P.; Ramasamy, M.; Nivorlis, A.; Mkandawire, M. Combined DC resistivity and induced polarization (DC-IP) for mapping the internal composition of a mine waste rock pile in Nova Scotia, Canada. J. Appl. Geophys. 2018, 150, 40–51. [Google Scholar] [CrossRef]
- Wu, R.; Martin, V.; McKenzie, J.; Broda, S.; Bussière, B.; Aubertin, M.; Kurylyk, B.L. Laboratory-scale assessment of a capillary barrier using fibre optic distributed temperature sensing (FO-DTS). Can. Geotech. J. 2020, 57, 115–126. [Google Scholar] [CrossRef]
- Li, Y.; Topal, E.; Ramazan, S. Optimising the long-term mine waste management and truck schedule in a large-scale open pit mine. Min. Technol. 2016, 125, 35–46. [Google Scholar] [CrossRef]
- Seigneur, N.; Ulrich Mayer, K.; Steefel, C.I. Reactive transport in evolving porous media. In Reviews in Mineralogy and Geochemistry; Mineralogical Society: Twickenham, UK, 2019; pp. 197–238. [Google Scholar]
- Beckingham, L.E.; Mitnick, E.H.; Steefel, C.I.; Zhang, S.; Voltolini, M.; Swift, A.M.; Yang, L.; Cole, D.R.; Sheets, J.M.; Ajo-Franklin, J.B.; et al. Evaluation of mineral reactive surface area estimates for prediction of reactivity of a multi-mineral sediment. Geochim. Cosmochim. Acta 2016, 188, 310–329. [Google Scholar] [CrossRef]
- Steefel, C.I.; Lichtner, P.C. Multicomponent reactive transport in discrete fractures: I. Controls on reaction front geometry. J. Hydrol. 1998, 209, 186–199. [Google Scholar] [CrossRef]
- Wunderly, D.M.; Blowes, D.W.; Frind, O.E.; Ptacek, C. Sulfide mineral oxidation and subsequent reactive transport of oxidation products in mine tailings impoundments: A numerical model. Water Resour. Res. 1996, 32, 3173–3187. [Google Scholar] [CrossRef]
- Maest, A.S.; Kuipers, J.R.; Travers, C.L.; Atkins, D.A. Predicting Water Quality at Hardrock Mines; 90 pages, Kuipers & Associates and Buka Environmental. Available online: http://pebblescience.org/Pebble-Mine/acid-drainage-pdfs/PredictionsReportFinal.pdf (accessed on 1 July 2020).
- Box, G.E.P. Science and statistics. J. Am. Stat. Assoc. 1976, 71, 791–799. [Google Scholar] [CrossRef]
- Muniruzzaman, M.; Kauppila, P.M.; Karlsson, T. Water Quality Prediction of Mining Waste Facilities Based on Predictive Models; GTK Open File Research Report 16/2018; GTK: Espoo, Finland, 2018; Available online: http://tupa.gtk.fi/raportti/arkisto/16_2018.pdf (accessed on 1 August 2020).
- Beckie, R. Analysis of Scale Effects in Large-Scale Solute-Transport Models. In Scale Dependence and Scale Invariance in Hydrology; Sposito, G., Ed.; Cambridge University Press: Cambridge, UK, 1998; pp. 314–334. ISBN 9780521571258. [Google Scholar]
- Malmström, M.E.; Destouni, G.; Banwart, S.A.; Strömberg, B.H.E. Resolving the scale-dependence of mineral weathering rates. Environ. Sci. Technol. 2000, 34, 1375–1378. [Google Scholar] [CrossRef]
- Morin, K.A.; Hutt, N.M. Environmental Geochemistry of Minesite Drainage: Practical Theory and Case Studies, Digital Edition; MDAG Publishing: Surrey, BC, Canada, 2001. [Google Scholar]
- Molins, S.; Knabner, P. Multiscale approaches in reactive transport modeling. Rev. Mineral. Geochem. 2019, 85, 27–48. [Google Scholar] [CrossRef]
- Molson, J.; Aubertin, M.; Bussière, B. Reactive transport modelling of acid mine drainage within discretely fractured porous media: Plume evolution from a surface source zone. Environ. Model. Softw. 2012, 38, 259–270. [Google Scholar] [CrossRef]
- Pabst, T.; Molson, J.; Aubertin, M.; Bussière, B. Reactive transport modelling of the hydro-geochemical behaviour of partially oxidized acid-generating mine tailings with a monolayer cover. Appl. Geochem. 2017, 78, 219–233. [Google Scholar] [CrossRef]
- Tomiyama, S.; Igarashi, T.; Tabelin, C.B.; Tangviroon, P.; Hiroyuki, I. Modeling of the groundwater flow system in excavated areas of an abandoned mine. J. Cont. Hydr. 2020, 230, 103617. [Google Scholar] [CrossRef]
- Mayer, K.U.; Frind, E.O.; Blowes, D.W. Multicomponent reactive transport modeling in variably saturated porous media using a generalized formulation for kinetically controlled reactions. Water Resour. Res. 2002, 38, 13–21. [Google Scholar] [CrossRef]
- Parkhurst, D.L.; Appelo, C.A.J. User’s guide to PHREEQC (Version 2): A computer program for speciation, batch-reaction, one-dimensional transport, and inverse geochemical calculations. USGS Water Resources Investigations Report 99-4259. Available online: https://pubs.er.usgs.gov/publication/wri994259 (accessed on 1 July 2020).
- van der Lee, J.; De Windt, L.; Lagneau, V.; Goblet, P. Module-oriented modeling of reactive transport with HYTEC. Comput. Geosci. 2003, 29, 265–275. [Google Scholar] [CrossRef]
- Xu, T.; Sonnenthal, E.; Spycher, N.; Pruess, K. TOUGHREACT User’s Guide: A Simulation Program for Non-Isothermal Multiphase Reactive Geochemical Transport in Variable Saturated Geologic Media; University of California: Berkeley, CA, USA, 2004. [Google Scholar] [CrossRef]
- Steefel, C.I.; Appelo, C.A.J.; Arora, B.; Jacques, D.; Kalbacher, T.; Kolditz, O.; Lagneau, V.; Lichtner, P.C.; Mayer, K.U.; Meeussen, J.C.L.; et al. Reactive transport codes for subsurface environmental simulation. Comput. Geosci. 2015, 19, 445–478. [Google Scholar] [CrossRef]
- Raymond, K.; Seigneur, N.; Su, D.; Plante, B.; Poaty, B.; Bussiere, B.; Mayer, K. Numerical modeling of a laboratory-scale waste rock pile featuring an engineered cover system. Minerals 2020, 10, 652. [Google Scholar] [CrossRef]
- Li, L.; Maher, K.; Navarre-Sitchler, A.; Druhan, J.; Meile, C.; Lawrence, C.; Moore, J.; Perdrial, J.; Sullivan, P.; Thompson, A.; et al. Expanding the role of reactive transport models in critical zone processes. Earth-Sci. Rev. 2017, 165, 280–301. [Google Scholar] [CrossRef]
- Wösten, J.H.M.; van Genuchten, M.T. Using texture and other soil properties to predict the unsaturated soil hydraulic functions. Soil Sci. Soc. Am. J. 1988, 52, 1762–1770. [Google Scholar] [CrossRef]
- Arya, L.M.; Leij, F.J.; Shouse, P.J.; van Genuchten, M.T. Relationship between the Hydraulic Conductivity Function and the Particle-Size Distribution. Soil Sci. Soc. Am. J. 1999, 63, 1063–1070. [Google Scholar] [CrossRef]
- Mishra, S.; Parker, J.C.; Singhal, N. Estimation of soil hydraulic properties and their uncertainty from particle size distribution data. J. Hydrol. 1989, 108, 1–18. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).