M-Polynomials and Topological Indices of Titania Nanotubes
Abstract
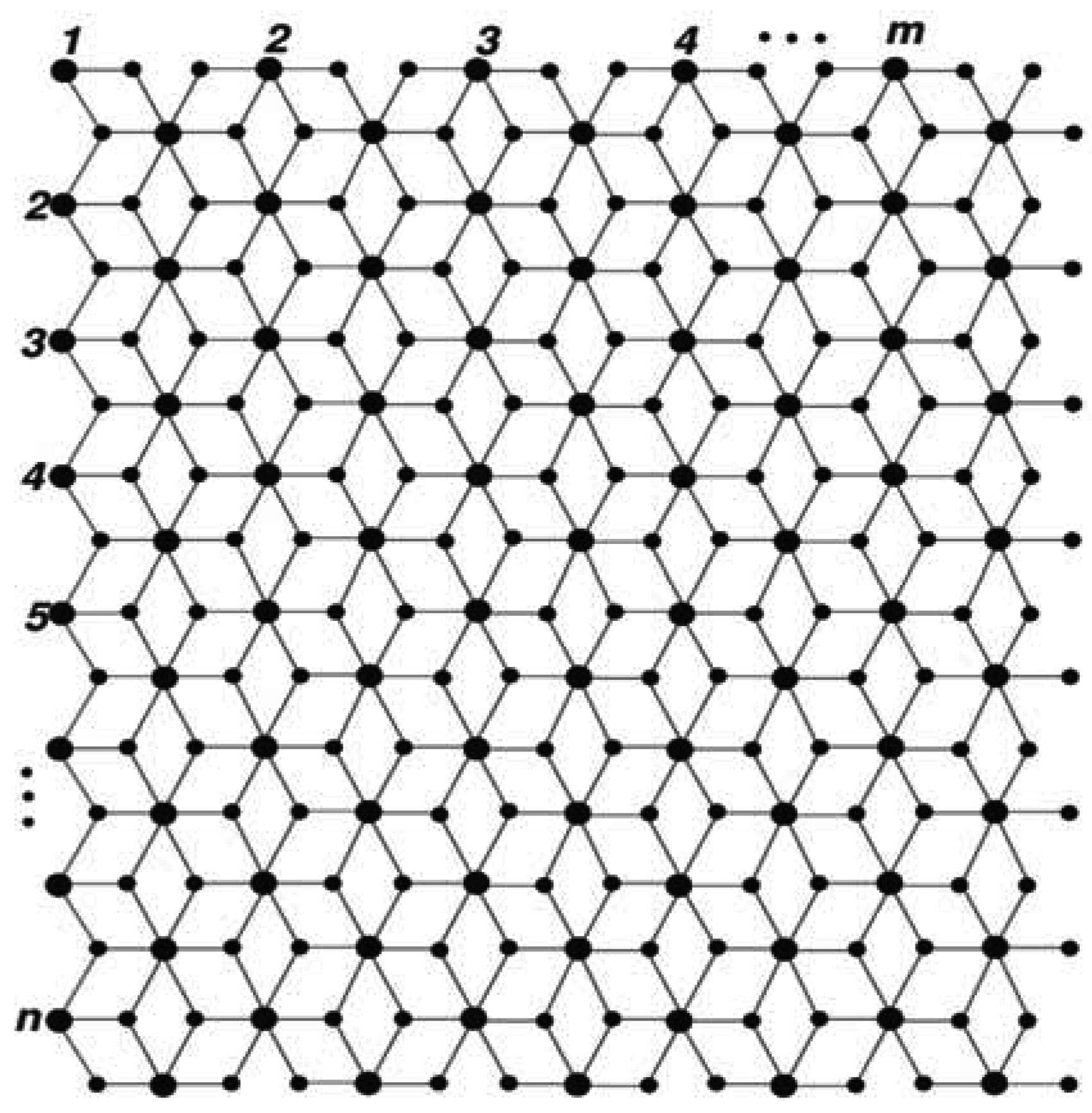
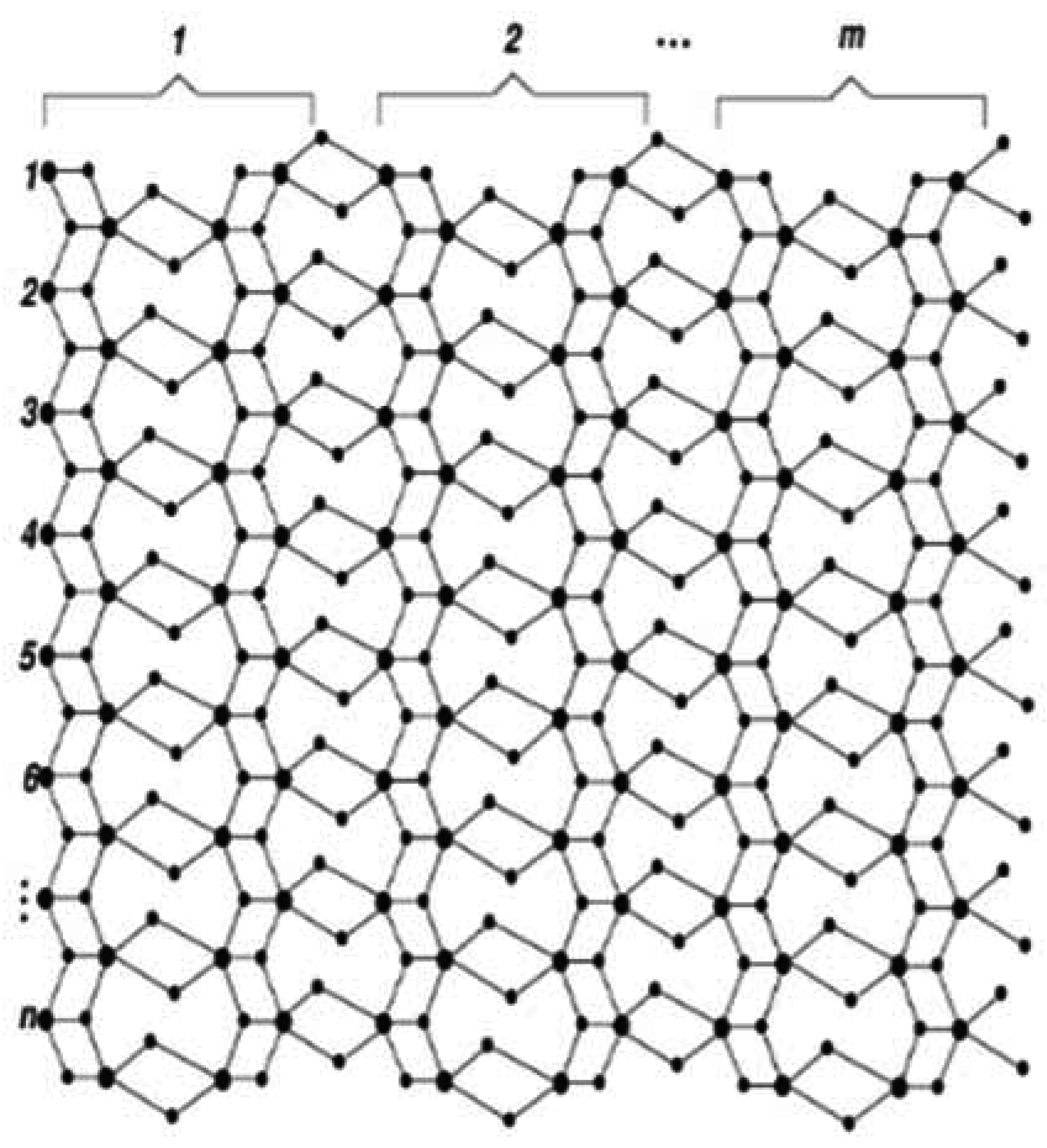
:1. Introduction
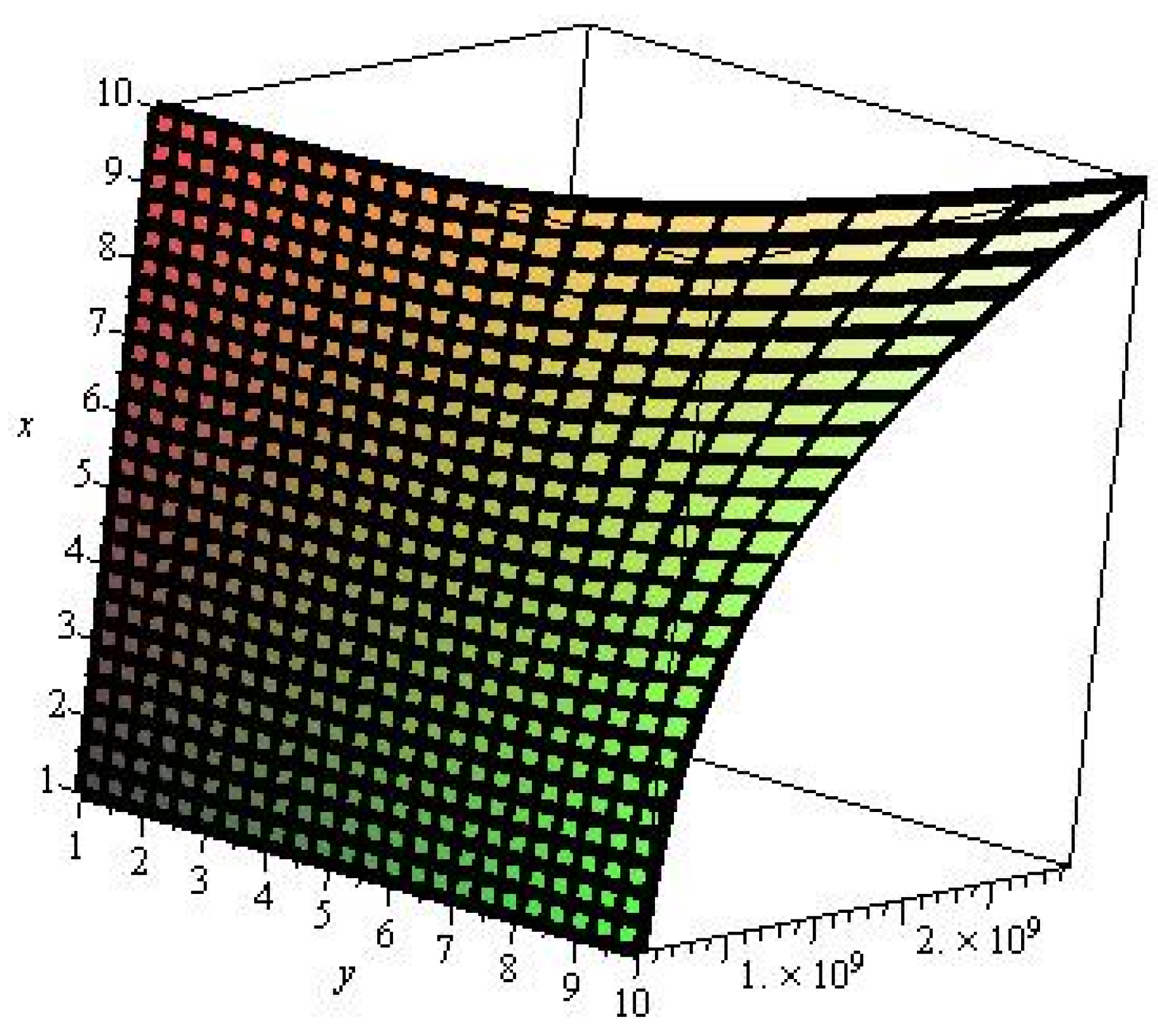
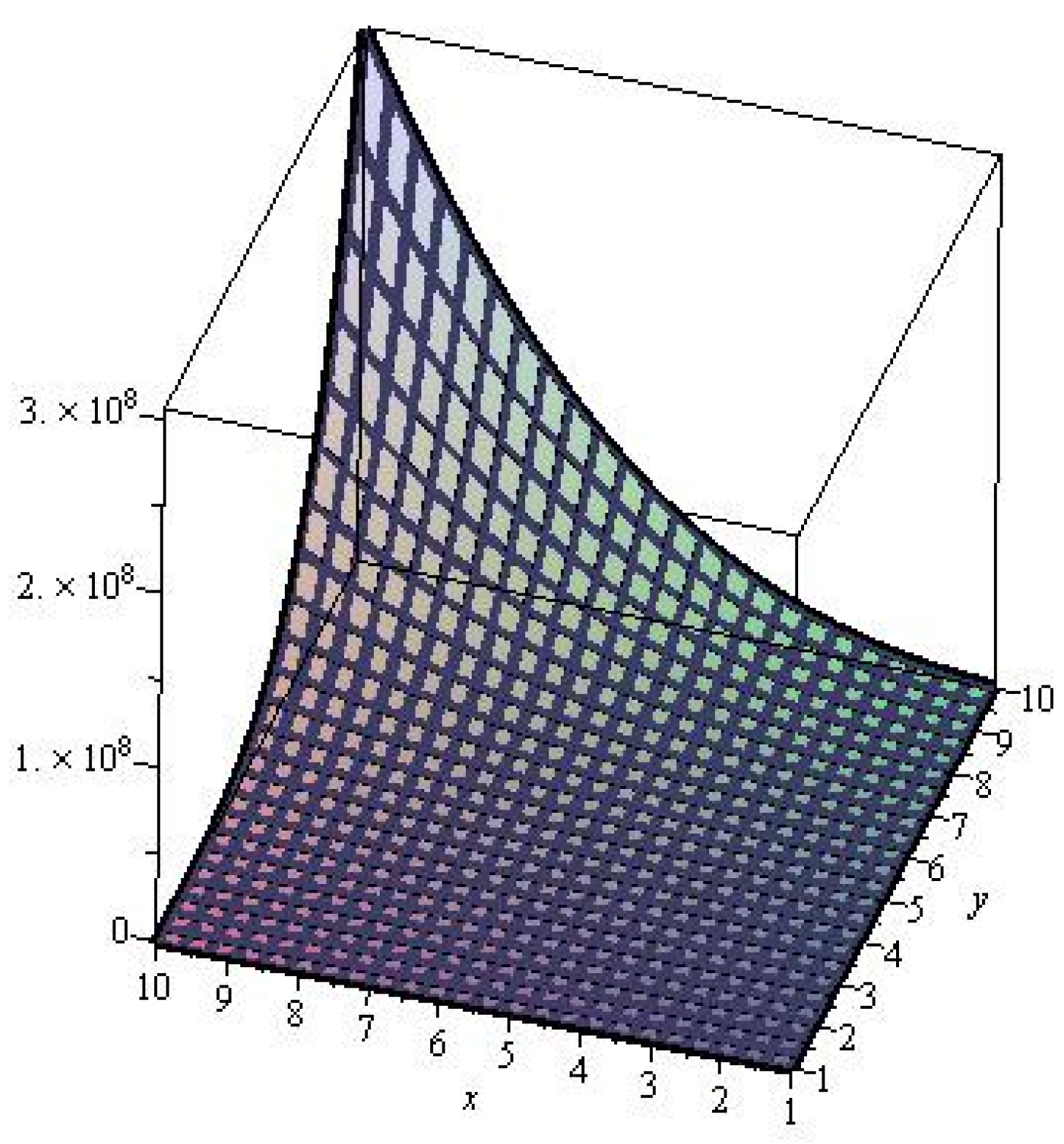
2. Results
3. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Li, Y.Z.; Lee, N.H.; Lee, E.G.; Song, J.S.; Kim, S.J. The characterization and photocatalytic properties of mesoporous rutile TiO2 powder synthesized through cell assembly of nanocrystals. Chem. Phys. Lett. 2004, 389, 124–128. [Google Scholar] [CrossRef]
- Bavykin, D.V.; Friedrich, J.M.; Walash, F.C. Protonated titanates and TiO2 nanostructured materials: Synthesis, properties and applications. Adv. Mater. 2006, 18, 2807–2824. [Google Scholar] [CrossRef]
- Wang, W.; Varghese, O.K.; Paulsose, M.; Grimes, C.A. A study on the growth and structure of Titania nonotubes. J. Mater. Res. 2004, 19, 417–422. [Google Scholar] [CrossRef]
- Evarestov, R.A.; Bandura, A.V.; Losev, M.V.; Piskunov, S.; Zhukovskii, Y.F. Titania nanotubes modeled from 3-layered and 6-layered(101) anatase sheets: Line group symmetry and comparative ab initio LCAO calculataions. Physica E 2010, 43, 266–278. [Google Scholar] [CrossRef]
- Gutman, I. Degree based topological indices. Croat. Chem. Acta 2013, 89, 351–361. [Google Scholar] [CrossRef]
- Wiener, H.J. Structural determination of paraffin boiling points. J. Am. Chem. Soc. 1947, 69, 17–20. [Google Scholar] [CrossRef] [PubMed]
- Gutman, I.; Trinajstic, N. Graph theory and molecular orbitals total f-electron energy of alternant hydrocarbons. Chem. Phys. Lett. 1972, 17, 535–538. [Google Scholar] [CrossRef]
- Hao, J. Theorems about Zagreb Indices and Modified Zagreb Indices. MATCH Commun. Math. Comput. Chem. 2011, 65, 659–670. [Google Scholar]
- Li, X.; Shi, Y. A Survey on the Randic Index. MATCH Commun. Math. Comput. Chem. 2008, 59, 127–156. [Google Scholar]
- Xu, K.; Liu, M.; Das, K.C.; Gutman, I.; Furtula, B. A survey on graphs extremal with respect to distance-based topological indices. MATCH Commun. Math. Comput. Chem. 2014, 71, 461–508. [Google Scholar]
- Munir, M.; Nazeer, W.; Nizami, A.R.; Rafique, S.; Kang, S.M. M-polynomial and degree-based topological indices of Buckytubes. Symmetry 2016. [Google Scholar] [CrossRef]
- Eliasi, M.; Iranmanesh, A.; Gutman, I. Multiplicative version of first Zagreb index. MATCH Commun. Math. Comput. Chem. 2012, 68, 217–230. [Google Scholar]
- Furtula, B.; Gutman, I.; Dehmer, M. On structure-sensitivity of degree-based topological indices. Appl. Math. Comput. 2013, 219, 8973–8978. [Google Scholar] [CrossRef]
- Gutman, I.; Das, K.C. Some properties of the Second Zagreb Index. MATCH Commun. Math. Comput. Chem. 2004, 50, 103–112. [Google Scholar]
- Yan, L.; Gao, W.; Li, J.S. General harmonic index and general sum connectivity index of polyomino chains and nanotubes. J. Comput. Theor. Nanosci. 2015, 12, 3940–3944. [Google Scholar] [CrossRef]
- Gao, W.; Shi, L. Wiener index of gear fan graph and gear wheel graph. Asian J. Chem. 2014, 26, 3397–3400. [Google Scholar]
- Gao, W.; Wang, W. Second atom-bond connectivity index of special chemical molecular structures. J. Chem. 2014, 2014. [Google Scholar] [CrossRef]
- Gao, W.; Wang, W.F. The vertex version of weighted wiener number for bicyclic molecular structures. Comput. Math. Methods Med. 2015, 2015. [Google Scholar] [CrossRef] [PubMed]
- Munir, M.; Nazeer, W.; Rafique, S.; Kang, S.M. M-polynomial and degree-based topological indices of Nanostar dendrimers. Symmetry 2016, 8. [Google Scholar] [CrossRef]
- Liou, K.H.; Kang, D.Y. Defective Single-Walled Aluminosilicate Nanotubes: Structural Stability and Mechanical Properties. ChemNanoMat 2016, 2, 189–195. [Google Scholar] [CrossRef]
- Klavzar, S.; Deutsch, E. M-Polynomial and Degree-Based Topological Indices. Iran. J. Math. Chem. 2015, 6, 93–102. [Google Scholar]
- Todeschini, R.; Consonni, V.; Mannhold, R.; Kubinyi, H.; Timmerman, H. Handbook of Molecular Descriptors; Wiley-VCH: Weinheim, Germany, 2002. [Google Scholar]
- Hall, L.H.; Kier, L.B. Molecular Connectivity in Chemistry and Drug Research; Academic Press: Boston, MA, USA, 1976. [Google Scholar]
- Liou, K.H.; Tsou, N.T.; Kang, D.Y. Relationships among the structural topology, bond strength, and mechanical properties of single-walled aluminosilicate nanotubes. Nanoscale 2015, 7, 16222–16229. [Google Scholar] [CrossRef] [PubMed]
- Paineau, E.; Krapf, M.E.M.; Amara, M.S.; Matskova, N.V.; Dozov, I.; Rouzie`re, S.; Thill, A.; Launois, P.; Davidson, P. A liquid-crystalline hexagonal columnar phase in highly-dilute suspensions of imogolite nanotubes. Nat. Commun. 2016, 7. [Google Scholar] [CrossRef] [PubMed]
- Thill, A.; Maillet, P.; Guiose, B.; Spalla, O.; Belloni, L.; Chaurand, P.; Auffan, M.; Olivi, L.; Rose, J. Physico-chemical Control over the Single- or Double-Wall Structure of Aluminogermanate Imogolite-like Nanotubes. J. Am. Chem. Soc. 2012, 134, 3780–3786. [Google Scholar] [CrossRef] [PubMed]
| Topological Index | Derivation from | |
|---|---|---|
| First Zagreb | ||
| Second Zagreb | ||
| Second Modified Zagreb | ||
| General Randi | ||
| General Randi | ||
| Symmetric Division Index |
| Number of vertices |
| Number of edges |
| Number of edges |
| 2 | 3 | 4 | 5 | |
|---|---|---|---|---|
| Number of vertices |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Munir, M.; Nazeer, W.; Nizami, A.R.; Rafique, S.; Kang, S.M. M-Polynomials and Topological Indices of Titania Nanotubes. Symmetry 2016, 8, 117. https://doi.org/10.3390/sym8110117
Munir M, Nazeer W, Nizami AR, Rafique S, Kang SM. M-Polynomials and Topological Indices of Titania Nanotubes. Symmetry. 2016; 8(11):117. https://doi.org/10.3390/sym8110117
Chicago/Turabian StyleMunir, Mobeen, Waqas Nazeer, Abdul Rauf Nizami, Shazia Rafique, and Shin Min Kang. 2016. "M-Polynomials and Topological Indices of Titania Nanotubes" Symmetry 8, no. 11: 117. https://doi.org/10.3390/sym8110117
APA StyleMunir, M., Nazeer, W., Nizami, A. R., Rafique, S., & Kang, S. M. (2016). M-Polynomials and Topological Indices of Titania Nanotubes. Symmetry, 8(11), 117. https://doi.org/10.3390/sym8110117






