Given the velocity field
, as determined in the preceding section, we now address Equation (
3) for the description of the combustion field. Among all the parameters involved, we have fixed
and
, which correspond to the combustion reaction of typical hydrocarbon fuels under standard conditions, and examined the influence of the remaining parameters. These include the Damköhler number
D, the fuel and oxygen Lewis numbers
and
, the initial strain rate ratio
, the initial mixture strength
, the thermo-physical properties of the splitter plate,
and
, and the volumetric heat-loss coefficient
H, which will be examined separately. For consistency, the combustion field was determined numerically in the same rectangular domain and on the same mesh as the flow field. The governing equations and boundary conditions were discretized via a second-order finite-difference scheme and solved using the Gauss–Seidel iteration method with successive over-relaxation. For steady solutions, an intermediate temperature value was chosen and fixed at a specified grid point to capture all possible solutions, stable and unstable. The stability of the computed states was assessed by examining the time evolution of small perturbations superimposed to a given steady state. The time-dependent calculations were carried out with appropriate time steps
, ensuring that the required temporal precision was satisfied. Further details can be found in [
23] and the references cited therein.
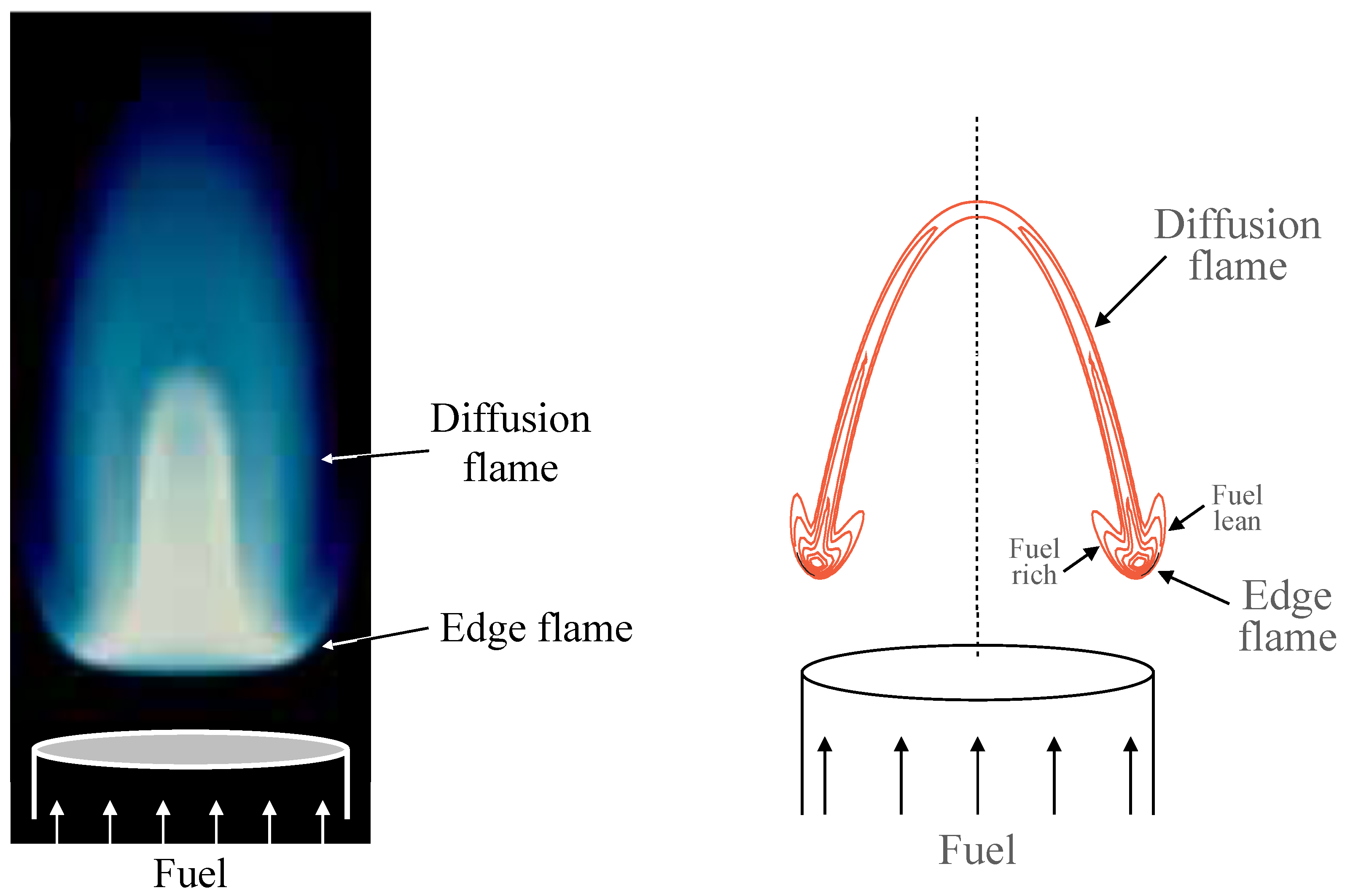
5.1. Symmetric Edge Flames
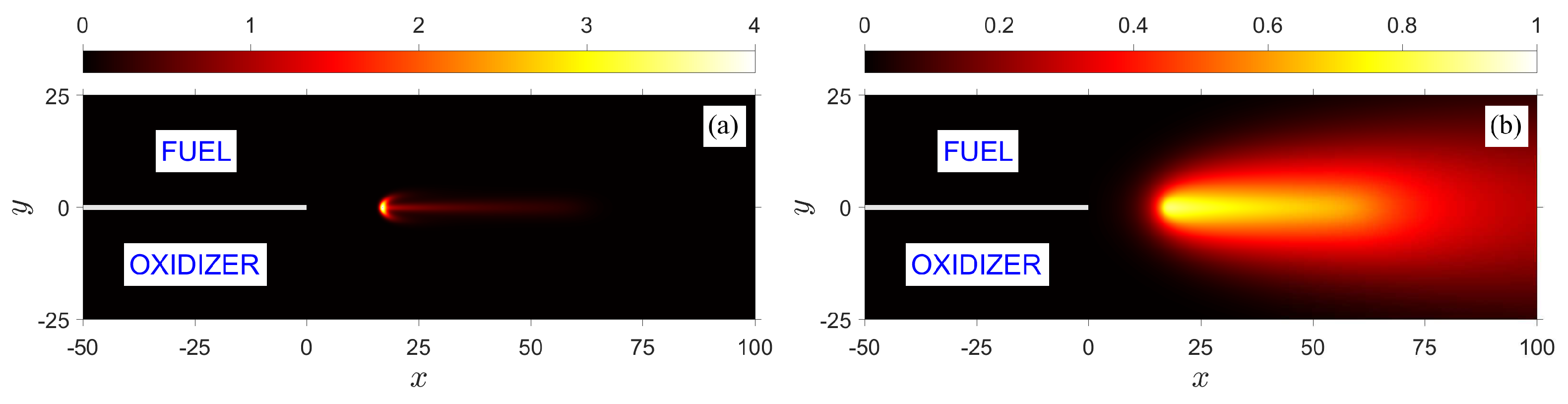
Figure 5 illustrates the structure of a symmetric flame, stabilized in the near wake of two merging streams of equal strain rates,
, and a flow rate corresponding to
, with the parameter values:
,
,
,
. We refer to this case as the
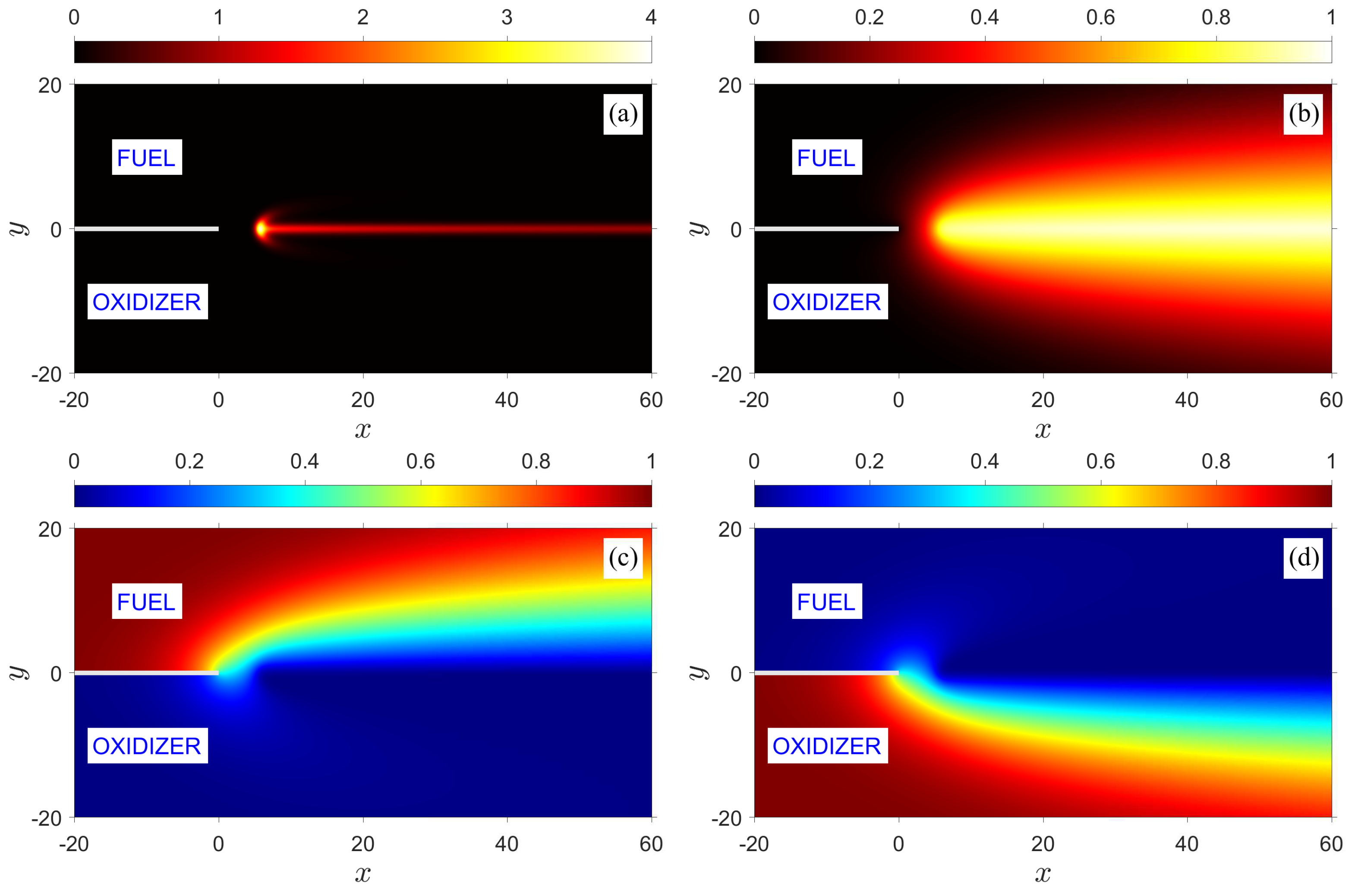
baseline case and, unless otherwise stated, hereinafter when varying one of the parameters it is implicitly assumed that all the other parameters remain unchanged. The flame structure in the figure is represented by contours of reaction rate
, temperature
, and fuel and oxidizer mass fractions,
and
, respectively. Because the flow field is symmetric with respect to the
x-axis, and the fuel and oxidizer of equal diffusivities are supplied in the incoming streams in stoichiometric proportion, the resulting edge flame formed in the wake of the splitter plate is also symmetric. The flame has an apparent tribrachial structure; it consists of a curved premixed flame at the front with fuel-rich and fuel-lean branches extending above and below, which is referred to as the
edge flame, and a straight diffusion flame trailing downstream along the centerline. An enlarged picture of an edge flame can be seen in one of the figures shown below. The temperature and mass fraction distributions shown in the figure illustrate the extent and width of the thermal and mixing layers. For the isothermal plate considered here, the thermal layer develops beyond the trailing edge of the plate, while the mixing layer extends behind the plate as a result of upstream diffusion.
The discussion below necessitates a precise and unambiguous definition of the edge-flame position. To this end, we define its coordinates, and , as the location where the reaction rate attains its maximum value. The axial distance between the edge flame and the tip of the plate, i.e., , will be referred to as the flame-standoff distance.
Figure 6 shows that the flame-standoff distance is strongly affected by the overall flow rate, i.e., variations in the Damköhler number
D, and by the disparity between heat and mass diffusivities, or variations in the Lewis number
. In general, when decreasing
D from relatively large values, the edge flame moves towards the plate, i.e., down the velocity gradient, in order to reach a new balance with the local flow velocity for its stabilization. The standoff distance, however, reaches a minimum value because heat loss to the cold plate prevents the edge flame from getting closer. The response of the edge flame when approaching the plate and the minimum standoff distance depend on the Lewis number. To retain the symmetry, the fuel and oxidizer Lewis numbers were assumed to have a common value, i.e.,
. The figure shows the dependence of the standoff distance
of steady solutions on the Damköhler number, for
. Depending on the Lewis number, the response curves exhibit one of two behaviors: a C-shaped response for relatively low Lewis numbers, as exemplified by
,
, and
, and a U-shaped response for higher values, as exemplified by
and
. The C-shaped curve consists of two branches with a turning point corresponding to a minimum Damköhler number,
, below which no solution exists. For
, the solution is multivalued. Steady solutions along the lower branch (solid curve) are stable with respect to small perturbations; when perturbed, the flame recedes back in time to its initial state. Solutions along the upper branch (dotted curve) are unstable; when perturbed, the flame advances or retreats from its initial equilibrium position indefinitely. Since only stable states are realized physically, the turning point
defines an
adiabatic extinction limit. In practical situations, when increasing the mass-flow rate by decreasing the Damköhler number starting with large values, the edge flame will first approach the plate along the stable lower branch, reach a minimum standoff distance, and then lift slightly up to
. When further increasing the mass-flow rate, the flame gets blown off. Both the Damköhler number and the flame-standoff distance at extinction show a decreasing trend when
decreases, suggesting that under otherwise similar conditions flames with lower Lewis numbers (e.g., light fuels) can survive higher flow rates.
Different from the C-shaped response, the U-shaped curve associated with high-Lewis-number flames shows that a single steady solution exists for all
D; the response curve does not have a turning point or an explicit extinction limit. In practical situations, when increasing the mass-flow rate by decreasing the Damköhler number starting with a large value, the edge flame will first approach the plate, reach a minimum standoff distance, and then lift off and stabilize further downstream. The flame lifts off to significantly large distances by increasing the mass-flow rate only slightly. Both the minimum standoff distance and the lift-off distance show an increasing trend when
increases, suggesting that, under otherwise similar conditions, flames with higher Lewis numbers (e.g., heavy fuels) can stabilize sufficiently far from the plate. The stabilization mode, however, depends on the Lewis number. For
, the entire response curve corresponds to stable steady states. For
, an intermediate range of Damköhler numbers associated with unstable states exist (dashed curve) between the stable states corresponding to smaller and larger values of
D. When a steady state within this range is slightly perturbed, the edge flame is observed to move back and forth along the centerline relative to its initial equilibrium position. Because these oscillations persist in time, the flame remains practically stabilized near the plate but not in a steady fashion. The marginal states separating stable steady states from stable oscillatory states are marked along the response curve by a ‘•’ symbol.
Figure 7 shows typical oscillations for four representative values of the Damköhler number marked in
Figure 6 by (a)–(d). The amplitude and frequency, as well as the nature of the oscillations, vary with
D. For example, oscillations of smaller amplitude are seen in (a), while a complex oscillation pattern of multiple frequencies is seen in (b). A closer examination shows that during its motion, the edge flame drags along the trailing diffusion flame, but oscillations along the diffusion flame decay quickly when moving further downstream [
23,
36]. The oscillations, which begin in the premixed segment of the edge flame, are presumed to share a similar underlying mechanism with the well-known large-Lewis-number pulsation of premixed flames attributed to a diffusive-thermal instability. Further results show that the ranges of both the Damköhler number
D and the flame-standoff distance
corresponding to oscillatory states expand on increasing the Lewis number [
23].
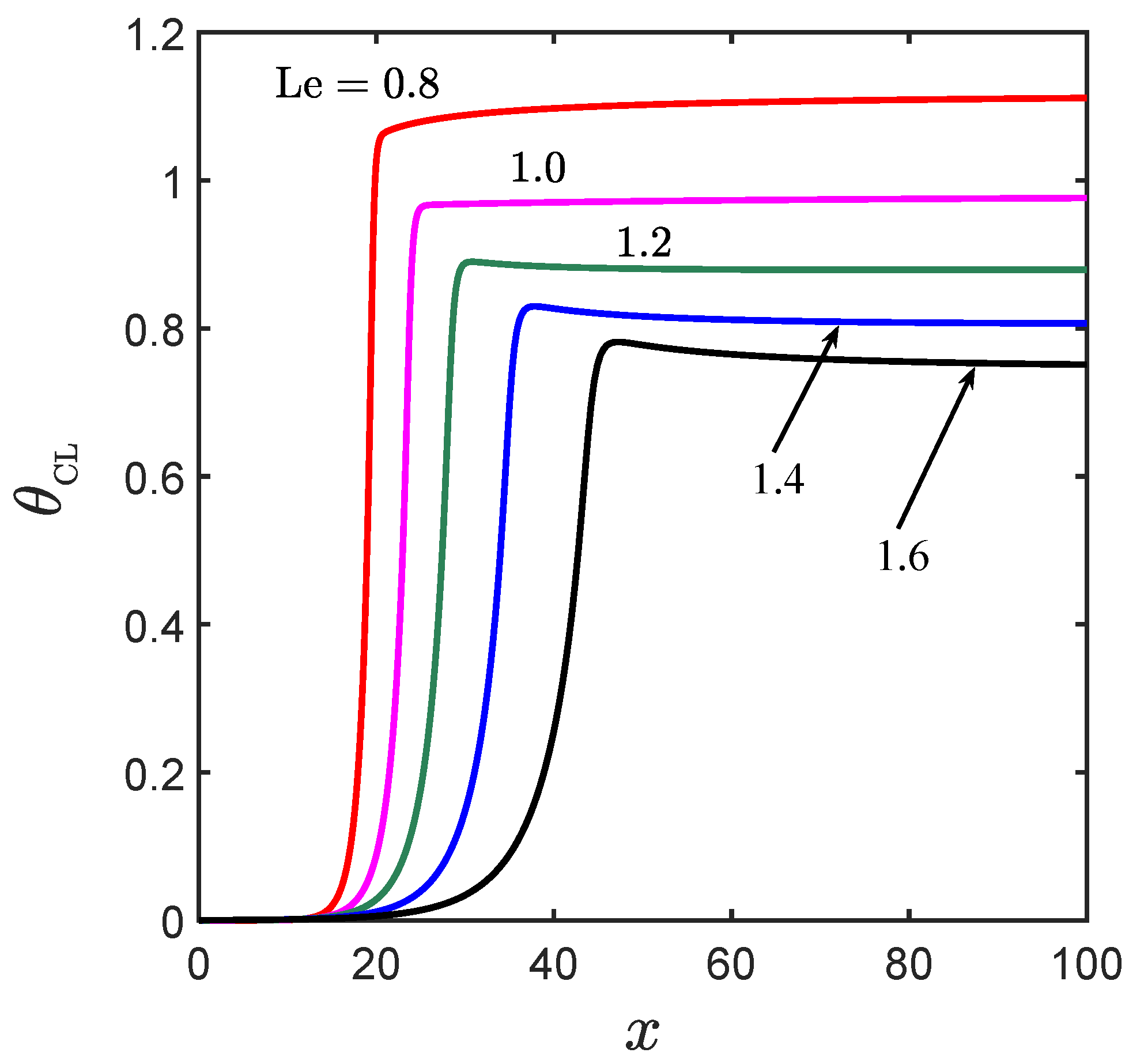
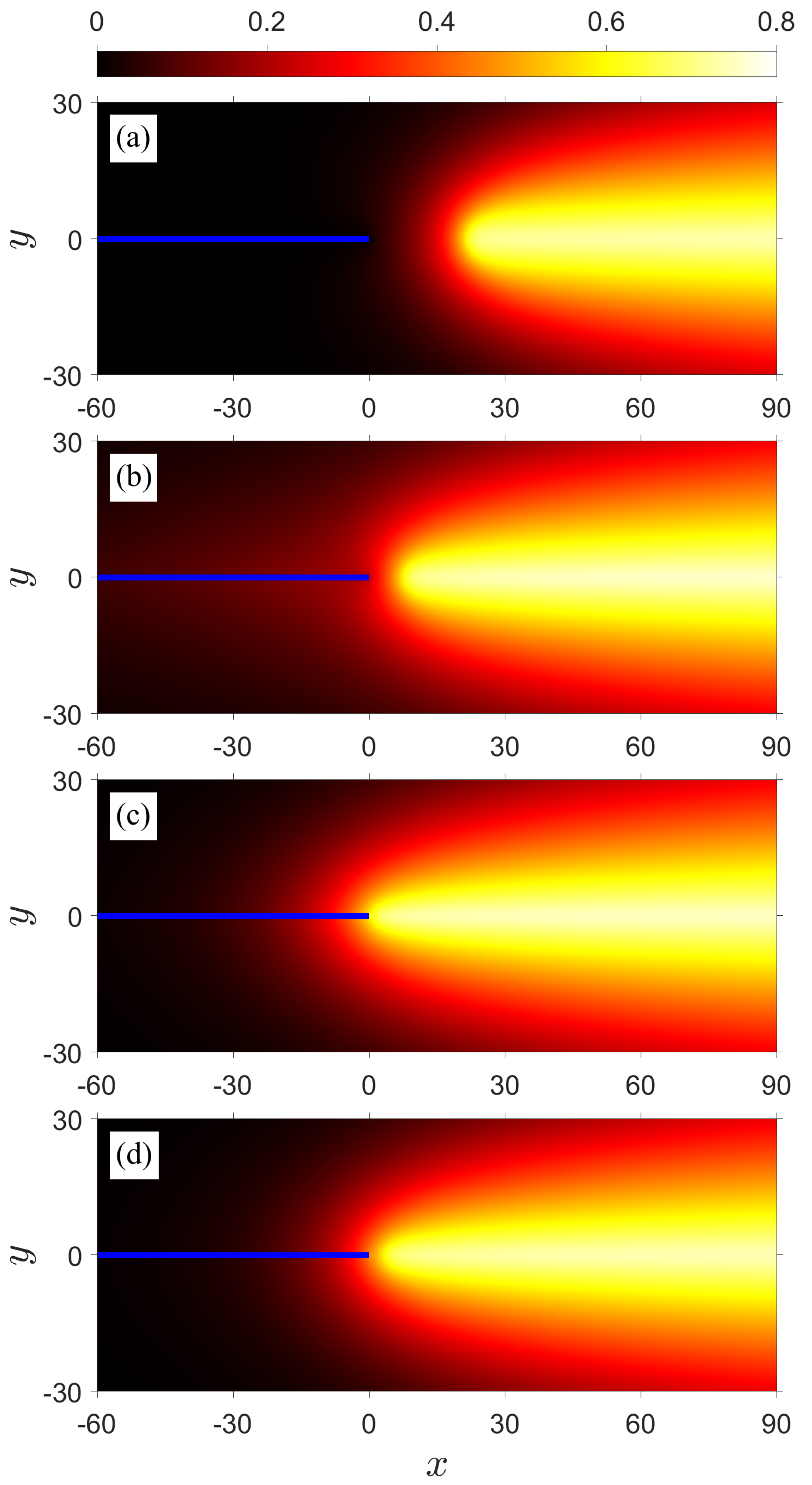
The influence of the Lewis number on the structural characteristics of edge flames is further illustrated in
Figure 8, where temperature profiles along the centerline are plotted for the same five representative Lewis numbers used earlier in
Figure 6. For the sake of comparison, the Damköhler number corresponding to each of these five cases was selected such that the temperature decays to the ambient value (
) at approximately the same position (
). We observe that when increasing
the temperature on the trailing diffusion flame falls consistently while the thickness of the preheat zone preceding the edge flame increases. The reason lies in the different effects that the Lewis number has on the temperature of diffusion and premixed flames, as discussed in
Section 3. Unlike the temperature of a premixed flame which remains the adiabatic flame temperature for all Lewis numbers, the flame temperature of a diffusion flame, or the stoichiometric temperature, decreases when increasing
. The structure of an edge flame, which combines characteristics of both premixed and diffusion flames, is influenced by the Lewis number through the thermal interaction between the leading premixed segment and the trailing diffusion flame. When
, the temperature of both the edge flame and the diffusion flame, is the adiabatic flame temperature. When
, the temperature of the diffusion flame exceeds the adiabatic flame temperature; the inherent heat transfer from the diffusion flame toward the premixed segment yields a super-adiabatic edge temperature. The opposite is true when
; heat is directed from the premixed segment towards the diffusion flame resulting in a sub-adiabatic edge temperature.
The significant variations in edge temperature associated with the Lewis number have implications on both the edge propagation speed
and the thickness of the preheat zone preceding the edge flame. The edge speed, similar to the edge temperature, is expected to increase with decreasing
. Because a stable stationary edge flame assumes the location where the edge speed
balances the incoming flow, a larger flow rate (smaller
D) would be required to stabilize the low-Lewis-number flames corresponding to a smaller standoff distance. Indeed, the flow rate required to stabilize the edge flame with
is significantly higher than that required to stabilize an edge flame with
, as indicated in the caption of
Figure 8. The same reasoning applies to the thickness of the preheat zone preceding the edge flame. Assuming an exponential decay
, which is the typical profile of a premixed flame, the characteristic thickness of the preheat zone proportional to
increases on increasing the Lewis number, as observed in
Figure 8.
The insights gained from the two distinct types of responses identified for edge flames and the role played by the Lewis number may be exploited to understand the stabilization of laminar jet diffusion flames relative to the nozzle rim. As evident from
Figure 6, the flame-standoff distance of low-Lewis-number edge flames characterized by C-shaped response curves is generally small in magnitude, on the order of the flame thickness. From a practical perspective, the edge flame is effectively attached to the tip of the plate. This attachment behavior persists when continuously increasing the flow rate until blow-off occurs. This implies that substantial lift-off is virtually impossible for low-Lewis-number edge flames. By contrast, for high-Lewis-number flames characterized by U-shaped response curves, the edge flame may be lifted and stabilized at substantially large distances from the plate by gradually increasing the flow rate. Note that both stabilization modes, namely the stationary mode which corresponds to an edge flame held stationary at a well-defined distance, and the oscillatory mode which corresponds to an edge flame moving back and forth relative to a mean position, can be realized in practice and thus are considered
globally stable. Such distinct attachment and lift-off behaviors of edge flames have been experimentally observed by Chung and Lee [
1], who proposed a Schmidt-number-based criterion to predict the transition between the attached and lifted regimes of jet diffusion flames. Their prediction based on flow and concentration profiles of cold jets, naturally involves a Schmidt number. When converted to a Lewis number, using the relation
, their condition is in good agreement with the transition between attached and lifted edge flames predicted in our mixing layer model, as discussed in [
23]. Choosing the Lewis number as the primary control parameter describing the stabilization of jet diffusion flames seems more appropriate, considering the fundamental and direct role it plays in determining the characteristics of edge flames, as discussed in the preceding paragraph. It is also worth mentioning that experiments on jet diffusion flames have also observed U-shaped responses of flame-standoff distance with respect to the flow rate [
37] and sustained flame oscillations [
38,
39]. These experimental findings have been attributed to buoyancy effects, which are fundamentally different from the present numerical predictions exclusively associated with diffusive-thermal influences. Systematic microgravity experiments in conjunction with numerical simulations accounting for buoyancy convection are therefore desired to elucidate the unique influence of these different mechanisms.
5.2. Asymmetric Edge Flames
The symmetric structure of the edge and trailing diffusion flames described above was obtained for selected conditions that constitute the baseline case and its symmetric variants. Streams of unequal initial strain rates (), fuel and oxidizer supplied in off-stoichiometric proportions (), preferential diffusion of fuel and oxidizer () would all lead to asymmetric flames. In the following, we examine some of these cases.
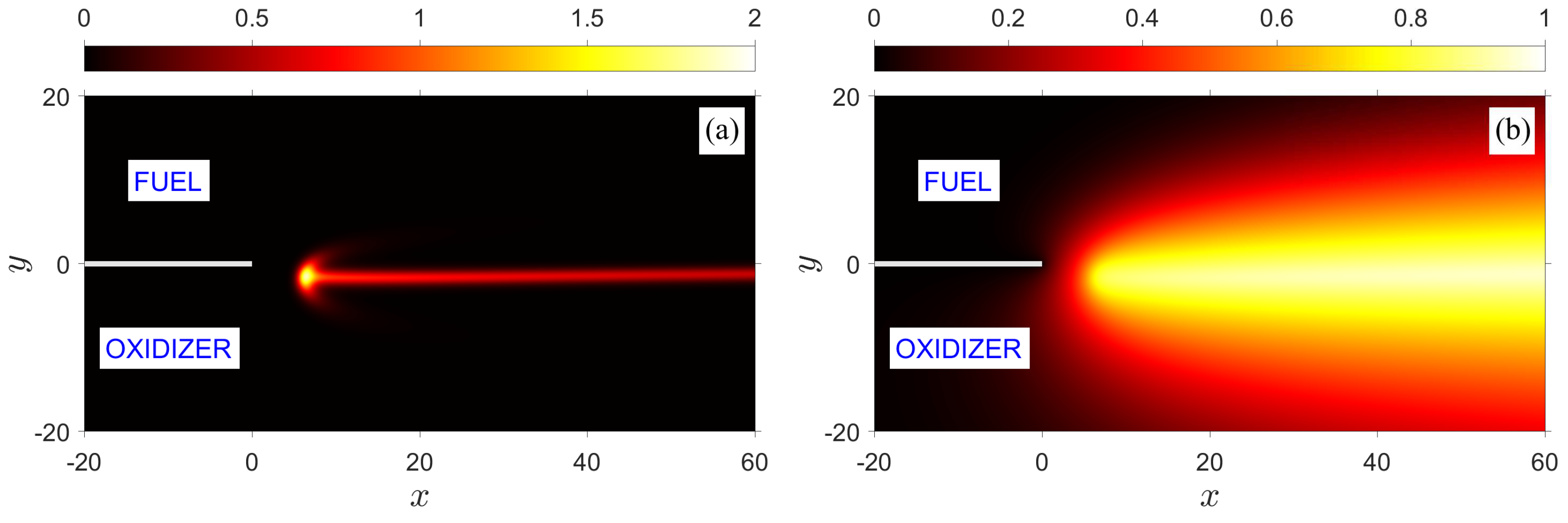
Figure 9 illustrates an asymmetric edge flame stabilized within the flow and resulting purely from preferential diffusion, i.e., unequal diffusivities of fuel and oxidizer, with
and
. The significantly larger diffusivity of the oxidizer offsets the balanced transport of the two reactants such that the trailing diffusion flame as a whole deviates from the centerline toward the fuel side. For the same reason, the premixed flame branches of the tribrachial edge structure are no longer symmetrically distributed on the two sides of the diffusion flame; the fuel-rich branch appears longer and the entire edge, measured relative to the normal direction, is deflected toward the fuel side. Meanwhile, in line with the trends identified in
Figure 6 and
Figure 8, for a given overall mass-flow rate (same Damköhler number), an increase in the fuel Lewis number leads to an appreciable increase in the flame-standoff distance and decrease in flame temperature.
Figure 10 shows an asymmetric edge flame stabilized within the flow and resulting purely from unequal strain rates in the incoming streams, i.e., with
. As the characteristic flow velocity originating from the oxidizer stream is considerably lower than that originating from the fuel stream, the diffusion flame deviates from the centerline toward the oxidizer stream to facilitate its intake of oxygen and the two premixed flame branches of the tribrachial structure are, for the same reason, no longer symmetric. The flame-standoff distance and temperature distribution along the diffusion flame appear, for this equal-Lewis-number case, to be scarcely influenced by a change in the flow field at the given Damköhler number. However, from an overall perspective, diffusion will play a more significant role when decreasing the strain rate ratio
, leading to a leftward shift of the entire edge flame response curve, and implying a delay in the blow-off conditions for low-Lewis-number flames and in the lift-off conditions of high-Lewis-number flames [
23].
The extent to which unequal strain rates combined with inter-diffusion of reactants influence the edge-flame position is shown in
Figure 11 for
and unequal Lewis numbers. The oxidizer Lewis number is assumed equal to one, as appropriate for air, and two values of the fuel Lewis number are considered; a low value
corresponding to a relatively light fuel, and a high value
corresponding to a relatively heavy fuel. It should be noted that diluting one or both streams with an inert one can significantly affect the corresponding Lewis number due to changes in the mixture thermal diffusivity; see, for example, [
24]. In addition to the edge flame, illustrated in the figure by reaction-rate contours, the stoichiometric surface
is shown as a dashed line. Although for
considered here the oxidizer is entrained into the mixing layer and the dividing streamline is deflected towards the fuel region, the edge and diffusion flames deviate towards the oxidizer region when
due to the high mobility of the fuel. When
, the inclination is towards the fuel region. The diffusion flame trailing behind the edge flame always aligns itself with the stoichiometric surface. We note parenthetically that due to preferential diffusion (
) the mixture fraction
Z is not a conserved scalar and, as a result,
is not a smooth curve; its determination that accounts for the sharp deflection observed across the curved part of the edge flame will be discussed below in
Section 6.
Figure 12 illustrates a situation of asymmetry resulting from fuel and oxidizer supplied in the incoming streams in off-stoichiometric proportions, with an initial mixture strength
. Because the fuel is in excess, the reaction is dominated by the supply of oxygen to the reaction zone. As a response, the diffusion flame adjusts its shape and position and finally stabilizes itself in the oxidizer stream, in an effort to balance the transport of the two reactants. A notable difference compared to the previous two non-symmetric cases is that now the edge flame loses its tribrachial structure; instead, it takes on a hook-like shape with only the fuel-rich branch retained, while the fuel-lean branch appears to completely degenerate into the diffusion flame.
Asymmetry in the combustion field can also result from chemistry imbalance, for example, unequal reaction orders as demonstrated by Juanos and Sirignano [
7] for a propane–air mixture with
and
, or from the interaction of two adjacent flames as illustrated by Kurdyumov and Jimenez [
40].