Dynamics of a COVID-19 Model with a Nonlinear Incidence Rate, Quarantine, Media Effects, and Number of Hospital Beds
Abstract
1. Introduction
2. The Dimensional Model
3. Analysis of the Model
Positivity
4. Equilibria Existence and Classification
- 1.
- possesses one steady state if Cases 2, 4, 8, and 16 are satisfied;
- 2.
- can possess more than one steady state if Cases 6, 10, and 12 are satisfied;
- 3.
- can possess two steady states if Cases 3, 5, 7, 9, 11, 13, and 15 are satisfied.
Local Stability of the Disease-Free Solution
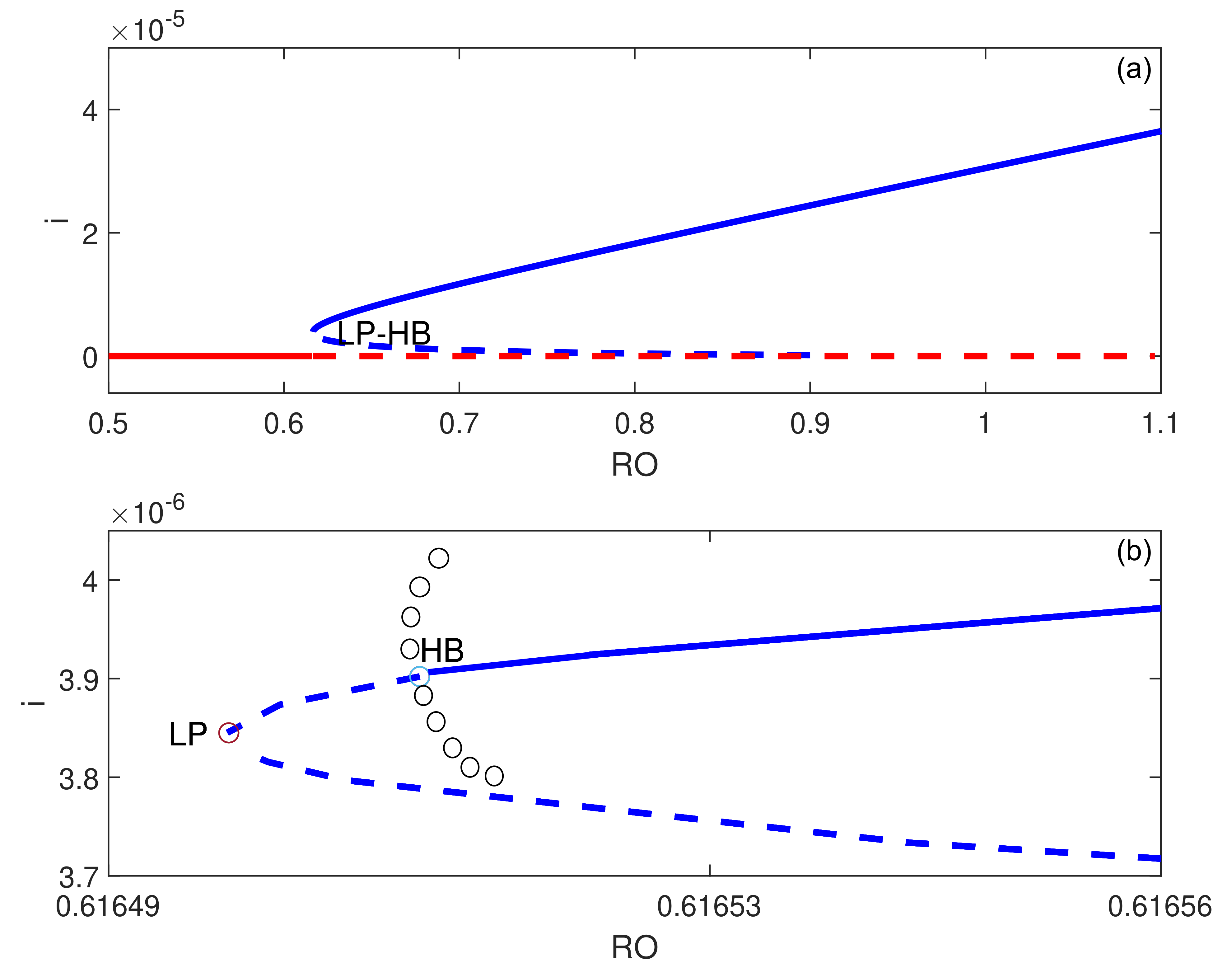
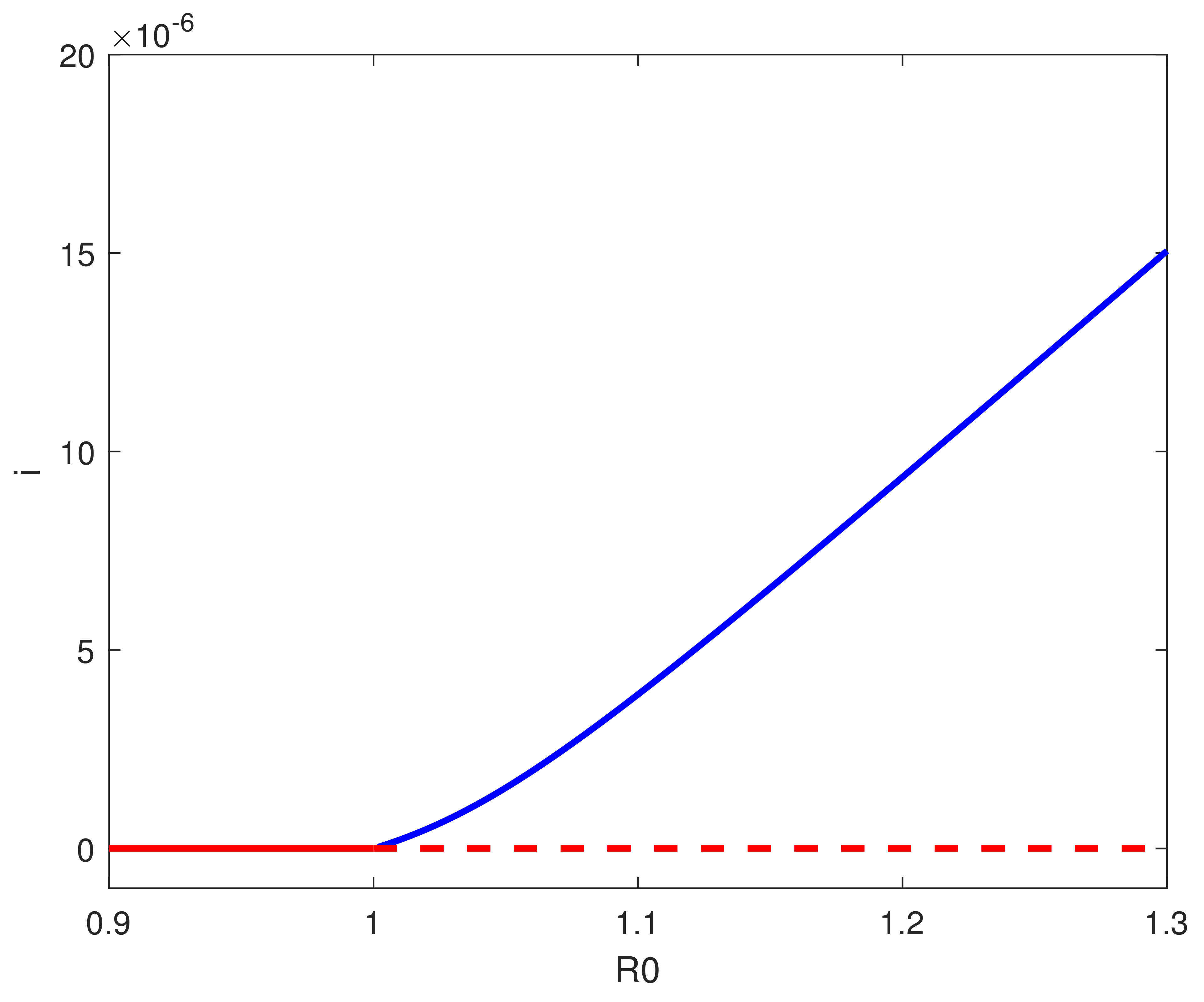
5. Backward Bifurcation
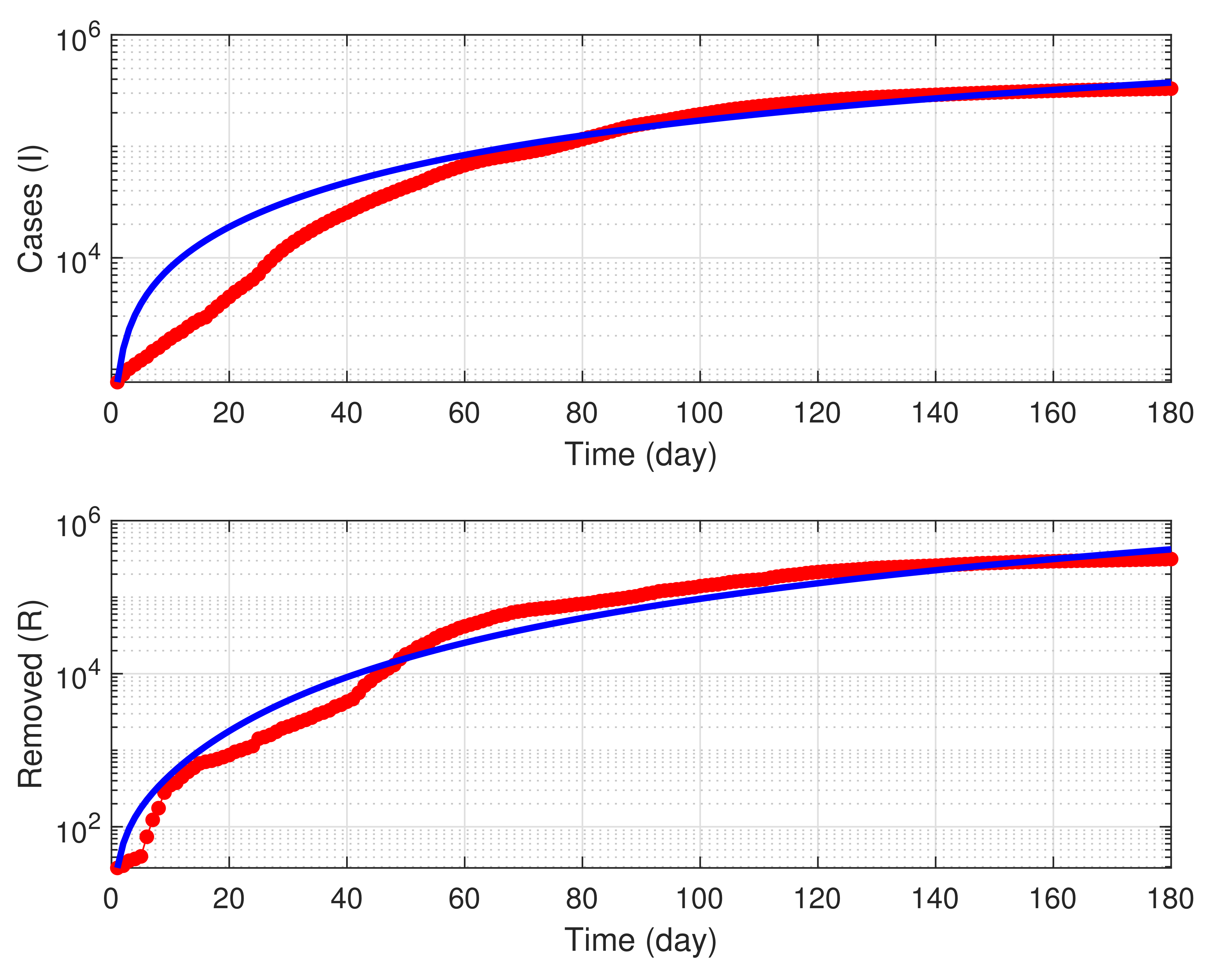
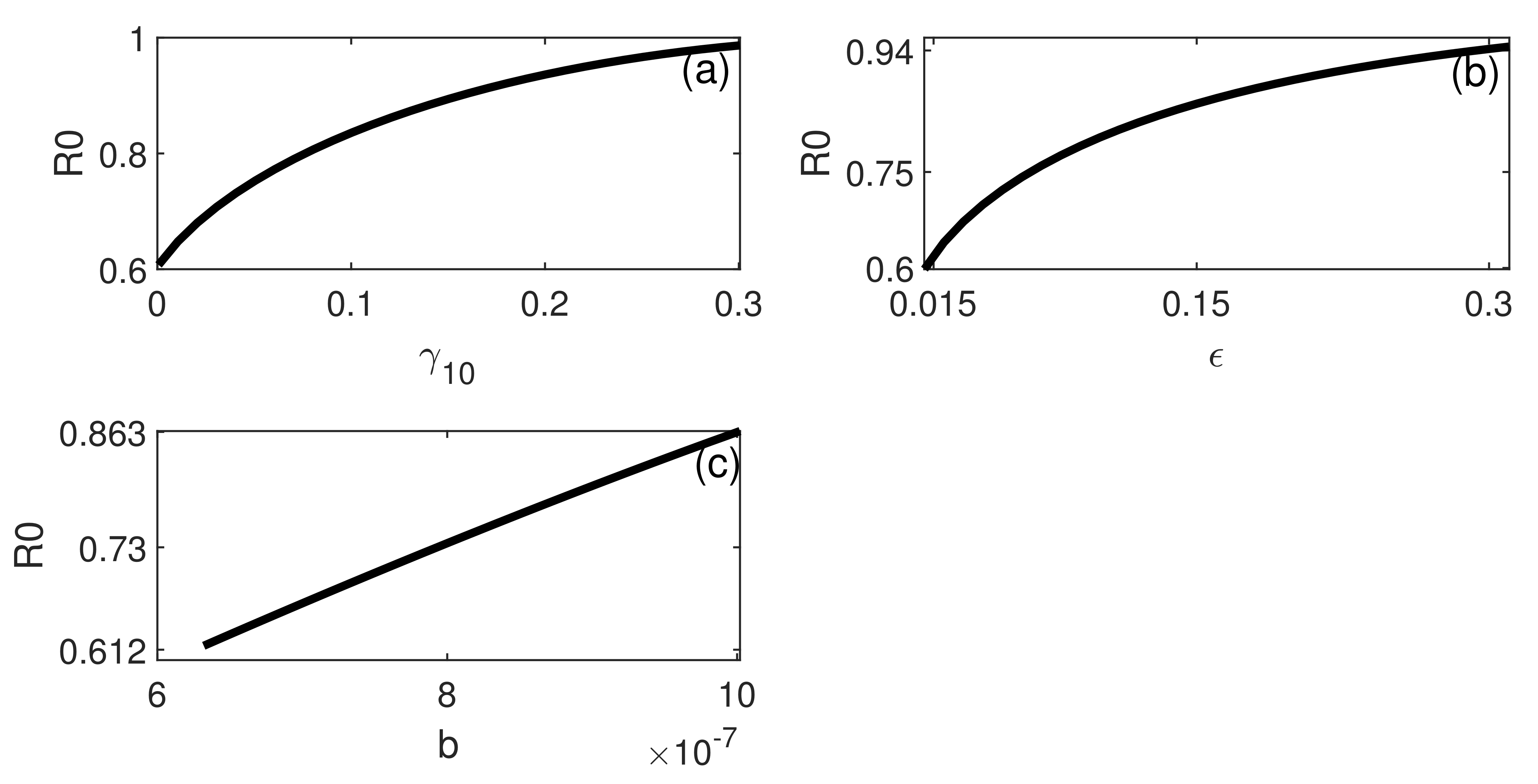
6. Numerical Simulations
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Anirudh, A. Mathematical modeling and the transmission dynamics in predicting the Covid-19—What next in combating the pandemic. Infect. Dis. Model. 2020, 5, 366–374. [Google Scholar] [CrossRef] [PubMed]
- Cooper, I.; Mondal, A.; Antonopoulos, C.G. A SIR model assumption for the spread of COVID-19 in different communities. Chaos Soliton. Fract. 2020, 139, 110057. [Google Scholar] [CrossRef]
- Koziol, K.; Stanislawski, R.; Bialic, G. Fractional-Order SIR epidemic model for transmission prediction of COVID-19 disease. Appl. Sci. 2020, 10, 8316. [Google Scholar] [CrossRef]
- He, S.; Peng, Y.; Sun, K. SEIR modeling of the COVID-19 and its dynamics. Nonlinear Dyn. 2020, 101, 1667–1680. [Google Scholar] [CrossRef] [PubMed]
- Godio, A.; Pace, F.; Vergnano, A. SEIR Modeling of the Italian Epidemic of SARS-CoV-2 Using Computational Swarm Intelligence. Int. J. Environ. Res. Public Health 2020, 17, 3535. [Google Scholar] [CrossRef] [PubMed]
- Zisad, S.N.; Hossain, M.S.; Hossain, M.S.; Andersson, K. An Integrated Neural Network and SEIR Model to Predict COVID-19. Algorithms 2021, 14, 94. [Google Scholar] [CrossRef]
- Prathumwan, D.; Trachoo, K.; Chaiya, I. Mathematical Modeling for Prediction Dynamics of the Coronavirus Disease 2019 (COVID-19) Pandemic, Quarantine Control Measures. Symmetry 2020, 12, 1404. [Google Scholar] [CrossRef]
- Feng, L.-X.; Jing, S.-L.; Hu, S.-K.; Wang, D.-F. Modelling the effects of media coverage and quarantine on the COVID-19 infections in the UK. Math. Biosci. Eng. 2020, 17, 3618–3636. [Google Scholar] [CrossRef] [PubMed]
- Mohsen, A.A.; Al-Husseiny, H.F.; Zhou, X.; Hattaf, K. Global stability of COVID-19 model involving the quarantine strategy and media coverage effects. AIMS Public Health 2020, 7, 587–605. [Google Scholar] [CrossRef]
- Sardar, T.; Nadim, S.; Rana, S.; Chattopadhyay, J. Assessment of lockdown effect in some states and overall India: A predictive mathematical study on COVID-19 outbreak. Chaos Soliton Fract. 2020, 139, 110078. [Google Scholar] [CrossRef]
- Linka, K.; Peirlinck, M.; Costabal, F.S.; Kuhl, E. Outbreak dynamics of COVID-19 in Europe and the effect of travel restrictions. Comput. Methods Biomech. Biomed. Eng. 2020, 23, 710–717. [Google Scholar] [CrossRef] [PubMed]
- Kwuimy, C.A.K.; Nazari, F.; Jiao, X.; Rohani, P.; Nataraj, C. Nonlinear dynamic analysis of an epidemiological model for COVID 19 including public behavior and government action. Nonlinear Dyn. 2020, 101, 1545–1559. [Google Scholar] [CrossRef] [PubMed]
- Ajbar, A.; Alqahtani, R.T. Bifurcation analysis of a SEIR epidemic system with governmental action and individual reaction. Adv. Differ. Equ. 2020, 2020, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Ghostine, R.; Gharamti, M.; Hassrouny, S.; Hoteit, I. An extended SEIR Model with vaccination for forecasting the COVID-19 pandemic in Saudi Arabia using an ensemble Kalman filter. Mathematics 2021, 9, 636. [Google Scholar] [CrossRef]
- Ruan, S.; Wang, W. Dynamical behavior of an epidemic model with a nonlinear incidence rate. J. Differ. Equ. 2003, 188, 135–163. [Google Scholar] [CrossRef]
- Tang, Y.; Huang, D.; Ruan, S.; Zhang, W. Coexistence of limit cycles and homoclinic loops in a SIRS model with a nonlinear incidence rate. SIAM J. Appl. Math. 2008, 69, 621–639. [Google Scholar] [CrossRef]
- Hu, Z.; Ma, W.; Ruan, S. Analysis of SIR epidemic models with nonlinear incidence rate and treatment. Math. Biosci. 2012, 238, 12–20. [Google Scholar] [CrossRef]
- Wang, W. Backward bifurcation of an epidemic model with treatment. Math. Biosci. 2006, 201, 58–71. [Google Scholar] [CrossRef]
- Chan, C.; Zhu, H. Bifurcations and complex dynamics of an SIR model with the impact of the number of hospital beds. J. Diff. Equ. 2014, 257, 1662–1688. [Google Scholar] [CrossRef]
- Cui, Q.; Qiu, Z.; Liu, W.; Hu, Z. Complex dynamics of an SIR epidemic model with nonlinear saturate incidence and recovery rate. Entropy 2017, 19, 305. [Google Scholar] [CrossRef]
- Saudi Ministry of Health. Available online: https://www.moh.gov.sa/en/Pages/Default.aspx (accessed on 2 November 2020).
- De la Sen, M.; Ibeas, A.; Agarwal, R.P. On Confinement and Quarantine Concerns on an SEIAR Epidemic Model with Simulated Parameterizations for the COVID-19 Pandemic. Symmetry 2020, 12, 1646. [Google Scholar] [CrossRef]
- Corduneanu, C. Principles of Differential and Integral Equations; Allyn and Bacon: Boston, MA, USA, 1971. [Google Scholar]
- Van den Driessche, P.; Watmough, J. Reproduction numbers and subthreshold endemic equilibria for compartmental models of disease transmission. Math. Biosci. 2002, 180, 29–48. [Google Scholar] [CrossRef]
- Castillo-Chavez, C.; Song, B. Dynamical models of tuberculosis and their applications. Math. Biosci. Eng. 2004, 1, 361–404. [Google Scholar] [CrossRef] [PubMed]
- General Authority for Statistics, Saudi Arabia. Available online: https://www.stats.gov.sa/en/5305 (accessed on 2 November 2020).
- Lina, Q.; Zhao, S.; Gao, D.; Lou, Y.; Yang, S.; Musa, S.S.; Wang, M.H.; Cai, Y.; Wang, W.; Yang, L.; et al. A conceptual model for the coronavirus disease 2019 (COVID 19) outbreak in Wuhan, China with individual reaction and governmental action. Int. J. Infect. Dis. 2020, 93, 211–216. [Google Scholar] [CrossRef] [PubMed]
- COVID-19 Dashbard. Available online: https://covid19.moh.gov.sa/ (accessed on 2 November 2020).
- Alharbi, Y.; Alqahtani, A.; Albalawi, O.; Bakouri, M. Epidemiological modeling of COVID-19 in Saudi Arabia: Spread projection, awareness, and impact of treatment. Appl Sci. 2020, 10, 5895. [Google Scholar] [CrossRef]
- Alrasheed, H.; Althnian, A.; Kurdi, H.; Al-Mgren, H.; Alharbi, S. COVID-19 Spread in Saudi Arabia: Modeling, simulation and analysis. Int. J. Environ. Res. Public Health 2020, 17, 7744. [Google Scholar] [CrossRef]
| Case | Number of Sign Changes | Number of Positive Roots | ||||||
|---|---|---|---|---|---|---|---|---|
| 1 | + | + | + | + | + | 0 | 0 | |
| 2 | + | + | + | + | − | 1 | 1 | |
| 3 | + | + | + | − | + | 2 | 0, 2 | |
| 4 | + | + | + | − | − | 1 | 1 | |
| 5 | + | + | − | + | + | 2 | 0, 2 | |
| 6 | + | + | − | + | − | 3 | 1, 3 | |
| 7 | + | + | − | − | + | 2 | 0,2 | |
| 8 | + | + | − | − | − | 1 | 1 | |
| 9 | + | − | + | + | + | 2 | 0, 2 | |
| 10 | + | − | + | + | − | 3 | 1, 3 | |
| 11 | + | − | + | − | + | 4 | 2, 4 | |
| 12 | + | − | + | − | − | 3 | 1, 3 | |
| 13 | + | − | − | + | + | 2 | 0, 2 | |
| 14 | + | − | − | + | − | 3 | 1, 3 | |
| 15 | + | − | − | − | + | 2 | 0,2 | |
| 16 | + | − | − | − | − | 1 | 1 |
| Parameter | Definition | Value | Source |
|---|---|---|---|
| b | Dimensionless hospital-bed-to-population ratio | [21] | |
| Half-saturation of incidence rate | fitted | ||
| Half-saturation of media effects | fitted | ||
| Awareness rate | fitted | ||
| Minimum recovery rate | 0.0026 | fitted | |
| Maximum recovery rate | 0.750 | fitted | |
| Recovery rate of quarantine at home | [27] | ||
| Natural death rate | [26] | ||
| Quarantine rate | fitted | ||
| Inverse of mean latent period | 0.333 | [27] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ajbar, A.; Alqahtani, R.T.; Boumaza, M. Dynamics of a COVID-19 Model with a Nonlinear Incidence Rate, Quarantine, Media Effects, and Number of Hospital Beds. Symmetry 2021, 13, 947. https://doi.org/10.3390/sym13060947
Ajbar A, Alqahtani RT, Boumaza M. Dynamics of a COVID-19 Model with a Nonlinear Incidence Rate, Quarantine, Media Effects, and Number of Hospital Beds. Symmetry. 2021; 13(6):947. https://doi.org/10.3390/sym13060947
Chicago/Turabian StyleAjbar, Abdelhamid, Rubayyi T. Alqahtani, and Mourad Boumaza. 2021. "Dynamics of a COVID-19 Model with a Nonlinear Incidence Rate, Quarantine, Media Effects, and Number of Hospital Beds" Symmetry 13, no. 6: 947. https://doi.org/10.3390/sym13060947
APA StyleAjbar, A., Alqahtani, R. T., & Boumaza, M. (2021). Dynamics of a COVID-19 Model with a Nonlinear Incidence Rate, Quarantine, Media Effects, and Number of Hospital Beds. Symmetry, 13(6), 947. https://doi.org/10.3390/sym13060947





