The Multi-Compartment SI(RD) Model with Regime Switching: An Application to COVID-19 Pandemic
Abstract
:1. Introduction
- We study the model proposed—a susceptible-infected-(recovered-dead) model with regime switching—proving that there are two ways to achieve the regime switching (the rough way and the smooth way), and that these are equivalent for all practical purposes;
- Given a bivariate set of data—(daily cases-lethality)—we propose a new simple method to estimate the parameters of the model specially suited to the jagged character of the observations using a binomial model; this amounts to hypothesise that there is a mechanism that corrupts, by noise, the observed trajectories of the ordinary differential equations of the model.
- We show that, taking into account important qualitative information, there are secondary parameters—that modify the primary parameters—that can be set according to reasonable scenarios, such that in one of these scenarios, the lethality rate can be recovered with a very small error.
2. The SI(RD) Model
- the daily infection rate of the susceptible population;
- the recovery rate is a rate which controls how fast members progress into the I and R groups, respectively;
- is the death rate among those infected;
- is a composite parameter, which—in case —is often used, and is referred to as the contact number.
- If the fraction of the population in the infected group is initially increasing (i.e., ), an epidemic has begun. For any in a neighbourhood of zero—such that —and if almost all population is susceptible—that is, —we have that:As a consequence, for the SI(RD) model, we define the composite parameter , corresponding to in the SIR model—since in SIR model, there is no parameter—by:
- At the peak point of an epidemic—with —we should have that , by Formula (2), and so, the peak point of an epidemic occurs at a time such that:
- T—the transmissibility which is the probability an individual infecting another given there was contact between them;
- —the average rate of contact between susceptible and infected individuals;
- —the duration of the infection in individuals; in the SI(RD) model, the infection ends with two possible outcomes: either recovered or dead.
3. SI(RD) Models with Regime Switching (Two Different Values for the Parameter )
- (j)
- The function is a continuous function.
- (jj)
- For some constant , the uniform Lipschitz condition in the variable t given by,is verified for all values of the variables .
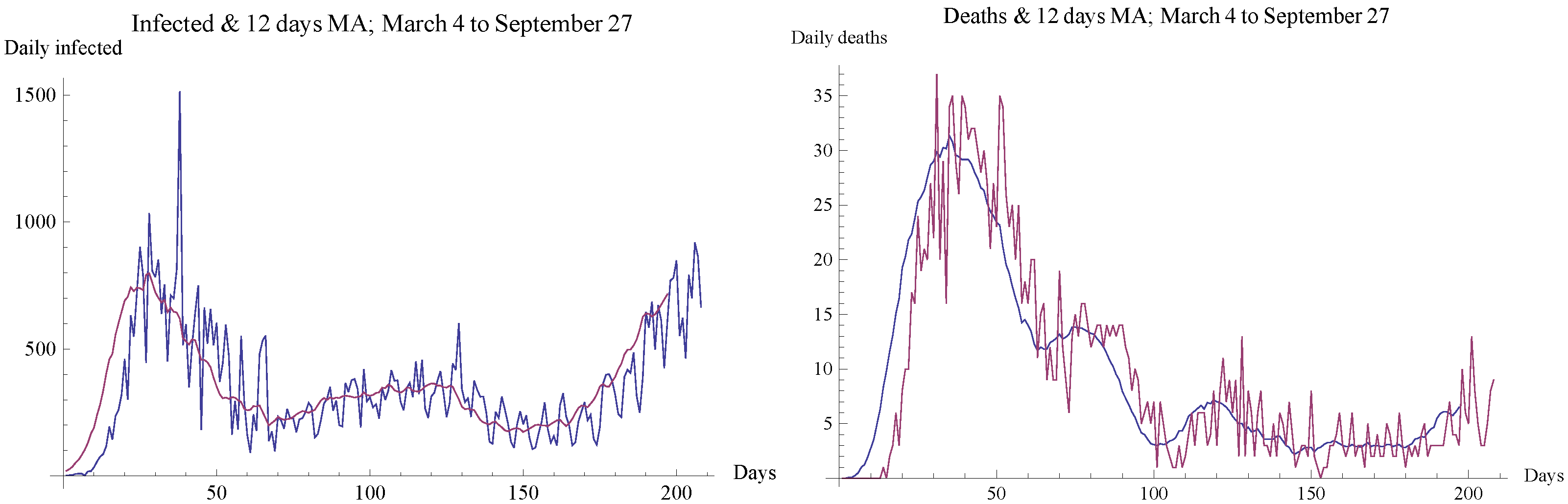
4. Data and Parameter Estimation
4.1. On the Assumed Value for the Duration of the Disease
4.2. Determination of the Initial Date of the Decreasing Trend Period
4.3. Estimation of the Contact Number
- (1)
- The length of the period is determined and we have a sequence of daily observations of the number of infected. There is a sequence of random variables such that the initial is arbitrary but the remaining observed data is a realisation of this sample.
- (2)
- There exists a random variable , taking two values —with u representing the magnitude of the upward jump and d representing the magnitude of a downward jump—such that and with and . For a sample of we have, for , that:The values are the parameters to be estimated and they depend on the period considered.
- (1)
- The maximum likelihood estimator of , as a consequence of the distribution of , is given by:
- (2)
- By the law of large numbers we have that for large enough,that is, may be taken as a good approximation of —the expectation of —and is a good approximation of , the variance of .
- (3)
- By the method of moments, we can determine the couple of estimators for the parameter couple as the solutions of the equations derived from the law of given by,considering the estimator of given by Formula (12) and the estimator of (respectively ) given by Formula (13). There are two sets of solutions—Formulas (14) and (15)—and we have to choose the set that corresponds to the period characteristic, that is, in the first period we should have the greatest u with the highest probability and in the second period the greatest d with the higher probability.
4.4. Estimation of the Lethality Rate
4.5. Further Hypothesis on the Results of the Estimation Procedure
- We will consider that the real number of infected is 20 times higher than the number reported. As a consequence, the model parameters and are to be modified to take this assumption in consideration.
- The estimated value for the parameter has to be replaced by the value given by,with , a secondary parameter, being the correction needed to take into account that the non diagnosed infected cases must correspond to non symptomatic cases and, as such, with a contagion force inferior to those infected with symptoms and tested with a positive outcome. Having no prior information available, we will consider that correspond to our basis scenario, and we will base our main conclusions on the observations of the outcomes of the model under this particular scenario.
- In the same way, the estimated value for the parameter should be replaced by the value given by,with , another secondary parameter, being the correction needed to take into account the mortality rate due to the infection in the asymptomatic and not tested with a positive outcome. Having no prior information available, we will suppose that correspond to our basis scenario and and to alternative scenarios.
- For the initially immune we consider that in the first period these are supposed to be 2.5% of the population. In the second period the initially immune are those who either recovered or died at the end of the preceding period.
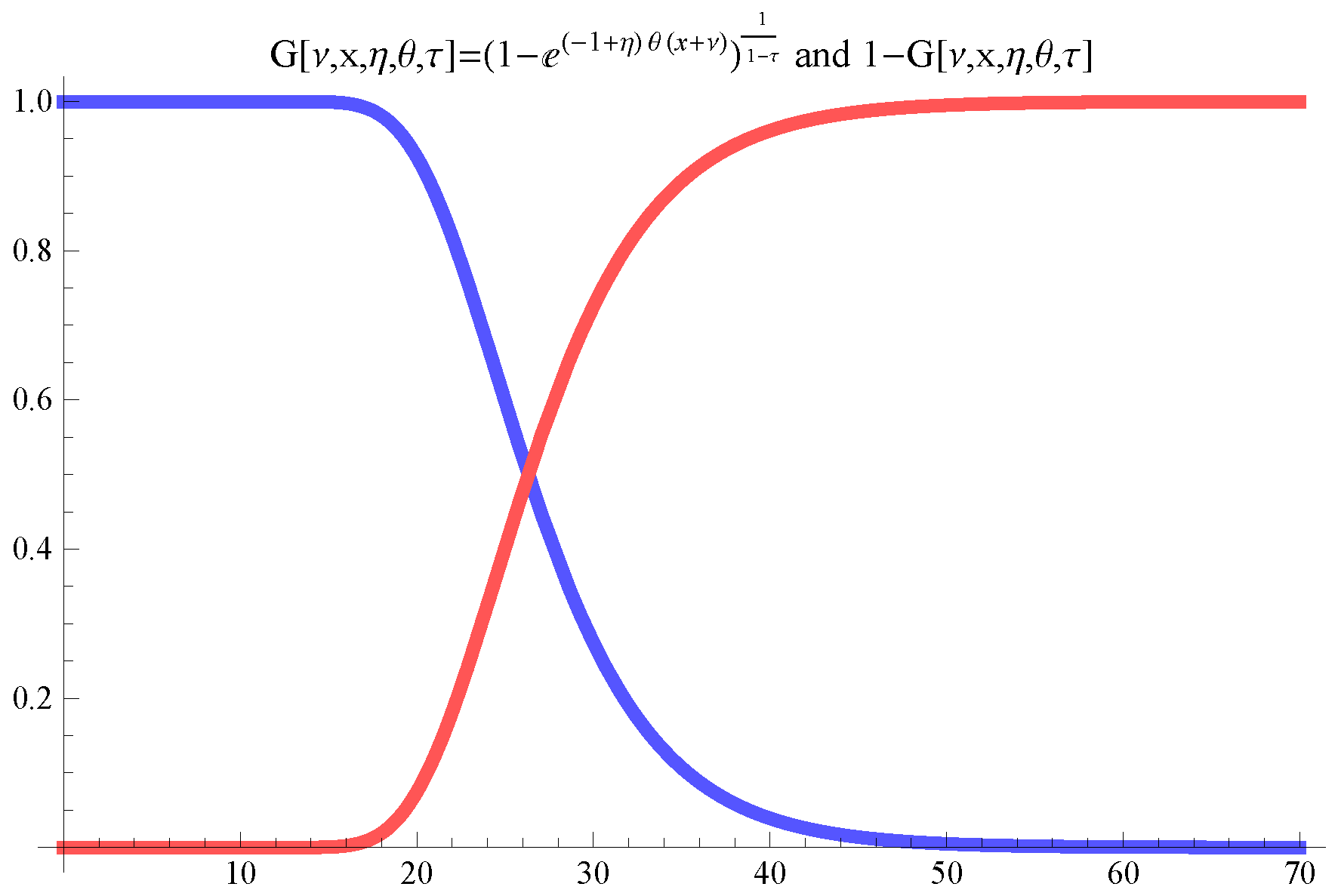
- For the smooth regime switching model drive function G, shown in Figure 1, the parameter choice ensures that for we have , for we have , for we gave and that for we gave and so, the smooth regime switching unfolds, essentially, between day 16, the declaration of starting time of the lockdown, and day 40.
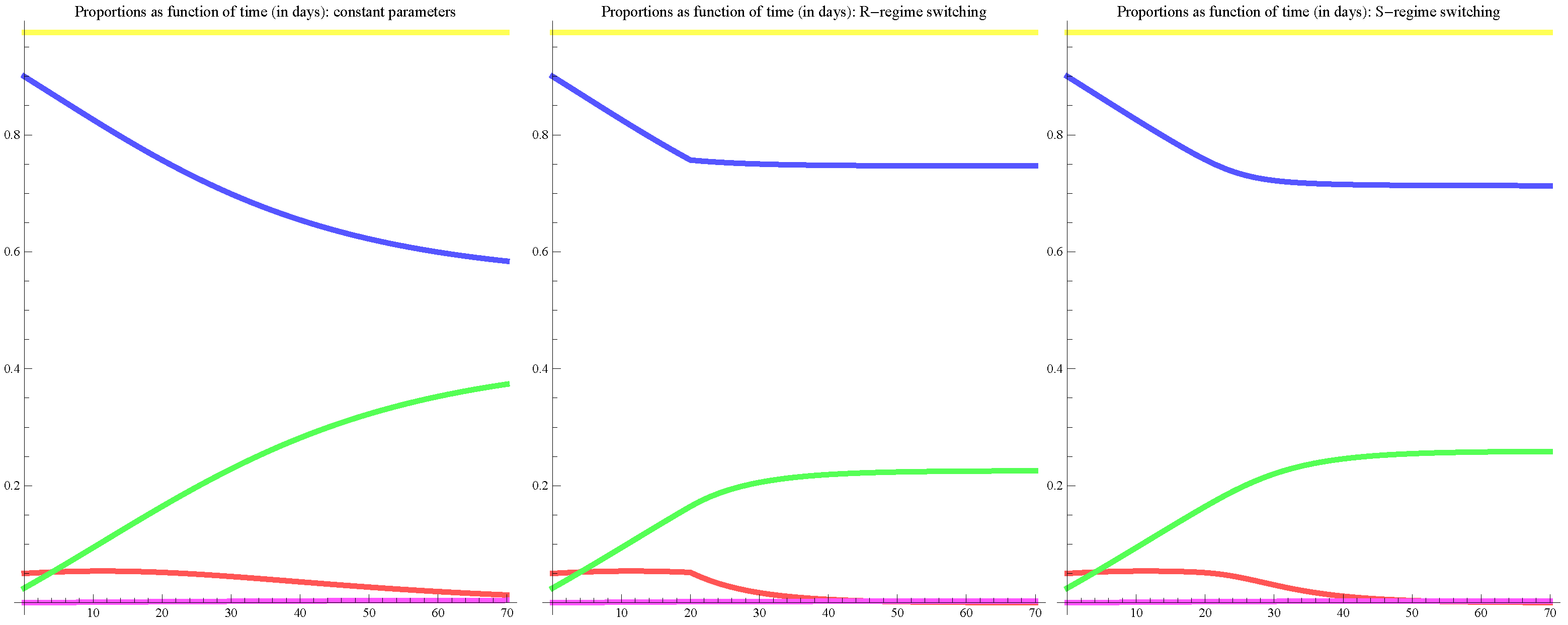
5. Results of the Model
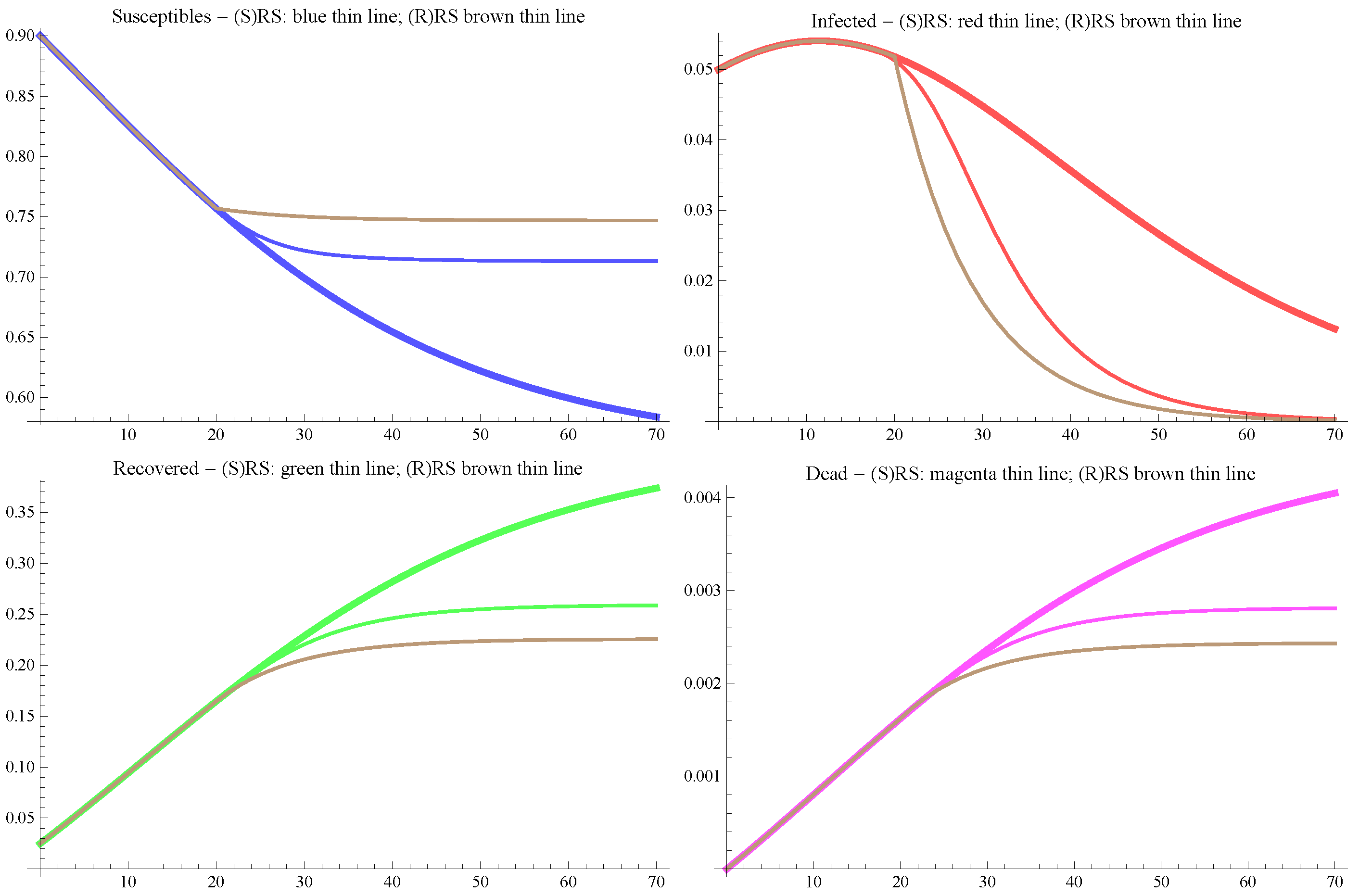
5.1. Results of the Model in the Basis Scenario
5.2. Results of the Model in the Extreme Scenarios I and II
6. On the Regime Switching SI(RD) ODE Models: Existence and Unicity Results
- (i)
- is measurable in the variable t, for fixed , and continuous in the variable , for fixed t.
- (ii)
- there exists a Lebesgue integrable function , defined on a neighbourhood of the initial time, let us say I, such that for .
7. Discussion, Conclusions and Further Work
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kermack, W.O.; McKendrick, A.G.; Walker, G.T. A contribution to the mathematical theory of epidemics. Proc. R. Soc. Lond. Ser. A Contain. Pap. A Math. Phys. Character 1927, 115, 700–721. [Google Scholar] [CrossRef] [Green Version]
- Martcheva, M. An Introduction to Mathematical Epidemiology; Texts in Applied Mathematics; Springer: New York, NY, USA, 2015; Volume 61. [Google Scholar] [CrossRef]
- Keeling, M.J.; Rohani, P. Modeling Infectious Diseases in Humans and Animals. Princeton University Press: Princeton, NJ, USA, 2008. [Google Scholar]
- Brauer, F.; Castillo-Chavez, C. Mathematical Models in Population Biology and Epidemiology, 2nd ed.; Texts in Applied Mathematics; Springer: New York, NY, USA, 2011. [Google Scholar]
- Giordano, G.; Blanchini, F.; Bruno, R.; Colaneri, P.; Di Filippo, A.; Di Matteo, A.; Colaneri, M. Modelling the COVID-19 epidemic and implementation of population-wide interventions in Italy. Nat. Med. 2020, 26, 855–860. [Google Scholar] [CrossRef]
- Bertozzi, A.L.; Franco, E.; Mohler, G.; Short, M.B.; Sledge, D. The challenges of modeling and forecasting the spread of COVID-19. Proc. Natl. Acad. Sci. USA 2020, 117, 16732–16738. [Google Scholar] [CrossRef]
- Carcione, J.M.; Santos, J.E.; Bagaini, C.; Ba, J. A Simulation of a COVID-19 Epidemic Based on a Deterministic SEIR Model. Front. Public Health 2020, 8, 230. [Google Scholar] [CrossRef] [PubMed]
- Shankar, S.; Mohakuda, S.S.; Kumar, A.; Nazneen, P.S.; Yadav, A.K.; Chatterjee, K.; Chatterjee, K. Systematic review of predictive mathematical models of COVID-19 epidemic. Med. J. Armed Forces India 2021, 77, S385–S392. [Google Scholar] [CrossRef]
- Brauer, F.; Castillo-Chávez, C. Mathematical Models in Population Biology and Epidemiology; Texts in Applied Mathematics; Springer: New York, NY, USA, 2001; Volume 40. [Google Scholar] [CrossRef]
- Moein, S.; Nickaeen, N.; Roointan, A.; Borhani, N.; Heidary, Z.; Javanmard, S.H.; Ghaisari, J.; Gheisari, Y. Inefficiency of SIR models in forecasting COVID-19 epidemic: A case study of Isfahan. Sci. Rep. 2021, 11, 4725. [Google Scholar] [CrossRef]
- De la Sen, M.; Ibeas, A.; Alonso-Quesada, S.; Nistal, R. On a New Epidemic Model with Asymptomatic and Dead-Infective Subpopulations with Feedback Controls Useful for Ebola Disease. Discret. Dyn. Nat. Soc. 2017, 2017, 4232971. [Google Scholar] [CrossRef] [Green Version]
- ud Din, R.; Algehyne, E.A. Mathematical analysis of COVID-19 by using SIR model with convex incidence rate. Results Phys. 2021, 23, 103970. [Google Scholar] [CrossRef]
- Sameni, R. Mathematical Modeling of Epidemic Diseases; A Case Study of the COVID-19 Coronavirus. arXiv 2020, arXiv:2003.11371. [Google Scholar]
- Asamoah, J.K.K.; Jin, Z.; Sun, G.Q.; Seidu, B.; Yankson, E.; Abidemi, A.; Oduro, F.; Moore, S.E.; Okyere, E. Sensitivity assessment and optimal economic evaluation of a new COVID-19 compartmental epidemic model with control interventions. Chaos Solitons Fractals 2021, 146, 110885. [Google Scholar] [CrossRef] [PubMed]
- Xue, L.; Jing, S.; Miller, J.C.; Sun, W.; Li, H.; Estrada-Franco, J.G.; Hyman, J.M.; Zhu, H. A data-driven network model for the emerging COVID-19 epidemics in Wuhan, Toronto and Italy. Math. Biosci. 2020, 326, 108391. [Google Scholar] [CrossRef] [PubMed]
- Glass, D.H. European and US lockdowns and second waves during the COVID-19 pandemic. Math. Biosci. 2020, 330, 108472. [Google Scholar] [CrossRef] [PubMed]
- Esquível, M.L.; Patrício, P.; Guerreiro, G.R. From ODE to Open Markov Chains, via SDE: An application to models for infections in individuals and populations. Comput. Math. Biophys. 2020, 8, 180–197. [Google Scholar] [CrossRef]
- Petrica, M.; Leordeanu, M.; Stochitoiu, R.D.; Popescu, I. A regime switching on Covid-19 analysis and prediction in Romania. arXiv 2020, arXiv:2007.13494. [Google Scholar]
- Martínez, V. A Modified SIRD Model to Study the Evolution of the COVID-19 Pandemic in Spain. Symmetry 2021, 13, 723. [Google Scholar] [CrossRef]
- Esquível, M.L. The Multi-Compartment SI(RD) Model with Regime Switching: An Application to COVID-19 Pandemic. Presentation at Seminário de Estatística e Gestão do Risco do Centro de Matemática e Aplicações, FCT Nova, New University of Lisbon, in November. 2020. Available online: https://drive.google.com/file/d/15sZZXUiB20OfeoHX5RFfYsLEWLyOvA_Y/view (accessed on 1 December 2020).
- Esquível, M.; Krasii, N.; Guerreiro, G.; Patrício, P. The Multi-Compartment SI(RD) Model with Regime Switching: An Application to COVID-19 Pandemic. Paper Submission to Journal of Applied Statistics in November. 2020. Available online: https://drive.google.com/file/d/1KKa4NEAH9O2HY38EACNfOlBTkavbCMFH/view?usp=sharing (accessed on 27 November 2020).
- Ndaïrou, F.; Torres, D.F.M. Mathematical Analysis of a Fractional COVID-19 Model Applied to Wuhan, Spain and Portugal. Axioms 2021, 10, 135. [Google Scholar] [CrossRef]
- Vaz, S.; Torres, D.F.M. A Discrete-Time Compartmental Epidemiological Model for COVID-19 with a Case Study for Portugal. Axioms 2021, 10, 314. [Google Scholar] [CrossRef]
- Doutor, P.; Rodrigues, P.; Soares, M.d.C.; Chalub, F.A.C.C. Optimal vaccination strategies and rational behaviour in seasonal epidemics. J. Math. Biol. 2016, 73, 1437–1465. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Machado, B.; Antunes, L.; Caetano, C.; Pereira, J.F.; Nunes, B.; Patrício, P. The impact of vaccination on the evolution of COVID-19 in Portugal. Math. Biosci. Eng. 2022, 19, 936–952. [Google Scholar] [CrossRef]
- Caetano, C.; Morgado, M.L.; Patrício, P.; Pereira, J.F.; Nunes, B. Mathematical Modelling of the Impact of Non-Pharmacological Strategies to Control the COVID-19 Epidemic in Portugal. Mathematics 2021, 9, 1084. [Google Scholar] [CrossRef]
- Brauer, F.; Castillo-Chavez, C.; Feng, Z. Mathematical Models in Epidemiology; Texts in Applied Mathematics; Springer: New York, NY, USA, 2019; Volume 69. [Google Scholar] [CrossRef]
- Sun, C.; Zhao, H.; Lin, Y.; Dai, Y. An SIRS Epidemic Model Incorporating Media Coverage with Time Delay. Comput. Math. Methods Med. 2014, 2014, 680743. [Google Scholar]
- Birkhoff, G.; Rota, G.C. Ordinary Differential Equations, 3rd ed.; John Wiley & Sons: New York, NY, USA; Chichester, UK; Brisbane, Australia, 1978. [Google Scholar]
- Cartan, H. Cours de Calcul Différentiel; Hermann: Paris, France, 1977. [Google Scholar]
- Hartman, P. Ordinary Differential Equations; Classics in Applied Mathematics; Society for Industrial and Applied Mathematics (SIAM): Philadelphia, PA, USA, 2002; Volume 38. [Google Scholar] [CrossRef]
- Wolfram Research, Inc. Mathematica, Version 12.3.1; Wolfram Research, Inc.: Champaign, IL, USA, 2021. [Google Scholar]
- Colson, P.; Levasseur, A.; Delerce, J.; Chaudet, V.; Bossi, M.; Ben Khedher, P.E.; Fournier, J.C.; Raoult, D. Dramatic increase in the SARS-CoV-2 mutation rate and low mortality rate during the second epidemic in summer in Marseille. Pre-Prints Covid Du IHU 2020, 1, 1–20. [Google Scholar] [CrossRef]
- Walsh, K.A.; Jordan, K.; Clyne, B.; Rohde, D.; Drummond, L.; Byrne, P.; Ahern, S.; Carty, P.G.; O’Brien, K.K.; O’Murchu, E.; et al. SARS-CoV-2 detection, viral load and infectivity over the course of an infection. J. Infect. 2020, 81, 357–371. [Google Scholar] [CrossRef]
- La Scola, B.; Le Bideau, M.; Andreani, J.; Hoang, V.T.; Grimaldier, C.; Colson, P.; Gautret, P.; Raoult, D. Viral RNA load as determined by cell culture as a management tool for discharge of SARS-CoV-2 patients from infectious disease wards. Eur. J. Clin. Microbiol. Infect. Dis. 2020, 39, 1059–1061. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, N.M.; Laydon, D.; Nedjati-Gilani, G.; Imai, N.; Ainslie, K.; Baguelin, M.; Bhatia, S.; Boonyasiri, A.; Cucunubá, Z.; Cuomo-Dannenburg, G.; et al. Impact of Nonpharmaceutical Interventions (NPIs) to Reduce COVID-19 Mortality and Healthcare Demand; Imperial College COVID-19 Response Team, Imperial College: London, UK, 2020. [Google Scholar] [CrossRef]
- Zhang, S.; Diao, M.; Yu, W.; Pei, L.; Lin, Z.; Chen, D. Estimation of the reproductive number of novel coronavirus (COVID-19) and the probable outbreak size on the Diamond Princess cruise ship: A data-driven analysis. Int. J. Infect. Dis. 2020, 93, 201–204. [Google Scholar] [CrossRef]
- Konishi, S.; Kitagawa, G. Information Criteria and Statistical Modeling; Springer: New York, NY, USA, 2008. [Google Scholar]
- Salpeter, E.E.; Salpeter, S.R. Mathematical Model for the Epidemiology of Tuberculosis, with Estimates of the Reproductive Number and Infection-Delay Function. Am. J. Epidemiol. 1998, 147, 398–406. [Google Scholar] [CrossRef] [PubMed]
- Kajiwara, T.; Sasaki, T.; Takeuchi, Y. Construction of Lyapunov functionals for delay differential equations in virology and epidemiology. Nonlinear Anal. Real World Appl. 2012, 13, 1802–1826. [Google Scholar] [CrossRef]
- Fridman, E. Introduction to Time-Delay Systems; Systems & Control: Foundations & Applications; Birkhäuser/Springer: Cham, Switzerland, 2014. [Google Scholar] [CrossRef]
- Böhning, D.; Rocchetti, I.; Maruotti, A.; Holling, H. Estimating the undetected infections in the COVID-19 outbreak by harnessing capture, recapture methods. Int. J. Infect. Dis. 2020, 97, 197–201. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.L.; Mertens, A.N.; Crider, Y.S.; Nguyen, A.; Pokpongkiat, N.N.; Djajadi, S.; Seth, A.; Hsiang, M.S.; Colford, J.M.; Reingold, A.; et al. Substantial underestimation of SARS-CoV-2 infection in the United States. Nat. Commun. 2020, 11, 4507. [Google Scholar] [CrossRef]
- Hasell, J.; Mathieu, E.; Beltekian, D.; Macdonald, B.; Giattino, C.; Ortiz-Ospina, E.; Roser, M.; Ritchie, H. A cross-country database of COVID-19 testing. Sci. Data 2020, 7, 345. [Google Scholar] [CrossRef] [PubMed]
- The Economist. Fatal Flaws: COVID-19 death toll appears higher than official figures suggest. Economist, 4 April 2020.
- Coddington, E.A.; Levinson, N. Theory of Ordinary Differential Equations; McGraw-Hill Book Company, Inc.: New York, NY, USA; Toronto, ON, USA; London, UK, 1955. [Google Scholar]
- Rudin, W. Real and Complex Analysis, 3rd ed.; McGraw-Hill Book Co.: New York, NY, USA, 1987. [Google Scholar]
- Kurzweil, J. Ordinary Differential Equations; Studies in Applied Mechanics; Elsevier Scientific Publishing Co.: Amsterdam, The Netherlands, 1986; Volume 13, p. 440. [Google Scholar]
- Teschl, G. Ordinary Differential Equations and Dynamical Systems; Graduate Studies in Mathematics; American Mathematical Society: Providence, RI, USA, 2012; Volume 140. [Google Scholar] [CrossRef]
- Nevanlinna, F.; Nevanlinna, R. Absolute Analysis; Springer: New York, NY, USA; Heidelberg, Germany, 1973. [Google Scholar]
- Stokes, A. The Application of a fixed-point theorem to a variety of nonlinear stability problems. Proc. Natl. Acad. Sci. USA 1959, 45, 231–235. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Butcher, J.C. Numerical Methods for Ordinary Differential Equations, 3rd ed.; John Wiley & Sons, Ltd.: Chichester, UK, 2016. [Google Scholar] [CrossRef]
- Bluman, G.W.; Anco, S.C. Symmetry and Integration Methods for Differential Equations; Applied Mathematical Sciences; Springer: New York, NY, USA, 2002; Volume 154. [Google Scholar]
- Ouhadan, A.; El Kinani, E.H.; Hajar, M. Lie symmetries analysis for SIR model of epidemiology. Appl. Math. Sci. (Ruse) 2013, 7, 4595–4604. [Google Scholar] [CrossRef]
- Matadi, M.B. On the integrability of the SIRD epidemic model. Commun. Math. Biol. Neurosci. 2020, 2020, 55. [Google Scholar]
- Leach, P.G.L.; Andriopoulos, K. Application of symmetry and symmetry analyses to systems of first-order equations arising from mathematical modelling in epidemiology. In Symmetry in Nonlinear Mathematical Physics. Part 1, 2, 3; Natsīonal. Akad. Nauk Ukraïni, Īnst. Mat. Kiev: Kiev, Ukraine, 2004; Volume 3, pp. 159–169. [Google Scholar]
- Nucci, M.C. Using Lie symmetries in epidemiology. In Proceedings of the 2004 Conference on Differential Equations and Applications in Mathematical Biology, Nanaimo, BC, Canada, 18–23 July 2004. [Google Scholar]
- Golubitsky, M.; Schaeffer, D.G. Singularities and Groups in Bifurcation Theory. Vol. I; Applied Mathematical Sciences; Springer: New York, NY, USA, 1985; Volume 51. [Google Scholar] [CrossRef]
- Golubitsky, M.; Stewart, I.; Schaeffer, D.G. Singularities and Groups in Bifurcation Theory. Vol. II; Applied Mathematical Sciences; Springer: New York, NY, USA, 1988; Volume 69. [Google Scholar] [CrossRef]
| Periods | u | d | |||
|---|---|---|---|---|---|
| 0.833333 | 2.33474 | 0.166667 | −2.04242 | 2.33474 | |
| 0.487805 | 1.94555 | 0.512195 | 0.411811 | 0.411811 |
| Periods | St. Error | t-Stat | p-Value | Adj. | BIC | AIC | ||
|---|---|---|---|---|---|---|---|---|
| 3 | 0.0306118 | 0.00155902 | 19.6353 | 0.938957 | 123.008 | 120.57 | ||
| 11 | 0.0350414 | 0.00210826 | 16.621 | 0.867616 | 302.68 | 299.21 |
| Parameters | Extreme Scenario I | Basis Scenario | Extreme Scenario II |
|---|---|---|---|
| 0.3 | 0.5 | 0.7 | |
| 0.25 | 0 | 0.5 | |
| 5 | 7.5 | 10 |
| Regime | Susceptible | Infected | Recovered | Deaths | |||
|---|---|---|---|---|---|---|---|
| Constant | 0.5 | 0 | 7.5 | 0.588081 | 0.0147572 | 0.368176 | 0.00398521 |
| Smooth RS | 0.5 | 0 | 7.5 | 0.71313 | 0.000537805 | 0.258525 | 0.00280666 |
| Rough RS | 0.5 | 0 | 7.5 | 0.746942 | 0.000271394 | 0.225357 | 0.00243014 |
| Regime | Susceptible | Infected | Recovered | Deaths | |||
|---|---|---|---|---|---|---|---|
| Constant | 0.3 | 0.25 | 5 | 0.801685 | 0.000413846 | 0.107069 | 0.0658329 |
| e Smooth RS | 0.3 | 0.25 | 5 | 0.815603 | 0.0981462 | 0.0612436 | |
| Rough RS | 0.3 | 0.25 | 5 | 0.822341 | 0.0937749 | 0.058881 |
| Regime | Susceptible | Infected | Recovered | Deaths | |||
|---|---|---|---|---|---|---|---|
| Constant | 0.7 | 0.5 | 10 | 0.361501 | 0.0420978 | 0.0849807 | 0.48642 |
| Smooth RS | 0.7 | 0.5 | 10 | 0.604511 | 0.00413745 | 0.0279379 | 0.338413 |
| Rough RS | 0.7 | 0.5 | 10 | 0.67301 | 0.00232243 | 0.0214171 | 0.27825 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Esquível, M.L.; Krasii, N.P.; Guerreiro, G.R.; Patrício, P. The Multi-Compartment SI(RD) Model with Regime Switching: An Application to COVID-19 Pandemic. Symmetry 2021, 13, 2427. https://doi.org/10.3390/sym13122427
Esquível ML, Krasii NP, Guerreiro GR, Patrício P. The Multi-Compartment SI(RD) Model with Regime Switching: An Application to COVID-19 Pandemic. Symmetry. 2021; 13(12):2427. https://doi.org/10.3390/sym13122427
Chicago/Turabian StyleEsquível, Manuel L., Nadezhda P. Krasii, Gracinda R. Guerreiro, and Paula Patrício. 2021. "The Multi-Compartment SI(RD) Model with Regime Switching: An Application to COVID-19 Pandemic" Symmetry 13, no. 12: 2427. https://doi.org/10.3390/sym13122427
APA StyleEsquível, M. L., Krasii, N. P., Guerreiro, G. R., & Patrício, P. (2021). The Multi-Compartment SI(RD) Model with Regime Switching: An Application to COVID-19 Pandemic. Symmetry, 13(12), 2427. https://doi.org/10.3390/sym13122427







