Significance of Non-Linear Terms in the Relativistic Coupled-Cluster Theory in the Determination of Molecular Properties
Abstract
1. Introduction
2. Theory and Implementation
3. Results and Discussions
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Eliav, E.; Kaldor, U.; Ishikawa, Y. Relativistic coupled cluster theory based on the no-pair dirac–coulomb–breit hamiltonian: Relativistic pair correlation energies of the xe atom. Int. J. Quantum Chem. 1994, 52, 205–214. [Google Scholar] [CrossRef]
- Nataraj, H.S.; Sahoo, B.K.; Das, B.P.; Mukherjee, D. Intrinsic Electric Dipole Moments of Paramagnetic Atoms: Rubidium and Cesium. Phys. Rev. Lett. 2008, 101, 033002. [Google Scholar] [CrossRef] [PubMed]
- Bishop, R.F. Microscopic Quantum Many-Body Theories and Their Applications; Springer: Berlin/Heidelberg, Germany, 1998; Chapter 1. [Google Scholar]
- Sahoo, B.K.; Kumar, P. Relativistic coupled-cluster-theory analysis of unusually large correlation effects in the determination of gj factors in Ca+. Phys. Rev. A 2017, 96, 012511. [Google Scholar] [CrossRef]
- Abe, M.; Gopakumar, G.; Hada, M.; Das, B.P.; Tatewaki, H.; Mukherjee, D. Application of relativistic coupled-cluster theory to the effective electric field in YbF. Phys. Rev. A 2014, 90, 022501. [Google Scholar] [CrossRef]
- Landau, L. On the conservation laws for weak interactions. Nucl. Phys. 1957, 3, 127. [Google Scholar] [CrossRef]
- Luders, G. Proof of the TCP theorem. Ann. Phys. 2000, 281, 1004. [Google Scholar] [CrossRef]
- Prasannaa, V.S.; Vutha, A.C.; Abe, M.; Das, B.P. Mercury Monohalides: Suitability for Electron Electric Dipole Moment Searches. Phys. Rev. Lett. 2015, 114, 183001. [Google Scholar] [CrossRef] [PubMed]
- Abe, M.; Prasannaa, V.S.; Das, B.P. Application of the finite-field coupled-cluster method to calculate molecular properties relevant to electron electric-dipole-moment searches. Phys. Rev. A 2018, 97, 032515. [Google Scholar] [CrossRef]
- Shuman, E.S.; Barry, J.F.; DeMille, D. Laser cooling of a diatomic molecule. Nature 2010, 467, 820. [Google Scholar] [CrossRef]
- Andreev, V.; Ang, D.G.; DeMille, D.; Doyle, J.M.; Gabrielse, G.; Haefner, J.; Hutzler, N.R.; Lasner, Z.; Meisenhelder, C.; Leary, B.R.; et al. Improved limit on the electric dipole moment of the electron. Nature 2018, 562, 355. [Google Scholar]
- Cesarotti, C.; Lu, Q.; Nakai, Y.; Parikh, A.; Reece, M. Interpreting the electron EDM constraint. J. High Energy Phys. 2019, 59, 2019. [Google Scholar] [CrossRef]
- Fuyuto, K.; Hisano, J.; Senaha, E. Toward verification of electroweak baryogenesis by electric dipole moments. Phys. Lett. B 2016, 755, 491. [Google Scholar] [CrossRef]
- Das, B.P. Aspects of Many-Body Effects in Molecules and Extended Systems; Mukherjee, D., Ed.; Springer: Berlin/Heidelberg, Germany, 1989; p. 411. [Google Scholar]
- Sasmal, S.; Pathak, H.; Nayak, M.K.; Vaval, N.; Pal, S. Calculation of P,T-odd interaction constant of PbF using Z-vector method in the relativistic coupled-cluster framework. J. Chem. Phys. 2015, 143, 084119. [Google Scholar] [CrossRef] [PubMed]
- Sasmal, S.; Pathak, H.; Nayak, M.K.; Vaval, N.; Pal, S. Relativistic coupled-cluster study of RaF as a candidate for the parity- and time-reversal-violating interaction. Phys. Rev. A 2016, 93, 062506. [Google Scholar] [CrossRef]
- Sunaga, A.; Prasannaa, V.S.; Abe, M.; Hada, M.; Das, B.P. Enhancement factors of parity- and time-reversal-violating effects for monofluorides. Phys. Rev. A 2018, 98, 042511. [Google Scholar] [CrossRef]
- Sunaga, A.; Prasannaa, V.S.; Abe, M.; Hada, M.; Das, B.P. Ultracold mercury alkali-metal molecules for electron-electric-dipole-moment searches. Phys. Rev. A 2019, 99, 040501(R). [Google Scholar] [CrossRef]
- Fazil, N.M.; Prasannaa, V.S.; Latha, K.V.P.; Abe, M.; Das, B.P. RaH as a potential candidate for electron electric-dipole-moment searches. Phys. Rev. A 2019, 99, 052502. [Google Scholar] [CrossRef]
- Prasannaa, V.S.; Shitara, N.; Sakurai, A.; Abe, M.; Das, B.P. Enhanced sensitivity of the electron electric dipole moment from YbOH: The role of theory. Phys. Rev. A 2019, 99, 062502. [Google Scholar] [CrossRef]
- Singh, Y.; Sahoo, B.K.; Das, B.P. Correlation trends in the ground-state static electric dipole polarizabilities of closed-shell atoms and ions. Phys. Rev. A 2013, 88, 062504. [Google Scholar] [CrossRef]
- Shavitt, I.; Bartlett, R.J. Many Body Methods in Chemistry and Physics; Cambridge University Press: Cambridge, UK, 2009. [Google Scholar]
- Yang, Z.; Li, J.; Lin, Q.; Xu, L.; Wang, H.; Yang, T.; Yin, J. Laser-cooled HgF as a promising candidate to measure the electric dipole moment of the electron. Phys. Rev. A 2019, 99, 032502. [Google Scholar] [CrossRef]
- Aggarwal, P.; Bethlem, H.L.; Borschevsky, A.; Denis, M.; Esajas, K.; Haase, P.A.B.; Hao, Y.; Hoekstra, S.; Jungmann, K.; Meijknecht, T.B.; et al. The NL-eEDM collaboration. Measuring the electric dipole moment of the electron in BaF. Eur. Phys. J. D 2018, 72, 197. [Google Scholar] [CrossRef]
- Vutha, A.C.; Horbatsch, M.; Hessels, E.A. Oriented Polar Molecules in a Solid Inert-Gas Matrix: A Proposed Method for Measuring the Electric Dipole Moment of the Electron. Atoms 2018, 6, 3. [Google Scholar] [CrossRef]
- Ernst, W.E.; Kandler, J.; Kindt, S.; Torring, T. Electric dipole moment of SrF X2Σ+ from high-precision stark effect measurements. Chem. Phys. Lett. 1985, 113, 351. [Google Scholar] [CrossRef]
- Cizek, J. Advances in Chemical Physics, Volume XIV: Correlation Effects in Atoms and Molecules; Lefebvre, W.C., Moser, C., Eds.; Interscience Publishers: New York, NY, USA, 1969. [Google Scholar]
- Yanai, T.; Nakano, H.; Nakajima, T.; Tsuneda, T.; Hirata, S.; Kawashima, Y.; Nakao, Y.; Kamiya, M.; Sekino, H.; Hirao, K. UTCHEM: A Program for ab initio Quantum Chemistry; Lecture Notes in Computer Science; Goos, G., Hartmanis, J., van Leeuwen, J., Eds.; Springer: Berlin/Heidelberg, Germany, 2003; Volume 2660, p. 84. [Google Scholar]
- Yanai, T.; Nakajima, T.; Ishikawa, Y.; Hirao, K. A new computational scheme for the Dirac Hartree Fock method employing an efficient integral algorithm. J. Chem. Phys. 2001, 114, 6526. [Google Scholar] [CrossRef]
- Abe, M.; Yanai, T.; Nakajima, T.; Hirao, K. A four-index transformation in Dirac’s four-component relativistic theory. Chem. Phys. Lett. 2004, 388, 68. [Google Scholar] [CrossRef]
- Visscher, L.; Jensen, H.J.A.; Saue, T.; Bast, R.; Dubillard, S.; Dyall, K.G.; Ekstrom, U.; Eliav, E.; Fleig, T.; Gomes, A.S.P.; et al. DIRAC: A Relativistic ab initio Electronic Structure Program. Release DIRAC08 2008. [Google Scholar]
- Kvasnicka, V.; Laurinc, V.; Biskupic, S. Coupled-cluster approach in electronic structure theory of molecules. Phys. Rep. 1982, 90, 159. [Google Scholar] [CrossRef]
- Prasannaa, V.S.; Sreerekha, S.; Abe, M.; Bannur, V.M.; Das, B.P. Permanent electric dipole moments of alkaline-earth-metal monofluorides: Interplay of relativistic and correlation effects. Phys. Rev. A 2016, 93, 042504. [Google Scholar] [CrossRef]
- Slater, J.C. The Theory of Complex Spectra. Phys. Rev. 1929, 34, 1293. [Google Scholar] [CrossRef]
- Condon, E.U. The Theory of Complex Spectra. Phys. Rev. 1930, 36, 1121. [Google Scholar] [CrossRef]
- Yanai, T.; Harrison, R.J.; Nakajima, T.; Ishikawa, Y.; Hirao, K. New implementation of molecular double point group symmetry in four component relativistic Gaussian type spinors. Int. J. Quantum Chem. 2007, 107, 1382. [Google Scholar] [CrossRef]
- Knecht, S.; Fux, S.; van Meer, R.; Visscher, L.; Reiher, M.; Saue, T. Mossbauer spectroscopy for heavy elements: A relativistic benchmark study of mercury. Theor. Chem. Acc. 2011, 129, 631. [Google Scholar] [CrossRef]
- Cheung, N.-H.; Cool, T.A. Franck-Condon Factors and r-Centroids for the B2ΣX2Σ Systems of HgCl, HgBr, and MgI. J. Quant. Spectrosc. Radiat. Transf. 1979, 21, 397. [Google Scholar] [CrossRef]
- Langhoff, S.R.; Bauschlicher, C.W., Jr.; Partridge, H.; Ahlrichs, R. Theoretical study of the dipole moments of selected alkaline earth halides. J. Chem. Phys. 1986, 84, 5025. [Google Scholar] [CrossRef]
- Huber, K.P.; Herzberg, G. (Eds.) Molecular Spectra and Molecular Structure, IV. Constants of Diatomic Molecules; Reinhold: New York, NY, USA, 1979. [Google Scholar]
- Mestdagh, J.M.; Visticot, J.P. Semiempirical electrostatic polarization model of the ionic bonding in alkali and alkaline earth hydroxides and halides. Chem. Phys. 1991, 155, 79. [Google Scholar] [CrossRef]
- Ryzlewicz, C.; Torring, T. Formation and microwave spectrum of the 2Σ-radical barium-monofluoride. Chem. Phys. 1980, 51, 329. [Google Scholar] [CrossRef]
- Dyall, K.G. Relativistic Quadruple-Zeta and Revised Triple-Zeta and Double-Zeta Basis Sets for the 4p, 5p, and 6p Elements. Theor. Chem. Acc. 2006, 115, 441. [Google Scholar] [CrossRef]
- Schuchardt, K.L.; Didier, B.T.; Elsethagen, T.; Sun, L.; Gurumoorthi, V.; Chase, J.; Li, J.; Windus, T.L. Basis Set Exchange: A Community Database for Computational Sciences. J. Chem. Inf. Model 2007, 47, 1045. [Google Scholar] [CrossRef]
- Noro, T.; Sekiya, M.; Koga, T. Segmented contracted basis sets for atoms H through Xe: Sapporo-(DK)-nZP sets (n = D, T, Q). Theor. Chem. Acc. 2012, 131, 1124. [Google Scholar] [CrossRef]
- Jardali, F.; Korek, M.; Younes, G. Theoretical calculation of the low-lying doublet electronic states of the SrF molecule. Canad. J. Phys. 2014, 92, 1223. [Google Scholar] [CrossRef]
- Sasmal, S.; Pathak, H.; Nayak, M.K.; Vaval, N.; Pal, S. Implementation of the Z-vector method in the relativistic-coupled-cluster framework to calculate first-order energy derivatives: Application to the SrF molecule. Phys. Rev. A 2015, 91, 030503(R). [Google Scholar] [CrossRef]
- Hao, Y.; Pasteka, L.F.; Visscher, L.; Aggarwal, P.; Bethlem, H.L.; Boeschoten, A.; Borschevsky, A.; Denis, M.; Esajas, K.; Hoekstra, S.; et al. High accuracy theoretical investigations of CaF, SrF, and BaF and implications for laser-cooling. J. Chem. Phys. 2019, 151, 034302. [Google Scholar] [CrossRef] [PubMed]
- Tohme, S.N.; Korek, M. Theoretical study of the electronic structure with dipole moment calculations of barium monofluoride. J. Quant. Spectrosc. Radiat. Transf. 2015, 167, 82. [Google Scholar] [CrossRef]
- Talukdar, K.; Nayak, M.K.; Vaval, N.; Pal, S. Relativistic coupled-cluster study of BaF in search of CP violation. arXiv 2020, arXiv:2002.02505. [Google Scholar] [CrossRef]
- Kozlov, M.G.; Titov, A.V.; Mosyagin, N.S.; Souchko, P.V. Enhancement of the electric dipole moment of the electron in the BaF molecule. Phys. Rev. A 1997, 56, R3326(R). [Google Scholar] [CrossRef]
- Nayak, M.K.; Chaudhuri, R.K. Ab initio calculation of P, T-odd interaction constant in BaF: A restricted active space configuration interaction approach. J. Phys. B At. Mol. Opt. Phys. 2006, 39, 1231. [Google Scholar] [CrossRef]
- Meyer, E.R.; Bohn, J.L.; Deskevich, M.P. Candidate molecular ions for an electron electric dipole moment experiment. Phys. Rev. A 2006, 73, 062108. [Google Scholar] [CrossRef]
- Meyer, E.R.; Bohn, J.L. Prospects for an electron electric-dipole moment search in metastable ThO and ThF+. Phys. Rev. A 2008, 78, 010502(R). [Google Scholar] [CrossRef]
- Ernst, W.E.; Kandler, J.; Torring, T. Hyperfine structure and electric dipole moment of BaF X2Σ+. J. Chem. Phys. 1986, 84, 4769. [Google Scholar] [CrossRef]
- Dmitriev, Y.Y.; Khait, Y.G.; Kozlov, M.G.; Labzovsky, L.N.; Mitrushenkov, A.O.; Shtoff, A.V.; Titov, A.V. Calculation of the spin-rotational Hamiltonian including P- and P, T-odd weak interaction terms for HgF and PbF molecules. Phys. Lett. A 1992, 167, 280. [Google Scholar] [CrossRef]
- Prasannaa, V.S.; Abe, M.; Das, B.P. The role of molecular electric dipole moments of mercury monohalides in the search for the electron electric dipole moment. Asian J. Phys. 2016, 25, 1259. [Google Scholar]
- Wadt, W.R. The electronic structure of HgCl and HgBr. Appl. Phys. Lett. 1979, 34, 658. [Google Scholar] [CrossRef]
- Torring, T.; Ernst, W.E.; Kindt, S. Dipole moments and potential energies of the alkaline earth monohalides from an ionic model. J. Chem. Phys. 1984, 81, 4614. [Google Scholar] [CrossRef]
- Allouche, A.R.; Wannous, G.; Aubert-Frecon, M. A ligand-field approach for the low-lying states of Ca, Sr and Ba monohalides. Chem. Phys. 1993, 170, 11. [Google Scholar] [CrossRef]
- Kobus, J.; Moncrieff, D.; Wilson, S. Comparison of the electric moments obtained from finite basis set and finite-difference Hartree-Fock calculations for diatomic. Phys. Rev. A 2000, 62, 062503. [Google Scholar] [CrossRef]
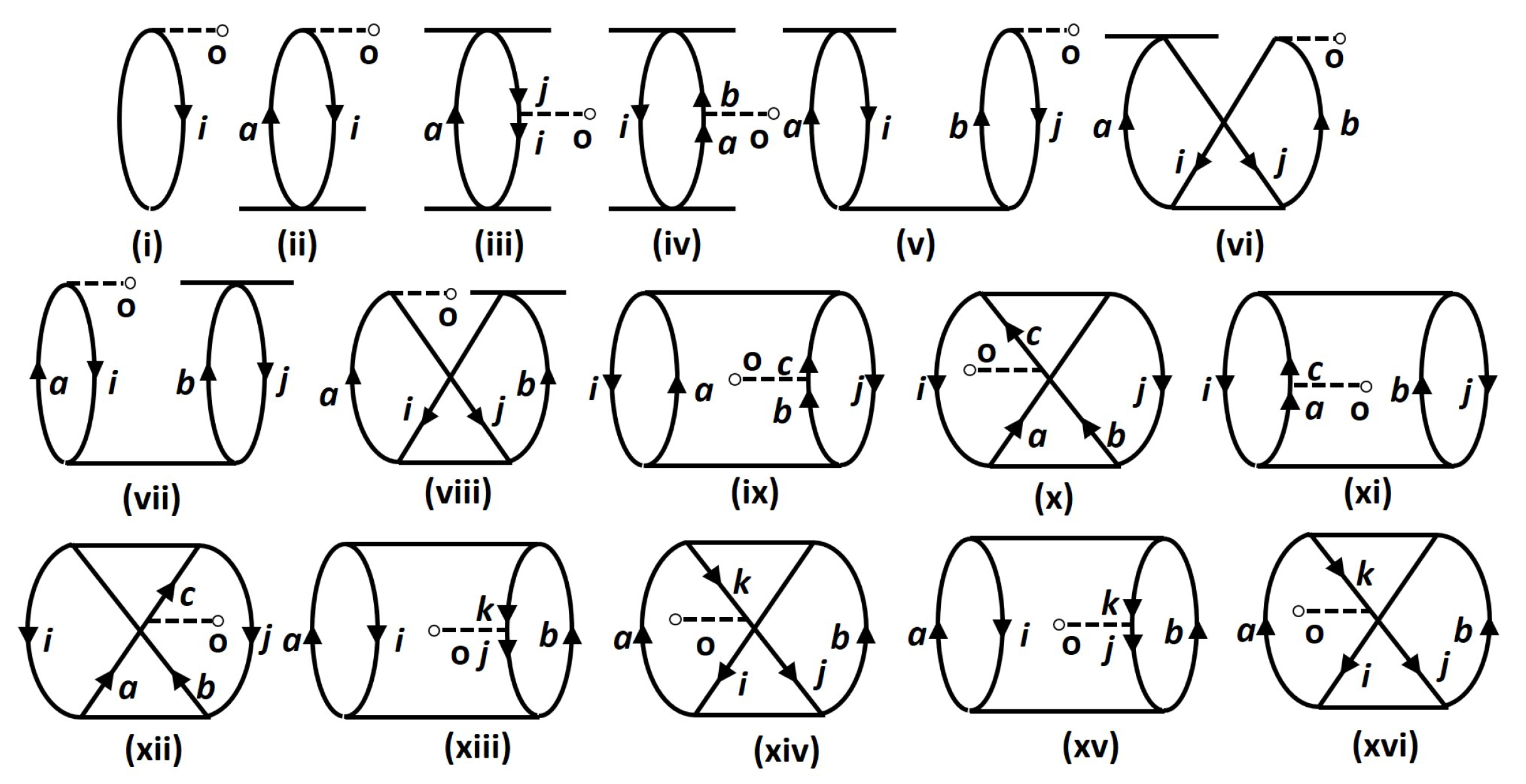

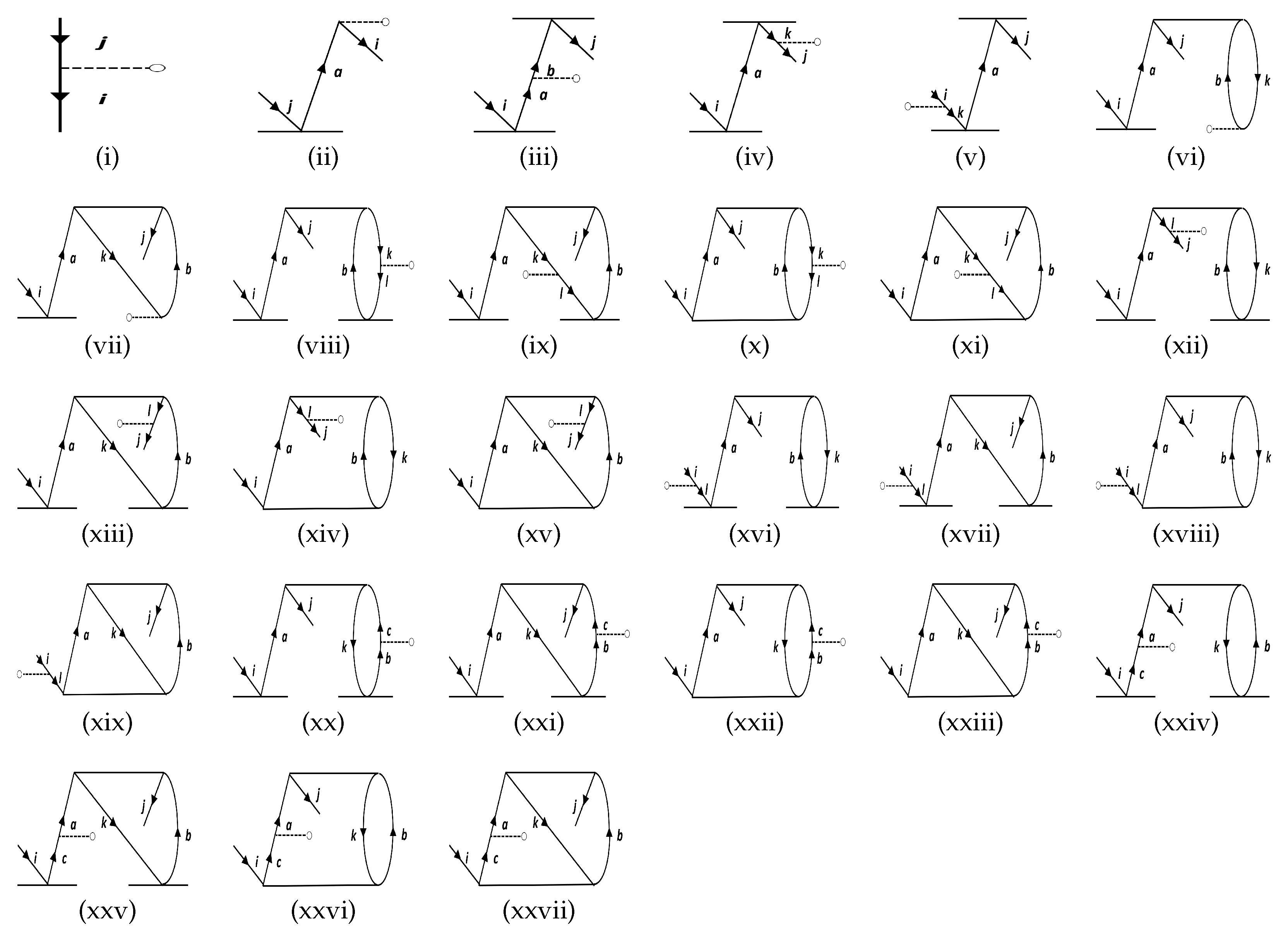
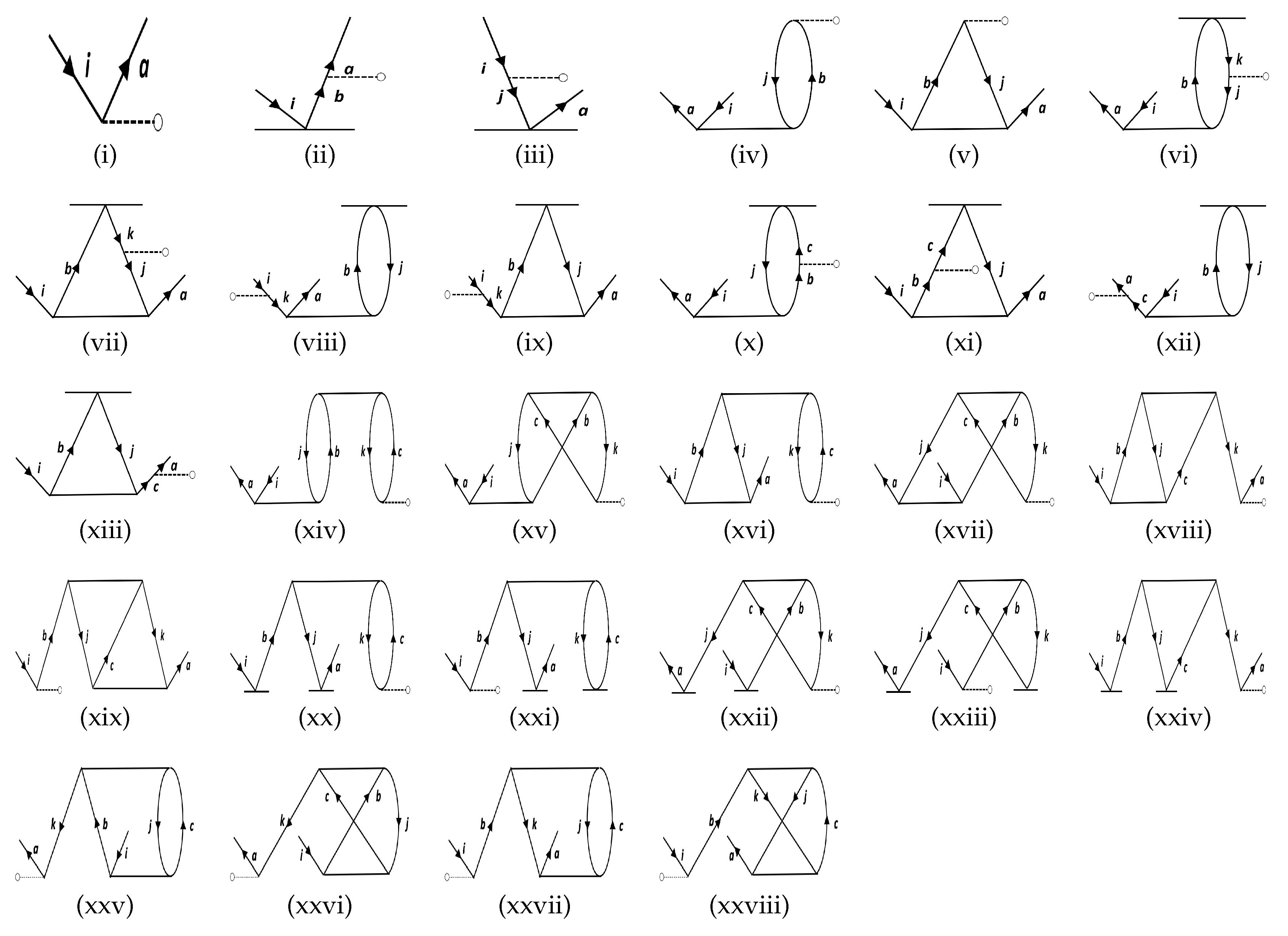

| Molecule | Method | PDM | |
|---|---|---|---|
| SrF | CASSCF-MRCI [46] | 3.36 | |
| CASSCF-RSPT2 [46] | 3.61 | ||
| Z-vector [47] | 3.45 | ||
| LERCCSD [9,33] | 3.6 | 2.17 | |
| FFCCSD [9] | 3.62 | 2.16 | |
| X2C-MRCI [48] | 3.20 | ||
| X2C-FSCC [48] | 3.46 | ||
| DF (This work) | 2.99 | 1.54 | |
| LERCCSD (This work ) | 3.57 | 2.15 | |
| nLERCCSD (This work) | 3.60 | 2.16 | |
| Experiment [26] | 3.4676(1) | ||
| BaF | MRCI [49] | 2.96 | |
| LERCCSD [33] | 3.4 | 6.50 | |
| FFCCSD [9] | 3.41 | 6.46 | |
| X2C-MRCI [48] | 2.90 | ||
| X2C-FSCC [48] | 3.23 | ||
| Z-vector [50] | 3.08 | ||
| ECP-RASSCF [51] | 7.5 | ||
| RASCI [52] | 7.28 | ||
| MRCI [53] | 5.1 | ||
| MRCI [54] | 6.1 | ||
| DF (This work) | 2.61 | 4.81 | |
| LERCCSD (This work) | 3.32 | 6.45 | |
| nLERCCSD (This work) | 3.37 | 6.39 | |
| Experiment (PDM) [55] | 3.17(3) | ||
| HgF | CI [56] | 4.15 | 99.26 |
| LERCCSD [57] | 2.61 | ||
| MRCI [53] | 68 | ||
| MRCI [54] | 95 | ||
| DF (This work) | 4.11 | 105.69 | |
| LERCCSD [8] | 115.42 | ||
| FFCCSD [9] | 2.92 | 116.37 | |
| LERCCSD (This work) | 3.25 | 114.93 | |
| nLERCCSD (This work) | 3.45 | 113.77 | |
| HgCl | CI [58] | 3.28 | |
| LERCCSD [57] | 2.72 | ||
| LERCCSD [8] | 113.56 | ||
| FFCCSD [9] | 2.96 | 114.31 | |
| DF (This work) | 4.30 | 104.33 | |
| LERCCSD (This work) | 3.26 | 112.51 | |
| nLERCCSD (This work) | 3.45 | 110.94 | |
| HgBr | CI [58] | 2.62 | |
| LERCCSD [57] | 2.36 | ||
| LERCCSD [8] | 109.29 | ||
| FFCCSD [9] | 2.71 | 109.56 | |
| DF (This work) | 4.14 | 99.72 | |
| LERCCSD (This work) | 2.62 | 109.38 | |
| nLERCCSD (This work) | 2.94 | 107.42 | |
| HgI | LERCCSD [57] | 1.64 | |
| LERCCSD [8] | 109.3 | ||
| FFCCSD [9] | 2.06 | 109.56 | |
| DF (This work) | 3.61 | 99.27 | |
| LERCCSD (This work) | 1.50 | 110.00 | |
| nLERCCSD (This work) | 2.01 | 107.38 |
| Molecule | HgF | HgCl | HgBr | HgI | SrF | BaF | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Term | Diagram | L | nL | L | nL | L | nL | L | nL | L | nL | L | nL |
| DF | Figure 1i | 105.69 | 104.33 | 99.72 | 99.27 | 1.54 | 4.81 | ||||||
| Figure 1ii | 17.09 | 13.11 | 17.05 | 12.21 | 19.83 | 14.76 | 23.85 | 15.71 | 0.63 | 0.61 | 1.79 | 1.60 | |
| Figure 1iii | −1.85 | −0.28 | −2.01 | −0.25 | −2.65 | −0.62 | −3.66 | −0.41 | −1.86 | −1.00 | −7.65 | −2.00 | |
| Figure 1iv | −1.41 | 0.16 | −1.40 | 0.28 | −1.21 | 0.47 | −1.56 | 1.16 | −9.01 | 4.80 | −6.47 | 7.60 | |
| Figure 1v | 1.19 | 0.93 | 0.65 | 0.29 | 0.38 | −0.11 | 0.38 | −0.27 | 2.73 | 1.02 | 9.46 | 2.51 | |
| Figure 1vi | 0.05 | 0.08 | 0.06 | 0.05 | −0.01 | −0.07 | −0.03 | −0.09 | −4.91 | −7.93 | −1.49 | −2.39 | |
| Figure 1vii | 0.61 | 0.58 | 0.92 | 0.85 | 0.66 | 0.32 | 0.57 | 0.19 | 1.43 | 1.48 | 7.04 | 7.13 | |
| Figure 1viii | −1.31 | −1.27 | −1.24 | −1.18 | −0.91 | −0.63 | −1.26 | −0.98 | 9.63 | 9.91 | −2.49 | −2.32 | |
| Figure 1ix | −2.50 | −2.46 | −2.54 | −2.49 | −2.68 | −2.65 | −2.93 | −2.89 | 8.58 | 6.15 | 3.22 | 2.17 | |
| Figure 1x | −0.17 | −0.17 | −0.15 | −0.14 | −0.14 | −0.13 | −0.13 | −0.11 | −2.17 | −1.93 | −6.87 | −6.83 | |
| Figure 1xi | −1.22 | −1.40 | −1.50 | −1.47 | −1.65 | −1.85 | −1.96 | −1.99 | −1.96 | −2.17 | −7.54 | −7.71 | |
| Figure 1xii | −0.17 | −0.17 | −0.15 | −0.14 | −0.14 | −0.13 | −0.13 | −0.11 | −2.17 | −1.93 | −6.87 | −6.83 | |
| Figure 1xiii | −1.64 | −1.57 | −1.67 | −1.57 | −1.70 | −1.58 | −1.84 | −1.69 | −1.38 | −1.96 | −3.20 | −9.61 | |
| Figure 1xiv | −0.10 | −0.10 | −0.10 | −0.10 | −0.10 | −0.10 | −0.10 | −0.10 | −5.39 | −5.53 | −2.28 | −2.33 | |
| Figure 1xv | 0.77 | 0.74 | 0.36 | 0.37 | 0.08 | 0.12 | −0.37 | −0.21 | 4.42 | 4.51 | 2.82 | 3.18 | |
| Figure 1xvi | −0.10 | −0.10 | −0.10 | −0.10 | −0.10 | −0.10 | −0.10 | −0.10 | −5.39 | −5.53 | −2.28 | −2.33 | |
| Total | 114.93 | 113.77 | 112.51 | 110.94 | 109.38 | 107.42 | 110.00 | 107.38 | 2.15 | 2.16 | 6.45 | 6.39 | |
| Molecule | HgF | HgCl | HgBr | HgI | SrF | BaF | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Term | Diagram | L | nL | L | nL | L | nL | L | nL | L | nL | L | nL |
| DF | Figure 1i | −767.04 | −925.61 | −1002.31 | −1075.83 | −375.75 | −578.39 | ||||||
| Figure 1ii | −0.60 | −0.78 | −0.83 | −1.01 | −1.26 | −1.54 | −1.92 | −2.33 | 0.63 | 0.65 | 0.80 | 0.82 | |
| Figure 1iii | 0.21 | 0.04 | 0.26 | 0.06 | 0.34 | 0.11 | 0.48 | 0.23 | 0.14 | −0.01 | 0.19 | −0.02 | |
| Figure 1iv | −0.45 | 0.05 | −0.48 | 0.07 | −0.62 | 0.13 | −0.79 | 0.26 | −0.18 | −0.01 | −0.23 | −0.02 | |
| Figure 1v | 0.10 | 0.11 | 0.13 | 0.13 | 0.20 | 0.19 | 0.30 | 0.29 | 2.44 | 2.53 | 3.04 | 3.13 | |
| Figure 1vi | 0.01 | 0.01 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | −1.99 | 2.13 | 2.40 | 2.55 | |
| Figure 1vii | 0.01 | 0.01 | 0.01 | −0.01 | −0.01 | −0.03 | 0.01 | −0.01 | 9.48 | 9.15 | 9.46 | 9.42 | |
| Figure 1viii | 0.02 | 0.01 | 0.01 | 0.01 | 0.02 | 0.03 | 0.04 | 0.04 | 1.47 | 1.45 | −4.02 | 4.10 | |
| Figure 1ix | 1.19 | 1.19 | 1.48 | 1.47 | 1.66 | 1.66 | 1.84 | 1.85 | 0.82 | 0.82 | 0.98 | 0.97 | |
| Figure 1x | −0.01 | −0.01 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 9.92 | 1.57 | −2.43 | −2.99 | |
| Figure 1xi | 1.14 | 1.16 | 1.40 | 1.41 | 1.57 | 1.62 | 1.73 | 1.81 | 0.79 | 0.78 | 0.95 | 0.94 | |
| Figure 1xii | −0.01 | −0.01 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 9.22 | 1.57 | −2.43 | −2.99 | |
| Figure 1xiii | −1.26 | −1.25 | −1.53 | −1.52 | −1.72 | −1.70 | −1.91 | −1.89 | −0.84 | −0.84 | −1.01 | −1.01 | |
| Figure 1xiv | 0.01 | 0.01 | 0.01 | 0.01 | 0.00 | 0.00 | 0.01 | 0.00 | 7.35 | 7.37 | −8.38 | 0.01 | |
| Figure 1xv | −1.23 | −1.21 | −1.51 | −1.48 | −1.70 | −1.67 | −1.91 | −1.85 | −0.83 | −0.83 | −0.99 | −0.98 | |
| Figure 1xvi | 0.01 | 0.01 | 0.01 | 0.01 | 0.00 | 0.00 | 0.01 | 0.00 | 7.35 | 7.37 | −8.38 | 0.01 | |
| NC | 771.15 | 929.91 | 1006.45 | 1079.44 | 378.74 | 581.00 | |||||||
| Total | 3.25 | 3.45 | 3.26 | 3.45 | 2.62 | 2.94 | 1.50 | 2.01 | 3.57 | 3.60 | 3.32 | 3.37 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Prasannaa, V.S.; Sahoo, B.K.; Abe, M.; Das, B.P. Significance of Non-Linear Terms in the Relativistic Coupled-Cluster Theory in the Determination of Molecular Properties. Symmetry 2020, 12, 811. https://doi.org/10.3390/sym12050811
Prasannaa VS, Sahoo BK, Abe M, Das BP. Significance of Non-Linear Terms in the Relativistic Coupled-Cluster Theory in the Determination of Molecular Properties. Symmetry. 2020; 12(5):811. https://doi.org/10.3390/sym12050811
Chicago/Turabian StylePrasannaa, V. Srinivasa, Bijaya K. Sahoo, Minori Abe, and Bhanu P. Das. 2020. "Significance of Non-Linear Terms in the Relativistic Coupled-Cluster Theory in the Determination of Molecular Properties" Symmetry 12, no. 5: 811. https://doi.org/10.3390/sym12050811
APA StylePrasannaa, V. S., Sahoo, B. K., Abe, M., & Das, B. P. (2020). Significance of Non-Linear Terms in the Relativistic Coupled-Cluster Theory in the Determination of Molecular Properties. Symmetry, 12(5), 811. https://doi.org/10.3390/sym12050811





