Functional Asymmetry and Fingerprint Features of Left-Handed and Right-Handed Young Yakuts (Mongoloid Race, North-Eastern Siberia)
Abstract
1. Introduction
2. Materials and Methods
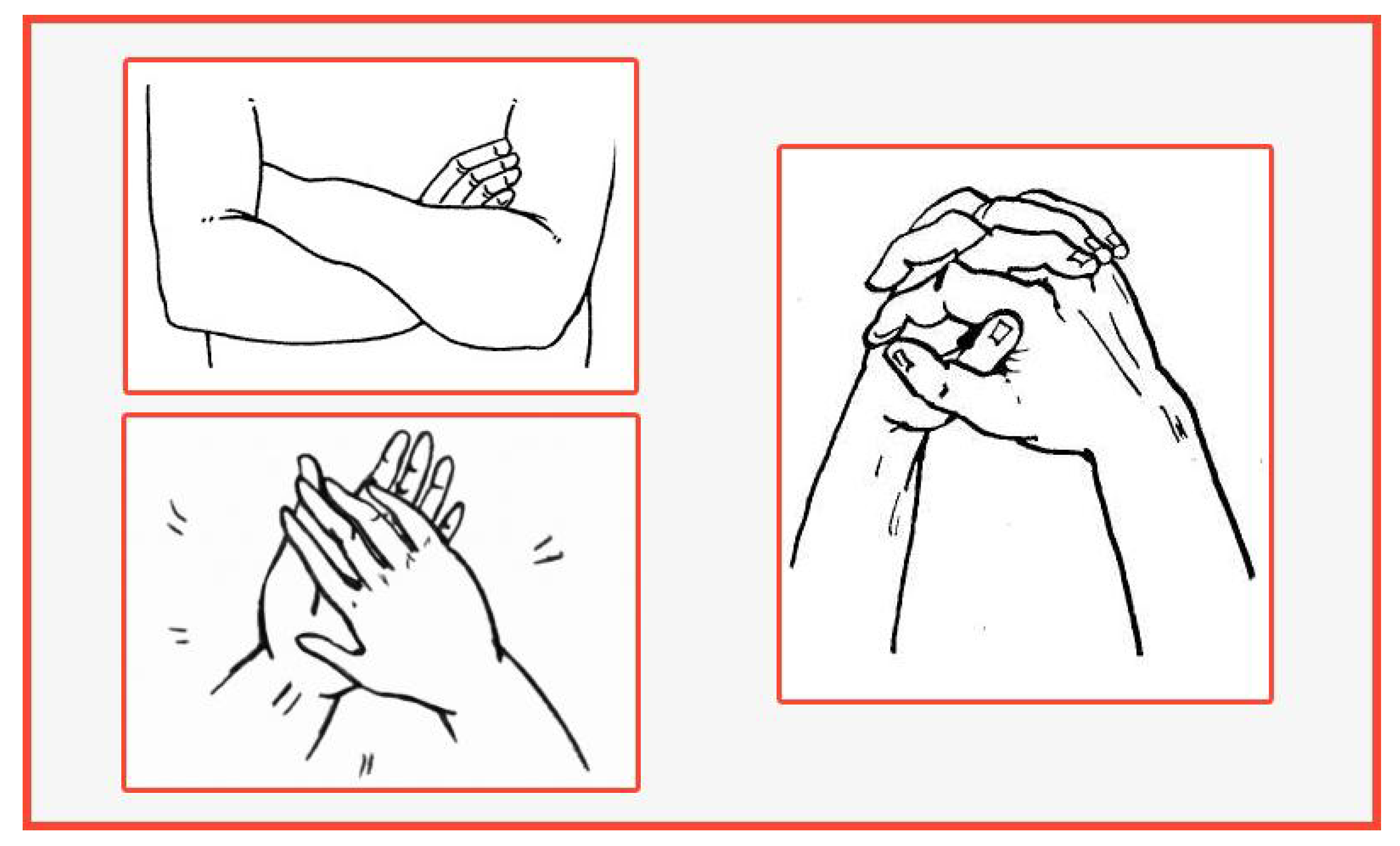
- Hand clasping;
- Arm folding;
- Applauding;
- Untying knots on a lace;
- Eye preference;
- Drawing of circles, squares, and triangles simultaneously with the right and left hands.
3. Results and Discussion
4. Fingerprint Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zakharov, V.M. Asimmetriya zhivotnykh (Asymmetry in Animals); Nauka: Moscow, Russia, 1987; p. 216. (In Russian) [Google Scholar]
- Van Valen, L. Study of fluctuating asymmetry. Evolution 1962, 16, 125–146. [Google Scholar] [CrossRef]
- Leary, R.F.; Allendorf, F.W. Fluctuating asymmetry as an indicator stress. Chance use in protect nature. Acta Zool. Fenica 1989, 4, 214–217. [Google Scholar]
- Parsons, P.A. Fluctuating asymmetry: An epigenetic measure of stress. Biol. Rev. 1990, 65, 131–145. [Google Scholar] [CrossRef] [PubMed]
- Parsons, P.A. Fluctuating asymmetry: A biological monitor of environmental and genomic stress. Heredity 1992, 68, 361–364. [Google Scholar] [CrossRef]
- Graham, J.H.; Özener, B. Fluctuating Asymmetry of Human Populations: A Review. Symmetry 2016, 8, 154. [Google Scholar] [CrossRef]
- Ravich-Shcherbo, I.V.; Maryutina, T.M.; Grigorenko, E.L. Psychogenetics; Aspect-Press: Moscow, Russia, 2000; p. 447. (In Russian) [Google Scholar]
- Geodakyan, V.A. Asynchronous asymmetry (sexual and lateral differentiation, the consequence of asynchronous evolution). ZHurn. Vysshej nervnoj deyatel’nosti im. I.P. Pavlova 1993, 43, 543–561. (In Russian) [Google Scholar]
- Annett, M. The Right-shift Theory of a Genetic Balanced Polymorphism for Cerebral Dominance and Cognitive Processing. Curr. Psychol. Cogn. 1995, 14, 427–480. [Google Scholar]
- Levy, J.; Nagylaki, T. A Model for the Genetics of Handedness. Genetics 1972, 72, 117–128. [Google Scholar]
- McManus, I.C.; Silk, G.; Cote, D.R.; Mellon, A.F.; Wong, J.; Kloss, J. The Development of Handedness in Children. Br. J. Dev. Psychol. 1988, 6, 257–273. [Google Scholar] [CrossRef]
- Michel, G.F.; Nelson, E.L.; Babik, I.; Campbell, J.; Marcinowski, E.C. Multiple Trajectories in the Developmental Psychobiology of Human Handedness. Advances in Child Development and Behavior. In Embodiment and Epigenesis: Theoretical and Methodological Issues in Understanding the Role of Biology within the Relational Developmental System. Part B: Ontogenetic Dimensions; Lerner, R.M., Benson, J.B., Eds.; Elsevier Inc.: Amsterdam, The Netherlands; Academic Press: Cambridge, MA, USA, 2013; Volume 45, pp. 227–260. [Google Scholar]
- McManus, I.C. The history and geography of human handedness. In Language Lateralization and Psychosis; Sommer, I.E.C., Kahn, R.S., Eds.; Cambridge University Press: Cambridge, UK, 2009; pp. 37–57. [Google Scholar]
- Raymond, M.; Pontier, D.; Dufour, A.-B.; Møller, A.P. Frequency-dependent maintenance of left handedness in humans. Proc. Roial Soc. B Biol. Sci. 1996, 263, 1627–1633. [Google Scholar] [CrossRef]
- Nikolaeva, E.I. Left-Handed Child: Diagnosis, Training, Correction; Detstvo-Press: St. Petersburg, Russia, 2005; p. 128. [Google Scholar]
- Laland, K.; Kumm, J.; Van Horn, J.; Feldman, M. A Gene—Culture Model of Human Handedness. Behav. Genet. 1995, 25, 433–445. [Google Scholar] [CrossRef] [PubMed]
- Ajrapetyanc, V.A. Left-Handed Children and Teenagers; Nauka: Moscow, Russia, 1987; p. 275. (In Russian) [Google Scholar]
- Lediaeva, E.V. The teaching of left-handed students as a psychological and pedagogical problem. Vestnik Mordovskogo Universiteta 2011, 2, 226–230. (In Russian) [Google Scholar]
- Denisova, Z.V. Mechanisms of Emotional Behavior of the Child; Nauka: Lenjngrad, Russia, 1978; p. 143. (In Russian) [Google Scholar]
- Dobrohotova, T.A.; Bragina, N.N. Lefthanders; Kniga: Moscow, Russia, 1994; p. 230. (In Russian) [Google Scholar]
- Sitnikova, M.A. The Psychological Aspects of Left-Handedness And Sinistrality Among Schoolchildren. Nauchnyi resultat. Ser. Pedagogika I Psichologia Obrazovania 2014, 2, 76–83. (In Russian) [Google Scholar] [CrossRef]
- Brandler, W.M.; Paracchini, S. The genetic relationship between handedness and neurodevelopmental disorders. Trends Mol. Med. 2014, 20, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Geschwind, N.; Behan, P. Left-handedness: Association with immune disease, migraine, and developmental learning disorder. Proc. Natl. Acad. Sci. USA 1982, 79, 97–100. [Google Scholar] [CrossRef]
- Llaurens, V.; Raymond, M.; Faurie, C. Why are some people left-handed? An evolutionary perspective. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2009, 364, 881–894. [Google Scholar] [CrossRef]
- Bryden, M.P. Handedness and cerebral organization. In Duality and Unity of the Brain; Ottoson, D., MacMillan, L., Eds.; Springer: Boston, MA, USA, 1987; pp. 55–70. [Google Scholar]
- Grouious, G.; Tsorbatzoudis, H.; Alexandris, K.; Barkoukis, V. Do left-handed competitors have an innate superiority in sports? Percept. Mot. Skills 2000, 90, 1273–1282. [Google Scholar] [CrossRef]
- Moskvina, N.V.; Moskvin, V.A. Left-handed in the sport of high achievements. Sportivnyjpsiholog 2010, 2, 25–29. (In Russian) [Google Scholar]
- Rhoads, J.G.; Damon, A. Some genetic traits in Solomon Island populations. II. Hand clasping, arm folding, and handedness. Am. J. Phys. Anthropol. 1973, 39, 179–183. [Google Scholar] [CrossRef]
- Kobyliansky, E.; Micle, S.; Arensburg, B. Handedness, hand-clasping arm-folding in males. Ann. Hum. Biol. 1978, 5, 247. [Google Scholar] [CrossRef]
- Downey, J.E. Further observations on the manner of clasping the hands. Am. Nat. 1926, 60, 387–390. [Google Scholar] [CrossRef]
- Kawabe, M. A study of the mode of clasping the hands. Trans. Sapporo Nat. Hist. Soc. 1949, 18, 49–52. [Google Scholar]
- Beckman, L.; Elston, R. Data on bilateral variation in man: Handedness, hand clasping and arm folding in Swedes. Hum. Biol. 1962, 34, 99–103. [Google Scholar] [PubMed]
- Polyuhov, A.M. “Hand-clasping” and “arm-folding”. Population, genetic and neurophysiological aspects. Russ. J. Genet. 1980, 7, 1294–1300. (In Russian) [Google Scholar]
- Tichy, J.; Belacek, J. Laterality in children: Cerebellar dominance, handedness, footedness and hair whorl. Act. Nerv. Super. Rediviva 2009, 51, 9–20. [Google Scholar]
- Hit, G.L.; Dolinova, N.A. Racial Classification of Humankind; Nauka: Moscow, Russia, 1990; p. 205. (In Russian) [Google Scholar]
- Gladkova, T.D. Phenomena of symmetry and asymmetry in humans in signs of dermatoglyphics. Voprosy Antropologii 1962, 10, 44–54. (In Russian) [Google Scholar]
- Guseva, I.S. Morphogenesis and Genetics of Human Ridge Skin; Belarus’: Minsk, Russia, 1986; p. 158. (In Russian) [Google Scholar]
- Bogdanov, N.N. Dermatoglyphics left handers. Voprosy Psikhologii 1997, 2, 76–87. (In Russian) [Google Scholar]
- Newmann, H.H. Dermatoglyphics and the problem of handedness. Am. J. Anat. 1934, 55, 277–322. [Google Scholar] [CrossRef]
- Nedz’ved’, G.K.; Usoev, S.S. Using of dermatoglyphics in the evaluation of latent left-handedness in organic diseases of the nervous system. In Rationalization and Invention in Medical Practice of Belarus’ SSR; Belarus’: Mink, Russia, 1972; pp. 82–83. (In Russian) [Google Scholar]
- Coren, S. Are fingerprints a genetic marker for handedness? Behav. Genet. 1994, 24, 141–148. [Google Scholar] [CrossRef]
- Kerkadze, L.A.; Pagava, K.I.; Gogberashvili, K.Y.A. Dermatoglyphic features of healthy children hands independing on the functional asymmetry. Izvestiya AN GSSR. Ser. Biol. 1989, 15, 154–158. (In Russian) [Google Scholar]
- Leche, S.M. Handedness and bimanual dermatoglyphic differences. Am. J. Anat. 1933, 53, 1–53. [Google Scholar] [CrossRef]
- Cromwell, H.; Rife, D.C. Dermatoglyphics in relation to functional handedness. Hum. Biol. 1942, 14, 516. [Google Scholar]
- Kimura, D.; Carson, M.W. Dermatoglyphic asymmetry: Relation to sex, handedness and cognitive pattern. Personal. Individ. Differ. 1995, 19, 471–478. [Google Scholar] [CrossRef]
- Sinha, C.K.; Meel, M.; Bayan, B. Using dermatoglyphics pattern to identify the left handed unique patterns and its biological significans-if any. World Appl. Sci. J. 2012, 20, 1107–1113. [Google Scholar] [CrossRef]
- Jantz, R.L. Population Variation in Asymmetry and Diversity from Finger to Finger for Digital Ridge-counts. Am. J. Phys. Anthrop. 1998, 42, 215–224. [Google Scholar] [CrossRef] [PubMed]
- Ten Broek, C.M.A.; Bots, J.; Bugiani, M.; Galis, F.; van Dongen, S. Developmental Origins of Limb Developmental Instability in Human Fetuses: Many Abnormalities Make the Difference. Symmetry 2017, 9, 51. [Google Scholar] [CrossRef]
- Van Dongen, S. Human Bodily Asymmetry Relates to Behavioral Lateralization and May not Reliably Reflect Developmental Instability. Symmetry 2018, 10, 117. [Google Scholar] [CrossRef]
- Naugler, C.T.; Ludman, M.D. A case-control study of fluctuating dermatoglyphic asymmetry as a risk marker for developmental delay. Am. J. Med. Genet. 1996, 66, 11–14. [Google Scholar] [CrossRef]
- King, S.; Mancini-Marie, A.; Brunet, A.; Walker, E.; Meaney, M.J.; Laplante, D.P. Prenatal maternal stress from a natural disaster predicts dermatoglyphic asymmetry in humans. Dev. Psychopathol. 2009, 21, 343–353. [Google Scholar] [CrossRef]
- Mellor, C.S. Dermatoglyphic Evidence of Fluctuating Asymmetry in Schizophrenia. Br. J. Psychiatry 1992, 160, 467–472. [Google Scholar] [CrossRef] [PubMed]
- Markow, T.A.; Wandler, K. Fluctuating Dermatoglyphic Asymmetry of Liability to Schizophrenia. Psychiatry Res. 1986, 19, 323–328. [Google Scholar] [CrossRef]
- Reilly, J.L.; Murphy, P.T.; Byrne, M.; Larkin, C.; Gill, M.; O’Callaghan, E.; Lane, A. Dermatoglyphic fluctuating asymmetry and atypical handedness in schizophrenia. Schizophr. Res. 2001, 50, 159–168. [Google Scholar] [CrossRef]
- Bruin, E.I.; Graham, J.H.; Louwerse, A.; Huizink, A.C. Mild Dermatoglyphic Deviations in Adolescents with Autism Spectrum Disorders and Average Intellectual Abilities as Compared to Typically Developing Boys. Autism Res. Treat. 2014. [Google Scholar] [CrossRef] [PubMed]
- Shakibaei, F.; Asadollahi, G.A.; Tabibi, A. Dermatoglyphics in patients with schizophrenia. J. Res. Med. Sci. 2011, 16, 1055–1061. [Google Scholar]
- Woolf, C.M.; Gianas, A. A study of fluctuating dermatoglyphic asymmetry in the sibs and parents of cleft lip propositi. Am. J. Hum. Genet. 1977, 28, 503–507. [Google Scholar]
- Scott, N.M.; Weinberg, S.M.; Neiswanger, K.; Brandon, C.A.; Daack-Hirsch, S.; Murray, J.C.; Liu, Y.-E.; Marazita, M.L. Dermatoglyphic fingerprint heterogeneity among individuals with nonsyndromic cleft lip with or without cleft palate and their unaffected relatives in China and the Philippines. Hum. Biol. 2005, 77, 257–266. [Google Scholar] [CrossRef]
- Floris, G.; Marini, E.; Mulliri, G. Dermatoglyphic asymmetry and diversity in some pathologies. Int. J. Anthropol. 2005, 20, 139. [Google Scholar] [CrossRef]
- Natekar, P.; DeSouza, F. Fluctuating asymmetry in dermatoglyphics of carcinoma of breast. Indian J. Hum. Genet. 2006, 12, 77–81. [Google Scholar] [CrossRef]
- Benderlioglu, Z. Fluctuating Asymmetry and Steroid Hormones: A Review. Symmetry 2010, 2, 541–553. [Google Scholar] [CrossRef]
- Wijerathne, B.T.B.; Meier, R.J.; Salgado, S.S.; Agampodi, S.B. Dermatoglyphics in kidney diseases: A review. SpringerPlus 2016, 5, 290. [Google Scholar] [CrossRef]
- Abilasha, S.; Harisudha, R.; Janaki, C.S. Dermatoglyphics: A Predictor Tool To Analyze The Occurrence Of Breast Cancer. Int. J. Med. Res. Health Sci. 2014, 3, 28–31. [Google Scholar] [CrossRef]
- Rosa, A.; Gutiérrez, B.; Guerra, A.; Arias, B.; Fañanás, L. Dermatoglyphics and abnormal palmar flexion creases as markers of early prenatal stress in children with idiopathic intellectual disability. J. Intell. Disabil. Res. 2001, 45, 416–423. [Google Scholar] [CrossRef]
- Bramon, E.; Walshe, M.; McDonald, C.; Martín, B.; Toulopoulou, T.; Wickham, H.; van Os, J.; Fearon, P.; Sham, P.C.; Fañanás, L.; et al. Dermatoglyphics and schizophrenia: A meta-analysis and investigation of the impact of obstetric complications upon a-b ridge count. Schizophr. Res. 2005, 75, 399–404. [Google Scholar] [CrossRef] [PubMed]
- Wijerathne, B.T.B.; Meier, R.J.; Agampodi, T.C.; Agampodi, S.B. Dermatoglyphics in hypertension: A review. J. Physiol. Anthropol. 2015, 34, 29. [Google Scholar] [CrossRef] [PubMed]
- Kaur, J.; Batra, A.P.S. Role of Dermatoglyphics In Medical Disorders. Indian J. Fundam. Appl. Life Sci. Online Int. J. 2013, 3, 536–539. [Google Scholar]
- Gogolev, A. Ethnic History of the Peoples of Yakutia; Yakutskijgosudarstvennyjuniversitet: Yakutsk, Russia, 2004; p. 104. (In Russian) [Google Scholar]
- Crubézy, E.; Amory, S.; Keyser, C.; Bouakaze, C.; Bodner, M.; Gibert, M.; Röck, A.; Parson, W.; Alexeev, A.; Ludes, B. Human evolution in Siberia: From frozen bodies to ancient DNA. BMC Evol. Biol. 2010, 10, 25. [Google Scholar] [CrossRef] [PubMed]
- Bezrukih, M.M. Left-Handed Child at School and at Home; Ventana-Graf: Moscow, Russia, 2005; p. 230. (In Russian) [Google Scholar]
- Zakharov, V.M.; Chubinishvili, A.T.; Baranov, A.S.; Valetskii, A.V.; Kryazheva, N.G.; Chistyakova, E.K.; Chubinishvili, A.T. Zdorov’e sredy: Metodika otsenki (Health of the Environment: Practical Evaluation). In Tsentrekolog; Politiki Rossii: Moscow, Russia, 2000; p. 68. (In Russian) [Google Scholar]
- Shadrina, E.; Vol’pert, Y. Fluctuating Asymmetry of Craniological Features of Small Mammals as a Reflection of Heterogeneity of Natural Populations. Symmetry 2016, 8, 142. [Google Scholar] [CrossRef]
- Dvirskij, A.E. The frequency of left-handedness and its anthropological characteristic. Vrachebnoedelo 1975, 9, 121–123. (In Russian) [Google Scholar]
- Remneva, E.V.; Morozova, D.S. Asimmetriya nekotoryh dermatoglificheskih priznakov russkogo naseleniya Omska. (Asymmetry of some dermatoglyphic features of the Russian population of Omsk). Vestn. Om. Unta 2013, 4, 173–177. (In Russian) [Google Scholar]
- Shadrina, E.G.; Tret’yakova, A.M. Application of dermatoglyphic parameters in intrapopulation groups investigations (on the example of the population of Yakutia). In IX All-Russia populational Colloquium “Individual and Population, Life Strategies”; Izdatel’skij dom: Ufa, Russia, 2006; Volume 1, pp. 528–533. (In Russian) [Google Scholar]
- Andreeva, A.A.; Shadrina, E.G. Phenetic characteristics of the population of Yakutia (by features of dermatoglyphic patterns). Int. J. Appl. Fundam. Res. 2012, 1, 78–79. (In Russian) [Google Scholar]
- Fedorova, S.A.; Barashkov, N.A.; Ushnitsky, V.V.; Kononova, S.K.; Bravina, R.I.; Alekseev, A.N.; Tomskiy, M.I.; Khusnutdinova, E.K. Ethnoterritorial groups of the Yakuts: The genetic structure features. Yakutskij Medicinskij Zhurnal 2014, 2, 28–31. [Google Scholar]
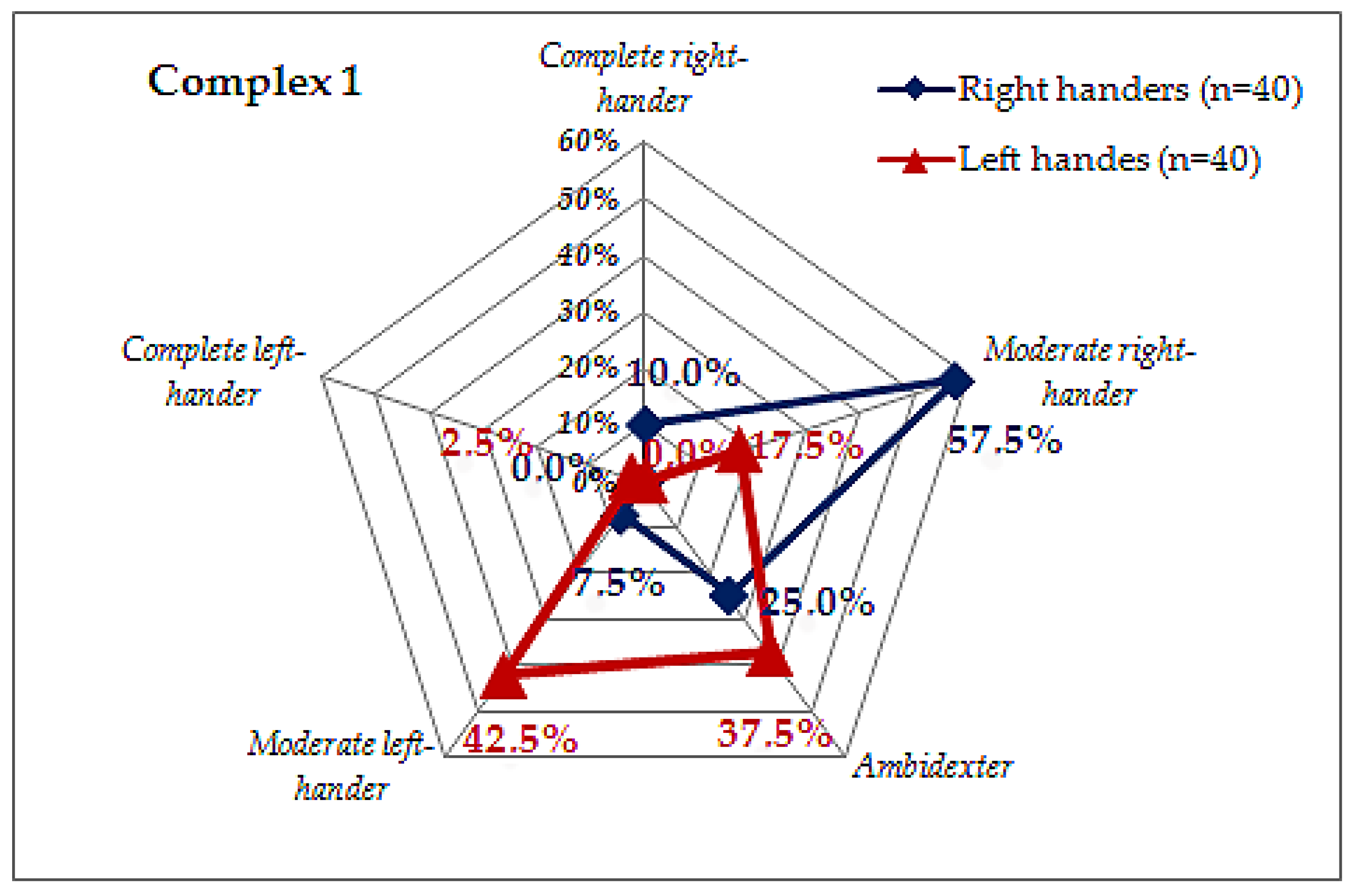
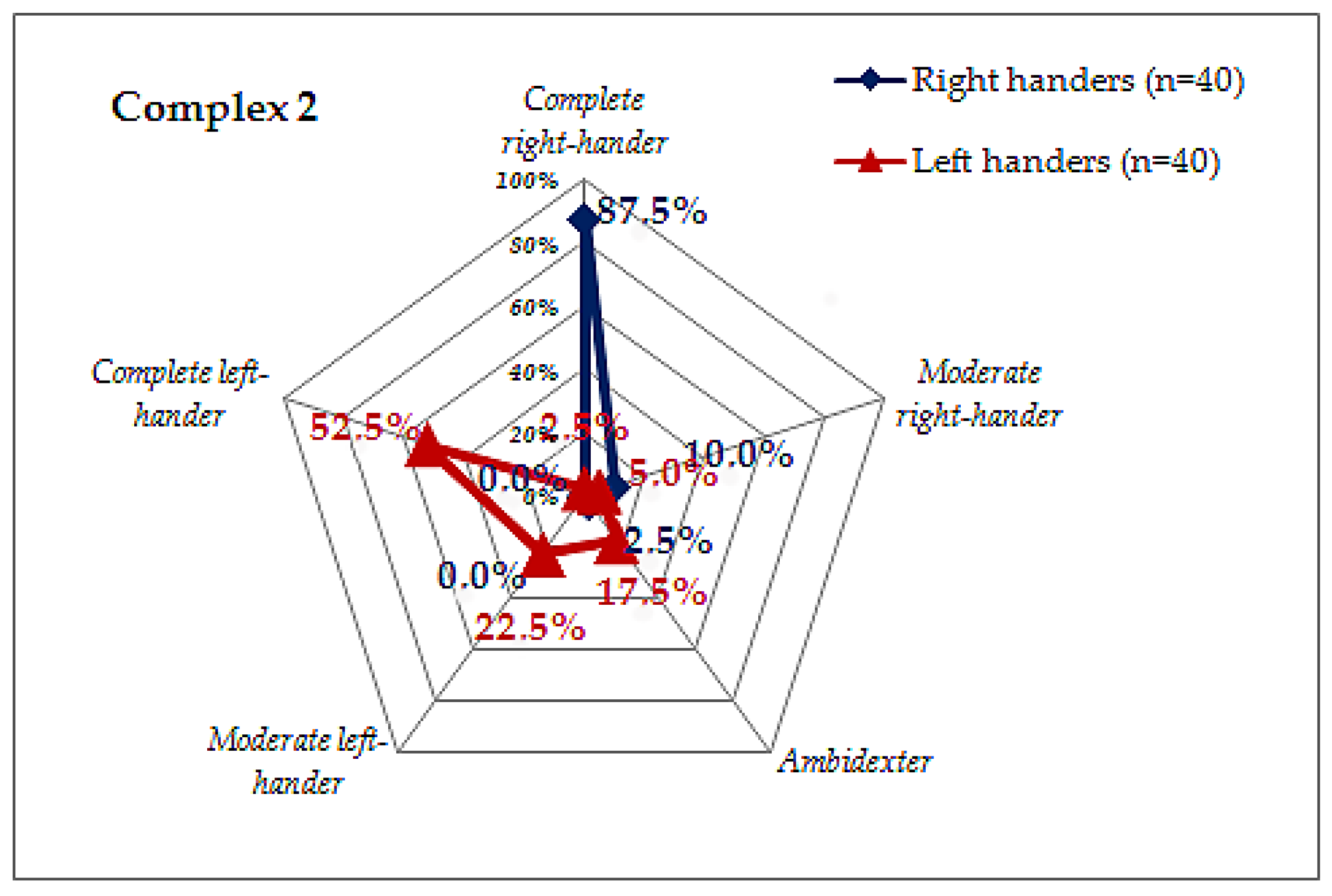
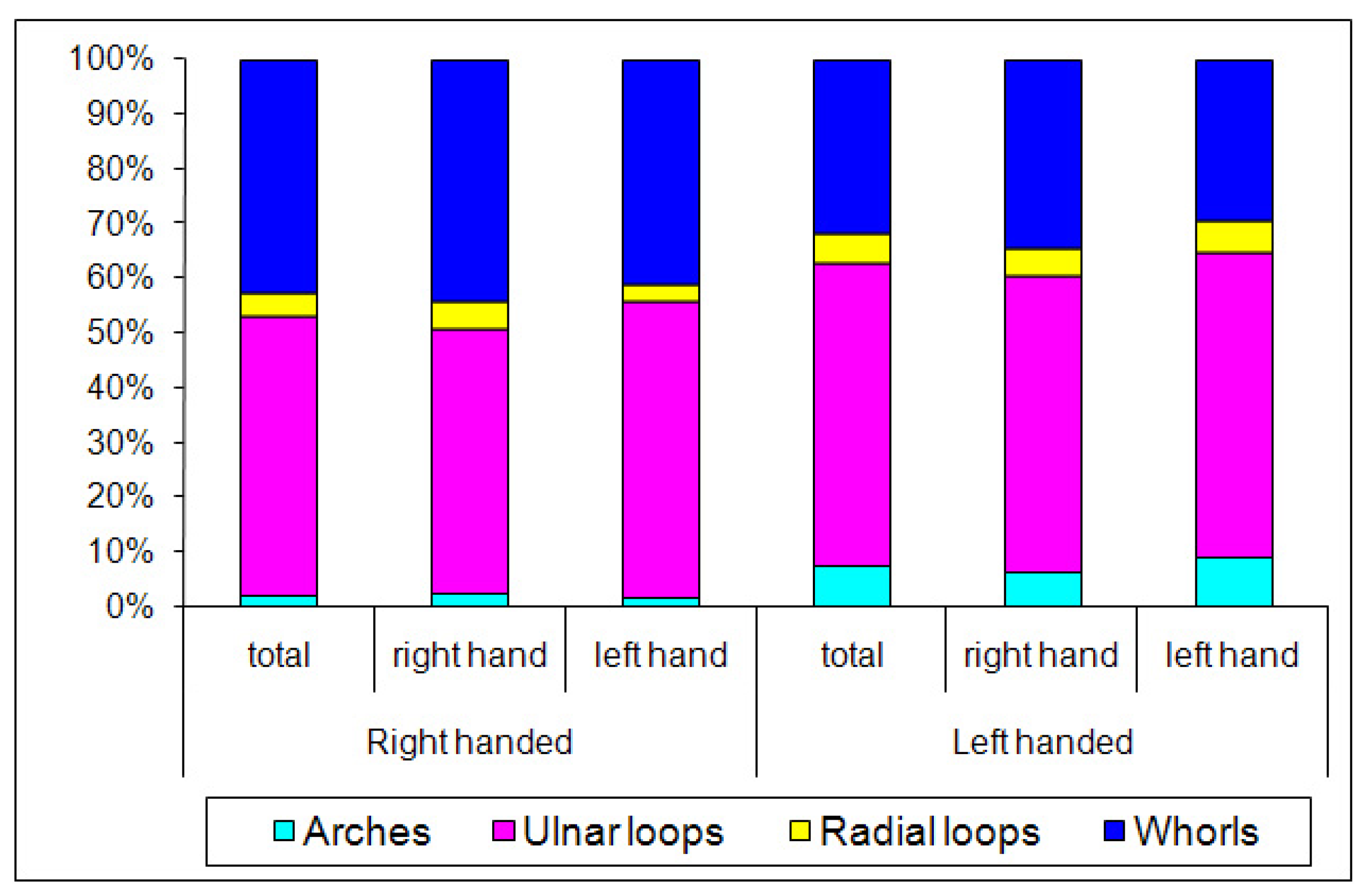
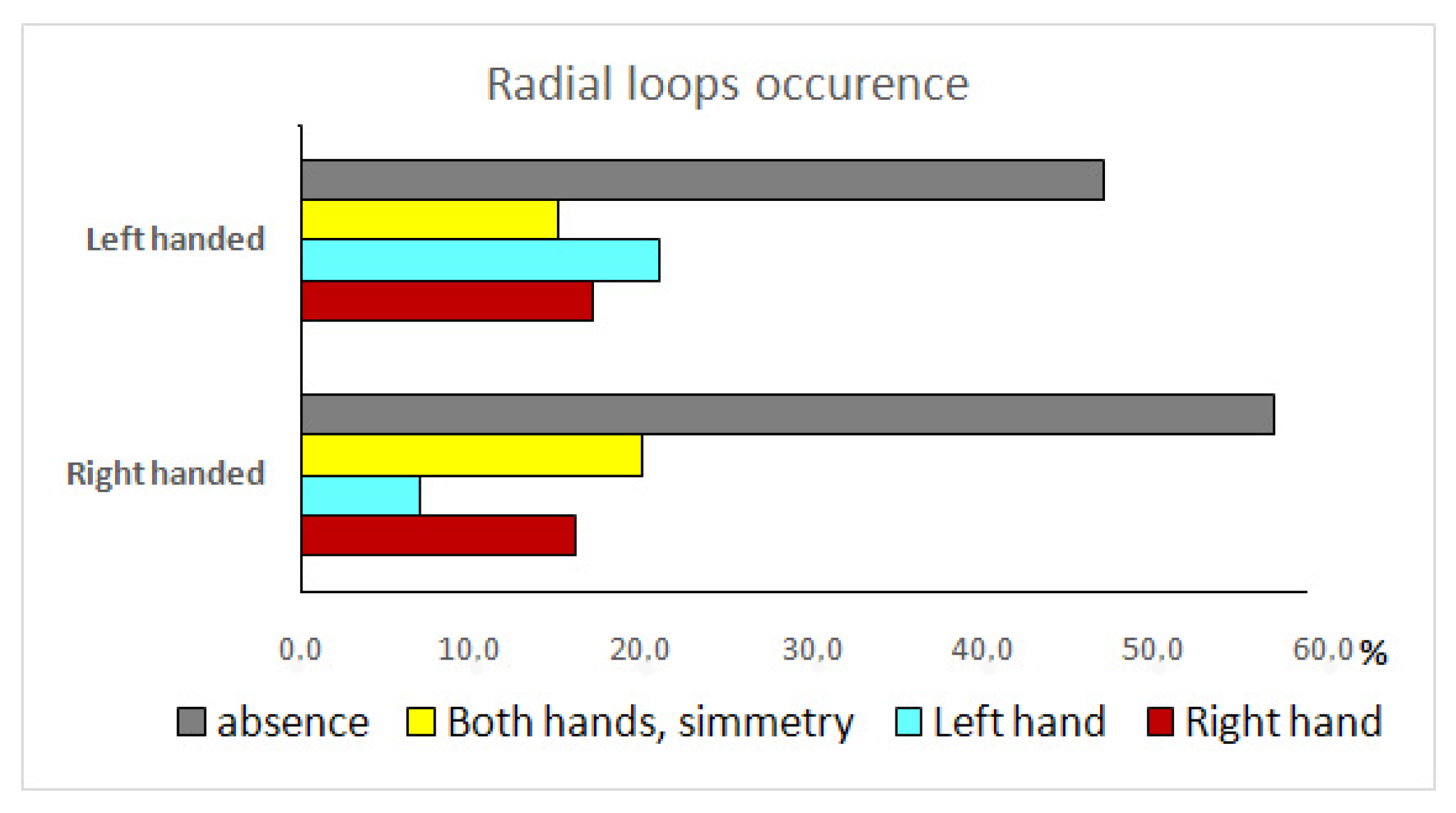
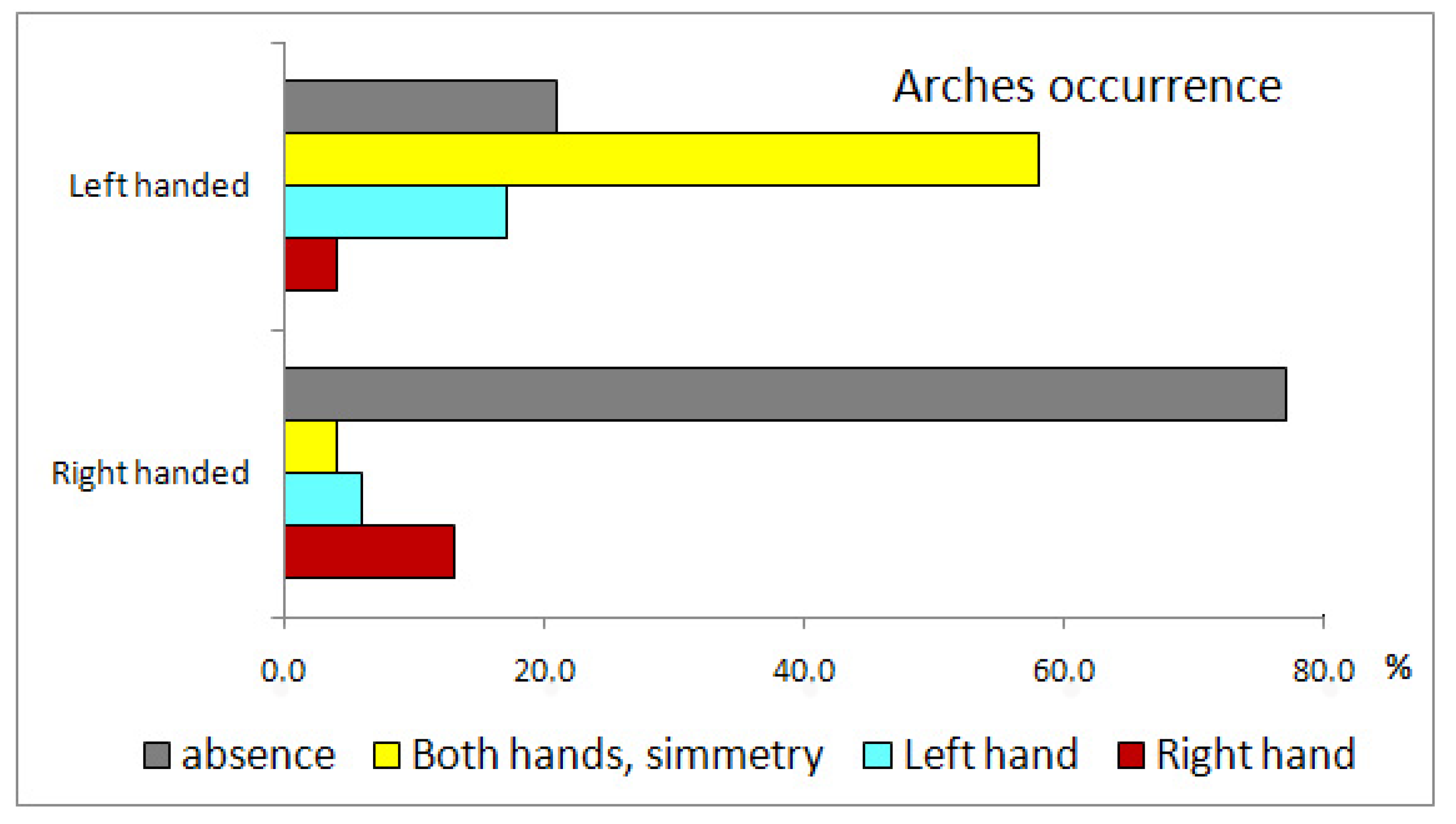
| Tests | Classification Principle | Result | Outcome |
|---|---|---|---|
| Complex 1 | Answer ratio. Tendency for right-handedness/left-handedness | 6/0 | Complete right-hander |
| 5/1 | Moderate right-hander | ||
| 4/2 | |||
| 3/3 | Ambidextrous | ||
| 2/4 | Moderate left-hander | ||
| 1/5 | |||
| 0/6 | Complete left-hander | ||
| Complex 2 | Score: 3 points—right-handed, 2 points—neutral, 1 point—left-handed | 33–36 | Complete right-hander |
| 27–32 | Moderate right-hander | ||
| 22–26 | Ambidextrous | ||
| 16–21 | Moderate left-hander | ||
| 12–15 | Complete left-hander |
| Tests | Sex | Sum Score | Average Score | ||
|---|---|---|---|---|---|
| Right-Handers | Left-Handers | Right-Handers | Left-Handers | ||
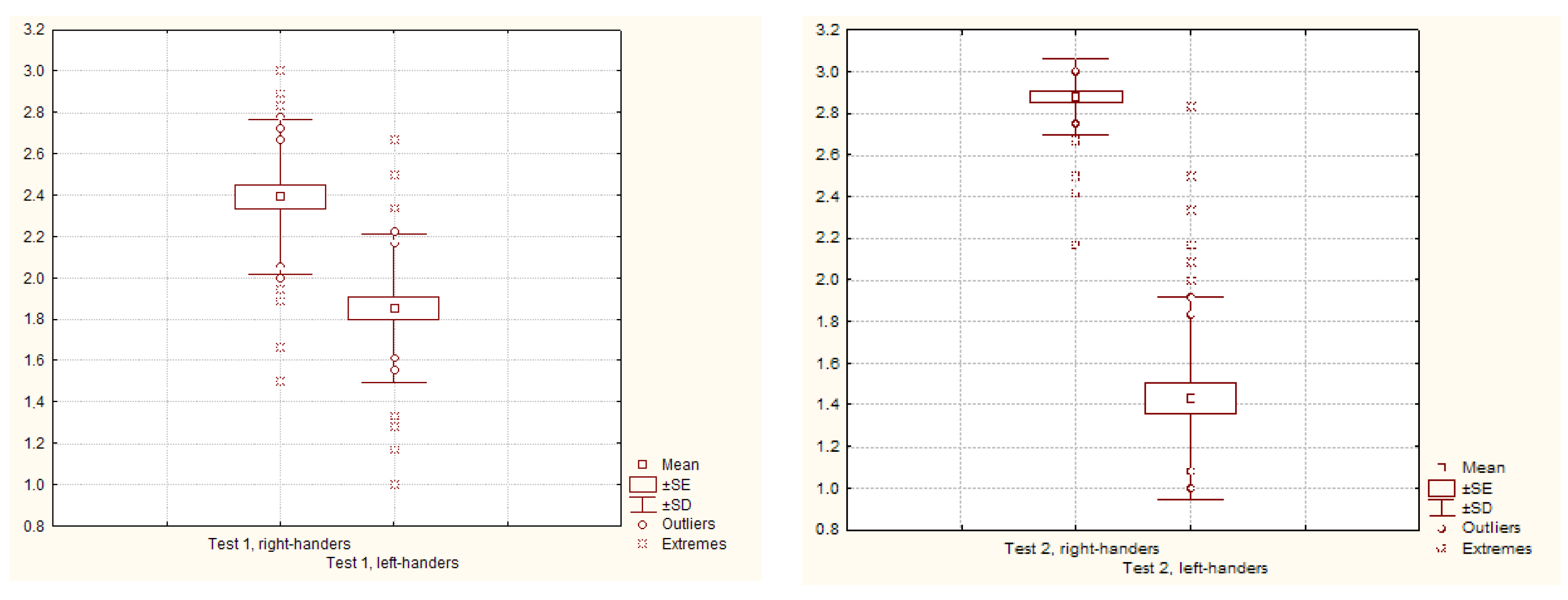
| Complex 1 | Males | 14.30 ± 0.53 | 11.33 ± 0.46 | 2.4 ± 0.09 | 1.9 ± 0.08 |
| Females | 14.42 ± 0.48 | 10.90 ± 0.51 | 2.4 ± 0.08 | 1.8 ± 0.08 | |
| Average | 14.36 ± 0.35 | 11.12 ± 0.34 | 2.4 ± 0.06 | 1.9 ± 0.06 | |
| Limit | 9.00–18.00 | 6.00–16.00 | 1.5–3.0 | 1.0–2.7 | |
| Mod value | 16.0 | 10.7 | 2.7 | 1.8 | |
| Difference between right-handers and left-handers, Student’s t-test | 6.61 p < 0.0001 | ||||
| Complex 2 | Males | 34.00 ± 0.58 | 17.60 ± 1.53 | 2.8 ± 0.05 | 1.5 ± 0.13 |
| Females | 35.10 ± 0.35 | 16.75 ± 1.09 | 2.9 ± 0.03 | 1.4 ± 0.09 | |
| Average | 34.55 ± 0.35 | 17.18 ± 0.93 | 2.9 ± 0.03 | 1.4 ± 0.08 | |
| Limit | 26.00–36.00 | 12.00–34.00 | 2.2–3.0 | 1.0–2.8 | |
| Mod value | 36.0 | 12.0 | 3.0 | 1.0 | |
| Difference between right-handers and left-handers, Student’s t-test | 17.50 p < 0.0001 | ||||
| Test | Sex | Handedness | Student’s t-test | |
|---|---|---|---|---|
| Right-Handers | Left-Handers | |||
| Complex 1 TKas1 | Males | 0.38 ± 0.10 | −0.09 ± 0.07 | 3.87 p < 0.0004 |
| Females | 0.43 ± 0.08 | −0.19 ± 0.09 | 5.24 p < 0.0001 | |
| Average | 0.40 ± 0.06 | −0.14 ± 0.06 | 6.48 p < 0.0001 | |
| Complex 2 TKas2 | Males | 0.81 ± 0.05 | −0.53 ± 0.13 | 9.81 p < 0.0001 |
| Females | 0.95 ± 0.02 | −0.53 ± 0.10 | 13.91 p < 0.0001 | |
| Average | 0.88 ± 0.03 | −0.53 ± 0.08 | 16.35 p < 0.0001 | |
| Sex | Right Hand M ± m | Left Hand M ± m | Limit | Coefficient of Variation | EKas |
|---|---|---|---|---|---|
| Right-handed | |||||
| Males (n = 30) | 44.63 ± 1.35 | 40.95 ± 1.40 | 29.37–56.10 | 10.18 ± 1.47 | 0.045 ± 0.010 |
| Females (n = 30) | 26.22 ± 0.56 | 24.53 ± 0.73 | 17.20–33.80 | 8.23 ± 1.21 | 0.036 ± 0.008 |
| Sexual difference EKas, Student’s t-test | 1.25, not significant | ||||
| Left-handed | |||||
| Males (n = 20) | 39.68 ± 1.84 | 41.63 ± 1.71 | 24.40–55.07 | 9.94 ± 1.16 | −0.012 ± 0.012 |
| Females (n = 28) | 24.63 ± 0.92 | 25.17 ± 0.90 | 14.83–37.27 | 10.37 ± 1.17 | −0.026 ± 0.012 |
| Sexual difference EKas, Student’s t-test | 3.17, p < 0.01 | ||||
| Patterns | Right Hand | Left Hand | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1st | 2nd | 3rd | 4th | 5th | 1st | 2nd | 3rd | 4th | 5th | |
| Right-handed males | ||||||||||
| Arches | 0.0 | 10.0 | 2.0 | 0.0 | 0.0 | 2.0 | 4.0 | 0.0 | 0.0 | 0.0 |
| Ulnar loops | 42.0 | 26.0 | 50.0 | 32.0 | 78.0 | 40.0 | 36.0 | 66.0 | 48.0 | 72.0 |
| Radial loops | 0.0 | 24.0 | 6.0 | 0.0 | 2.0 | 0.0 | 14.0 | 2.0 | 0.0 | 2.0 |
| Whorls | 58.0 | 40.0 | 42.0 | 68.0 | 20.0 | 58.0 | 46.0 | 32.0 | 52.0 | 26.0 |
| Right-handed females | ||||||||||
| Arches | 2.0 | 4.0 | 4.0 | 4.0 | 2.0 | 2.0 | 6.0 | 6.0 | 0.0 | 0.0 |
| Ulnar loops | 50.0 | 30.0 | 50.0 | 40.0 | 82.0 | 54.0 | 30.0 | 52.0 | 54.0 | 86.0 |
| Radial loops | 0.0 | 20.0 | 0.0 | 0.0 | 0.0 | 0.0 | 14.0 | 2.0 | 0.0 | 0.0 |
| Whorls | 48.0 | 46.0 | 46.0 | 56.0 | 16.0 | 44.0 | 50.0 | 40.0 | 46.0 | 14.0 |
| Left-handed males | ||||||||||
| Arches | 10.0 | 20.0 | 5.0 | 0.0 | 0.0 | 10.0 | 20.0 | 10.0 | 0.0 | 0.0 |
| Ulnar loops | 40.0 | 15.0 | 65.0 | 60.0 | 70.0 | 50.0 | 20.0 | 75.0 | 65.0 | 80.0 |
| Radial loops | 0.0 | 30.0 | 5.0 | 5.0 | 5.0 | 0.0 | 15.0 | 0.0 | 0.0 | 5.0 |
| Whorls | 50.0 | 35.0 | 25.0 | 35.0 | 25.0 | 40.0 | 45.0 | 15.0 | 35.0 | 15.0 |
| Left-handed females | ||||||||||
| Arches | 3.1 | 12.5 | 9.4 | 3.1 | 3.1 | 6.3 | 15.6 | 15.6 | 9.4 | 3.1 |
| Ulnar loops | 50.0 | 31.3 | 62.5 | 56.3 | 84.4 | 40.6 | 25.0 | 59.4 | 62.5 | 84.4 |
| Radial loops | 0.0 | 12.5 | 0.0 | 0.0 | 0.0 | 0.0 | 15.6 | 9.4 | 3.1 | 6.3 |
| Whorls | 46.9 | 43.8 | 28.1 | 40.6 | 12.5 | 53.1 | 43.8 | 15.6 | 25.0 | 6.3 |
| Patterns | Hand | Reduction in Occurrence in Different Finger-Patterns | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
 | ||||||||||
| Right-handers | ||||||||||
| Arches | Right | II | > | III | ≥ | IV | ≥ | I | ≥ | V |
| Left | II | > | III | ≥ | I | > | IV | V | ||
| Radial loops | Right | II | > | III | > | V | > | I | IV | |
| Left | II | > | III | > | V | > | I | IV | ||
| Ulnar loops | Right | V | > | III | ≥ | I | > | IV | > | II |
| Left | V | > | III | ≥ | IV | ≥ | I | > | II | |
| Whorls | Right | IV | ≥ | I | > | III | ≥ | II | > | V |
| Left | I | ≥ | IV | ≥ | II | > | III | > | V | |
| Left-handers | ||||||||||
| Arches | Right | II | > | III | ≥ | I | > | IV | ≈ | V |
| Left | II | > | III | > | I | > | IV | V | ||
| Radial loops | Right | II | > | III | ≈ | IV | ≈ | V | > | I |
| Left | II | > | III | ≈ | V | > | IV | > | I | |
| Ulnar loops | Right | V | > | III | ≥ | IV | > | I | > | II |
| Left | V | > | III | ≥ | IV | > | I | > | II | |
| Whorls | Right | I | > | II | > | IV | > | III | > | V |
| Left | I | > | II | > | IV | > | III | > | V | |
| Table of Symbols: | |||
| Fingers: | Occurrence | ||
| I | Thumb | > | Higher |
| II | Index | ≥ | Higher or equal |
| III | Middle | ≈ | Approximately equal |
| IV | Ring | The pattern was not found | |
| V | Little | ||
| Sex, Handedness | Average Number of Loops per Individual | Individuals, % Occurrence | ||
| Right Hand | Left Hand | Total | ||
| Loops, total * | ||||
| Right-handed males (n = 50) | 2.52 ± 0.21 | 2.80 ± 0.24 | 5.32 ± 0.40 | 90.00 |
| Right-handed females (n = 50) | 2.68 ± 0.25 | 2.92 ± 0.24 | 5.60 ± 0.46 | 94.00 |
| Right-handed total (n = 100) ** | 2.60 ± 0.16 | 2.86 ± 0.17 | 5.46 ± 0.30 | 92.00 |
| Left-handed males (n = 20) | 2.95 ± 0.32 | 3.10 ± 0.32 | 6.05 ± 0.60 | 90.00 |
| Left-handed females (n = 32) | 2.97 ± 0.23 | 3.06 ± 0.25 | 6.03 ± 0.43 | 96.88 |
| Left-handed total (n = 52) ** | 2.96 ± 0.19 | 3.08 ± 0.19 | 6.04 ± 0.35 | 94.23 |
| Ulnar loops * | ||||
| Right-handed males (n = 50) | 2.20 ± 0.22 | 2.62 ± 0.22 | 4.82 ± 0.38 | 90.00 |
| Right-handed females (n = 50) | 2.48 ± 0.24 | 2.76 ± 0.23 | 5.24 ± 0.44 | 94.00 |
| Right-handed total (n = 100) ** | 2.34 ± 0.16 | 2.69 ± 0.16 | 5.03 ± 0.29 | 92.00 |
| Left-handed males (n = 20) | 2.50 ± 0.33 | 2.90 ± 0.30 | 5.40 ± 0.59 | 90.00 |
| Left-handed females (n = 32) | 2.84 ± 0.22 | 2.72 ± 0.24 | 5.56 ± 0.39 | 96.88 |
| Left-handed total (n = 52) ** | 2.71 ± 0.18 | 2.79 ± 0.19 | 5.50 ± 0.33 | 94.23 |
| Radial loops | ||||
| Sex, Handedness | Average Number of Loops | Occurrence on the Fingers 3–5 | ||
| Right Hand * | Left Hand * | Total * | ||
| Right-handed males (n = 50) | 0.32 ± 0.08 | 0.18 ± 0.06 | 0.50 ± 0.11 | 0.06 ± 0.04 |
| Right-handed females (n = 50) | 0.20 ± 0.06 | 0.16 ± 0.05 | 0.36 ± 0.09 | 0.02 ± 0.02 |
| Right-handed total (n = 100) ** | 0.26 ± 0.05 | 0.17 ± 0.04 | 0.43 ± 0.07 | 0.04 ± 0.02 |
| Left-handed males (n = 20) | 0.45 ± 0.17 | 0.20 ± 0.09 | 0.65 ± 0.18 | 0.20 ± 0.11 |
| Left-handed females (n = 32) | 0.13 ± 0.06 | 0.34 ± 0.15 | 0.47 ± 0.17 | 0.19 ± 0.10 |
| Left-handed total (n = 52) ** | 0.25 ± 0.08 | 0.29 ± 0.10 | 0.54 ± 0.13 | 0.19 ± 0.06 |
| Difference between right- and left-handers, Student’s t-test | Not significant | Not significant | Not significant | 3.32, p < 0.001 |
| Handedness, Sex | Radial Loops FPas | Arches FPAs | ||
|---|---|---|---|---|
| n | M ± m | n | M ± m | |
| Right-handed males | 10 | +0.53 ± 0.19 | 5 | +0.50 ± 0.27 |
| Right-handed females | 8 | +0.15 ± 0.22 | 7 | +0.38 ± 0.53 |
| Right-handers, total | 18 | +0.37 ± 0.15 | 12 | +0.44 ± 0.29 |
| Left-handed males | 17 | +0.30 ± 0.30 | 8 | –0.20 ± 0.37 |
| Left-handed females | 13 | –0.88 ± 0.52 | 8 | –0.86 ± 0.46 |
| Left-handers, total | 30 | –0.22 ± 0.31 | 16 | –0.58 ± 0.31 |
| Differences between left-handers and right-handers, Student’s t-test | 2.21 p < 0.05 | 2.40 p < 0.05 | ||
| Sex, Handedness * | Average Number of Whorls Per Individual | Individuals, % | |||
|---|---|---|---|---|---|
| Right Hand | Left Hand | Total | Occurrence | On 10 Fingers | |
| Right-handed men | 2.32 ± 0.23 | 2.14 ± 0.24 | 4.46 ± 0.42 | 92.0 | 10.0 |
| Right-handed women | 2.14 ± 0.26 | 1.96 ± 0.24 | 4.10 ± 0.49 | 72.00 | 6.00 |
| Right-handed total | 2.23 ± 0.18 | 2.05 ± 0.17 | 4.28 ± 0.32 | 82.00 | 8.00 |
| Left-handed men | 1.70 ± 0.36 | 1.50 ± 0.36 | 3.20 ± 0.70 | 75.00 | 10.00 |
| Left-handed women | 1.72 ± 0.25 | 1.44 ± 0.27 | 3.16 ± 0.48 | 84.38 | 3.13 |
| Left-handed total | 1.71 ± 0.21 | 1.46 ± 0.21 | 3.17 ± 0.40 | 80.77 | 5.77 |
| Differences between left-handers and right-handers, Student’s t-test | Not significant | 2.17 p < 0.05 | 2.17 p < 0.05 | Not significant | 12.33 p < 0.001 |
| Sex, Handedness * | Average Number of Arches Per Individual | On Both Hands | 3–4 Arches on 1 Hand | ||
|---|---|---|---|---|---|
| Right Hand | Left Hand | Total | |||
| Right-handed men | 0.14 ± 0.05 | 0.06 ± 0.03 | 0.20 ± 0.07 | 0.04 ± 0.03 | 0 |
| Right-handed women | 0.16 ± 0.08 | 0.10 ± 0.05 | 0.26 ± 0.10 | 0.02 ± 0.02 | 0 |
| Right-handed total ** | 0.15 ± 0.05 | 0.08 ± 0.03 | 0.23 ± 0.06 | 0.03 ± 0.02 | 0 |
| Left-handed men | 0.35 ± 0.18 | 0.40 ± 0.18 | 0.75 ± 0.35 | 0.15 ± 0.08 | 0.05 ± 0.05 |
| Left-handed women | 0.31 ± 0.15 | 0.50 ± 0.19 | 0.81 ± 0.33 | 0.19 ± 0.07 | 0.13 ± 0.06 |
| Left-handed total ** | 0.33 ± 0.12 | 0.46 ± 0.14 | 0.79 ± 0.24 | 0.17 ± 0.05 | 0.10 ± 0.04 |
| Differences between left-handers and right-handers, Student’s t-test | Not significant | 2.74 p < 0.01 | 2.25 p < 0.05 | 2.57 p < 0.05 | 2.33 p < 0.05 |
| Handedness, Sex | Right Hand | Left Hand | Summary DI |
|---|---|---|---|
| Male right-handers (n = 50) | 1.43 ± 0.05 | 1.42 ± 0.05 | 1.42 ± 0.05 |
| Female right-handers (n = 50) | 1.39 ± 0.06 | 1.36 ± 0.05 | 1.38 ± 0.05 |
| Right-handers, total (n = 100) | 1.41 ± 0.04 | 1.39 ± 0.04 | 1.40 ± 0.04 |
| Male left-handers (n = 20) | 1.27 ± 0.10 | 1.22 ± 0.10 | 1.25 ± 0.09 |
| Female left-handers (n = 32) | 1.28 ± 0.07 | 1.19 ± 0.08 | 1.23 ± 0.07 |
| Left-handers, total (n = 52) | 1.28 ± 0.06 | 1.20 ± 0.06 | 1.24 ± 0.06 |
| Differences between left-handers and right-handers, Student’s t-test | 1.80, not significant | 2.63, p < 0.01 | 2.45, p < 0.05 |
| Hand, Finger | Males | Females | Total |
|---|---|---|---|
| M ± m | M ± m | M ± m | |
| Right-handers | |||
| Thumb | 0.18 ± 0.05 | 0.12 ± 0.05 | 0.15 ± 0.04 |
| Index | 0.48 ± 0.07 | 0.38 ± 0.07 | 0.43 ± 0.05 |
| Middle | 0.32 ± 0.07 | 0.22 ± 0.06 | 0.27 ± 0.04 |
| Ring | 0.36 ± 0.07 | 0.30 ± 0.07 | 0.33 ± 0.05 |
| Little | 0.24 ± 0.06 | 0.10 ± 0.04 | 0.17 ± 0.04 |
| OFAM | 0.32 ± 0.03 | 0.22 ± 0.03 | 0.27 ± 0.02 |
| Left-handers | |||
| Thumb | 0.15 ± 0.08 | 0.25 ± 0.08 | 0.21 ± 0.06 |
| Index | 0.50 ± 0.11 | 0.34 ± 0.09 | 0.40 ± 0.07 |
| Middle | 0.20 ± 0.09 | 0.31 ± 0.08 | 0.27 ± 0.06 |
| Ring | 0.15 ± 0.08 | 0.31 ± 0.08 | 0.25 ± 0.06 |
| Little | 0.15 ± 0.08 | 0.13 ± 0.06 | 0.13 ± 0.05 |
| OFAM | 0.23 ± 0.04 | 0.27 ± 0.05 | 0.25 ± 0.03 |
| Group | Ridge Count | FA | ||
|---|---|---|---|---|
| Right Hand | Left Hand | Total | ||
| Females | 71.15 ± 9.75 | 73.46 ± 3.71 | 144.60 ± 18.07 | 0.14 ± 0.04 |
| Males | 88.75 ± 8.97 | 74.44 ± 7.13 | 163.19 ± 16.14 | 0.10 ± 0.02 |
| Right-handers, total | 78.19 ± 6.92 | 73.85 ± 6.19 | 152.04 ± 12.56 | 0.13 ± 0.02 |
| Females | 71.55 ± 6.78 | 75.89 ± 7.14 | 147.44 ± 13.54 | 0.12 ± 0.03 |
| Males | 86.25 ± 10.52 | 86.00 ± 9.20 | 160.14 ± 16.41 | 0.07 ± 0.01 |
| Left-handers, total | 74.27 ± 5.63 | 77.12 ± 5.68 | 151.12 ± 11.02 | 0.10 ± 0.02 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shadrina, E.; Vol’pert, Y. Functional Asymmetry and Fingerprint Features of Left-Handed and Right-Handed Young Yakuts (Mongoloid Race, North-Eastern Siberia). Symmetry 2018, 10, 728. https://doi.org/10.3390/sym10120728
Shadrina E, Vol’pert Y. Functional Asymmetry and Fingerprint Features of Left-Handed and Right-Handed Young Yakuts (Mongoloid Race, North-Eastern Siberia). Symmetry. 2018; 10(12):728. https://doi.org/10.3390/sym10120728
Chicago/Turabian StyleShadrina, Elena, and Yakov Vol’pert. 2018. "Functional Asymmetry and Fingerprint Features of Left-Handed and Right-Handed Young Yakuts (Mongoloid Race, North-Eastern Siberia)" Symmetry 10, no. 12: 728. https://doi.org/10.3390/sym10120728
APA StyleShadrina, E., & Vol’pert, Y. (2018). Functional Asymmetry and Fingerprint Features of Left-Handed and Right-Handed Young Yakuts (Mongoloid Race, North-Eastern Siberia). Symmetry, 10(12), 728. https://doi.org/10.3390/sym10120728






