Evaluation of Nitrogen and Carbon Stable Isotopes in Filter Feeding Bivalves and Surficial Sediment for Assessing Aquatic Condition in Lakes and Estuaries
Abstract
:1. Introduction
2. Methods
3. Results
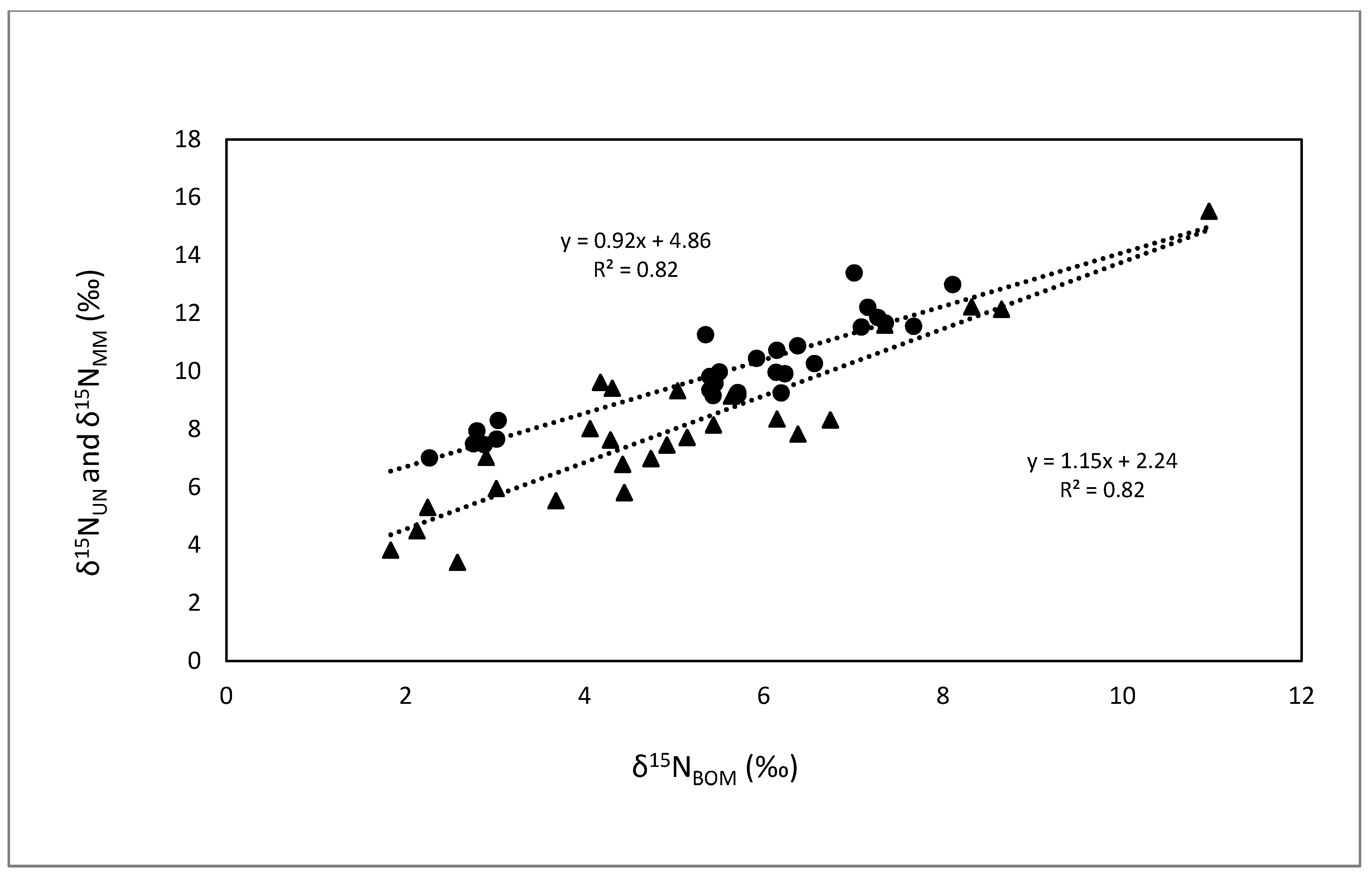
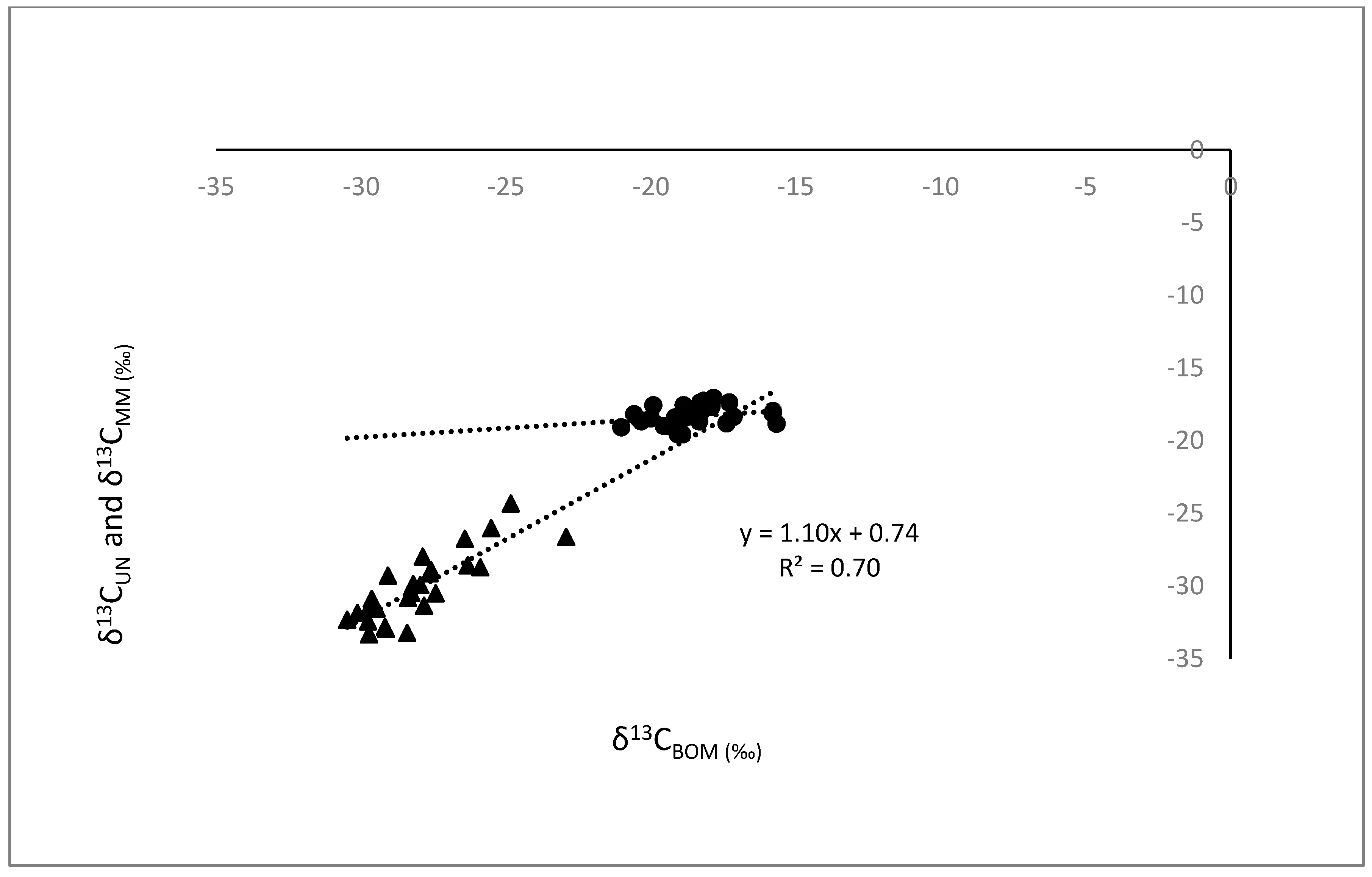
3.1. Comparisons of Bivalves with BOM
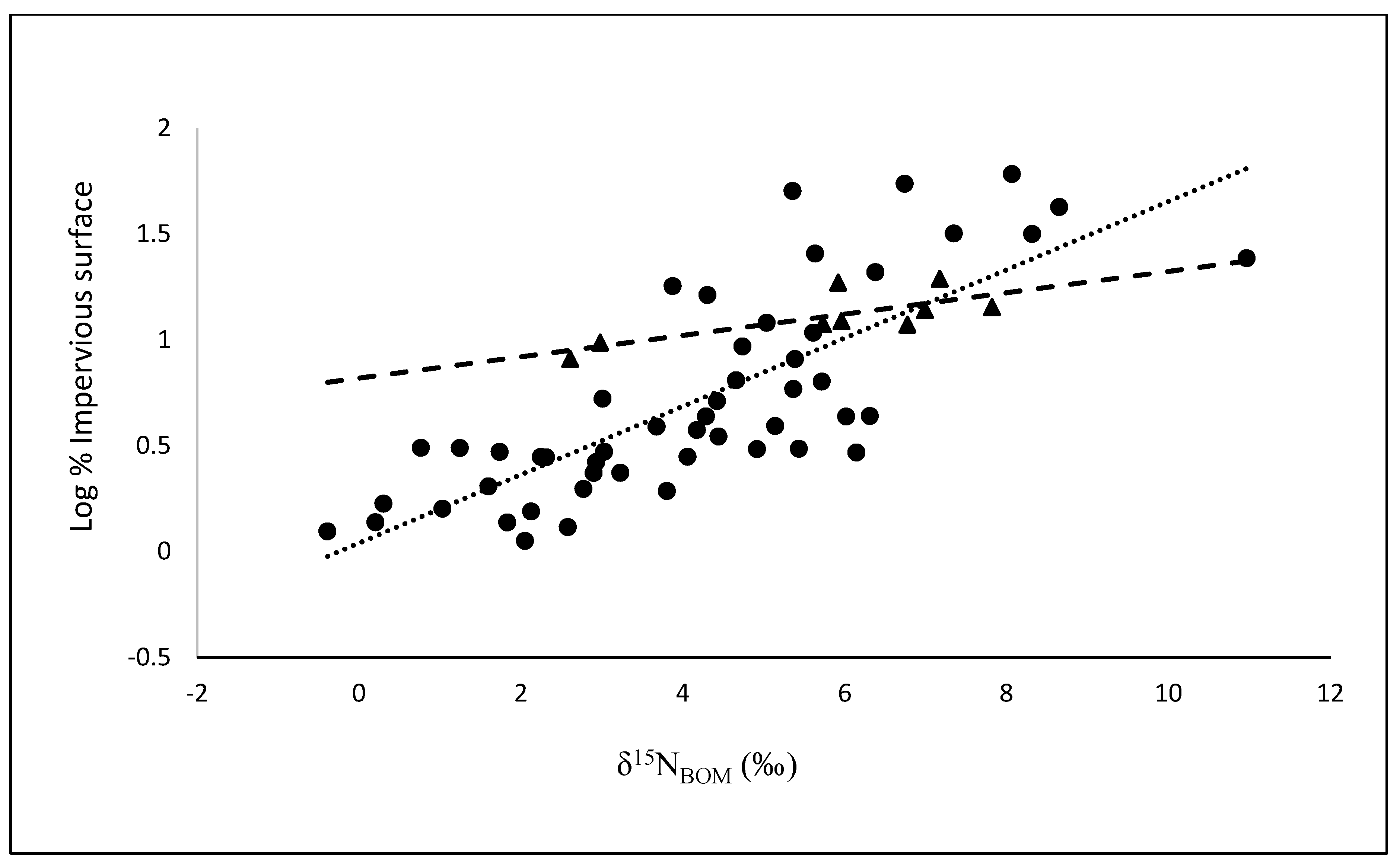
3.2. Comparisons of BOM with Land Cover and Population
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- United States Environmental Protection Agency. National Lakes Assessment 2012: A Collaborative Survey of Lakes in the United States; Technical Report EPA 841-R-16-113; Office of Water, Office of Research and Development: Washington, DC, USA, 2016.
- United States Environmental Protection Agency. National Rivers and Streams Assessment 2008–2009: A Collaborative Survey; Technical Report EPA/841/R-16/007; Office of Water, Office of Research and Development: Washington, DC, USA, 2016.
- United States Environmental Protection Agency. National Coastal Condition Assessment 2015: A Collaborative Survey of the Nation’s Estuaries and Great Lakes Nearshore Waters; Technical Report EPA 841-R-21-001; Office of Water, Office of Research and Development: Washington, DC, USA, 2021.
- Sondergaard, M.; Jeppesen, E. Anthropogenic impacts on lake and stream ecosystems, and approaches to restoration. J. Appl. Ecol. 2007, 44, 1089–1094. [Google Scholar] [CrossRef]
- Wurtsbaugh, W.A.; Paerl, H.W.; Dodds, W.K. Nutrients, eutrophication and harmful algal blooms along the freshwater to marine continuum. WIREs Water 2019, 6, e1373. [Google Scholar] [CrossRef]
- Smucker, N.J.; Beaulieu, J.J.; Nietch, C.T.; Young, J.L. Increasingly severe cyanobacterial blooms and deep-water hypoxia coincide with warming water temperatures in reservoirs. Glob. Chang. Biol. 2021, 27, 2507–2519. [Google Scholar] [CrossRef] [PubMed]
- Howarth, R.W.; Sharpley, A.; Walker, D. Sources of nutrient pollution to coastal waters in the United States: Implications for achieving coastal water quality goals. Estuaries 2002, 25, 656–676. [Google Scholar] [CrossRef]
- Seitzinger, S.P.; Harrison, J.A.; Dumont, E.; Beusen, A.H.W.; Bouwman, A.F. Sources and delivery of carbon, nitrogen, and phosphorus to the coastal zone: An overview of Global Nutrient Export from Watersheds (NEWS) models and their application. Glob. Biogeochem. Cycles 2005, 19, GB4S01. [Google Scholar] [CrossRef] [Green Version]
- Boyer, E.W.; Howarth, R.W.; Galloway, J.N.; Dentener, F.J.; Green, P.A.; Vörösmarty, C.J. Riverine nitrogen export from the continents to the coasts. Glob. Biogeochem. Cycles 2006, 20, GB1S91. [Google Scholar] [CrossRef] [Green Version]
- Groffman, P.M.; Law, N.L.; Belt, K.T.; Band, L.E.; Fisher, G.T. Nitrogen fluxes and retention in urban watershed ecosystems. Ecosystems 2020, 7, 393–403. [Google Scholar] [CrossRef]
- Alexander, R.B.; Smith, R.A.; Schwarz, G.E.; Boyer, E.W.; Nolan, J.V.; Brakebill, J.W. Differences in phosphorus and nitrogen delivery to the Gulf of Mexico from the Mississippi River Basin. Environ. Sci. Technol. 2008, 42, 822–830. [Google Scholar] [CrossRef] [Green Version]
- Kaushal, S.S.; Mayer, P.M.; Vidon, P.G.; Smith, R.M.; Pennino, M.J.; Newcomer, T.A.; Duan, S.; Welty, C.; Belt, K.T. Land use and climate variability amplify carbon, nutrient, and contaminant pulses: A review with management implications. J. Am. Water Resour. Assoc. 2014, 50, 585–614. [Google Scholar] [CrossRef] [Green Version]
- Loecke, T.D.; Burgin, A.J.; Riveros-Iregui, D.A.; Ward, A.S.; Thomas, S.A.; Davis, C.A.; St Clair, M.A. Weather whiplash in agricultural regions drives deterioration of water quality. Biogeochemistry 2017, 133, 7–15. [Google Scholar] [CrossRef]
- Balint, S.J.; Joyce, E.; Pennino, S.; Oczkowski, A.; McKinney, R.; Hastings, M.G. Identifying sources and impacts of precipitation-derived nitrogen in Narragansett Bay, RI. Estuaries Coasts 2021, 45, 1287–1304. [Google Scholar] [CrossRef]
- Smucker, N.J.; Pilgrim, E.M.; Wu, H.; Nietch, C.T.; Darling, J.A.; Molina, M.; Johnson, B.R.; Yuan, L.L. Characterizing temporal variability in streams supports nutrient indicator development using diatom and bacterial DNA metabarcoding. Sci. Total Environ. 2022, 831, 154960. [Google Scholar] [CrossRef] [PubMed]
- Yuan, L.L.; Smucker, N.J.; Nietch, C.T.; Pilgrim, E.M. Quantifying spatial and temporal relationships between diatoms and nutrients in streams strengthens evidence of nutrient effects from monitoring data. Freshw. Sci. 2022, 41, 100–112. [Google Scholar] [CrossRef] [PubMed]
- Karube, Z.; Sakai, Y.; Takeyama, T.; Okuda, N.; Kohzu, A.; Yoshimizu, C.; Nagata, T.; Tayasu, I. Carbon and nitrogen stable isotope ratios of macroinvertebrates in the littoral zone of Lake Biwa as indicators of anthropogenic activities in the watershed. Ecol. Res. 2010, 25, 847–855. [Google Scholar]
- Smucker, N.J.; Kuhn, A.; Cruz-Quinones, C.J.; Serbst, J.R.; Lake, J.L. Stable isotopes of algae and macroinvertebrates in streams respond to watershed urbanization, inform management goals, and indicate food web relationships. Ecol. Indic. 2018, 90, 295–304. [Google Scholar] [CrossRef]
- Dalu, T.; Cuthbert, R.N.; Taylor, J.C.; Magoro, M.L.; Weyl, O.L.F.; Froneman, P.W.; Wasserman, R.J. Benthic diatom-based indices and isotopic biomonitoring of nitrogen pollution in a warm temperate Australian river system. Sci. Tot. Environ. 2020, 748, 142452. [Google Scholar] [CrossRef]
- Cabana, G.; Rasmussen, J.B. Comparison of aquatic food chains using nitrogen isotopes. Proc. Natl. Acad. Sci. USA 1996, 93, 10844–10847. [Google Scholar] [CrossRef] [Green Version]
- Vander Zanden, M.J.; Rasmussen, J.B. Primary consumer δ13C and δ15N and the trophic position of aquatic consumers. Ecology 1999, 80, 1395–1404. [Google Scholar] [CrossRef]
- Lake, J.L.; McKinney, R.A.; Osterman, F.A.; Pruell, R.J.; Kiddon, J.; Ryba, S.A.; Libby, A.D. Stable nitrogen isotopes as indicators of anthropogenic activities in small freshwater systems. Can. J. Fish Aquat. Sci. 2001, 58, 870–878. [Google Scholar] [CrossRef]
- McKinney, R.A.; Lake, J.L.; Charpentier, M.A.; Ryba, S. Using mussel isotope ratios to assess anthropogenic nitrogen inputs to freshwater ecosystems. Environ. Monit. Assess. 2002, 74, 167–192. [Google Scholar] [CrossRef]
- Atkinson, C.L.; Christian, A.D.; Spooner, D.E.; Vaughn, C.C. Long-lived organisms provide an integrative footprint of agricultural land use. Ecol. Appl. 2014, 24, 375–384. [Google Scholar] [CrossRef] [PubMed]
- Brooks, J.R.; Compton, J.E.; Lin, J.; Herlihy, A.; Nahlik, A.M.; Rugh, W.; Weber, M. d15N of Chironomidae: An index of nitrogen sources and processing within watersheds for national aquatic monitoring programs. Sci. Total Environ. 2022, 20, 151867. [Google Scholar] [CrossRef] [PubMed]
- Carmichael, R.H.; Annett, B.; Valiela, I. Nitrogen loading to pleasant bay, cape cod:application of models and stable isotopes to detect incipient nutrient enrichment of estuaries. Mar. Pollut. Bull. 2004, 48, 137–143. [Google Scholar] [CrossRef]
- Carmichael, R.H.; Hattenrath, T.; Ivan Valiela, I.; Michener, R.H. Nitrogen stable isotopes in the shell of Mercenaria mercenaria trace wastewater inputs from watersheds to estuarine ecosystems. Aquat. Biol. 2008, 4, 99–111. [Google Scholar] [CrossRef]
- Lake, J.L.; Serbst, J.R.; Kuhn, A.; Smucker, N.J.; Edwards, P.; Libby, A.; Charpentier, M.; Miller, K. Use of stable isotopes in benthic organic material as a baseline for estimating fish trophic positions in lakes. Can. J. Fish Aquat. Sci. 2019, 76, 1227–1237. [Google Scholar] [CrossRef] [Green Version]
- Watanabe, S.; Kodama, M.; Fukuda, M. Nitrogen stable isotope ratio in the manila clam, Ruditapes philippinarum, reflects eutrophication levels in tidal flats. Mar. Pollut. Bull. 2009, 58, 1447–1453. [Google Scholar] [CrossRef]
- Carmichael, R.H.; Rutecki, D.; Annett, B.; Gaines, E.; Valiela, I. Position of horseshoe crabs in estuarine food webs: N and C stable isotopic study of foraging ranges and diet composition. J. Exp. Mar. Biol. Ecol. 2004, 299, 231–253. [Google Scholar] [CrossRef]
- Mitchell, J.S.; Bailey, R.C.; Knapton, R.W. Abundance of Dreissena polymorpha and Dreissena bugensis in a warmwater plume: Effects of depth and temperature. Can. J. Fish. Aquat. Sci. 1996, 53, 1705–1712. [Google Scholar] [CrossRef]
- Hodell, D.A.; Schelske, C.L. Production, sedimentation, and isotopic composition of organic matter in Lake Ontario. Limnol. Oceanogr 1998, 43, 200–214. [Google Scholar] [CrossRef]
- Post, D.M. Using stable isotopes to estimate trophic position: Models, methods, and assumptions. Ecology 2002, 83, 703–718. [Google Scholar] [CrossRef]
- Vander Zanden, M.J.; Shuter, B.J.; Lester, N.; Rasmussen, J.B. Patterns of food chain length in lakes: A stable isotope study. Am. Nat. 1999, 154, 406–416. [Google Scholar] [CrossRef] [PubMed]
- McConnaughey, T.A.; Gillikin, D.P. Carbon isotopes in mollusk shell carbonates. Geo. Mar. Lett. 2008, 28, 287–299. [Google Scholar] [CrossRef]
- Komada, T.; Anderson, M.R.; Dorfmeier, C.L. Carbonate removal from coastal sediments for the determination of organic carbon and its isotopic signatures, δ13C and δ14C: Comparison of fumigation and direct acidification by hydrochloric acid. Limnol. Oceanogr. Methods. 2008, 6, 254–262. [Google Scholar] [CrossRef] [Green Version]
- Boothman, W.; (U.S. Environmental Protection Agency Office of Research and Development, 27 Tarzwell Drive, Narragansett RI, USA. Personal communication, 2021. Manuscript in preparation.
- Oczkowski, A.; Nixon, S.; Henry, K.; DiMilla, P.; Pilson, M.; Granger, S.; Buckley, B.; Thornber, C.; McKinney, R.; Chaves, J. Distribution and Trophic Importance of Anthropogenic Nitrogen in Narragansett Bay: An Assessment Using Stable Isotopes, Estuaries and Coasts. J. CERF 2008, 31, 53–69. [Google Scholar] [CrossRef]
- RIGIS (2011) Rhode Island Geographic Information System. Land Cover/Land Use for Rhode Island. 1988, 2003–2004, 2011. Accessed on 15 July 2016 for Watershed Population and Lake Data; Accessed on 3 December 2019 for Estuaries. 2011. Available online: www.rigis.org (accessed on 6 October 2022).
- Mass GIS (2005) Massachusetts Geographic Information System (Mass-GIS). Massachusetts Land Use 1985, 1999, and 2005. (2005) Massachusetts Geographic Information System (Mass-GIS). Massachusetts Land Use Support/Application-Serv/Office-of-Geographic-Information-Massgis. Accessed on 15 July 2016 for Watershed Population and Lake Data; Accessed on 3 December 2019 for Estuaries. Available online: www.mass.gov/anf/research-and-tech/it-serv-and-MassGIS (accessed on 6 October 2022).
- Sleeter, R.; Gould, M. Geographic Information System Software to Remodel Population Data Using Dasymetric Mapping Methods: U.S. Geological Survey Techniques and Methods 11-C2; US Department of the Interior, US Geological Survey: Denver, CO, USA, 2007; 15p.
- United States Census Bureau. Historical Data: 2010. 2010. Available online: www.census.gov/programs-surveys/popest.html (accessed on 3 December 2019).
- McClelland, J.W.; Valiela, I.; Michener, R.H. Nitrogen-stable isotope signatures in estuarine food webs: A record of increasing urbanization in coastal watersheds. Limnol. Oceanogr. 1997, 42, 930–937. [Google Scholar] [CrossRef]
- Vuorio, K.; Tarvainen, M.; Sarvala, J. Unionid mussels as stable isotope baseline indicators for long-lived secondary consumers in pelagic food web comparisons. Fundam. Appl. Limnol. 2007, 169, 237–245. [Google Scholar] [CrossRef]
- Yasuno, N.; Shindo, K.; Takagi, Y.; Kanaya, G.; Shikano, S.; Fujimoto, Y.; Shimada, T.; Kikuchi, E. Ontogenetic changes in the trophic position of a freshwater Unionidae mussel. Fundam. Appl. Limnol. 2014, 184, 341–349. [Google Scholar] [CrossRef]
- Novais, A.; Dias, E.; Sousa, R. Inter- and intraspecific variation of carbon and nitrogen stable isotope ratios in freshwater bivalves. Hydrobiologia 2016, 765, 149–158. [Google Scholar] [CrossRef]
- Roegner, G.C.; Roger, L.M. Hard Clam Mercenaria mercenaria. In VIMS Books and Book Chapters 20; Virginia Institute of Marine Science: Gloucester Point, VA, USA, 1991; Available online: https://scholarworks.wm.edu/vimsbooks/20 (accessed on 8 November 2022).
- Pelletier, M.C.; Gillett, D.J.; Hamilton, A.; Grayson, T.; Hansen, V.; Leppo, E.W.Q.; Wiesberg, S.B.; Borja, A. Adaptation and application of multivariate (M-AMBI) in US coastal waters. Ecol. Indic. 2018, 89, 818–827. [Google Scholar] [CrossRef]
- Pinto, R.; Patricio, J.; Baeta, A.; Neto, B.D.F.J.M.; Marques, J.C. Review and evaluation of estuarine biotic indices to assess benthic condition. Ecol. Indic. 2009, 9, 1–25. [Google Scholar] [CrossRef] [Green Version]
- Pruden, M.J.; Dietl, G.P.; Handley, J.C.; Smith, J.A. Using molluscs to assess ecological quality status of soft-bottom habitats along the Atlantic coastline of the United States. Ecol. Indic. 2021, 129, 107910. [Google Scholar] [CrossRef]
- Botrel, M.; Gregory-Eaves, I.; Maranger, R. Defining drivers of nitrogen stable isotopes (δ15N) of surface sediments in temperate lakes. J. Paleolimnol. 2014, 52, 419–433. [Google Scholar] [CrossRef]
- Sabo, R.D.; Clark, C.M.; Bash, J.; Sobota, D.; Cooter, E.; Dobrowolski, J.P.; Compton, J.E. Decadal shift in nitrogen inputs and fluxes across the contiguous United States: 2002–2012. J. Geophys. Res. Biogeosciences 2019, 124, 3104–3124. [Google Scholar] [CrossRef]
| Lakes | ||||||||
|---|---|---|---|---|---|---|---|---|
| Spearman Correlations | ||||||||
| Log%Impsur | % UrRes | Logpop | % Forest | % Agr | % WetL | % Barren | % Water | |
| δ15NBOM | 0.81 | 0.78 | 0.54 | −0.71 | 0.15 | −0.26 | 0.31 | −0.18 |
| p | <0.001 | <0.001 | <0.001 | <0.001 | NS | NS | 0.03 | NS |
| Pearson Correlations | ||||||||
| Log%Impsur | % UrRes | Logpop | % Forest | % Agr | % WetL | % Barren | % Water | |
| δ15NBOM | 0.79 | 0.69 | 0.59 | −0.71 | 0.14 | −0.23 | 0.35 | −0.27 |
| p | <0.001 | <0.001 | <0.001 | <0.001 | NS | NS | 0.01 | NS |
| Estuaries | ||||||||
| Spearman Correlations | ||||||||
| Log%Impsur | %UrRes | Logpop | % Forest | % Agr | % WetL | % Barren | % Water | |
| δ15NBOM | 0.73 | 0.5 | 0.33 | 0.02 | 0.25 | −0.17 | −0.57 | 0.05 |
| p | 0.02 | NS | NS | NS | NS | NS | NS | NS |
| Pearson Correlations | ||||||||
| Log%Impsur | % UrRes | Logpop | % Forest | % Agr | % WetL | % Barren | % Water | |
| δ15NBOM | 0.75 | 0.48 | 0.67 | 0.04 | 0.31 | −0.03 | −0.13 | −0.07 |
| p | 0.02 | NS | 0.05 | NS | NS | NS | NS | NS |
| Lakes | ||||
|---|---|---|---|---|
| Log%Impsur | % UrRes | Logpop | % Forest | |
| δ15NBOM | 0.79 | 0.74 | 0.49 | −0.73 |
| p | <0.001 | <0.001 | 0.011 | <0.001 |
| δ15NUN | 0.75 | 0.66 | 0.63 | −0.67 |
| p | <0.001 | <0.001 | <0.001 | <0.001 |
| Estuaries | ||||
| Log%Impsur | % UrRes | Logpop | % Forest | |
| δ15NBOM | 0.75 | 0.48 | 0.67 | 0.04 |
| p | 0.019 | NS | NS | NS |
| δ15NMM | 0.61 | 0.36 | 0.55 | 0.19 |
| p | NS | NS | NS | NS |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lake, J.L.; Serbst, J.R.; Kuhn, A.; Charpentier, M.; Smucker, N.J. Evaluation of Nitrogen and Carbon Stable Isotopes in Filter Feeding Bivalves and Surficial Sediment for Assessing Aquatic Condition in Lakes and Estuaries. Water 2022, 14, 3712. https://doi.org/10.3390/w14223712
Lake JL, Serbst JR, Kuhn A, Charpentier M, Smucker NJ. Evaluation of Nitrogen and Carbon Stable Isotopes in Filter Feeding Bivalves and Surficial Sediment for Assessing Aquatic Condition in Lakes and Estuaries. Water. 2022; 14(22):3712. https://doi.org/10.3390/w14223712
Chicago/Turabian StyleLake, James L., Jonathan R. Serbst, Anne Kuhn, Michael Charpentier, and Nathan J. Smucker. 2022. "Evaluation of Nitrogen and Carbon Stable Isotopes in Filter Feeding Bivalves and Surficial Sediment for Assessing Aquatic Condition in Lakes and Estuaries" Water 14, no. 22: 3712. https://doi.org/10.3390/w14223712






