Assessment of Heavy Metal Pollution in Suburban River Sediment of Nantong (China) and Preliminary Exploration of Solidification/Stabilization Scheme
Abstract
:1. Introduction
2. Materials and Methods
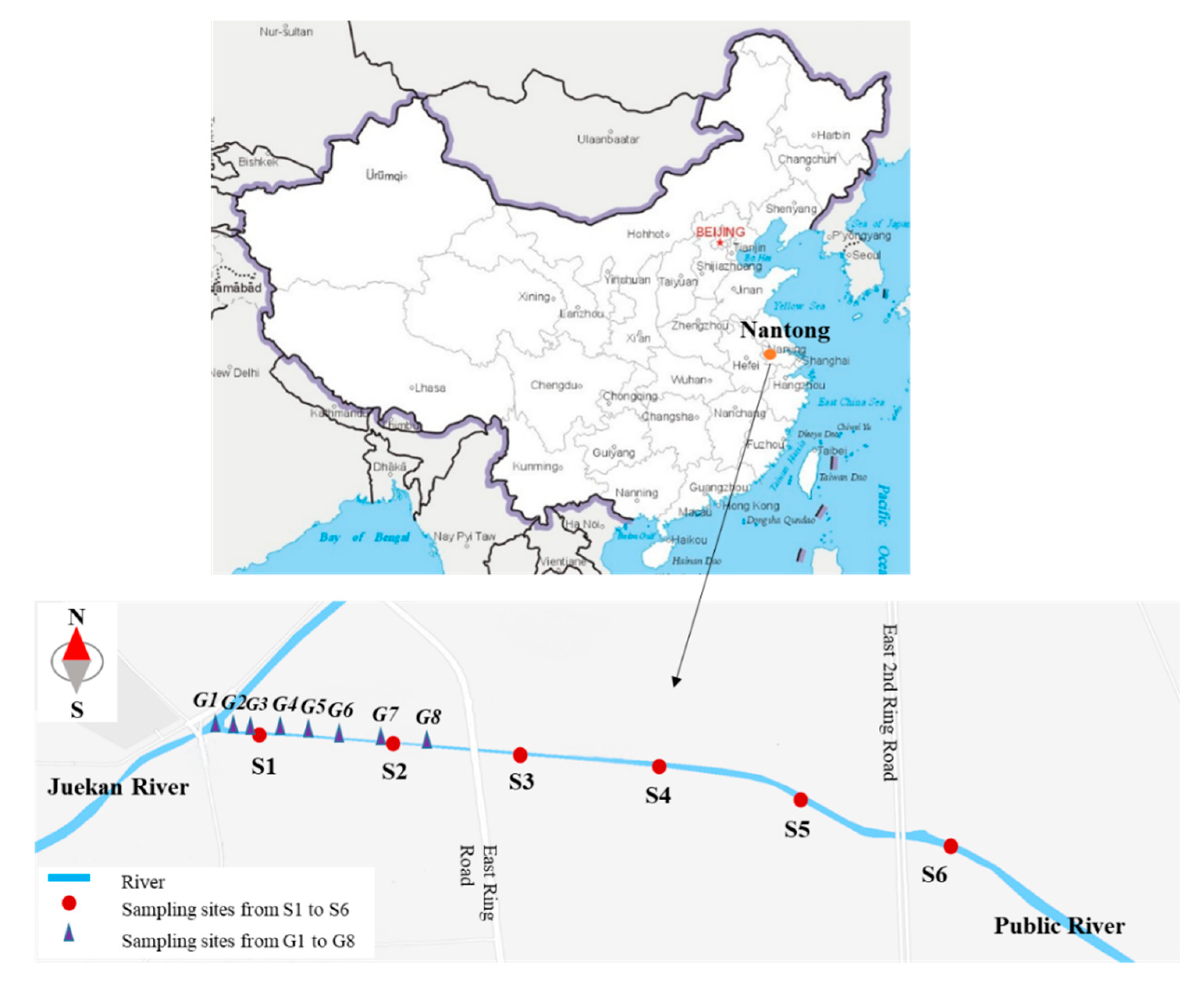
2.1. Study Area
2.2. Sample Collection
2.3. Solidification/Stabilization Experiment
2.4. Chemical Analysis
2.5. Quality Assurance and Quality Control (QA/QC)
2.6. Statistical Analysis
3. Results and discussion
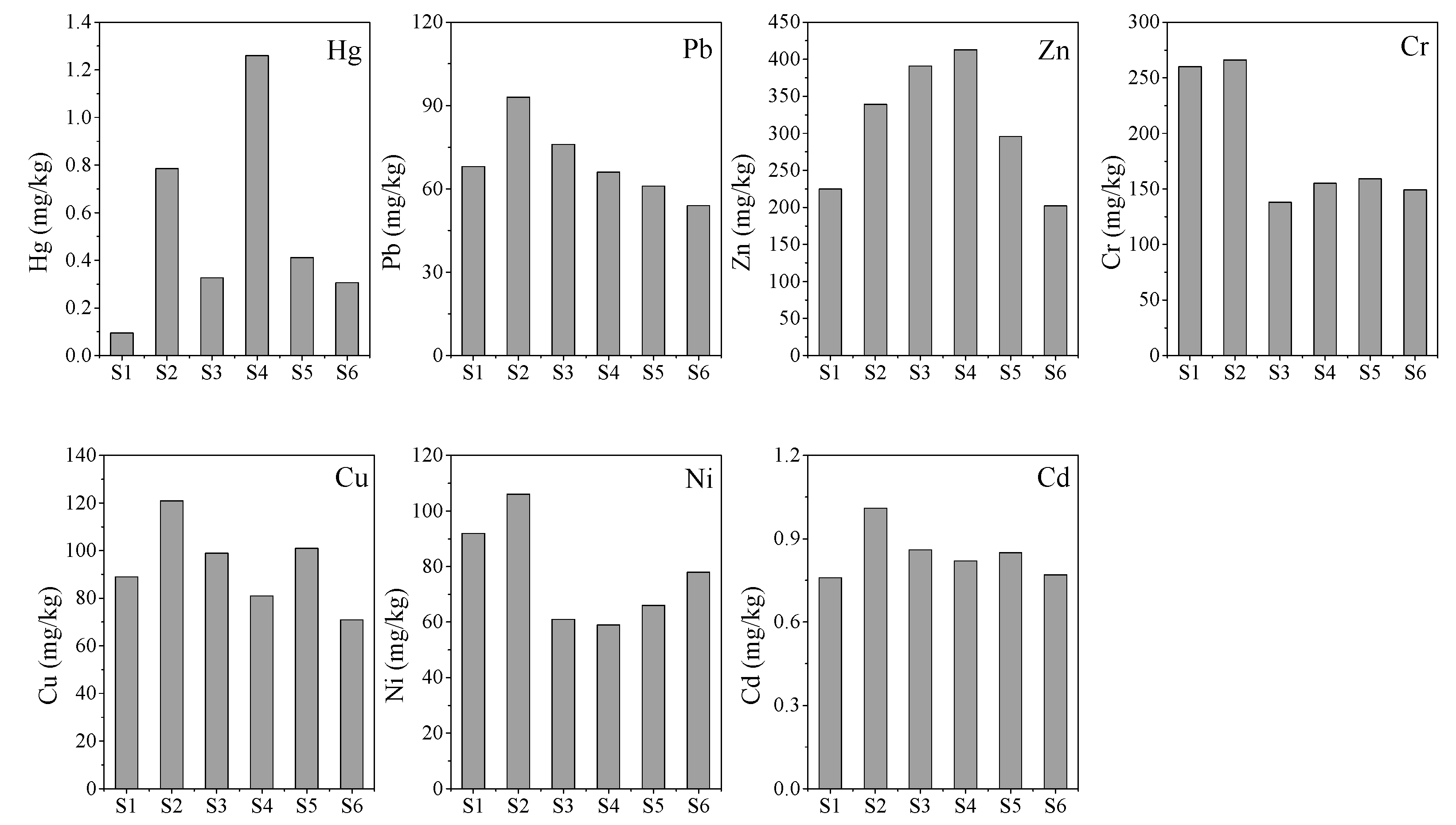
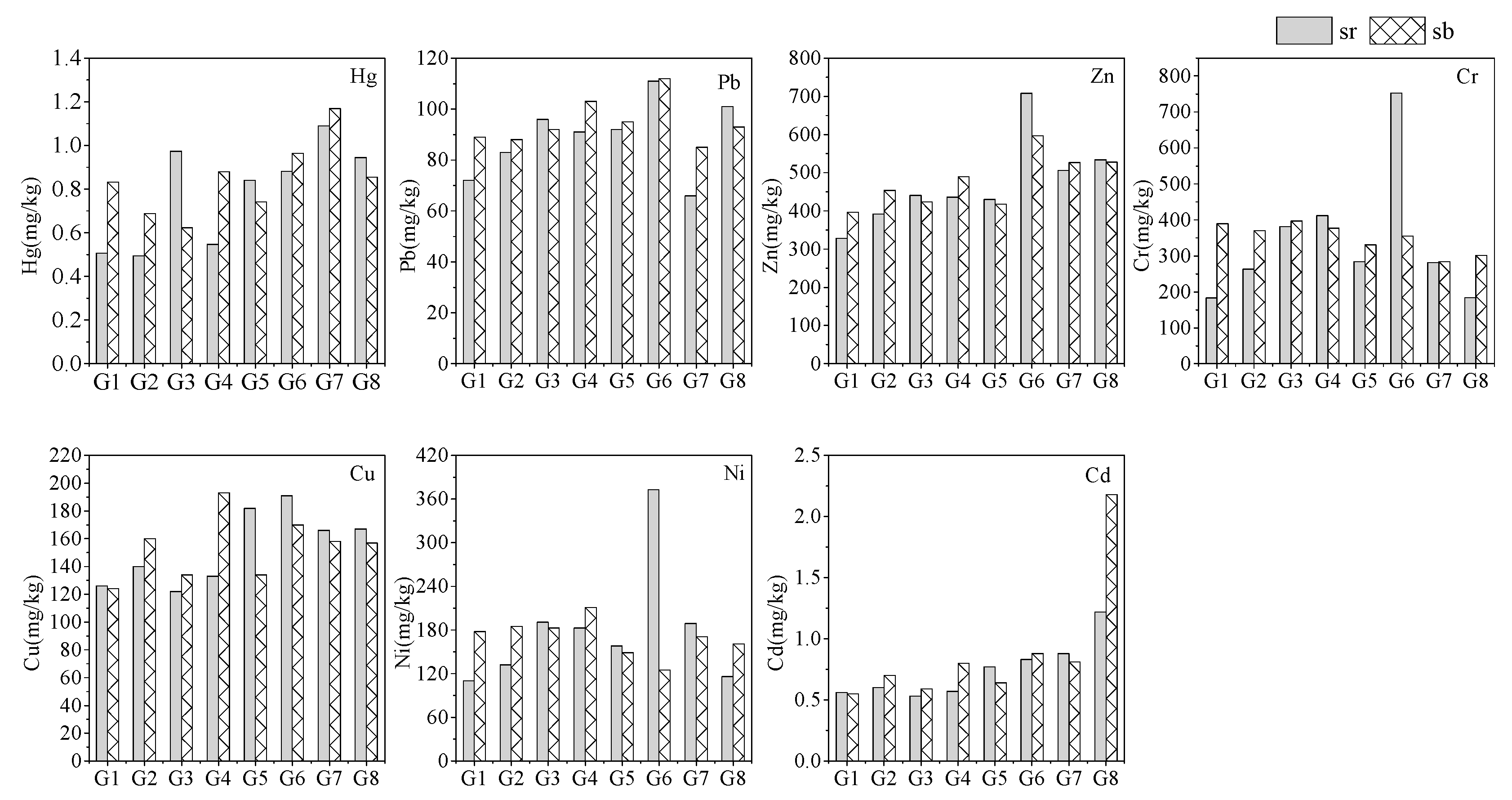
3.1. Distribution of Heavy Metals in Sediment
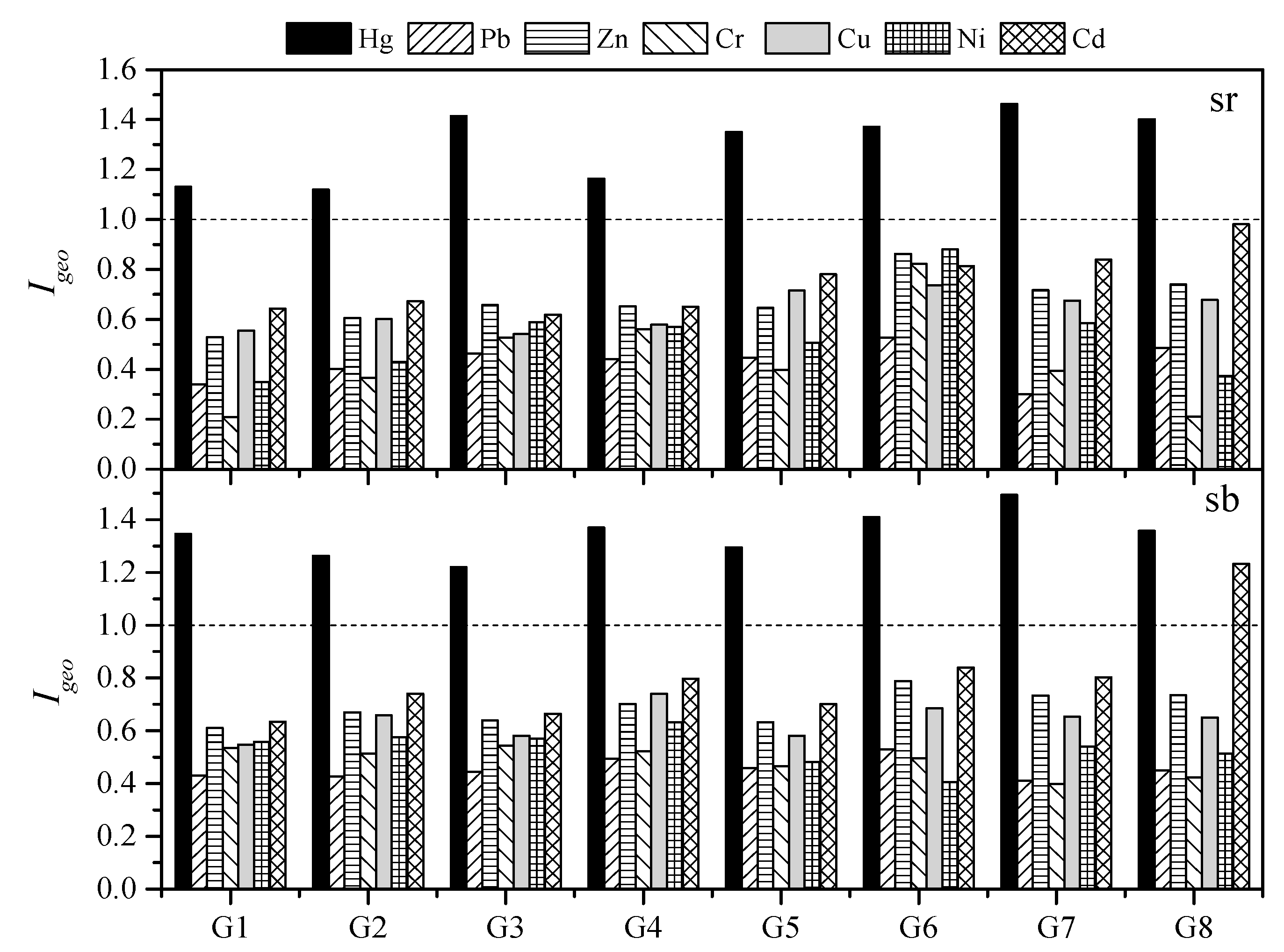
3.2. Geo-Accumulation Index (Igeo)
3.3. Comparison with Relevant Screening Levels
3.4. Leaching Behavior of Heavy Metals
3.5. Preliminary Exploration of Solidification/Stabilization Scheme
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Su, C.; Jiang, L.Q.; Zhang, W.J. A review on heavy metal contamination in the soil worldwide: Situation, impact and remediation techniques. Environ. Skept. Crit. 2014, 3, 24–38. [Google Scholar]
- Aradpour, S.; Noori, R.; Naseh, M.R.V.; Hosseinzadeh, M.; Safavi, S.; Ghahraman-Rozegar, F.; Maghrebi, M. Alarming carcinogenic and non-carcinogenic risk of heavy metals in Sabalan dam reservoir, Northwest of Iran. Environ. Pollut. Bioavailab. 2021, 33, 278–291. [Google Scholar] [CrossRef]
- Singh, K.P.; Mohan, D.; Singh, V.K.; Malik, A. Studies on distribution and fractionation of heavy metals in Gomti river sediments—A tributary of the Ganges, India. J. Hydrol. 2005, 312, 14–27. [Google Scholar] [CrossRef]
- Salomons, W.; Stigliani, W.M. Biogeodynamics of Pollutants in Soils and Sediments: Risk Assessment of Delayed and Non-Linear Responses; Springer: New York, NY, USA, 1995; pp. 331–343. [Google Scholar]
- Lei, Y.; Wang, P.F.; Zhang, W.M. The Effects of Aquatic Plant Vallisneria Natans on the Distribution of Metals under Different Hydrodynamic Conditions. Appl. Mech. Mater. 2015, 737, 362–366. [Google Scholar] [CrossRef]
- Hill, N.A.; Simpson, S.L.; Johnston, E.L. Beyond the bed: Effects of metal contamination on recruitment to bedded sediments and overlying substrata. Environ. Pollut. 2013, 173, 182–191. [Google Scholar] [CrossRef]
- Canli, M.; Atli, G. The relationships between heavy metal (Cd, Cr, Cu, Fe, Pb, Zn) levels and the size of six Mediterranean fish species. Environ. Pollut. 2003, 121, 129–136. [Google Scholar] [CrossRef]
- João, P.V.; Artur, J.M.V.; Luisa, D. Assessment of heavy metal pollution from anthropogenic activities and remediation strategies: A review. J. Environ. Manag. 2019, 246, 101–118. [Google Scholar]
- Mulligan, C.N.; Yong, R.N.; Gibbs, B.F. An evaluation of technologies for the heavy metal remediation of dredged sediments. J. Hazard. Mater. 2001, 85, 145–163. [Google Scholar] [CrossRef]
- Peng, J.F.; Song, Y.H.; Yuan, P.; Cui, X.Y.; Qiu, G.L. The remediation of heavy metals contaminated sediment. J. Hazard. Mater. 2009, 161, 633–640. [Google Scholar] [CrossRef]
- Abollino, O.; Aceto, M.; Malandrino, M.; Sarzanini, C.; Mentasti, E. Adsorption of heavy metals on Na-montmorillonite. Effect of pH and organic substances. Water Res. 2003, 37, 1619–1627. [Google Scholar] [CrossRef]
- Chen, Y.; Zhang, Y.; Chen, G. Remediation of Heavy Metal Contaminated Soils by Lime: A Review. Ecol. Environ. Sci. 2016, 25, 1419–1424. [Google Scholar]
- Dries, J.; Bastiaens, L.; Springael, D.; Kuypers, S.; Agathos, S.N.; Diels, L. Effect of humic acids on heavy metal removal by zero-valent iron in batch and continuous flow column systems. Water Res. 2005, 39, 3531–3540. [Google Scholar] [CrossRef] [PubMed]
- Hosny, W.M.; Hadi, A.K.A.; El-Saied, H.; Basta, A.H. Metal chelates with some cellulose derivatives. Part III. Synthesis and structural chemistry of nickel (II) and copper (II) complexes with carboxymethyl cellulose. Polym. Int. 2010, 37, 93–96. [Google Scholar] [CrossRef]
- Tang, P.P.; Zhang, W.L.; Chen, Y.H.; Ghen, G.; Xu, J. Stabilization/solidification and recycling of sediment from Taihu Lake in China: Engineering behavior and environmental impact. Waste Manag. 2020, 116, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.; Arora, V.K.; Biswas, S. Contaminated dredged soil stabilization using cement and bottom ash for use as highway subgrade fill. Int. J. Geo-Eng. 2017, 8, 20. [Google Scholar] [CrossRef] [Green Version]
- Senneca, O.; Cortese, L.; Martino, R.D.; Fabbricino, M.; Scopino, A. Mechanisms affecting the delayed efficiency of cement based stabilization/solidification processes. J. Clean. Prod. 2020, 261, 1–9. [Google Scholar] [CrossRef]
- Chrysochoou, M.; Dermatas, D. Evaluation of ettringite and hydrocalumite formation for heavy metal immobilization: Literature review and experimental study. J. Hazard. Mater. 2006, 136, 20–33. [Google Scholar] [CrossRef]
- Guo, Y.F. Present Situation Analysis and Control Countermeasures of Water Pollution in Rudong Rural Area. In Agricultural Extension; Yangzhou University: Yangzhou, China, 2006. [Google Scholar]
- Sakan, S.; Sakan, N.; Anđelković, I.; Trifunović, S.; Đorđević, D. Study of potential harmful elements (arsenic, mercury and selenium) in surface sediments from Serbian rivers and artificial lakes. J. Geochem. Explor. 2017, 180, 24–34. [Google Scholar] [CrossRef] [Green Version]
- Tüzen, M. Determination of heavy metals in soil, mushroom and plant samples by atomic absorption spectrometry. Microchem. J. 2003, 74, 289–297. [Google Scholar] [CrossRef]
- Mann, S.S.; Rate, A.W. Determination of cadmium in soil extracts containing high levels of iron and aluminum by graphite furnace atomic absorption spectrophotometry. Commun. Soil Sci. Plant Anal. 1998, 29, 2725–2737. [Google Scholar] [CrossRef]
- Ministry of Environmental Protection; National Standards of China. Solid Waste-Extraction Procedure for Leaching Toxicity-Sulphuric Acid & Nitric Acid Method; China Environmental Science Press: Beijing, China, 2007; pp. 1–10.
- Ministry of Environmental Protection; National Standards of China. Solid Waste-Determination of Metals-Inductively Coupled Plasma Mass Spectrome-Trey (ICP-MS); China Environmental Science Press: Beijing, China, 2015; pp. 1–10.
- Bartoli, G.; Papa, S.; Sagnella, E.; Fioretto, A. Heavy metal content in sediments along the Calore river: Relationships with physical–chemical characteristics. J. Environ. Manag. 2012, 95, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Al-Khashman, O.A.; Shawabkeh, R.A. Metals distribution in soils around the cement factory in southern Jordan. Environ. Pollut. 2006, 140, 387–394. [Google Scholar] [CrossRef] [PubMed]
- Steiner, F.; Lana, M.d.C. Effect of pH on boron adsorption in some soils of Paraná, Brazil. Chil. J. Agric. Res. 2013, 73, 181–186. [Google Scholar] [CrossRef] [Green Version]
- Liao, Q.L.; Liu, C.; Xu, Y.; Jin, Y.; Wu, Y.Z.; Hua, M.; Zhu, B.W.; Weng, Z.H. Geochemical baseline values of elements in soil of Jiangsu Province. Geol. China 2011, 28, 1363–1378. [Google Scholar]
- Aradpour, S.; Noori, R.; Tang, Q.H.; Bhattarai, R.; Klve, B. Metal contamination assessment in water column and surface sediments of a warm monomictic man-made lake: Sabalan Dam Reservoir, Iran. Hydrol. Res. 2020, 51, 799–814. [Google Scholar] [CrossRef]
- Bazrafshan, E.; Mostafapour, F.K.; Esmaelnejad, M.; Ebrahimzadeh, G.R.; Mahvi, A.H. Concentration of heavy metals in surface water and sediments of Chah Nimeh water reservoir in Sistan and Baluchestan province, Iran. Desalination Water Treat. 2015, 57, 9332–9342. [Google Scholar] [CrossRef]
- Tang, Q.; Bao, Y.; He, X.; Zhou, H.; Cao, Z.; Gao, P.; Zhong, R.; Hu, Y.; Zhang, X. Sedimentation and associated trace metal enrichment in the riparian zone of the Three Gorges Reservoir, China. Sci. Total Environ. 2014, 479–480, 258–266. [Google Scholar] [CrossRef]
- Bibi, M.H.; Ahmed, F.; Ishiga, H. Assessment of metal concentrations in lake sediments of southwest Japan based on sediment quality guidelines. Environ. Geol. 2007, 52, 625–639. [Google Scholar] [CrossRef]
- Zahran, M.A.; El-Amier, Y.A.; Elnaggar, A.A.; Mohamed, H.A.; El-Alfy, E.H. Assessment and Distribution of Heavy Metals Pollutants in Manzala Lake, Egypt. J. Geosci. Environ. Prot. 2015, 3, 107–122. [Google Scholar] [CrossRef] [Green Version]
- Makokha, V.A.; Qi, Y.; Shen, Y.; Wang, J. Concentrations, Distribution, and Ecological Risk Assessment of Heavy Metals in the East Dongting and Honghu Lake, China. Expo. Health 2016, 8, 31–41. [Google Scholar] [CrossRef]
- Yang, Y.; Chen, F.; Zhang, L.; Liu, J.; Wu, S.; Kang, M. Comprehensive assessment of heavy metal contamination in sediment of the Pearl River Estuary and adjacent shelf. Mar. Pollut. Bull. 2012, 64, 1947–1955. [Google Scholar] [CrossRef] [PubMed]
- Al-Najjar, T.; Rasheed, M.; Ababneh, Z.; Ababneh, A.; Al-Omarey, H. Heavy metals pollution in sediment cores from the Gulf of Aqaba, Red Sea. Nat. Sci. 2011, 3, 775–782. [Google Scholar] [CrossRef] [Green Version]
- Zeng, F.; Ali, S.; Zhang, H.; Ouyang, Y.; Qiu, B.; Wu, F.; Zhang, G. The influence of pH and organic matter content in paddy soil on heavy metal availability and their uptake by rice plants. Environ. Pollut. 2011, 159, 84–91. [Google Scholar] [CrossRef] [PubMed]
- Zhao, K.; Liu, X.; Xu, J.; Selim, H.M. Heavy metal contaminations in a soil–rice system: Identification of spatial dependence in relation to soil properties of paddy fields. J. Hazard. Mater. 2010, 181, 778–787. [Google Scholar] [CrossRef]
- Zhai, M.; Kampunzu, H.; Modisi, M.P.; Totolo, O. Distribution of heavy metals in Gaborone urban soils (Botswana) and its relationship to soil pollution and bedrock composition. Environ. Geol. 2003, 45, 171–180. [Google Scholar] [CrossRef]
- Shi, G.; Chen, Z.; Bi, C.; Li, Y.; Teng, J.; Wang, L.; Xu, S. Comprehensive assessment of toxic metals in urban and suburban street deposited sediments (SDSs) in the biggest metropolitan area of China. Environ. Pollut. 2010, 158, 694–703. [Google Scholar] [CrossRef]
- Martinez, L.L.G.; Poleto, C. Assessment of diffuse pollution associated with metals in urban sediments using the geoaccumulation index (Igeo). J. Soil Sediments 2014, 14, 1251–1257. [Google Scholar] [CrossRef]
- USEPA. Guidance for Developing Ecological Soil Screening Levels (Eco-SSLs). In Review of Existing Soil Screening Benchmarks; American Petroleum Institute Biomonitoring Task Force: Washington, DC, USA, 2003; pp. 1–91. [Google Scholar]
- USEPA. Ecological Soil Screening Levels for Zinc; Office of Solid Waste and Emergency Response: Washington, DC, USA, 2007; pp. 1–808.
- USEPA. Ecological Soil Screening Levels for Nickel; Office of Solid Waste and Emergency Response: Washington, DC, USA, 2007; pp. 1–133.
- USEPA. Ecological Soil Screening Levels for Lead; Office of Solid Waste and Emergency Response: Washington, DC, USA, 2005; pp. 1–242.
- USEPA. Ecological Soil Screening Levels for Copper; Office of Solid Waste and Emergency Response: Washington, DC, USA, 2007; pp. 1–313.
- USEPA. Ecological Soil Screening Levels for Chromium; Office of Solid Waste and Emergency Response: Washington, DC, USA, 2008; pp. 1–106.
- USEPA. Ecological Soil Screening Levels for Cadmium; Office of Solid Waste and Emergency Response: Washington, DC, USA, 2005; pp. 1–236.
- Ministry of Housing and Urban-Rural Development. Planting Soil for Greening; Standards Press of China: Beijing, China, 2016; pp. 1–16.
- Ministry of Ecological and Environment. Soil Environmental Quality: Risk Control Standard for Soil Contamination of Agricultural Land; China Environmental Science Press: Beijing, China, 2018; pp. 1–7.
- Ministry of Ecological and Environment. Soil Environmental Quality: Risk Control Standard for Soil Contamination of Development Land; China Environmental Science Press: Beijing, China, 2018; pp. 1–14.
- General Administration of Quality Supervision I.a.Q., China S.A.o. Standard for Groundwater Quality; China Environmental Science Press: Beijing, China, 2017; pp. 1–16. [Google Scholar]
- Ministry of Environmental Protection; General Administration of Quality Supervision I.a.Q. Standard for Surface Water Quality; China Environmental Science Press: Beijing, China, 2002; pp. 1–34.
- Wu, C.; Liu, X.; Wang, X.; Huang, J. Synthesis and Structural Chemistry of Sodium Carboxymethyl Cellulose and Its Metal Complexes. New Technol. New Process 2009, 10, 64–69. [Google Scholar]
- Zhen, Z.; Zhiwei, H.; Wenli, L.; Xinhua, X. Synchronous Treatment of Heavy Metal Ions and Nitrate by Zero-valent Iron. Environ. Sci. 2009, 30, 775–779. [Google Scholar]
- Franco, D.V.; Silva, L.; Jardim, W.F. Reduction of Hexavalent Chromium in Soil and Ground Water Using Zero-Valent Iron Under Batch and Semi-Batch Conditions. Water Air Soil Pollut. 2009, 197, 49–60. [Google Scholar] [CrossRef]
- He, Y.J.; Wang, W.X.; Lv, L.N.; Wang, F.Z.; Hu, S.G. Curing of Cr~(3+),Pb~(2+) in Different Cement Pastes. J. Mater. Sci. Eng. 2018, 36, 530–534. [Google Scholar]
- Berardi, R.; Cioffi, R.; Santoro, L. Matrix stability and leaching behaviour in ettringite-based stabilization systems doped with heavy metals. Waste Manag. 1998, 17, 535–540. [Google Scholar] [CrossRef]
- Chen, Y.H. The Immobilization and Stability of Heavy Metal Ions by Ettringite. In College of Material Science and Engineering; Chongqing University: Chongqing, China, 2017. [Google Scholar]
- Zhong, L.; Qu, J.; Li, X.; He, X.; Zhang, Q. Simultaneous synthesis of ettringite and absorbate incorporation by aqueous agitation of a mechanochemically prepared precursor. RSC Adv. 2016, 6, 35203–35209. [Google Scholar] [CrossRef]

| Hg | Pb | Zn | Cr | Cu | Ni | Cd | |
|---|---|---|---|---|---|---|---|
| average | 0.737 | 85.8 | 431 | 303 | 137 | 149 | 0.826 |
| SD | 0.290 | 23.22 | 144 | 166 | 41.2 | 81.6 | 0.500 |
| min | 0.095 | 54 | 202 | 138 | 71 | 59 | 0.53 |
| max | 1.26 | 112 | 708 | 753 | 193 | 373 | 2.18 |
| Species | S1 | G1 | G2 | G3 | G4 | ||||
|---|---|---|---|---|---|---|---|---|---|
| sr | sb | sr | sb | sr | sb | sr | sb | ||
| Zn | - | - | - | - | - | - | - | - | |
| Cr (6+) | 4.7 | - | <2.0 | <2.0 | 3.8 | <2.0 | <2.0 | <2.0 | <2.0 |
| Ni | - | 25.5 | - | 125 | 99.5 | 43.4 | 76.7 | 51.6 | |
| Cd | - | - | - | - | - | - | - | - | |
| Species | S2 | G5 | G6 | G7 | G8 | ||||
| sr | sb | sr | sb | sr | sb | sr | sb | ||
| Zn | - | - | <6.4 | <6.4 | 19.7 | <6.4 | <6.4 | 12 | |
| Cr (6+) | 3.8 | 2.7 | <2.0 | <2.0 | <2.0 | <2.0 | <2.0 | - | 6.9 |
| Ni | 43.5 | - | 120 | - | 61 | 56.3 | - | 59.4 | |
| Cd | - | - | - | - | - | - | <1.2 | <1.2 | |
| No. | Regents | Adding Proportions | G2-sb | G6-sr |
|---|---|---|---|---|
| Ni (μg/L) | Ni (μg/L) | |||
| 1 | MMT + LM | 2% + 8% | 1.19 × 103 | 1.59 × 103 |
| 2 | MMT + LM | 4% + 6% | 530 | 1.05 × 103 |
| 3 | MMT + LM | 8% + 2% | 68.5 | 437 |
| 4 | MMT + ZVI | 2% + 8% | 50.1 | 331 |
| 5 | MMT + ZVI | 4% + 6% | 57.0 | 484 |
| 6 | MMT + ZVI | 8% + 2% | 158 | 705 |
| 7 | MMT + CMC | 9.5% + 0.5% | 221 | 1.56 × 103 |
| 8 | LM + ZVI | 2% + 8% | 23.3 | 192 |
| 9 | LM + ZVI | 4% + 6% | 51.5 | 135 |
| 10 | LM + ZVI | 8% + 2% | 404 | 1.96 × 103 |
| 11 | LM + CMC | 9.5% + 0.5% | 315 | 1.94 × 103 |
| 12 | ZVI + CMC | 9.5% + 0.5% | 53.7 | 8.2 |
| No. | Reagents | Adding Proportions | G2-sb | G6-sr |
|---|---|---|---|---|
| Ni (μg/L) | Ni (μg/L) | |||
| 1 | SAC + CMC | 1% + 0.3% | 72.3 | 64.1 |
| 2 | SAC + ZVI + CMC | 1% + 0.5% + 0.3% | 33.4 | 79.1 |
| 3 | SAC + KP + CMC | 1% + 0.5% + 0.3% | 239 | 758 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, Q.; Zhao, F.; Wu, B.; Fang, X.; Chen, J.; Yang, T.; Chai, X.; Yuan, L. Assessment of Heavy Metal Pollution in Suburban River Sediment of Nantong (China) and Preliminary Exploration of Solidification/Stabilization Scheme. Water 2022, 14, 2247. https://doi.org/10.3390/w14142247
Xu Q, Zhao F, Wu B, Fang X, Chen J, Yang T, Chai X, Yuan L. Assessment of Heavy Metal Pollution in Suburban River Sediment of Nantong (China) and Preliminary Exploration of Solidification/Stabilization Scheme. Water. 2022; 14(14):2247. https://doi.org/10.3390/w14142247
Chicago/Turabian StyleXu, Qinqin, Fengbin Zhao, Boran Wu, Xin Fang, Jun Chen, Tao Yang, Xiaoli Chai, and Liqun Yuan. 2022. "Assessment of Heavy Metal Pollution in Suburban River Sediment of Nantong (China) and Preliminary Exploration of Solidification/Stabilization Scheme" Water 14, no. 14: 2247. https://doi.org/10.3390/w14142247
APA StyleXu, Q., Zhao, F., Wu, B., Fang, X., Chen, J., Yang, T., Chai, X., & Yuan, L. (2022). Assessment of Heavy Metal Pollution in Suburban River Sediment of Nantong (China) and Preliminary Exploration of Solidification/Stabilization Scheme. Water, 14(14), 2247. https://doi.org/10.3390/w14142247






