Effect of the Presence of Virus-like Particles on Bacterial Growth in Sunlit Surface and Dark Deep Ocean Environments in the Southern East China Sea
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Site and Samplings
2.2. VLP Production
2.3. Effect of VLP Presence on Bacterial Growth
2.4. Enumeration of VLPs and Host Density by Flow Cytometry (FCM)
2.5. Statistical Analysis
3. Results and Discussion
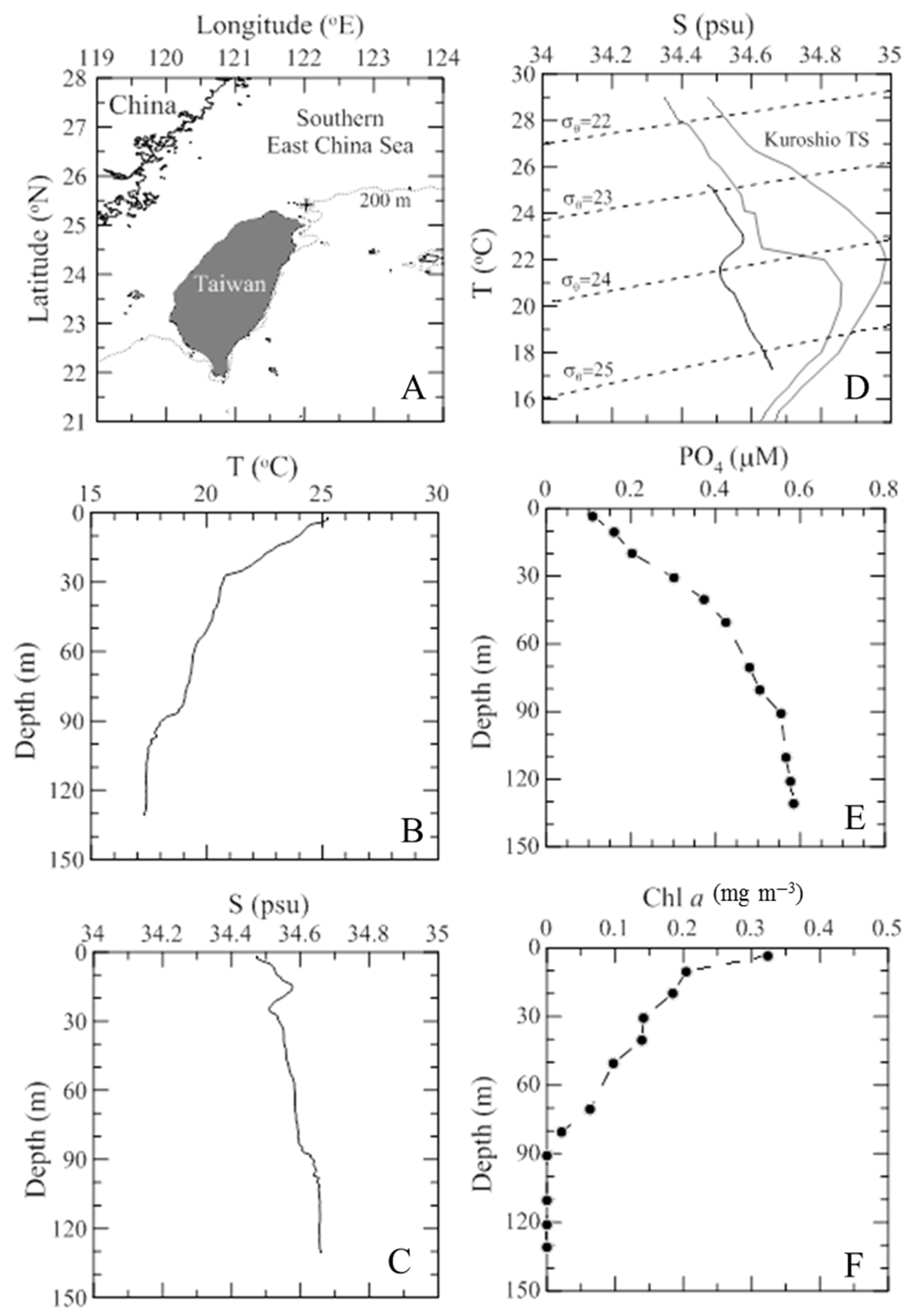
3.1. Environmental Dynamics
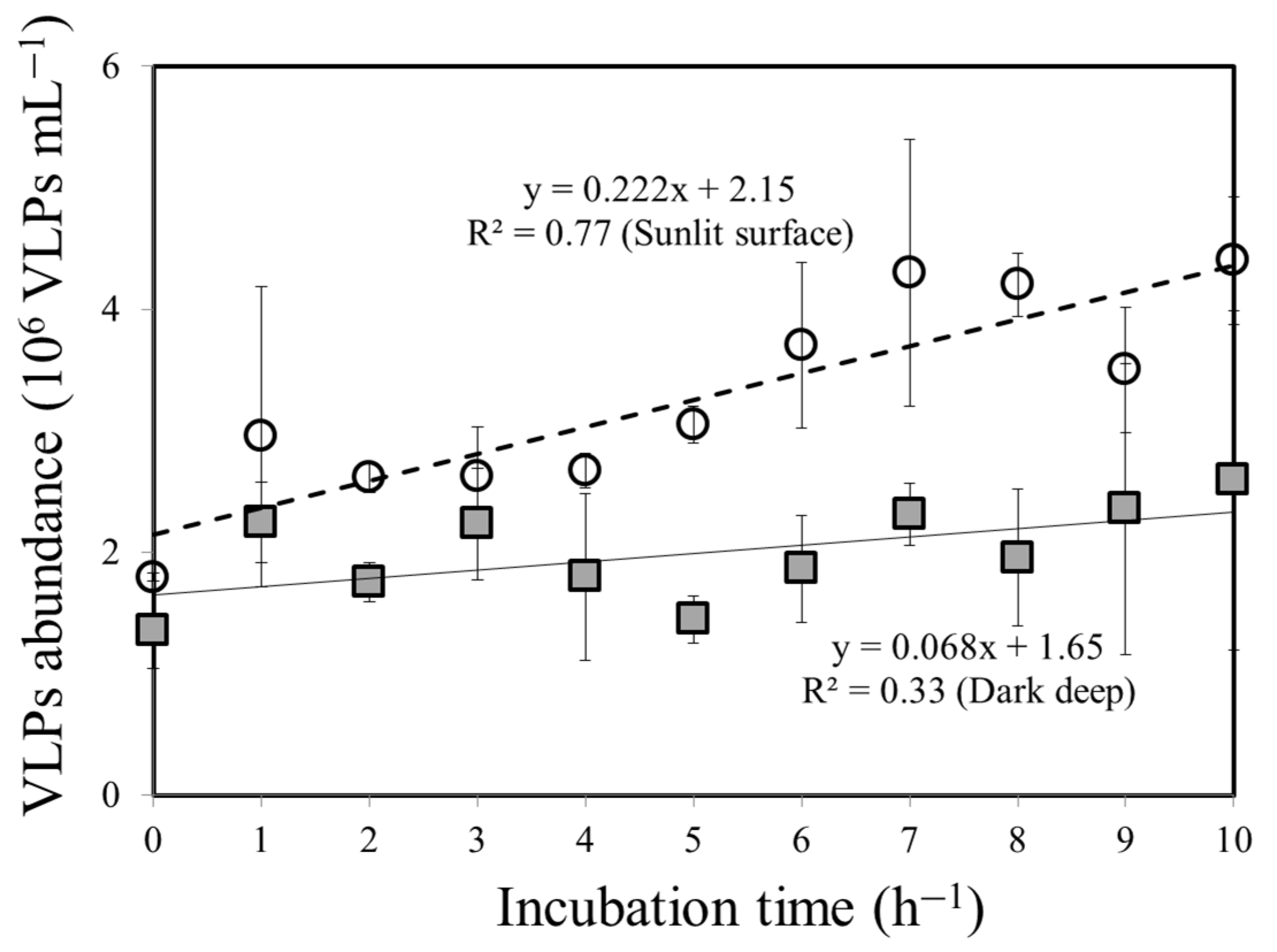
3.2. Bacteria–VLP Interaction
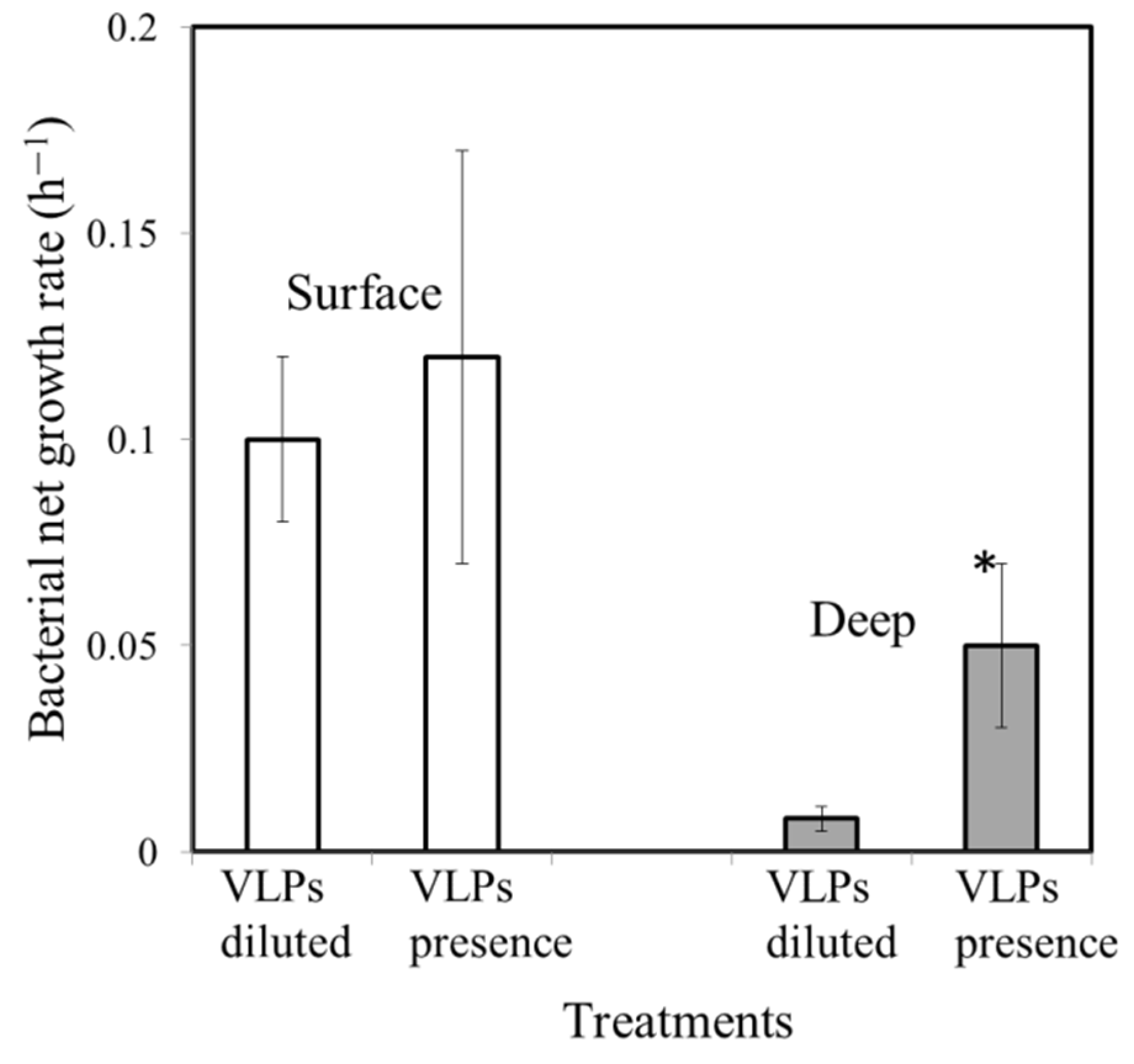
3.3. Effect of VLP Presence on Bacterial Growth Rate
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Fischer, U.R.; Velimirov, B. High control of bacterial production by viruses in a eutrophic oxbowlake. Aquat. Microb. Ecol. 2002, 27, 1–12. [Google Scholar] [CrossRef]
- Weinbauer, M.G. Ecology of prokaryotic viruses. FEMS Microbiol. Rev. 2004, 28, 127–181. [Google Scholar] [CrossRef]
- Suttle, C.A. Marine viruses—Major players in the global ecosystem. Nat. Rev. Microbiol. 2007, 5, 801–812. [Google Scholar] [CrossRef]
- Weinbauer, M.G.; Rassoulzadegan, F. Are viruses driving microbial diversification and diversity? Environ. Microbiol. 2004, 6, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Haaber, J.; Middelboe, M. Viral lysis of Phaeocystis pouchetii: Implications for algal population dynamics and heterotrophic C, N and P cycling. ISME J. 2009, 3, 430–441. [Google Scholar] [CrossRef] [PubMed]
- Shelford, E.J.; Middelboe, M.; Moller, E.F.; Suttle, C.A. Virus-driven nitrogen cycling enhances phytoplankton growth. Aquat. Microb. Ecol. 2012, 66, 41–46. [Google Scholar] [CrossRef]
- Riemann, L.; Middelboe, M. Stability of bacterialand viral community compositions in Danish coastal watersas depicted by DNA fingerprinting techniques. Aquat. Microb. Ecol. 2002, 27, 219–232. [Google Scholar] [CrossRef]
- Cochlan, W.P.; Wikner, J.; Steward, G.F.; Smith, D.C.; Azam, F. Spatial distribution of viruses, bacteria and chlorophyll a in nertic, oceanic and estuarine environments. Mar. Ecol. Prog. Ser. 1993, 92, 77–87. [Google Scholar] [CrossRef]
- Bettarel, Y.; Sime-Ngando, T.; Amblard, C.; Carrias, J.F.; Portelli, C. Virioplankton and microbial communities in aquatic systems: A seasonal study in two lakes of differing trophy. Freshw. Biol. 2003, 48, 810–822. [Google Scholar] [CrossRef]
- Peduzzi, P.; Schiemer, F. Bacteria and viruses in the water column of tropical freshwater reservoirs. Environ. Microbiol. 2004, 6, 707–715. [Google Scholar] [CrossRef]
- Fernandes, A.; Arau’jo, F.; Godinho, M.J.L. Short-term variations of virus-like particles in a tropical lake: Relationship with microbial communities (bacteria, ciliates and flagellates). Microbiol Res. 2009, 164, 411–419. [Google Scholar]
- Tsai, A.Y.; Gong, G.C.; Liu, H. Seasonal variations in virioplankton and picoplankton in semi-enclosed and open coastal waters. Terr. Atmos. Ocean. Sci. 2018, 29, 465–472. [Google Scholar] [CrossRef]
- Mathias, C.B.; Kirschner, K.T.; Velimirov, B. Seasonal variations of virus abundance and viral control of the bacterial population in backwater system of the Danube River. Appl. Environ. Microbiol. 1995, 61, 3734–3740. [Google Scholar] [CrossRef] [PubMed]
- Weinbauer, M.G.; Fuks, D.; Puskaric, S.; Peduzzi, P. Diel, seasonal and depth-related variability of viruses and dissolved DNA in the northern Adriatic Sea. Microb. Ecol. 1995, 30, 25–41. [Google Scholar] [CrossRef] [PubMed]
- Weinbauer, M.G.; Höfle, M.G. Significance of viral lysis and flagellate grazing as factors controlling bacterioplankton production in a eutrophic lake. Appl. Environ. Microbiol. 1998, 64, 431–438. [Google Scholar] [CrossRef] [PubMed]
- Wommack, K.E.; Hill, R.T.; Muller, T.A.; Colwell, R.R. Effects of sunlight on bacteriophage viability and structure. Appl. Environ. Microbiol. 1996, 62, 1336–1341. [Google Scholar] [CrossRef] [PubMed]
- Sime-Ngando, T.; Bettarel, Y.; Chartogne, C.; Sean, K. The imprint of wild viruses on freshwater microbial ecology. Recent Res. Dev. Microbiol. 2003, 7, 481–497. [Google Scholar]
- Magiopoulos, I.; Pitta, P. Viruses in a deep oligotrophic sea: Seasonal distribution of marine viruses in the epi-, meso- and bathypelagic waters of the Eastern Mediterranean Sea. Deep. Sea Res. Part I 2012, 66, 1–10. [Google Scholar] [CrossRef]
- Clasen, J.L.; Brigden, S.M.; Payet, J.P.; Suttle, C.A. Evidence that viral abundance across oceans and lakes is driven by different biological factors. Freshw. Biol. 2008, 53, 1090–1100. [Google Scholar] [CrossRef]
- Parada, V.; Sintes, E.; van Aken, H.M.; Weinbauer, M.G.; Herndl, G.J. Viral abundance, decay and diversity in the meso- and bathypelagic waters of the North Atlantic. Appl. Environ. Microbiol. 2007, 73, 4429–4438. [Google Scholar] [CrossRef]
- Shelford, E.J.; Suttle, C.A. Virus-mediated transfer of nitrogen from heterotrophic bacteria to phytoplankton. Biogeosciences 2018, 15, 809–819. [Google Scholar] [CrossRef]
- Gong, G.C.; Tsai, A.Y. Reduced daytime net growth rate of Synechococcus spp. in the East China Sea in summer estimated using a dilution approach. Estuar. Coast. Shelf Sci. 2019, 219, 90–96. [Google Scholar] [CrossRef]
- Brum, J.R. Concentration, production and turnover of viruses and dissolved DNA pools at Stn ALOHA, North Pacific subtropical gyre. Aquat. Microb. Ecol. 2005, 41, 103–113. [Google Scholar] [CrossRef]
- Granéli, E.; Wallström, K.; Larsson, U.; Granéli, W.; Elmgren, R. Nutrient limitation of primary production in the Baltic Sea area. AMBIO 1990, 19, 142–151. [Google Scholar]
- Middelboe, M. Bacterial growth rate and marine virus–host dynamics. Microb. Ecol. 2000, 40, 114–124. [Google Scholar] [CrossRef] [PubMed]
- Gong, G.C.; Shiah, F.K.; Liu, K.K.; Wen, Y.H.; Liang, M.H. Spatial and temporal variation of chlorophyll a, primary productivity and chemical hydrography in the southern East China Sea. Cont. Shelf. Res. 2000, 20, 411–436. [Google Scholar] [CrossRef]
- Pai, S.C.; Yang, C.C.; Riley, J.P. Effects of acidity and molybdate concentration on the kinetics of the formation of the phosphoantimonyl molybdenum blue complex. Anal. Chim. Acta 1990, 229, 115–120. [Google Scholar]
- Mei, M.L.; Danovaro, R. Virus production and life strategies in aquatic sediments. Limnol. Oceanogr. 2004, 49, 459–470. [Google Scholar] [CrossRef]
- Brussaard, C.P.D. Optimization of procedures for counting viruses by flow cytometry. Appl. Environ. Microbiol. 2004, 70, 1506–1513. [Google Scholar] [CrossRef]
- Hammes, F.; Egli, T. Cytometric methods for measuring bacteria in water: Advantages, pitfalls and applications. Anal. Bioanal. Chem. 2010, 397, 1083–1095. [Google Scholar] [CrossRef]
- Gong, G.C.; Liu, K.K.; Pai, S.-C. Prediction of nitrate concentration from two end member mixing in the southern East China Sea. Cont. Shelf. Res. 1995, 5, 827–842. [Google Scholar] [CrossRef]
- Williamson, S.J.; Paul, J.H. Nutrient stimulation of lytic phage production in bacterial populations of the Gulf of Mexico. Aquat. Microb. Ecol. 2004, 36, 9–17. [Google Scholar] [CrossRef]
- Wilson, W.; Joint, I.; Carr, N.G.; Mann, N.H. The effect of phosphorus status on the kinetics of cyanophage infection in the oceanic cyanobacterium Synechococcus sp. WH7803. J. Phycol. 1996, 32, 506–516. [Google Scholar] [CrossRef]
- Wommack, K.E.; Colwell, R.R. Virioplankton: Viruses in aquatic ecosystems. Microbiol. Mol. Biol. Rev. 2000, 64, 69–114. [Google Scholar] [CrossRef]
- Boehme, J.; Frischer, M.E.; Jiang, S.C.; Kellogg, C.A.; Pichard, S.; Rose, J.B.; Steinway, C.; Paul, J.H. Viruses, bacterioplankton, and phytoplankton in the southeastern Gulf of Mexico: Distribution and contribution to oceanic DNA pools. Mar. Ecol. Prog. Ser. 1993, 97, 1–10. [Google Scholar] [CrossRef]
- Taylor, G.T.; Hein, C.; Iabichella, M. Temporal variations in viral distributions in the anoxic Cariaco Basin. Aquat. Microb. Ecol. 2003, 30, 103–116. [Google Scholar] [CrossRef]
- Maranger, R.; Bird, D.F. Viral abundances in aquatic systems: A comparison between marine and fresh waters. Mar. Ecol. Prog. Ser. 1995, 121, 217–226. [Google Scholar] [CrossRef]
- Weinbauer, M.G.; Brettar, I.; Höfle, M.G. Lysogeny and virus-induced mortality of bacterioplankton in surface, deep, and anoxic marine waters. Limnol. Oceanogr. 2003, 48, 1457–1465. [Google Scholar] [CrossRef]
- Yager, P.L.; Connelly, T.L.; Mortazavi, B.; Wommack, K.E.; Bano, N.; Bauer, J.E.; Opsahl, S.; Hollibaugh, J.T. Dynamic bacterial and viral response to an algal bloom at subzero temperatures. Limnol. Oceanogr. 2001, 46, 790–801. [Google Scholar] [CrossRef]
- Colombet, J.; Sime-Ngando, T.; Cauchie, H.M.; Fonty, G.; Hoffmann, L.; Demeure, G. Depth-related gradients of viral activity in Lake Pavin. Appl. Environ. Microbiol. 2006, 72, 4440–4445. [Google Scholar] [CrossRef][Green Version]
- Wigington, C.H.; Sonderegger, D.; Brussaard, C.P.; Buchan, A.; Finke, J.F.; Fuhrman, J.A.; Lennon, J.T.; Middelboe, M.; Suttle, C.A.; Stock, C.; et al. Re-examination of the relationship between marine virus and microbial cell abundances. Nat. Microbiol. 2016, 1, 15024. [Google Scholar] [CrossRef] [PubMed]
- Laybourn-Parry, J.; Marshall, W.A.; Marchant, H.J. Nutritional versatility as a key to survival in Antarctic phytoflagellates in two contrasting saline lakes. Freshw. Biol. 2005, 50, 830–838. [Google Scholar] [CrossRef]
- Bongiorni, L.; Magagnini, M.; Armeni, M.; Noble, R.; Danovaro, R. Viral production, decay rates, and life strategies along a trophic gradient in the north Adriatic sea. Appl. Environ. Microbiol. 2005, 71, 6644–6650. [Google Scholar] [CrossRef]
- Hara, S.; Koike, I.; Terauchi, K.; Kamiya, H.; Tanoue, E. Abundance of viruses in deep oceanic waters. Mar. Ecol. Prog. Ser. 1996, 145, 269–277. [Google Scholar] [CrossRef]
- Tsai, A.Y.; Gong, G.C.; Chao, C.F. Contribution of viral lysis and nanoflagellate grazing to bacterial mortality at surface waters and deeper depths in the coastal ecosystem of subtropical western Pacific. Estuaries Coasts 2016, 39, 1357–1366. [Google Scholar] [CrossRef]
- Bettarel, Y.; Sime-Ngando, T.; Amblard, C.; Dolan, J. Viral activity in two contrasting lake ecosystems. Appl. Environ. Microbiol. 2004, 70, 2941–2951. [Google Scholar] [CrossRef]
- Pradeep Ram, A.S.; Nishimura, Y.; Tomaru, Y.; Nagasaki, K.; Nagata, T. Seasonal variation in viral-induced mortality of bacterioplankton in the water column of a large mesotrophic lake (Lake Biwa, Japan). Aquat. Microb. Ecol. 2010, 58, 249–259. [Google Scholar] [CrossRef]
- Middelboe, M.; Jorgensen, N.O.G. Viral lysis of bacteria: An important source of dissolved amino acids and cell wall compounds. J. Mar. Biol. Assoc. UK 2006, 86, 605–612. [Google Scholar] [CrossRef]
| Temperature | PO4 | Chl a | Bacterial Abundance | VLPs Abundance | VBR | VLPs Production | Net Growth Rate in 20% Diluted Water | Net Growth Rate in <2 μm Filtered Water | Contribution of VLPs to Bacterial Growth (%) | |
|---|---|---|---|---|---|---|---|---|---|---|
| (°C) | (μM) | (mg m−3) | (105 cells ml−1) | (106 viruses ml−1) | (106 viruses ml−1 h−1) | (h−1) | (h−1) | (%) | ||
| Surface water | 25.2 | 0.11 | 0.375 | 0.8±0.3 | 1.8 ± 0.4 | 22.5 ± 2.1 | 0.222 | 0.10 ± 0.02 | 0.12±0.05 | 17% |
| Deep water | 17.3 | 0.58 | nd | 0.4±0.1 | 1.4 ± 0.3 | 35 ± 5.6 | 0.068 | 0.01 ± 0.005 | 0.05±0.02 | 80% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsai, A.-Y.; Lin, Y.-T.; Gong, G.-C. Effect of the Presence of Virus-like Particles on Bacterial Growth in Sunlit Surface and Dark Deep Ocean Environments in the Southern East China Sea. Water 2021, 13, 2934. https://doi.org/10.3390/w13202934
Tsai A-Y, Lin Y-T, Gong G-C. Effect of the Presence of Virus-like Particles on Bacterial Growth in Sunlit Surface and Dark Deep Ocean Environments in the Southern East China Sea. Water. 2021; 13(20):2934. https://doi.org/10.3390/w13202934
Chicago/Turabian StyleTsai, An-Yi, Ying-Tsong Lin, and Gwo-Ching Gong. 2021. "Effect of the Presence of Virus-like Particles on Bacterial Growth in Sunlit Surface and Dark Deep Ocean Environments in the Southern East China Sea" Water 13, no. 20: 2934. https://doi.org/10.3390/w13202934
APA StyleTsai, A.-Y., Lin, Y.-T., & Gong, G.-C. (2021). Effect of the Presence of Virus-like Particles on Bacterial Growth in Sunlit Surface and Dark Deep Ocean Environments in the Southern East China Sea. Water, 13(20), 2934. https://doi.org/10.3390/w13202934






