Taxonomic Diversity of Fungi and Bacteria in Azoé-NP® Vertical Flow Constructed Wetlands
Abstract
:1. Introduction
2. Materials and Methods
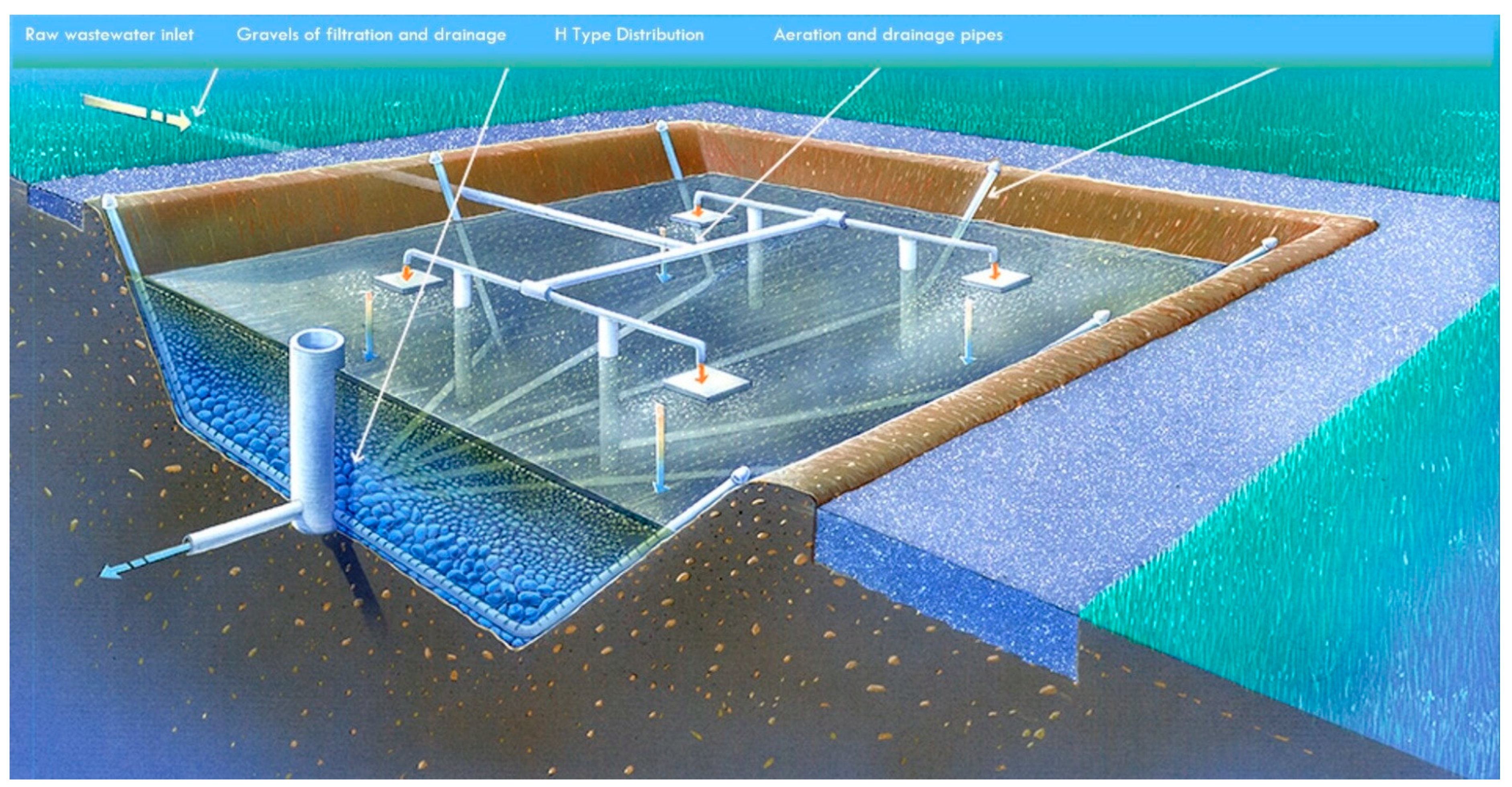
2.1. Treatment Plants and Sampling
2.2. DNA Extraction and Sequencing
2.3. Bioinformatic Analysis
3. Results
3.1. Chemical Analysis
3.2. Taxonomic Analysis of Bacterial Community
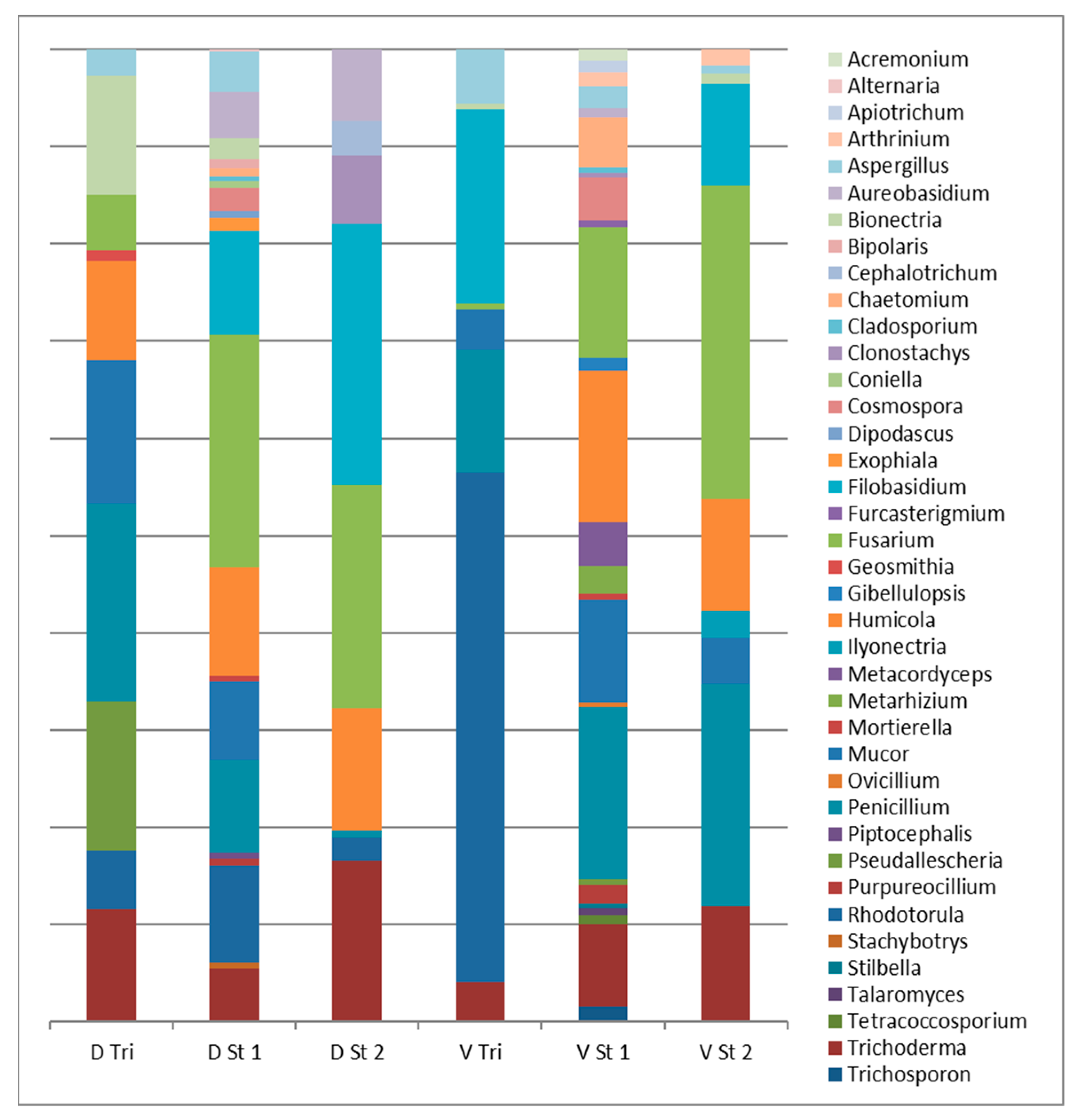
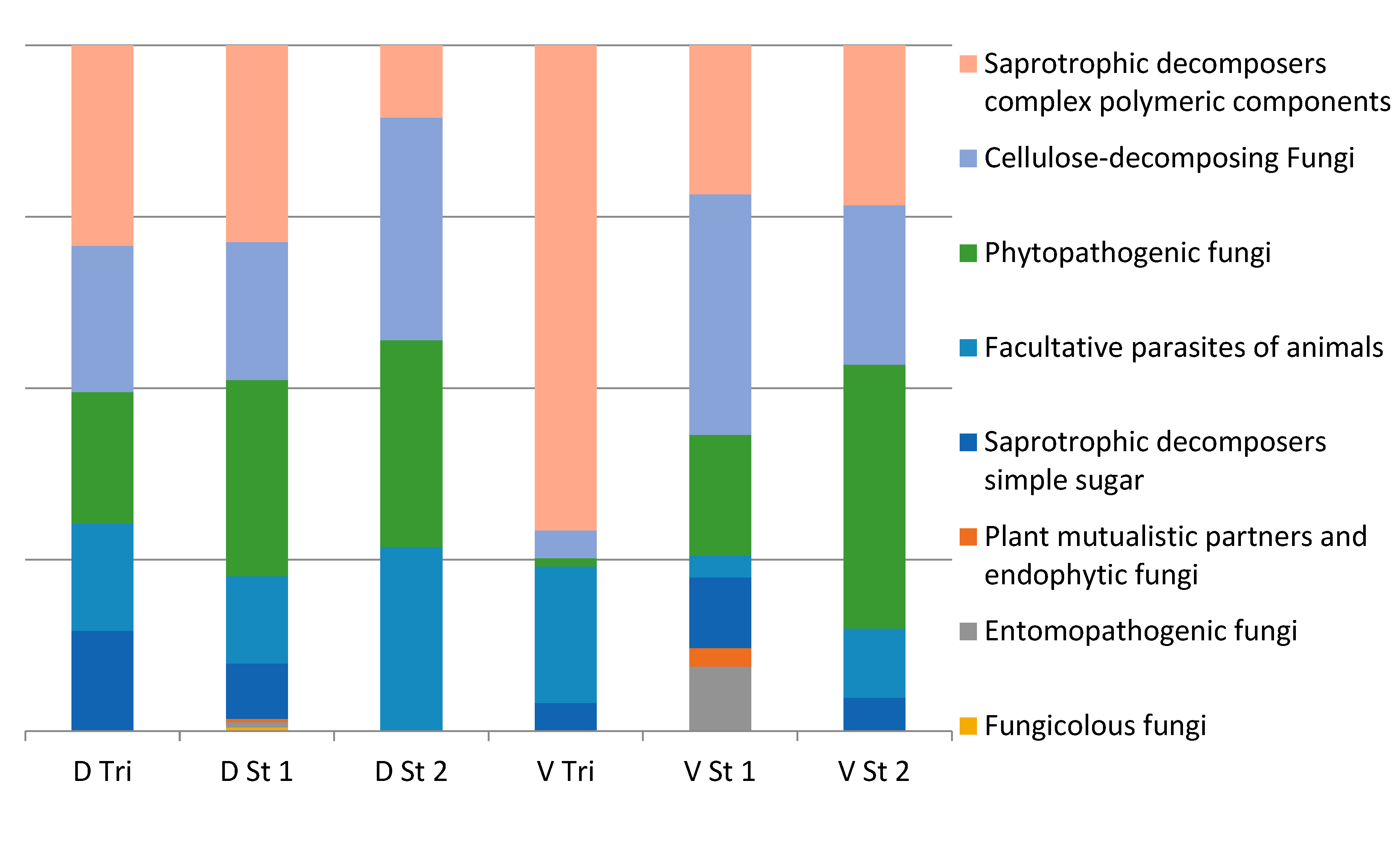
3.3. Fungi Biodiversity
4. Discussion
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Waqas, S.; Bilad, M.R.; Man, Z.B.; Suleman, H.; Nordin, N.A.H.; Jaafar, J.; Othman, M.H.D.; Elma, M. An Energy-Efficient Membrane Rotating Biological Contactor for Wastewater Treatment. J. Clean. Prod. 2021, 282, 124544. [Google Scholar] [CrossRef]
- Wang, C.; Huang, R.; Sun, R.; Yang, J.; Sillanpää, M. A review on persulfates activation by functional biochar for organic contaminants removal: Synthesis, characterizations, radical determination, and mechanism. J. Environ. Chem. Eng. 2021, 9, 106267. [Google Scholar] [CrossRef]
- Dang, J.; Wang, H.; Wang, C. Adsorption of Toxic Zinc by Functionalized Lignocellulose Derived from Waste Biomass: Kinetics, Isotherms and Thermodynamics. Sustainability 2021, 13, 10673. [Google Scholar] [CrossRef]
- Gourdon, R.; Kania, M.; Gautier, M.; Kim, B.; Michel, P. Treatment of domestic wastewater from small cities on vertical flow constructed wetlands (VFCWs). In Proceedings of the 4th Congrès International de Géotechnique—Ouvrages -Structures, Ho Chi Minh City, Vietnam, 26–27 October 2017; pp. 1066–1073. [Google Scholar] [CrossRef]
- Abou-Elela, S.I.; Hellal, M. Municipal wastewater treatment using vertical flow constructed wetlands planted with Canna, Phragmites and Cyprus. Ecol. Eng. 2012, 47, 209–213. [Google Scholar] [CrossRef]
- Torrens, A.; Molle, P.; Boutin, C.; Salgot, M. Impact of design and operation variables on the performance of vertical-flow constructed wetlands and intermittent sand filters treating pond effluent. Water Res. 2009, 43, 1851–1858. [Google Scholar] [CrossRef]
- Molle, P.; Liénard, A.; Grasmick, A.; Iwema, A. Effect of reeds and feeding operations on hydraulic behaviour of vertical flow constructed wetlands under hydraulic overloads. Water Res. 2006, 40, 606–612. [Google Scholar] [CrossRef]
- Brix, H.; Arias, C.A. The use of vertical flow constructed wetlands for on-site treatment of domestic wastewater: New Danish guidelines. Ecol. Eng. 2005, 25, 491–500. [Google Scholar] [CrossRef]
- Kadlec, R.; Knight, R.; Vymazal, J.; Brix, H.; Cooper, P.; Haberl, R. Constructed Wetlands for Pollution Control; IWA Publishing: London, UK, 2000. [Google Scholar]
- Kim, B.; Gautier, M.; Prost-Boucle, S.; Molle, P.; Michel, P.; Gourdon, R. Performance evaluation of partially saturated vertical-flow constructed wetland with trickling filter and chemical precipitation for domestic and winery wastewaters treatment. Ecol. Eng. 2014, 71, 41–47. [Google Scholar] [CrossRef] [Green Version]
- Molle, P. French vertical flow constructed wetlands: A need of a better understanding of the role of the deposit layer. Water Sci. Technol. 2013, 69, 106–112. [Google Scholar] [CrossRef]
- Molle, P.; Liénard, A.; Boutin, C.; Merlin, G.; Iwema, A. How to treat raw sewagewith constructed wetlands: An overview of the French systems. Water Sci. Technol. 2005, 51, 11–21. [Google Scholar] [CrossRef]
- Stottmeister, U.; Wießner, A.; Kuschk, P.; Kappelmeyer, U.; Kästner, M.; Bederski, O.; Müller, R.A.; Moormann, H. Effects of plants and microorganisms in constructed wetlands for wastewater treatment. Biotechnol. Adv. 2003, 22, 93–117. [Google Scholar] [CrossRef]
- Faulwetter, J.L.; Gagnon, V.; Sundberg, C.; Chazarenc, F.; Burr, M.D.; Brisson, J.; Camper, A.K.; Stein, O.R. Microbial processes influencing performance of treatment wetlands: A review. Ecol. Eng. 2009, 35, 987–1004. [Google Scholar] [CrossRef]
- Scholz, M.; Lee, B.H. Constructed wetlands: A review. Int. J. Environ. Res. 2005, 62, 1256–1261. [Google Scholar] [CrossRef]
- Huang, D.L.; Zeng, G.M.; Feng, C.L.; Hu, S.; Jiang, X.Y.; Tang, L.; Su, F.F.; Zhang, Y.; Zeng, W.; Liu, H.L. Degradation of lead-contaminated lignocellulosic waste by Phanerochaete chrysosporium and the reduction of lead toxicity. Environ. Sci. Technol. 2008, 42, 4946–4951. [Google Scholar] [CrossRef]
- Cheng, M.; Huang, C.; Wu, H.P.; Qin, L. Synthesis of surface molecular imprinted TiO2/graphene photocatalyst and its highly efficient photocatalytic degradation of target pollutant under visible light irradiation. Appl. Surf. Sci. 2016, 390, 368–376. [Google Scholar]
- Wallace, S.; Austin, D. Emerging models for nitrogen removal in treatment wetlands. J. Environ. Health 2008, 71, 10–16. [Google Scholar] [PubMed]
- Oehl, F.; Sieverding, E.; Dubois, D.; Ineichen, K.; Boller, T.; Wiemken, A. Impact of long-term conventional and organic farming on the diversity of arbuscular mycorrhizal fungi. Oecologia 2004, 138, 574–583. [Google Scholar] [CrossRef]
- Samer, M. Biological and chemical wastewater treatment processes. Wastewater Treat. Eng. 2015, 150, 1–50. [Google Scholar] [CrossRef] [Green Version]
- Deshmukh, R.; Khardenavis, A.A.; Purohit, H.J. Diverse Metabolic Capacities of Fungi for Bioremediation. Indian J. Microbiol. 2016, 56, 247–264. [Google Scholar] [CrossRef] [Green Version]
- Singh, R.K.; Tripathi, R.; Ranjan, A.; Srivastava, A.K. Fungi as potential candidates for bioremediation. In Abatement of Environmental Pollutants: Trends and Strategies; Singh, P., Kumar, A., Borthakur, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2020; pp. 177–191. [Google Scholar] [CrossRef]
- Gazem, M.A.H.; Nazareth, S. Sorption of lead and copper from an aqueous phase system by marine-derived Aspergillus species. Ann. Microbiol. 2013, 63, 503–511. [Google Scholar] [CrossRef]
- Singh, M.P.; Vishwakarma, S.K.; Srivastava, A.K. Bioremediation of Direct Blue 14 and Extracellular Ligninolytic Enzyme Production by White Rot Fungi: Pleurotus Spp. BioMed Res. Int. 2013, 2013, 1–4. [Google Scholar] [CrossRef] [Green Version]
- Pietro-Souza, W.; de Campos Pereira, F.; Mello, I.S.; Stachack, F.F.F.; Terezo, A.J.; da Cunha, C.N.; Whitee, J.F.; Lif, H.; Soares, M.A. Mercury resistance and bioremediation mediated by endophytic fungi. Chemosphere 2020, 240, 124–874. [Google Scholar] [CrossRef] [PubMed]
- Hu, B.; Hu, S.; Chen, Z.; Vymazal, J. Employ of arbuscular mycorrhizal fungi for pharmaceuticals ibuprofen and diclofenac removal in mesocosm-scale constructed wetlands. J. Hazard. Mater. 2021, 409, 124524. [Google Scholar] [CrossRef] [PubMed]
- Viga, T.K.R.; Aguilar, R.; Castillo-Argüero, S.; Chiappa-Carrara, X.; Guadarrama, P.; Ramos-Zapata, J. Wetland plant species improve performance when inoculated with arbuscular mycorrhizal fungi: A meta-analysis of experimental pot studies. Mycorrhiza 2018, 28, 477–493. [Google Scholar] [CrossRef] [PubMed]
- Kendrick, B. Fungi: Ecological importance and impact on humans. eLS 2011. [Google Scholar] [CrossRef]
- Rousk, J.; Bååth, E. Growth of saprotrophic fungi and bacteria in soil. FEMS Microbiol. Ecol. 2011, 78, 17–30. [Google Scholar] [CrossRef] [Green Version]
- Shchegolkova, N.; Krasnov, G.S.; Belova, A.A.; Dmitriev, A.; Kharitonov, S.; Klimina, K.; Melnikova, N.V.; Kudryavtseva, A. Microbial Community Structure of Activated Sludge in Treatment Plants with Different Wastewater Compositions. Front. Microbiol. 2016, 7, 90. [Google Scholar] [CrossRef] [Green Version]
- Crous, P.W.; Verkley, G.J.M.; Groenewald, J.Z.; Samson, R.A. Fungal Biodiversity; CBS Laboratory Manual Series; Westerdijk Fungal Biodiversity Institute: Utrecht, The Netherlands, 2009; p. 269. [Google Scholar]
- Domsch, K.H.; Gams, W.; Anderson, T. Compendium of Soil Fungi, 2nd ed.; IHW-Verlag: Ehing, Germany, 2007; p. 672. [Google Scholar]
- Gardes, M.; Bruns, T.D. ITS primers with enhanced specificity for basidiomycetes—Application to the identification of mycorrhizae and rusts. Mol. Ecol. 1993, 2, 113–118. [Google Scholar] [CrossRef]
- Manter, D.K.; Vivanco, J.M. Use of the ITS primers, ITS1F and ITS4, to characterize fungal abundance and diversity in mixed-template samples by qPCR and length heterogeneity analysis. J. Microbiol. Methods 2007, 71, 7–14. [Google Scholar] [CrossRef]
- Callahan, B.J.; Mcmurdie, P.J.; Rosen, M.J.; Han, A.W.; Johnson, A.J.A.; Holmes, S.P. DADA2: High-resolution sample inference from Illumina amplicon data. Nat. Methods 2016, 13, 581–583. [Google Scholar] [CrossRef] [Green Version]
- Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet. J. 2011, 17, 10–12. [Google Scholar] [CrossRef]
- Mulder, M.; Radjabzadeh, D.; Jong, J.C.K.-D.; Uitterlinden, A.G.; Kraaij, R.; Stricker, B.H.; Verbon, A. Long-term effects of antimicrobial drugs on the composition of the human gut microbiota. Gut Microbes 2020, 12, 1795492. [Google Scholar] [CrossRef] [PubMed]
- Maciejewski, K.; Gautier, M.; Kim, B.; Michel, P.; Gourdon, R. Effect of trickling filter on carbon and nitrogen removal in vertical flow treatment wetlands: A full-scale investigation. J. Environ. Manag. 2022, 303, 114159. [Google Scholar] [CrossRef] [PubMed]
- Chi, Z.; Hou, L.; Li, H. Effects of pollution load and salinity shock on nitrogen removal and bacterial community in two-stage vertical flow constructed wetlands. Bioresour. Technol. 2021, 342, 126031. [Google Scholar] [CrossRef]
- Calheiros, C.S.C.; Pereira, S.I.A.; Franco, A.R.; Castro, P.M.L. Diverse Arbuscular Mycorrhizal Fungi (AMF) Communities Colonize Plants Inhabiting a Constructed Wetland for Wastewater Treatment. Water 2019, 11, 1535. [Google Scholar] [CrossRef] [Green Version]
- Xu, Z.; Ban, Y.; Jiang, Y.; Zhang, X.; Liu, X. Arbuscular Mycorrhizal Fungi in Wetland Habitats and Their Application in Constructed Wetland: A Review. Pedosphere 2016, 26, 592–617. [Google Scholar] [CrossRef]
- Hu, S.; Chen, Z.; Vosátka, M.; Vymazal, J. Arbuscular mycorrhizal fungi colonization and physiological functions toward wetland plants under different water regimes. Sci. Total Environ. 2020, 716, 137040. [Google Scholar] [CrossRef]
- Frąc, M.; Hannula, S.E.; Bełka, M.; Jędryczka, M. Fungal Biodiversity and Their Role in Soil Health. Front. Microbiol. 2018, 9, 707. [Google Scholar] [CrossRef] [Green Version]
- Naranjo-Ortiz, M.A.; Gabaldón, T. Fungal evolution: Major ecological adaptations and evolutionary transitions. Biol. Rev. 2019, 94, 1443–1476. [Google Scholar] [CrossRef] [Green Version]
- Divya, L.M.; Prasanth, G.K.; Sadasivan, C. Potential of the salt-tolerant laccase-producing strain Trichoderma viride Pers. NFCCI-2745 from an estuary in the bioremediation of phenol-polluted environments. J. Basic Microbiol. 2013, 54, 542–547. [Google Scholar] [CrossRef]
- Vacondio, B.; Birolli, W.; Ferreira, I.M.; Seleghim, M.H.; Gonçalves, S.; Vasconcellos, S.P.; Porto, A.L.M. Biodegradation of pentachlorophenol by marine-derived fungus Trichoderma harzianum CBMAI 1677 isolated from ascidian Didemnun ligulum. Biocatal. Agric. Biotechnol. 2015, 4, 266–275. [Google Scholar] [CrossRef]
- Hickey, P. Toxicity of water-soluble fractions of crude oil on some bacteria and fungi Isolated from marine water. Am. J. Anim. Res. 2013, 3, 24–29. [Google Scholar]
- Maruthi, Y.; Hossain, K.; Thakre, S. Aspergillus flavus: A potential Bioremediator for oil contaminated soils. Eur. J. Sustain. Dev. 2013, 2, 57. [Google Scholar] [CrossRef] [Green Version]
- Chang, Y.-T.; Lee, J.-F.; Liu, K.-H.; Liao, Y.-F.; Yang, V. Immobilization of fungal laccase onto a nonionic surfactant-modified clay material: Application to PAH degradation. Environ. Sci. Pollut. Res. 2016, 23, 4024–4035. [Google Scholar] [CrossRef]
- Balaji, V.; Arulazhagan, P.; Ebenezer, P. Enzymatic bioremediation of polyaromatic hydrocarbons by fungal consortia enriched from petroleum contaminated soil and oil seeds. J. Environ. Biol. 2014, 35, 521–529. [Google Scholar]
- Lladó, S.; Covino, S.; Solanas, A.; Viñas, M.; Petruccioli, M.; D’Annibale, A. Comparative assessment of bioremediation approaches to highly recalcitrant PAH degradation in a real industrial polluted soil. J. Hazard. Mater. 2013, 248, 407–414. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bennett, R.; Cordero, P.R.; Bautista, G.S.; Dedeles, G.R. Reduction of hexavalent chromium using fungi and bacteria isolated from contaminated soil and water samples. Chem. Ecol. 2013, 29, 320–328. [Google Scholar] [CrossRef]
- Sousa, N.R.; Ramos, M.A.; Marques, A.P.; Castro, P.M. A genotype dependent-response to cadmium contamination in soil is displayed by Pinus pinaster in symbiosis with different mycorrhizal fungi. Appl. Soil Ecol. 2014, 76, 7–13. [Google Scholar] [CrossRef]
- Krishnamurthy, Y.L.; Naik, B.S. Endophytic fungi bioremediation. In Endophytes: Crop Productivity and Protection; Maheshwari, D.K., Annapurna, K., Eds.; Springer: Cham, Switzerland, 2017; pp. 47–60. [Google Scholar] [CrossRef]
- Michielse, C.B.; Rep, M. Pathogen profile update: Fusarium oxysporum. Mol. Plant Pathol. 2009, 10, 311–324. [Google Scholar] [CrossRef]
| Station | Sample | TSS | BOD5 | COD | NH4-N | NO3-N | TN |
|---|---|---|---|---|---|---|---|
| Vercia (V) Azoé-NP® | Inflow | 517.1 | 302.7 | 805 | 31.6 | 1.9 | 74.9 |
| Outflow | 4.9 | 7.2 | 33.3 | 12.9 | 12.3 | 16.6 | |
| St Désert (D) Azoé-NP® | Inflow | 258.6 | 205 | 541.4 | 38.5 | 0.5 | 60.5 |
| Outflow | 5.8 | 3 | 16.2 | 1.5 | 10.8 | 13.6 |
| Taxon | D Inflow | D Tri | D St 1 | D St 2 | D Outflow | V Inflow | V Tri | V St 1 | V St 2 | V Outflow |
|---|---|---|---|---|---|---|---|---|---|---|
| Acidobacteria | 12 | 1669 | 2266 | 987 | 51 | 13 | 110 | 4266 | 4400 | 124 |
| Actinobacteria | 4817 | 161 | 639 | 418 | 193 | 2703 | 659 | 1135 | 1099 | 1662 |
| Bacteroidetes | 2077 | 5959 | 4150 | 941 | 5066 | 20,167 | 11,651 | 2196 | 5551 | 3246 |
| Chlamydiae | 2 | 21 | 26 | 0 | 18 | 4 | 0 | 138 | 45 | 19 |
| Chloroflexi | 7 | 4144 | 1696 | 314 | 38 | 4 | 0 | 2162 | 3399 | 87 |
| Cyanobacteria | 7 | 426 | 88 | 2 | 28 | 23 | 10,915 | 25 | 291 | 60 |
| Dependentiae | 3 | 106 | 30 | 4 | 3 | 0 | 2 | 45 | 46 | 17 |
| Epsilonbacteraeota | 334 | 4873 | 16 | 3 | 153 | 1723 | 52 | 27 | 174 | 211 |
| Euryarchaeota | 353 | 19 | 0 | 0 | 5 | 124 | 0 | 4 | 15 | 64 |
| Firmicutes | 32,995 | 190 | 132 | 8 | 465 | 12,462 | 43 | 286 | 911 | 2210 |
| Fusobacteria | 801 | 0 | 0 | 0 | 4 | 184 | 0 | 0 | 8 | 4 |
| Gemmatimonadetes | 0 | 274 | 1154 | 12 | 0 | 0 | 2 | 1140 | 859 | 3 |
| Hydrogenedentes | 0 | 159 | 176 | 8 | 0 | 8 | 2 | 17 | 197 | 0 |
| Kiritimatiellaeota | 4 | 216 | 21 | 3 | 7 | 31 | 0 | 25 | 337 | 72 |
| Latescibacteria | 0 | 206 | 58 | 24 | 0 | 0 | 0 | 1109 | 370 | 0 |
| Nitrospirae | 0 | 47 | 294 | 404 | 3 | 0 | 0 | 1018 | 487 | 7 |
| Patescibacteria | 202 | 3031 | 454 | 48 | 405 | 570 | 193 | 813 | 1587 | 1863 |
| Planctomycetes | 12 | 845 | 981 | 402 | 40 | 0 | 7 | 1834 | 3942 | 73 |
| Proteobacteria | 37,326 | 36,941 | 12,926 | 4915 | 64,974 | 57,231 | 21,270 | 8497 | 19,896 | 5634 |
| Synergistetes | 456 | 0 | 0 | 0 | 4 | 98 | 0 | 0 | 0 | 85 |
| Thaumarchaeota | 0 | 2 | 16 | 72 | 40 | 0 | 3 | 1194 | 333 | 0 |
| Verrucomicrobia | 1986 | 955 | 1315 | 345 | 207 | 769 | 3289 | 1386 | 2966 | 584 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kharitonov, S.; Shchegolkova, N.; Alexandrova, A.; Saynchuk, A.; Michel, P.; Maciejewski, K.; Gautier, M.; Gourdon, R.; Semenov, M.; Krasnov, G. Taxonomic Diversity of Fungi and Bacteria in Azoé-NP® Vertical Flow Constructed Wetlands. Water 2022, 14, 698. https://doi.org/10.3390/w14050698
Kharitonov S, Shchegolkova N, Alexandrova A, Saynchuk A, Michel P, Maciejewski K, Gautier M, Gourdon R, Semenov M, Krasnov G. Taxonomic Diversity of Fungi and Bacteria in Azoé-NP® Vertical Flow Constructed Wetlands. Water. 2022; 14(5):698. https://doi.org/10.3390/w14050698
Chicago/Turabian StyleKharitonov, Sergey, Nataliya Shchegolkova, Alina Alexandrova, Alexandra Saynchuk, Philippe Michel, Kevin Maciejewski, Mathieu Gautier, Remy Gourdon, Mikhail Semenov, and George Krasnov. 2022. "Taxonomic Diversity of Fungi and Bacteria in Azoé-NP® Vertical Flow Constructed Wetlands" Water 14, no. 5: 698. https://doi.org/10.3390/w14050698
APA StyleKharitonov, S., Shchegolkova, N., Alexandrova, A., Saynchuk, A., Michel, P., Maciejewski, K., Gautier, M., Gourdon, R., Semenov, M., & Krasnov, G. (2022). Taxonomic Diversity of Fungi and Bacteria in Azoé-NP® Vertical Flow Constructed Wetlands. Water, 14(5), 698. https://doi.org/10.3390/w14050698







