Abstract
UV/H2O2 process and TiO2-based photocatalysis were studied in the present work. The effectiveness of these methods was tested in the treatment of effluents taken from handicraft factories. Microorganisms, dyes, and different organic pollutants were detected in the industrial effluents. The experimental procedure for the wastewater treatment was carried out in a patented sunlight reactor on a pilot plant scale. From this study, UV/H2O2 was found to be the best treatment for dye elimination. The optimal peroxide dosage for the degradation of dyes and the elimination of bacteria was 0.07 M. In this case, 70.80% of discoloration was achieved after 7 h of sunlight exposure, under an average sunlight intensity of 3.42 W/m2. The photocatalytic treatment based on TiO2 achieved the highest elimination of coliform bacteria and the lowest TOC value; however, the presence of this material in the reactor had a detrimental effect on the overall elimination of dyes. A combination of both UV/H2O2 and TiO2 treatments significantly improves the dyes discoloration, the elimination of bacteria, and the organic compounds degradation. Some of the results of this study were presented at the 4th Congreso Colombiano de Procesos Avanzados de Oxidación, 4CCPAOx.
1. Introduction
Wastewater coming from small-scale industries currently represents an important source of environmental pollution in Latin American countries. These effluents must be treated before releasing them into the drain, but in the real context of small industries, it is not a priority [1]. Many conventional treatments for industrial effluents can be very expensive [2] and in some cases not effective enough.
Advanced oxidation processes (AOPs) are alternatives to traditional treatments. These processes can use sunlight as an energy source for oxidant species production, thus reducing the global economic cost for treatment or recovery of industrial effluents [3]. AOPs include methods such as UV/O3, UV/H2O2, Fenton, photo-Fenton, non-thermic plasma, sonolysis, and heterogeneous photocatalysis, among others.
TiO2-based photocatalysts has been evaluated around the world [4]; however, this heterogeneous process has different disadvantages mainly related to semiconductor recovering at the end of the treatments [5]. The potential toxicity of this oxide must also be considered in its application on a large scale [6]. This high energy gap (3.23 eV) material needs ultraviolet light to be excited to produce hydroxyl radicals [7], but sunlight only consists of 5% ultraviolet light [8], resulting in a challenge for using TiO2 under environmental conditions. However, in the present study we selected this oxide only as a reference material to achieve the removal of heavy metals by photochemical reduction.
Homogeneous AOPs currently represent a suitable alternative to heterogeneous processes. Different authors have evaluated UV/H2O2 for water treatment and degradation of lab-prepared solutions containing pollutants [9,10,11].
D.H. Tseng et al. [12] evaluated the effect of the addition of oxygen and/or H2O2 in the photocatalytic degradation of monochlorobenzene in a TiO2 aqueous suspension. These authors found that a lower H2O2 dosage acted as an electron acceptor to enhance the degradation efficiency of this molecule. The highest H2O2 content inside the reaction medium had a detrimental effect on photodegradation, mainly due to the capture of •OH radicals and the competitive adsorption of peroxide. T. Hirakawa et al. [13] have also demonstrated the positive effect of peroxide addition over the efficiency of •OH radical generation on the TiO2 surface.
As widely noted, the electron-hole pair lifetime is very short, and the recombination process of these species can reduce photocatalytic efficiency [14]. Different alternatives have been employed to avoid the recombination; for example, oxygen and/or H2O2 can act as electron collectors, thus improving the electron-hole separation [15]. In addition, new •OH radicals can be produced by interaction of H2O2 with photoinduced electrons and oxygen. This results in a better photocatalytic performance, but a suitable concentration of peroxide should be combined with a semiconductor photocatalyst inside the reactor [16].
AOPs can be very effective in the elimination of bacteria and organic pollutants at the laboratory scale; however, it is very important to study the effectiveness of these processes in the treatment of high volumes of industrial effluents [17,18]. Different researchers such as Mueses et al. [19] have studied the reactor design based on hydrodynamics, molecular adsorption processes, mass transfer of reactants and oxidants (oxygen, H2O2, O3, etc.), and other variables. They reported the potential of heterogeneous photocatalysis in the treatment of wastewater.
On the other hand, researchers around the world have been interested in the design of solar photocatalytic reactors for water treatment (PWT) since it appears as a logical extension of solar thermal collectors [20]. It was found that thin film slurries are the most suitable geometry for the optimum utilization of the incident photons in PWTs. In these reactors, the TiO2 suspensions were forced into a liquid distribution system and allowed to freefall through a vertically mounted hollow column. Likewise, the study of PWT has demonstrated that a misbalance between the radiation field and fluid residence time leads to inefficient reactors. This is because photocatalytic processes are kinetically slow and need long periods of time for total pollutant elimination from water samples [21,22,23]. So, in order to obtain greater efficiency in water treatment, UV PWT reactors with serpentine-like geometry have been developed.
In the present study, the effectiveness of UV/H2O2 and TiO2 photocatalysis was evaluated. The novelty of this research was focused on the use of effluents coming from small Colombian producers of handicrafts and stained natural fibers. The photocatalytic treatment of these effluents at the pilot plant scale was carried out under sunlight.
2. Materials and Methods
Wastewater treatment was carried out in a sunlight reactor using a patented design (Figure 1) with a 120 L capacity [24]. The main operation parameters for this pilot plant scale reactor are listed as follows: volumetric flow rate 0.50 L/s, length 22.5 m, and a total residence time of 36 s.

Figure 1.
Sunlight reactor for wastewater treatment.
For these assays, 30 L of industrial effluents coming from local handicrafts factories (Location: Nobsa, Boyacá, Colombia, coordinates 5°46′11″ N, 72°56′24″ O) was used. The total reaction time was 7 h, which was selected based on the total hours of sunlight per day available in the study area.
Firstly, photolysis tests under direct sunlight and without peroxide or titania in the reaction medium were carried out. Then, the homogeneous treatment combining UV light and H2O2 (UV/H2O2) was evaluated. For these wastewater treatment tests, the total volume of polluted water used an H2O2 concentration that was set to 0.05 and 0.07 M.
In the case of the photocatalytic tests, 0.50 g/L of commercial TiO2 (Sigma Aldrich, assay 99.7%) was used. The main physicochemical properties of this oxide were: 3.23 eV as the band gap value and 51 m2/g for the specific surface area; only the anatase phase was detected in this material.
In the search for the best results, combined treatments were evaluated, i.e., TiO2 and UV-H2O2 (0.07 M peroxide).
In order to evaluate the overall effectiveness of the treatments applied, total coliform concentration, dyestuff degradation, and total organic carbon (TOC) elimination were evaluated every hour.
Sampling and evaluation of the analytic parameters were carried out by following the Standard Methods for the Examination of Water and Wastewater [25]. The bacteria content was calculated after taking the initial concentration in UFC for each experiment as a reference. The bacterial elimination was calculated using Equation (1):
where:
Y = (UFC at the end of the reaction)/(UFC Initial) × 100
Y = % bacteria content at the end of the reaction.
Different samples were taken from the reactor every hour for 7 h. The discoloration percentage was measured by UV-Vis spectrophotometry in a Thermo Scientific Evolution 300 spectrophotometer. The wavelengths evaluated were between 300 and 450 nm, where the highest absorption of dye was observed. The discoloration percentage was calculated by using Equation (2).
where:
Discoloration (%) = (A − B)/A × 100
A = Absorbance value of the initial sample at 450 nm.
B = Absorbance value of sample after the determined reaction time.
TOC analysis was performed in a TOC/TNb multi N/C 2100 analyzer, and bacteria were identified using the membrane filter technique for the coliform group (9222 method) [25]. In order to ensure the reproducibility and reliability of the reported results, all assays and reactions were carried out twice with the arithmetic average and standard deviation reported in the results.
3. Results and Discussion
3.1. Bacteria Elimination
Figure 2 represents the bacteria elimination percentage as a function of the reaction time. From this figure, it is possible to note the bactericidal effect of direct sunlight, where the total coliform bacteria content decreases almost 20%. It is important to take into account that under the weather conditions of this study, the average sunlight intensity oscillates in a day (between 0.60 and 13.70 W/m2). It is also important to note that different tests were performed on a laboratory scale which had similar results to those observed at the pilot plant scale. The bactericidal effect of UV light by itself is mainly due to its absorption by cellular components called intracellular chromophores, such as L–tryptophan. In the presence of oxygen, UV irradiation contributes to the generation of reactive oxygen species (ROS), such as •OH, and , which induces oxidative stresses that damage the cell membranes and components leading to the death of the bacteria [26,27].
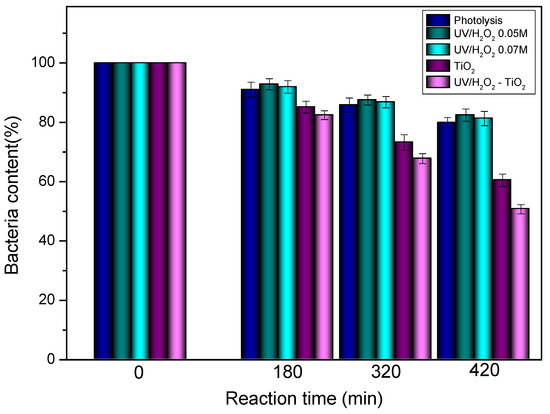
Figure 2.
Total coliform bacteria percentage in handicraft factory effluents under different AOPs.
On the other hand, when 0.05 M H2O2 was added into the reaction medium, the bactericidal effect slightly decreased; however, this difference can be inside of the error percentage of this analysis. After 0.05 M H2O2 addition, peroxide itself contributes to bacterial damage; in addition, this compound can also contribute to the ROS generation under illumination, thus leading to a slight improvement in bacteria elimination (Figure 2).
As also seen in Figure 2, heterogeneous photocatalysis induced by TiO2 is the more effective alternative for reducing bacteria concentration in the effluents. This resulted in removing 40% of the bacteria after 7 h of treatment. Different authors have reported that TiO2 photocatalysis promotes the peroxidation of the E. coli membrane phospholipids and induces major disorders in the cell membrane which could be the main mechanism for killing bacteria using this treatment [28]. Previous publications showed the destruction of bacterial cells during photocatalytic treatment using high-resolution microscopy [29,30]. The effectiveness of TiO2 in E. coli inactivation can also be related to different factors such as the size and shape of these microorganisms (this is because the bacteria are more than 30 times larger than TiO2 particles), concentration of TiO2 and microorganisms, nature, and intensity of UV irradiation [31].
UV-H2O2 and TiO2 combined processes were also evaluated. In general, it was observed that the overall effectiveness of the treatment significantly increases, thus leading to a decrease in the bacteria content of 51%. This could be caused by the addition of peroxide, which can contribute to decreasing the electron-hole recombination during the photocatalytic process on the titania surface. This is the result of photogenerated charges that are used for the production of •OH radicals as seen in other studies [13,32,33,34]: (i) these radicals are formed by the reduction of H2O2 with conduction band electrons; and (ii) furthermore, H2O2 is oxidized to O2•− by the valence band hole. These factors lead to improvement in photocatalytic efficiency in bacteria elimination from the effluents.
It has been reported that after the addition of H2O2, the rate of •OH radical formation is better for TiO2 in its rutile phase than in the anatase one. A mix of the Anatase:rutile phases can also improve the radicals formation [13]. In the current study, commercial TiO2 was evaluated, which only presents the anatase phase, so performing further research by using other materials containing the rutile phase could be interesting.
3.2. Dyestuffs Discoloration
Figure 3 shows selected images of the reactor, before and after UV/H2O2 treatment. In this figure it is possible to observe the changes in color of the wastewater sample. In previous studies, the characterization of the wastewater samples coming from handicraft factories [35] was found to primarily contain azo dyes. The degradation mechanism of these pollutants mediated by TiO2, under light and by using O2 as oxidant, has been reported by Zhiyong et al.; these authors have employed methyl orange (MO) as the model molecule [36]. In general, it has been described that dye photodegradation takes place by producing a singlet-excited state in which an electron is injected from the excited state of the adsorbed dyestuff onto the TiO2 conduction band. This leads to the formation of a MO cation which subsequently decays and the electron in the conduction band is scavenged by the O2 adsorbed on the TiO2 surface generating the superoxide radical. The (–N=N–) bond can be oxidized by •OH radicals or can be reduced by electrons on the conduction band. The breakup of the –N=N– group leads to the discoloration of the dyestuffs. Azoic dye degradation can also lead to the formation of intermediate compounds such as aromatic amines, phenolic compounds, and short-chain organic acids. The production of these acids is due to the opening of the aromatic ring in the dye molecule due to the exposure of successive oxidations which eventually form CO2 and water [37].

Figure 3.
Sunlight reactor images before (left) and after (right) UV/H2O2 treatment.
Figure 4 shows the wastewater discoloration as a function of the reaction time. As can be seen in the figure, the addition of peroxide is a good alternative for the degradation of dyes, eliminating 61.26% of the color in the water after 7 h of illumination by using 0.07 M of H2O2.
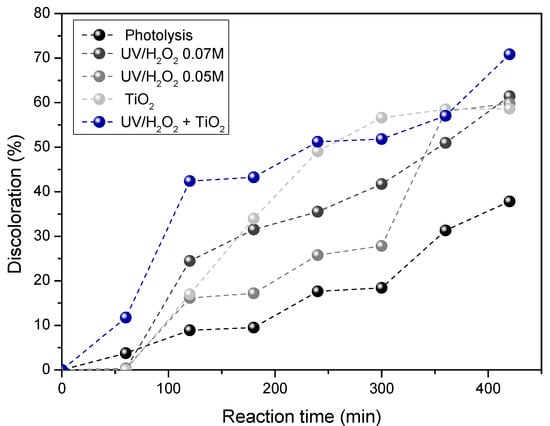
Figure 4.
Stained wastewater discoloration under different AOPs.
On the other hand, by using TiO2 as photocatalyst, the elimination of dyes after 7 h of illumination slightly decreases (58.63% of discoloration) compared to the results obtained by using 0.07 M peroxide. This is mainly due to the opacity generated inside the reactor given by the titania powders employed in the heterogeneous process. These powders cause a screening effect which reduces overall photonic efficiency [38].
Finally, by combining 0.07 M of H2O2 and TiO2, the highest discoloration (i.e., 70.80%) was achieved. This is due to the greater production of •OH radicals induced by a reaction with the peroxide [39].
3.3. Organic Pollutant Mineralization
Taking into account that: (i) dyestuffs can be transformed into new and more dangerous intermediary compounds during the photocatalytic processes; and (ii) different dyes can also absorb light in the same wavelength, TOC analyses were performed to study the mineralization phenomena of the effluents after treatment.
Figure 5 represents TOC elimination as a function of the treatment time. As seen in the figure, the highest TOC value was obtained by using the UV/H2O2 treatment. This behavior can be explained by the potential formation of intermediary organic compounds resulting from the breakup of the dye molecules.
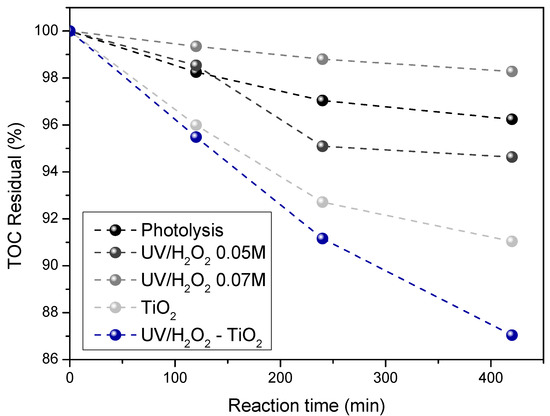
Figure 5.
TOC evolution as a function of the reaction time under different AOPs.
On the other hand, by using TiO2 photocatalysis, a lower TOC value compared to UV/H2O2 assays, was observed, thus showing the mineralization of other organic compounds present in the effluents. The mechanism for organic pollutant degradation under photocatalysis is based on the interaction between light and semiconductor particles. This produces ROS strong enough to destroy chemical pollutants in water by oxidation of the carbon chain mainly given by •OH radicals. As a result, the formation of short chain organic acids and the subsequent mineralization of them to obtain CO2 and water occur. A similar mechanism has been reported by Guo et al. by using phenol as a model molecule [40].
Bearing in mind that the wastewater samples treated in this study were real, more than 10 different dyes can coexist in the sample. As a result, the behavior observed in the TOC analyses by using the UV/H2O2 treatment can be explained by the potential formation of an unknown quantity of intermediate organic compounds caused by dye molecules breaking.
Furthermore, due to the low availability of sunlight during the UV/H2O2 treatments (i.e., 3.4 W/m2), it makes possible the generation of intermediate molecules from dyes in wastewater, which could be mainly due to the catalyzed action of peroxide, not to the direct action of sunlight. This could explain the discoloration but not mineralization [41,42]. On the contrary, the highest mineralization was obtained by using TiO2, it is due to the environmental conditions on the day of treatment (high light intensity), thus showing the mineralization of other organic compounds present in the effluents.
The standard deviation for all the results presented in the manuscript has been attached as Supplementary Material in Tables S1–S3.
Several studies related to E. coli inactivation, dye degradation, and organic pollutant mineralization have been addressed by studying these water pollutants individually. Most of these studies include the simulation of environmental conditions at a lab scale by using bacteria strains and commercial compounds. These studies have been very important to predict the behavior of the AOPs. However, in order to determine the true effectiveness of these processes, it is very important to test them using real wastewater sources. These effluents are usually polluted by a combination of different contaminants at the same time. This study creates an important bridge to previous studies performed on the applications of AOPs on a pilot plant scale in the treatment of effluents coming from small local industries. Thus, further research can be broadened by understanding the primary limitations and advantages of employing AOPs under real environmental conditions.
The main advantage of employing AOPs for the treatment of wastewater is the possibility to degrade many pollutants simultaneously in any given effluent. A key thing to note is that these processes are secondary or tertiary treatments. In order to effectively employ these methods, it is necessary to select one or more of these AOPs to be combined with other methods to obtain better results on a larger scale.
4. Conclusions
UV/H2O2 and TiO2 photocatalysis was successfully tested on a pilot plant scale in the treatment of industrial wastewater, thus showing the potential of AOPs in environmental remediation.
The individual treatments show different effectiveness depending on the pollutant to be treated; thus, the greatest discoloration of dyes was achieved by UV/H2O2. In contrast, the highest elimination of bacteria and decreased TOC value was obtained under photocatalytic treatment based on TiO2.
In general, only small differences were observed in the overall effectiveness of the independent treatments employed for recovering wastewater from handicraft factories. The combination of UV, H2O2, and TiO2 AOPs significantly improves the final results obtained, thus leading to an improvement of 70.80% for dye discoloration, a 51% reduction of bacteria, and the elimination of 87% of organic compounds.
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/w13192705/s1, Table S1: Dyes degradation Standard Deviation, Table S2: Bacteria Removal Standard deviation, and Table S3: TOC Standard deviation.
Author Contributions
Conceptualization, methodology, supervision, and funding acquisition, J.J.M.M.; investigation, J.S.H.N. and W.G., project administration, H.R.; writing—original draft preparation, M.C.H., writing—original draft preparation, J.A.N. All authors have read and agreed to the published version of the manuscript.
Funding
Gobernación de Boyacá, by patrimonio autónomo Fondo Nacional de Financiamiento para la Ciencia, la Tecnología y la Innovación “Fransisco José de Caldas—Minciencias”, Project 789—2020, and Universidad Pedagógica y Tecnológica de Colombia (UPTC) Project SGI 3007.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
This work was financed by the Gobernación de Boyacá, by patrimonio autónomo Fondo Nacional de Financiamiento para la Ciencia, la Tecnología y la Innovación “Fransisco José de Caldas—Minciencias”, Project 789—2020, and Universidad Pedagógica y Tecnológica de Colombia (UPTC) Project SGI 3007. VIE-UPTC for the concession of a young researcher grant 2021, project SGI 3021.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Murcia, J.J.; Hernández, J.; Rojas, H.; Moreno-Cascante, J.; Sánchez-Cid, P.; Hidalgo, M.; Navío, J.A.; Jaramillo-Páez, C. Evaluation of Au–ZnO, ZnO/Ag2CO3 and Ag–TiO2 as Photocatalyst for Wastewater Treatment. Top. Catal. 2020, 63, 1286–1301. [Google Scholar] [CrossRef]
- Leonhauser, J.; Pawar, J.; Birkenbeul, U. Chapter 5—Novel Technologies for the Elimination of Pollutants and Hazardous Substances in the Chemical and Pharmaceutical Industries. In Industrial Wastewater Treatment, Recycling and Reuse; Ranade, V.V., Bhandari, V.M., Eds.; Butterworth-Heinemann: Oxford, UK, 2014; pp. 215–234. [Google Scholar]
- Manickavachagam, M.; Sillanpaa, M.; Swaminathan, M.; Ahmmad, B. Advanced Oxidation Processes for Wastewater Treatment 2014. Int. J. Photoenergy 2015, 2015, 1. [Google Scholar] [CrossRef]
- Serpone, N. Heterogeneous Photocatalysis and Prospects of TiO2-Based Photocatalytic DeNOxing the Atmospheric Environment. Catalysts 2018, 8, 553. [Google Scholar] [CrossRef] [Green Version]
- Dong, H.; Zeng, G.; Tang, L.; Fan, C.; Zhang, C.; He, X.; He, Y. An overview on limitations of TiO2-based particles for photocatalytic degradation of organic pollutants and the corresponding countermeasures. Water Res. 2015, 79, 128–146. [Google Scholar] [CrossRef]
- Skocaj, M.; Filipic, M.; Petkovic, J.; Novak, S. Titanium dioxide in our everyday life; is it safe? Radiol. Oncol. 2011, 45, 227–247. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Valencia, S.; Marín, J.M.; Restrepo, G. Study of the Bandgap of Synthesized Titanium Dioxide Nanoparticules Using the Sol-Gel Method and a Hydrothermal Treatment. Open Mater. Sci. 2010, 4, 9–14. [Google Scholar] [CrossRef]
- Abdullah, A.M.; Gracia-Pinilla, M.Á.; Pillai, S.C.; O’Shea, K. UV and Visible Light-Driven Production of Hydroxyl Radicals by Reduced Forms of N, F, and P Codoped Titanium Dioxide. Molecules 2019, 24, 2147. [Google Scholar] [CrossRef] [Green Version]
- Rosario-Ortiz, F.L.; Wert, E.C.; Snyder, S.A. Evaluation of UV/H2O2 treatment for the oxidation of pharmaceuticals in wastewater. Water Res. 2010, 44, 1440–1448. [Google Scholar] [CrossRef]
- Chidambara Raj, C.B.; Quen, H.L. Advanced oxidation processes for wastewater treatment: Optimization of UV/H2O2 process through a statistical technique. Chem. Eng. Sci. 2005, 60, 5305–5311. [Google Scholar] [CrossRef]
- Ulliman, S.L.; Miklos, D.B.; Hübner, U.; Drewes, J.E.; Linden, K.G. Improving UV/H2O2 performance following tertiary treatment of municipal wastewater. Environ. Sci. Water Res. Technol. 2018, 4, 1321–1330. [Google Scholar] [CrossRef]
- Tseng, D.-H.; Juang, L.-C.; Huang, H.-H. Effect of Oxygen and Hydrogen Peroxide on the Photocatalytic Degradation of Monochlorobenzene in TiO2 Aqueous Suspension. Int. J. Photoenergy 2012, 2012, 328526. [Google Scholar] [CrossRef]
- Hirakawa, T.; Yawata, K.; Nosaka, Y. Photocatalytic reactivity for O2•− and OH• radical formation in anatase and rutile TiO2 suspension as the effect of H2O2 addition. App. Catal. A 2007, 325, 105–111. [Google Scholar] [CrossRef]
- Ameta, R.; Solanki, M.S.; Benjamin, S.; Ameta, S.C. Chapter 6—Photocatalysis. In Advanced Oxidation Processes for Waste Water Treatment; Ameta, S.C., Ameta, R., Eds.; Academic Press: Cambridge, MA, USA, 2018; pp. 135–175. [Google Scholar]
- Li, Z.; Kong, C.; Lu, G. Visible Photocatalytic Water Splitting and Photocatalytic Two-Electron Oxygen Formation over Cu- and Fe-Doped g-C3N4. J. Phys. Chem. C 2016, 120, 56–63. [Google Scholar] [CrossRef]
- Nosaka, Y.; Nosaka, A. Understanding Hydroxyl Radical (•OH) Generation Processes in Photocatalysis. ACS Energy Lett. 2016, 1, 356–359. [Google Scholar] [CrossRef] [Green Version]
- Malato, S.; Blanco, J.; Vidal, A.; Richter, C. Photocatalysis with solar energy at a pilot-plant scale: An overview. Appl. Catal. B 2002, 37, 1–15. [Google Scholar] [CrossRef]
- Malato, S.; Fernández-Ibáñez, P.; Maldonado, M.I.; Blanco, J.; Gernjak, W. Decontamination and disinfection of water by solar photocatalysis: Recent overview and trends. Catal. Today 2009, 147, 1–59. [Google Scholar] [CrossRef]
- Mueses, M.A.; Colina-Márquez, J.; Machuca-Martínez, F.; Puma, G.L. Recent advances on modeling of solar heterogeneous photocatalytic reactors applied for degradation of pharmaceuticals and emerging organic contaminants in water. Curr. Opin. Green Sustain. Chem. 2021, 30, 100486. [Google Scholar] [CrossRef]
- Malato, S.; Maldonado, M.I.; Fernández-Ibáñez, P.; Oller, I.; Polo, I.; Sánchez-Moreno, R. Decontamination and disinfection of water by solar photocatalysis: The pilot plants of the Plataforma Solar de Almeria. Mater. Sci. Semicond. Process. 2016, 42, 15–23. [Google Scholar] [CrossRef]
- Puma, G.L.; Yue, P.L. A laminar falling film slurry photocatalytic reactor. Part I—model development. Chem. Eng. Sci. 1998, 53, 2993–3006. [Google Scholar] [CrossRef]
- Wang, D.; Li, Y.; Zhang, W.; Wang, Q.; Wang, P.; Wang, C. Development and modeling of a flat plate serpentine reactor for photocatalytic degradation of 17-ethinylestradiol. Environ. Sci. Pollut. Res. 2013, 20, 2321–2329. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Mueses, M.A.; Márquez, J.A.C.; Machuca-Martínez, F.; Grčić, I.; Moreira, R.P.M.; Puma, G.L. Engineering and modeling perspectives on photocatalytic reactors for water treatment. Water Res. 2021, 202, 117421. [Google Scholar] [CrossRef] [PubMed]
- Universidad Pedagógica y Tecnológica de Colombia; Mesa, J.J.M.; Cely, W.G.; Sarmiento, H.A.R.; Lobo, J.A.C. Patente “Planta Para el Tratamiento de Aguas Residuales con Función dual Floculación/Fotocatálisis Impulsada por Energía Solar y un Reactor de Tubos Soportado Sobre una Lámina Inclinada”; Superintendencia de Industria y Comercio: Bogotá, Colombia, 2020. [Google Scholar]
- American Public Health Association (APHA). Standard Methods for Examination of Water and Wastewater, 22nd ed.; American Public Health Association/American Water Works Association/Water Environment Federation: Washington, DC, USA, 2012. [Google Scholar]
- Costa, A.; Lepretti, M.; Rizzo, L.; Caputo, I.; Vaiano, V.; Sacco, O.; Silva, W.; Sannino, D. Surface water disinfection by chlorination and advanced oxidation processes: Inactivation of an antibiotic resistant E. coli strain and cytotoxicity evaluation. Sci. Total Environ. 2016, 1, 554–555. [Google Scholar]
- Kacem, M.; Plantard, G.; Wery, N.; Goetz, V. Kinetics and efficiency displayed by supported and suspended TiO2 catalysts applied to the disinfection of Escherichia coli. Chin. J. Catal. 2014, 35, 1571–1577. [Google Scholar] [CrossRef]
- Maness, P.-C.; Smolinski, S.; Blake, D.M.; Huang, Z.; Wolfrum, E.J.; Jacobi, W.A. Bactericidal Activity of Photocatalytic TiO2 Reaction: Toward an Understanding of Its Killing Mechanism. Appl. Environ. Microbiol. 1999, 65, 4094–4098. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lu, Z.-X.; Zhou, L.; Zhang, Z.-L.; Shi, W.-L.; Xie, Z.-X.; Xie, H.-Y.; Pang, D.-W.; Shen, P. Cell Damage Induced by Photocatalysis of TiO2 Thin Films. Langmuir 2003, 19, 8765–8768. [Google Scholar] [CrossRef]
- Aminedi, R.; Wadhwa, G.; Das, N.; Pal, B. Shape-dependent bactericidal activity of TiO2 for the killing of Gram-negative bacteria Agrobacterium tumefaciens under UV torch irradiation. Environ. Sci. Pollut. Res. 2013, 20, 6521–6530. [Google Scholar] [CrossRef]
- Benabbou, A.K.; Derriche, Z.; Felix, C.; Lejeune, P.; Guillard, C. Photocatalytic inactivation of Escherischia coli Effect of concentration of TiO2 and microorganism, nature, and intensity of UV irradiation. App. Catal. B 2007, 76, 257–263. [Google Scholar] [CrossRef]
- Hoffmann, M.R.; Martin, S.T.; Choi, W.; Bahnemann, D.W. Environmental Applications of Semiconductor Photocatalysis. Chem. Rev. 1995, 95, 69. [Google Scholar] [CrossRef]
- Hirakawa, T.; Nosaka, Y. Properties of O2•− and OH• Formed in TiO2 Aqueous Suspensions by Photocatalytic Reaction and the Influence of H2O2 and Some Ions. Langmuir 2002, 18, 3247–3254. [Google Scholar] [CrossRef]
- Nosaka, Y.; Komori, S.; Yawata, K.; Hirakawa, T.; Nosaka, A.Y. Photocatalytic ˙OH radical formation in TiO2 aqueous suspension studied by several detection methods. Phys. Chem. Chem. Phys. 2003, 5, 4731. [Google Scholar] [CrossRef]
- Murcia, J.J.; Cely, Á.C.; Rojas, H.; Hidalgo, M.C.; Navío, J.A. Fluorinated and Platinized Titania as Effective Materials in the Photocatalytic Treatment of Dyestuffs and Stained Wastewater Coming from Handicrafts Factories. Catalysts 2019, 9, 179. [Google Scholar] [CrossRef] [Green Version]
- Zhiyong, Y.; Keppner, H.; Laub, D.; Mielczarski, E.; Mielczarski, J.; Kiwi-Minsker, L.; Renken, A.; Kiwi, J. Photocatalytic discoloration of Methyl Orange on innovative parylene–TiO2 flexible thin films under simulated sunlight. Appl. Catal. B 2008, 79, 63–71. [Google Scholar] [CrossRef] [Green Version]
- He, Y.; Grieser, F.; Ashokkumar, M. The mechanism of sonophotocatalytic degradation of methyl orange and its products in aqueous solutions. Ultrason. Sonochem. 2011, 18, 974–980. [Google Scholar] [CrossRef] [PubMed]
- Hegazy, A.; Prouzet, E. Effect of physical chemistry parameters in photocatalytic properties of TiO2 nanocrystals. Comptes Rendus Chim. 2013, 16, 651–659. [Google Scholar] [CrossRef]
- Lara-Ramos, J.A.; Llanos-Diaz, G.D.; Diaz-Angulo, J.; Machuca-Martínez, F. Evaluation of Caffeine Degradation by Sequential Coupling of TiO2/O3/H2O2/UV Processes. Top. Catal. 2020, 63, 1361–1373. [Google Scholar] [CrossRef]
- Guo, Z.; Ma, R.; Li, G. Degradation of phenol by nanomaterial TiO2 in wastewater. Chem. Eng. J. 2006, 119, 55–59. [Google Scholar] [CrossRef]
- Metz, D.H.; Meyer, M.; Dotson, A.; Beerendonk, E.; Dionysiou, D.D. The effect of UV/H2O2 treatment on disinfection by-product formation potential under simulated distribution system conditions. Water Res. 2011, 45, 3969–3980. [Google Scholar] [CrossRef]
- Vilhunen, S.; Vilve, M.; Vepsäläinen, M.; Sillanpää, M. Removal of organic matter from a variety of water matrices by UV photolysis and UV/H2O2 method. J. Hazard. Mater. 2010, 179, 776–782. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).