Land Use Change Influences Ecosystem Function in Headwater Streams of the Lowland Amazon Basin
Abstract
1. Introduction
2. Materials and Methods
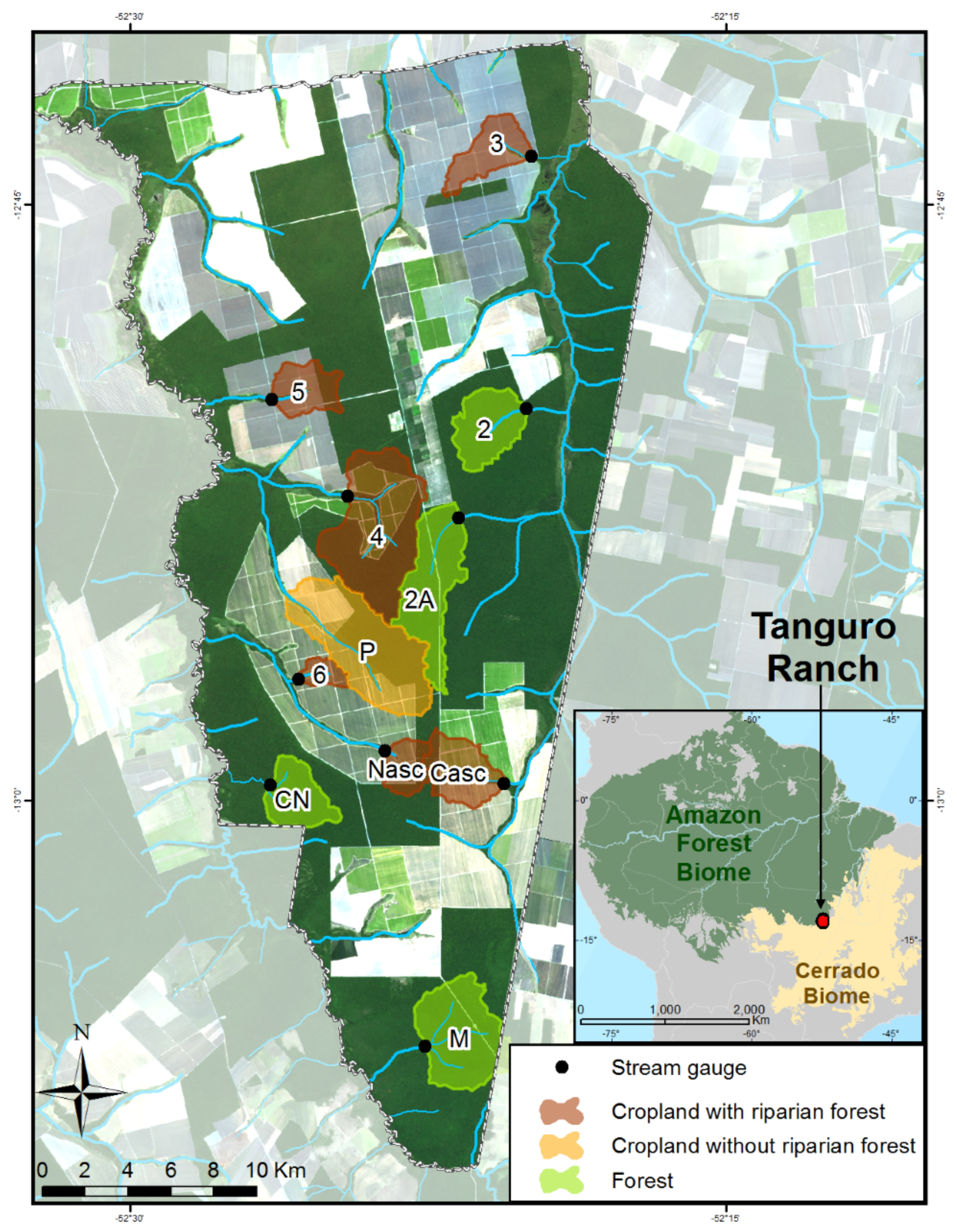
2.1. Site Description
2.2. Stream Physical-Chemical Characteristics
2.3. Litterfall Inputs to Streams
2.4. Stream Habitat and OM Standing Stocks
2.5. Stream Metabolism
2.6. Estimating Nutrient Uptake
2.7. Data Analysis
3. Results
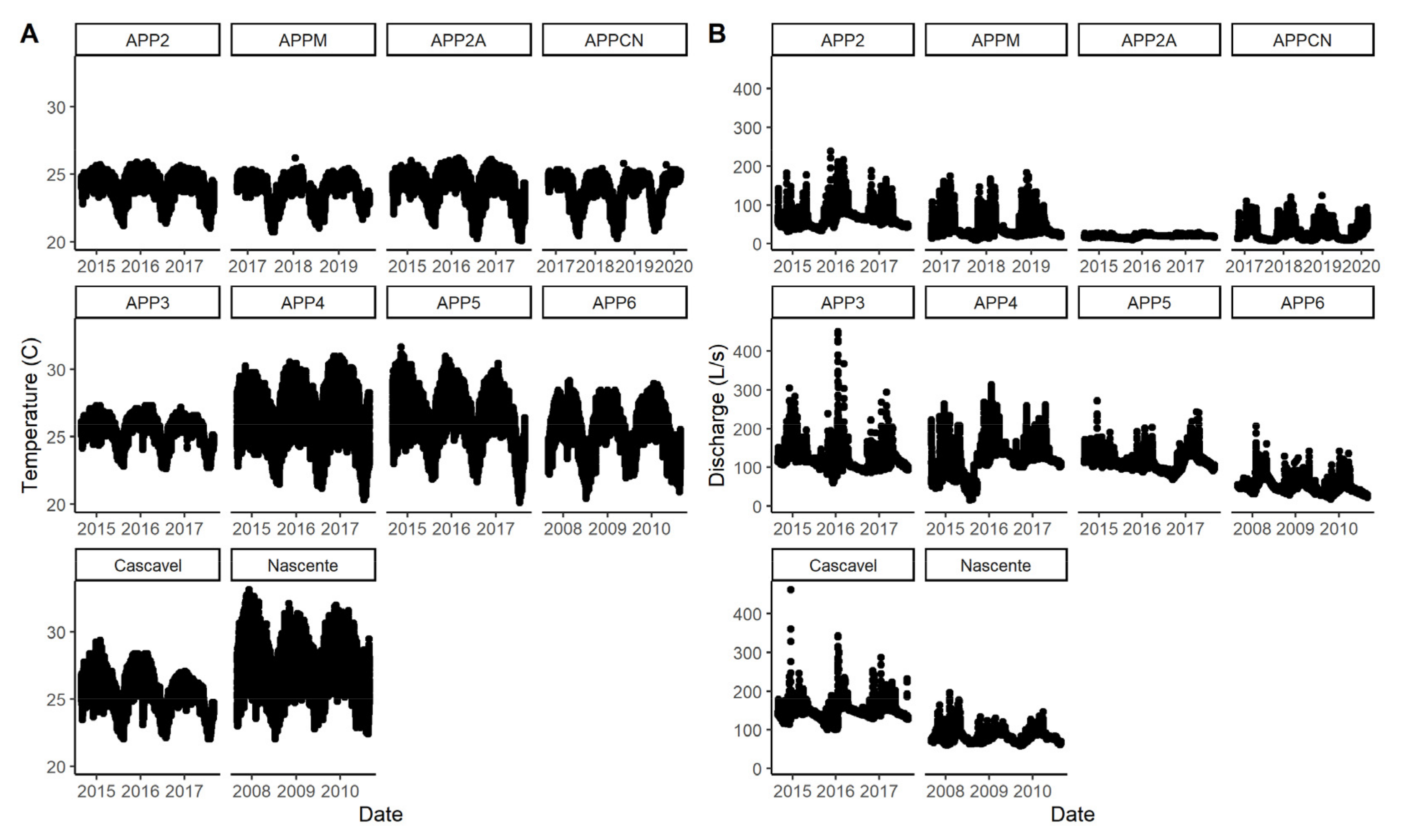
3.1. Stream Characteristics
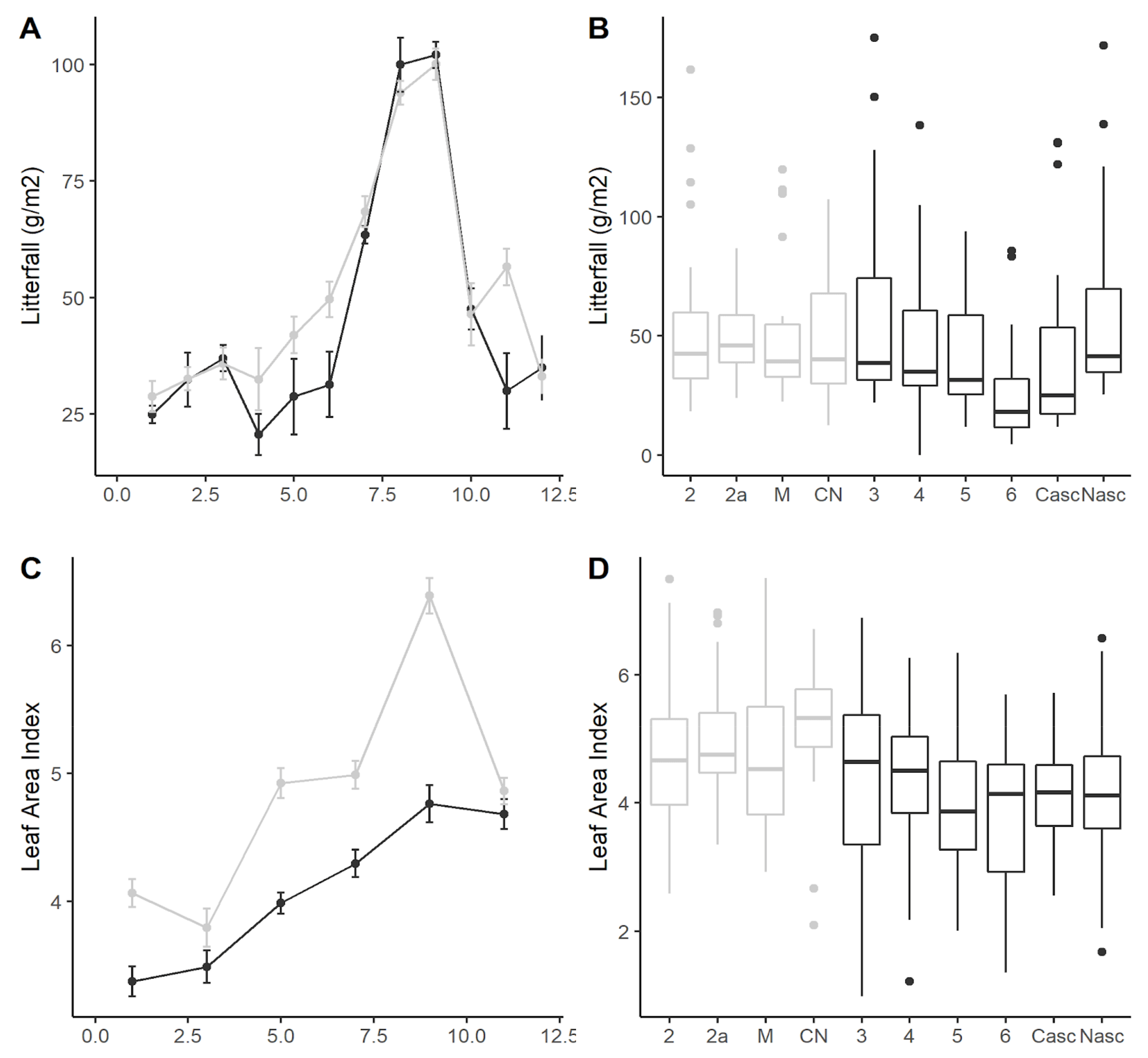
3.2. Litterfall
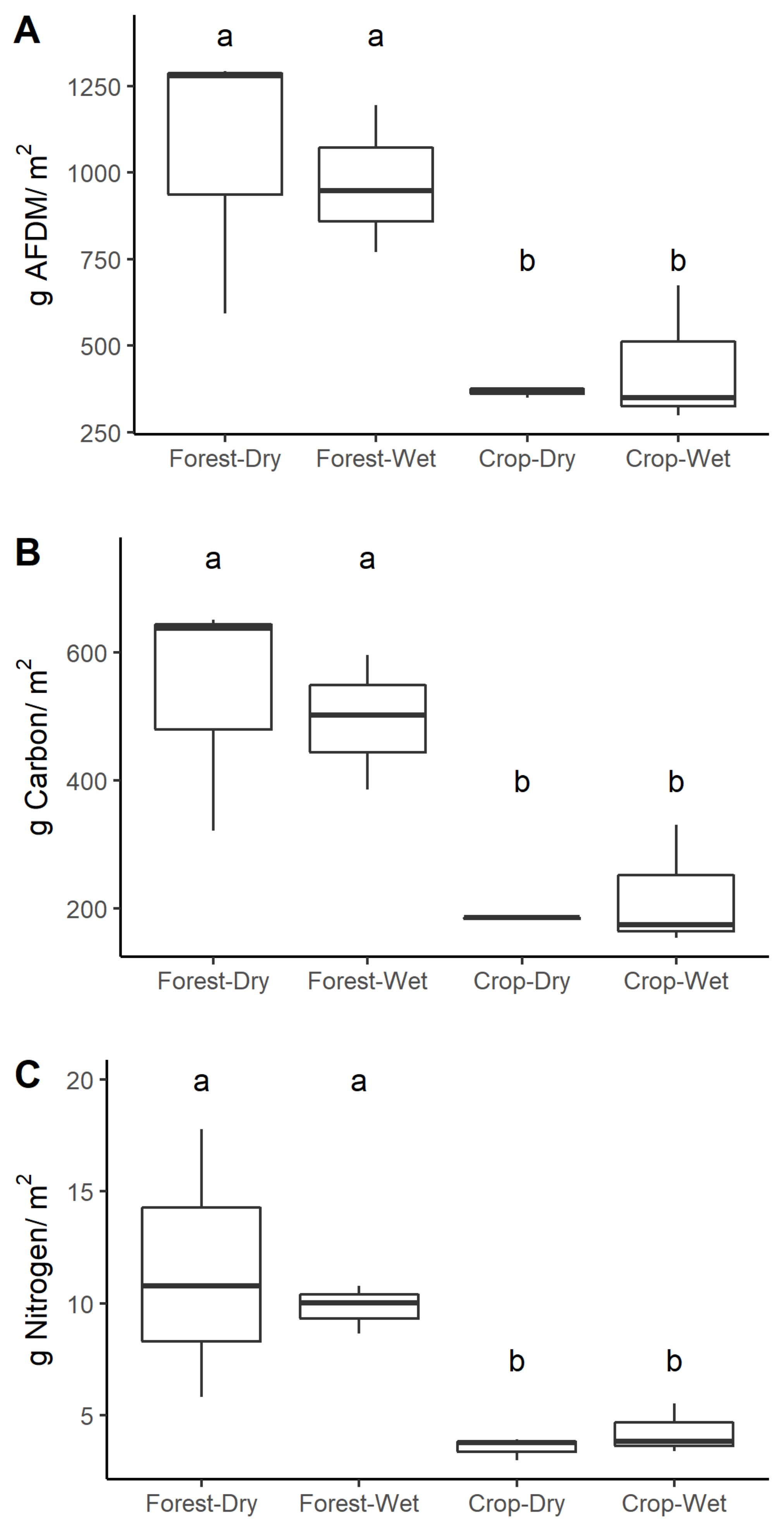
3.3. Stream OM
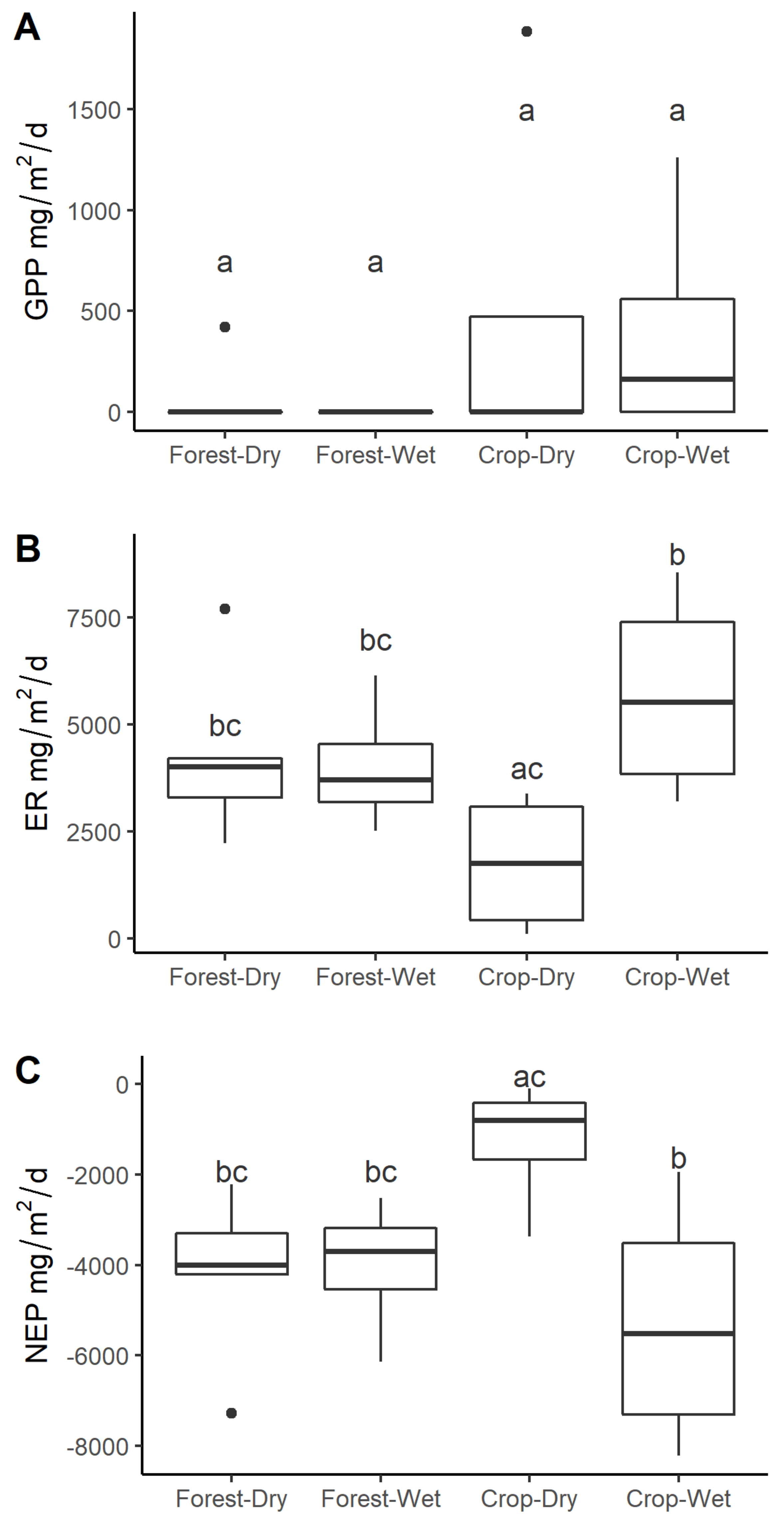
3.4. Stream Metabolism
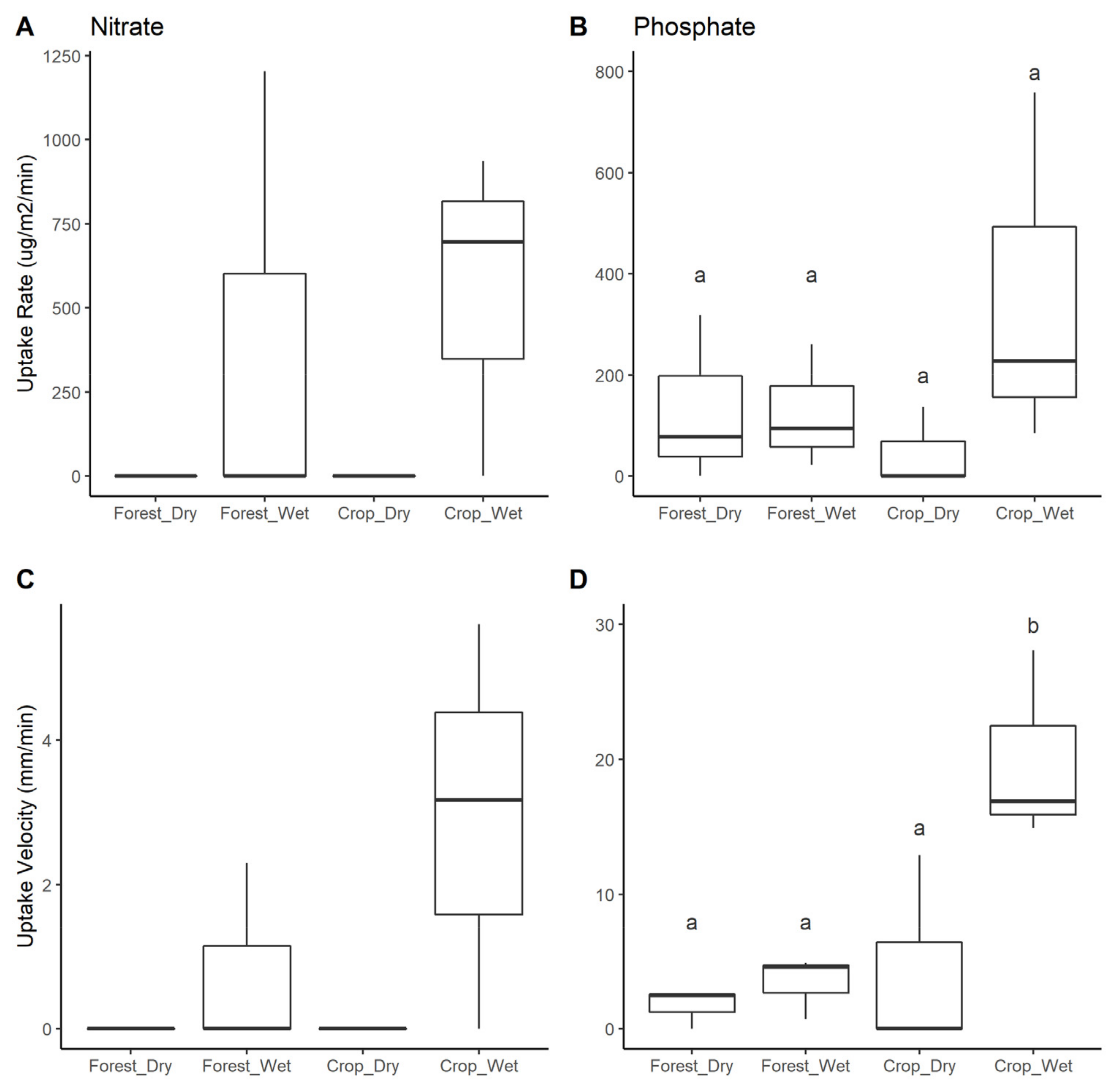
3.5. Nutrient Uptake
4. Discussion
4.1. Litterfall
4.2. OM Standing Stocks
4.3. Stream Metabolism
4.4. Nutrient Uptake
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Allan, J. Landscapes and Riverscapes: The Influence of Land Use on Stream Ecosystems. Annu. Rev. Ecol. Evol. Syst. 2004, 35, 257–284. [Google Scholar] [CrossRef]
- Roser, M.; Ritchie, H. Yields and Land Use in Agriculture [Internet]. 2018. Available online: OurWorldInData.org (accessed on 11 April 2021).
- Carpenter, S.R.; Caraco, N.F.; Correll, D.L.; Howarth, R.W.; Sharpley, A.N.; Smith, V.H. Nonpoint Pollution of Surface Waters with Phosphorus and Nitrogen. Ecol. Appl. 1998, 8, 559–568. [Google Scholar] [CrossRef]
- Turner, R.E.; Rabalais, N.N. Linking Landscape and Water Quality in the Mississippi River Basin for 200 Years. Bioscience 2003, 53, 563–572. [Google Scholar] [CrossRef]
- Mulholland, P.J.; Helton, A.M.; Poole, G.C.; Hall, R.O., Jr.; Hamilton, S.K.; Peterson, B.J.; Tank, J.L.; Ashkenas, L.R.; Cooper, L.W.; Dahm, C.N.; et al. Stream Denitrification across Biomes and Its Response to Anthropogenic Nitrate Loading. Nature 2008, 452, 202-U46. [Google Scholar] [CrossRef] [PubMed]
- Bernot, M.J.; Sobota, D.J.; Hall, R.O.; Mulholland, P.J.; Dodds, W.K.; Webster, J.R.; Tank, J.L.; Ashkenas, L.R.; Cooper, L.W.; Dahm, C.N.; et al. Inter-Regional Comparison of Land-Use Effects on Stream Metabolism. Freshw. Biol. 2010, 55, 1874–1890. [Google Scholar] [CrossRef]
- Ruegg, J.; Conn, C.C.; Anderson, E.P.; Battin, T.J.; Bernhardt, E.S.; Boix Canadell, M.; Bonjour, S.M.; Hosen, J.D.; Marzolf, N.S.; Yackulic, C.B. Thinking like a Consumer: Linking Aquatic Basal Metabolism and Consumer Dynamics. Limnol. Oceanogr. Lett. 2021, 6, 1–17. [Google Scholar] [CrossRef]
- Keenan, R.J.; Reams, G.A.; Achard, F.; de Freitas, J.V.; Grainger, A.; Lindquist, E. Dynamics of Global Forest Area: Results from the FAO Global Forest Resources Assessment 2015. For. Ecol. Manag. 2015, 352, 9–20. [Google Scholar] [CrossRef]
- Marzolf, N.S.; Ardon, M. Ecosystem Metabolism in Tropical Streams and Rivers: A Review and Synthesis. Limnol. Oceanogr. 2021. [Google Scholar] [CrossRef]
- Davies, P.M.; Bunn, S.E.; Hamilton, S.K.; David, D. Primary production in tropical streams and rivers. In Tropical Stream Ecology; Elsevier: Amsterdam, The Netherlands, 2008. [Google Scholar]
- Boulton, A.J.; Boyero, L.; Covich, A.P.; Dobson, M.; Lake, S.; Pearson, R. Are tropical streams ecologically different from temperate streams? In Tropical Stream Ecology; Elsevier: Amsterdam, The Netherlands, 2008; pp. 257–284. [Google Scholar]
- Gentry, A.H. Four Neotropical Rainforests; Yale University Press: New Haven, CT, USA, 1993. [Google Scholar]
- Lowe-McConnell, R. Ecological Studies in Tropical Fish Communities; Cambridge University Press: Cambridge, UK, 1987. [Google Scholar]
- Foley, J.A.; Ramankutty, N.; Brauman, K.A.; Cassidy, E.S.; Gerber, J.S.; Johnston, M.; Mueller, N.D.; O’Connell, C.; Ray, D.K.; West, P.C.; et al. Solutions for a Cultivated Planet. Nature 2011, 478, 337–342. [Google Scholar] [CrossRef]
- Bierschenk, A.M.; Mueller, M.; Pander, J.; Geist, J. Impact of Catchment Land Use on Fish Community Composition in the Headwater Areas of Elbe, Danube and Main. Sci. Total Environ. 2019, 652, 66–74. [Google Scholar] [CrossRef]
- Mueller, M.; Bierschenk, A.M.; Bierschenk, B.M.; Pander, J.; Geist, J. Effects of Multiple Stressors on the Distribution of Fish Communities in 203 Headwater Streams of Rhine, Elbe and Danube. Sci. Total Environ. 2020, 703. [Google Scholar] [CrossRef] [PubMed]
- Gomi, T.; Sidle, R.; Richardson, J. Understanding Processes and Downstream Linkages of Headwater Systems. Bioscience 2002, 52, 905–916. [Google Scholar] [CrossRef]
- McClain, M.E.; Elsenbeer, H. Terrestrial inputs to Amazonian streams and internal biogeochemical processing. In The Biogeochemistry of the Amazon Basin; Oxford University Press: New York, NY, USA, 2001; pp. 209–234. [Google Scholar]
- Peterson, B.; Wollheim, W.; Mulholland, P.; Webster, J.; Meyer, J.; Tank, J.; Marti, E.; Bowden, W.; Valett, H.; Hershey, A.; et al. Control of Nitrogen Export from Watersheds by Headwater Streams. Science 2001, 292, 86–90. [Google Scholar] [CrossRef]
- Koenig, L.E.; Helton, A.M.; Savoy, P.; Bertuzzo, E.; Heffernan, J.B.; Hall, R.O.; Bernhardt, E.S. Emergent Productivity Regimes of River Networks. Limnol. Oceanogr. Lett. 2019, 4, 173–181. [Google Scholar] [CrossRef]
- Richardson, J.S. Biological Diversity in Headwater Streams. Water 2019, 11, 366. [Google Scholar] [CrossRef]
- Sweeney, B. Streamside Forests and the Physical, Chemical, and Trophic Characteristics of Piedmont Streams in Eastern North America. Water Sci. Technol. 1992, 26, 2653–2673. [Google Scholar] [CrossRef]
- Tank, J.L.; Rosi-Marshall, E.J.; Griffiths, N.A.; Entrekin, S.A.; Stephen, M.L. A Review of Allochthonous Organic Matter Dynamics and Metabolism in Streams. J. N. Am. Benthol. Soc. 2010, 29, 118–146. [Google Scholar] [CrossRef]
- Macedo, M.N.; Coe, M.T.; DeFries, R.; Uriarte, M.; Brando, P.M.; Neill, C.; Walker, W.S. Land-Use-Driven Stream Warming in Southeastern Amazonia. Philos. Trans. R. Soc. B Biol. Sci. 2013, 368. [Google Scholar] [CrossRef]
- Kominoski, J.S.; Marczak, L.B.; Richardson, J.S. Riparian Forest Composition Affects Stream Litter Decomposition despite Similar Microbial and Invertebrate Communities. Ecology 2011, 92, 151–159. [Google Scholar] [CrossRef] [PubMed]
- Webster, J.R.; Benfield, E.F. Vascular Plant Breakdown in Freshwater Ecosystems. Annu. Rev. Ecol. Evol. Syst. 1986, 17, 567–594. [Google Scholar] [CrossRef]
- Boyero, L.; Graca, M.A.S.; Tonin, A.M.; Perez, J.; Swafford, A.J.; Ferreira, V.; Landeira-Dabarca, A.; Alexandrou, M.A.; Gessner, M.O.; Mckie, B.G.; et al. Riparian Plant Litter Quality Increases with Latitude. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef]
- Webster, J.; Benfield, E.; Ehrman, T.; Schaeffer, M.; Tank, J.; Hutchens, J.; D’Angelo, D. What Happens to Allochthonous Material That Falls into Streams? A Synthesis of New and Published Information from Coweeta. Freshw. Biol. 1999, 41, 687–705. [Google Scholar] [CrossRef]
- Boyero, L.; Perez, J.; Lopez-Rojo, N.; Tonin, A.M.; Correa-Araneda, F.; Pearson, R.G.; Bosch, J.; Albarino, R.J.; Anbalagan, S.; Barmuta, L.A.; et al. Latitude Dictates Plant Diversity Effects on Instream Decomposition. Sci. Adv. 2021, 7. [Google Scholar] [CrossRef]
- Seitzinger, S.; Harrison, J.A.; Bohlke, J.K.; Bouwman, A.F.; Lowrance, R.; Peterson, B.; Tobias, C.; Van Drecht, G. Denitrification across Landscapes and Waterscapes: A Synthesis. Ecol. Appl. 2006, 16, 2064–2090. [Google Scholar] [CrossRef]
- Kreiling, R.M.; Thoms, M.C.; Richardson, W.B. Beyond the Edge: Linking Agricultural Landscapes, Stream Networks, and Best Management Practices. J. Environ. Qual. 2018, 47, 42–53. [Google Scholar] [CrossRef] [PubMed]
- Knott, J.; Mueller, M.; Pander, J.; Geist, J. Effectiveness of Catchment Erosion Protection Measures and Scale-Dependent Response of Stream Biota. Hydrobiologia 2019, 830, 77–92. [Google Scholar] [CrossRef]
- Hayhoe, S.J.; Neill, C.; Porder, S.; Mchorney, R.; Lefebvre, P.; Coe, M.T.; Elsenbeer, H.; Krusche, A.V. Conversion to Soy on the Amazonian Agricultural Frontier Increases Streamflow without Affecting Stormflow Dynamics. Glob. Chang. Biol. 2011, 17, 1821–1833. [Google Scholar] [CrossRef]
- Likens, G.E.; Bormann, F.H. Lingages between Terrestrial and Aquatic Ecosystems. Bioscience 1974, 24, 447–456. [Google Scholar] [CrossRef]
- Bormann, F.H.; Likens, G.E.; Fisher, D.W.; Pierce, R.S. Nutrient Loss Accelerated by Clear Cutting of a Forest Ecosystem. Science 1968, 159, 882–884. [Google Scholar] [CrossRef] [PubMed]
- Likens, G.E.; Bormann, F.H.; Johnson, N.M.; Fisher, D.W.; Pierce, R.S. Effects of Forest Cutting and Herbicide Treatment on Nutrient Budgets in the Hubbard Brook Watershed-Ecosystem. Ecol. Monogr. 1970, 40, 23–47. [Google Scholar] [CrossRef]
- Neill, C.; Coe, M.T.; Riskin, S.H.; Krusche, A.V.; Elsenbeer, H.; Macedo, M.N.; McHorney, R.; Lefebvre, P.; Davidson, E.A.; Scheffler, R.; et al. Watershed Responses to Amazon Soya Bean Cropland Expansion and Intensification. Philos. Trans. R. Soc. B Biol. Sci. 2013, 368, 20120425. [Google Scholar] [CrossRef] [PubMed]
- Mulholland, P.J.; Fellows, C.S.; Tank, J.L.; Grimm, N.B.; Webster, J.R.; Hamilton, S.K.; Marti, E.; Ashkenas, L.; Bowden, W.B.; Dodds, W.K.; et al. Inter-Biome Comparison of Factors Controlling Stream Metabolism. Freshw. Biol. 2001, 46, 1503–1517. [Google Scholar] [CrossRef]
- Jordan, T.; Correll, D.; Weller, D. Nutrient Interception by a Riparian Forest Receiving Inputs from Adjacent Cropland. J. Environ. Qual. 1993, 22, 467–473. [Google Scholar] [CrossRef]
- Kuhn, J.; Casas-Mulet, R.; Pander, J.; Geist, J. Assessing Stream Thermal Heterogeneity and Cold-Water Patches from UAV-Based Imagery: A Matter of Classification Methods and Metrics. Remote Sens. 2021, 13, 1379. [Google Scholar] [CrossRef]
- Nagy, R.C.; Porder, S.; Neill, C.; Brando, P.; Quintino, R.M.; do Nascimento, S.A. Structure and Composition of Altered Riparian Forests in an Agricultural Amazonian Landscape. Ecol. Appl. 2015, 25, 1725–1738. [Google Scholar] [CrossRef] [PubMed]
- Maracahipes-Santos, L.; Silverio, D.V.; Macedo, M.N.; Maracahipes, L.; Jankowski, K.J.; Paolucci, L.N.; Neill, C.; Brando, P.M. Agricultural Land-Use Change Alters the Structure and Diversity of Amazon Riparian Forests. Biol. Conserv. 2020, 252. [Google Scholar] [CrossRef]
- Broussard, W.; Turner, R.E. A Century of Changing Land-Use and Water-Quality Relationships in the Continental US. Front. Ecol. Environ. 2009, 7, 302–307. [Google Scholar] [CrossRef]
- Marques, N.C.S.; Jankowski, K.J.; Macedo, M.N.; Juen, L.; Luiza-Andrade, A.; Deegan, L.A. Riparian Forests Buffer the Negative Effects of Cropland on Macroinvertebrate Diversity in Lowland Amazonian Streams. Hydrobiologia 2021, 1–18. [Google Scholar] [CrossRef]
- Ilha, P.; Rosso, S.; Schiesari, L. Effects of Deforestation on Headwater Stream Fish Assemblages in the Upper Xingu River Basin, Southeastern Amazonia. Neotrop. Ichthyol. 2019, 17. [Google Scholar] [CrossRef]
- Nepstad, D.C.; Stickler, C.M.; Soares-Filho, B.; Merry, F. Interactions among Amazon Land Use, Forests and Climate: Prospects for a near-Term Forest Tipping Point. Philos. Trans. R. Soc. B Biol. Sci. 2008, 363, 1737–1746. [Google Scholar] [CrossRef]
- Brando, P.M.; Coe, M.T.; DeFries, R.; Azevedo, A.A. Ecology, Economy and Management of an Agroindustrial Frontier Landscape in the Southeast Amazon. Philos. Trans. R. Soc. B Biol. Sci. 2013, 368. [Google Scholar] [CrossRef]
- Macedo, M.N.; DeFries, R.S.; Morton, D.C.; Stickler, C.M.; Galford, G.L.; Shimabukuro, Y.E. Decoupling of Deforestation and Soy Production in the Southern Amazon during the Late 2000s. Proc. Natl. Acad. Sci. USA 2012, 109, 1341–1346. [Google Scholar] [CrossRef] [PubMed]
- Spera, S.A.; Cohn, A.S.; VanWey, L.K.; Mustard, J.F.; Rudorff, B.F.; Risso, J.; Adami, M. Recent Cropping Frequency, Expansion, and Abandonment in Mato Grosso, Brazil Had Selective Land Characteristics. Environ. Res. Lett. 2014, 9, 064010. [Google Scholar] [CrossRef]
- Neill, C.; Macedo, M.N. The rise of Brazil’s globally-connected Amazon soybean agriculture. In Global Latin America: Into the Twenty-First Century; Gutman, M., Lesser, J., Eds.; University of California Press: Berkeley, CA, USA, 2016; pp. 167–186. [Google Scholar]
- Riskin, S.H.; Neill, C.; Jankowski, K.; Krusche, A.V.; McHorney, R.; Elsenbeer, H.; Macedo, M.N.; Nunes, D.; Porder, S. Solute and Sediment Export from Amazon Forest and Soybean Headwater Streams. Ecol. Appl. 2017, 27, 193–207. [Google Scholar] [CrossRef]
- Maracahipes-Santos, L.; dos Santos, J.O.; Reis, S.M.; Lenza, E. Temporal Changes in Species Composition, Diversity, and Woody Vegetation Structure of Savannas in the Cerrado-Amazon Transition Zone. Acta Bot. Bras. 2018, 32, 254–263. [Google Scholar] [CrossRef]
- Riskin, S.H.; Porder, S.; Neill, C.; Figueira, A.M.e.S.; Tubbesing, C.; Mahowald, N. The Fate of Phosphorus Fertilizer in Amazon Soya Bean Fields. Philos. Trans. R. Soc. B Biol. Sci. 2013, 368, 20120154. [Google Scholar] [CrossRef] [PubMed]
- Jankowski, K.; Neill, C.; Davidson, E.A.; Macedo, M.N.; Costa, C., Jr.; Galford, G.L.; Santos, L.M.; Lefebvre, P.; Nunes, D.; Cerri, C.E.P.; et al. Deep Soils Modify Environmental Consequences of Increased Nitrogen Fertilizer Use in Intensifying Amazon Agriculture. Sci. Rep. 2018, 8. [Google Scholar] [CrossRef]
- Galford, G.L.; Melillo, J.M.; Kicklighter, D.W.; Cronin, T.W.; Cerri, C.E.P.; Mustard, J.F.; Cerri, C.C. Greenhouse Gas Emissions from Alternative Futures of Deforestation and Agricultural Management in the Southern Amazon. Proc. Natl. Acad. Sci. USA 2010, 107, 19649–19654. [Google Scholar] [CrossRef]
- Clark, D.; Brown, S.; Kicklighter, D.; Chambers, J.; Thomlinson, J.; Ni, J. Measuring Net Primary Production in Forests: Concepts and Field Methods. Ecol. Appl. 2001, 11, 356–370. [Google Scholar] [CrossRef]
- Holtgrieve, G.W.; Schindler, D.E.; Branch, T.A.; A’mar, Z.T. Simultaneous Quantification of Aquatic Ecosystem Metabolism and Reaeration Using a Bayesian Statistical Model of Oxygen Dynamics. Limnol. Oceanogr. 2010, 55, 1047–1063. [Google Scholar] [CrossRef]
- Schindler, D.E.; Jankowski, K.; A’mar, Z.T.; Holtgrieve, G.W. Two-Stage Metabolism Inferred from Diel Oxygen Dynamics in Aquatic Ecosystems. Ecosphere 2017, 8, e01867. [Google Scholar] [CrossRef]
- Jankowski, K.J.; Schindler, D.E. Watershed Geomorphology Modifies the Sensitivity of Aquatic Ecosystem Metabolism to Temperature. Sci. Rep. 2019, 9. [Google Scholar] [CrossRef]
- Raymond, P.A.; Zappa, C.J.; Butman, D.; Bott, T.L.; Potter, J.; Mulholland, P.; Laursen, A.E.; McDowell, W.H.; Newbold, D. Scaling the Gas Transfer Velocity and Hydraulic Geometry in Streams and Small Rivers: Gas Transfer Velocity and Hydraulic Geometry. Limnol. Oceanogr. Fluids Environ. 2012, 2, 41–53. [Google Scholar] [CrossRef]
- Plummer, M.; Best, N.; Cowles, K.; Vines, K.; Sarkar, D.; Bates, D.; Almond, R.; Magnusson, A. Output Analysis and Diagnostics for MCMC. R News 2006, 6, 7–11. [Google Scholar]
- Tank, J.L.; Rosi-Marshall, E.J.; Baker, M.A.; Hall, R.O., Jr. Are Rivers Just Big Streams? A Pulse Method to Quantify Nitrogen Demand in a Large River. Ecology 2008, 89, 2935–2945. [Google Scholar] [CrossRef]
- Covino, T.P.; McGlynn, B.L.; McNamara, R.A. Tracer Additions for Spiraling Curve Characterization (TASCC): Quantifying Stream Nutrient Uptake Kinetics from Ambient to Saturation. Limnol. Oceanogr. Methods 2010, 8, 484–498. [Google Scholar] [CrossRef]
- Wollheim, W.M.; Vörösmarty, C.J.; Peterson, B.J.; Seitzinger, S.P.; Hopkinson, C.S. Relationship between River Size and Nutrient Removal. Geophys. Res. Lett. 2006, 33. [Google Scholar] [CrossRef]
- Bates, D.; Maechler, M.; Bolker, B.; Walker, S. Fitting Linear Mixed-Effects Models Using Lme4. J. Stat. Softw. 2015, 67, 1–48. [Google Scholar] [CrossRef]
- Burnham, K.P.; Anderson, D.R. Model Selection and Multimodel Inference; Springer: New York, NY, USA, 2002. [Google Scholar]
- Nakagawa, S.; Schielzeth, H. A General and Simple Method for Obtaining R2 from Generalized Linear Mixed-Effects Models. Methods Ecol. Evol. 2013, 4, 133–142. [Google Scholar] [CrossRef]
- Lenth, R.V.; Buerkner, P.; Herve, M.; Love, J.; Riebl, H.; Singmann, H. Estimated Marginal Means, Aka Least-Squares Means; R Core Team: Vienna, Austria, 2021. [Google Scholar]
- Selva, E.C.; Couto, E.G.; Johnson, M.S.; Lehmann, J. Litterfall Production and Fluvial Export in Headwater Catchments of the Southern Amazon. J. Trop. Ecol. 2007, 23, 329–335. [Google Scholar] [CrossRef][Green Version]
- Tonin, A.M.; Goncalves, J.F., Jr.; Bambi, P.; Couceiro, S.R.M.; Feitoza, L.A.M.; Fontana, L.E.; Hamada, N.; Hepp, L.U.; Lezan-Kowalczuk, V.G.; Leite, G.F.M.; et al. Plant Litter Dynamics in the Forest-Stream Interface: Precipitation Is a Major Control across Tropical Biomes. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef]
- Wantzen, K.M.; Yule, C.; Tockner, K.; Junk, W.J. Riparian wetlands of tropical streams. In Tropical Stream Ecology; Elsevier: London, UK, 2008; pp. 199–218. [Google Scholar]
- Goncalves Junior, J.F.; Franca, J.S.; Callisto, M. Dynamics of Allochthonous Organic Matter in a Tropical Brazilian Headstream. Braz. Arch. Biol. Technol. 2006, 49, 967–973. [Google Scholar] [CrossRef]
- Taiz, L.; Zeiger, E. Fisiologia Vegetal, 3rd ed.; Artmed: Porto Alegre, Brazil, 2004. [Google Scholar]
- Stout, B.; Benfield, E.; Webster, J. Effects of a Forest Disturbance on Shredder Production in Southern Appalachian Headwater Streams. Freshw. Biol. 1993, 29, 59–69. [Google Scholar] [CrossRef]
- Sponseller, R.; Benfield, E. Influences of Land Use on Leaf Breakdown in Southern Appalachian Headwater Streams: A Multiple-Scale Analysis. J. N. Am. Benthol. Soc. 2001, 20, 44–59. [Google Scholar] [CrossRef]
- Magana, A. Litter Input from Riparian Vegetation to Streams: A Case Study of the Njoro, River, Kenya. Hydrobiologia 2001, 458, 141–149. [Google Scholar] [CrossRef]
- Benstead, J.; Pringle, C. Deforestation Alters the Resource Base and Biomass of Endemic Stream Insects in Eastern Madagascar. Freshw. Biol. 2004, 49, 490–501. [Google Scholar] [CrossRef]
- Suberkropp, K. Annual Production of Leaf Decaying Fungi in a Woodland Stream. Freshw. Biol. 1997, 38, 169–178. [Google Scholar] [CrossRef]
- Laitung, B.; Pretty, J.; Chauvet, E.; Dobson, M. Response of Aquatic Hyphomycete Communities to Enhanced Stream Retention in Areas Impacted by Commercial Forestry. Freshw. Biol. 2002, 47, 313–323. [Google Scholar] [CrossRef]
- Entrekin, S.A.; Tank, J.L.; Rosi-Marshall, E.J.; Hoellein, T.J.; Lamberti, G.A. Responses in Organic Matter Accumulation and Processing to an Experimental Wood Addition in Three Headwater Streams. Freshw. Biol. 2008, 53, 1642–1657. [Google Scholar] [CrossRef]
- Wallace, J.B.; Eggert, S.L.; Meyer, J.L.; Webster, J.R. Stream Invertebrate Productivity Linked to Forest Subsidies: 37 Stream-Years of Reference and Experimental Data. Ecology 2015, 96, 1213–1228. [Google Scholar] [CrossRef]
- Leroy, C.; Marks, J. Litter Quality, Stream Characteristics and Litter Diversity Influence Decomposition Rates and Macroinvertebrates. Freshw. Biol. 2006, 51, 605–617. [Google Scholar] [CrossRef]
- Wallace, J.; Eggert, S.; Meyer, J.; Webster, J. Multiple Trophic Levels of a Forest Stream Linked to Terrestrial Litter Inputs. Science 1997, 277, 102–104. [Google Scholar] [CrossRef]
- Angermeier, P.L.; Karr, J.R. Fish Communities along Environmental Gradient in a System of Tropical Streams. Environ. Biol. Fishes 1983, 9, 117–135. [Google Scholar] [CrossRef]
- Scheffler, R.; Neill, C.; Krusche, A.V.; Elsenbeer, H. Soil Hydraulic Response to Land-Use Change Associated with the Recent Soybean Expansion at the Amazon Agricultural Frontier. Agric. Ecosyst. Environ. 2011, 144, 281–289. [Google Scholar] [CrossRef]
- Tuchman, N.; King, R. Changes in Mechanisms of Summer Detritus Processing between Wooded and Agricultural Sites in a Michigan Headwater Stream. Hydrobiologia 1993, 268, 115–127. [Google Scholar] [CrossRef]
- D’Angelo, D.; Webster, J. Natural and Constrainment-Induced Factors Influencing Breakdown of Dogwood and Oak Leaves. Hydrobiologia 1992, 237, 39–46. [Google Scholar] [CrossRef]
- Boyero, L.; Pearson, R.G.; Hui, C.; Gessner, M.O.; Perez, J.; Alexandrou, M.A.; Graca, M.A.S.; Cardinale, B.J.; Albarino, R.J.; Arunachalam, M.; et al. Biotic and Abiotic Variables Influencing Plant Litter Breakdown in Streams: A Global Study. Proc. R. Soc. B Biol. Sci. 2016, 283. [Google Scholar] [CrossRef]
- Shah, J.J.F.; Kominoski, J.S.; Ardon, M.; Dodds, W.K.; Gessner, M.O.; Griffiths, N.A.; Hawkins, C.P.; Johnson, S.L.; Lecerf, A.; Leroy, C.J.; et al. Global Synthesis of the Temperature Sensitivity of Leaf Litter Breakdown in Streams and Rivers. Glob. Chang. Biol. 2017, 23, 3064–3075. [Google Scholar] [CrossRef]
- Landeira-Dabarca, A.; Perez, J.; Graca, M.A.S.; Boyero, L. Joint Effects of Temperature and Litter Quality on Detritivore-Mediated Breakdown in Streams. Aquat. Sci. 2019, 81. [Google Scholar] [CrossRef]
- Spencer, R.G.M.; Kellerman, A.M.; Podgorski, D.C.; Macedo, M.N.; Jankowski, K.; Nunes, D.; Neill, C. Identifying the Molecular Signatures of Agricultural Expansion in Amazonian Headwater Streams. J. Geophys. Res. Biogeosci. 2019, 124, 1637–1650. [Google Scholar] [CrossRef]
- Carlson, K.M.; Curran, L.M.; Ponette-González, A.G.; Ruspita; Lisnawati, N.; Lisnawati, N.; Purwanto, Y.; Brauman, K.A.; Raymond, P.A. Influence of Watershed-Climate Interactions on Stream Temperature, Sediment Yield, and Metabolism along a Land Use Intensity Gradient in Indonesian Borneo. J. Geophys. Res. Biogeosci. 2014, 119, 1110–1128. [Google Scholar] [CrossRef]
- Ortega-Pieck, A.; Fremier, A.K.; Orr, C.H. Agricultural Influences on the Magnitude of Stream Metabolism in Humid Tropical Headwater Streams. Hydrobiologia 2017, 799, 49–64. [Google Scholar] [CrossRef]
- Tromboni, F.; Dodds, W.K.; Neres-Lima, V.; Zandonà, E.; Moulton, T.P. Heterogeneity and Scaling of Photosynthesis, Respiration, and Nitrogen Uptake in Three Atlantic Rainforest Streams. Ecosphere 2017, 8, e01959. [Google Scholar] [CrossRef]
- Saltarelli, W.A.; Dodds, W.K.; Tromboni, F.; do Carmo Calijuri, M.; Neres-Lima, V.; Jordão, C.E.; Palhares, J.C.P.; Cunha, D.G.F. Variation of Stream Metabolism along a Tropical Environmental Gradient. J. Limnol. 2018, 77, 3. [Google Scholar] [CrossRef]
- Silva-Junior, E.F.; Moulton, T.P.; Boechat, I.G.; Guecker, B. Leaf Decomposition and Ecosystem Metabolism as Functional Indicators of Land Use Impacts on Tropical Streams. Ecol. Indic. 2014, 36, 195–204. [Google Scholar] [CrossRef]
- Bott, T.L.; Newbold, J.D. Ecosystem Metabolism and Nutrient Uptake in Peruvian Headwater Streams: Metabolism and Nutrient Spiraling in Peruvian Streams. Int. Rev. Hydrobiol. 2013, 98, 117–131. [Google Scholar] [CrossRef]
- Ortiz-Zayas, J.; Lewis, W.; Saunders, J.; McCutchan, J.; Scatena, F. Metabolism of a Tropical Rainforest Stream. J. N. Am. Benthol. Soc. 2005, 24, 769–783. [Google Scholar] [CrossRef]
- Guecker, B.; Boechat, I.G.; Giani, A. Impacts of Agricultural Land Use on Ecosystem Structure and Whole-Stream Metabolism of Tropical Cerrado Streams. Freshw. Biol. 2009, 54, 2069–2085. [Google Scholar] [CrossRef]
- Fugère, V.; Jacobsen, D.; Finestone, E.H.; Chapman, L.J. Ecosystem Structure and Function of Afrotropical Streams with Contrasting Land Use. Freshw. Biol. 2018, 63, 1498–1513. [Google Scholar] [CrossRef]
- Masese, F.O.; Salcedo-Borda, J.S.; Gettel, G.M.; Irvine, K.; McClain, M.E. Influence of Catchment Land Use and Seasonality on Dissolved Organic Matter Composition and Ecosystem Metabolism in Headwater Streams of a Kenyan River. Biogeochemistry 2017, 132, 1–22. [Google Scholar] [CrossRef]
- Laurance, W.; Laurance, S.; Ferreira, L.; RankindeMerona, J.; Gascon, C.; Lovejoy, T. Biomass Collapse in Amazonian Forest Fragments. Science 1997, 278, 1117–1118. [Google Scholar] [CrossRef]
- Finlay, J.C. Stream Size and Human Influences on Ecosystem Production in River Networks. Ecosphere 2011, 2, art87. [Google Scholar] [CrossRef]
- Raymond, P.A.; Saiers, J.E.; Sobczak, W.V. Hydrological and Biogeochemical Controls on Watershed Dissolved Organic Matter Transport: Pulse-Shunt Concept. Ecology 2016, 97, 5–16. [Google Scholar] [CrossRef]
- Payn, R.A.; Hall, R.O.; Kennedy, T.A.; Poole, G.C.; Marshall, L.A. A Coupled Metabolic-Hydraulic Model and Calibration Scheme for Estimating Whole-River Metabolism during Dynamic Flow Conditions: Estimating River Metabolism during Dynamic Flow. Limnol. Oceanogr. Methods 2017, 15, 847–866. [Google Scholar] [CrossRef]
- O’Donnell, B.; Hotchkiss, E.R. Coupling Concentration- and Process-Discharge Relationships Integrates Water Chemistry and Metabolism in Streams. Water Resour. Res. 2019, 55, 10179–10190. [Google Scholar] [CrossRef]
- Davidson, E.A.; de Carvalho, C.J.R.; Figueira, A.M.; Ishida, F.Y.; Ometto, J.P.H.B.; Nardoto, G.B.; Saba, R.T.; Hayashi, S.N.; Leal, E.C.; Vieira, I.C.G.; et al. Recuperation of Nitrogen Cycling in Amazonian Forests Following Agricultural Abandonment. Nature 2007, 447, 995-U6. [Google Scholar] [CrossRef] [PubMed]
- Vitousek, P.M.; Porder, S.; Houlton, B.Z.; Chadwick, O.A. Terrestrial Phosphorus Limitation: Mechanisms, Implications, and Nitrogen-Phosphorus Interactions. Ecol. Appl. 2010, 20, 5–15. [Google Scholar] [CrossRef]
- Ensign, S.H.; Doyle, M.W. Nutrient Spiraling in Streams and River Networks. J. Geophys. Res. Biogeosci. 2006, 111. [Google Scholar] [CrossRef]
- Neill, C.; Deegan, L.A.; Thomas, S.M.; Haupert, C.L.; Krusche, A.V.; Ballester, V.M.; Victoria, R.L. Deforestation Alters the Hydraulic and Biogeochemical Characteristics of Small Lowland Amazonian Streams. Hydrol. Process. 2006, 20, 2563–2580. [Google Scholar] [CrossRef]
- Finkler, N.R.; Tromboni, F.; Boechat, I.G.; Gucker, B.; Fernandes Cunha, D.G. Nitrogen and Phosphorus Uptake Dynamics in Tropical Cerrado Woodland Streams. Water 2018, 10, 1080. [Google Scholar] [CrossRef]
- Tromboni, F.; Thomas, S.A.; Gucker, B.; Neres-Lima, V.; Lourenco-Amorim, C.; Moulton, T.P.; Silva-Junior, E.F.; Feijo-Lima, R.; Boechat, I.G.; Zandona, E. Nutrient Limitation and the Stoichiometry of Nutrient Uptake in a Tropical Rain Forest Stream. J. Geophys. Res. Biogeosci. 2018, 123, 2154–2167. [Google Scholar] [CrossRef]
- Demars, B.O.L. Whole-Stream Phosphorus Cycling: Testing Methods to Assess the Effect of Saturation of Sorption Capacity on Nutrient Uptake Length Measurements. Water Res. 2008, 42, 2507–2516. [Google Scholar] [CrossRef]
- Powers, S.M.; Johnson, R.A.; Stanley, E.H. Nutrient Retention and the Problem of Hydrologic Disconnection in Streams and Wetlands. Ecosystems 2012, 15, 435–449. [Google Scholar] [CrossRef]
- Stream Solute Workshop Concepts and Methods for Assessing Solute Dynamics in Stream Ecosystems. J. N. Am. Benthol. Soc. 1990, 9, 95–119. [CrossRef]
- Schade, J.D.; MacNeill, K.; Thomas, S.A.; McNeely, F.C.; Welter, J.R.; Hood, J.; Goodrich, M.; Power, M.E.; Finlay, J.C. The Stoichiometry of Nitrogen and Phosphorus Spiralling in Heterotrophic and Autotrophic Streams. Freshw. Biol. 2011, 56, 424–436. [Google Scholar] [CrossRef]
- Fernandes Cunha, D.G.; Finkler, N.R.; do Carmo Calijuri, M.; Covino, T.P.; Tromboni, F.; Dodds, W.K. Nutrient Uptake in a Simplified Stream Channel: Experimental Manipulation of Hydraulic Residence Time and Transient Storage. Ecohydrology 2018, 11. [Google Scholar] [CrossRef]
- Newbold, J.; Elwood, J.W.; O’Neill, R.V.; Sheldon, A.L. Phosphorus Dynamics in a Woodland Stream Ecosystem: A Study of Nutrient Spiralling. Ecology 1983, 64, 1249–1265. [Google Scholar] [CrossRef]
- Meyer, J. The Role of Sediments and Bryophytes in Phosphorus Dynamics in a Headwater Stream Ecosystem. Limnol. Oceanogr. 1979, 24, 365–375. [Google Scholar] [CrossRef]
- Bernot, M.; Tank, J.; Royer, T.; David, M. Nutrient Uptake in Streams Draining Agricultural Catchments of the Midwestern United States. Freshw. Biol. 2006, 51, 499–509. [Google Scholar] [CrossRef]
- Wymore, A.S.; Coble, A.A.; Rodriguez-Cardona, B.; McDowell, W.H. Nitrate Uptake across Biomes and the Influence of Elemental Stoichiometry: A New Look at LINX II. Glob. Biogeochem. Cycles 2016, 30, 1183–1191. [Google Scholar] [CrossRef]
- Gucker, B.; Boechat, I.G. Measurement Uncertainty in Stream Nutrient Uptake: Detecting Land-Use Impacts on Tropical Streams. Ecol. Indic. 2019, 106. [Google Scholar] [CrossRef]
- Deegan, L.A.; Neill, C.; Haupert, C.L.; Ballester, M.V.R.; Krusche, A.V.; Victoria, R.L.; Thomas, S.M.; de Moor, E. Amazon Deforestation Laters Small Stream Structure, Nitrogen Biogeochemistry, and Connectivity to Larger Rivers. Biogeochemistry 2011, 105, 53–74. [Google Scholar] [CrossRef]
- Fernandes Cunha, D.G.; Finkler, N.R.; Gomez, N.; Cochero, J.; Donadelli, J.L.; Saltarelli, W.A.; do Carmo Calijuri, M.; Poli Miwa, A.C.; Tromboni, F.; Dodds, W.K.; et al. Agriculture Influences Ammonium and Soluble Reactive Phosphorus Retention in South American Headwater Streams. Ecohydrology 2020, 13. [Google Scholar] [CrossRef]
| Forest | Cropland | Source of Variation | |||||
|---|---|---|---|---|---|---|---|
| Rainy | Dry | Rainy | Dry | Pland use | Pseason | Pinteraction | |
| Basin area (km2) | 1107 ± 203 | 813 ± 299 | 0.171 | ||||
| Forest cover (%) | 95 ± 2 | 23 ± 1 | 0.013 * | ||||
| Agricultural cover (%) | 2 ± 1 | 75 ± 1 | 0.013 * | ||||
| Discharge (L s−1) | 82 ± 25 | 43 ± 10 | 296 ± 69 | 114 ± 33 | 0.027 * | 0.019 * | 0.495 |
| Stream velocity (m s−1) | 0.12 ± 0.03 | 0.09 ± 0.02 | 0.15 ± 0.08 | 0.14 ± 0.09 | 0.106 | 0.445 | 0.719 |
| Stream slope (m m−1) | na | 0.002 ± 0.000 | na | 0.005 ± 0.001 | <0.001 * | na | na |
| Channel depth (cm) | 33 ± 4 | 24 ± 2 | 32 ± 3 | 31 ± 4 | 0.212 | 0.279 | 0.159 |
| Channel width (cm) | 210 ± 37 | 91 ± 6 | 201 ± 18 | 85 ± 32 | 0.326 | <0.001 * | 0.100 |
| Channel width: depth | 6.2 ± 0.47 | 3.7 ± 0.23 | 6.4 ± 1.19 | 3.1 ± 1.57 | 0.850 | 0.023 * | 0.685 |
| Canopy cover (LAI) | 3.8 ± 0.5 | 6.4 ± 0.3 | 3.1 ± 0.4 | 4.8 ± 0.3 | 0.059 | 0.001 * | 0.131 |
| Water temperature (°C) | 24.6 ± 0.1 | 24.4 ± 0.1 | 26.2 ± 0.3 | 25.7 ± 0.2 | <0.001 * | 0.122 | 0.644 |
| pH | 4.9 ± 0.2 | 4.2 ± 0.2 | 5.3 ± 0.0 | 4.6 ± 0.5 | 0.277 | 0.075 | 0.998 |
| Conductivity (uS cm−1) | 7.4 ± 1.3 | 6.3 ± 0.3 | 5.1 ± 0.2 | 5.4 ± 0.2 | 0.007 * | 0.796 | 0.237 |
| Dissolved oxygen (%) | 62.9 ± 5.5 | 75.8 ± 7.2 | 62.4 ± 10.7 | 80.7 ± 6.5 | 0.957 | 0.081 | 0.680 |
| FBOM (%) | 10 ± 6 | 11 ± 3 | 15 ± 8 | 39 ± 14 | 0.100 | 0.156 | 0.236 |
| Coarse litter (%) | 18 ± 9 | 8 ± 4 | 16 ± 16 | 14 ± 4 | 0.924 | 0.621 | 0.656 |
| Leaf pack (%) | 20 ± 9 | 42 ± 2 | 13 ± 7 | 4 ± 1 | 0.059 | 0.489 | 0.831 |
| Sand (%) | 23 ± 16 | 19 ± 10 | 26 ± 4 | 5 ± 5 | 0.815 | 0.518 | 0.225 |
| Wood (%) | 9 ± 1 | 13 ± 6 | 3 ± 2 | 10 ± 8 | 0.278 | 0.147 | 0.949 |
| Standing Stock | Forest | Cropland | Source of variation | ||||
|---|---|---|---|---|---|---|---|
| (g m−2) | Rainy | Dry | Rainy | Dry | Pland use | Pseason | Pinteraction |
| Leaves | |||||||
| AFDM | 413 ± 73 | 584 ± 155 | 224 ± 84 | 193 ± 5 | 0.011 * | 0.547 | 0.631 |
| C | 213 ± 44 | 303 ± 102 | 103 ± 34 | 93 ± 9 | 0.005 * | 0.522 | 0.609 |
| N | 5.4 ± 0.6 | 8.2 ± 3.3 | 2.6 ± 0.7 | 2.4 ± 0.3 | 0.004 * | 0.524 | 0.610 |
| Wood | |||||||
| AFDM | 503 ± 42 | 433 ± 204 | 217 ± 63 | 167 ± 25 | 0.031 * | 0.370 | 0.757 |
| C | 255 ± 20 | 217 ± 124 | 107 ± 30 | 82 ± 14 | 0.025 * | 0.360 | 0.751 |
| N | 4.5 ± 0.4 | 3.7 ± 1.6 | 1.5 ± 0.3 | 1.1 ± 0.01 | 0.004 * | 0.320 | 0.733 |
| Seed | |||||||
| AFDM | 32 ± 13 | 11 ± 3 | 10 ± 3 | 11 ± 5 | 0.360 | 0.460 | 0.520 |
| C | 16 ± 6.9 | 5.5 ± 1.8 | 5 ± 1.6 | 5 ± 2.7 | 0.327 | 0.429 | 0.520 |
| N | 0.2 ± 0.1 | 0.1 ± 0.03 | 0.1 ± 0.05 | 0.1 ± 0.09 | 0.457 | 0.537 | 0.510 |
| Coarse Benthic Organic Matter (CBOM) | |||||||
| AFDM | 948 ± 125 | 1028 ± 229 | 452 ± 132 | 371 ± 15 | 0.002 * | 0.835 | 0.707 |
| C | 485 ± 61 | 526 ± 142 | 215 ± 56 | 180 ± 3.4 | 0.001 * | 0.846 | 0.701 |
| N | 10 ± 0.2 | 11.9 ± 3.8 | 4.1 ± 0.7 | 3.6 ± 0.37 | <0.001 * | 0.959 | 0.586 |
| Fine Benthic organic matter (FBOM) | |||||||
| AFDM | 590 ± 292 | 929 ± 271 | 492 ± 252 | 471 ± 26 | 0.587 | 0.347 | 0.580 |
| C | 280 ± 180 | 429 ± 142 | 195 ± 102 | 181 ± 5.3 | 0.459 | 0.388 | 0.613 |
| N | 16 ± 10 | 25 ± 8.5 | 12 ± 7.4 | 11.1 ± 1 | 0.506 | 0.400 | 0.622 |
| Stream | Season | Land Use | Discharge (L/s) | Background Chloride (mg/L) | Background Nutrient (mg/L) | Uptake Length (m) | Uptake Rate (g/m2/min) | Uptake Velocity (mm/min) | Proportion Removed |
|---|---|---|---|---|---|---|---|---|---|
| Nitrate (NO3-N) | |||||||||
| APP2 | Wet | Forest | 82.3 | 0.345 | 0.003 | nm | nm | nm | nm |
| APP2a | Wet | Forest | 26.9 | 0.259 | 0.012 | nm | nm | nm | nm |
| APPM | Wet | Forest | 139.1 | 0.370 | 0.001 | 1170.1 | 1203.4 | 2.295 | 0.2 |
| Cascavel | Wet | Cropland | 118.4 | 0.374 | 0.001 | nm | nm | nm | nm |
| Nascente | Wet | Cropland | 105.7 | 0.476 | 0.004 | 546 | 697.2 | 5.6 | 0.17 |
| APP6 | Wet | Cropland | 48.3 | 0.303 | 0.001 | 374.7 | 936.8 | 3.17 | 0.2 |
| APP2 | Dry | Forest | 35.4 | 0.746 | 0.010 | nm | nm | nm | nm |
| APP2a | Dry | Forest | 11.1 | 0.431 | 0.030 | nm | nm | nm | nm |
| APPM | Dry | Forest | 47.1 | 0.438 | 0.027 | nm | nm | nm | nm |
| Cascavel | Dry | Cropland | 41 | 0.243 | 0.010 | nm | nm | nm | nm |
| Nascente | Dry | Cropland | 41.2 | 0.260 | 0.010 | nm | nm | nm | nm |
| APP6 | Dry | Cropland | 16.6 | 0.385 | 0.010 | nm | nm | nm | nm |
| Phosphate (PO4-P) | |||||||||
| APP2 | Wet | Forest | 82.3 | 0.345 | 0.006 | 4585 | 22.11 | 0.74 | 0.02 |
| APP2a | Wet | Forest | 26.9 | 0.259 | 0.004 | 239.4 | 94.3 | 4.59 | 0.47 |
| APPM | Wet | Forest | 139.1 | 0.370 | 0.005 | 564.6 | 260.9 | 4.9 | 0.36 |
| Cascavel | Wet | Cropland | 118.4 | 0.374 | 0.009 | 289 | 85.1 | 14.9 | 0.65 |
| Nascente | Wet | Cropland | 105.7 | 0.476 | 0.009 | 116.3 | 758.1 | 28.1 | 0.58 |
| APP6 | Wet | Cropland | 48.3 | 0.303 | 0.004 | 70.4 | 227.4 | 16.87 | 0.57 |
| APP2 | Dry | Forest | 35.4 | 0.746 | 0.003 | 526.6 | 78.2 | 2.64 | 0.25 |
| APP2a | Dry | Forest | 11.1 | 0.431 | 0.005 | 164.6 | 318.3 | 2.49 | 0.46 |
| APPM | Dry | Forest | 47.1 | 0.438 | 0.006 | nm | nm | nm | nm |
| Cascavel | Dry | Cropland | 41 | 0.243 | 0.053 | 56.6 | 136.6 | 12.9 | 0.80 |
| Nascente | Dry | Cropland | 41.2 | 0.260 | 0.005 | nm | nm | nm | nm |
| APP6 | Dry | Cropland | 16.6 | 0.385 | 0.005 | nm | nm | nm | nm |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jankowski, K.J.; Deegan, L.A.; Neill, C.; Sullivan, H.L.; Ilha, P.; Maracahipes-Santos, L.; Marques, N.; Macedo, M.N. Land Use Change Influences Ecosystem Function in Headwater Streams of the Lowland Amazon Basin. Water 2021, 13, 1667. https://doi.org/10.3390/w13121667
Jankowski KJ, Deegan LA, Neill C, Sullivan HL, Ilha P, Maracahipes-Santos L, Marques N, Macedo MN. Land Use Change Influences Ecosystem Function in Headwater Streams of the Lowland Amazon Basin. Water. 2021; 13(12):1667. https://doi.org/10.3390/w13121667
Chicago/Turabian StyleJankowski, Kathi Jo, Linda A. Deegan, Christopher Neill, Hillary L. Sullivan, Paulo Ilha, Leonardo Maracahipes-Santos, Nubia Marques, and Marcia N. Macedo. 2021. "Land Use Change Influences Ecosystem Function in Headwater Streams of the Lowland Amazon Basin" Water 13, no. 12: 1667. https://doi.org/10.3390/w13121667
APA StyleJankowski, K. J., Deegan, L. A., Neill, C., Sullivan, H. L., Ilha, P., Maracahipes-Santos, L., Marques, N., & Macedo, M. N. (2021). Land Use Change Influences Ecosystem Function in Headwater Streams of the Lowland Amazon Basin. Water, 13(12), 1667. https://doi.org/10.3390/w13121667








