Metallic Pollution and the Use of Antioxidant Enzymes as Biomarkers in Bellamya unicolor (Olivier, 1804) (Gastropoda: Bellamyinae)
Abstract
:1. Introduction
2. Materials and Methods
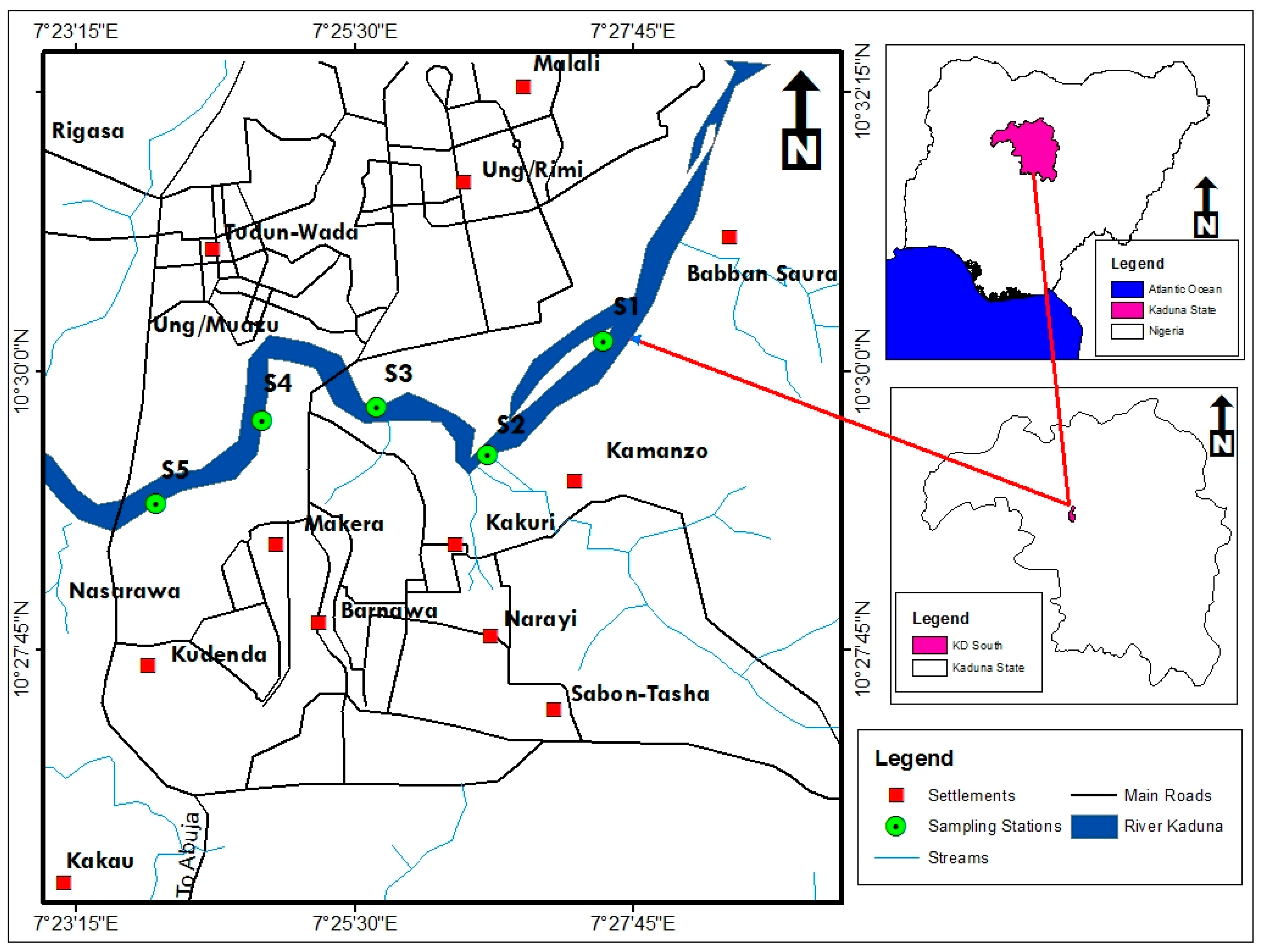
2.1. Study Area
2.2. Collection of Samples
2.3. Treatment of Samples and Determination of Heavy Metals
2.4. Assay of Antioxidant Enzymes Activities
2.4.1. Assay of Catalase (CAT)
2.4.2. Assay of Superoxide Dismutase (SOD)
2.5. Sediment Contamination and Ecological Risk Assessment
2.6. Data Analysis
3. Results
3.1. Concentrations of Heavy Metals in Sediment and Freshwater Snail
3.2. Ecological Risk Assessment of Heavy Metals
3.2.1. Geo-Accumulation Index (Igeo)
3.2.2. Ecological Risk Factor (ErF)
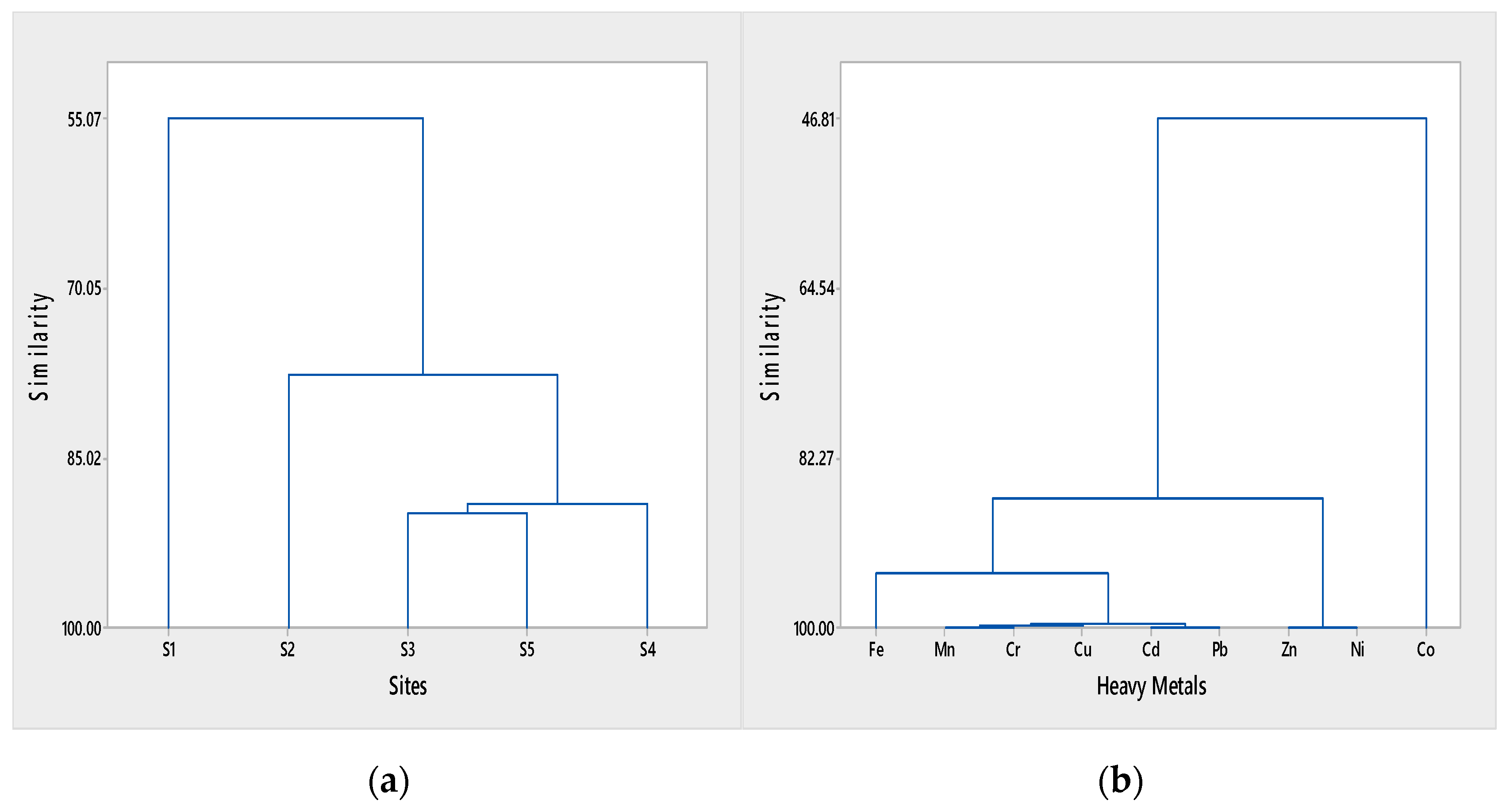
3.3. Identification of Pollution Sources
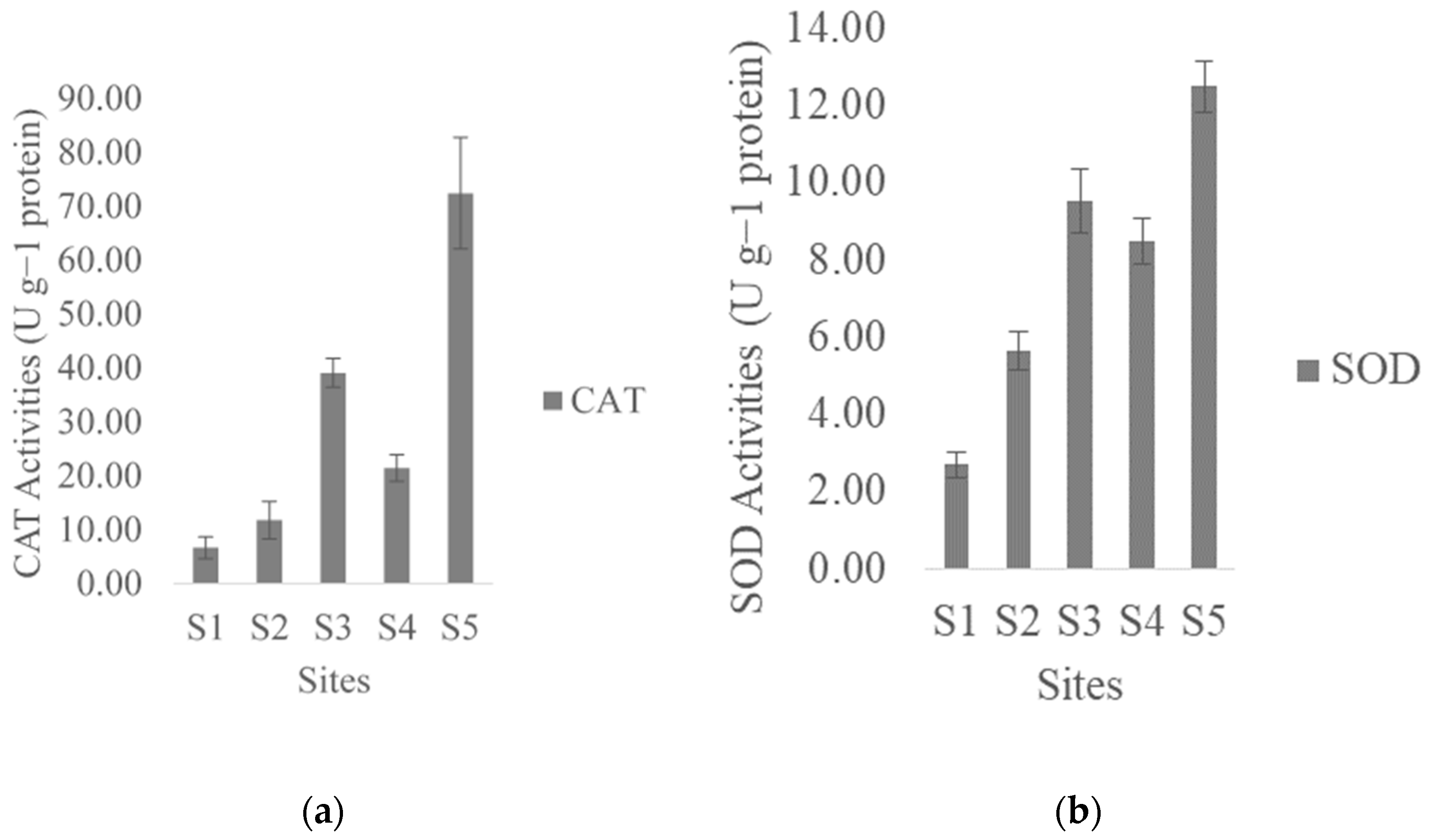
3.4. Antioxidant Enzyme Activities
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bakshi, M.; Ghosh, S.; Chakraborty, D.; Hazra, S.; Chaudhuri, P. Assessment of potentially toxic metal (PTM) pollution in mangrove habitats using biochemical markers: A case study on Avicennia officinalis L. in and around Sundarban, India. Mar. Pollut. Bull. 2018, 133, 157–172. [Google Scholar] [CrossRef]
- Bastami, K.D.; Bagheri, H.; Kheirabadi, V.; Zaferani, G.G.; Teymori, M.B.; Hamzehpoor, A.; Soltani, F.; Haghparast, S.; Harami, S.R.M.; Ghorghani, N.F. Distribution and ecological risk assessment of heavy metals in surface sediments along southeast coast of the Caspian Sea. Mar. Pollut. Bull. 2014, 81, 262–267. [Google Scholar] [CrossRef] [PubMed]
- Xiao, R.; Bai, J.; Lu, Q.; Zhao, Q.; Gao, Z.; Wen, X.; Liu, X. Fractionation, transfer, and ecological risks of heavy metals in riparian and ditch wetlands across a 100-year chronosequence of reclamation in an estuary of China. Sci. Total Environ. 2015, 517, 66–75. [Google Scholar] [CrossRef] [PubMed]
- Ke, X.; Gui, S.; Huang, H.; Zhang, H.; Wang, C.; Guo, W. Ecological risk assessment and source identification for heavy metals in surface sediment from the Liaohe River protected area, China. Chemosphere 2017, 175, 473–481. [Google Scholar] [CrossRef]
- Suresh, G.; Ramasamy, V.; Sundarrajan, M.; Paramasivam, K. Spatial and vertical distributions of heavy metals and their potential toxicity levels in various beach sediments from high-background-radiation area, Kerala, India. Mar. Pollut. Bull. 2015, 91, 389–400. [Google Scholar] [CrossRef] [PubMed]
- Xu, M.; Sun, W.; Wang, R. Spatial Distribution and Ecological Risk Assessment of Potentially Harmful Trace Elements in Surface Sediments from Lake Dali, North China. Water 2019, 11, 2544. [Google Scholar] [CrossRef] [Green Version]
- Hill, N.A.; Simpson, S.L.; Johnston, E.L. Beyond the bed: Effects of metal contamination on recruitment to bedded sediments and overlying substrata. Environ. Pollut. 2013, 173, 182–191. [Google Scholar] [CrossRef] [PubMed]
- Bermejo, J.S.; Beltrán, R.; Ariza, J.G. Spatial variations of heavy metals contamination in sediments from Odiel river (Southwest Spain). Environ. Int. 2003, 29, 69–77. [Google Scholar] [CrossRef]
- Kükrer, S.; Şeker, S.; Abacı, Z.T.; Kutlu, B. Ecological risk assessment of heavy metals in surface sediments of northern littoral zone of Lake Çıldır, Ardahan, Turkey. Environ. Monit. Assess. 2014, 186, 3847–3857. [Google Scholar] [CrossRef]
- Yang, Z.; Wang, Y.; Shen, Z.; Niu, J.; Tang, Z. Distribution and speciation of heavy metals in sediments from the mainstream, tributaries, and lakes of the Yangtze River catchment of Wuhan, China. J. Hazard. Mater. 2009, 166, 1186–1194. [Google Scholar] [CrossRef]
- Yu, G.; Liu, Y.; Yu, S.; Wu, S.; Leung, A.; Luo, X.; Xu, B.; Li, H.; Wong, M.H. Inconsistency and comprehensiveness of risk assessments for heavy metals in urban surface sediments. Chemosphere 2011, 85, 1080–1087. [Google Scholar] [CrossRef] [PubMed]
- Zahra, A.; Hashmi, M.Z.; Malik, R.N.; Ahmed, Z. Enrichment and geo-accumulation of heavy metals and risk assessment of sediments of the Kurang Nallah—Feeding tributary of the Rawal Lake Reservoir, Pakistan. Sci. Total Environ. 2014, 470, 925–933. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.N.; Yuan, X.Z.; Zeng, G.M.; Jiang, M.; Liang, J.; Zhang, C.; Juan, Y.; Huang, H.J.; Liu, Z.F.; Jiang, H.W. Ecological risk assessment of heavy metals in sediments of Xiawan Port based on modified potential ecological risk index. Trans. Nonferrous Met. Soc. China 2012, 22, 1470–1477. [Google Scholar] [CrossRef]
- Rahman, M.S.; Hossain, M.B.; Babu, S.O.F.; Rahman, M.; Ahmed, A.S.; Jolly, Y.; Choudhury, T.; Begum, B.; Kabir, J.; Akter, S. Source of metal contamination in sediment, their ecological risk, and phytoremediation ability of the studied mangrove plants in ship breaking area, Bangladesh. Mar. Pollut. Bull. 2019, 141, 137–146. [Google Scholar] [CrossRef]
- Waykar, B.; Petare, R. Studies on monitoring the heavy metal contents in water, sediment and snail species in Latipada reservoir. J. Environ. Biol. 2016, 37, 585. [Google Scholar]
- Zhou, Q.; Zhang, J.; Fu, J.; Shi, J.; Jiang, G. Biomonitoring: An appealing tool for assessment of metal pollution in the aquatic ecosystem. Anal. Chim. Acta 2008, 606, 135–150. [Google Scholar] [CrossRef]
- Chen, M.; Boyle, E.A.; Switzer, A.D.; Gouramanis, C. A century long sedimentary record of anthropogenic lead (Pb), Pb isotopes and other trace metals in Singapore. Environ. Pollut. 2016, 213, 446–459. [Google Scholar] [CrossRef]
- Shalaby, B.; Samy, Y.M.; Mashaly, A.O.; El Hefnawy, M.A.A. Comparative Geochemical Assessment of Heavy Metal Pollutants among the Mediterranean Deltaic Lakes Sediments (Edku, Burullus and Manzala), Egypt. Egypt. J. Chem. 2017, 60, 361–378. [Google Scholar] [CrossRef] [Green Version]
- Halliwell, B.; Gutteridge, J.M. Free Radicals in Biology and Medicine; Oxford University Press: Oxford, UK, 2015. [Google Scholar]
- Gawad, S.S.A. Concentrations of heavy metals in water, sediment and mollusc gastropod, Lanistes carinatus from Lake Manzala, Egypt. Egypt. J. Aquat. Res. 2018, 44, 77–82. [Google Scholar] [CrossRef]
- Buhler, D.R.; Williams, D.E. The role of biotransformation in the toxicity of chemicals. Aquat. Toxicol. 1988, 11, 19–28. [Google Scholar] [CrossRef]
- Regoli, F.; Principato, G.; Bertoli, E.; Nigro, M.; Orlando, E. Biochemical characterization of the antioxidant system in the scallop Adamussium colbecki, a sentinel organism for monitoring the Antarctic environment. Polar Biol. 1997, 17, 251–258. [Google Scholar] [CrossRef]
- Siwela, A.H.; Nyathi, C.; Naik, Y.S. A comparison of metal levels and antioxidant enzymes in freshwater snails, Lymnaea natalensis, exposed to sediment and water collected from Wright Dam and Lower Mguza Dam, Bulawayo, Zimbabwe. Ecotoxicol. Environ. Saf. 2010, 73, 1728–1732. [Google Scholar] [CrossRef] [PubMed]
- NIMET. Nigeria Meteorological Agency; CRC Press: Kaduna, Nigeria, 2010. [Google Scholar]
- KEPA. Kaduna State Environmental Protection Authority, Revised in 1998:1998. Available online: http://www.kepa.org.ng/ (accessed on 31 December 2019).
- Brown, D.; Kristensen, T. A Field Guide to African Freshwater Snails I. West African species. Dan. Bilharz. Lab. Charlottenlund 1993, 32. [Google Scholar]
- United States Environmental Protection Agency (USEPA). Method 3051 A. Microwave Assisted Acid Digestion of Sediments, Sludge’s, Soils and Oils; USEPA, U.S. Government Printing Office: Washington, DC, USA, 1997. Available online: http://www.epa.gov/SW-846/pdfs/3051a (accessed on 31 December 2019).
- Banaee, M.; Sureda, A.; Taheri, S.; Hedayatzadeh, F. Sub-lethal effects of dimethoate alone and in combination with cadmium on biochemical parameters in freshwater snail, Galba truncatula. Comp. Biochem. Physiol. Part C Toxicol. Pharm. 2019, 220, 62–70. [Google Scholar] [CrossRef] [PubMed]
- Chance, B.; Maehly, A. [136] Assay of catalases and peroxidases. Methods Enzymol. 1955, 2, 764–775. [Google Scholar]
- Kakkar, P.; Das, B.; Viswanathan, P. A modified spectrophotometric assay of superoxide dismutase. Indian J. Biochem. Biophys. 1984, 21, 130–132. [Google Scholar]
- Cheng, H.; Li, M.; Zhao, C.; Yang, K.; Li, K.; Peng, M.; Yang, Z.; Liu, F.; Liu, Y.; Bai, R. Concentrations of toxic metals and ecological risk assessment for sediments of major freshwater lakes in China. J. Geochem. Explor. 2015, 157, 15–26. [Google Scholar] [CrossRef]
- Muller, G. Schwermetalle in den sedimenten des Rheins-Veranderungen seit. Umschav 1979, 79, 133–149. [Google Scholar]
- Wang, Y.; Yang, L.; Kong, L.; Liu, E.; Wang, L.; Zhu, J. Spatial distribution, ecological risk assessment and source identification for heavy metals in surface sediments from Dongping Lake, Shandong, East China. Catena 2015, 125, 200–205. [Google Scholar] [CrossRef]
- Hakanson, L. An ecological risk index for aquatic pollution control. A sedimentological approach. Water Res. 1980, 14, 975–1001. [Google Scholar] [CrossRef]
- Loska, K.; Wiechuła, D. Application of principal component analysis for the estimation of source of heavy metal contamination in surface sediments from the Rybnik Reservoir. Chemosphere 2003, 51, 723–733. [Google Scholar] [CrossRef]
- Ma, X.; Zuo, H.; Tian, M.; Zhang, L.; Meng, J.; Zhou, X.; Min, N.; Chang, X.; Liu, Y. Assessment of heavy metals contamination in sediments from three adjacent regions of the Yellow River using metal chemical fractions and multivariate analysis techniques. Chemosphere 2016, 144, 264–272. [Google Scholar] [CrossRef] [PubMed]
- Chai, M.; Shi, F.; Li, R.; Shen, X. Heavy metal contamination and ecological risk in Spartina alterniflora marsh in intertidal sediments of Bohai Bay, China. Mar. Pollut. Bull. 2014, 84, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Cui, J.; Zang, S.; Zhai, D.; Wu, B. Potential ecological risk of heavy metals and metalloid in the sediments of Wuyuer River basin, Heilongjiang Province, China. Ecotoxicology 2014, 23, 589–600. [Google Scholar] [CrossRef]
- Lin, Q.; Liu, E.; Zhang, E.; Nath, B.; Shen, J.; Yuan, H.; Wang, R. Reconstruction of atmospheric trace metals pollution in Southwest China using sediments from a large and deep alpine lake: Historical trends, sources and sediment focusing. Sci. Total Environ. 2018, 613, 331–341. [Google Scholar] [CrossRef]
- Sutherland, R. Bed sediment-associated trace metals in an urban stream, Oahu, Hawaii. Environ. Geol. 2000, 39, 611–627. [Google Scholar] [CrossRef]
- Liu, E.; Birch, G.F.; Shen, J.; Yuan, H.; Zhang, E.; Cao, Y. Comprehensive evaluation of heavy metal contamination in surface and core sediments of Taihu Lake, the third largest freshwater lake in China. Environ. Earth Sci. 2012, 67, 39–51. [Google Scholar] [CrossRef]
- Zhang, R.; Zhou, L.; Zhang, F.; Ding, Y.; Gao, J.; Chen, J.; Yan, H.; Shao, W. Heavy metal pollution and assessment in the tidal flat sediments of Haizhou Bay, China. Mar. Pollut. Bull. 2013, 74, 403–412. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, S.; Xie, Y.; Wang, X.; Sheng, X.; Chen, J. Distribution and pollution character of heavy metals in the surface sediments of Liao River. Huan Jing Ke Xue Huanjing Kexue 2008, 29, 2413–2418. [Google Scholar]
- Mummullage, N.; Wasanthi, S. Source Characterisation of Urban Road Surface Pollutants for Enhanced Water Quality Predictions. Ph.D. Thesis, Queensland University of Technology, Brisbane, Australia, 2015. [Google Scholar]
- Brady, J.P.; Kinaev, I.; Goonetilleke, A.; Ayoko, G.A. Comparison of partial extraction reagents for assessing potential bioavailability of heavy metals in sediments. Mar. Pollut. Bull. 2016, 106, 329–334. [Google Scholar] [CrossRef]
- Duodu, G.O.; Goonetilleke, A.; Ayoko, G.A. Potential bioavailability assessment, source apportionment and ecological risk of heavy metals in the sediment of Brisbane River estuary, Australia. Mar. Pollut. Bull. 2017, 117, 523–531. [Google Scholar] [CrossRef] [PubMed]
- Brady, J.P. Heavy Metals in the Sediments of Northern Moreton Bay, Queensland, Australia. Ph.D. Thesis, Queensland University of Technology, Brisbane, Australia, 2015. [Google Scholar]
- Liu, R.; Bao, K.; Yao, S.; Yang, F.; Wang, X. Ecological risk assessment and distribution of potentially harmful trace elements in lake sediments of Songnen Plain, NE China. Ecotoxicol. Environ. Saf. 2018, 163, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Hu, J.; Xiong, K.; Huang, X.; Duan, S. Distribution of heavy metals in core sediments from Baihua Lake. Procedia Environ. Sci. 2012, 16, 51–58. [Google Scholar] [CrossRef] [Green Version]
- Suresh, G.; Ramasamy, V.; Meenakshisundaram, V.; Venkatachalapathy, R.; Ponnusamy, V. Influence of mineralogical and heavy metal composition on natural radionuclide concentrations in the river sediments. Appl. Radiat. Isot. 2011, 69, 1466–1474. [Google Scholar] [CrossRef]
- Mohiuddin, K.; Otomo, K.; Ogawa, Y.; Shikazono, N. Seasonal and spatial distribution of trace elements in the water and sediments of the Tsurumi River in Japan. Environ. Monit. Assess. 2012, 184, 265–279. [Google Scholar] [CrossRef]
- Chandra, R.; Yadav, S.; Yadav, S. Phytoextraction potential of heavy metals by native wetland plants growing on chlorolignin containing sludge of pulp and paper industry. Ecol. Eng. 2017, 98, 134–145. [Google Scholar] [CrossRef]
- Varol, M.; Şen, B. Assessment of nutrient and heavy metal contamination in surface water and sediments of the upper Tigris River, Turkey. Catena 2012, 92, 1–10. [Google Scholar] [CrossRef]
- Chung, C.Y.; Chen, J.J.; Lee, C.G.; Chiu, C.Y.; Lai, W.L.; Liao, S.W. Integrated estuary management for diffused sediment pollution in Dapeng Bay and neighboring rivers (Taiwan). Environ. Monit. Assess. 2011, 173, 499–517. [Google Scholar] [CrossRef]
- Li, J.; He, M.; Han, W.; Gu, Y. Analysis and assessment on heavy metal sources in the coastal soils developed from alluvial deposits using multivariate statistical methods. J. Hazard. Mater. 2009, 164, 976–981. [Google Scholar] [CrossRef]
- Peijnenburg, W.J.; Zablotskaja, M.; Vijver, M.G. Monitoring metals in terrestrial environments within a bioavailability framework and a focus on soil extraction. Ecotoxicol. Environ. Saf. 2007, 67, 163–179. [Google Scholar] [CrossRef]
- Harish, S.; Murugan, K. Oxidative stress indices in natural populations of Avicennia alba Blume. as biomarker of environmental pollution. Environ. Res. 2011, 111, 1070–1073. [Google Scholar] [CrossRef] [PubMed]
- Shahid, M.; Pourrut, B.; Dumat, C.; Nadeem, M.; Aslam, M.; Pinelli, E. Heavy-metal-induced reactive oxygen species: Phytotoxicity and physicochemical changes in plants. In Reviews of Environmental Contamination and Toxicology; Springer: New York, NY, USA, 2014; Volume 232, pp. 1–44. [Google Scholar]
- Asaeda, T.; Barnuevo, A. Oxidative stress as an indicator of niche-width preference of mangrove Rhizophora stylosa. Forest Ecol. Manag. 2019, 432, 73–82. [Google Scholar] [CrossRef]
| Fe | Mn | Cu | Zn | Cd | Cr | Pb | Ni | Co | |
|---|---|---|---|---|---|---|---|---|---|
| CSd | 90.8225 | 42.2875 | 31.285 | 33.5475 | 29.4975 | 46.67625 | 67.66875 | 31.6575 | 34.6275 |
| Range | 34.54–165.32 | 6.36–78.98 | 1.63–59.01 | 4.50–61.48 | 0.96–58.84 | 16.09–79.20 | 39.43–96.08 | 2.64–60.08 | 11.42–62.61 |
| CSn | 20.8825 | 9.0625 | 7.295 | 7.555 | 7.4225 | 10.3725 | 15.0375 | 7.27 | 7.435 |
| Range | 6.61–33.30 | 1.34–22.75 | 0.54–16.51 | 1.74–18.63 | 0.92–17.52 | 4.26–17.24 | 6.24–24.20 | 0.63–14.17 | 1.6–21.46 |
| TEL | NA | NA | 35.7 | 123 | 0.596 | 37.3 | 35 | 18 | NA |
| PEL | NA | NA | 197 | 315 | 3.53 | 90 | 91.3 | 36 | NA |
| SQGs | Fe | Mn | Cu | Zn | Cd | Cr | Pb | Ni | Co |
|---|---|---|---|---|---|---|---|---|---|
| % of samples < TEL | NA | NA | 53.6 | 100 | 0 | 42.6 | 0 | 38.8 | NA |
| % of samples btw TEL-PEL | NA | NA | 46.4 | 0 | 20.8 | 57.4 | 100 | 61.2 | NA |
| % of samples > PEL | NA | NA | 0 | 0 | 79.2 | 0 | 0 | 0 | NA |
| Igeo | ||||||||
|---|---|---|---|---|---|---|---|---|
| Sites | Mn | Cu | Zn | Cd | Cr | Pb | Ni | Co |
| S1 | 11.81 | 5.61 | 8.15 | −2.39 | 9.89 | 9.00 | 6.42 | 5.78 |
| S2 | 12.47 | 7.04 | 8.40 | −2.91 | 10.31 | 9.03 | 7.51 | 7.13 |
| S3 | 15.34 | 10.79 | 11.96 | 3.53 | 12.09 | 10.31 | 11.40 | 9.62 |
| S4 | 13.68 | 8.18 | 10.07 | 0.33 | 10.96 | 9.72 | 9.24 | 7.84 |
| S5 | 15.44 | 10.79 | 11.93 | 3.56 | 12.21 | 10.32 | 11.41 | 9.63 |
| Mean | 13.75 | 8.48 | 10.10 | 0.42 | 11.09 | 9.68 | 9.19 | 8.00 |
| Level | 6 | 6 | 6 | 1 | 6 | 6 | 6 | 6 |
| (Individual Metal) | (Multi-Metal) ErF | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Sites | Mn | Cu | Zn | Cd | Cr | Pb | Ni | Co | ErF |
| S1 | 0.01 | 0.02 | 0.03 | 35.86 | 0.32 | 2.27 | 0.38 | 1.32 | 40.21 |
| S2 | 0.02 | 0.07 | 0.04 | 25.31 | 0.43 | 2.32 | 0.58 | 3.26 | 32.03 |
| S3 | 0.16 | 0.62 | 0.44 | 2160.00 | 1.45 | 5.60 | 8.51 | 17.74 | 2194.52 |
| S4 | 0.09 | 0.32 | 0.24 | 1103.20 | 0.88 | 4.07 | 4.56 | 9.26 | 1122.61 |
| S5 | 0.17 | 0.62 | 0.45 | 2206.41 | 1.58 | 5.65 | 8.58 | 17.89 | 2241.35 |
| Mean | 0.09 | 0.33 | 0.24 | 1106.16 | 0.93 | 3.98 | 4.52 | 9.89 | 1126.14 |
| Fe | Mn | Cu | Zn | Cd | Cr | Pb | Ni | Co | SOD | CAT | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Fe | 1 | ||||||||||
| Mn | 0.584 | 1 | |||||||||
| Cu | 0.777 ** | 0.567 | 1 | ||||||||
| Zn | 0.671 * | 0.984 ** | 0.619 | 1 | |||||||
| Cd | 0.728 * | 0.971 ** | 0.660 * | 0.985 ** | 1 | ||||||
| Cr | 0.632 | 0.988 ** | 0.555 | 0.993 ** | 0.981 ** | 1 | |||||
| Pb | 0.765 ** | 0.592 | 0.994 ** | 0.638 * | 0.671 * | 0.572 | 1 | ||||
| Ni | 0.672 * | 0.978 ** | 0.622 | 0.993 ** | 0.988 ** | 0.992 ** | 0.634 * | 1 | |||
| Co | 0.640 * | 0.982 ** | 0.585 | 0.993 ** | 0.983 ** | 0.987 ** | 0.608 | 0.986 ** | 1 | ||
| SOD | 0.809 ** | 0.822 ** | 0.845 ** | 0.827 ** | 0.877 ** | 0.809 ** | 0.860 ** | 0.832 ** | 0.822 ** | 1 | |
| CAT | 0.567 | 0.779 ** | 0.758 * | 0.765 ** | 0.769 ** | 0.735 * | 0.801 ** | 0.759 * | 0.777 ** | 0.904 ** | 1 |
| Component | |||
|---|---|---|---|
| Heavy Metal | PC1 | PC2 | PC3 |
| Fe_S | 0.74 | 0.40 | 0.03 |
| Mn_S | 0.97 | −0.20 | −0.02 |
| Cu_S | 0.73 | −0.23 | 0.01 |
| Zn_S | 0.98 | −0.17 | −0.02 |
| Cd_S | 0.99 | −0.10 | 0.00 |
| Cr_S | 0.97 | −0.23 | −0.02 |
| Pb_S | 0.75 | −0.17 | 0.05 |
| Ni_S | 0.98 | −0.16 | 0.03 |
| Co_S | 0.97 | −0.22 | 0.30 |
| Fe_Sd | 0.85 | 0.26 | 0.21 |
| Mn_Sd | 0.97 | −0.18 | 0.07 |
| Cu_Sd | 0.97 | 0.63 | −0.01 |
| Zn_Sd | 0.68 | 0.36 | −0.05 |
| Cd_Sd | 0.98 | −0.18 | −0.05 |
| Cr_Sd | 0.98 | −0.16 | 0.01 |
| Pb_Sd | 0.97 | 0.60 | −0.09 |
| Ni_Sd | 0.67 | 0.33 | −0.52 |
| Co_Sd | 0.24 | 0.07 | 0.95 |
| Eigenvalue | 13.82 | 1.59 | 1.24 |
| Total variance (%) | 76.80 | 8.81 | 6.89 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aljahdali, M.O.; Alhassan, A.B. Metallic Pollution and the Use of Antioxidant Enzymes as Biomarkers in Bellamya unicolor (Olivier, 1804) (Gastropoda: Bellamyinae). Water 2020, 12, 202. https://doi.org/10.3390/w12010202
Aljahdali MO, Alhassan AB. Metallic Pollution and the Use of Antioxidant Enzymes as Biomarkers in Bellamya unicolor (Olivier, 1804) (Gastropoda: Bellamyinae). Water. 2020; 12(1):202. https://doi.org/10.3390/w12010202
Chicago/Turabian StyleAljahdali, Mohammed Othman, and Abdullahi Bala Alhassan. 2020. "Metallic Pollution and the Use of Antioxidant Enzymes as Biomarkers in Bellamya unicolor (Olivier, 1804) (Gastropoda: Bellamyinae)" Water 12, no. 1: 202. https://doi.org/10.3390/w12010202
APA StyleAljahdali, M. O., & Alhassan, A. B. (2020). Metallic Pollution and the Use of Antioxidant Enzymes as Biomarkers in Bellamya unicolor (Olivier, 1804) (Gastropoda: Bellamyinae). Water, 12(1), 202. https://doi.org/10.3390/w12010202






