Amendment of Husk Biochar on Accumulation and Chemical Form of Cadmium in Lettuce and Pak-Choi Grown in Contaminated Soil
Abstract
1. Introduction
2. Materials and Methods
3. Results
3.1. Soil Properties
3.2. Soil Cd Fraction and Growth Exhibition
3.3. Cd Accumulation
3.4. Chemical Form
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hseu, Z.Y.; Su, S.W.; Lai, H.Y.; Guo, H.Y.; Chen, T.C.; Chen, Z.S. Remediation techniques and heavy metal uptake by different rice varieties in metal-contaminated soils of Taiwan: New aspects for food safety regulation and sustainable agriculture. Soil Sci. Plant Nutr. 2010, 56, 31–52. [Google Scholar] [CrossRef]
- SGWPR Act (Soil and Groundwater Pollution Remediation Act). Available online: https://sgw.epa.gov.tw/en/laws_policy/laws/458db6eb-5602-46b2-9471-745d58078aaf (accessed on 5 March 2020).
- Di Toppi, L.S.; Gabbrielli, R. Response to cadmium in higher plants. Environ. Exp. Bot. 1999, 41, 105–130. [Google Scholar] [CrossRef]
- Chen, D.; Guo, H.; Li, R.; Li, L.; Pan, G.; Chang, A.; Joseph, S. Low uptake affinity cultivars with biochar to tackle Cd-tainted rice—A field study over four rice seasons in Hunan, China. Sci. Total Environ. 2016, 541, 1489–1498. [Google Scholar] [CrossRef]
- Ali, H.; Khan, E.; Ilahi, I. Environmental chemistry and ecotoxicology of hazardous heavy metals: Environmental persistence, toxicity, and bioaccumulation. J. Chem. 2019, 2019, 1–14. [Google Scholar] [CrossRef]
- Barraza, F.; Schreck, E.; Lévêque, T.; Uzu, G.; López, F.; Ruales, J.; Prunier, J.; Marquet, A.; Maurice, L. Cadmium bioaccumulation and gastric bioaccessibility in cacao: A field study in areas impacted by oil activities in Ecuador. Environ. Pollut. 2017, 229, 950–963. [Google Scholar] [CrossRef] [PubMed]
- Kovacevic, G.; Kastori, R.; Merkulov, L. Dry matter and leaf structure in young wheat plants as affected by cadmium, lead, and nickel. Biol. Plant. 1999, 42, 119–123. [Google Scholar] [CrossRef]
- Liu, Y.T.; Chen, Z.S.; Hong, C.Y. Cadmium-induced physiological response and antioxidant enzyme changes in the novel cadmium accumulator, Tagetes patula. J. Hazard. Mater. 2011, 189, 724–731. [Google Scholar] [CrossRef]
- Xu, D.Y.; Chen, Z.F.; Sun, K.; Yan, D.; Kang, M.J.; Zhao, Y. Effect of cadmium on the physiological parameters and the subcellular cadmium localization in the potato (Solanum tuberosum L.). Ecotox. Environ. Safe. 2013, 97, 147–153. [Google Scholar] [CrossRef]
- Lasat, M.M.; Baker, A.J.M.; Kochian, L.V. Physiological characterization of root Zn2+ absorption and translocation to shoots in Zn hyperaccumulator and nonaccumulator species of Thlaspi. Plant Physiol. 1996, 112, 1715–1722. [Google Scholar] [CrossRef]
- Lasat, M.M.; Fuhrmann, M.; Ebbs, S.D.; Cornish, J.E.; Kochian, L.V. Phytoremediation of a radiocesium-contaminated soil: Evaluation of cesium-137 bioaccumulation in the shoots of tree plant species. J. Environ. Qual. 1998, 7, 165–169. [Google Scholar] [CrossRef]
- Küpper, H.; Zhao, F.J.; McGrath, S.P. Cellular compartmentation of zinc in leaves of the hyperaccumulator Thlaspi caerulescens. Plant Physiol. 1999, 119, 305–311. [Google Scholar] [CrossRef] [PubMed]
- Ge, W.; Jiao, Y.Q.; Sun, B.L.; Qin, R.; Jiang, W.S.; Liu, D.H. Cadmium-mediated oxidative stress and ultrastructural changes in root cells of poplar cultivars. S. Afr. J. Bot. 2012, 83, 98–108. [Google Scholar] [CrossRef][Green Version]
- Haynes, R.J. Ion exchange properties of roots and ionic interactions within root apoplasm: Their role in ion accumulation by plants. Bot. Rev. 1980, 46, 75–99. [Google Scholar] [CrossRef]
- Zhao, Y.F.; Wu, J.F.; Shang, D.R.; Ning, J.S.; Zhai, Y.X.; Shend, X.F.; Ding, H.Y. Subcellular distribution and chemical forms of cadmium in the edible seaweed, Porphyra yezoensis. Food Chem. 2015, 168, 48–54. [Google Scholar] [CrossRef] [PubMed]
- Wu, F.B.; Dong, J.; Qian, Q.Q.; Zhang, G.P. Subcellular distribution and chemical form of Cd and Ca-Zn interaction in different barley genotypes. Chemosphere 2005, 60, 1437–1446. [Google Scholar] [CrossRef]
- Su, Y.; Liu, J.L.; Lu, Z.W.; Wang, X.M.; Zhang, Z.; Shi, G.G. Effects of iron deficiency on subcellular distribution and chemical forms of cadmium in peanut roots in relation to its translocation. Environ. Exp. Bot. 2014, 97, 40–48. [Google Scholar] [CrossRef]
- Fu, X.; Dou, C.; Chen, Y.; Chen, X.; Shi, J. Subcellular distribution and chemical forms of cadmium in Phytplacca americana L. J. Hazard. Mater. 2011, 186, 103–107. [Google Scholar] [CrossRef]
- Wang, X.; Liu, Y.G.; Zeng, G.M.; Chai, L.Y.; Song, X.C.; Min, Z.Y.; Xiao, X. Subcellular distribution and chemical forms of cadmium in Bechmeria nivea (L.) Gaud. Environ. Exp. Bot. 2008, 62, 389–395. [Google Scholar] [CrossRef]
- Qiu, Q.; Wang, Y.; Yang, Z.; Yuan, J. Effects of phosphorus supplied in soil on subcellular distribution and chemical forms in two Chinese flowering cabbage (Brassica parachinensis L.) cultivars differing in cadmium accumulation. Food Chem. Toxicol. 2011, 49, 2260–2267. [Google Scholar] [CrossRef]
- Lai, H.Y. Subcellular distribution and chemical forms of cadmium in Impatiens walleriana in relation to its phytoextraction potential. Chemosphere 2015, 138, 370–376. [Google Scholar] [CrossRef]
- Garau, G.; Castaldi, C.; Santona, L.; Deiana, P.; Melis, P. Influence of red mud, zeolite and lime on heavy metal immobilization, culturable heterotrophic microbial populations and enzyme activities in a contaminated soil. Geoderma 2007, 142, 47–57. [Google Scholar] [CrossRef]
- Cao, X.; Dermatas, D.; Xu, X.; Shen, G. Immobilization of lead in shooting range soils by means of cement, quicklime, and phosphate amendments. Environ. Sci. Pollut. Res. 2008, 15, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Liu, N.; Jiang, Z.; Li, X.; Liu, H.; Li, N.; Wei, S. Mitigation of rice cadmium (Cd) accumulation by joint application of organic amendments and selenium (Se) in high-Cd-contaminated soils. Chemosphere 2020, 241, 125106. [Google Scholar] [CrossRef] [PubMed]
- Shuman, L. Organic waste amendments effect on zinc fraction of two soils. J. Environ. Qual. 1999, 28, 1442–1447. [Google Scholar] [CrossRef]
- Egene, C.E.; Van Poucke, R.; OK, Y.S.; Meers, E.; Tack, F.M.G. Impact of organic amendments (biochar, compost and peat) on Cd and Zn mobility and solubility in contaminated soil of the Campine region after three years. Sci. Total Environ. 2018, 626, 195–202. [Google Scholar] [CrossRef]
- Ahmad, M.; Rajapaksha, A.U.; Lim, J.E.; Zhang, M.; Bolan, N.; Mohan, D.; Vithanage, M.; Lee, S.S.; Ok, Y.S. Biochar as a sorbent for contaminant management in soil and water: A review. Chemosphere 2014, 99, 19–33. [Google Scholar] [CrossRef]
- Moreno-Jiménez, E.; Esteban, E.; Carpena-Ruiz, R.O.; Lobo, M.C.; Rénalos, J.M. Phytostabilisation with Mediterranean shrubs and liming improved soil quality in a pot experiment with a pyrite mine soil. J. Hazard. Mater. 2012, 201–202, 52–59. [Google Scholar] [CrossRef]
- Xu, C.; Chen, H.X.; Xiang, Q.; Zhu, H.H.; Wang, S.; Zhu, Q.H.; Huang, D.Y.; Zhang, Y.Z. Effect of peanut shell and wheat straw biochar on the availability of Cd and Pb in a soil-rice (Oryza sativa L.) system. Environ. Sci. Pollut. Res. 2017, 25, 1147–1156. [Google Scholar] [CrossRef]
- Wang, Y.M.; Tang, D.D.; Zhang, X.H.; Uchimiya, M.; Yuan, X.Y.; Li, M.; Chen, Y.Z. Effects of soil amendments on cadmium transfer along the lettuce-snail food chain: Influence of chemical speciation. Sci. Total Environ. 2019, 649, 801–807. [Google Scholar] [CrossRef]
- Zhang, X.Y.; Zhang, Y.M.; Liu, X.Y.; Zhang, C.Y.; Dong, S.D.; Liu, Q.; Deng, M. Cd uptake by Phytolacca americana L. promoted by cornstalk biochar amendments in Cd-contaminated soil. Int. J. Phytorem. 2019, 22, 251–258. [Google Scholar] [CrossRef]
- Chen, Z.S.; Lee, D.Y. Evaluation of remediation techniques on two cadmium-polluted soils in Taiwan. In Remediation of Soils Contaminated with Metals; Iskander, I.K., Adriano, D.C., Eds.; Science Reviews: Northwood, UK, 1997; pp. 209–223. [Google Scholar]
- Thomas, G.W. Soil pH and soil acidity. In Methods of Soil Analysis. Part. 3 Chemical Methods; Sparks, D.L., Page, A.L., Helmke, P.A., Loeppert, R.H., Soltanpour, P.N., Tabatabai, M.A., Johnston, C.T., Sumner, M.E., Eds.; SSSA Inc./ASA Inc.: Madison, WI, USA, 1996; pp. 475–490. [Google Scholar]
- Rhoades, J.D. Salinity: Electrical conductivity and total dissolved solids. In Methods of Soil Analysis. Part 3. Chemical Methods; Sparks, D.L., Page, A.L., Helmke, P.A., Loeppert, R.H., Soltanpour, P.N., Tabatabai, M.A., Johnston, C.T., Sumner, M.E., Eds.; SSSA Inc./ASA Inc.: Madison, WI, USA, 1996; pp. 417–435. [Google Scholar]
- Nelson, D.W.; Sommers, L.E. Total carbon, organic carbon, and organic matter. In Methods of Soil Analysis. Part 3. Chemical Methods; Sparks, D.L., Page, A.L., Helmke, P.A., Loeppert, R.H., Soltanpour, P.N., Tabatabai, M.A., Johnston, C.T., Sumner, M.E., Eds.; SSSA Inc./ASA Inc.: Madison, WI, USA, 1996; pp. 961–1010. [Google Scholar]
- Sumners, M.E.; Miller, W.P. Cation exchange capacity and exchange coefficients. In Methods of Soil Analysis. Part 3. Chemical Methods; Sparks, D.L., Page, A.L., Helmke, P.A., Loeppert, R.H., Soltanpour, P.N., Tabatabai, M.A., Johnston, C.T., Sumner, M.E., Eds.; SSSA Inc./ASA Inc.: Madison, WI, USA, 1996; pp. 1201–1229. [Google Scholar]
- Gee, G.W.; Bauder, J.W. Particle-size analysis. In Methods of Soil Analysis. Part 1. Physical and Mineralogical Method, 2nd ed.; Klute, A., Ed.; SSSA Inc./ASA Inc.: Madison, WI, USA, 1986; pp. 383–412. [Google Scholar]
- EPA/Taiwan. Method Code No: NIEA S321.65B; Environmental Protection Administration of Taiwan ROC: Taipei, Taiwan, 2018.
- Bremner, J.M. Nitrogen–Total. In Methods of Soil Analysis. Part 3. Chemical Methods; Sparks, D.L., Page, A.L., Helmke, P.A., Loeppert, R.H., Soltanpour, P.N., Tabatabai, M.A., Johnston, C.T., Sumner, M.E., Eds.; SSSA Inc./ASA Inc.: Madison, WI, USA, 1996; pp. 1085–1121. [Google Scholar]
- Kuo, S. Phosphorus. In Methods of Soil Analysis. Part 3. Chemical Methods; Sparks, D.L., Page, A.L., Helmke, P.A., Loeppert, R.H., Soltanpour, P.N., Tabatabai, M.A., Johnston, C.T., Sumner, M.E., Eds.; SSSA Inc./ASA Inc.: Madison, WI, USA, 1996; pp. 869–919. [Google Scholar]
- Helmke, P.A.; Sparks, D.L. Lithium, Sodium, Potassium, Rubidium, and Cesium. In Methods of Soil Analysis. Part 3. Chemical Methods; Sparks, D.L., Page, A.L., Helmke, P.A., Loeppert, R.H., Soltanpour, P.N., Tabatabai, M.A., Johnston, C.T., Sumner, M.E., Eds.; SSSA Inc./ASA Inc.: Madison, WI, USA, 1996; pp. 551–574. [Google Scholar]
- De Medici, D.; Komínková, D.; Race, M.; Fabbricino, M.; Součková, L. Evaluation of the potential for caesium transfer from contaminated soil to the food chain as a consequence of uptake by edible vegetables. Ecotox. Environ. Safe 2019, 171, 558–563. [Google Scholar] [CrossRef] [PubMed]
- Yu, T.H.; Hsieh, S.P.; Su, C.M.; Huang, F.J.; Hung, C.C.; Yiin, L.M. Analysis of Leafy Vegetable Nitrate Using a Modified Spectrometric Method. Int. J. Anal. Chem. 2018, 2018, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Baker, D.E.; Amacher, M.C. Nickel, copper, zinc, and cadmium. In Methods of Soil Analysis. Part 2. Chemical and Microbiological Properties, 2nd ed.; Page, A.L., Millers, R.H., Keeney, D.R., Eds.; SSSA Inc./ASA Inc.: Madison, WI, USA, 1982; pp. 323–336. [Google Scholar]
- Mench, M.J.; Didier, V.L.; Loffler, M.; Gomez, A.; Masson, P. A mimicked in-situ remediation study of metal-contaminated soils with emphasis on cadmium and lead. J. Environ. Qual. 1994, 23, 58–63. [Google Scholar] [CrossRef]
- Ure, A.M. Methods of analysis of heavy metals in soils. In Heavy Metals in Soils, 2nd ed.; Alloway, B.J., Ed.; Blackie Academic and Professional: London, UK, 1995; pp. 58–102. [Google Scholar]
- Lam, C.M.; Lai, H.Y. Effect of inoculation with arbuscular mycorrhizal fungi and blanching on the bioaccessibility of heavy metals in water spinach (Ipomoea aquatica Forsk.). Ecotox. Environ. Safe 2018, 162, 563–570. [Google Scholar] [CrossRef]
- European Commission (EC). Commission Regulation (EU) No 420/2011 of 29 April 2011 amending Regulation (EC) No 1881/2006 setting maximum levels for certain contaminants in foodstuffs. Off. J. Eur. Union 2011, 11, 3–6. [Google Scholar]
- Lai, H.Y.; Chen, B.C. The dynamic growth exhibition and accumulation of cadmium of pak choi grown in contaminated soils. Int. J. Environ. Res. Public Health 2013, 10, 5284–5298. [Google Scholar] [CrossRef]
- Antoniadis, V.; Shaheen, S.M.; Boersch, J.; Frohne, T.; Laing, G.D.; Rinklebe, J. Bioavailability and risk assessment of potentially toxic elements in garden edible vegetables and soils around a highly contaminated former mining area in Germany. J. Environ. Manag. 2017, 186, 192–200. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority. Scientific Opinion of the Panel on Contaminants in the Food Chain on a request from the European Commission of cadmium in food. ESFA J. 2009, 980, 1–139. [Google Scholar]
- Kim, K.; Melough, M.M.; Vance, T.M.; Noh, H.; Koo, S.I.; Chun, O.K. Dietary cadmium intake and sources in the US. Nutrients 2019, 11, 2. [Google Scholar] [CrossRef]
- Lam, C.M.; Chen, K.S.; Lai, H.Y. Chemical forms and health risk of cadmium in water spinach grown in contaminated soil with an increased level of phosphorus. Int. J. Environ. Res. Public Health 2019, 16, 3322. [Google Scholar] [CrossRef]
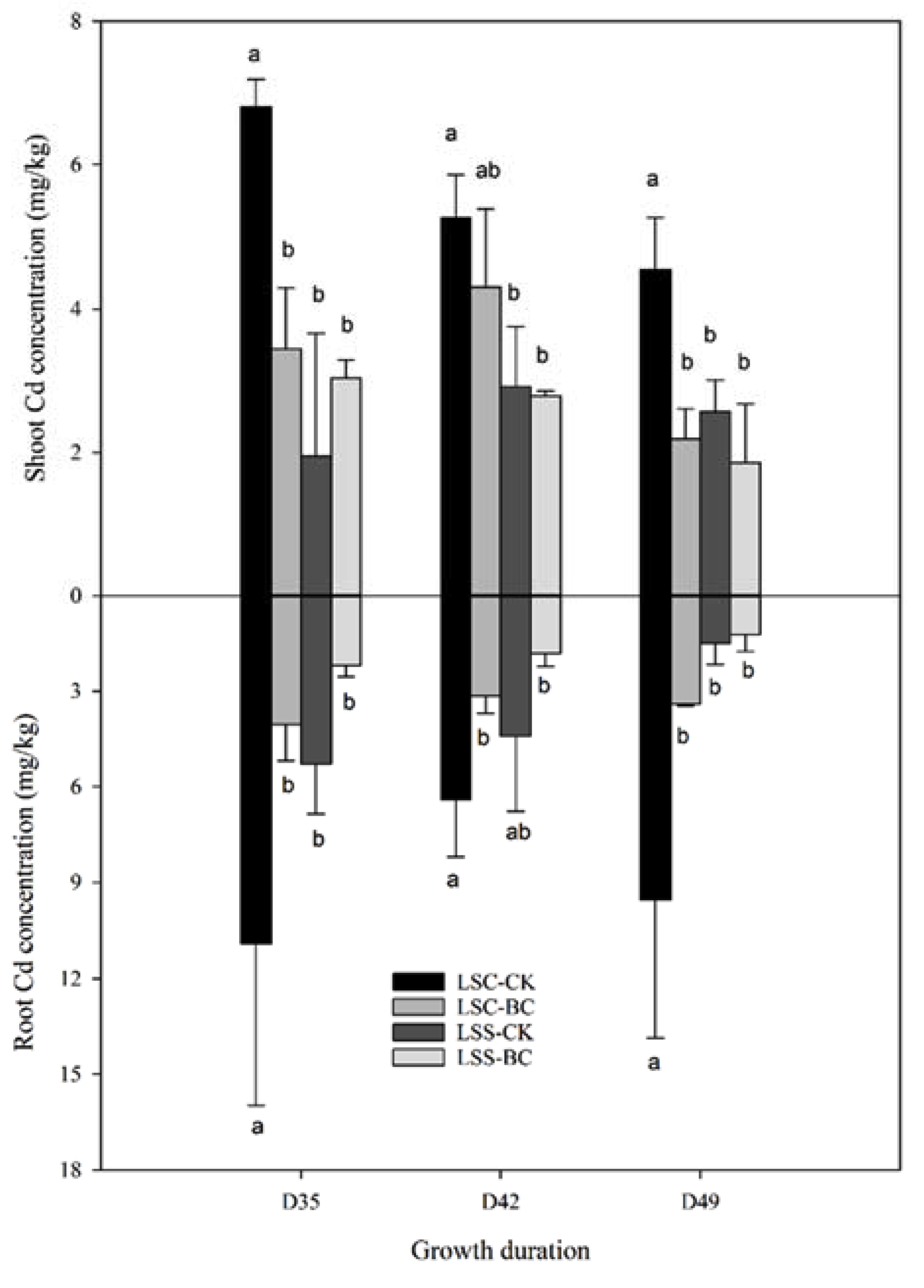
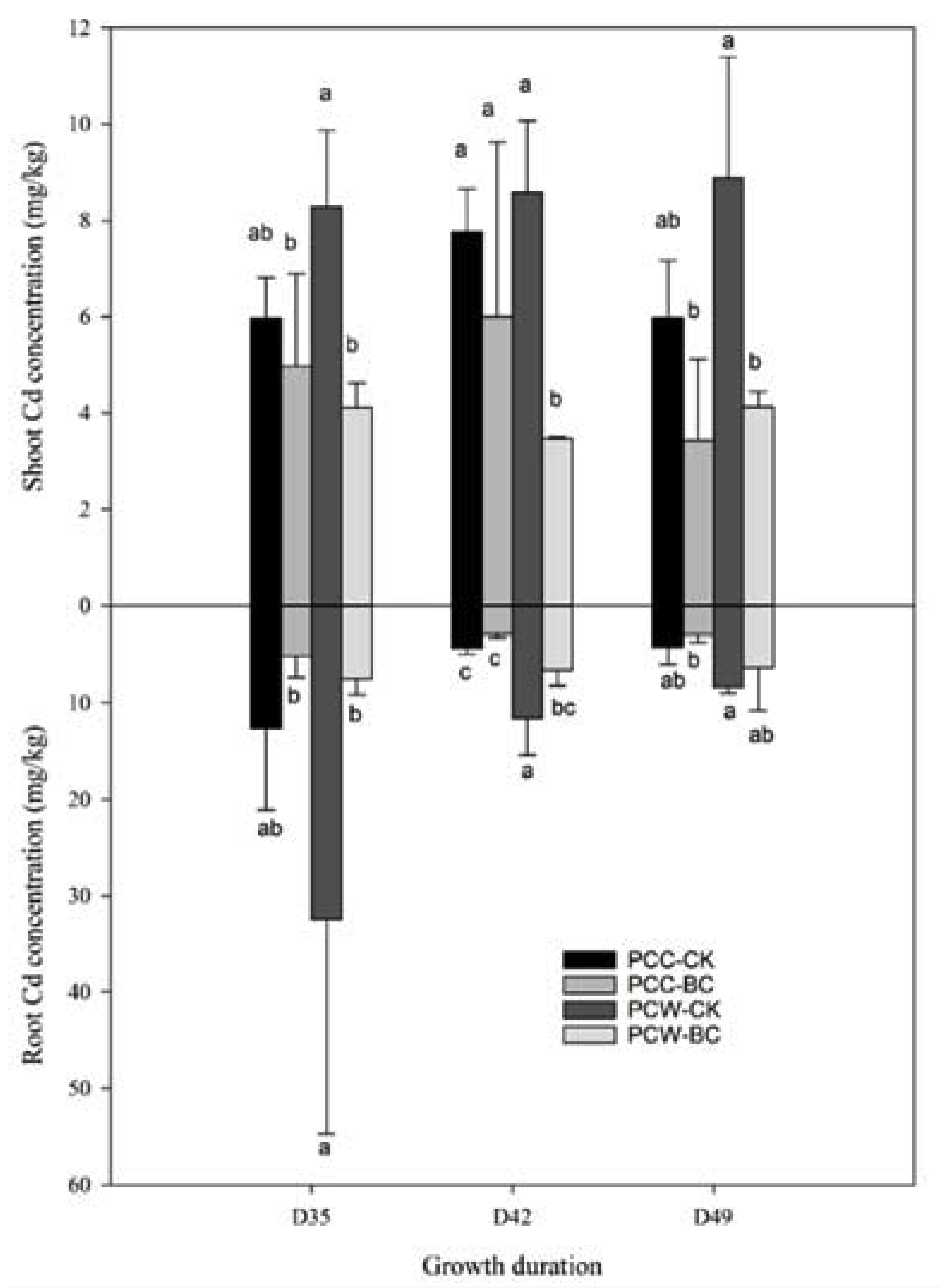
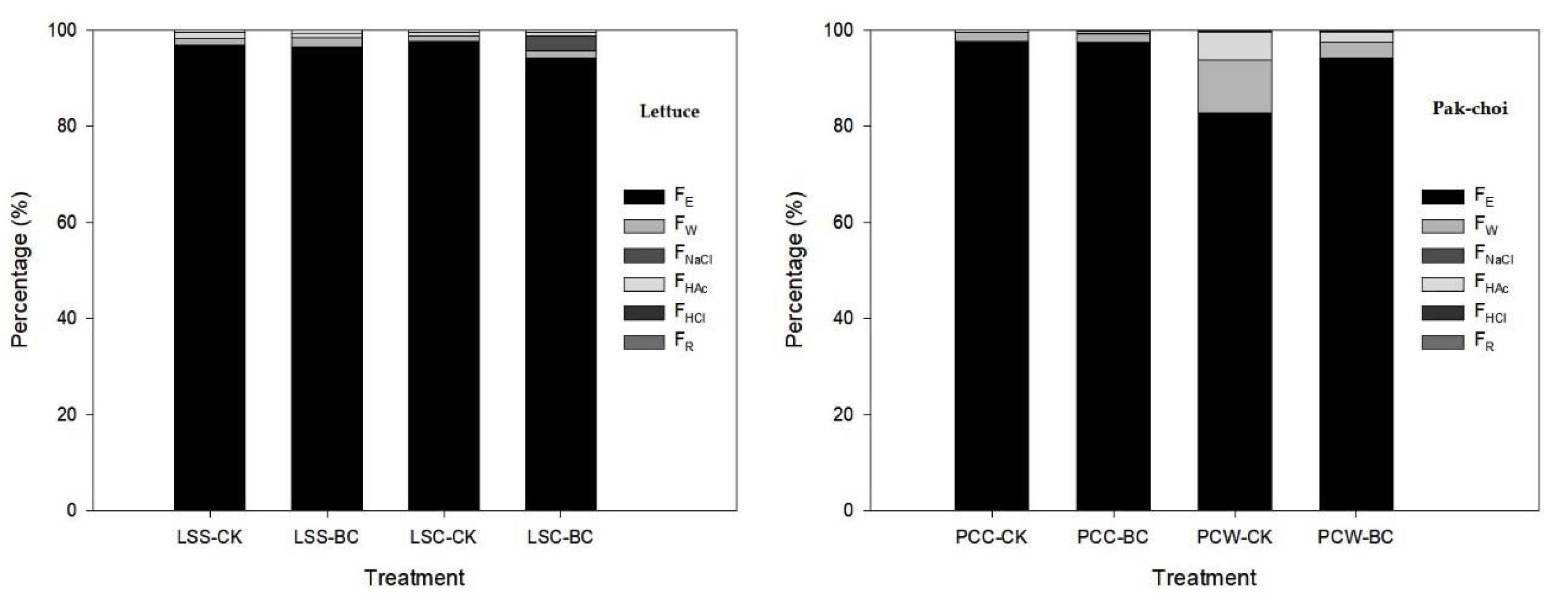
| Soil Property and Cd Concentration 1 | ||||||||
|---|---|---|---|---|---|---|---|---|
| Treatment 2 | pH | Electrical Conductivity | Extracting Agent | BCR Sequestration Extraction 3 | ||||
| 0.1 N HCl | 0.05 N EDTA | F-I | F-II | F-III | F-IV | |||
| dS/m | mg/kg | % | ||||||
| PCC-CK | 6.90 ± 0.15b | 0.03 ± 0.00b | 6.63 ± 0.24a | 6.81 ± 0.25a | 55.99 ± 0.39b | 26.37 ± 0.23c | 16.18 ± 0.22b | 1.46 ± 0.04b |
| PCC-BC | 7.93 ± 0.18a | 0.12 ± 0.02a | 6.98 ± 0.61a | 7.03 ± 0.38a | 64.67 ± 0.31a | 26.61 ± 0.07c | 7.63 ± 0.10c | 1.08 ± 0.03c |
| PCW-CK | 6.90 ± 0.20b | 0.05 ± 0.01b | 7.23 ± 0.68a | 7.01 ± 0.39a | 52.57 ± 0.34c | 31.00 ± 0.10a | 16.44 ± 0.17ab | ND |
| PCW-BC | 7.62 ± 0.46a | 0.17 ± 0.06ab | 6.91 ± 0.56a | 6.27 ± 0.34b | 51.93 ± 0.29d | 29.91 ± 0.15b | 16.56 ± 0.22a | 1.60 ± 0.03a |
| LSC-CK | 6.69 ± 0.11a | 0.07 ± 0.02a | 7.70 ± 0.62a | 7.41 ± 0.85a | 61.34 ± 0.27a | 20.86 ± 0.19c | 17.80 ± 0.23b | ND |
| LSC-BC | 6.81 ± 0.16a | 0.16 ± 0.08a | 7.00 ± 0.64a | 6.82 ± 0.56a | 37.70 ± 0.39d | 45.26 ± 0.11a | 17.04 ± 0.22c | ND |
| LSS-CK | 6.77 ± 0.11a | 0.05 ± 0.01a | 6.90 ± 0.88a | 7.62 ± 0.30a | 38.90 ± 0.28c | 36.51 ± 0.32b | 21.84 ± 0.16a | 2.76 ± 0.05 |
| LSS-BC | 7.02 ± 0.23a | 0.06 ± 0.00a | 6.55 ± 1.18a | 6.64 ± 0.94a | 57.32 ± 0.44b | 20.88 ± 0.12c | 21.80 ± 0.25a | ND |
| Growth Exhibition | Pak-choi 2 | Lettuce 2 | ||||||
|---|---|---|---|---|---|---|---|---|
| PCC-CK | PCC-BC | PCW-CK | PCW-BC | LSC-CK | LSC-BC | LSS-CK | LSS-BC | |
| Root length (cm) | 4.61 ± 1.49a | 8.26 ± 3.52a | 3.59 ± 0.20a | 4.66 ± 0.95a | 7.59 ± 1.30a | 6.96 ± 0.21a | 7.71 ± 0.45a | 8.09 ± 1.38a |
| Shoot height (cm) | 10.32 ± 2.75a | 12.19 ± 0.67a | 12.19 ± 0.94a | 11.56 ± 1.79a | 8.74 ± 0.38bc | 7.58 ± 0.88c | 15.47 ± 0.39a | 17.99 ± 2.19ab |
| Shoot fresh weight (g/plant) | 0.48 ± 0.07a | 1.03 ± 0.12a | 0.51 ± 0.28a | 0.78 ± 0.56a | 0.52 ± 0.09c | 0.90 ± 0.23b | 0.35 ± 0.08c | 1.51 ± 0.22a |
| SPAD reading 3 | 10.48 ± 1.70ab | 7.84 ± 0.60c | 9.71 ± 0.92bc | 11.96 ± 1.14a | 14.32 ± 1.97a | 14.41 ± 1.74a | 12.42 ± 0.50a | 14.91 ± 0.30a |
| Treatment 2 | BCFR 3 | BCFS 3 | TF 3 | Average Daily Dose 4 (ADDv; μg/kg·BW/day) | Hazard Quotient (HQv) 4 | ||||
|---|---|---|---|---|---|---|---|---|---|
| ADDv-TC | ADDv-CF | ADDv-BF | HQv-TC | HQv-CF | HQv-BF | ||||
| PCC-CK | 0.40 ± 0.17ab | 0.57 ± 0.11a | 1.43 ± 0.67a | 0.66 | 0.002 | 0.15 | 7.88 | 0.02 | 1.73 |
| PCC-BC | 0.25 ± 0.06b | 0.29 ± 0.14a | 1.15 ± 0.64a | 0.37 | 0.001 | 0.08 | 4.42 | 0.02 | 0.97 |
| PCW-CK | 0.78 ± 0.06a | 0.82 ± 0.23a | 1.06 ± 0.23a | 0.60 | 0.019 | 0.13 | 7.16 | 0.22 | 1.57 |
| PCW-BC | 0.57 ± 0.40ab | 0.37 ± 0.03a | 0.65 ± 0.49a | 0.29 | 0.004 | 0.06 | 3.43 | 0.05 | 0.75 |
| LSC-CK | 0.89 ± 0.40a | 0.42 ± 0.07a | 0.47 ± 0.37b | 0.34 | 0.002 | 0.08 | 4.07 | 0.03 | 0.90 |
| LSC-BC | 0.28 ± 0.01b | 0.18 ± 0.04b | 0.64 ± 0.13b | 0.20 | 0.004 | 0.04 | 2.41 | 0.05 | 0.53 |
| LSS-CK | 0.14 ± 0.06b | 0.25 ± 0.04b | 1.70 ± 0.49a | 0.26 | 0.002 | 0.06 | 3.04 | 0.03 | 0.67 |
| LSS-BC | 0.11 ± 0.05b | 0.17 ± 0.07b | 1.53 ± 0.79a | 0.29 | 0.002 | 0.06 | 3.46 | 0.03 | 0.76 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, K.-S.; Pai, C.-Y.; Lai, H.-Y. Amendment of Husk Biochar on Accumulation and Chemical Form of Cadmium in Lettuce and Pak-Choi Grown in Contaminated Soil. Water 2020, 12, 868. https://doi.org/10.3390/w12030868
Chen K-S, Pai C-Y, Lai H-Y. Amendment of Husk Biochar on Accumulation and Chemical Form of Cadmium in Lettuce and Pak-Choi Grown in Contaminated Soil. Water. 2020; 12(3):868. https://doi.org/10.3390/w12030868
Chicago/Turabian StyleChen, Kuei-San, Chun-Yu Pai, and Hung-Yu Lai. 2020. "Amendment of Husk Biochar on Accumulation and Chemical Form of Cadmium in Lettuce and Pak-Choi Grown in Contaminated Soil" Water 12, no. 3: 868. https://doi.org/10.3390/w12030868
APA StyleChen, K.-S., Pai, C.-Y., & Lai, H.-Y. (2020). Amendment of Husk Biochar on Accumulation and Chemical Form of Cadmium in Lettuce and Pak-Choi Grown in Contaminated Soil. Water, 12(3), 868. https://doi.org/10.3390/w12030868






