Efficient Removal of Cr(VI) from Water by Biochar and Activated Carbon Prepared through Hydrothermal Carbonization and Pyrolysis: Adsorption-Coupled Reduction Mechanism
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of Biochar and Activated Carbon
2.2. Characterization of Biochar and Activated Carbon
2.3. Batch Adsorption Study
2.4. Adsorption Isotherm Model
3. Results and Discussion
3.1. Property of Prepared Biochar and Activated Carbon
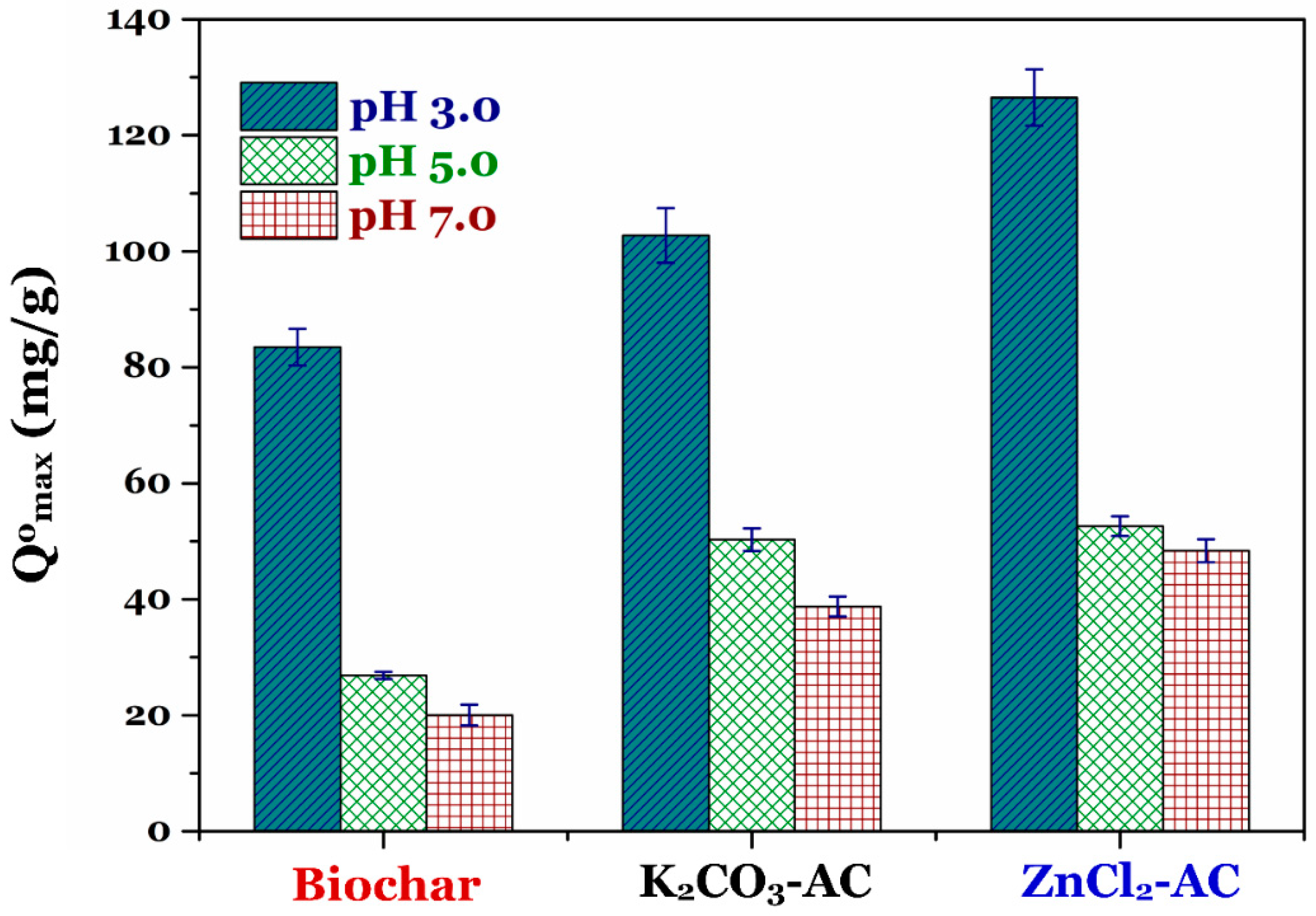
3.2. Effect of pH on Adsorption Capacity of Prepared Biochar and Activated Carbon
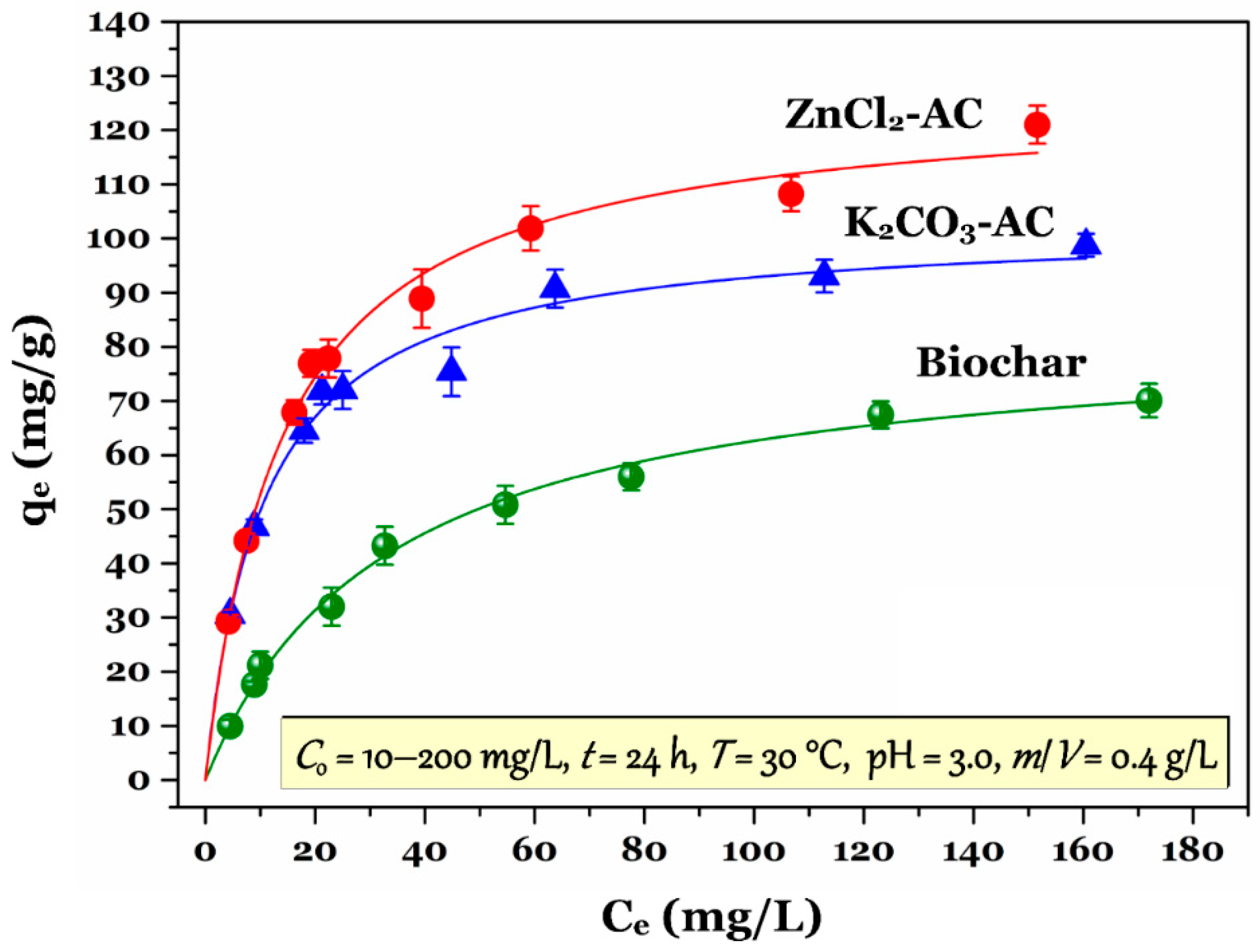
3.3. Adsorption Isotherm
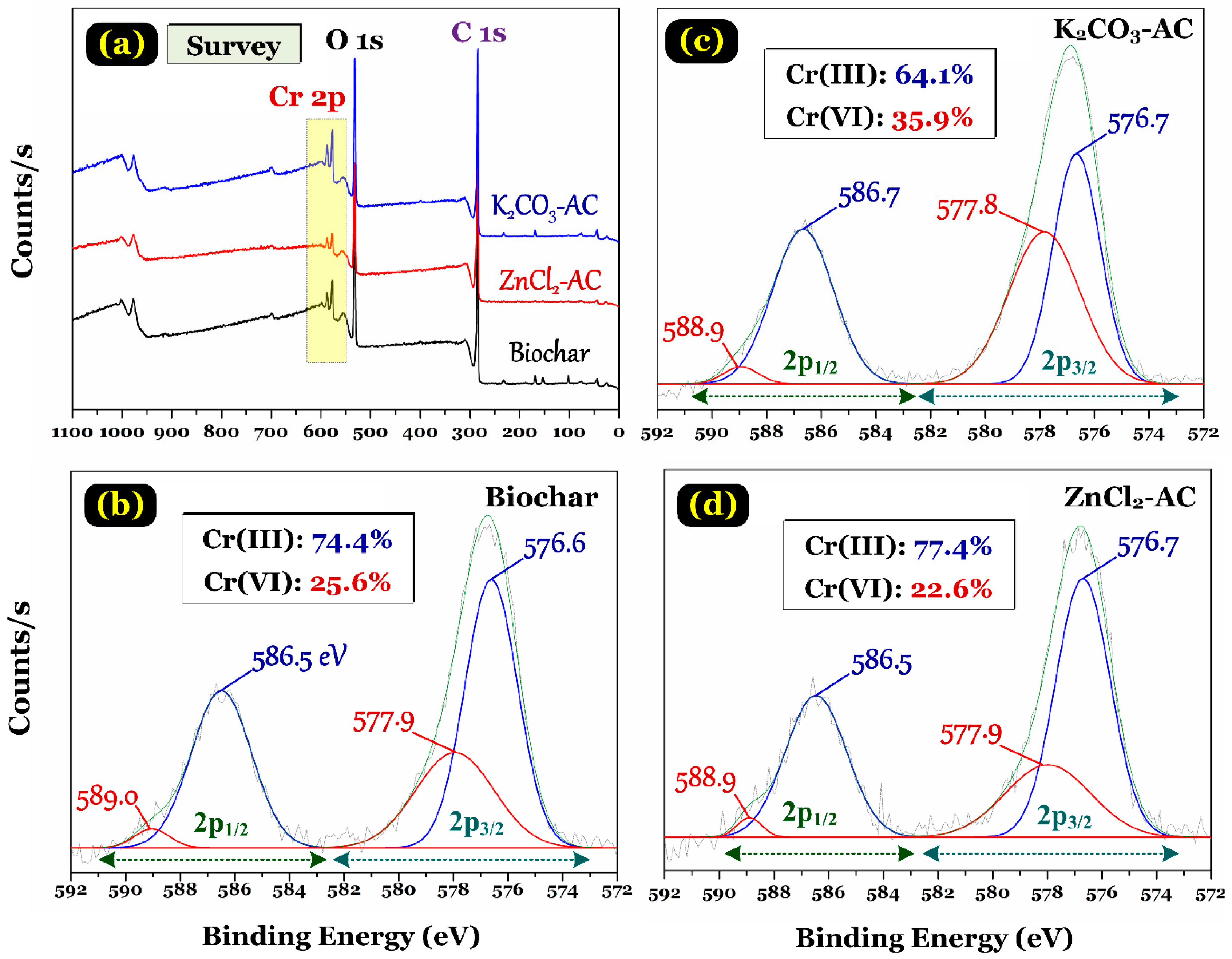
3.4. Adsorption Mechanism
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Korak, J.A.; Huggins, R.; Arias-Paic, M. Regeneration of pilot-scale ion exchange columns for hexavalent chromium removal. Water Res. 2017, 118, 141–151. [Google Scholar] [CrossRef] [PubMed]
- Mohan, D.; Pittman, C.U. Activated carbons and low cost adsorbents for remediation of tri- and hexavalent chromium from water. J. Hazard. Mater. 2006, 137, 762–811. [Google Scholar] [CrossRef]
- Aharchaou, I.; Py, J.-S.; Cambier, S.; Loizeau, J.-L.; Cornelis, G.; Rousselle, P.; Battaglia, E.; Vignati, D.A.L. Chromium hazard and risk assessment: New insights from a detailed speciation study in a standard test medium. Environ. Toxicol. Chem. 2018, 37, 983–992. [Google Scholar] [CrossRef] [PubMed]
- Yan, K.; Liu, Z.; Li, Z.; Yue, R.; Guo, F.; Xu, Z. Selective separation of chromium from sulphuric acid leaching solutions of mixed electroplating sludge using phosphate precipitation. Hydrometallurgy 2019, 186, 42–49. [Google Scholar] [CrossRef]
- Martín-Domínguez, A.; Rivera-Huerta, M.L.; Pérez-Castrejón, S.; Garrido-Hoyos, S.E.; Villegas-Mendoza, I.E.; Gelover-Santiago, S.L.; Drogui, P.; Buelna, G. Chromium removal from drinking water by redox-assisted coagulation: Chemical versus electrocoagulation. Sep. Purif. Technol. 2018, 200, 266–272. [Google Scholar] [CrossRef]
- Tu, J.; Gan, Y.; Liang, T.; Wan, H.; Wang, P. A miniaturized electrochemical system for high sensitive determination of chromium(VI) by screen-printed carbon electrode with gold nanoparticles modification. Sens. Actuators B Chem. 2018, 272, 582–588. [Google Scholar] [CrossRef]
- Mamais, D.; Noutsopoulos, C.; Kavallari, I.; Nyktari, E.; Kaldis, A.; Panousi, E.; Nikitopoulos, G.; Antoniou, K.; Nasioka, M. Biological groundwater treatment for chromium removal at low hexavalent chromium concentrations. Chemosphere 2016, 152, 238–244. [Google Scholar] [CrossRef]
- Zolfaghari, G.; Kargar, M. Nanofiltration and microfiltration for the removal of chromium, total dissolved solids, and sulfate from water. MethodsX 2019, 6, 549–557. [Google Scholar] [CrossRef]
- Tran, H.N.; Nguyen, D.T.; Le, G.T.; Tomul, F.; Lima, E.C.; Woo, S.H.; Sarmah, A.K.; Nguyen, H.Q.; Nguyen, P.T.; Nguyen, D.D.; et al. Adsorption mechanism of hexavalent chromium onto layered double hydroxides-based adsorbents: A systematic in-depth review. J. Hazard. Mater. 2019, 373, 258–270. [Google Scholar] [CrossRef]
- Wang, H.; Zhang, M.; Lv, Q. Removal efficiency and mechanism of Cr(VI) from aqueous solution by maize straw biochars derived at different pyrolysis temperatures. Water 2019, 11, 781. [Google Scholar] [CrossRef]
- Tytłak, A.; Oleszczuk, P.; Dobrowolski, R. Sorption and desorption of Cr(VI) ions from water by biochars in different environmental conditions. Environ. Sci. Pollut. Res. 2015, 22, 5985–5994. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.; Li, L.; Liu, Z.; Lu, M.; Wang, C.; Li, H.; Ma, W.; Wang, S. Preparation and characterization of activated carbons from tobacco stem by chemical activation. J. Air Waste Manag. Assoc. 2017, 67, 713–724. [Google Scholar] [CrossRef] [PubMed]
- Ma, H.; Yang, J.; Gao, X.; Liu, Z.; Liu, X.; Xu, Z. Removal of chromium (VI) from water by porous carbon derived from corn straw: Influencing factors, regeneration and mechanism. J. Hazard. Mater. 2019, 369, 550–560. [Google Scholar] [CrossRef] [PubMed]
- Tran Hai, N.; Huang, F.-C.; Lee, C.-K.; Chao, H.-P. Activated carbon derived from spherical hydrochar functionalized with triethylenetetramine: Synthesis, characterizations, and adsorption application. Green Process. Synth. 2017, 6, 565. [Google Scholar] [CrossRef]
- Xu, S.; Yu, W.; Liu, S.; Xu, C.; Li, J.; Zhang, Y. Adsorption of hexavalent chromium using banana pseudostem biochar and its mechanism. Sustainability 2018, 10, 4250. [Google Scholar] [CrossRef]
- Huang, F.-C.; Lee, C.-K.; Han, Y.-L.; Chao, W.-C.; Chao, H.-P. Preparation of activated carbon using micro-nano carbon spheres through chemical activation. J. Taiwan Inst. Chem. Eng. 2014, 45, 2805–2812. [Google Scholar] [CrossRef]
- Boakye, P.; Tran, H.N.; Lee, D.S.; Woo, S.H. Effect of water washing pretreatment on property and adsorption capacity of macroalgae-derived biochar. J. Environ. Manag. 2019, 233, 165–174. [Google Scholar] [CrossRef]
- Freedonia. Global Activated Carbon-Demand and Sales Forecasts, Market Share, Market Size, Market Leaders; The Freedonia Group: Cleveland, OH, USA, 2018. [Google Scholar]
- Tran, H.N.; You, S.-J.; Chao, H.-P. Effect of pyrolysis temperatures and times on the adsorption of cadmium onto orange peel derived biochar. Waste Manag. Res. 2016, 34, 129–138. [Google Scholar] [CrossRef]
- Tran, H.N.; Wang, Y.; You, S.; Chao, H. Sustainable biochar derived from agricultural wastes for removal of methylene green 5 from aqueous solution: Adsorption kinetics, isotherms, thermodynamics, and mechanism analysis. In Air, Gas, and Water Pollution Control Using Industrial and Agricultural Solid Wastes Adsorbents, 1st ed.; CRC Press: Boca Raton, FL, USA, 2017; pp. 255–291. [Google Scholar]
- Tran, H.N.; You, S.-J.; Chao, H.-P. Fast and efficient adsorption of methylene green 5 on activated carbon prepared from new chemical activation method. J. Environ. Manag. 2017, 188, 322–336. [Google Scholar] [CrossRef]
- Tran, H.N.; Wen, Y.-C.; Wang, Y.-F.; You, S.-J. Highly efficient removal of hazardous aromatic pollutants by micro-nano spherical carbons synthesized from different chemical activation methods: A comparison study. Environ. Technol. 2019, 40, 1376–1391. [Google Scholar] [CrossRef]
- Tran, H.N.; Lee, C.-K.; Nguyen, T.V.; Chao, H.-P. Saccharide-derived microporous spherical biochar prepared from hydrothermal carbonization and different pyrolysis temperatures: Synthesis, characterization, and application in water treatment. Environ. Technol. 2018, 39, 2747–2760. [Google Scholar] [CrossRef] [PubMed]
- Sevilla, M.; Fuertes, A.B.; Mokaya, R. High density hydrogen storage in superactivated carbons from hydrothermally carbonized renewable organic materials. Energy Environ. Sci. 2011, 4, 1400–1410. [Google Scholar] [CrossRef]
- Tran, H.N.; Lee, C.-K.; Vu, M.T.; Chao, H.-P. Removal of copper, lead, methylene green 5, and acid red 1 by saccharide-derived spherical biochar prepared at low calcination temperatures: Adsorption kinetics, isotherms, and thermodynamics. Water Air Soil Pollut. 2017, 228, 401–417. [Google Scholar] [CrossRef]
- Tran, H.N.; Wang, Y.-F.; You, S.-J.; Chao, H.-P. Insights into the mechanism of cationic dye adsorption on activated charcoal: The importance of π–π interactions. Process. Saf. Environ. Prot. 2017, 107, 168–180. [Google Scholar] [CrossRef]
- Langmuir, I. The adsorption of gases on plane surfaces of glass, mica and platinum. J. Am. Chem. Soc. 1918, 40, 1361–1403. [Google Scholar] [CrossRef]
- Freundlich, H. Über die adsorption in lösungen. Z. Phys. Chem. 1907, 57, 385–470. [Google Scholar] [CrossRef]
- Redlich, O.; Peterson, D.L. A useful adsorption isotherm. J. Phys. Chem. 1959, 63, 1024. [Google Scholar] [CrossRef]
- Tran, H.N.; You, S.-J.; Hosseini-Bandegharaei, A.; Chao, H.-P. Mistakes and inconsistencies regarding adsorption of contaminants from aqueous solutions: A critical review. Water Res. 2017, 120, 88–116. [Google Scholar] [CrossRef] [PubMed]
- Thommes, M.; Kaneko, K.; Neimark Alexander, V.; Olivier James, P.; Rodriguez-Reinoso, F.; Rouquerol, J.; Sing Kenneth, S.W. Physisorption of gases, with special reference to the evaluation of surface area and pore size distribution (IUPAC Technical Report). Pure Appl. Chem. 2015, 87, 1051–1069. [Google Scholar] [CrossRef]
- Chen, Y.; An, D.; Sun, S.; Gao, J.; Qian, L. Reduction and Removal of Chromium VI in Water by Powdered Activated Carbon. Materials 2018, 11, 269. [Google Scholar] [CrossRef]
- Tran, H.N.; You, S.-J.; Chao, H.-P. Thermodynamic parameters of cadmium adsorption onto orange peel calculated from various methods: A comparison study. J. Environ. Chem. Eng. 2016, 4, 2671–2682. [Google Scholar] [CrossRef]
- Tran, H.N.; Chao, H.-P. Adsorption and desorption of potentially toxic metals on modified biosorbents through new green grafting process. Environ. Sci. Pollut. Res. 2018, 25, 12808–12820. [Google Scholar] [CrossRef]
- Sengupta, A.K.; Clifford, D. Chromate ion exchange mechanism for cooling water. Ind. Eng. Chem. Fundam. 1986, 25, 249–258. [Google Scholar] [CrossRef]
- Chao, H.-P.; Wang, Y.-C.; Tran, H.N. Removal of hexavalent chromium from groundwater by Mg/Al-layered double hydroxides using characteristics of in-situ synthesis. Environ. Pollut. 2018, 243, 620–629. [Google Scholar] [CrossRef] [PubMed]
- Volesky, B. Biosorption and me. Water Res. 2007, 41, 4017–4029. [Google Scholar] [CrossRef] [PubMed]
- Tran, H.N.; Lin, C.-C.; Chao, H.-P. Amino acids-intercalated Mg/Al layered double hydroxides as dual-electronic adsorbent for effective removal of cationic and oxyanionic metal ions. Sep. Purif. Technol. 2018, 192, 36–45. [Google Scholar] [CrossRef]
- Kumar, A.; Jena, H.M. Adsorption of Cr(VI) from aqueous phase by high surface area activated carbon prepared by chemical activation with ZnCl2. Process. Saf. Environ. Prot. 2017, 109, 63–71. [Google Scholar] [CrossRef]
- Enniya, I.; Rghioui, L.; Jourani, A. Adsorption of hexavalent chromium in aqueous solution on activated carbon prepared from apple peels. Sustain. Chem. Pharm. 2018, 7, 9–16. [Google Scholar] [CrossRef]
- Hamadi, N.K.; Chen, X.D.; Farid, M.M.; Lu, M.G.Q. Adsorption kinetics for the removal of chromium(VI) from aqueous solution by adsorbents derived from used tyres and sawdust. Chem. Eng. J. 2001, 84, 95–105. [Google Scholar] [CrossRef]
- Krishna Kumar, A.S.; Jiang, S.-J.; Tseng, W.-L. Effective adsorption of chromium(VI)/Cr(III) from aqueous solution using ionic liquid functionalized multiwalled carbon nanotubes as a super sorbent. J. Mater. Chem. A 2015, 3, 7044–7057. [Google Scholar] [CrossRef]
- Moreno-Castilla, C. Adsorption of organic molecules from aqueous solutions on carbon materials. Carbon 2004, 42, 83–94. [Google Scholar] [CrossRef]
- Park, D.; Yun, Y.-S.; Park, J.M. Studies on hexavalent chromium biosorption by chemically-treated biomass of Ecklonia sp. Chemosphere 2005, 60, 1356–1364. [Google Scholar] [CrossRef] [PubMed]
- Park, D.; Yun, Y.-S.; Park, J.M. Mechanisms of the removal of hexavalent chromium by biomaterials or biomaterial-based activated carbons. J. Hazard. Mater. 2006, 137, 1254–1257. [Google Scholar] [CrossRef] [PubMed]
- Park, D.; Yun, Y.-S.; Kim, J.Y.; Park, J.M. How to study Cr(VI) biosorption: Use of fermentation waste for detoxifying Cr(VI) in aqueous solution. Chem. Eng. J. 2008, 136, 173–179. [Google Scholar] [CrossRef]
- Choudhary, B.; Paul, D.; Singh, A.; Gupta, T. Removal of hexavalent chromium upon interaction with biochar under acidic conditions: Mechanistic insights and application. Environ. Sci. Pollut. Res. 2017, 24, 16786–16797. [Google Scholar] [CrossRef] [PubMed]
- Tran, H.N. Comment on “simultaneous and efficient removal of Cr(VI) and methyl orange on LDHs decorated porous carbons”. Chem. Eng. J. 2019, 359, 810–812. [Google Scholar] [CrossRef]
- Tran, H.N.; Pham, V.V.; Vo, D.-V.N.; Nguyen, P.T. Comment on “Removal of hexavalent chromium by biochar supported nZVI composite: Batch and fixed-bed column evaluations, mechanisms, and secondary contamination prevention”. Chemosphere 2019. [Google Scholar] [CrossRef]
- Liu, R.; Zhu, X.; Chen, B. A New Insight of Graphene oxide-Fe(III) Complex Photochemical Behaviors under Visible Light Irradiation. Sci. Rep. 2017, 7, 40711. [Google Scholar] [CrossRef]
- Park, D.; Yun, Y.-S.; Park, J.M. XAS and XPS studies on chromium-binding groups of biomaterial during Cr(VI) biosorption. J. Colloid Interface Sci. 2008, 317, 54–61. [Google Scholar] [CrossRef]
- Rivera-Utrilla, J.; Sánchez-Polo, M. Adsorption of Cr(III) on ozonised activated carbon. Importance of Cπ—cation interactions. Water Res. 2003, 37, 3335–3340. [Google Scholar] [CrossRef]
- Arim, A.L.; Quina, M.J.; Gando-Ferreira, L.M. Uptake of trivalent chromium from aqueous solutions by xanthate pine bark: Characterization, batch and column studies. Process. Saf. Environ. Prot. 2019, 121, 374–386. [Google Scholar] [CrossRef]
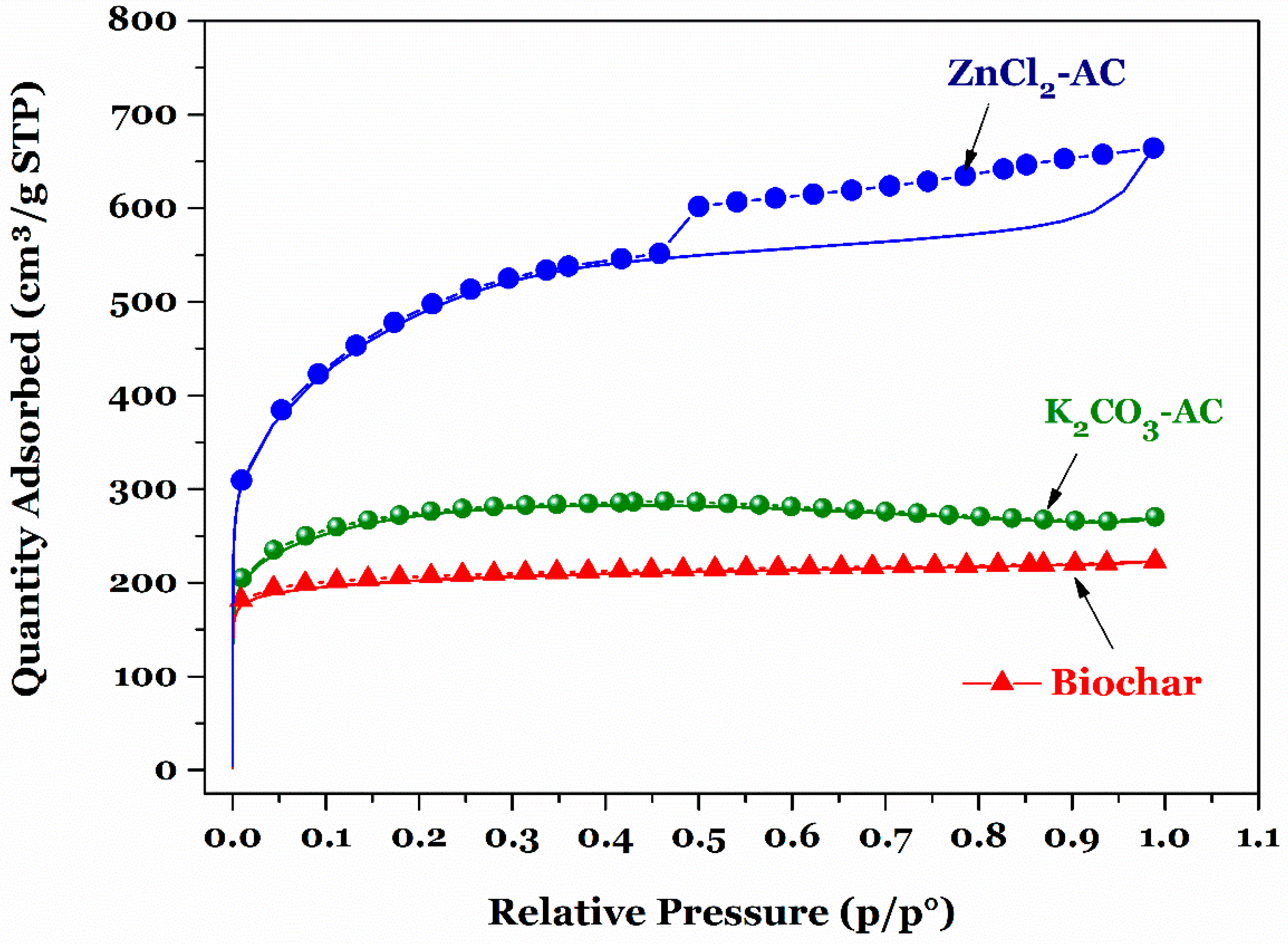


| Adsorbent | SBET (m2/g) | VTotal (cm3/g) | Pore Width (nm) |
|---|---|---|---|
| ZnCl2-AC | 1757 | 1.027 | 2.34 |
| K2CO3-AC | 1013 | 0.418 | 1.65 |
| Commercial AC | 1026 | 0.502 | 1.95 |
| Biochar | 792 | 0.345 | 1.74 |
| Unit | Biochar | K2CO3-AC | ZnCl2-AC | |
|---|---|---|---|---|
| 1. Langmuir model | ||||
| Qomax | (mg/g) | 83.5 | 103 | 127 |
| KL | (L/mg) | 0.030 | 0.094 | 0.071 |
| R2 | — | 0.995 | 0.984 | 0.990 |
| χ2 | — | 3.373 | 20.9 | 18.6 |
| 2. Freundlich model | ||||
| KF | (mg/g)(L/mg)n | 15.4 | 43.0 | 41.3 |
| n | — | 0.300 | 0.165 | 0.212 |
| R2 | — | 0.996 | 0.989 | 0.997 |
| χ2 | — | 2.40 | 14.2 | 6.34 |
| 3. Redlich–Peterson model | ||||
| KRP | (L/g) | 5.86 | 216 | 71.2 |
| aRP | (mg/L) | 0.212 | 4.86 | 1.54 |
| g | — | 0.802 | 0.841 | 0.809 |
| R2 | — | 0.996 | 0.986 | 0.996 |
| χ2 | — | 2.66 | 18.9 | 8.24 |
| Stage | Activating Agent | SBET (m2/g) | VTotal (cm3/g) | Qomax (mg/g) | References | |
|---|---|---|---|---|---|---|
| 1. Activated carbon | ||||||
| Corn straw | One | KOH | 2131 | 1.128 | 176 | [13] |
| Tectona grandis sawdust | Two | ZnCl2 | 1757 | 0.418 | 127 | This study |
| Tectona grandis sawdust | Two | K2CO3 | 1013 | 1.027 | 103 | This study |
| Multiwalled carbon nanotubes | — | — | 87.4 | 0.82 | 85.8 | [42] |
| Waste tires | Two | CO2 | 832 | — | 55.2 | [41] |
| NORIT RB4C (CAC) | Two | Steam | 1026 | 0.502 | 52.6 | This study |
| F400 (CAC) | — | — | — | — | 48.5 | [41] |
| Nutshells | One | ZnCl2 | 2869 | 1.96 | 46.2 | [39] |
| Apple peels | One | H3PO4 | — | — | 36.0 | [40] |
| 2. Biochar | ||||||
| Tectona grandis sawdust | Two | No | 792 | 0.345 | 83.5 | This study |
| Wheat straw | One | No | 26.3 | — | 24.6 | [11] |
| Wicke | One | No | 11.4 | — | 23.6 | [11] |
| Banana pseudostem | One | No | 8.53 | 0.044 | 21.5 | [15] |
| Sawdust | One | No | 320 | — | 2.15 | [41] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vo, A.T.; Nguyen, V.P.; Ouakouak, A.; Nieva, A.; Doma, B.T., Jr.; Tran, H.N.; Chao, H.-P. Efficient Removal of Cr(VI) from Water by Biochar and Activated Carbon Prepared through Hydrothermal Carbonization and Pyrolysis: Adsorption-Coupled Reduction Mechanism. Water 2019, 11, 1164. https://doi.org/10.3390/w11061164
Vo AT, Nguyen VP, Ouakouak A, Nieva A, Doma BT Jr., Tran HN, Chao H-P. Efficient Removal of Cr(VI) from Water by Biochar and Activated Carbon Prepared through Hydrothermal Carbonization and Pyrolysis: Adsorption-Coupled Reduction Mechanism. Water. 2019; 11(6):1164. https://doi.org/10.3390/w11061164
Chicago/Turabian StyleVo, Anh Tuan, Van Phuong Nguyen, Abdelkader Ouakouak, Aileen Nieva, Bonifacio T. Doma, Jr., Hai Nguyen Tran, and Huan-Ping Chao. 2019. "Efficient Removal of Cr(VI) from Water by Biochar and Activated Carbon Prepared through Hydrothermal Carbonization and Pyrolysis: Adsorption-Coupled Reduction Mechanism" Water 11, no. 6: 1164. https://doi.org/10.3390/w11061164
APA StyleVo, A. T., Nguyen, V. P., Ouakouak, A., Nieva, A., Doma, B. T., Jr., Tran, H. N., & Chao, H.-P. (2019). Efficient Removal of Cr(VI) from Water by Biochar and Activated Carbon Prepared through Hydrothermal Carbonization and Pyrolysis: Adsorption-Coupled Reduction Mechanism. Water, 11(6), 1164. https://doi.org/10.3390/w11061164






