Is the Hyporheic Zone Relevant beyond the Scientific Community?
Abstract
1. Introduction
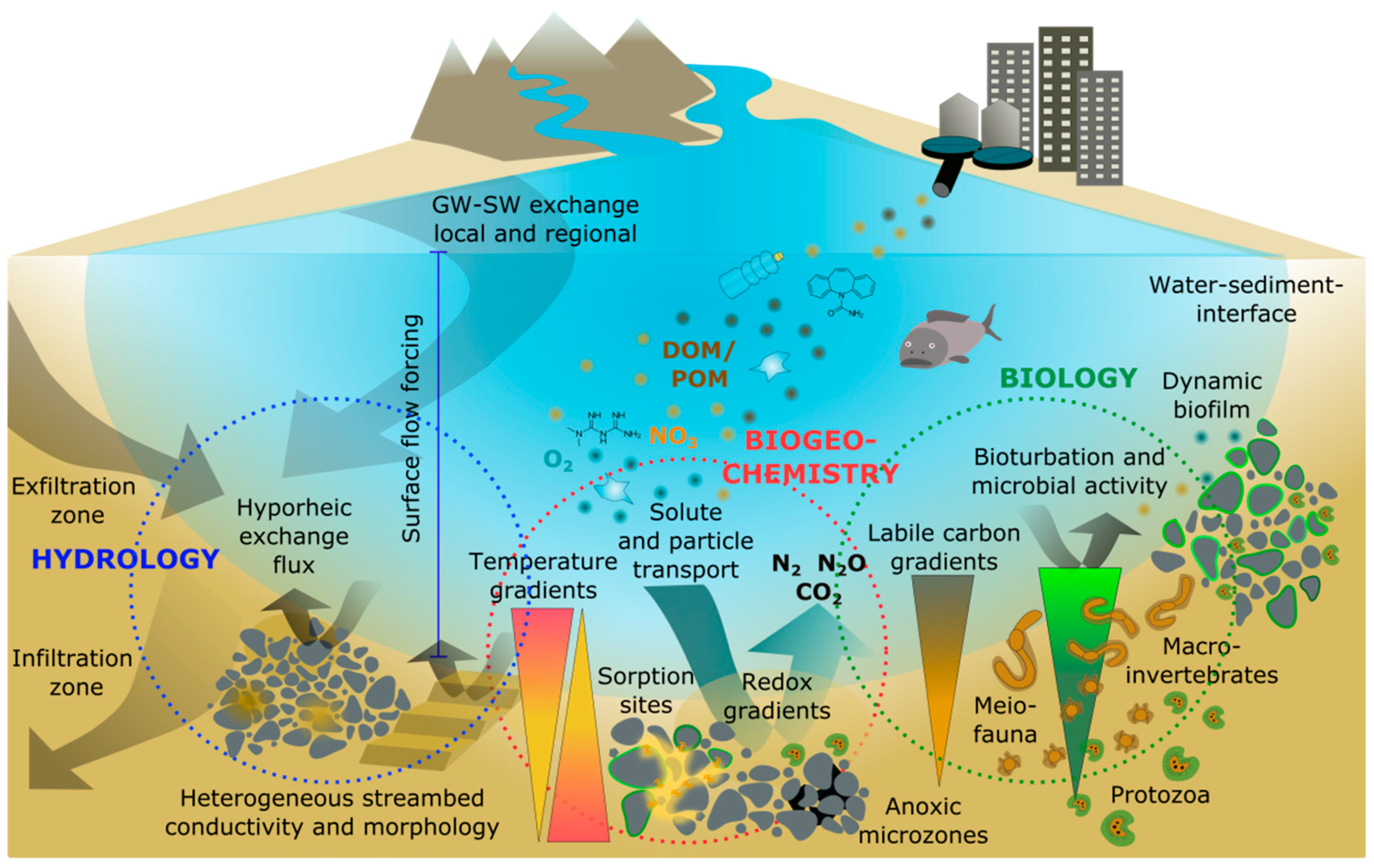
2. Hyporheic Zone Drivers and Processes
2.1. Physical Drivers of Hyporheic Exchange Flows
2.2. Hyporheic Assemblages and Biological Processes
2.3. Biogeochemical Processes
3. Relevance of the Hyporheic Zone
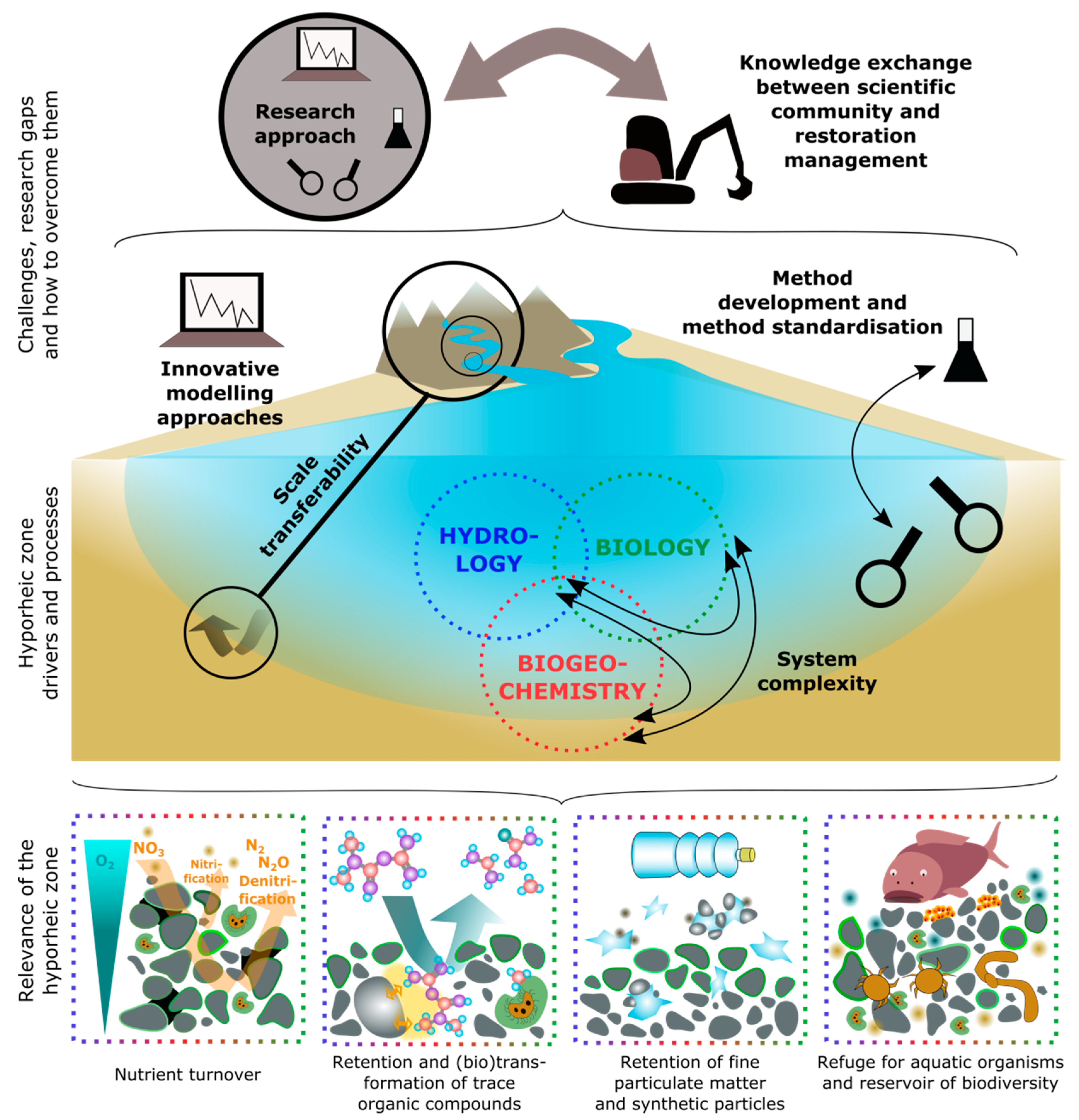
3.1. Nutrient Turnover
3.2. Retention and (Bio)transformation of Trace Organic Compounds
3.3. Retention of Fine Particulate Matter and Synthetic Particles
3.4. Refuge for Aquatic Organisms and Reservoir of Biodiversity
4. Challenges, Research Gaps, and How to Overcome Them
4.1. System Complexity
4.2. Scale Transferability
4.3. Research Approach
4.4. Method Development and Method Standardization
4.5. Innovative Modelling Approaches
4.6. Knowledge Exchange Between the Scientific Community and Restoration Practitioners
5. Conclusions
- to understand geomorphic-climatic controls that underlie spatial patterns of streamflow dynamics to quantify hydrologically critical drivers of HEF across different scales [255,256]. A proper description of the spatial variability of hydrological processes would help clarify how the ecosystem services provided by HEFs can be extended and upscaled to entire river networks.
- to enlighten the role of HEF, hyporheic sediments, and processes in cycling of microplastics, as HEF has the potential to retain large amounts of microplastics.
- to develop methods of incorporating stream restoration structures into site-specific designs that optimize retention of local priority pollutants.
- to clarify the relative contribution of the HZ to overall in-stream removal of reactive compounds in various stream systems differing with respect to their hydrological characteristics.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Boano, F.; Harvey, J.W.; Marion, A.; Packman, A.I.; Revelli, R.; Ridolfi, L.; Wörman, A. Hyporheic flow and transport processes: Mechanisms, models, and biogeochemical implications. Rev. Geophys. 2014, 52, 603–679. [Google Scholar] [CrossRef]
- Orghidan, T. A new habitat of subsurface waters: The hyporheic biotope. Fundam. Appl. Limnol. Arch. Hydrobiol. 2010, 176, 291–302. [Google Scholar] [CrossRef]
- Dahm, C.N.; Valett, H.M.; Baxter, C.V.; Woessner, W.W. Hyporheic zones. In Methods in Stream Ecology, 2nd ed.; Hauer, F.R., Lamberti, G., Eds.; Academic Press: San Diego, CA, USA, 2007; pp. 119–236. [Google Scholar]
- Peralta-Maraver, I.; Galloway, J.; Posselt, M.; Arnon, S.; Reiss, J.; Lewandowski, J.; Robertson, A.L. Environmental filtering and community delineation in the streambed ecotone. Sci. Rep. 2018, 8, 15871. [Google Scholar] [CrossRef] [PubMed]
- Gooseff, M.N. Defining hyporheic zones–advancing our conceptual and operational definitions of where stream water and groundwater meet. Geogr. Compass 2010, 4, 945–955. [Google Scholar] [CrossRef]
- Ward, A.S. The evolution and state of interdisciplinary hyporheic research. Wiley Interdiscip. Rev. Water 2016, 3, 83–103. [Google Scholar] [CrossRef]
- Gomez-Velez, J.D.; Wilson, J.L.; Cardenas, M.B.; Harvey, J.W. Flow and residence times of dynamic river bank storage and sinuosity-driven hyporheic exchange. Water Resour. Res. 2017, 53, 8572–8595. [Google Scholar] [CrossRef]
- Fox, A.; Boano, F.; Arnon, S. Impact of losing and gaining streamflow conditions on hyporheic exchange fluxes induced by dune-shaped bed forms. Water Resour. Res. 2014, 50, 1895–1907. [Google Scholar] [CrossRef]
- Gomez-Velez, J.D.; Harvey, J.; Cardenas, M.B.; Kiel, B. Denitrification in the Mississippi River network controlled by flow through river bedforms. Nat. Geosci. 2015, 8, 941–945. [Google Scholar] [CrossRef]
- Boulton, A.J.; Hancock, P.J. Rivers as groundwater-dependent ecosystems: A review of degrees of dependency, riverine processes and management implications. Aust. J. Bot. 2006, 54, 133–144. [Google Scholar] [CrossRef]
- Magliozzi, C.; Grabowski, R.C.; Packman, A.I.; Krause, S. Toward a conceptual framework of hyporheic exchange across spatial scales. Hydrol. Earth Syst. Sci. 2018, 22, 6163–6185. [Google Scholar] [CrossRef]
- Brunke, M.; Gonser, T. The ecological significance of exchange processes between rivers and groundwater. Freshw. Biol. 1997, 37, 1–33. [Google Scholar] [CrossRef]
- Hancock, P.J. Human impacts on the stream-groundwater exchange zone. Environ. Manag. 2002, 29, 763–781. [Google Scholar] [CrossRef] [PubMed]
- Boulton, A.J.; Findlay, S.; Marmonier, P.; Stanley, E.H.; Valett, H.M. The functional significance of the hyporheic zone in streams and rivers. Annu. Rev. Ecol. Evol. Syst. 1998, 29, 59–81. [Google Scholar] [CrossRef]
- Fischer, H.; Kloep, F.; Wilzcek, S.; Pusch, M.T. A river’s liver–microbial processes within the hyporheic zone of a large lowland river. Biogeochemistry 2005, 76, 349–371. [Google Scholar] [CrossRef]
- Krause, S.; Hannah, D.M.; Fleckenstein, J.H.; Heppell, C.M.; Kaeser, D.; Pickup, R.; Pinay, G.; Robertson, A.L.; Wood, P.J. Inter-disciplinary perspectives on processes in the hyporheic zone. Ecohydrology 2011, 4, 481–499. [Google Scholar] [CrossRef]
- Krause, S.; Lewandowski, J.; Grimm, N.B.; Hannah, D.M.; Pinay, G.; McDonald, K.; Marti, E.; Argerich, A.; Pfister, L.; Klaus, J.; et al. Ecohydrological interfaces as hot spots of ecosystem processes. Water Resour. Res. 2017, 53, 6359–6376. [Google Scholar] [CrossRef]
- Cranswick, R.H.; Cook, P.G. Scales and magnitude of hyporheic, river-aquifer and bank storage exchange fluxes. Hydrol. Process. 2015, 29, 3084–3097. [Google Scholar] [CrossRef]
- Stubbington, R. The hyporheic zone as an invertebrate refuge: A review of variability in space, time, taxa and behavior. Mar. Freshw. Res. 2012, 63, 293–311. [Google Scholar] [CrossRef]
- Dole-Olivier, M.J. The hyporheic refuge hypothesis reconsidered: A review of hydrological aspects. Mar. Freshw. Res. 2011, 62, 1281–1302. [Google Scholar] [CrossRef]
- Peralta-Maraver, I.; Reiss, J.; Robertson, A.L. Interplay of hydrology, community ecology and pollutant attenuation in the hyporheic zone. Sci. Total Environ. 2018, 610, 267–275. [Google Scholar] [CrossRef]
- Anderson, M.P. Heat as a ground water tracer. Ground Water 2005, 43, 951–968. [Google Scholar] [CrossRef] [PubMed]
- Rau, G.C.; Andersen, M.S.; McCallum, A.M.; Roshan, H.; Acworth, R.I. Heat as a tracer to quantify water flow in near-surface sediments. Earth Sci. Rev. 2014, 129, 40–58. [Google Scholar] [CrossRef]
- Ren, J.; Cheng, J.Q.; Yang, J.; Zhou, Y.J. A review on using heat as a tool for studying groundwater-surface water interactions. Environ. Earth Sci. 2018, 77, 756. [Google Scholar] [CrossRef]
- Knapp, J.L.A.; González-Pinzón, R.; Haggerty, R. The resazurin-resorufin system: Insights from a decade of “Smart” tracer development for hydrologic applications. Water Resour. Res. 2018, 54, 6877–6889. [Google Scholar] [CrossRef]
- Kalbus, E.; Reinstorf, F.; Schirmer, M. Measuring methods for groundwater–surface water interactions: A review. Hydrol. Earth Syst. Sci. 2006, 10, 873–887. [Google Scholar] [CrossRef]
- Brunner, P.; Therrien, R.; Renard, P.; Simmons, C.T.; Franssen, H.J.H. Advances in understanding river-groundwater interactions. Rev. Geophys. 2017, 55, 818–854. [Google Scholar] [CrossRef]
- Hill, A.R. Nitrate removal in stream riparian zones. J. Environ. Qual. 1996, 25, 743–755. [Google Scholar] [CrossRef]
- Birgand, F.; Skaggs, R.W.; Chescheir, G.M.; Gilliam, J.W. Nitrogen removal in streams of agricultural catchments -A literature review. Crit. Rev. Environ. Sci. Technol. 2007, 37, 381–487. [Google Scholar] [CrossRef]
- Gandy, C.J.; Smith, J.W.N.; Jarvis, A.P. Attenuation of mining-derived pollutants in the hyporheic zone: A review. Sci. Total Environ. 2007, 273, 435–446. [Google Scholar] [CrossRef]
- Klein, S.; Worch, E.; Knepper, T.P. Occurrence and spatial distribution of microplastics in river shore sediments of the Rhine-Main area in Germany. Environ. Sci. Technol. 2015, 49, 6070–6076. [Google Scholar] [CrossRef]
- Boutron, O.; Margoum, C.; Chovelon, J.-M.; Guillemain, C.; Gouy, V. Effect of the submergence, the bed form geometry, and the speed of the surface water flow on the mitigation of pesticides in agricultural ditches. Water Resour. Res. 2011, 47, 1–13. [Google Scholar] [CrossRef]
- Peter, K.T.; Herzog, S.; Tian, Z.Y.; Wu, C.; McCray, J.E.; Lynch, K.; Kolodziej, E.P. Evaluating emerging organic contaminant removal in an engineered hyporheic zone using high resolution mass spectrometry. Water Res. 2019, 150, 140–152. [Google Scholar] [CrossRef] [PubMed]
- Riml, J.; Wörman, A.; Kunkel, U.; Radke, M. Evaluating the fate of six common pharmaceuticals using a reactive transport model: Insights from a stream tracer test. Sci. Total Environ. 2013, 458–460, 344–354. [Google Scholar] [CrossRef] [PubMed]
- Schaper, J.L.; Posselt, M.; McCallum, J.L.; Banks, E.W.; Hoehne, A.; Meinikmann, K.; Shanafield, M.A.; Batelaan, O.; Lewandowski, J. Hyporheic exchange controls fate of trace organic compounds in an urban stream. Environ. Sci. Technol. 2018, 52, 12285–12294. [Google Scholar] [CrossRef] [PubMed]
- Conant, B.; Robinson, C.E.; Hinton, M.J.; Russell, H.A. A framework for conceptualizing groundwater-surface water interactions and identifying potential impacts on water quality, water quantity, and ecosystems. J. Hydrol. 2019, 574, 609–627. [Google Scholar] [CrossRef]
- Ward, A.S.; Packman, A.I. Advancing our predictive understanding of river corridor exchange. Wiley Interdiscip. Rev. Water 2018, 6, e1327. [Google Scholar] [CrossRef]
- Millennium Ecosystem Assessment. Ecosystems and Human Well-Being: Synthesis; Island Press: Washington, DC, USA, 2005; p. 160. [Google Scholar]
- Gomez-Velez, J.D.; Harvey, J.W. A hydrogeomorphic river network model predicts where and why hyporheic exchange is important in large basins. Geophys. Res. Lett. 2014, 41, 6403–6412. [Google Scholar] [CrossRef]
- Caruso, A.; Ridolfi, L.; Boano, F. Impact of watershed topography on hyporheic exchange. Adv. Water Resour. 2016, 94, 400–411. [Google Scholar] [CrossRef]
- Mojarrad, B.B.; Riml, J.; Wörman, A.; Laudon, H. Fragmentation of the hyporheic zone due to regional groundwater circulation. Water Resour. Res. 2019, 55, 1242–1262. [Google Scholar] [CrossRef]
- Kurth, A.-M.; Dawes, N.; Selker, J.; Schirmer, M. Autonomous distributed temperature sensing for long-term heated applications in remote areas. Geosci. Instrum. Meth. 2013, 2, 71–77. [Google Scholar] [CrossRef]
- Boano, F.; Revelli, R.; Ridolfi, L. Quantifying the impact of groundwater discharge on the surface-subsurface exchange. Hydrol. Process. 2009, 23, 2108–2116. [Google Scholar] [CrossRef]
- Shanafield, M.; Cook, P.G. Transmission losses, infiltration and groundwater recharge through ephemeral and intermittent streambeds: A review of applied methods. J. Hydrol. 2014, 511, 518–529. [Google Scholar] [CrossRef]
- Elliott, A.H.; Brooks, N.H. Transfer of nonsorbing solutes to a streambed with bed forms: Theory. Water Resour. Res. 1997, 33, 123–136. [Google Scholar] [CrossRef]
- Elliott, A.H.; Brooks, N.H. Transfer of nonsorbing solutes to a streambed with bed forms: Laboratory experiments. Water Resour. Res. 1997, 33, 137–151. [Google Scholar] [CrossRef]
- Krause, S.; Klaar, M.; Hannah, D.; Mant, J.; Bridgeman, J.; Trimmer, M.; Manning-Jones, S. The potential of large woody debris to alter biogeochemical processes and ecosystem services in lowland rivers. Wiley Interdiscip. Rev. Water 2014, 1, 263–275. [Google Scholar] [CrossRef]
- Dixon, S.J.; Sear, D.A. The influence of geomorphology on large wood dynamics in a low gradient headwater stream. Water Resour. Res. 2014, 50, 9194–9210. [Google Scholar] [CrossRef]
- Blaen, P.J.; Kurz, M.J.; Drummond, J.D.; Knapp, J.L.A.; Mendoza-Lera, C.; Schmadel, N.M.; Klaar, M.J.; Jäger, A.; Folegot, S.; Lee-Cullin, J.; et al. Woody debris is related to reach-scale hotspots of lowland stream ecosystem respiration under baseflow conditions. Ecohydrology 2018, e1952. [Google Scholar] [CrossRef]
- Packman, A.; Salehin, M.; Zaramella, M. Hyporheic exchange with gravel beds: Basic hydrodynamic interactions and bedform-induced advective flows. J. Hydraul. Eng. 2004, 130, 1647–1656. [Google Scholar] [CrossRef]
- Roche, K.R.; Blois, G.; Best, J.L.; Christensen, K.T.; Aubeneau, A.F.; Packman, A.I. Turbulence links momentum and solute exchange in coarse-grained streambeds. Water Resour. Res. 2018, 54, 3225–3242. [Google Scholar] [CrossRef]
- Wroblicky, G.; Campana, M.; Valett, H.; Dahm, C. Seasonal variation in surface-subsurface water exchange and lateral hyporheic area of two stream-aquifer systems. Water Resour. Res. 1998, 34, 317–328. [Google Scholar] [CrossRef]
- Salehin, M.; Packman, A.I.; Paradis, M. Hyporheic exchange with heterogeneous streambeds: Laboratory experiments and modelling. Water Resour. Res. 2004, 40, W11504. [Google Scholar] [CrossRef]
- Vaux, W.G. Intragravel flow and interchange of water in a streambed. Fish. Bull. NOAA 1968, 66, 479–489. [Google Scholar]
- Pryshlak, T.T.; Sawyer, A.H.; Stonedahl, S.H.; Soltanian, M.R. Multiscale hyporheic exchange through strongly heterogeneous sediments. Water Resour. Res. 2015, 51, 9127–9140. [Google Scholar] [CrossRef]
- Kalbus, E.; Schmidt, C.; Molson, J.W.; Reinstorf, F.; Schirmer, M. Influence of aquifer and streambed heterogeneity on the distribution of groundwater discharge. Hydrol. Earth Syst. Sci. 2009, 13, 69–77. [Google Scholar] [CrossRef]
- Raghavendra Naganna, S.; Chandra Deka, P. Variability of streambed hydraulic conductivity in an intermittent stream reach regulated by vented dams: A case study. J. Hydrol. 2018, 562, 477–491. [Google Scholar] [CrossRef]
- Schirmer, M.; Leschik, S.; Musolff, A. Current research in urban hydrogeology—A review. Adv. Water Resour. 2013, 51, 280–291. [Google Scholar] [CrossRef]
- Bourke, S.A.; Cook, P.G.; Shanafield, M.; Dogramaci, S.; Clark, J.F. Characterisation of hyporheic exchange in a losing stream using radon-222. J. Hydrol. 2014, 519, 94–105. [Google Scholar] [CrossRef]
- Cranswick, R.H.; Cook, P.G.; Lamontagne, S. Hyporheic zone exchange fluxes and residence times inferred from riverbed temperature and radon data. J. Hydrol. 2014, 519, 1870–1881. [Google Scholar] [CrossRef]
- Schmidt, C.; Bayer-Raich, M.; Schirmer, M. Characterization of spatial heterogeneity of groundwater-stream water interactions using multiple depth streambed temperature measurements at the reach scale. Hydrol. Earth Syst. Sci. 2006, 10, 849–859. [Google Scholar] [CrossRef]
- Gordon, R.P.; Lautz, L.K.; Briggs, M.A.; McKenzie, J.M. Automated calculation of vertical pore-water flux from field temperature time series using the VFLUX method and computer program. J. Hydrol. 2012, 420, 142–158. [Google Scholar] [CrossRef]
- Mojarrad, B.B.; Betterle, A.; Singh, T.; Olid, C.; Wörman, A. The effect of stream discharge on hyporheic exchange. Water 2019, 11, 1436. [Google Scholar] [CrossRef]
- Vandersteen, G.; Schneidewind, U.; Anibas, C.; Schmidt, C.; Seuntjens, P.; Batelaan, O. Determining groundwater-surface water exchange from temperature-time series: Combining a local polynomial method with a maximum likelihood estimator. Water Resour. Res. 2015, 51, 922–939. [Google Scholar] [CrossRef]
- Schneidewind, U.; van Berkel, M.; Anibas, C.; Vandersteen, G.; Schmidt, C.; Joris, I.; Seuntjens, P.; Batelaan, O.; Zwart, H.J. LPMLE3: A novel 1-D approach to study water flow in streambeds using heat as a tracer. Water Resour. Res. 2016, 52, 6596–6610. [Google Scholar] [CrossRef]
- Lewandowski, J.; Angermann, L.; Nützmann, G.; Fleckenstein, J.H. A heat pulse technique for the determination of small-scale flow directions and flow velocities in the streambed of sand-bed streams. Hydrol. Process. 2011, 25, 3244–3255. [Google Scholar] [CrossRef]
- Angermann, L.; Krause, S.; Lewandowski, J. Application of heat pulse injections for investigating shallow hyporheic flow in a lowland river. Water Resour. Res. 2012, 48, W00P02. [Google Scholar] [CrossRef]
- Banks, E.W.; Shanafield, M.A.; Noorduijn, S.; McCallum, J.; Lewandowski, J.; Batelaan, O. Active heat pulse sensing of 3D-flow fields in streambeds. Hydrol. Earth Syst. Sci. 2018, 22, 1917–1929. [Google Scholar] [CrossRef]
- Selker, J.; Van de Giesen, N.; Westhoff, M.; Luxemburg, W.; Parlange, M.B. Fiber optics opens window on stream dynamics. Geophys. Res. Lett. 2006, 33, L24401. [Google Scholar] [CrossRef]
- Lowry, C.S.; Walker, J.F.; Hunt, R.J.; Anderson, M.P. Identifying spatial variability of groundwater discharge in a wetland stream using a distributed temperature sensor. Water Resour. Res. 2007, 43, W10408. [Google Scholar] [CrossRef]
- Tyler, S.W.; Selker, J.S.; Hausner, M.B.; Hatch, C.E.; Torgersen, T.; Thodal, C.E.; Schladow, S.G. Environmental temperature sensing using Raman spectra DTS fiber-optic methods. Water Resour. Res. 2009, 45, W00D23. [Google Scholar] [CrossRef]
- Vogt, T.; Schneider, P.; Hahn-Woernle, L.; Cirpka, O.A. Estimation of seepage rates in a losing stream by means of fiber-optic high-resolution vertical temperature profiling. J. Hydrol. 2010, 380, 154–164. [Google Scholar] [CrossRef]
- Shanafield, M.; McCallum, J.L.; Cook, P.G.; Noorduijn, S. Using basic metrics to analyze high-resolution temperature data in the subsurface. Hydrogeol. J. 2017, 25, 1501–1508. [Google Scholar] [CrossRef]
- Gaona, J.; Meinikmann, K.; Lewandowski, J. Identification of groundwater exfiltration, interflow discharge, and hyporheic exchange flows by fibre optic distributed temperature sensing supported by electromagnetic induction geophysics. Hydrol. Process. 2019, 33, 1390–1402. [Google Scholar] [CrossRef]
- Hare, D.K.; Briggs, M.A.; Rosenberry, D.O.; Boutt, D.F.; Lane, J.W. A comparison of thermal infrared to fiber-optic distributed temperature sensing for evaluation of groundwater discharge to surface water. J. Hydrol. 2015, 530, 153–166. [Google Scholar] [CrossRef]
- Ward, A.S.; Gooseff, M.N.; Singha, K. Imaging hyporheic zone solute transport using electrical resistivity. Hydrol. Process. 2010, 24, 948–953. [Google Scholar] [CrossRef]
- McLachlan, P.J.; Chambers, J.E.; Uhlemann, S.S.; Binley, A. Geophysical characterisation of the groundwater-surface water interface. Adv. Water Resour. 2017, 109, 302–319. [Google Scholar] [CrossRef]
- Wu, L.; Singh, T.; Gomez-Velez, J.; Nützmann, G.; Wörman, A.; Krause, S.; Lewandowski, J. Impact of dynamically changing discharge on hyporheic exchange processes under gaining and losing groundwater conditions. Water Resour. Res. 2018, 54, 10076–10093. [Google Scholar] [CrossRef]
- Singh, T.; Wu, L.; Gomez-Velez, J.D.; Lewandowski, J.; Hannah, D.M.; Krause, S. Dynamic hyporheic zones: Exploring the role of peak flow events on bedform-induced hyporheic exchange. Water Resour. Res. 2019, 55, 218–235. [Google Scholar] [CrossRef]
- Zarnetske, J.P.; Haggerty, R.; Wondzell, S.M.; Baker, M.A. Dynamics of nitrate production and removal as a function of residence time in the hyporheic zone. J. Geophys. Res. 2011, 116, G01025. [Google Scholar] [CrossRef]
- Harvey, J.W.; Bohlke, J.K.; Voytek, M.A.; Scott, D.; Tobias, C.R. Hyporheic zone denitrification: Controls on effective reaction depth and contribution to whole-stream mass balance. Water Resour. Res. 2013, 49, 6298–6316. [Google Scholar] [CrossRef]
- Johnson, Z.C.; Warwick, J.J.; Schumer, R. Factors affecting hyporheic and surface transient storage in a western U.S. river. J. Hydrol. 2014, 510, 325–339. [Google Scholar] [CrossRef]
- Bencala, K.E.; Walters, R.A. Simulation of solute transport in a mountain pool-and-riffle stream: A transient storage model. Water Resour. Res. 1983, 19, 718–724. [Google Scholar] [CrossRef]
- Wörman, A.; Packman, A.I.; Johansson, H.; Jonsson, K. Effect of flow-induced exchange in hyporheic zones on longitudinal transport of solutes in streams and rivers. Water Resour. Res. 2002, 38, 2-1–2-15. [Google Scholar] [CrossRef]
- Gooseff, M.N.; Wondzell, S.M.; Haggerty, R.; Anderson, J. Comparing transient storage modeling and residence time distribution (RTD) analysis in geomorphically varied reaches in the Lookout Creek basin, Oregon, USA. Adv. Water Resour. 2003, 26, 925–937. [Google Scholar] [CrossRef]
- Jury, W.A. Simulation of solute transport using a transfer function model. Water Resour. Res. 1982, 18, 363–368. [Google Scholar] [CrossRef]
- Sardin, M.; Schweich, D.; Leu, F.J.; Van Genuchten, M.T. Modeling the nonequilibrium transport of linearly interacting solutes in porous media: A review. Water Resour. Res. 1991, 27, 2287–2307. [Google Scholar] [CrossRef]
- Knapp, J.L.A.; Cirpka, O.A. Determination of hyporheic travel time distributions and other parameters from concurrent conservative and reactive tracer tests by local-in-global optimization. Water Resour. Res. 2017, 53, 4984–5001. [Google Scholar] [CrossRef]
- Liao, Z.; Cirpka, O.A. Shape-free inference of hyporheic traveltime distributions from synthetic conservative and “smart” tracer tests in streams. Water Resour. Res. 2011, 47, W07510. [Google Scholar] [CrossRef]
- Liao, Z.; Lemke, D.; Osenbrück, K.; Cirpka, O.A. Modeling and inverting reactive stream tracers undergoing two-site sorption and decay in the hyporheic zone. Water Resour. Res. 2013, 49, 3406–3422. [Google Scholar] [CrossRef]
- Battin, T.J.; Besemer, K.; Bengtsson, M.M.; Romani, A.M.; Packmann, A.I. The ecology and biogeochemistry of stream biofilms. Nat. Rev. Microbiol. 2016, 14, 251–263. [Google Scholar] [CrossRef]
- Williams, D.D.; Hynes, H.B.N. The occurrence of benthos deep in the substratum of a stream. Freshw. Biol. 1974, 4, 233–256. [Google Scholar] [CrossRef]
- Zeglin, H.L. Stream microbial diversity responds to environmental changes: Review and synthesis of existing research. Front. Microbiol. 2015, 6, 454. [Google Scholar] [CrossRef] [PubMed]
- Singer, G.; Besemer, K.; Schmitt-Kopplin, P.; Hödl, I.; Battin, T.J. Physical heterogeneity increases biofilm resource use and its molecular diversity in stream mesocosms. PLoS ONE 2010, 5, e9988. [Google Scholar] [CrossRef] [PubMed]
- Romaní, A.M.; Fund, K.; Artigas, J.; Schwartz, T.; Sabater, S.; Obst, U. Relevance of polymeric matrix enzymes during biofilm formation. Microb. Ecol. 2008, 56, 427–436. [Google Scholar] [CrossRef] [PubMed]
- Battin, T.J.; Kaplan, L.A.; Findlay, S.; Hopkinson, C.S.; Marti, E.; Packman, A.I.; Newbold, J.D.; Sabater, F. Biophysical controls on organic carbon fluxes in fluvial networks. Nat. Geosci. 2008, 1, 95–100. [Google Scholar] [CrossRef]
- Mulholland, P.J.; Helton, A.M.; Poole, G.C.; Hall, R.O.; Hamilton, S.K.; Peterson, B.J.; Tank, J.L.; Ashkenas, L.R.; Cooper, L.W.; Dahm, C.N.; et al. Stream denitrification across biomes and its response to anthropogenic nitrate loading. Nature 2008, 452, 202–205. [Google Scholar] [CrossRef]
- Storey, R.G.; Fulthorpe, R.R.; Williams, D.D. Perspectives and predictions on the microbial ecology of the hyporheic zone. Freshw. Biol. 1999, 41, 119–130. [Google Scholar] [CrossRef]
- Robertson, A.L.; Wood, P.J. Ecology of the hyporheic zone: Origins, current knowledge and future directions. Fundam. Appl. Limnol Arch. Hydrobiol. 2010, 176, 279–289. [Google Scholar] [CrossRef]
- Wood, P.J.; Boulton, A.J.; Little, S.; Stubbington, R. Is the hyporheic zone a refugium for aquatic macroinvertebrates during severe low flow conditions? Fundam. Appl. Limnol./Arch. Hydrobiol. 2010, 176, 377–390. [Google Scholar] [CrossRef]
- Eichhorn, M. Natural Systems: The Organisation of Life; John Wiley and Sons: Chichester, UK, 2016; p. 359. [Google Scholar]
- Dole-Olivier, M.J. Surface water-groundwater exchanges in three dimensions on a backwater of the Rhône River. Freshw. Biol. 1998, 40, 93–109. [Google Scholar] [CrossRef]
- Fraser, B.G.; Williams, D.D. Seasonal boundary dynamics of a groundwater/surface-water ecotone. Ecology 1998, 79, 2019–2031. [Google Scholar] [CrossRef]
- Miyake, Y.; Nakano, S. Effects of substratum stability on diversity of stream invertebrates during baseflow at two spatial scales. Freshw. Biol. 2002, 47, 219–230. [Google Scholar] [CrossRef]
- Sliva, L.; Williams, D.D. Responses of hyporheic meiofauna to habitat manipulation. Hydrobiologia 2005, 548, 217–232. [Google Scholar] [CrossRef]
- Davy-Bowker, J.; Sweeting, W.; Wright, N.; Clarke, R.T.; Arnott, S. The distribution of benthic and hyporheic macroinvertebrates from the heads and tails of riffles. Hydrobiologia 2006, 563, 109–123. [Google Scholar] [CrossRef]
- Andrushchyshyn, O.P.; Wilson, K.P.; Williams, D.D. Ciliate communities in shallow groundwater: Seasonal and spatial characteristics. Freshw. Biol. 2007, 52, 1745–1761. [Google Scholar] [CrossRef]
- Magliozzi, C.; Usseglio-Polatera, P.; Meyer, A.; Grabowski, R.C. Functional traits of hyporheic and benthic invertebrates reveal importance of wood-driven geomorphological processes in rivers. Funct. Ecol. 2019, 33, 1758–1770. [Google Scholar] [CrossRef]
- Smock, L.A.; Gladden, J.E.; Riekenberg, J.L.; Smith, L.C.; Black, C.R. Lotic macroinvertebrate production in three dimensions: Channel surface, hyporheic, and floodplain environments. Ecology 1992, 73, 875–886. [Google Scholar] [CrossRef]
- Post, D.M.; Doyle, M.W.; Sabo, J.L.; Finlay, J.C. The problem of boundaries in defining ecosystems: A potential landmine for uniting geomorphology and ecology. Geomorphology 2007, 89, 111–126. [Google Scholar] [CrossRef]
- Peralta-Maraver, I.; Perkins, D.M.; Thompson, M.S.; Fussmann, K.; Reiss, J.; Robertson, A.L. Comparing biotic drivers of litter breakdown across streams compartments. J. Anim. Ecol. 2019, 88, 1146–1157. [Google Scholar] [CrossRef]
- Smith, J.J.; Lake, P.S. The breakdown of buried and surface–placed leaf litter in an upland stream. Hydrobiologia 1993, 271, 141–148. [Google Scholar] [CrossRef]
- Cornut, J.; Elger, A.; Lambrigot, D.; Marmonier, P.; Chauvet, E. Early stages of leaf decomposition are mediated by aquatic fungi in the hyporheic zone of woodland streams. Freshw. Biol. 2010, 55, 2541–2556. [Google Scholar] [CrossRef]
- Peralta-Maraver, I.; Robertson, A.L.; Perkins, D.M. Depth and vertical hydrodynamics constrain the size structure of a lowland streambed community. Biol. Lett. 2019, 15, 20190317. [Google Scholar] [CrossRef] [PubMed]
- Stumm, W.; Morgan, J.J. Redox conditions in natural waters. In Aquatic Chemistry: Chemical Equilibria and Rates in Natural Waters, 3rd ed.; Schnoor, J.L., Zehnder, A., Eds.; Wiley: New York, NY, USA, 1996; pp. 464–489. [Google Scholar] [CrossRef]
- Jones, J.B.; Holmes, R.M.; Fisher, S.G.; Grimm, N.B.; Greene, D.M. Methanogenesis in Arizona, USA dryland streams. Biogeochemistry 1995, 31, 155–173. [Google Scholar] [CrossRef]
- Zarnetske, J.P.; Haggerty, R.; Wondzell, S.M.; Baker, M.A. Labile dissolved organic carbon supply limits hyporheic denitrification. J. Geophys. Res. 2011, 116, G04036. [Google Scholar] [CrossRef]
- Briggs, M.A.; Day-Lewis, F.D.; Zarnetske, J.P.; Harvey, J.W. A physical explanation for the development of redox microzones in hyporheic flow. Geophys. Res. Lett. 2015, 42, 4402–4410. [Google Scholar] [CrossRef]
- Triska, F.J.; Duff, J.H.; Avanzino, R.J. The role of water exchange between a stream channel and its hyporheic zone in nitrogen cycling at the terrestrial-aquatic interface. Hydrobiologia 1993, 251, 167–184. [Google Scholar] [CrossRef]
- Triska, F.J.; Duff, J.H.; Avanzino, R.J. Patterns of hydrological exchange and nutrient transformation in the hyporheic zone of a gravel-bottom stream: Examining terrestrial-aquatic linkages. Freshw. Biol. 1993, 29, 259–274. [Google Scholar] [CrossRef]
- Christensen, P.B.; Nielsen, L.P.; Sørensen, J.; Revsbech, N.P. Denitrification in nitrate-rich streams: Diurnal and seasonal variation related to benthic oxygen metabolism. Limnol. Oceanogr. 1990, 35, 640–651. [Google Scholar] [CrossRef]
- Schaper, J.L.; Seher, W.; Nützmann, G.; Putschew, A.; Jekel, M.; Lewandowski, J. The fate of polar trace organic compounds in the hyporheic zone. Water Res. 2018, 140, 158–166. [Google Scholar] [CrossRef]
- Lewandowski, J.; Nützmann, G. Nutrient retention and release in a floodplain’s aquifer and in the hyporheic zone of a lowland river. Ecol. Eng. 2010, 36, 1156–1166. [Google Scholar] [CrossRef]
- Trauth, N.; Schmidt, C.; Vieweg, M.; Maier, U.; Fleckenstein, J.H. Hyporheic transport and biogeochemical reactions in pool-riffle systems under varying ambient groundwater flow conditions. J. Geophys. Res. Biogeosci. 2014, 119, 910–928. [Google Scholar] [CrossRef]
- De Falco, N.; Boano, F.; Bogler, A.; Bar-Zeev, E.; Arnon, S. Influence of stream-subsurface exchange flux and bacterial biofilms on oxygen consumption under nutrient-rich conditions. J. Geophys. Res. Biogeosci. 2018, 123, 2021–2034. [Google Scholar] [CrossRef]
- Haggerty, R.; Argerich, A.; Marti, E. Development of a “smart” tracer for the assessment of microbiological activity and sediment-water interaction in natural waters: The resazurin-resorufin system. Water Resour. Res. 2008, 44, W00D01. [Google Scholar] [CrossRef]
- Haggerty, R.; Marti, E.; Argerich, A.; von Schiller, D.; Grimm, N.B. Resazurin as a “smart” tracer for quantifying metabolically active transient storage in stream ecosystems. J. Geophys. Res. Biogeosci. 2009, 114, G03014. [Google Scholar] [CrossRef]
- Schirmer, M.; Luster, J.; Linde, N.; Perona, P.; Mitchell, E.A.D.; Barry, D.A.; Hollender, J.; Cirpka, O.A.; Schneider, P.; Vogt, T.; et al. Morphological, hydrological, biogeochemical and ecological changes and challenges in river restoration–The Thur River case study. Hydrol. Earth Syst. Sci. 2014, 18, 1–14. [Google Scholar] [CrossRef]
- Lewandowski, J.; Putschew, A.; Schwesig, D.; Neumann, C.; Radke, M. Fate of organic micropollutants in the hyporheic zone of a eutrophic lowland stream: Results of a preliminary field study. Sci. Total Environ. 2011, 409, 1824–1835. [Google Scholar] [CrossRef]
- Lapworth, D.J.; Gooddy, D.C.; Jarvie, H.P. Understanding phosphorus mobility and bioavailability in the hyporheic zone of a chalk stream. Water Air Soil Pollut. 2010, 218, 213–226. [Google Scholar] [CrossRef]
- Butturini, A.; Sabater, F. Importance of transient storage zones for ammonium and phosphate retention in a sandy-bottom Mediterranean stream. Freshw. Biol. 1999, 41, 593–603. [Google Scholar] [CrossRef]
- Arnon, S.; Yanuka, K.; Nejidat, A. Impact of overlying water velocity on ammonium uptake by benthic biofilms. Hydrol. Process. 2013, 27, 570–578. [Google Scholar] [CrossRef]
- Harvey, J.J.; Gomez-Velez, J.; Schmadel, N.; Scott, D.; Boyer, E.; Alexander, R.; Eng, K.; Golden, H.; Kettner, A.; Konrad, C.; et al. How Hydrologic connectivity regulates water quality in river corridors. J. Am. Water Resour. Assoc. 2019, 55, 369–381. [Google Scholar] [CrossRef]
- Ocampo, C.J.; Oldham, C.E.; Sivapalan, M. Nitrate attenuation in agricultural catchments: Shifting balances between transport and reaction. Water Resour. Res. 2006, 42, W01408. [Google Scholar] [CrossRef]
- Gordon, R.P.; Lautz, L.K.; Daniluk, T.L. Spatial patterns of hyporheic exchange and biogeochemical cycling around cross-vane restoration structures: Implications for stream restoration design. Water Resour. Res. 2013, 49, 2040–2055. [Google Scholar] [CrossRef]
- Hester, E.T.; Hammond, B.; Scott, D.T. Effects of inset floodplains and hyporheic exchange induced by in-stream structures on nitrate removal in a headwater stream. Ecol. Eng. 2016, 97, 452–464. [Google Scholar] [CrossRef]
- Morén, I.; Riml, J.; Wörman, A. In-Stream Water Management Strategies for Reducing Nutrient Loads to the Baltic Sea; BONUS Soils2Sea Deliverable 4.4; KTH Royal Institute of Technology: Stockholm, Sweden, March 2018; Available online: www.Soils2Sea.eu (accessed on 23 October 2019).
- Refsgaard, J.C.; Chubarenko, B.; Donnely, C.; de Jonge, H.; Olesen, J.E.; Steljes, N.; Wachniew, P.; Wörman, A. Spatially differentiated regulation measures-can it save the Baltic Sea from excessive N-loads? Ambio 2019. [Google Scholar] [CrossRef] [PubMed]
- Botter, G.; Basu, N.B.; Zanardo, S.; Rao, P.S.C.; Rinaldo, A. Stochastic modeling of nutrient losses in streams: Interactions of climatic, hydrologic and biogeochemical controls. Water Resour. Res. 2010, 46, W08509. [Google Scholar] [CrossRef]
- Kidd, K.A.; Blanchfield, P.J.; Mills, K.H.; Palace, V.P.; Evans, R.E.; Lazorchak, J.M.; Flick, R.W. Collapse of a fish population after exposure to a synthetic estrogen. Proc. Natl. Acad. Sci. USA 2007, 104, 8897–8901. [Google Scholar] [CrossRef] [PubMed]
- Galus, M.; Rangarajan, S.; Lai, A.; Shaya, L.; Balshine, S.; Wilson, J.Y. Effects of chronic, parental pharmaceutical exposure on zebrafish (Danio rerio) offspring. Aquat. Toxicol. 2014, 151, 124–134. [Google Scholar] [CrossRef]
- Cruz-Rojas, C.; SanJuan-Reyes, N.; Fuentes-Benites, M.P.A.G.; Dublan-García, O.; Galar-Martínez, M.; Islas-Flores, H.; Gómez-Oliván, L.M. Acesulfame potassium: Its ecotoxicity measured through oxidative stress biomarkers in common carp (Cyprinus carpio). Sci. Total Environ. 2019, 647, 772–784. [Google Scholar] [CrossRef]
- Kubec, J.; Hossain, S.; Grabicová, K.; Randák, T.; Kouba, A.; Grabic, R.; Roje, S.; Buřič, M. Oxazepam alters the behavior of crayfish at diluted concentrations, venlafaxine does not. Water 2019, 11, 196. [Google Scholar] [CrossRef]
- Luo, Y.; Guo, W.; Ngo, H.H.; Nghiem, L.D.; Hai, F.I.; Zhang, J.; Liang, S.; Wang, X.C. A review on the occurrence of micropollutants in the aquatic environment and their fate and removal during wastewater treatment. Sci. Total Environ. 2014, 473–474, 619–641. [Google Scholar] [CrossRef]
- Melvin, S.; Leusch, F. Removal of trace organic contaminants from domestic wastewater: A meta-analysis comparison of sewage treatment technologies. Environ. Int. 2016, 92–93, 183–188. [Google Scholar] [CrossRef]
- Phillips, P.; Chalmers, A.; Gray, J.; Kolpin, D.; Foreman, W.; Wall, G. Combined Sewer Overflows: An Environmental Source of Hormones and Wastewater Micropollutants. Environ. Sci. Technol. 2012, 46, 563–579. [Google Scholar] [CrossRef] [PubMed]
- Roehrdanz, P.R.; Feraud, M.; Lee, D.G.; Means, J.C.; Snyder, S.A.; Holden, P.A. Spatial models of sewer pipe leakage predict the occurrence of wastewater indicators in shallow urban groundwater. Environ. Sci. Technol. 2017, 51, 1213–1223. [Google Scholar] [CrossRef] [PubMed]
- Schaider, L.A.; Ackerman, J.M.; Rudel, R.A. Septic systems as sources of organic wastewater compounds in domestic drinking water wells in a shallow sand and gravel aquifer. Sci. Total Environ. 2016, 547, 470–481. [Google Scholar] [CrossRef] [PubMed]
- Eggen, T.; Moeder, M.; Arukwe, A. Municipal landfill leachates: A significant source for new and emerging pollutants. Sci. Total Environ. 2010, 408, 5147–5157. [Google Scholar] [CrossRef]
- Masoner, J.R.; Kolpin, D.W.; Cozzarelli, I.M.; Barber, L.B.; Burden, D.S.; Foreman, W.T.; Forshay, K.J.; Furlong, E.T.; Groves, J.F.; Hladik, M.L.; et al. Urban stormwater: An overlooked pathway of extensive mixed contaminants to surface and groundwaters in the United States. Environ. Sci. Technol. 2019, 53, 10070–10081. [Google Scholar] [CrossRef]
- Jekel, M.; Ruhl, A.S.; Meinel, F.; Zietzschmann, F.; Lima, S.P.; Baur, N.; Wenzel, M.; Gnirß, R.; Sperlich, A.; Dünnbier, U.; et al. Anthropogenic organic micro-pollutants and pathogens in the urban water cycle: Assessment, barriers and risk communication (ASKURIS). Environ. Sci. Eur. 2013, 25, 1–8. [Google Scholar] [CrossRef]
- Schwarzenbach, R.P.; Escher, B.I.; Fenner, K.; Hofstetter, T.B.; Johnson, C.A.; Von Gunten, U.; Wehrli, B. The challenge of micropollutants in aquatic systems. Science 2006, 313, 1072–1077. [Google Scholar] [CrossRef]
- Boulton, A.J.; Datry, T.; Kasahara, T.; Mutz, M.; Stanford, J.A. Ecology and management of the hyporheic zone: Stream-groundwater interactions of running waters and their floodplains. Freshw. Sci. 2010, 29, 26–40. [Google Scholar] [CrossRef]
- Heberer, T.; Massmann, G.; Fanck, B.; Taute, T.; Dunnbier, U. Behaviour and redox sensitivity of antimicrobial residues during bank filtration. Chemosphere 2008, 73, 451–460. [Google Scholar] [CrossRef]
- Huntscha, S.; Singer, H.P.; McArdell, C.S.; Frank, C.E.; Hollender, J. Multiresidue analysis of 88 polar organic micropollutants in ground, surface and wastewater using online mixed-bed multilayer solid-phase extraction coupled to high performance liquid chromatography-tandem mass spectrometry. J. Chromatogr. A 2012, 1268, 74–83. [Google Scholar] [CrossRef]
- Lawrence, J.E.; Skold, M.E.; Hussain, F.A.; Silverman, D.R.; Resh, V.H.; Sedlak, D.L.; Luthy, R.G.; McCray, J.E. Hyporheic zone in urban streams: A review and opportunities for enhancing water quality and improving aquatic habitat by active management. Environ. Eng. Sci. 2013, 30, 480–501. [Google Scholar] [CrossRef]
- Regnery, J.; Barringer, J.; Wing, A.D.; Hoppe-Jones, C.; Teerlink, J.; Drewes, J.E. Start-up performance of a full-scale riverbank filtration site regarding removal of DOC, nutrients, and trace organic chemicals. Chemosphere 2015, 127, 136–142. [Google Scholar] [CrossRef] [PubMed]
- Hollender, J.; Rothardt, J.; Radny, D.; Loos, M.; Epting, J.; Huggenberger, P.; Borer, P.; Singer, H. Target and Non-target Screening of micropollutants at riverbank filtration sites with short residence times using LC-HRMS/MS. Water Res. X 2018, 1, 100007. [Google Scholar] [CrossRef]
- Burke, V.; Greskowiak, J.; Asmuß, T.; Bremermann, R.; Taute, T.; Massmann, G. Temperature dependent redox zonation and attenuation of wastewater-derived organic micropollutants in the hyporheic zone. Sci. Total Environ. 2014, 482–483, 53–61. [Google Scholar] [CrossRef]
- Schaper, J.L.; Posselt, M.; Bouchez, C.; Jaeger, A.; Nützmann, G.; Putschew, A.; Singer, G.; Lewandowski, J. Fate of trace organic compounds in the hyporheic zone: Influence of retardation, the benthic bio-layer and organic carbon. Environ. Sci. Technol. 2019, 53, 4224–4234. [Google Scholar] [CrossRef]
- Jaeger, A.; Coll, C.; Posselt, M.; Rutere, C.; Mechelke, J.; Betterle, A.; Raza, M.; Meinikmann, K.; Mehrtens, A.; Portmann, A.; et al. Using recirculating flumes and a response surface model to investigate the role of hyporheic exchange and bacterial diversity on micropollutant half lives. Environ. Sci. Process. Impacts 2019. accepted. [Google Scholar] [CrossRef]
- Shapiro, O.H.; Kushmaro, A.; Brenner, A. Bacteriophage predation regulates microbial abundance and diversity in a full-scale bioreactor treating industrial wastewater. ISME J. 2010, 4, 327–336. [Google Scholar] [CrossRef]
- Otto, S.; Harms, H.; Wick, L.Y. Effects of predation and dispersal on bacterial abundance and contaminant biodegradation. FEMS Microbiol. Ecol. 2017, 93, fiw241. [Google Scholar] [CrossRef]
- El-Athman, F.; Adrian, L.; Jekel, M.; Putschew, A. Deiodination in the presence of Dehalococcoides mccartyi strain CBDB1: Comparison of the native enzyme and co-factor vitamin B12. Environ. Sci. Pollut. Res. 2019. [Google Scholar] [CrossRef]
- El-Athman, F.; Jekel, M.; Putschew, A. Reaction kinetics of corrinoid-mediated deiodination of iodinated X-ray contrast media and other iodinated organic compounds. Chemosphere 2019, 234, 971–977. [Google Scholar] [CrossRef]
- El-Athman, F.; Adrian, L.; Jekel, M.; Putschew, A. Abiotic reductive deiodination of iodinated organic compounds and X-ray contrast media catalyzed by free corrinoids. Chemosphere 2019, 221, 212–218. [Google Scholar] [CrossRef] [PubMed]
- Jonsson, K.; Johansson, H.; Wörman, A. Sorption behaviour and long-term retention of reactive solutes in the hyporheic zone of streams. J. Environ. Eng. 2004, 130, 573–584. [Google Scholar] [CrossRef]
- Hebig, K.H.; Groza, L.G.; Sabourin, M.J.; Scheytt, T.J.; Ptacek, C.J. Transport behavior of the pharmaceutical compounds carbamazepine, sulfamethoxazole, gemfibrozil, ibuprofen, and naproxen, and the lifestyle drug caffeine, in saturated laboratory columns. Sci. Total Environ. 2017, 590, 708–719. [Google Scholar] [CrossRef] [PubMed]
- Nghiem, L.D.; Hawkes, S. Effects of membrane fouling on the nanofiltration of trace organic contaminants. Desalination 2009, 236, 273–281. [Google Scholar] [CrossRef]
- Packman, A.I.; Brooks, N.H.; Morgan, J.J. A physicochemical model for colloid exchange between a stream and a sand streambed with bed forms. Water Resour. Res. 2000, 36, 2351–2361. [Google Scholar] [CrossRef]
- Packman, A.I.; Brooks, N.H.; Morgan, J.J. Kaolinite exchange between a stream and streambed: Laboratory experiments and validation of a colloid transport model. Water Resour. Res. 2000, 36, 2363–2372. [Google Scholar] [CrossRef]
- Celiz, M.D.; Tso, J.; Aga, D.S. Pharmaceutical metabolites in the environment: Analytical challenges and ecological risks. Environ. Toxicol. Chem. 2009, 28, 2473–2484. [Google Scholar] [CrossRef]
- Henning, N.; Kunkel, U.; Wick, A.; Ternes, T.A. Biotransformation of gabapentin in surface water matrices under different redox conditions and the occurrence of one major TP in the aquatic environment. Water Res. 2018, 137, 290–300. [Google Scholar] [CrossRef]
- Nödler, K.; Hillebrand, O.; Idzik, K.; Strathmann, M.; Schiperski, F.; Zirlewagen, J.; Licha, T. Occurrence and fate of the angiotensin II receptor antagonist transformation product valsartan acid in the water cycle—A comparative study with selected β-blockers and the persistent anthropogenic wastewater indicators carbamazepine and acesulfame. Water Res. 2013, 47, 6650–6659. [Google Scholar] [CrossRef]
- Li, Z.; Sobek, A.; Radke, M. Flume experiments to investigate the environmental fate of pharmaceuticals and their transformation products in streams. Environ. Sci. Technol. 2015, 49, 6009–6017. [Google Scholar] [CrossRef]
- Writer, J.H.; Antweiler, R.C.; Ferrer, I.; Ryan, J.N.; Thurman, E.M. In-stream attenuation of neuro-active pharmaceuticals and their metabolites. Environ. Sci. Technol. 2013, 47, 9781–9790. [Google Scholar] [CrossRef] [PubMed]
- Mechelke, J.; Vermeirssen, E.L.M.; Hollender, J. Passive sampling of organic contaminants across the water-sediment interface of an urban stream. Water Res. 2019, 165, 114966. [Google Scholar] [CrossRef] [PubMed]
- Jaeger, A.; Posselt, M.; Betterle, A.; Schaper, J.; Mechelke, J.; Coll, C.; Lewandowski, J. Spatial and temporal variability in attenuation of polar trace organic compounds in an urban lowland stream. Environ. Sci. Technol. 2019, 53, 2383–2395. [Google Scholar] [CrossRef] [PubMed]
- Ren, J.H.; Packman, A.I. Stream-subsurface exchange of zinc in the presence of silica and kaolinite colloids. Environ. Sci. Technol. 2004, 38, 6571–6581. [Google Scholar] [CrossRef] [PubMed]
- Karwan, D.L.; Saiers, J.E. Hyporheic exchange and streambed filtration of suspended particles. Water Resour. Res. 2012, 48, W01519. [Google Scholar] [CrossRef]
- Fox, A.; Packman, A.I.; Boano, F.; Phillips, C.B.; Arnon, S. Interactions between suspended kaolinite Deposition and hyporheic exchange flux under losing and gaining flow conditions. Geophys. Res. Lett. 2018, 45, 4077–4085. [Google Scholar] [CrossRef]
- Wharton, G.; Mohajeri, S.H.; Righetti, M. The pernicious problem of streambed colmation: A multi-disciplinary reflection on the mechanisms, causes, impacts, and management challenges. Wiley Interdiscip. Rev. Water 2017, e1231. [Google Scholar] [CrossRef]
- Jones, I.; Growns, I.; Arnold, A.; McCall, S.; Bowes, M. The effects of increased flow and fine sediment on hyporheic invertebrates and nutrients in stream mesocosms. Freshw. Biol. 2015, 60, 813–826. [Google Scholar] [CrossRef]
- Descloux, S.; Datry, T.; Marmonier, P. Benthic and hyporheic invertebrate assemblages along a gradient of increasing streambed colmation by fine sediment. Aquat. Sci. 2013, 75, 493–507. [Google Scholar] [CrossRef]
- Mathers, K.L.; Millett, J.; Robertson, A.L.; Stubbington, R.; Wood, P.J. Faunal response to benthic and hyporheic sedimentation varies with direction of vertical hydrological exchange. Freshw. Biol. 2014, 59, 2278–2289. [Google Scholar] [CrossRef]
- Dunscombe, M.; Robertson, A.; Peralta Maraver, I.; Shaw, P. Community structure and functioning below the stream bed across contrasting geologies. Sci. Total Environ. 2018, 630, 1028–1035. [Google Scholar] [CrossRef] [PubMed]
- Kemp, P.; Sear, D.A.; Collins, A.L.; Naden, P.S.; Jones, J.I. The impacts of fine sediment on riverine fish. Hydrol. Process. 2011, 25, 1800–1821. [Google Scholar] [CrossRef]
- Sear, D.A.; Jones, J.I.; Collins, A.L.; Hulin, A.; Burke, N.; Bateman, S.; Pattison, I.; Naden, P.S. Does fine sediment source as well as quantity affect salmonid embryo mortality and development? Sci. Total Environ. 2016, 541, 957–968. [Google Scholar] [CrossRef] [PubMed]
- Larsen, L.; Harvey, J.; Skalak, K.; Goodman, M. Fluorescence-based source tracking of organic sediment in restored and unrestored urban streams. Limnol. Oceanogr. 2015, 60, 1439–1461. [Google Scholar] [CrossRef]
- Wagner, M.; Scherer, C.; Alvarez-Muñoz, D.; Brennholt, N.; Bourrain, X.; Buchinger, S.; Fries, E.; Grosbois, C.; Klasmeier, J.; Marti, T. Microplastics in freshwater ecosystems: What we know and what we need to know. Environ. Sci. Eur. 2014, 26, 12. [Google Scholar] [CrossRef]
- Dris, R.; Imhof, H.; Sanchez, W.; Gasperi, J.; Galgani, F.; Tassin, B.; Laforsch, C. Beyond the ocean: Contamination of freshwater ecosystems with (micro-) plastic particles. Environ. Chem. 2015, 12, 539–550. [Google Scholar] [CrossRef]
- Mani, T.; Hauk, A.; Walter, U.; Burkhardt-Holm, P. Microplastics profile along the Rhine River. Sci. Rep. 2015, 5, 17988. [Google Scholar] [CrossRef]
- Alimi, O.S.; Hernandez, L.M.; Tufenkji, N. Microplastics and nanoplastics in aquatic environments: Aggregation, deposition, and enhanced contaminant transport. Environ. Sci. Technol. 2018, 52, 1704–1724. [Google Scholar] [CrossRef]
- Browne, M.A.; Crump, P.; Niven, S.J.; Teuten, E.; Tonkin, A.; Galloway, T.; Thompson, R. Accumulation of microplastic on shorelines worldwide: Sources and sinks. Environ. Sci. Technol. 2011, 45, 9175–9179. [Google Scholar] [CrossRef]
- Magnusson, K.; Norén, F. Screening of Microplastic Particles in and Down-Stream a Wastewater Treatment Plant; Report C55; Swedish Environmental Research Institute: Stockholm, Sweeden, 2014; p. 22. Available online: http://www.diva-portal.org/smash/get/diva2:773505/FULLTEXT01.pdf (accessed on 23 October 2019).
- Vogelsang, C.; Lusher, A.L.; Dadkhah, M.E.; Sundvor, I.; Umar, M.; Ranneklev, S.B.; Eidsvoll, D.; Meland, S. Microplastics in Road Dust–Characteristics, Pathways and Measures; Research Report 7361-2019; Institute of Transport Economics, Norwegian Centre for Transport Research: Oslo, Norway, 2019; p. 174. Available online: https://niva.brage.unit.no/niva-xmlui/handle/11250/2493537 (accessed on 23 October 2019).
- Kooi, M.; Besseling, E.; Kroeze, C.; Van Wezel, A.P.; Koelmans, A.A. Modeling the fate and transport of plastic debris in freshwaters: Review and guidance. In Freshwater Microplastics; Springer: Cham, Switzerland, 2018; pp. 125–152. [Google Scholar] [CrossRef]
- Besseling, E.; Quik, J.T.; Sun, M.; Koelmans, A.A. Fate of nano-and microplastic in freshwater systems: A modeling study. Environ. Pollut. 2017, 220, 540–548. [Google Scholar] [CrossRef]
- Castañeda, R.A.; Avlijas, S.; Simard, M.A.; Ricciardi, A. Microplastic pollution in St. Lawrence river sediments. Can. J. Fish. Aquat. Sci. 2014, 71, 1767–1771. [Google Scholar] [CrossRef]
- Hoellein, T.J.; Shogren, A.J.; Tank, J.L.; Risteca, P.; Kelly, J.J. Microplastic deposition velocity in streams follows patterns for naturally occurring allochthonous particles. Sci. Rep. 2019, 9, 3740. [Google Scholar] [CrossRef] [PubMed]
- Nel, H.A.; Dalu, T.; Wasserman, R.J. Sinks and sources: Assessing microplastic abundance in river sediment and deposit feeders in an Austral temperate urban river system. Sci. Total Environ. 2018, 612, 950–956. [Google Scholar] [CrossRef]
- Hurley, R.; Woodward, J.; Rothwell, J.J. Microplastic contamination of river beds significantly reduced by catchment-wide flooding. Nat. Geosci. 2018, 11, 251–257. [Google Scholar] [CrossRef]
- Maazouzi, C.; Galassi, D.; Claret, C.; Cellot, B.; Fiers, F.; Martin, D.; Marmonier, P.; Dole-Olivier, M.J. Do benthic invertebrates use hyporheic refuges during streambed drying? A manipulative field experiment in nested hyporheic flowpaths. Ecohydrology 2017, 10, e1865. [Google Scholar] [CrossRef]
- Malcolm, I.A.; Soulsby, C.; Youngson, A.F.; Hannah, D.M.; McLaren, I.S.; Thorne, A. Hydrological influences on hyporheic water quality: Implications for salmon egg survival. Hydrol. Process. 2004, 18, 1543–1560. [Google Scholar] [CrossRef]
- Febria, C.M.; Beddoes, P.; Fulthorpe, R.R.; Williams, D.D. Bacterial community dynamics in the hyporheic zone of an intermittent stream. ISME J. 2012, 6, 1078–1088. [Google Scholar] [CrossRef]
- Datry, T.; Larned, S.T.; Tockner, K. Intermittent rivers: A challenge for freshwater ecology. BioScience 2014, 64, 229–235. [Google Scholar] [CrossRef]
- Bonada, N.; Doledec, S.; Statzner, B. Taxonomic and biological trait differences of stream macroinvertebrate communities between mediterranean and temperate regions: Implications for future climatic scenarios. Glob. Chang. Biol. 2007, 13, 1658–1671. [Google Scholar] [CrossRef]
- Ward, A.S.; Zarnetske, J.P.; Baranov, V.; Blaen, P.J.; Brekenfeld, N.; Chu, R.; Derelle, R.; Drummond, J.; Fleckenstein, J.; Garayburu-Caruso, V.; et al. Co-located contemporaneous mapping of morphological, hydrological, chemical, and biological conditions in a 5th order mountain stream network, Oregon, USA. Earth Syst. Sci. 2019. [Google Scholar] [CrossRef]
- Zheng, L.; Cardenas, M.B.; Wang, L.; Mohrig, D. Ripple effects: Bedform morphodynamics cascading into hyporheic zone biogeochemistry. Water Resour. Res. 2019, 55, 7320–7342. [Google Scholar] [CrossRef]
- Odum, H.T. Primary production in flowing waters. Limnol. Oceanogr. 1956, 1, 102–117. [Google Scholar] [CrossRef]
- Mulholland, P.J.; Houser, J.N.; Maloney, K.O. Stream diurnal dissolved oxygen profiles as indicators of in-stream metabolism and disturbance effects: Fort Benning as a case study. Ecol. Indic. 2005, 5, 243–252. [Google Scholar] [CrossRef]
- Roberts, B.J.; Mulholland, P.J.; Hill, W.R. Multiple scales of temporal variability in ecosystem metabolism rates: Results from 2 years of continuous monitoring in a forested headwater stream. Ecosystems 2007, 10, 588–606. [Google Scholar] [CrossRef]
- Rajwa-Kuligiewicz, A.; Bialik, R.J.; Rowiński, P.M. Dissolved oxygen and water temperature dynamics in lowland rivers over various timescales. J. Hydrol. Hydromech. 2015, 63, 353–363. [Google Scholar] [CrossRef]
- Brandt, T.; Vieweg, M.; Laube, G.; Schima, R.; Goblirsch, T.; Fleckenstein, J.H.; Schmidt, C. Automated in situ oxygen profiling at aquatic−terrestrial interfaces. Environ. Sci. Technol. 2017, 51, 9970–9978. [Google Scholar] [CrossRef]
- Gilbert, J.; Dole-Oliver, M.J.; Marmonier, P.; Vervier, P. Surface water-groundwater ecotones. In The Ecology and Management of Aquatic–Terrestrial Ecotones; Naiman, R.J., Décamps, H., Eds.; Unesco: Paris, France, 1990; pp. 199–225. [Google Scholar]
- Townsend, C.R.; Scarsbrook, M.R.; Dolédec, S. The intermediate disturbance hypothesis, refugia, and biodiversity in streams. Limnol. Oceanogr. 1997, 42, 938–949. [Google Scholar] [CrossRef]
- Robertson, A.L. Lotic meiofaunal community dynamics: Colonisation, resilience and persistence in a spatially and temporally heterogeneous environment. Freshw. Biol. 2000, 44, 135–147. [Google Scholar] [CrossRef]
- Morén, I.; Wörman, A.; Riml, J. Design of remediation actions for nutrient mitigation in the hyporheic zone. Water Resour. Res. 2017, 53, 8872–8899. [Google Scholar] [CrossRef]
- Grant, S.B.; Azizian, M.; Cook, P.; Boano, F.; Rippy, M.A. Factoring stream turbulence into global assessments of nitrogen pollution. Science 2018, 359, 1266–1269. [Google Scholar] [CrossRef]
- Wörman, A.; Packman, A.I.; Marklund, L.; Harvey, J.W.; Stone, S.H. Fractal topography and subsurface water flows from fluvial bedforms to the continental shield. Geophys. Res. Lett. 2007, 34, L07402. [Google Scholar] [CrossRef]
- Stonedahl, S.H.; Harvey, J.W.; Wörman, A.; Salehin, M.; Packman, A.I. A multiscale model for integrating hyporheic exchange from ripples to meanders. Water Resour. Res. 2010, 46, W12539. [Google Scholar] [CrossRef]
- Bridge, J.W. High Resolution In-Situ Monitoring of Hyporheic Zone Biogeochemistry; Science Report SC030155/SR3; Environment Agency: Bristol, UK, 2005; p. 51. Available online: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/291498/scho0605bjco-e-e.pdf (accessed on 23 October 2019).
- Posselt, M.; Jaeger, A.; Schaper, J.L.; Radke, M.; Benskin, J.P. Determination of polar organic micropollutants in surface and pore water by high-resolution sampling-direct injection-ultra high performance liquid chromatography-tandem mass spectrometry. Environ. Sci. Process. Impacts 2018, 20, 1716–1727. [Google Scholar] [CrossRef] [PubMed]
- Saenger, N.; Kitanidis, P.K.; Street, R. A numerical study of surface-subsurface exchange processes at a riffle-pool pair in the Lahn River, Germany. Water Resour. Res. 2005, 41, 12424. [Google Scholar] [CrossRef]
- Cardenas, M.B.; Wilson, J.L. Hydrodynamics of coupled flow above and below a sediment-water interface with triangular bed-forms. Adv. Water Resour. 2007, 30, 301–313. [Google Scholar] [CrossRef]
- Cardenas, M.B.; Wilson, J.L. Effects of current-bed form induced fluid flow on the thermal regime of sediments. Water Resour. Res. 2007, 43, W08431. [Google Scholar] [CrossRef]
- Cardenas, M.B.; Wilson, J.L. Dunes, turbulent eddies, and interfacial exchange with permeable sediments. Water Resour. Res. 2007, 43, W08412. [Google Scholar] [CrossRef]
- Bardini, L.; Boano, F.; Cardenas, M.B.; Revelli, R.; Ridolfi, L. Nutrient cycling in bedform induced hyporheic zones. Geochim. Cosmochim. Acta 2012, 84, 47–61. [Google Scholar] [CrossRef]
- Trauth, N.; Schmidt, C.; Maier, U.; Vieweg, M.; Fleckenstein, J.H. Coupled 3-D stream flow and hyporheic flow model under varying stream and ambient groundwater flow conditions in a pool-riffle system. Water Resour. Res. 2013, 49, 5834–5850. [Google Scholar] [CrossRef]
- Chen, X.B.; Cardenas, M.B.; Chen, L. Three-dimensional versus two-dimensional bed form-induced hyporheic exchange. Water Resour. Res. 2015, 51, 2923–2936. [Google Scholar] [CrossRef]
- Chen, X.; Cardenas, M.B.; Chen, L. Hyporheic exchange driven by three-dimensional sandy bed forms: Sensitivity to and prediction from bed form geometry. Water Resour. Res. 2018, 54. [Google Scholar] [CrossRef]
- Ren, J.; Wang, X.; Zhou, Y.; Chen, B.; Men, L. An analysis of the factors affecting hyporheic exchange based on numerical modeling. Water 2019, 11, 665. [Google Scholar] [CrossRef]
- Brunner, P.; Simmons, C.T. HydroGeoSphere: A fully integrated, physically based hydrological model. Groundwater 2012, 50, 170–176. [Google Scholar] [CrossRef]
- Oxtoby, O.; Heyns, J.; Suliman, R. A finite-volume solver for two-fluid flow in heterogeneous porous media based on OpenFOAM. Paper presented at the Open Source CFD International Conference, Hamburg, Germany, 24–25 October 2013. [Google Scholar]
- Broecker, T.; Teuber, K.; Elsesser, W.; Hinkelmann, R. Multiphase modeling of hydrosystems using OpenFOAM. In Advances in Hydroinformatics; Springer: Singapore, 2018; pp. 1013–1029. [Google Scholar] [CrossRef]
- Broecker, T.; Teuber, K.; Sobhi Gollo, V.; Nützmann, G.; Hinkelmann, R. Integral flow modelling approach for surface water-groundwater interaction along a rippled streambed. Water 2019, 11, 1517. [Google Scholar] [CrossRef]
- Honti, M.; Bischoff, F.; Moser, A.; Stamm, C.; Baranya, S.; Fenner, K. Relating Degradation of Pharmaceutical Active Ingredients in a Stream Network to Degradation in Water-Sediment Simulation Tests. Water Resour. Res. 2018, 54, 9207–9223. [Google Scholar] [CrossRef]
- Lindim, C.; van Gils, J.; Cousins, I.T. A large-scale model for simulating the fate & transport of organic contaminants in river basins. Chemosphere 2016, 144, 803–810. [Google Scholar] [CrossRef]
- Feijtel, T.; Boeije, G.; Matthies, M.; Young, A.; Morris, G.; Gandolfi, C.; Hansen, B.; Fox, K.; Holt, M.; Koch, V.; et al. Development of a geography-referenced regional exposure assessment tool for European rivers-great-er contribution to great-er #1. Chemosphere 1997, 34, 2351–2373. [Google Scholar] [CrossRef]
- Coll, C.; Lindim, C.; Sobek, A.; Sohn, M.D.; MacLeod, M. Prospects for finding Junge variability-lifetime relationships for micropollutants in the Danube river. Environ. Sci. Process. Impacts 2019. [Google Scholar] [CrossRef]
- Frei, S.; Peiffer, S. Exposure times rather than residence times control redox transformation efficiencies in riparian wetlands. J. Hydrol. 2016, 543, 182–196. [Google Scholar] [CrossRef]
- Pittroff, M.; Frei, S.; Gilfedder, B.S. Quantifying nitrate and oxygen reduction rates in the hyporheic zone using 222Rn to upscale biogeochemical turnover in rivers. Water Resour. Res. 2017, 53, 563–579. [Google Scholar] [CrossRef]
- Grabowski, R.C.; Gurnell, A.M. Diagnosing problems of fine sediment delivery and transfer in a lowland catchment. Aquat. Sci. 2016, 78, 95–106. [Google Scholar] [CrossRef]
- Grabowski, R.C.; Gurnell, A.M.; Burgess-Gamble, L.; England, J.; Holland, D.; Klaar, M.J.; Morrissey, I.; Uttley, C.; Wharton, G. The current state of the use of large wood in river restoration and management. Water Environ. J. 2019, 33, 366–377. [Google Scholar] [CrossRef]
- Azinheira, D.L.; Scott, D.T.; Hession, W.; Hester, E.T. Comparison of effects of inset floodplains and hyporheic exchange induced by in-stream structures on solute retention. Water Resour. Res. 2014, 50, 6168–6190. [Google Scholar] [CrossRef]
- Lammers, R.W.; Bledsoe, B.P. What role does stream restoration play in nutrient management? Crit. Rev. Environ. Sci. Technol. 2017, 47, 335–371. [Google Scholar] [CrossRef]
- Herzog, S.P.; Higgins, C.P.; Singha, K.; McCray, J.E. Performance of engineered streambeds for inducing hyporheic transient storage and attenuation of resazurin. Environ. Sci. Technol. 2018, 52, 10627–10636. [Google Scholar] [CrossRef] [PubMed]
- Ward, A.S.; Morgan, J.A.; White, J.R.; Royer, T.V. Streambed restoration to remove fine sediment alters reach-scale transient storage in a low-gradient fifth-order river, Indiana, USA. Hydrol. Process. 2018, 32, 1786–1800. [Google Scholar] [CrossRef]
- Herzog, S.P.; Higgins, C.P.; McCray, J.E. Engineered streambeds for induced hyporheic flow: Enhanced removal of nutrients, pathogens, and metals from urban streams. J. Environ. Eng. 2015, 142, 04015053. [Google Scholar] [CrossRef]
- Bernhardt, E.S.; Palmer, M.A. Restoring streams in an urbanizing world. Freshw. Biol. 2007, 52, 738–751. [Google Scholar] [CrossRef]
- Mutz, M.; Kalbus, E.; Meinecke, S. Effect of instream wood on vertical water flux in low-energy sand bed flume experiments. Water Resour. Res. 2007, 43, 1–10. [Google Scholar] [CrossRef]
- Thompson, M.S.A.; Brooks, S.J.; Sayer, C.D.; Woodward, G.; Axmacher, J.C.; Perkins, D.M.; Gray, C. Large woody debris “rewilding” rapidly restores biodiversity in riverine food webs. J. Appl. Ecol. 2018, 55, 895–904. [Google Scholar] [CrossRef]
- Palmer, M.A.; Menninger, H.L.; Bernhardt, E. River restoration, habitat heterogeneity and biodiversity: A failure of theory or practice? Freshw. Biol. 2010, 55, 205–222. [Google Scholar] [CrossRef]
- Magliozzi, C.; Coro, G.; Grabowski, R.C.; Packman, A.I.; Krause, S. A multiscale statistical method to identify potential areas of hyporheic exchange for river restoration planning. Environ. Model. Softw. 2019, 111, 311–323. [Google Scholar] [CrossRef]
- Betterle, A.; Schirmer, M.; Botter, G. Characterizing the spatial correlation of daily streamflows. Water Resour. Res. 2017, 53, 1646–1663. [Google Scholar] [CrossRef]
- Betterle, A.; Schirmer, M.; Botter, G. Flow dynamics at the continental scale: Streamflow correlation and hydrological similarity. Hydrol. Process. 2018, 33, 627–646. [Google Scholar] [CrossRef]
| Group | Definition |
|---|---|
| Biofilms | Unicellular consortia of prokaryotes (archaea and bacteria), fungi, and algae (in the top sediment layers) embedded in a porous extracellular matrix. |
| Protozoa | Eukaryotic single cell free-living organisms such as flagellates, ciliates, and amoeba. |
| Meiofauna | Eumetazoa invertebrates whose body size generally ranges between 0.45 μm and 500 μm. |
| Macroinvertebrates | Eumetazoa invertebrates whose body size is generally greater than 500 μm. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lewandowski, J.; Arnon, S.; Banks, E.; Batelaan, O.; Betterle, A.; Broecker, T.; Coll, C.; Drummond, J.D.; Gaona Garcia, J.; Galloway, J.; et al. Is the Hyporheic Zone Relevant beyond the Scientific Community? Water 2019, 11, 2230. https://doi.org/10.3390/w11112230
Lewandowski J, Arnon S, Banks E, Batelaan O, Betterle A, Broecker T, Coll C, Drummond JD, Gaona Garcia J, Galloway J, et al. Is the Hyporheic Zone Relevant beyond the Scientific Community? Water. 2019; 11(11):2230. https://doi.org/10.3390/w11112230
Chicago/Turabian StyleLewandowski, Jörg, Shai Arnon, Eddie Banks, Okke Batelaan, Andrea Betterle, Tabea Broecker, Claudia Coll, Jennifer D. Drummond, Jaime Gaona Garcia, Jason Galloway, and et al. 2019. "Is the Hyporheic Zone Relevant beyond the Scientific Community?" Water 11, no. 11: 2230. https://doi.org/10.3390/w11112230
APA StyleLewandowski, J., Arnon, S., Banks, E., Batelaan, O., Betterle, A., Broecker, T., Coll, C., Drummond, J. D., Gaona Garcia, J., Galloway, J., Gomez-Velez, J., Grabowski, R. C., Herzog, S. P., Hinkelmann, R., Höhne, A., Hollender, J., Horn, M. A., Jaeger, A., Krause, S., ... Wu, L. (2019). Is the Hyporheic Zone Relevant beyond the Scientific Community? Water, 11(11), 2230. https://doi.org/10.3390/w11112230











