Silica Storage, Fluxes, and Nutrient Stoichiometry in Different Benthic Primary Producer Communities in the Littoral Zone of a Deep Subalpine Lake (Lake Iseo, Italy)
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Monitoring Design and Sampling
2.3. Benthic Fluxes and Benthic Metabolism
2.4. Primary Producer Biomass and Sediment Features
2.5. Analytical Methods
2.6. Statistical Analyses
3. Results
3.1. Water and Sediment Characteristics
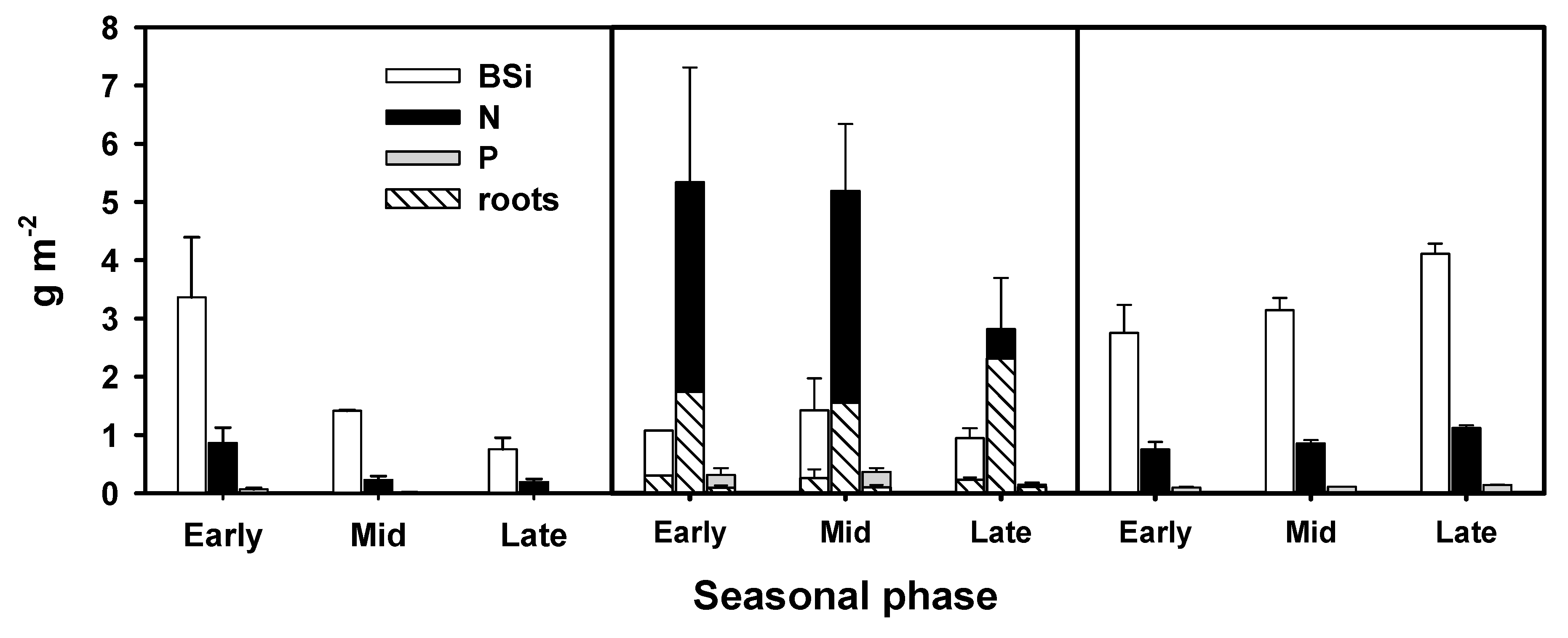
3.2. Primary Producer Biomass and Elemental Composition
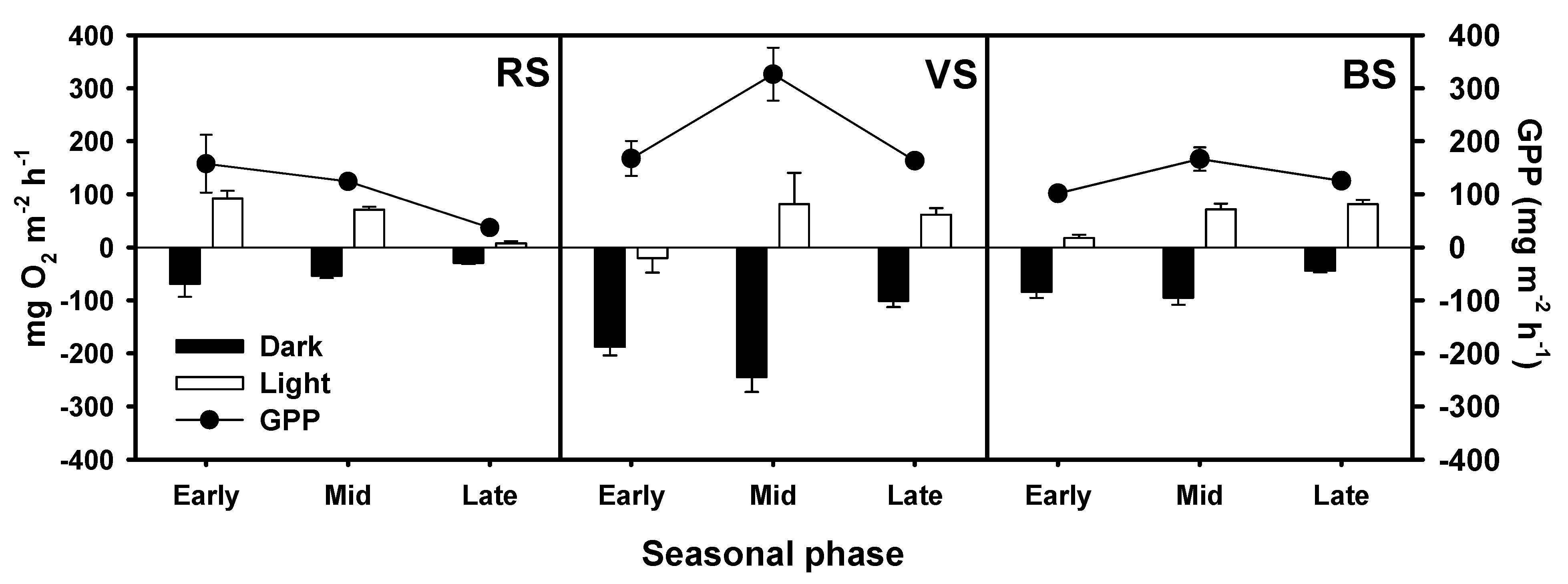
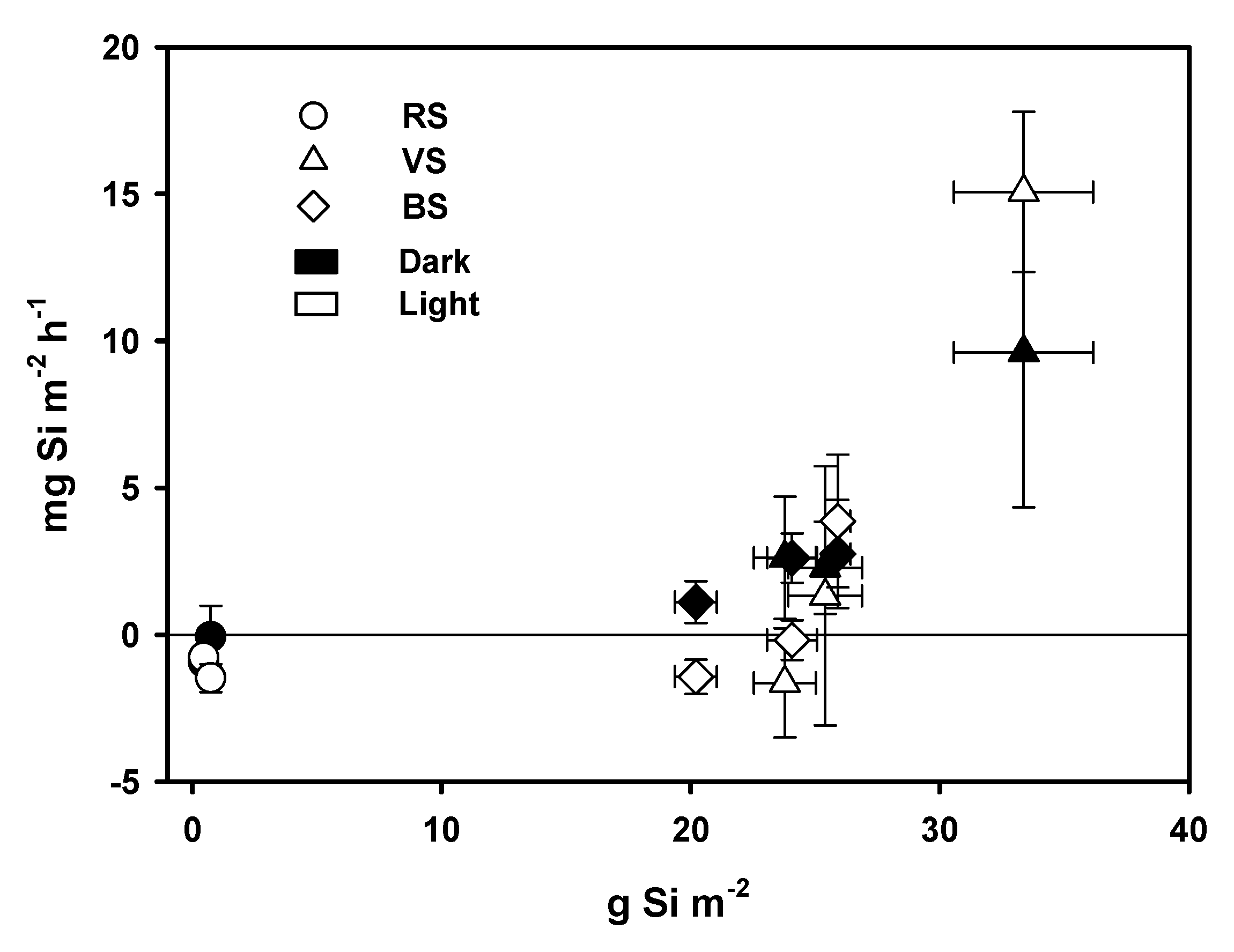
3.3. Oxygen Fluxes and Gross Primary Production
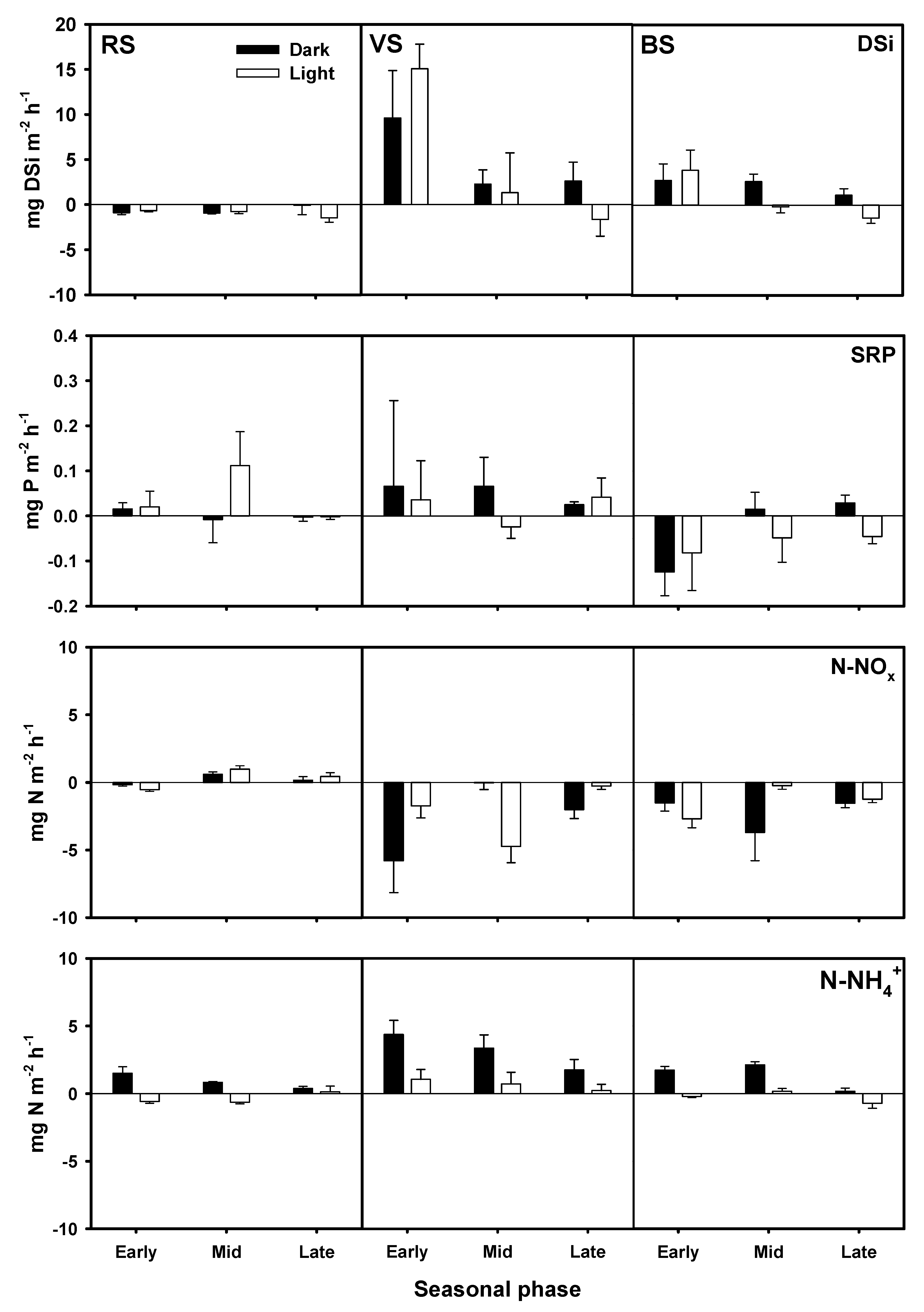
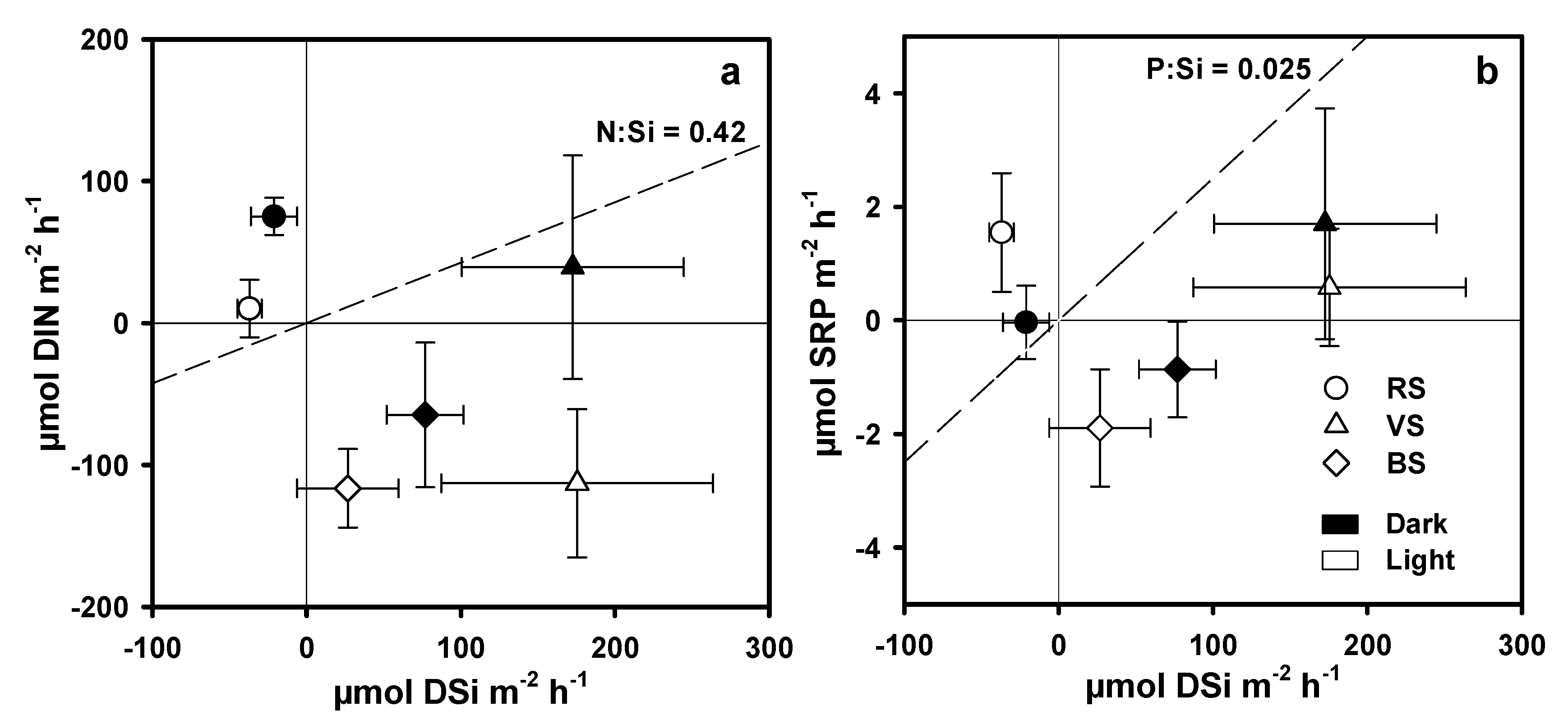
3.4. Nutrient Fluxes across the Water Sediment Interface
4. Discussion
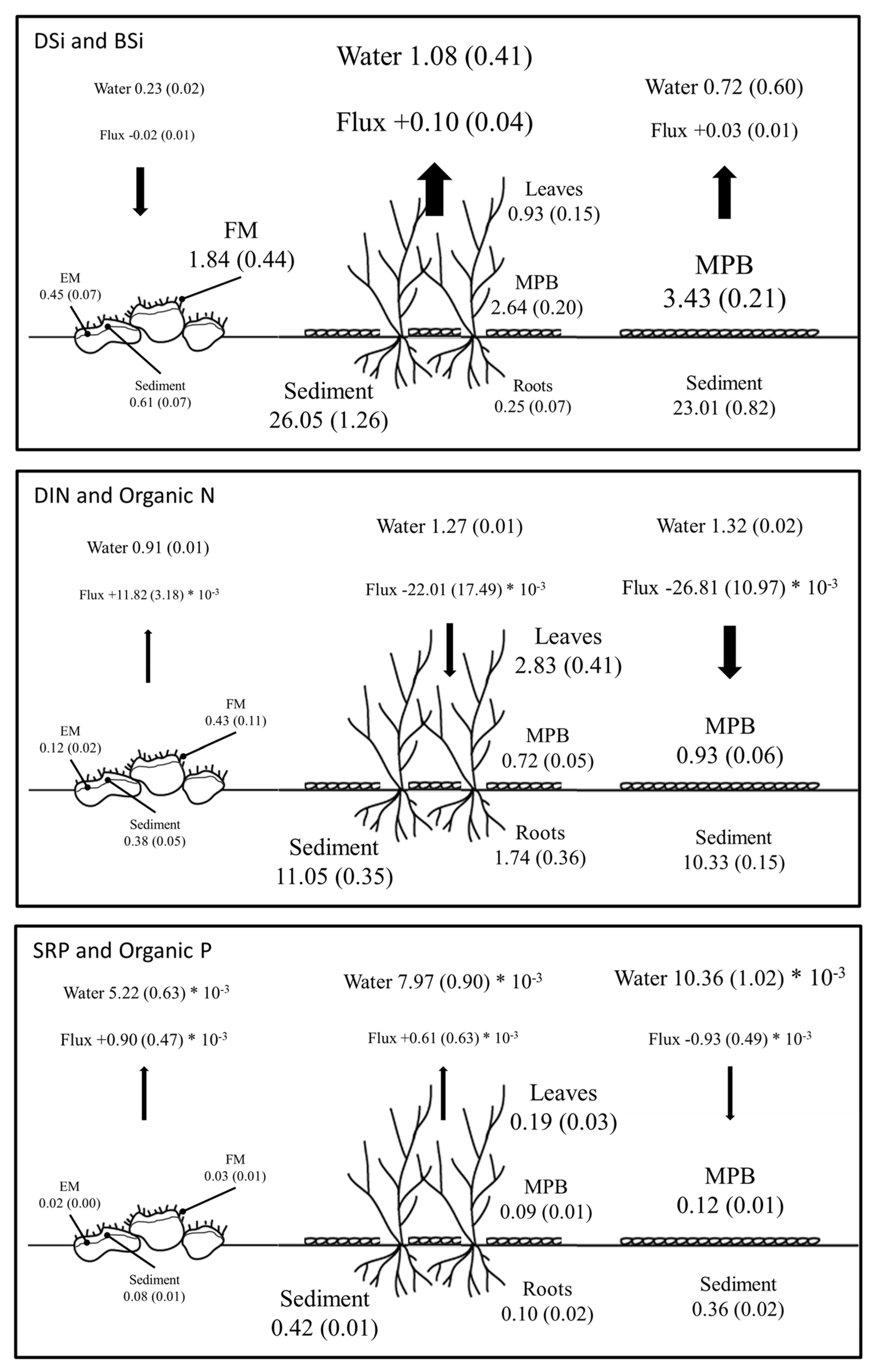
4.1. BSi Storage in Primary Producer Biomass and Surficial Sediment and BSi, N, and P Stoichiometry
4.2. DSi, DIN, and SRP Fluxes across the Water Sediment Interface
4.3. The Fate of DSi in the Littoral Zone in Relation to Submerged Aquatic Vegetation
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Redfield, A.; Ketchum, B.H.; Richards, F.A. The influence of organisms of the composition of seawater. In The Sea; Hill, M.N., Ed.; Wiley-Interscience: New York, NY, USA, 1963; Volume 2, pp. 26–77. [Google Scholar]
- Egge, J.K.; Aksnes, D.L. Silicate as regulating nutrient in phytoplankton competition. Mar. Ecol. Prog. Ser. 1992, 83, 281–289. [Google Scholar] [CrossRef]
- Struyf, E.; Conley, D.J. Emerging understanding of the ecosystem silica filter. Biogeochemistry 2012, 107, 9–18. [Google Scholar] [CrossRef]
- Frings, P.J.; Clymans, W.; Jeppesen, E.; Lauridsen, T.L.; Struyf, E.; Conley, D.J. Lack of steady-state in the global biogeochemical Si cycle: Emerging evidence from lake Si sequestration. Biogeochemistry 2014, 117, 225–277. [Google Scholar] [CrossRef]
- Vandevenne, F.; Struyf, E.; Clymans, W.; Meire, P. Agricultural silica harvest: Have human created a new loop in the global silica cycle? Front. Ecol. Environ. 2012, 10, 243–248. [Google Scholar] [CrossRef]
- Meybeck, M.; Vörösmarty, C. Fluvial filtering of land-to-ocean fluxes: From natural Holocene variations to Anthropocene. C. R. Geosci. 2005, 337, 107–123. [Google Scholar] [CrossRef]
- Harrison, J.A.; Maranger, R.J.; Alexander, R.B.; Giblin, A.E.; Jacinthe, P.; Mayorga, E.; Seitzinger, S.P.; Sobota, D.J.; Wollheim, W.M. The regional and global significance of nitrogen removal in lakes and reservoirs. Biogeochemistry 2009, 93, 143–157. [Google Scholar] [CrossRef]
- Struyf, E.; Conley, D.J. Silica: An essential nutrient in wetland biogeochemistry. Front. Ecol. Environ. 2009, 7, 88–94. [Google Scholar] [CrossRef]
- Verburg, P.; Horrox, J.; Chaney, E.; Rutherford, J.C.; Quinn, J.M.; Wilcock, R.J. Nutrient ratios, differential retention, and the effect on nutrient limitation in a deep oligotrophic lake. Hydrobiologia 2013, 718, 119–130. [Google Scholar] [CrossRef]
- Humborg, C.; Ittekkot, V.; Cociasu, A.; Bodungen, B.V. Effect of Danube river dam on Black Sea biogeochemistry and ecosystem structure. Nature 1997, 386, 385–388. [Google Scholar] [CrossRef]
- Ittekkot, V.; Humborg, C.; Schafer, P. Hydrological Alterations and Marine Biogeochemistry: A Silicate Issue? Bioscience 2000, 50, 776–782. [Google Scholar] [CrossRef]
- Scheffer, M. Ecology of Shallow Lakes, 1th ed.; Springer: Dordrecht, The Netherlands, 1997; Volume 22, pp. 1–357. [Google Scholar]
- Salmaso, N.; Anneville, O.; Straile, D. European large perialpine lakes under anthropogenic pressures and climate change: Present status, research gaps and future challenges. Hydrobiologia 2018, 824, 1–32. [Google Scholar] [CrossRef]
- Nizzoli, D.; Bartoli, M.; Azzoni, R.; Longhi, D.; Castaldelli, G.; Viaroli, P. Denitrification in a meromictic lake and its relevance to nitrogen flows within a moderately impacted forested catchment. Biogeochemistry 2018, 137, 143–161. [Google Scholar] [CrossRef]
- Viaroli, P.; Azzoni, R.; Bartoli, M.; Iacumin, P.; Salmaso, N.; Nizzoli, D. Persistence of meromixis and its effects on redox conditions and trophic status in Lake Idro (Southern Alps, Italy). Hydrobiologia 2018, 824, 51–69. [Google Scholar] [CrossRef]
- Vadeboncoeur, Y.; Steinman, A.D. Periphyton Function in Lake Ecosystems. Sci. World J. 2002, 2, 1449–1468. [Google Scholar] [CrossRef] [PubMed]
- Strayer, D.L.; Findlay, S.E.G. Ecology of freshwater shore zones. Aquat. Sci. 2010, 72, 127–163. [Google Scholar] [CrossRef]
- Zohary, T.; Ostrovsky, I. Ecological impacts of excessive water level fluctuations in stratified freshwater lakes. Inland Waters 2011, 1, 47–59. [Google Scholar] [CrossRef]
- Schoelynck, J.; Bal, K.; Backx, H.; Okruszko, T.; Meire, P.; Struyf, E. Silica uptake in aquatic and wetland macrophytes: A strategic choice between silica, lignin and cellulose? New Phytol. 2010, 186, 385–391. [Google Scholar] [CrossRef]
- Alfredsson, H.; Clymans, W.; Stadmark, J.; Conley, D.; Rousk, J. Bacterial and fungal colonization and decomposition of submerged plant litter: Consequences for biogenic. FEMS Microbiol. Ecol. 2016, 92, fiw011. [Google Scholar] [CrossRef]
- Borrelli, N.; Fernández, M.; Maris, S.; Osterrieth, M. Calcium and silica biomineralizations in leaves of eleven aquatic species of the Pampean Plain, Argentina. Aquat. Bot. 2011, 94, 29–36. [Google Scholar] [CrossRef]
- Carey, J.C.; Fulweiler, R.W. Nitrogen enrichment increases net silica accumulation in a temperate salt marsh. Limnol. Oceanogr. 2013, 58, 99–111. [Google Scholar] [CrossRef]
- Bartoli, M.; Nizzoli, D.; Viaroli, P. Microphytobenthos activity and fluxes at the sediment-water interface: Interactions and spatial variability. Aquat. Ecol. 2003, 37, 341–349. [Google Scholar] [CrossRef]
- Nizzoli, D.; Bartoli, M.; Cooper, M.; Welsh, D.T.; Underwood, G.J.C.; Viaroli, P. Implications for oxygen, nutrient fluxes and denitrification rates during the early stage of sediment colonisation by the polychaete Nereis spp. in four estuaries. Estuar. Coast. Shelf Sci. 2007, 75, 125–134. [Google Scholar] [CrossRef]
- Wetzel, R.G.; Likens, G.E. Limnological Analyses, 2nd ed.; Springer: New York, NY, USA, 1991; pp. 1–391. [Google Scholar]
- Den Heyer, C.; Kalff, J. Organic matter mineralization rates in sediments: A within- and among-lake study. Limnol. Oceanogr. 1998, 43, 695–705. [Google Scholar] [CrossRef]
- Bruesewitz, D.A.; Tank, J.L.; Hamilton, S.K. Incorporating spatial variation of nitrification and denitrification rates into whole-lake nitrogen dynamics. J. Geophys. Res. 2012, 117, 1–12. [Google Scholar] [CrossRef]
- Kelderman, P.; Lindeboom, H.J.; Klein, J. Light dependent sediment-water exchange of dissolved reactive phosphorus and silicon in a producing microflora mat. Hydrobiologia 1988, 159, 137–147. [Google Scholar] [CrossRef]
- Spears, B.M.; Carvalho, L.; Perkins, R.; Paterson, D.M. Effects of light on sediment nutrient flux and water column nutrient stoichiometry in a shallow lake. Water Res. 2008, 42, 977–986. [Google Scholar] [CrossRef]
- Glibert, P.M. Eutrophication, harmful algae and biodiversity—Challenging paradigms in a world of complex nutrient changes. Mar. Pollut. Bull. 2017, 124, 591–606. [Google Scholar] [CrossRef]
- Van Damme, S.; Frank, D.; Micky, T.; Olivier, B.; Eric, S.; Britta, G.; Oswald, V.C.; Patrick, M. Tidal exchange between a freshwater tidal marsh and an impacted estuary: The Scheldt estuary, Belgium. Estuar. Coast. Shelf Sci. 2009, 85, 197–207. [Google Scholar] [CrossRef]
- Carey, J.C.; Fulweiler, R.W. Salt marsh tidal exchange increases residence time of silica in estuaries. Limnol. Oceanogr. 2014, 59, 1203–1212. [Google Scholar] [CrossRef]
- Italian National Institute of Statistics (ISTAT). 2010. Available online: http://dati. istat.it/ (accessed on 21 April 2017).
- Salmaso, N.; Mosello, R. Limnological research in the deep southern subalpine lakes: Synthesis, directions and perspectives. Adv. Oceanogr. Limnol. 2010, 1, 29–66. [Google Scholar] [CrossRef]
- Nizzoli, D.; Welsh, D.T.; Longhi, D.; Viaroli, P. Influence of Potamogeton pectinatus and microphytobenthos on benthic metabolism, nutrient fluxes and denitrification in a freshwater littoral sediment in an agricultural landscape: N assimilation versus N removal. Hydrobiologia 2014, 737, 183–200. [Google Scholar] [CrossRef]
- Maasri, A.; Fayolle, S.; Gandouin, E.; Garnier, R.; Franquet, E. Epilithic chironomid larvae and water enrichment: Is larval distribution explained by epilithon quantity or quality? J. N. Am. Benthol. Soc. 2008, 27, 38–51. [Google Scholar] [CrossRef]
- Sigmon, D.E.; Cahoon, L.B. Comparative effects of benthic microalgae and phytoplankton on dissolved silica fluxes. Aquat. Microb. Ecol. 1997, 13, 275–284. [Google Scholar] [CrossRef]
- de Jonge, V.N. Fluctuations in the Organic Carbon to Chlorophyll a Ratios for Estuarine Benthic Diatom Populations. Mar. Ecol. Prog. Ser. 1980, 2, 345–353. [Google Scholar] [CrossRef]
- Kahlert, M. C:N:P ratios of freshwater benthic algae. Arch. Hydrobiol. Spec. Issue Adv. Limnol. 1998, 51, 105–114. [Google Scholar]
- Sundback, K.; Miles, A.; Goransson, E. Nitrogen fluxes, denitrification and the role of microphytobenthos in microtidal shallow-water sediments: An annual study. Mar. Ecol. Prog. Ser. 2000, 200, 59–76. [Google Scholar] [CrossRef]
- American Public Health Association (APHA). Standard Methods for the Examination of Water and Wastewaters, 20th ed.; APHA: Washington, DC, USA, 1998. [Google Scholar]
- Koroleff, F. Direct determination of ammonia in natural waters as indophenol blue. Information on techniques and methods for seawater analysis. ICES Interlab. Rep 1970, 3, 19–22. [Google Scholar]
- Valderrama, J.C. The simultaneous analysis of total nitrogen and total phosphorus in natural waters. Mar. Chem. 1981, 10, 109–122. [Google Scholar] [CrossRef]
- Golterman, H.L.; Clymo, R.S.; Ohnstand, M.A.M. Methods for Physical and Chemical Analysis of Freshwaters, 8th ed.; Blackwell: Oxford, UK, 1978; pp. 1–213. [Google Scholar]
- Lorenzen, C.J. Determination of chlorophyll and pheo-pigments: Spectrophotometric equations. Limnol. Oceanogr. Methods 1967, 12, 343–346. [Google Scholar] [CrossRef]
- Azzoni, R.; Nizzoli, D.; Bartoli, M.; Christian, R.R. Factors controlling benthic biogeochemistry in urbanized coastal systems: An example from Venice (Italy). Estuaries Coasts 2015, 38, 1016–1031. [Google Scholar] [CrossRef]
- Aspila, K.; Agemian, H.; Chau, A.S.Y. A semi-automated method for the determination of inorganic, organic and total phosphate in sediments. Analyst 1976, 101, 187–197. [Google Scholar] [CrossRef] [PubMed]
- Struyf, E.; Van Damme, S.; Gribsholt, B.; Middelburg, J.J.; Meire, P. Biogenic silica in tidal freshwater marsh sediments and vegetation (Schelde estuary, Belgium). Mar. Ecol. Prog. Ser. 2005, 303, 51–60. [Google Scholar] [CrossRef]
- DeMaster, D.J. The supply and accumulation of silica in the marine environment. Geochim. Cosmochim. Acta 1981, 45, 1715–1732. [Google Scholar] [CrossRef]
- Pinheiro, J.; Bates, D.; DebRoy, S.; Sarkar, D. Nlme: Linear and Nonlinear Mixed Effects Models 2016; R Package Version 3.1-126; R Foundation for Statistical Computing: Vienna, Austria, 2016. [Google Scholar]
- Lenth, R.; Singmann, H.; Love, J.; Buerkner, P.; Herve, M. Estimated Marginal Means, Aka Least-Squares Means 2018; R Package Version 1.3.1; R Foundation for Statistical Computing: Vienna, Austria, 2018. [Google Scholar]
- The R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019. [Google Scholar]
- Gommes, R.; Muntau, H. The chemical composition of limnophytes in lake maggiore. Mem. Ist. Ital. Idrobiol. 1981, 38, 237–307. [Google Scholar]
- Xing, W.; Wu, H.; Hao, B.; Liu, G. Stoichiometric characteristics and responses of submerged macrophytes to eutrophication in lakes along the middle and lower reaches of the Yangtze River. Ecol. Eng. 2013, 54, 16–21. [Google Scholar] [CrossRef]
- Schaller, J.; Schoelynck, J.; Struyf, E.; Meire, P. Silicon Affects Nutrient Content and Ratios of Wetland Plants. Silicon 2016, 8, 479–485. [Google Scholar] [CrossRef]
- Schoelynck, J.; Struyf, E. Silicon in aquatic vegetation. Funct. Ecol. 2016, 30, 1323–1330. [Google Scholar] [CrossRef]
- Schoelynck, J.; Bal, K.; Puijalon, S.; Meire, P.; Struyf, E. Hydrodynamically mediated macrophyte silica dynamics. Plant Biol. 2012, 14, 997–1005. [Google Scholar] [CrossRef]
- Newberry, T.L.; Schelske, C.L. Biogenic silica record in the sediments of Little Round Lake, Ontario. Hydrobiologia 1986, 143, 293–300. [Google Scholar] [CrossRef]
- Conley, D.J.; Quigley, M.A.; Schelske, C.L. Silica and phosphorus flux from sediments: Importance of internal recycling in Lake Michigan. Can. J. Fish. Aquat. Sci. 1988, 45, 1030–1035. [Google Scholar] [CrossRef]
- House, W.A.; Denison, F.H.; Warwick, M.S.; Zhmud, B.V. Dissolution of silica and the development of concentration profiles in freshwater sediments. Appl. Geochem. 2000, 15, 425–438. [Google Scholar] [CrossRef]
- Friedrich, J.; Dinkel, C.; Grieder, E.; Radan, S.; Secrieru, D.; Steingruber, S.; Wehrli, B. Nutrient uptake and benthic regeneration in Danube Delta Lakes. Biogeochemistry 2003, 64, 373–398. [Google Scholar] [CrossRef]
- Ferris, J.A.; Lehman, J.T. Interannual variation in diatom bloom dynamics: Roles of hydrology, nutrient limitation, sinking, and whole lake manipulation. Water Res. 2007, 41, 2551–2562. [Google Scholar] [CrossRef] [PubMed]
- Hobbs, W.O.; Lalonde, S.V.; Vinebrooke, R.D.; Konhauser, K.O.; Weidman, R.P.; Graham, M.D.; Wolfe, A.P. Algal-silica cycling and pigment diagenesis in recent alpine lake sediments: Mechanisms and paleoecological implications. J. Paleolimnol. 2010, 44, 613–628. [Google Scholar] [CrossRef]
- Trachsel, M.; Grosjean, M.; Larocque-tobler, I.; Schwikowski, M.; Blass, A.; Sturm, M. Quantitative summer temperature reconstruction derived from a combined biogenic Si and chironomid record from varved sediments of Lake Silvaplana (south-eastern Swiss Alps) back to AD 1177. Quat. Sci. Rev. 2010, 29, 2719–2730. [Google Scholar] [CrossRef]
- Tallberg, P.; Lehtoranta, J.; Hietanen, S. Silicate Release from Sand-Manipulated Sediment Cores: Biogenic or Adsorbed Si? Silicon 2013, 5, 67–74. [Google Scholar] [CrossRef]
- Aigars, J.; Poik, R.; Dalsgaard, T.; Egl, E.; Jansons, M. Biogeochemistry of N, P and SI in the Gulf of Riga surface sediments: Implications of seasonally changing factors. Cont. Shelf Res. 2015, 105, 112–120. [Google Scholar] [CrossRef]
- Tallberg, P.; Heiskanen, A.S.; Niemistö, J.; Hall, P.O.J.; Lehtoranta, J. Are benthic fluxes important for the availability of Si in the Gulf of Finland? J. Mar. Syst. 2017, 171, 89–100. [Google Scholar] [CrossRef]
- Racchetti, E.; Bartoli, M.; Soana, E.; Longhi, D.; Christian, R.R.; Pinardi, M.; Viaroli, P. Influence of hydrological connectivity of riverine wetlands on nitrogen removal via denitrification. Biogeochemistry 2011, 103, 335–354. [Google Scholar] [CrossRef]
- Soana, E.; Naldi, M.; Bonaglia, S.; Racchetti, E.; Castaldelli, G.; Bruchert, V.; Viaroli, P.; Bartoli, M. Benthic nitrogen metabolism in a macrophyte meadow (Vallisneria spiralis L.) under increasing sedimentary organic matter loads. Biogeochemistry 2015, 124, 387–404. [Google Scholar] [CrossRef]
- Carrick, H.J.; Lowe, R.L. Nutrient limitation of benthic algae in Lake Michigan: The role of silica. J. Phycol. 2007, 43, 228–234. [Google Scholar] [CrossRef]
- Malkin, S.Y.; Sorichetti, R.J.; Wiklund, J.A.; Hecky, R.E. Seasonal abundance, community composition, and silica content of diatoms epiphytic on Cladophora glomerata. J. Gt. Lakes Res. 2009, 35, 199–205. [Google Scholar] [CrossRef]
- Cao, Y.; Zhang, N.; Sun, J.; Li, W. Responses of periphyton on non-plant substrates to different macrophytes under various nitrogen concentrations: A mesocosm study. Aquat. Bot. 2019, 154, 53–59. [Google Scholar] [CrossRef]
- Müller, C. Uptake and accumulation of some nutrient elements in relation to the biomass of an epilithic community. In Periphyton of Freshwater Ecosystems; Springer: Dordrecht, The Netherlands, 1983; Volume 17, pp. 147–151. [Google Scholar]
- Riber, H.H.; Sørensen, J.P.; Kowalczewski, A. Exchange of phosphorus between water, macrophytes and epiphytic periphyton in the littoral of Mikolajskie Lake, Poland. In Periphyton of Freshwater Ecosystems; Springer: Dordrecht, The Netherlands, 1983; Volume 17, pp. 235–243. [Google Scholar]
- Swift, D.; Nicholas, R. Periphyton and Water Quality Relationships in the Everglades Water Conservation Areas, 1978–1982; South Florida Water Management District Tech. Pub.: West Palm Beach, FL, USA, 1987; Volume 87-2, p. 44. [Google Scholar]
- Grimshaw, H.J.; Rosen, M.; Swift, D.R.; Rodberg, K.; Noel, J.M. Marsh phosphorus concentrations, phosphorus content and species composition of Everglades periphyton communities. Arch. Hydrobiol. 1993, 128, 257–276. [Google Scholar]
- Hillebrand, H.; Sommer, U. The nutrient stoichiometry of benthic microalgal growth: Redfield proportions are optimal. Limnol. Oceanogr. 1999, 44, 440–446. [Google Scholar] [CrossRef]
- Pinardi, M.; Bartoli, M.; Longhi, D.; Marzocchi, U.; Laini, A.; Ribaudo, C.; Viaroli, P. Benthic metabolism and denitrification in a river reach: A comparison between vegetated and bare sediments. J. Limnol. 2009, 68, 133–145. [Google Scholar] [CrossRef]
- Ghirardi, N.; Bolpagni, R.; Bresciani, M.; Valerio, G.; Pilotti, M.; Giardino, C. Spatiotemporal Dynamics of Submerged Aquatic Vegetation in a Deep Lake from Sentinel-2 Data. Water 2019, 11, 563. [Google Scholar] [CrossRef]
- Toporowska, M.; Pawlik-Skowrońska, B.; Wojtal, A.Z. Epiphytic algae on Stratiotes aloides L., Potamogeton lucens L., Ceratophyllum demersum L. and Chara spp. in a macrophyte-dominated lake. Oceanol. Hydrobiol. Stud. 2008, 37, 51–63. [Google Scholar] [CrossRef]
- Pettit, N.E.; Ward, D.P.; Adame, M.F.; Valdez, D.; Bunn, S.E. Influence of aquatic plant architecture on epiphyte biomass on a tropical river floodplain. Aquat. Bot. 2016, 129, 35–43. [Google Scholar] [CrossRef]
- Burkholder, J.M.; Wetzel, R.G. Microbial colonization on natural and artificial macrophytes in a phosphorus-limited, hardwater lake. J. Phycol. 1898, 25, 55–65. [Google Scholar] [CrossRef]
- Cattaneo, A.; Galanti, G.; Gentinetta, S.; Romo, S. Epiphytic algae and macroinvertebrates on submerged and floating-leaved macrophytes in an Italian lake. Freshw. Biol. 1998, 39, 725–740. [Google Scholar] [CrossRef]
- Moore, P.A.; Reddy, K.R.; Fisher, M.M. Phosphorus Flux between Sediment and Overlying Water in Lake Okeechobee, Florida: Spatial and Temporal Variations. J. Environ. Qual. 1998, 1439, 1428–1439. [Google Scholar] [CrossRef]
- Reynolds, C.S.; Davies, P.S. Sources and bioavailability of phosphorus fractions in freshwaters: A British perspective. Biol. Rev. 2001, 76, 27–64. [Google Scholar] [CrossRef] [PubMed]
- Francis, T.B.; Schindler, D.E.; Fox, J.M.; Seminet-reneau, E. Effects of Urbanization on the Dynamics of Organic Sediments in Temperate Lakes. Ecosystems 2007, 10, 1057–1068. [Google Scholar] [CrossRef]
- Viaroli, P.; Bartoli, M.; Giordani, G.; Naldi, M.; Orfanidis, S.; Zaldivar, J.M. Community shifts, alternative stable states, biogeochemical controls and feedbacks in eutrophic coastal lagoons: A brief overview. Aquat. Conserv Mar. Freshw. Ecosyst. 2008, 18, 105–117. [Google Scholar] [CrossRef]
- Barone, L.; Pilotti, M.; Valerio, G.; Balistrocchi, M.; Milanesi, L.; Chapra, S.C.; Nizzoli, D. Analysis of the residual nutrient load from a combined sewer system in a watershed of a deep Italian lake. J. Hydrol. 2019, 571, 202–213. [Google Scholar] [CrossRef]
- Donnermeyer, G.N.; Smart, M.M. The biomass and nutritive potential of Vallisneria americana Michx in navigation pool 9 of the upper Mississippi river. Aquat. Bot. 1985, 22, 33–44. [Google Scholar] [CrossRef]
- Viaroli, P.; Bartoli, M.; Bondavalli, C.; Christian, R.R.; Giordani, G.; Naldi, M. Macrophyte communities and their impact on benthic fluxes of oxygen, sulphide and nutrients in shallow eutrophic environments. Hydrobiologia 1996, 329, 105–119. [Google Scholar] [CrossRef]
| Watershed | |
| Area, lake included (km2) | 1842 |
| Mean altitude (m asl) | 1429 |
| Lake | |
| Altitude (m asl) | 185 |
| Volume (km3) | 7.6 |
| Maximum depth (m) | 258 |
| Average depth (m) | 123 |
| Total Area (km2) | 60.9 |
| Littoral Area (km2) | 5.0 |
| Theoretical renewal time (years) | 4.5 |
| Water Characteristics | RS | VS | BS | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Early | Mid | Late | Early | Mid | Late | Early | Mid | Late | |
| DSi (µg Si L−1) | 57 (1) | 130 (5) | 225 (24) | 404 (20) | 400 (39) | 494 (13) | 137 (10) | 238 (10) | 490 (13) |
| SRP (µg P L−1) | 2 (0) | 5 (1) | 2 (0) | 4 (1) | 4 (0) | 2 (0) | 5 (1) | 5 (1) | 2 (0) |
| N-NOx (µg N L−1) | 592 (4) | 509 (3) | 538 (6) | 426 (7) | 376 (2) | 442 (8) | 497 (8) | 420 (6) | 485 (8) |
| N-NH4+ (µg N L−1) | 56 (4) | 85 (3) | 63 (3) | 100 (4) | 94 (6) | 89 (3) | 74 (4) | 55 (3) | 54 (5) |
| DIN (µg N L−1) | 648 (5) | 595 (4) | 601 (6) | 526 (4) | 470 (6) | 531 (9) | 571 (9) | 476 (6) | 539 (10) |
| DSi:DIN (mol:mol) | 0.04 (0.00) | 0.11 (0.00) | 0.19 (0.02) | 0.39 (0.02) | 0.42 (0.04) | 0.47 (0.01) | 0.12 (0.01) | 0.25 (0.01) | 0.46 (0.01) |
| DSi:SRP (mol:mol) | 38 (3) | 31 (4) | 109 (13) | 148 (20) | 155 (26) | 309 (29) | 36 (5) | 65 (5) | 262 (32) |
| DIN:SRP (mol:mol) | 863 (88) | 293 (38) | 596 (42) | 395 (55) | 348 (44) | 664 (57) | 307 (40) | 262 (18) | 564 (56) |
| Sediment Characteristics | RS | VS | BS | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Early | Mid | Late | Early | Mid | Late | Early | Mid | Late | |
| OM (% DW) | n.a. | 7.23 | 8.72 | 4.46 (0.33) | 4.00 (0.27) | 4.30 (0.04) | 2.57 (0.15) | 2.57 (0.05) | 2.78 (0.23) |
| BSi (% DW) | n.a. | 0.78 (0.02) | 1.08 (0.03) | 0.89 (0.04) | 0.72 (0.05) | 0.82 (0.06) | 0.54 (0.01) | 0.46 (0.02) | 0.41 (0.03) |
| P (% DW) | n.a. | 0.10 | 0.12 | 0.01 (0.00) | 0.01 (0.00) | 0.01 (0.00) | 0.01 (0.00) | 0.01 (0.00) | 0.01 (0.00) |
| N (% DW) | n.a. | 0.42 | 0.58 | 0.32 (0.01) | 0.31 (0.00) | 0.35 (0.00) | 0.21 (0.01) | 0.20 (0.01) | 0.21 (0.01) |
| BSi:N (mol:mol) | n.a. | 0.98 | 0.72 | 1.37 (0.01) | 1.16 (0.08) | 1.16 (0.06) | 1.27 (0.03) | 1.19 (0.05) | 0.98 (0.05) |
| BSi:P (mol:mol) | n.a. | 9.07 | 7.49 | 77 (1) | 66 (4) | 67 (3) | 91 (4) | 79 (2) | 56 (6) |
| N:P (mol:mol) | n.a. | 9.23 | 10.35 | 57 (2) | 58 (2) | 58 (2) | 72 (4) | 66 (1) | 57 (3) |
| Chl-a (mg m−2) | n.a. | 21 (3) | 42 (7) | 141 (5) | 190 (23) | 196 (22) | 193 (34) | 220 (15) | 288 (12) |
| Pha (mg m−2) | n.a. | 5 (1) | 7 (1) | 146 (1) | 97 (10) | 136 (12) | 55 (9) | 40 (3) | 79 (4) |
| Primary Producer | Seasonal phase | B | BSi | N | P | BSi:N | BSi:P | N:P | |
|---|---|---|---|---|---|---|---|---|---|
| Filamentous algae | Early | 62 (19) | 3.36 (1.03) | 0.86 (0.26) | 0.07 (0.02) | 0.98 | 62 | 63 | |
| Mid | 48 (21) | 1.41 (0.40) | 0.23 (0.06) | 0.01 (0.00) | 1.54 | 141 | 91 | ||
| Late | 30 (22) | 0.75 (0.20) | 0.19 (0.05) | 0.01 (0.00) | 0.98 | 106 | 108 | ||
| Vallisneria spiralis | Leaves | Early | 99 (36) | 0.72 | 3.64 | 0.22 | 0.10 | 4 | 37 |
| Mid | 206 (16) | 1.00 | 2.35 | 0.13 | 0.21 | 9 | 40 | ||
| Late | 117 (11) | 0.91 | 2.45 | 0.10 | 0.19 | 10 | 54 | ||
| Roots | Early | 60 (22) | 0.48 | 2.92 | 0.16 | 0.08 | 3 | 40 | |
| Mid | 125 (9) | 0.50 | 2.61 | 0.17 | 0.10 | 3 | 34 | ||
| Late | 70 (13) | 0.33 | 3.32 | 0.16 | 0.05 | 2 | 46 | ||
| Najas marina | Leaves | Early | 4 | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. |
| Mid | 97 (27) | 0.64 | 2.82 | 0.27 | 0.11 | 3 | 23 | ||
| Late | 54 (17) | 0.85 | 1.95 | 0.15 | 0.22 | 6 | 29 | ||
| Roots | Early | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | |
| Mid | 15 (4) | 0.61 | 1.83 | 0.12 | 0.17 | 6 | 34 | ||
| Late | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. |
| Source of Variation | DF | O2 | DSi | SRP | N-NOx | N-NH4+ | DIN |
|---|---|---|---|---|---|---|---|
| Site | 2 | <0.001 | <0.001 | <0.050 | <0.001 | <0.001 | <0.001 |
| Condition | 1 | <0.001 | 0.182 | 0.811 | 0.224 | <0.001 | <0.001 |
| Sampling date | 2 | 0.210 | 0.185 | 0.263 | <0.001 | <0.001 | 0.109 |
| Site*Condition | 2 | <0.001 | 0.703 | 0.304 | 0.699 | 0.067 | 0.601 |
| Site* Sampling date | 4 | <0.001 | <0.001 | 0.467 | 0.346 | <0.001 | 0.946 |
| Condition* Sampling date | 2 | <0.001 | 0.108 | 0.587 | 0.488 | <0.001 | <0.010 |
| Site*Condition* Sampling date | 4 | <0.001 | 0.550 | 0.533 | <0.001 | 0.879 | <0.050 |
| Reference | System | Depth (m) | Condition | DSi Flux (mg Si m−2 h−1) | BSi (%) | Method * |
|---|---|---|---|---|---|---|
| [58] | lake | Bare | 2.3–4.6 | 10.2–15.9 | 1 | |
| [28] | lake | 3 | MPB | −1.6 to 11.5 | ||
| [59] | lake | Bare | 2.6–11.8 | 1.0–22.0 | 2 | |
| [60] | river | Bare | 1.0–4.0 | |||
| [23] | coastal lagoon | MPB | light −0.01 ± 0.08 dark 1.40 ± 0.20 | |||
| [61] | coastal lagoon | 2–4 | Bare | 1.2–29.9 | ||
| Uptake | 3.8–28.7 | |||||
| [62] | lake | 5–10 | Bare | 3.1 ± 0.3 | 1.7 | 1 |
| [29] | lake | 3.9 | MPB | light −2.3–21.9 dark −2.0–15.13 | ||
| [63] | lake | 20 | Bare | 0.05 | 0.4–1.5 | 1 |
| [64] | lake | Bare | 0.1–3.4 | 0.2–1.4 | 1 | |
| [65] | coastal | 33 | Algal | 0.28–3.36 | ||
| [66] | coastal | 44–45 | Bare | 1.1–2.9 | ||
| [67] | costal | 2–7 | Bare | 0.7–5.7 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Scibona, A.; Nizzoli, D.; Cristini, D.; Longhi, D.; Bolpagni, R.; Viaroli, P. Silica Storage, Fluxes, and Nutrient Stoichiometry in Different Benthic Primary Producer Communities in the Littoral Zone of a Deep Subalpine Lake (Lake Iseo, Italy). Water 2019, 11, 2140. https://doi.org/10.3390/w11102140
Scibona A, Nizzoli D, Cristini D, Longhi D, Bolpagni R, Viaroli P. Silica Storage, Fluxes, and Nutrient Stoichiometry in Different Benthic Primary Producer Communities in the Littoral Zone of a Deep Subalpine Lake (Lake Iseo, Italy). Water. 2019; 11(10):2140. https://doi.org/10.3390/w11102140
Chicago/Turabian StyleScibona, Alessandro, Daniele Nizzoli, Domiziana Cristini, Daniele Longhi, Rossano Bolpagni, and Pierluigi Viaroli. 2019. "Silica Storage, Fluxes, and Nutrient Stoichiometry in Different Benthic Primary Producer Communities in the Littoral Zone of a Deep Subalpine Lake (Lake Iseo, Italy)" Water 11, no. 10: 2140. https://doi.org/10.3390/w11102140
APA StyleScibona, A., Nizzoli, D., Cristini, D., Longhi, D., Bolpagni, R., & Viaroli, P. (2019). Silica Storage, Fluxes, and Nutrient Stoichiometry in Different Benthic Primary Producer Communities in the Littoral Zone of a Deep Subalpine Lake (Lake Iseo, Italy). Water, 11(10), 2140. https://doi.org/10.3390/w11102140






