Exposure to Submicron Particles and Estimation of the Dose Received by Children in School and Non-School Environments
Abstract
1. Introduction
2. Methodology
2.1. Study Area and Monitoring Site
2.2. Study Design
2.3. Instrumentation and Its Quality Assurance
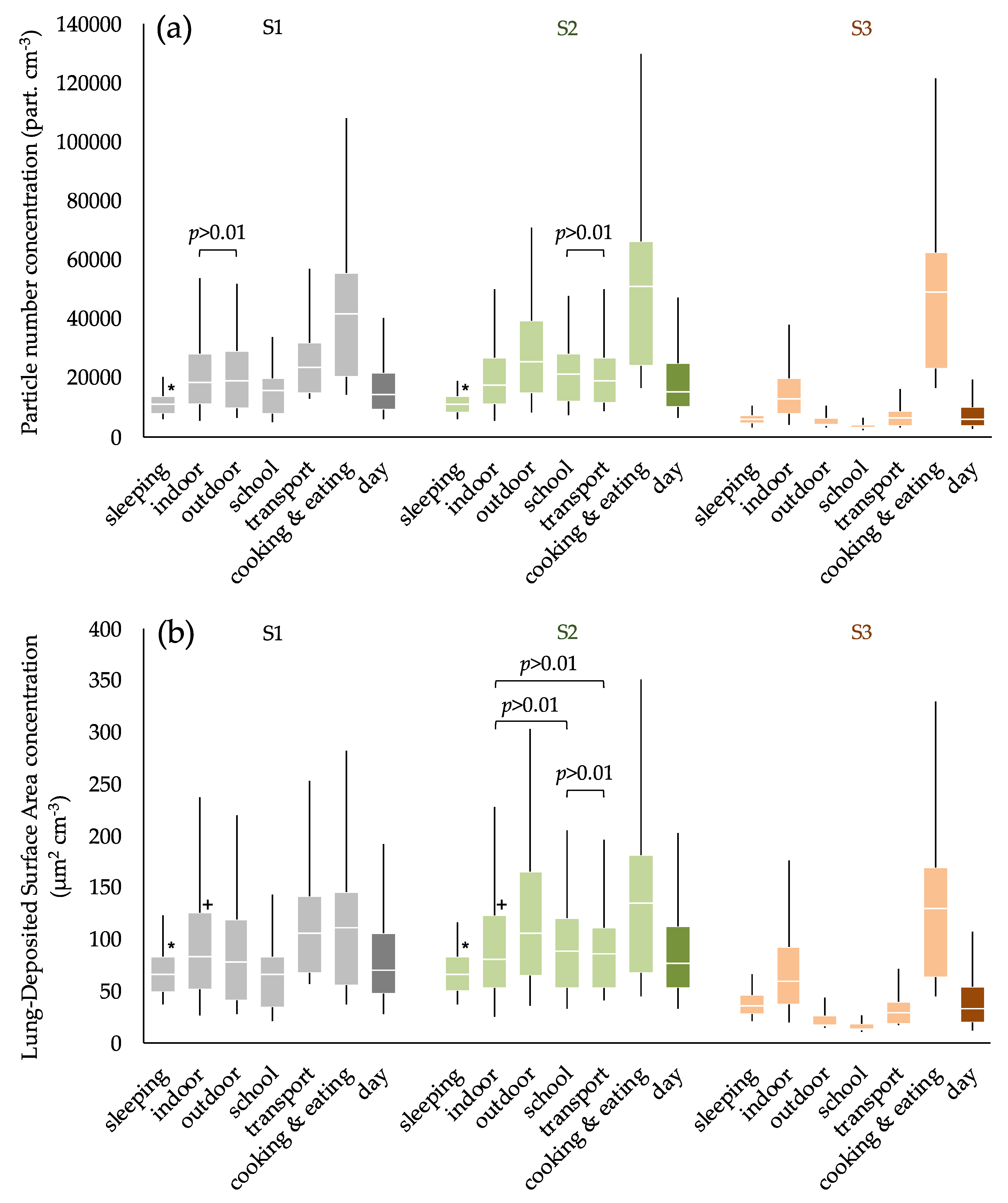
2.4. Statistical Analysis of the Data
3. Results
3.1. Time Activity Patterns
3.2. Exposure to Submicron Particles
3.3. Particle Doses Received by Children
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Schmid, O.; Möller, W.; Semmler-Behnke, M.; Ferron, G.A.; Karg, E.; Lipka, J.; Schulz, H.; Kreyling, W.G.; Stoeger, T. Dosimetry and toxicology of inhaled ultrafine particles. Biomarkers 2009, 14, 67–73. [Google Scholar] [CrossRef]
- Buteau, S.; Goldberg, M.S. A structured review of panel studies used to investigate associations between ambient air pollution and heart rate variability. Environ. Res. 2016, 148, 207–247. [Google Scholar] [CrossRef]
- Rizza, V.; Stabile, L.; Vistocco, D.; Russi, A.; Pardi, S.; Buonanno, G. Effects of the exposure to ultrafine particles on heart rate in a healthy population. Sci. Total Environ. 2019, 650, 2403–2410. [Google Scholar] [CrossRef]
- Brook, R.D.; Jerrett, M.; Brook, J.R.; Bard, R.L.; Finkelstein, M.M. The Relationship Between Diabetes Mellitus and Traffic-Related Air Pollution. J. Occup. Environ. Med. 2008, 50, 32–38. [Google Scholar] [CrossRef]
- Auchincloss, A.H.; Roux, A.V.D.; Dvonch, J.T.; Brown, P.L.; Barr, R.G.; Daviglus, M.L.; Goff, D.C.; Kaufman, J.D.; O’Neill, M.S. Associations between Recent Exposure to Ambient Fine Particulate Matter and Blood Pressure in the Multi-Ethnic Study of Atherosclerosis (MESA). Environ. Health Perspect. 2008, 116, 486–491. [Google Scholar] [CrossRef]
- Power, M.C.; Weisskopf, M.G.; Alexeeff, S.E.; Coull, B.A.; Spiro, A.; Schwartz, J. Traffic-Related Air Pollution and Cognitive Function in a Cohort of Older Men. Environ. Health Perspect. 2010, 119, 682–687. [Google Scholar] [CrossRef]
- World Health Organization. Mortality and Burden of Disease from Ambient Air Pollution: Situation and Trends; WHO: Geneva, Switzerland, 2014. [Google Scholar]
- Beelen, R.; Stafoggia, M.; Raaschou-Nielsen, O.; Andersen, Z.J.; Xun, W.W.; Katsouyanni, K.; Dimakopoulou, K.; Brunekreef, B.; Weinmayr, G.; Hoffmann, B.; et al. Long-term Exposure to Air Pollution and Cardiovascular Mortality. Epidemiology 2014, 25, 368–378. [Google Scholar] [CrossRef] [PubMed]
- International Agency for Research on Cancer. Outdoor Air Pollution a Leading Environmental Cause of Cancer Deaths; IARC: Lyon, Geneva, 2013. [Google Scholar] [CrossRef]
- Loomis, D.; Grosse, Y.; Lauby-Secretan, B.; El Ghissassi, F.; Bouvard, V.; Benbrahim-Tallaa, L.; Guha, N.; Baan, R.; Mattock, H.; Straif, K. The carcinogenicity of outdoor air pollution. Lancet Oncol. 2013, 14, 1262–1263. [Google Scholar] [CrossRef]
- Peters, A.; Von Klot, S.; Heier, M.; Trentinaglia, I.; Hörmann, A.; Wichmann, H.E.; Lowel, H. Exposure to Traffic and the Onset of Myocardial Infarction. N. Engl. J. Med. 2004, 351, 1721–1730. [Google Scholar] [CrossRef] [PubMed]
- Schins, R.P.; Lightbody, J.H.; Borm, P.J.; Shi, T.; Donaldson, K.; Stone, V. Inflammatory effects of coarse and fine particulate matter in relation to chemical and biological constituents. Toxicol. Appl. Pharmacol. 2004, 195, 1–11. [Google Scholar] [CrossRef]
- Weichenthal, S. Selected physiological effects of ultrafine particles in acute cardiovascular morbidity. Environ. Res. 2012, 115, 26–36. [Google Scholar] [CrossRef] [PubMed]
- Unfried, K.; Albrecht, C.; Klotz, L.-O.; Von Mikecz, A.; Grether-Beck, S.; Schins, R.P. Cellular responses to nanoparticles: Target structures and mechanisms. Nanotoxicology 2007, 1, 52–71. [Google Scholar] [CrossRef]
- International Commission on Radiological Protection. Human respiratory tract model for radiological protection. A report of a Task Group of the International Commission on Radiological Protection. Ann. ICRP 1994, 24, 1–482. [Google Scholar]
- Peters, A.; Veronesi, B.; Calderón-Garcidueñas, L.; Gehr, P.; Chen, L.C.; Geiser, M.; Reed, W.; Rothen-Rutishauser, B.; Schürch, S.; Schulz, H. Translocation and potential neurological effects of fine and ultrafine particles a critical update. Part. Fibre Toxicol. 2006, 3, 13. [Google Scholar] [CrossRef] [PubMed]
- Nakane, H. Translocation of particles deposited in the respiratory system: A systematic review and statistical analysis. Environ. Health Prev. Med. 2011, 17, 263–274. [Google Scholar] [CrossRef] [PubMed]
- Campagnolo, L.; Massimiani, M.; Vecchione, L.; Piccirilli, D.; Toschi, N.; Magrini, A.; Bonanno, E.; Scimeca, M.; Castagnozzi, L.; Buonanno, G.; et al. Silver nanoparticles inhaled during pregnancy reach and affect the placenta and the foetus. Nanotoxicology 2017, 11, 687–698. [Google Scholar] [CrossRef] [PubMed]
- Loomis, D. Sizing up air pollution research. Epidemiology 2000, 11, 2. [Google Scholar] [CrossRef]
- Pope, C.A. What do epidemiologic findings tell us about health effects of environmental aerosols? J. Aerosol Med. 2000, 13, 335–354. [Google Scholar] [CrossRef]
- Franck, U.; Odeh, S.; Wiedensohler, A.; Wehner, B.; Herbarth, O. The effect of particle size on cardiovascular disorders—The smaller the worse. Sci. Total Environ. 2011, 409, 4217–4221. [Google Scholar] [CrossRef]
- Kumar, P.; Morawska, L.; Birmili, W.; Paasonen, P.; Hu, M.; Kulmala, M.; Harrison, R.M.; Norford, L.; Britter, R. Ultrafine particles in cities. Environ. Int. 2014, 66, 1–10. [Google Scholar] [CrossRef]
- Buonanno, G.; Marks, G.B.; Morawska, L. Health effects of daily airborne particle dose in children: Direct association between personal dose and respiratory health effects. Environ. Pollut. 2013, 180, 246–250. [Google Scholar] [CrossRef] [PubMed]
- Giechaskiel, B.; Alföldy, B.; Drossinos, Y. A metric for health effects studies of diesel exhaust particles. J. Aerosol Sci. 2009, 40, 639–651. [Google Scholar] [CrossRef]
- Brown, D.; Wilson, M.; MacNee, W.; Stone, V.; Donaldson, K. Size-Dependent Proinflammatory Effects of Ultrafine Polystyrene Particles: A Role for Surface Area and Oxidative Stress in the Enhanced Activity of Ultrafines. Toxicol. Appl. Pharmacol. 2001, 175, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Hamoir, J.; Nemmar, A.; Halloy, D.; Wirth, D.; Vincke, G.; Vanderplasschen, A.; Nemery, B.; Gustin, P. Effect of polystyrene particles on lung microvascular permeability in isolated perfused rabbit lungs: Role of size and surface properties. Toxicol. Appl. Pharmacol. 2003, 190, 278–285. [Google Scholar] [CrossRef]
- Strak, M.; Boogaard, H.; Meliefste, K.; Oldenwening, M.; Zuurbier, M.; Brunekreef, B.; Hoek, G. Respiratory health effects of ultrafine and fine particle exposure in cyclists. Occup. Environ. Med. 2009, 67, 118–124. [Google Scholar] [CrossRef] [PubMed]
- Landkocz, Y.; LeDoux, F.; André, V.; Cazier, F.; Genevray, P.; Dewaele, D.; Martin, P.J.; Lepers, C.; Verdin, A.; Courcot, L.; et al. Fine and ultrafine atmospheric particulate matter at a multi-influenced urban site: Physicochemical characterization, mutagenicity and cytotoxicity. Environ. Pollut. 2017, 221, 130–140. [Google Scholar] [CrossRef] [PubMed]
- Longhin, E.; Gualtieri, M.; Capasso, L.; Bengalli, R.; Mollerup, S.; Holme, J.A.; Øvrevik, J.; Casadei, S.; Di Benedetto, C.; Parenti, P.; et al. Physico-chemical properties and biological effects of diesel and biomass particles. Environ. Pollut. 2016, 215, 366–375. [Google Scholar] [CrossRef] [PubMed]
- Sager, T.; Castranova, V. Surface area of particle administered versus mass in determining the pulmonary toxicity of ultrafine and fine carbon black: Comparison to ultrafine titanium dioxide. Part. Fibre Toxicol. 2009, 6, 15. [Google Scholar] [CrossRef]
- Schmid, O.; Stöger, T. Surface area is the biologically most effective dose metric for acute nanoparticle toxicity in the lung. J. Aerosol Sci. 2016, 99, 133–143. [Google Scholar] [CrossRef]
- Nygaard, U.C.; Samuelsen, M.; Aase, A.; Løvik, M. The Capacity of Particles to Increase Allergic Sensitization Is Predicted by Particle Number and Surface Area, Not by Particle Mass. Toxicol. Sci. 2004, 82, 515–524. [Google Scholar] [CrossRef]
- Cauda, E.G.; Ku, B.K.; Miller, A.L.; Barone, T.L. Toward Developing a New Occupational Exposure Metric Approach for Characterization of Diesel Aerosols. Aerosol Sci. Technol. 2012, 46, 1370–1381. [Google Scholar] [CrossRef] [PubMed]
- Oberdörster, G.; Oberdörster, E.; Oberdörster, J. Nanotoxicology: An Emerging Discipline Evolving from Studies of Ultrafine Particles. Environ. Health Perspect. 2005, 113, 823–839. [Google Scholar] [CrossRef]
- Tran, C.L.; Buchanan, D.; Cullen, R.T.; Searl, A.; Jones, A.D.; Donaldson, K. Inhalation of poorly soluble particles. II. Influence Of particle surface area on inflammation and clearance. Inhal. Toxicol. 2000, 12, 1113–1126. [Google Scholar] [CrossRef] [PubMed]
- Buonanno, G.; Stabile, L.; Morawska, L.; Giovinco, G.; Querol, X. Do air quality targets really represent safe limits for lung cancer risk? Sci. Total Environ. 2017, 580, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Sze-To, G.N.; Wu, C.L.; Chao, C.Y.H.; Wan, M.P.; Chan, T.C. Exposure and cancer risk toward cooking-generated ultrafine and coarse particles in Hong Kong homes. HVAC&R Res. 2012, 18, 204–216. [Google Scholar] [CrossRef]
- Stabile, L.; Buonanno, G.; Ficco, G.; Scungio, M. Smokers’ lung cancer risk related to the cigarette-generated mainstream particles. J. Aerosol Sci. 2017, 107, 41–54. [Google Scholar] [CrossRef]
- Pacitto, A.; Stabile, L.; Viana, M.; Scungio, M.; Reche, C.; Querol, X.; Alastuey, A.; Rivas, I.; Álvarez-Pedrerol, M.; Sunyer, J.; et al. Particle-related exposure, dose and lung cancer risk of primary school children in two European countries. Sci. Total Environ. 2018, 616–617, 720–729. [Google Scholar] [CrossRef]
- European Parliament and Council of the European Union. EU Directive 2008/50/EC of the European Parliament and of the Council of 21 May 2008 on ambient air quality and cleaner air for Europe, 2008. Off. J. Eur. Union 2008, L 152/1, 1–44.
- Buonanno, G.; Dell’Isola, M.; Stabile, L.; Viola, A. Uncertainty Budget of the SMPS–APS System in the Measurement of PM1, PM2.5, and PM10. Aerosol Sci. Technol. 2009, 43, 1130–1141. [Google Scholar] [CrossRef]
- Buonanno, G.; Dell’Isola, M.; Stabile, L.; Viola, A. Critical aspects of the uncertainty budget in the gravimetric PM measurements. Measurement 2011, 44, 139–147. [Google Scholar] [CrossRef]
- Moreno, T.; Pacitto, A.; Fernández, A.; Amato, F.; Marco, E.; Grimalt, J.; Buonanno, G.; Querol, X. Vehicle interior air quality conditions when travelling by taxi. Environ. Res. 2019, 172, 529–542. [Google Scholar] [CrossRef] [PubMed]
- Kaur, S.; Nieuwenhuijsen, M.; Colvile, R. Pedestrian exposure to air pollution along a major road in Central London, UK. Atmos. Environ. 2005, 39, 7307–7320. [Google Scholar] [CrossRef]
- Stabile, L.; Arpino, F.; Buonanno, G.; Russi, A.; Frattolillo, A. A simplified benchmark of ultrafine particle dispersion in idealized urban street canyons: A wind tunnel study. Build. Environ. 2015, 93, 186–198. [Google Scholar] [CrossRef]
- Manigrasso, M.; Stabile, L.; Avino, P.; Buonanno, G. Influence of measurement frequency on the evaluation of short-term dose of sub-micrometric particles during indoor and outdoor generation events. Atmos. Environ. 2013, 67, 130–142. [Google Scholar] [CrossRef]
- Scungio, M.; Arpino, F.; Cortellessa, G.; Buonanno, G. Detached eddy simulation of turbulent flow in isolated street canyons of different aspect ratios. Atmos. Pollut. Res. 2015, 6, 351–364. [Google Scholar] [CrossRef]
- Kumar, P.; Ketzel, M.; Vardoulakis, S.; Pirjola, L.; Britter, R. Dynamics and dispersion modelling of nanoparticles from road traffic in the urban atmospheric environment—A review. J. Aerosol Sci. 2011, 42, 580–603. [Google Scholar] [CrossRef]
- Scungio, M.; Buonanno, G.; Arpino, F.; Ficco, G. Influential parameters on ultrafine particle concentration downwind at waste-to-energy plants. Waste Manag. 2015, 38, 157–163. [Google Scholar] [CrossRef]
- Neft, I.; Scungio, M.; Culver, N.; Singh, S. Simulations of Aerosol Filtration by Vegetation: Validation of Existing Models with Available Lab data and Application to Near-Roadway Scenario. Aerosol Sci. Technol. 2016, 50, 937–946. [Google Scholar] [CrossRef]
- Buonanno, G.; Fuoco, F.C.; Stabile, L. Influential parameters on particle exposure of pedestrians in urban microenvironments. Atmos. Environ. 2011, 45, 1434–1443. [Google Scholar] [CrossRef]
- Rizza, V.; Stabile, L.; Buonanno, G.; Morawska, L. Variability of airborne particle metrics in an urban area. Environ. Pollut. 2017, 220, 625–635. [Google Scholar] [CrossRef]
- Pacitto, A.; Stabile, L.; Moreno, T.; Kumar, P.; Wierzbicka, A.; Morawska, L.; Buonanno, G. The influence of lifestyle on airborne particle surface area doses received by different Western populations. Environ. Pollut. 2018, 232, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Buonanno, G.; Stabile, L.; Morawska, L. Personal exposure to ultrafine particles: The influence of time-activity patterns. Sci. Total Environ. 2014, 468, 903–907. [Google Scholar] [CrossRef] [PubMed]
- Buonanno, G.; Marini, S.; Morawska, L.; Fuoco, F.C. Individual dose and exposure of Italian children to ultrafine particles. Sci. Total Environ. 2012, 438, 271–277. [Google Scholar] [CrossRef] [PubMed]
- Brent, R.L.; Weitzman, M. The Vulnerability, Sensitivity, and Resiliency of the Developing Embryo, Infant, Child, and Adolescent to the Effects of Environmental Chemicals, Drugs, and Physical Agents as Compared to the Adult: Preface. Pediatrics 2004, 113, 933–934. [Google Scholar]
- Makri, A.; Stilianakis, N.I. Vulnerability to air pollution health effects. Int. J. Hyg. Environ. Health 2008, 211, 326–336. [Google Scholar] [CrossRef] [PubMed]
- Ginsberg, G.; Foos, B.P.; Firestone, M.P. Review and Analysis of Inhalation Dosimetry Methods for Application to Children’s Risk Assessment. J. Toxicol. Environ. Health Part A 2005, 68, 573–615. [Google Scholar] [CrossRef]
- Heinrich, J.; Slama, R. Fine particles, a major threat to children. Int. J. Hyg. Environ. Health 2007, 210, 617–622. [Google Scholar] [CrossRef]
- Bateson, T.F.; Schwartz, J. Children’s Response to Air Pollutants. J. Toxicol. Environ. Health Part A Curr. Issues 2007, 71, 238–243. [Google Scholar] [CrossRef]
- Selgrade, M.K.; Plopper, C.G.; Gilmour, M.I.; Conolly, R.B.; Foos, B.S.P. Assessing The Health Effects and Risks Associated with Children’s Inhalation Exposures—Asthma and Allergy. J. Toxicol. Environ. Health Part A 2007, 71, 196–207. [Google Scholar] [CrossRef]
- Buonanno, G.; Giovinco, G.; Morawska, L.; Stabile, L. Tracheobronchial and alveolar dose of submicrometer particles for different population age groups in Italy. Atmos. Environ. 2011, 45, 6216–6224. [Google Scholar] [CrossRef]
- Buonanno, G.; Morawska, L.; Stabile, L.; Wang, L.; Giovinco, G. A comparison of submicrometer particle dose between Australian and Italian people. Environ. Pollut. 2012, 169, 183–189. [Google Scholar] [CrossRef] [PubMed]
- The Organisation for Economic Co-Operation and Development. How Long Do Students Spend in the Classroom? OECD: Paris, France, 2012. [Google Scholar]
- Stabile, L.; Dell’Isola, M.; Russi, A.; Massimo, A.; Buonanno, G. The effect of natural ventilation strategy on indoor air quality in schools. Sci. Total Environ. 2017, 595, 894–902. [Google Scholar] [CrossRef] [PubMed]
- Stephens, B.; Siegel, J.A. Penetration of ambient submicron particles into single-family residences and associations with building characteristics. Indoor Air 2012, 22, 501–513. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Hinds, W.C.; Krudysz, M.; Kuhn, T.; Froines, J.; Sioutas, C. Penetration of freeway ultrafine particles into indoor environments. J. Aerosol Sci. 2005, 36, 303–322. [Google Scholar] [CrossRef]
- Viana, M.; Díez, S.; Reche, C. Indoor and outdoor sources and infiltration processes of PM1 and black carbon in an urban environment. Atmos. Environ. 2011, 45, 6359–6367. [Google Scholar] [CrossRef]
- Tippayawong, N.; Khuntong, P.; Nitatwichit, C.; Khunatorn, Y.; Tantakitti, C. Indoor/outdoor relationships of size-resolved particle concentrations in naturally ventilated school environments. Build. Environ. 2009, 44, 188–197. [Google Scholar] [CrossRef]
- Health Effects Institute. HEI Perspectives 3. In Understanding the Health Effects of Ambient Ultrafine Particles; Health Effects Institute: Boston, MA, USA, 2013. [Google Scholar]
- Pacitto, A.; Amato, F.; Moreno, T.; Pandolfi, M.; Fonseca, A.; Mazaheri, M.; Stabile, L.; Buonanno, G.; Querol, X. Effect of ventilation strategies and air purifiers on the children’s exposure to airborne particles and gaseous pollutants in school gyms. Sci. Total Environ. 2020, 712, 135673. [Google Scholar] [CrossRef]
- Salimi, F.; Mazaheri, M.; Clifford, S.; Crilley, L.R.; Laiman, R.; Morawska, L.; Clifford, S.J. Spatial Variation of Particle Number Concentration in School Microscale Environments and Its Impact on Exposure Assessment. Environ. Sci. Technol. 2013, 47, 5251–5258. [Google Scholar] [CrossRef]
- Mazaheri, M.; Reche, C.; Rivas, I.; Crilley, L.R.; Alvarez-Pedrerol, M.; Viana, M.; Tobias, A.; Alastuey, A.; Sunyer, J.; Querol, X.; et al. Variability in exposure to ambient ultrafine particles in urban schools: Comparative assessment between Australia and Spain. Environ. Int. 2016, 88, 142–149. [Google Scholar] [CrossRef]
- Rivas, I.; Viana, M.; Moreno, T.; Bouso, L.; Pandolfi, M.; Alvarez-Pedrerol, M.; Forns, J.; Alastuey, A.; Sunyer, J.; Querol, X. Outdoor infiltration and indoor contribution of UFP and BC, OC, secondary inorganic ions and metals in PM2.5 in schools. Atmos. Environ. 2015, 106, 129–138. [Google Scholar] [CrossRef]
- Stabile, L.; Massimo, A.; Rizza, V.; D’Apuzzo, M.; Evangelisti, A.; Scungio, M.; Frattolillo, A.; Cortellessa, G.; Buonanno, G. A novel approach to evaluate the lung cancer risk of airborne particles emitted in a city. Sci. Total Environ. 2019, 656, 1032–1042. [Google Scholar] [CrossRef] [PubMed]
- Buonanno, G.; Johnson, G.; Morawska, L.; Stabile, L. Volatility Characterization of Cooking-Generated Aerosol Particles. Aerosol Sci. Technol. 2011, 45, 1069–1077. [Google Scholar] [CrossRef]
- Buonanno, G.; Morawska, L.; Stabile, L. Particle emission factors during cooking activities. Atmos. Environ. 2009, 43, 3235–3242. [Google Scholar] [CrossRef]
- See, S.W.; Balasubramanian, R. Chemical characteristics of fine particles emitted from different gas cooking methods. Atmos. Environ. 2008, 42, 8852–8862. [Google Scholar] [CrossRef]
- Wallace, L.A.; Ott, W.R.; Weschler, C.J. Ultrafine particles from electric appliances and cooking pans: Experiments suggesting desorption/nucleation of sorbed organics as the primary source. Indoor Air 2014, 25, 536–546. [Google Scholar] [CrossRef] [PubMed]
- Rivas, I.; Kumar, P.; Hagen-Zanker, A. Exposure to air pollutants during commuting in London: Are there inequalities among different socio-economic groups? Environ. Int. 2017, 101, 143–157. [Google Scholar] [CrossRef]
- Moreno, T.; Reche, C.; Rivas, I.; Minguillon, M.C.; Martins, V.; Vargas, C.; Buonanno, G.; Parga, J.; Pandolfi, M.; Brines, M.; et al. Urban air quality comparison for bus, tram, subway and pedestrian commutes in Barcelona. Environ. Res. 2015, 142, 495–510. [Google Scholar] [CrossRef]
- Fierz, M.; Houle, C.; Steigmeier, P.; Burtscher, H. Design, Calibration, and Field Performance of a Miniature Diffusion Size Classifier. Aerosol Sci. Technol. 2011, 45, 1–10. [Google Scholar] [CrossRef]
- Fernández, A.; Amato, F.; Moreno, N.; Pacitto, A.; Reche, C.; Marco, E.; Grimalt, J.O.; Querol, X.; Moreno, T. Chemistry and sources of PM2.5 and volatile organic compounds breathed inside urban commuting and tourist buses. Atmos. Environ. 2020, 223, 117234. [Google Scholar] [CrossRef]
- Stabile, L.; Trassierra, C.V.; Dell’Agli, G.; Buonanno, G. Ultrafine Particle Generation through Atomization Technique: The Influence of the Solution. Aerosol Air Qual. Res. 2013, 13, 1667–1677. [Google Scholar] [CrossRef]
- Goel, A.; Kumar, P. A review of fundamental drivers governing the emissions, dispersion and exposure to vehicle-emitted nanoparticles at signalised traffic intersections. Atmos. Environ. 2014, 97, 316–331. [Google Scholar] [CrossRef]
- Kumar, P.; Pirjola, L.; Ketzel, M.; Harrison, R.M. Nanoparticle emissions from 11 non-vehicle exhaust sources—A review. Atmos. Environ. 2013, 67, 252–277. [Google Scholar] [CrossRef]
- Marra, J.; Voetz, M.; Kiesling, H.-J. Monitor for detecting and assessing exposure to airborne nanoparticles. J. Nanopart. Res. 2009, 12, 21–37. [Google Scholar] [CrossRef]
- Kruskal, W.H.; Wallis, W.A. Errata: Use of Ranks in One-Criterion Variance Analysis. J. Am. Stat. Assoc. 1953, 48, 907. [Google Scholar] [CrossRef]
- Buonanno, G.; Giovinco, G.; Morawska, L.; Stabile, L. Lung cancer risk of airborne particles for Italian population. Environ. Res. 2015, 142, 443–451. [Google Scholar] [CrossRef]
- Stabile, L.; Buonanno, G.; Frattolillo, A.; Dell’Isola, M. The effect of the ventilation retrofit in a school on CO2, airborne particles, and energy consumptions. Build. Environ. 2019, 156, 1–11. [Google Scholar] [CrossRef]
- Buonanno, G.; Fuoco, F.; Morawska, L.; Stabile, L. Airborne particle concentrations at schools measured at different spatial scales. Atmos. Environ. 2013, 67, 38–45. [Google Scholar] [CrossRef]
- Stafoggia, M.; Schneider, A.; Cyrys, J.; Samoli, E.; Andersen, Z.J.; Bedada, G.B.; Bellander, T.; Cattani, G.; Eleftheriadis, K.; Faustini, A.; et al. Association Between Short-term Exposure to Ultrafine Particles and Mortality in Eight European Urban Areas. Epidemiology 2017, 28, 172–180. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Zhang, L.-W.; Huang, J.-J.; Song, F.; Zhang, L.-P.; Qian, Z.-M.; Trevathan, E.; Mao, H.-J.; Han, B.; Vaughn, M.; et al. Long-term exposure to urban air pollution and lung cancer mortality: A 12-year cohort study in Northern China. Sci. Total Environ. 2016, 571, 855–861. [Google Scholar] [CrossRef] [PubMed]
| School | City | Sampling Period | T (°C) | RH (%) | NO2 (μg·m−3) | PM10 (μg·m−3) | PM2.5 (μg·m−3) | Distance of the Closest Fsp to the School and Definition According to the Standard |
|---|---|---|---|---|---|---|---|---|
| S1 | Salerno | November–December 2018 | 15 (13–17) | 71 (n.a.) | 34 (8–66) | 21 (8–56) | 13 (5–44) | Distance: <100 m type of site: suburban type of station: background |
| S2 | Roma | February–March 2019 | 11 (6–16) | 66 (n.a.) | 42 (26–122) | 29 (7–63) | 16 (12–18) | Distance: 1 km type of site: urban type of station: traffic |
| S3 | Parma | April–May 2019 | 15 (10–18) | 71 (n.a.) | 12 (10–42) | 12 (<5–27) | 7.5 (<5–21) | Distance: 12 km type of site: rural type of station: background |
| Microenvironment | Activities |
|---|---|
| Transportation | Trip and use of time not specified, round-trip to work |
| School | All type of activities performed in school environments |
| Cooking & eating | Cooking, eating and drinking |
| Outdoor day | Gardening and animal care, restoration, sport and outdoor activities, physical workout, Productive exercise, Sports-connected activities |
| Indoor day | Personal care, studying not specified, studying in the free time, activities for home and family not specified, housework, purchasing goods and services, helping adult family members, helping other family members, active activities, social activities and entertainment, social life, entertainment and culture, inactivity, hobbies and computer science, art and hobbies, computing, playing, media, reading, watching TV, DVD or videos, listening to the radio or recording |
| Sleeping | Sleeping |
| Microenvironment | School | Time (min) | Time Fraction | PN conc. (104 part. cm3) | LDSA conc. (µm2·cm−3) | δ (mm2) | Daily Dose Fraction | i_δ (mm2·min−1) |
|---|---|---|---|---|---|---|---|---|
| Sleeping | S1 | 540 (480–590) | 38% | 1.11 (0.62–2.04) | 66 (37–123) | 182 (101–340) | 17% | 0.34 |
| S2 | 530 (485–560) | 37% | 1.12 (0.61–1.93) | 66 (37–117) | 180 (100–319) | 15% | 0.34 | |
| S3 | 597 (540–620) | 41% | 0.62 (0.33–1.08) | 36 (21–67) | 111 (63–204) | 17% | 0.19 | |
| Indoor | S1 | 320 (135–365) | 22% | 1.85 (0.59–7.70) | 84 (27–356) | 407 (128–1714) | 38% | 1.27 |
| S2 | 345 (140–395) | 24% | 1.79 (0.58–7.36) | 81 (26–342) | 426 (136–1777) | 36% | 1.23 | |
| S3 | 268 (110–320) | 19% | 1.32 (0.43–5.26) | 60 (20–248) | 245 (80–1002) | 38% | 0.91 | |
| Outdoor | S1 | 40 (18–68) | 3% | 1.91 (0.68–5.20) | 79 (28–220) | 48 (17–133) | 5% | 1.20 |
| S2 | 35 (13–58) | 2% | 2.58 (0.86–7.12) | 106 (36–304) | 57 (119–162) | 5% | 1.62 | |
| S3 | 36 (15–60) | 3% | 0.42 (0.36–1.18) | 17 (15–49) | 10 (8–27) | 2% | 00.27 | |
| School | S1 | 360 (295–375) | 25% | 1.57 (0.54–3.38) | 66 (22–144) | 163 (56–362) | 15% | 0.45 |
| S2 | 360 (290–385) | 25% | 2.13 (0.76–4.77) | 89 (33–205) | 222 (81–515) | 19% | 0.62 | |
| S3 | 360 (300–390) | 25% | 0.34 (0.28–0.76) | 14 (12–33) | 36 (28–82) | 6% | 0.10 | |
| Transport | S1 | 60 (16–138) | 4% | 2.38 (1.29–6.38) | 106 (57–274) | 62 (33–160) | 6% | 1.04 |
| S2 | 50 (12–106) | 3% | 1.93(0.91–5.55) | 86 (41–254) | 41 (20–122) | 4% | 0.82 | |
| S3 | 62 (15–131) | 4% | 0.66 (0.36–1.84) | 29 (17–84) | 17 (10–50) | 3% | 0.28 | |
| Cooking & Eating | S1 | 120 (97–148) | 8% | 4.20 (1.44–15.3) | 112 (38–412) | 200 (69–744) | 19% | 1.66 |
| S2 | 120 (93–156) | 8% | 5.11 (1.69–16.6) | 136 (46–453) | 244 (83–823) | 21% | 2.03 | |
| S3 | 117 (90–160) | 8% | 4.91 (1.67–19.3) | 130 (45–525) | 227 (80–923) | 35% | 1.94 | |
| Day | S1 | 1440 | 1.44 (0.61–5.99) | 71 (28–248) | 1062 | 0.74 | ||
| S2 | 1440 | 1.55 (0.65–6.33) | 77 (34–261) | 1169 | 0.81 | |||
| S3 | 1440 | 0.62 (0.30–5.07) | 34 (12–169) | 646 | 0.45 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pacitto, A.; Stabile, L.; Russo, S.; Buonanno, G. Exposure to Submicron Particles and Estimation of the Dose Received by Children in School and Non-School Environments. Atmosphere 2020, 11, 485. https://doi.org/10.3390/atmos11050485
Pacitto A, Stabile L, Russo S, Buonanno G. Exposure to Submicron Particles and Estimation of the Dose Received by Children in School and Non-School Environments. Atmosphere. 2020; 11(5):485. https://doi.org/10.3390/atmos11050485
Chicago/Turabian StylePacitto, Antonio, Luca Stabile, Stefania Russo, and Giorgio Buonanno. 2020. "Exposure to Submicron Particles and Estimation of the Dose Received by Children in School and Non-School Environments" Atmosphere 11, no. 5: 485. https://doi.org/10.3390/atmos11050485
APA StylePacitto, A., Stabile, L., Russo, S., & Buonanno, G. (2020). Exposure to Submicron Particles and Estimation of the Dose Received by Children in School and Non-School Environments. Atmosphere, 11(5), 485. https://doi.org/10.3390/atmos11050485







