A Spatial and Temporal Risk Assessment of the Impacts of El Niño on the Tropical Forest Carbon Cycle: Theoretical Framework, Scenarios, and Implications
Abstract
1. Introduction
2. Methods
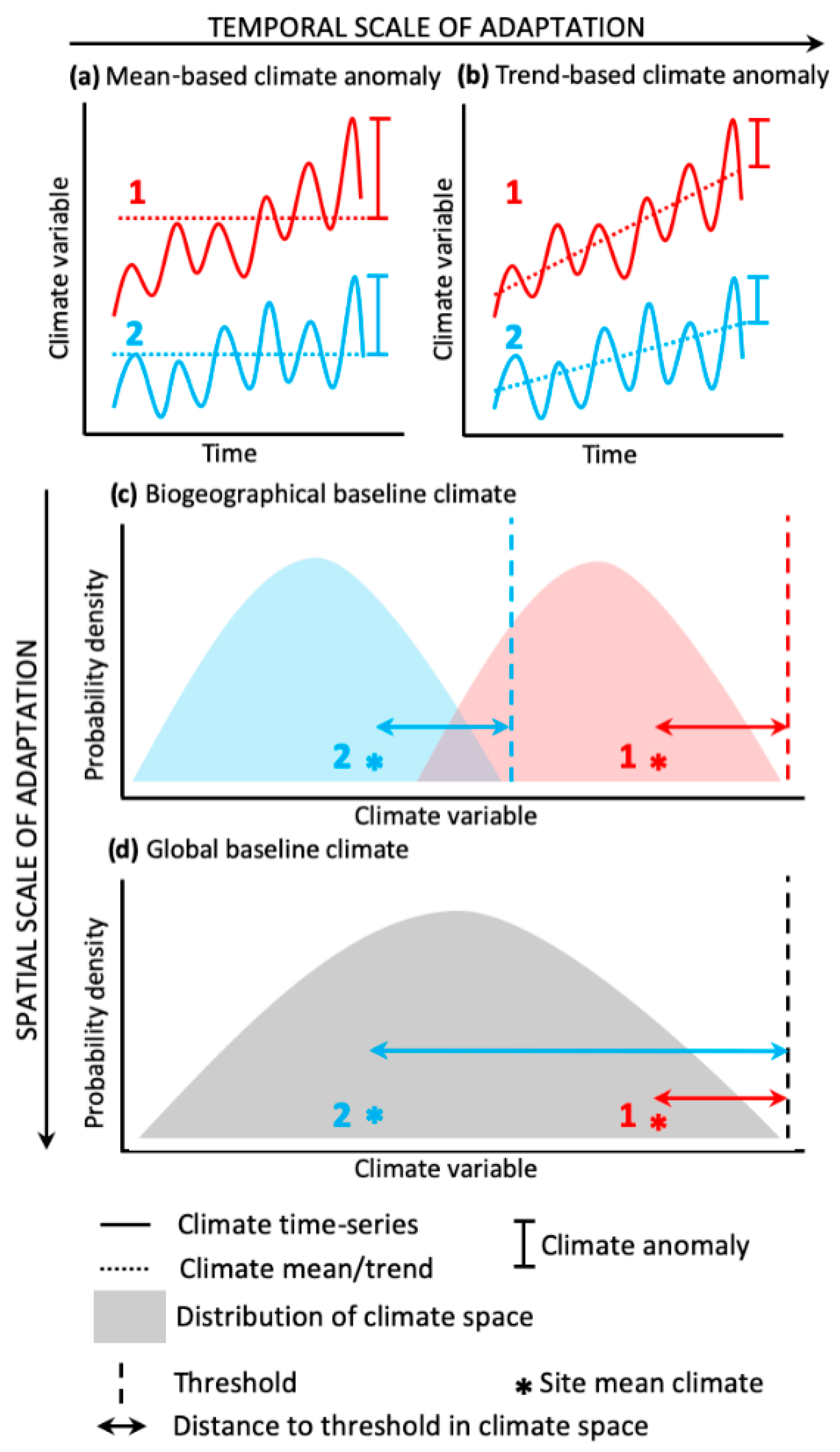
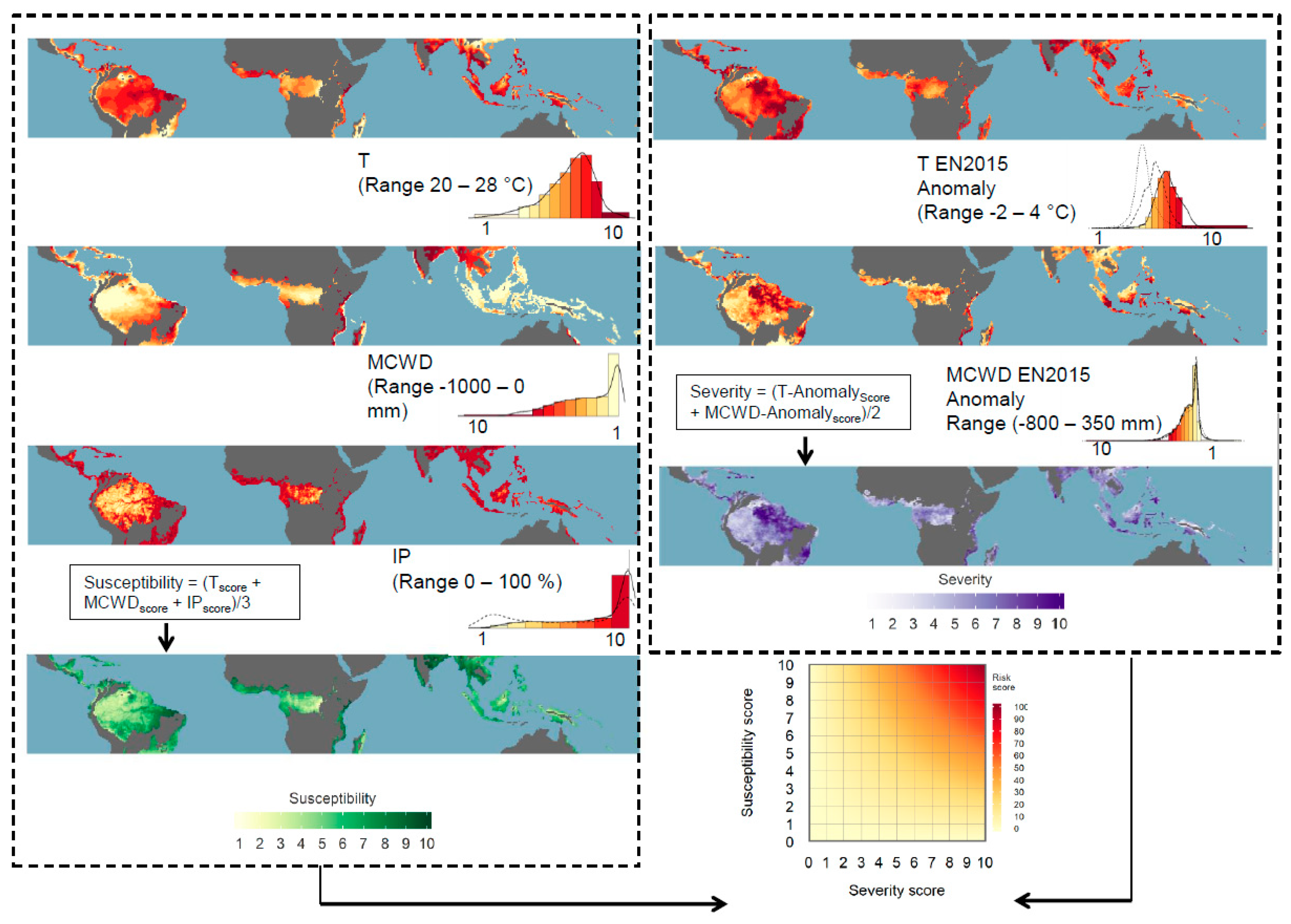
2.1. Susceptibility Scores
2.2. Severity Scores
2.3. Comparing El Niño Risk Scores
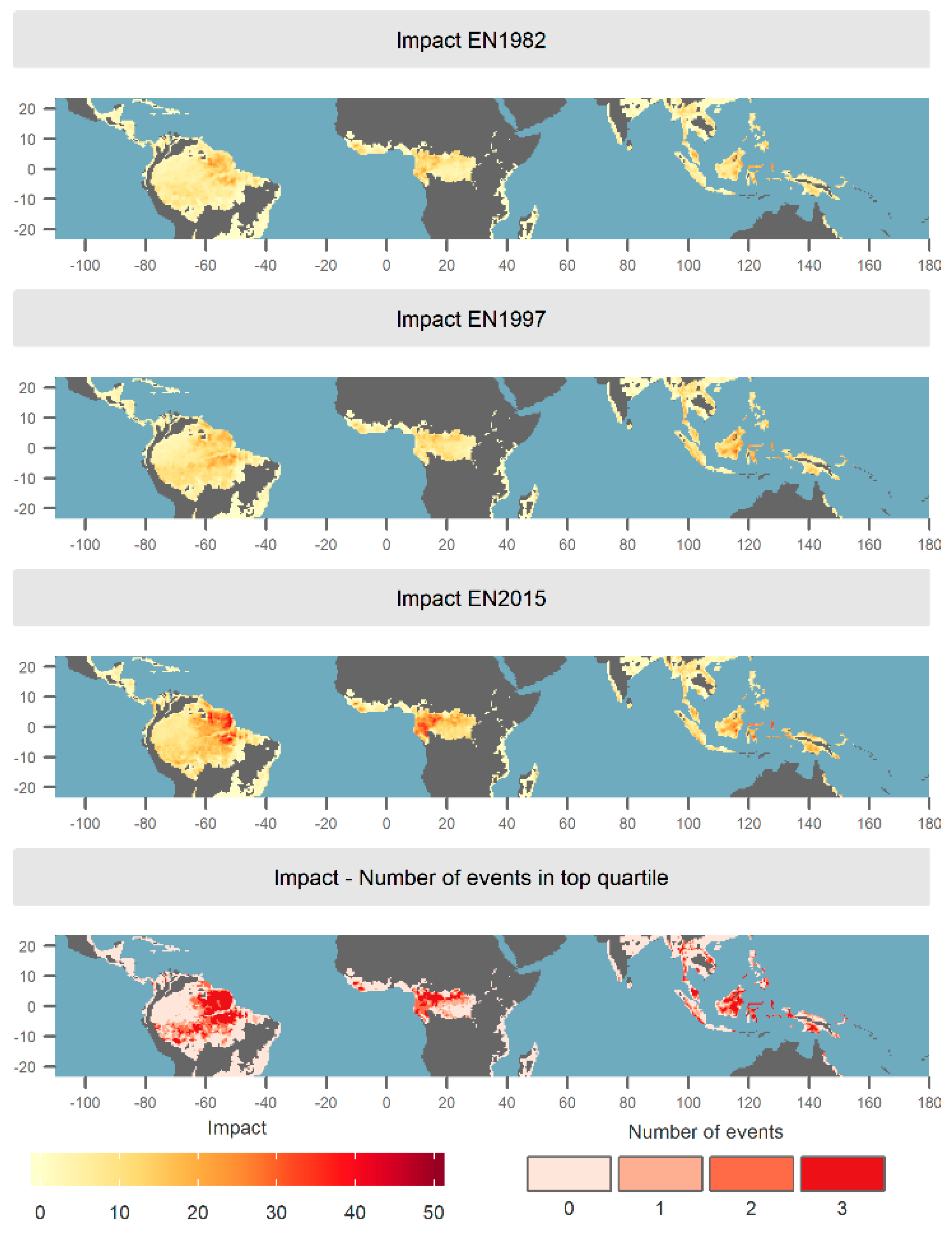
2.4. Estimating El Niño Impact
3. Results
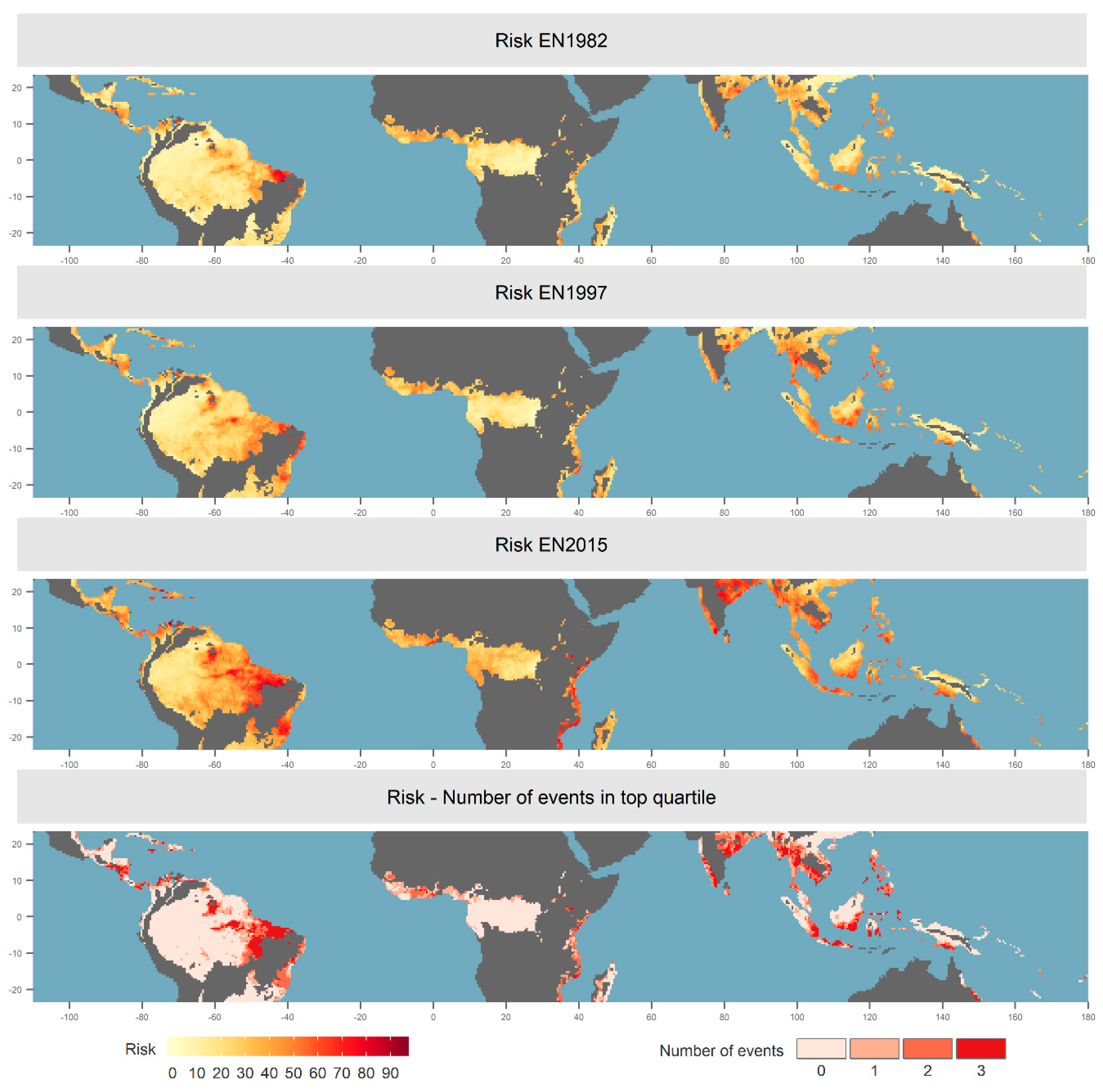
3.1. Spatial and Temporal Distribution of Risk and Impact
3.2. Drivers of Risk
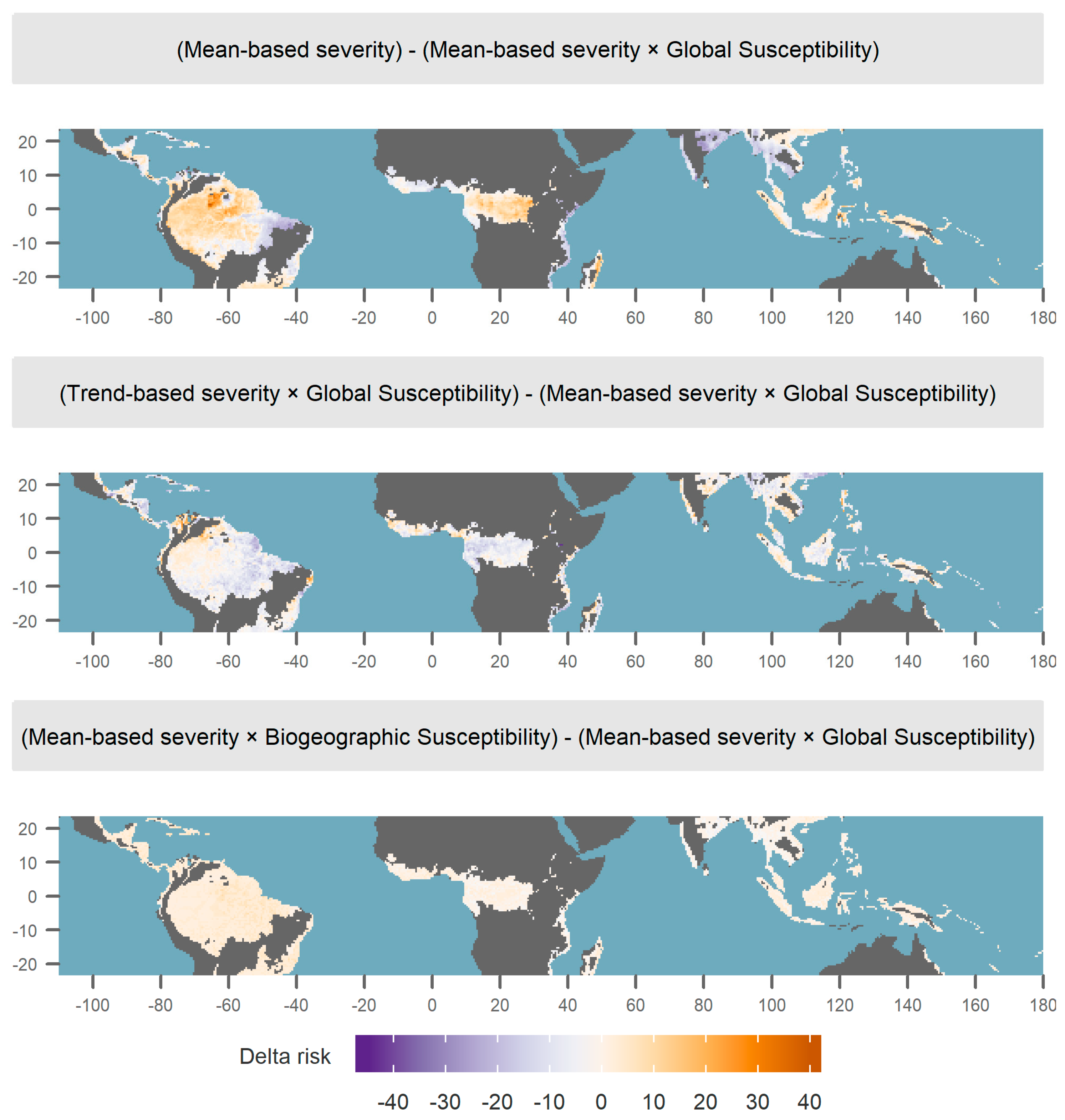
3.3. Alternative Definitions of Risk
4. Discussion
Consequences of El Niño and Implications for Conservation
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Malhi, Y.; Wright, J. Spatial Patterns and Recent Trends in the Climate of Tropical Rainforest Regions. Philos. Trans. R. Soc. Lond. Ser. B Biol. Sci. 2004, 359, 311–329. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Bowman, K.W.; Schimel, D.S.; Parazoo, N.C.; Jiang, Z.; Lee, M.; Bloom, A.A.; Wunch, D.; Frankenberg, C.; Sun, Y.; et al. Contrasting Carbon Cycle Responses of the Tropical Continents to the 2015–2016 El Niño. Science 2017, 358, eaam5690. [Google Scholar] [CrossRef] [PubMed]
- Lloyd, J.; Farquhar, G.D. Effects of Rising Temperatures and Co2 on the Physiology of Tropical Forest Trees. Philos. Trans. R. Soc. Lond. Ser. B Biol. Sci. 2008, 363, 1811–1817. [Google Scholar] [CrossRef] [PubMed]
- Heskel, M.A.; O’Sullivan, O.S.; Reich, P.B.; Tjoelker, M.G.; Weerasinghe, L.K.; Penillard, A.; Egerton, J.J.; Creek, D.; Bloomfield, K.J.; Xiang, J.; et al. Convergence in the Temperature Response of Leaf Respiration across Biomes and Plant Functional Types. Proc. Natl. Acad. Sci. USA 2016, 113, 3832–3837. [Google Scholar] [CrossRef] [PubMed]
- Slot, M.; Winter, K. In situ Temperature Response of Photosynthesis of 42 Tree and Liana Species in the Canopy of Two Panamanian Lowland Tropical Forests with Contrasting Rainfall Regimes. New Phytol. 2017, 214, 1103–1117. [Google Scholar] [CrossRef] [PubMed]
- Rowland, L.; da Costa, A.C.L.; Galbraith, D.R.; Oliveira, R.S.; Binks, O.J.; Oliveira, A.A.R.; Pullen, A.M.; Doughty, C.E.; Metcalfe, D.B.; Vasconcelos, S.S.; et al. Death from Drought in Tropical Forests Is Triggered by Hydraulics Not Carbon Starvation. Nature 2015, 528, 119–122. [Google Scholar] [CrossRef] [PubMed]
- Slik, J.F.; Arroyo-Rodríguez, V.; Aiba, S.I.; Alvarez-Loayza, P.; Alves, L.F.; Ashton, P.; Balvanera, P.; Bastian, M.L.; Bellingham, P.J.; Van Den Berg, E.; et al. An Estimate of the Number of Tropical Tree Species. Proc. Natl. Acad. Sci. USA 2015, 112, 7472–7477. [Google Scholar] [CrossRef]
- Slot, M.; Winter, K. In Situ Temperature Relationships of Biochemical and Stomatal Controls of Photosynthesis in Four Lowland Tropical Tree Species. Plant Cell Environ. 2017, 40, 3055–3068. [Google Scholar] [CrossRef]
- Clark, D.B.; Clark, D.A.; Oberbauer, S.F. Annual Wood Production in a Tropical Rain Forest in Ne Costa Rica Linked to Climatic Variation but Not to Increasing CO2. Glob. Chang. Biol. 2010, 16, 747–759. [Google Scholar] [CrossRef]
- McDowell, N.; Allen, C.D.; Anderson-Teixeira, K.; Brando, P.; Brienen, R.; Chambers, J.; Christoffersen, B.; Davies, S.; Doughty, C.; Duque, A.; et al. Drivers and Mechanisms of Tree Mortality in Moist Tropical Forests. New Phytol. 2018, 219, 851–869. [Google Scholar] [CrossRef]
- Condit, R.; Aguilar, S.; Hernandez, A.; Perez, R.; Lao, S.; Angehr, G.; Hubbell, S.P.; Foster, R.B. Tropical Forest Dynamics across a Rainfall Gradient and the Impact of an El Niño Dry Season. J. Trop. Ecol. 2004, 20, 51–72. [Google Scholar] [CrossRef]
- Chen, Y.; Morton, D.C.; Andela, N.; Van Der Werf, G.R.; Giglio, L.; Randerson, J.T. A Pan-Tropical Cascade of Fire Driven by El Niño/Southern Oscillation. Nat. Clim. Chang. 2017, 7, 906. [Google Scholar] [CrossRef]
- Fonseca, M.G.; Anderson, L.O.; Arai, E.; Shimabukuro, Y.E.; Xaud, H.A.; Xaud, M.R.; Madani, N.; Wagner, F.H.; Aragão, L.E. Climatic and Anthropogenic Drivers of Northern Amazon Fires during the 2015–2016 El Niño Event. Ecol. Appl. 2017, 27, 2514–2527. [Google Scholar] [CrossRef] [PubMed]
- Silva Junior, C.; Aragão, L.; Fonseca, M.; Almeida, C.; Vedovato, L.; Anderson, L. Deforestation-Induced Fragmentation Increases Forest Fire Occurrence in Central Brazilian Amazonia. Forests 2018, 9, 305. [Google Scholar] [CrossRef]
- Cochrane, M.A. Synergistic Interactions between Habitat Fragmentation and Fire in Evergreen Tropical Forests. Conserv. Biol. 2001, 15, 1515–1521. [Google Scholar] [CrossRef]
- Armenteras, D.; González, T.M.; Retana, J. Forest Fragmentation and Edge Influence on Fire Occurrence and Intensity under Different Management Types in Amazon Forests. Biol. Conserv. 2013, 159, 73–79. [Google Scholar] [CrossRef]
- Cano-Crespo, A.; Oliveira, P.J.; Boit, A.; Cardoso, M.; Thonicke, K. Forest Edge Burning in the Brazilian Amazon Promoted by Escaping Fires from Managed Pastures. J. Geophys. Res. Biogeosci. 2015, 120, 2095–2107. [Google Scholar] [CrossRef]
- Jiménez-Muñoz, J.C.; Mattar, C.; Barichivich, J.; Santamaría-Artigas, A.; Takahashi, K.; Malhi, Y.; Sobrino, J.A.; Van Der Schrier, G. Record-Breaking Warming and Extreme Drought in the Amazon Rainforest During the Course of El Niño 2015–2016. Sci. Rep. 2016, 6, 33130. [Google Scholar] [CrossRef]
- Yamori, W.; Hikosaka, K.; Way, D.A. Temperature Response of Photosynthesis in C3, C4, and Cam Plants: Temperature Acclimation and Temperature Adaptation. Photosynth. Res. 2014, 119, 101–117. [Google Scholar] [CrossRef]
- Fauset, S.; Baker, T.R.; Lewis, S.L.; Feldpausch, T.R.; Affum-Baffoe, K.; Foli, E.G.; Hamer, K.C.; Swaine, M.D. Drought-Induced Shifts in the Floristic and Functional Composition of Tropical Forests in Ghana. Ecol. Lett. 2012, 15, 1120–1129. [Google Scholar] [CrossRef]
- Esquivel-Muelbert, A.; Baker, T.R.; Dexter, K.G.; Lewis, S.L.; Brienen, R.J.; Feldpausch, T.R.; Lloyd, J.; Monteagudo-Mendoza, A.; Arroyo, L.; Álvarez-Dávila, E.; et al. Compositional Response of Amazon Forests to Climate Change. Glob. Chang. Biol. 2019, 25, 39–56. [Google Scholar] [CrossRef] [PubMed]
- Coelho, C.A.; Cavalcanti, I.A.; Costa, S.M.; Freitas, S.R.; Ito, E.R.; Luz, G.; Santos, A.F.; Nobre, C.A.; Marengo, J.A.; Pezza, A.B. Climate Diagnostics of Three Major Drought Events in the Amazon and Illustrations of Their Seasonal Precipitation Predictions. Meteorol. Appl. 2012, 19, 237–255. [Google Scholar] [CrossRef]
- Slik, J.F.; Franklin, J.; Arroyo-Rodríguez, V.; Field, R.; Aguilar, S.; Aguirre, N.; Ahumada, J.; Aiba, S.I.; Alves, L.F.; Anitha, K.; et al. Phylogenetic Classification of the World’s Tropical Forests. Proc. Natl. Acad. Sci. USA 2018, 115, 1837–1842. [Google Scholar] [CrossRef] [PubMed]
- Choat, B.; Jansen, S.; Brodribb, T.J.; Cochard, H.; Delzon, S.; Bhaskar, R.; Bucci, S.J.; Feild, T.S.; Gleason, S.M.; Hacke, U.G.; et al. Global Convergence in the Vulnerability of Forests to Drought. Nature 2012, 491, 752–755. [Google Scholar] [CrossRef] [PubMed]
- Olson, D.M.; Dinerstein, E.; Wikramanayake, E.D.; Burgess, N.D.; Powell, G.V.; Underwood, E.C.; D’amico, J.A.; Itoua, I.; Strand, H.E.; Morrison, J.C.; et al. Terrestrial Ecoregions of the World: A New Map of Life on Earth. BioScience 2001, 51, 933–938. [Google Scholar] [CrossRef]
- Aragao, L.E.O.; Malhi, Y.; Roman-Cuesta, R.M.; Saatchi, S.; Anderson, L.O.; Shimabukuro, Y.E. Spatial Patterns and Fire Response of Recent Amazonian Droughts. Geophys. Res. Lett. 2007, 34, 1–5. [Google Scholar] [CrossRef]
- Vedovato, L.B.; Fonseca, M.G.; Arai, E.; Anderson, L.O.; Aragao, L.E. The Extent of 2014 Forest Fragmentation in the Brazilian Amazon. Reg. Environ. Chang. 2016, 16, 2485–2490. [Google Scholar] [CrossRef]
- Armenteras, D.; Barreto, J.S.; Tabor, K.; Molowny-Horas, R.; Retana, J. Changing Patterns of Fire Occurrence in Proximity to Forest Edges, Roads and Rivers between Nw Amazonian Countries. Biogeosciences 2017, 14, 2755–2765. [Google Scholar] [CrossRef]
- Hansen, M.C.; Potapov, P.V.; Moore, R.; Hancher, M.; Turubanova, S.A.A.; Tyukavina, A.; Thau, D.; Stehman, S.V.; Goetz, S.J.; Loveland, T.R.; et al. High-Resolution Global Maps of 21st-Century Forest Cover Change. Science 2013, 342, 850–853. [Google Scholar] [CrossRef] [PubMed]
- Avitabile, V.; Herold, M.; Heuvelink, G.B.; Lewis, S.L.; Phillips, O.L.; Asner, G.P.; Armston, J.; Ashton, P.S.; Banin, L.; Bayol, N.; et al. An Integrated Pan-Tropical Biomass Map Using Multiple Reference Datasets. Glob. Chang. Biol. 2016, 22, 1406–1420. [Google Scholar] [CrossRef] [PubMed]
- Page, S.E.; Rieley, J.O.; Banks, C.J. Global and Regional Importance of the Tropical Peatland Carbon Pool. Glob. Chang. Biol. 2011, 17, 798–818. [Google Scholar] [CrossRef]
- Aragão, L.E.; Anderson, L.O.; Fonseca, M.G.; Rosan, T.M.; Vedovato, L.B.; Wagner, F.H.; Silva, C.V.; Junior, C.H.S.; Arai, E.; Aguiar, A.P.; et al. 21st Century Drought-Related Fires Counteract the Decline of Amazon Deforestation Carbon Emissions. Nat. Commun. 2018, 9, 536. [Google Scholar] [CrossRef] [PubMed]
- Silva Junior, C.H.; Anderson, L.O.; Silva, A.L.; Almeida, C.T.; Dalagnol, R.; Pletsch, M.A.; Penha, T.V.; Paloschi, R.A.; Aragão, L.E. Fire Responses to the 2010 and 2015/2016 Amazonian Droughts. Front. Earth Sci. 2019, 7, 97. [Google Scholar] [CrossRef]
- Engelbrecht, B.M.; Comita, L.S.; Condit, R.; Kursar, T.A.; Tyree, M.T.; Turner, B.L.; Hubbell, S.P. Drought Sensitivity Shapes Species Distribution Patterns in Tropical Forests. Nature 2007, 447, 80–82. [Google Scholar] [CrossRef] [PubMed]
- Zelazowski, P.; Malhi, Y.; Huntingford, C.; Sitch, S.; Fisher, J.B. Changes in the Potential Distribution of Humid Tropical Forests on a Warmer Planet. Philos. Trans. R. Soc. Lond. A Math. Phys. Eng. Sci. 2011, 369, 137–160. [Google Scholar] [CrossRef] [PubMed]
- Bennett, A.C.; McDowell, N.G.; Allen, C.D.; Anderson-Teixeira, K.J. Larger Trees Suffer Most During Drought in Forests Worldwide. Nat. Plants 2015, 1, 15139. [Google Scholar] [CrossRef] [PubMed]
- Brando, P.M.; Nepstad, D.C.; Balch, J.K.; Bolker, B.; Christman, M.C.; Coe, M.; Putz, F.E. Fire-Induced Tree Mortality in a Neotropical Forest: The Roles of Bark Traits, Tree Size, Wood Density and Fire Behavior. Glob. Chang. Biol. 2012, 18, 630–641. [Google Scholar] [CrossRef]
- Van Nieuwstadt, M.G.; Sheil, D. Drought, Fire and Tree Survival in a Borneo Rain Forest, East Kalimantan, Indonesia. J. Ecol. 2005, 93, 191–201. [Google Scholar] [CrossRef]
- Taylor, T.C.; Smith, M.N.; Slot, M.; Feeley, K.J. The Capacity to Emit Isoprene Differentiates the Photosynthetic Temperature Responses of Tropical Plant Species. Plant Cell Environ. 2019, 42, 2448–2457. [Google Scholar] [CrossRef]
- Esquivel-Muelbert, A.; Baker, T.R.; Dexter, K.G.; Lewis, S.L.; ter Steege, H.; Lopez-Gonzalez, G.; Monteagudo Mendoza, A.; Brienen, R.; Feldpausch, T.R.; Pitman, N.; et al. Seasonal Drought Limits Tree Species across the Neotropics. Ecography 2017, 40, 618–629. [Google Scholar] [CrossRef]
- Spracklen, D.V.; Arnold, S.R.; Taylor, C.M. Observations of Increased Tropical Rainfall Preceded by Air Passage over Forests. Nature 2012, 489, 282. [Google Scholar] [CrossRef] [PubMed]
- Cai, W.; Wang, G.; Dewitte, B.; Wu, L.; Santoso, A.; Takahashi, K.; Yang, Y.; Carréric, A.; McPhaden, M.J. Increased Variability of Eastern Pacific El Niño under Greenhouse Warming. Nature 2018, 564, 201–206. [Google Scholar] [CrossRef] [PubMed]
- Cochrane, M.A.; Schulze, M.D. Fire as a Recurrent Event in Tropical Forests of the Eastern Amazon: Effects on Forest Structure, Biomass, and Species Composition. Biotropica 1999, 31, 2–16. [Google Scholar] [CrossRef]
- Silva, C.V.; Aragão, L.E.; Barlow, J.; Espirito-Santo, F.; Young, P.J.; Anderson, L.O.; Berenguer, E.; Brasil, I.; Foster Brown, I.; Castro, B.; et al. Drought-Induced Amazonian Wildfires Instigate a Decadal-Scale Disruption of Forest Carbon Dynamics. Philos. Trans. R. Soc. B Biol. Sci. 2018, 373, 20180043. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Esquivel-Muelbert, A.; Bennett, A.C.; Sullivan, M.J.P.; Baker, J.C.A.; Gavish, Y.; Johnson, M.O.; Wang, Y.; Chambers-Ostler, A.; Lisli Giannichi, M.; Gomes, L.; et al. A Spatial and Temporal Risk Assessment of the Impacts of El Niño on the Tropical Forest Carbon Cycle: Theoretical Framework, Scenarios, and Implications. Atmosphere 2019, 10, 588. https://doi.org/10.3390/atmos10100588
Esquivel-Muelbert A, Bennett AC, Sullivan MJP, Baker JCA, Gavish Y, Johnson MO, Wang Y, Chambers-Ostler A, Lisli Giannichi M, Gomes L, et al. A Spatial and Temporal Risk Assessment of the Impacts of El Niño on the Tropical Forest Carbon Cycle: Theoretical Framework, Scenarios, and Implications. Atmosphere. 2019; 10(10):588. https://doi.org/10.3390/atmos10100588
Chicago/Turabian StyleEsquivel-Muelbert, Adriane, Amy C. Bennett, Martin J. P. Sullivan, Jessica C. A. Baker, Yoni Gavish, Michelle O. Johnson, Yunxia Wang, Alexander Chambers-Ostler, Marta Lisli Giannichi, Luciene Gomes, and et al. 2019. "A Spatial and Temporal Risk Assessment of the Impacts of El Niño on the Tropical Forest Carbon Cycle: Theoretical Framework, Scenarios, and Implications" Atmosphere 10, no. 10: 588. https://doi.org/10.3390/atmos10100588
APA StyleEsquivel-Muelbert, A., Bennett, A. C., Sullivan, M. J. P., Baker, J. C. A., Gavish, Y., Johnson, M. O., Wang, Y., Chambers-Ostler, A., Lisli Giannichi, M., Gomes, L., Kalamandeen, M., Charan Pattnayak, K., & Fauset, S. (2019). A Spatial and Temporal Risk Assessment of the Impacts of El Niño on the Tropical Forest Carbon Cycle: Theoretical Framework, Scenarios, and Implications. Atmosphere, 10(10), 588. https://doi.org/10.3390/atmos10100588




