The Yellow Stripe-Like (YSL) Gene Functions in Internal Copper Transport in Peanut
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Materials and Growth Conditions
2.2. Quantitative Real-Time PCR
2.3. Subcellular Localization of AhYSL3.1
2.4. In Situ Hybridization
2.5. Functional Complementation in Yeast
2.6. Generation of Transgenic Lines
2.7. Analyses of Transgenic Tobacco and Rice Plants
2.8. Transmembrane Protein Structure Prediction and Statistical Analysis
3. Results
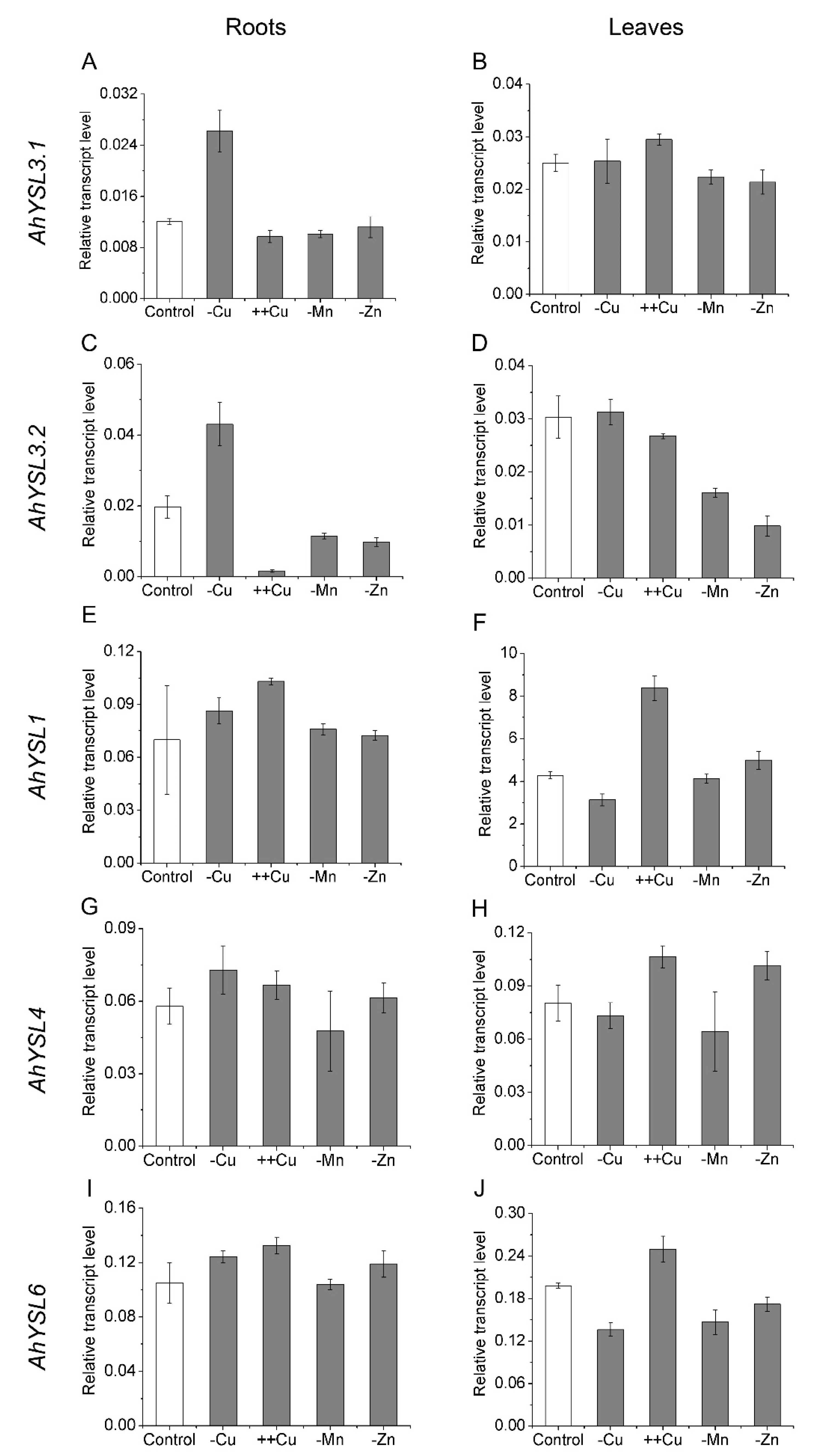
3.1. Expression of AhYSL Genes in Response to Metal Treatments
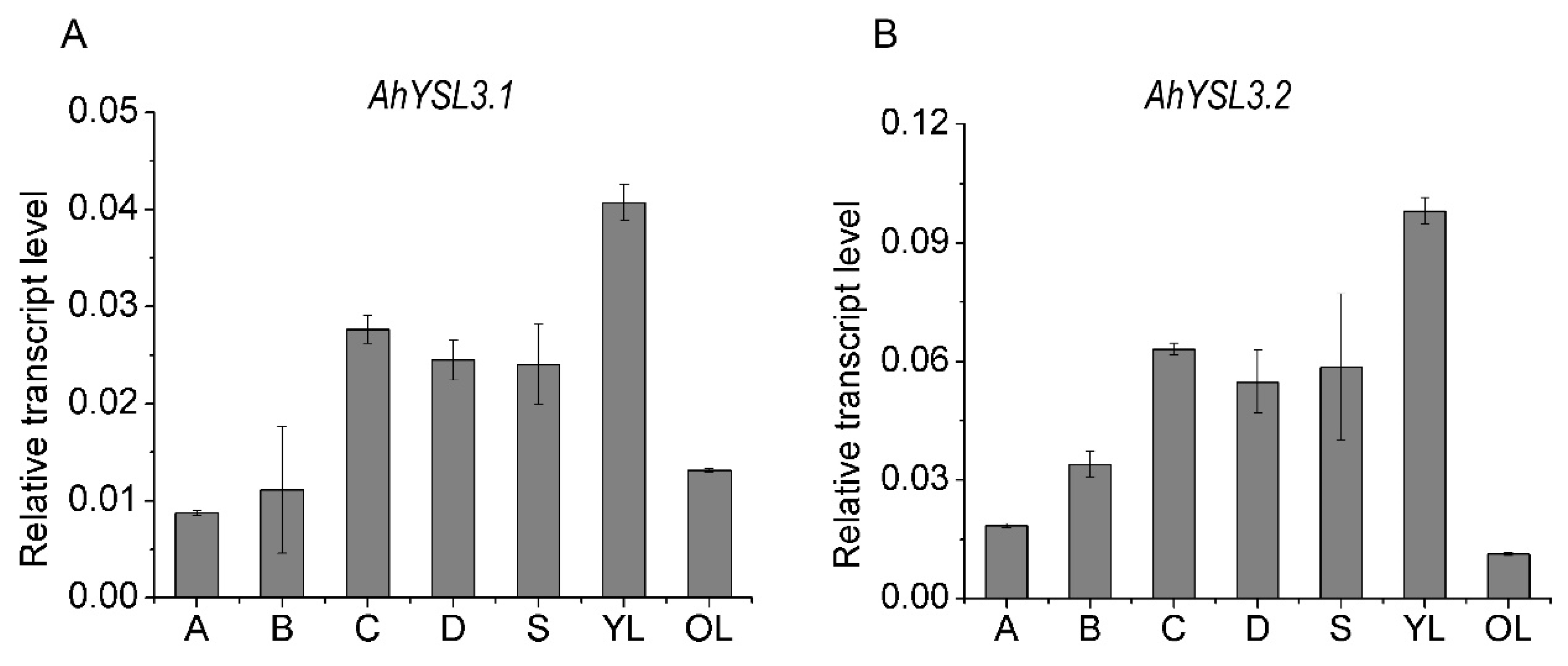
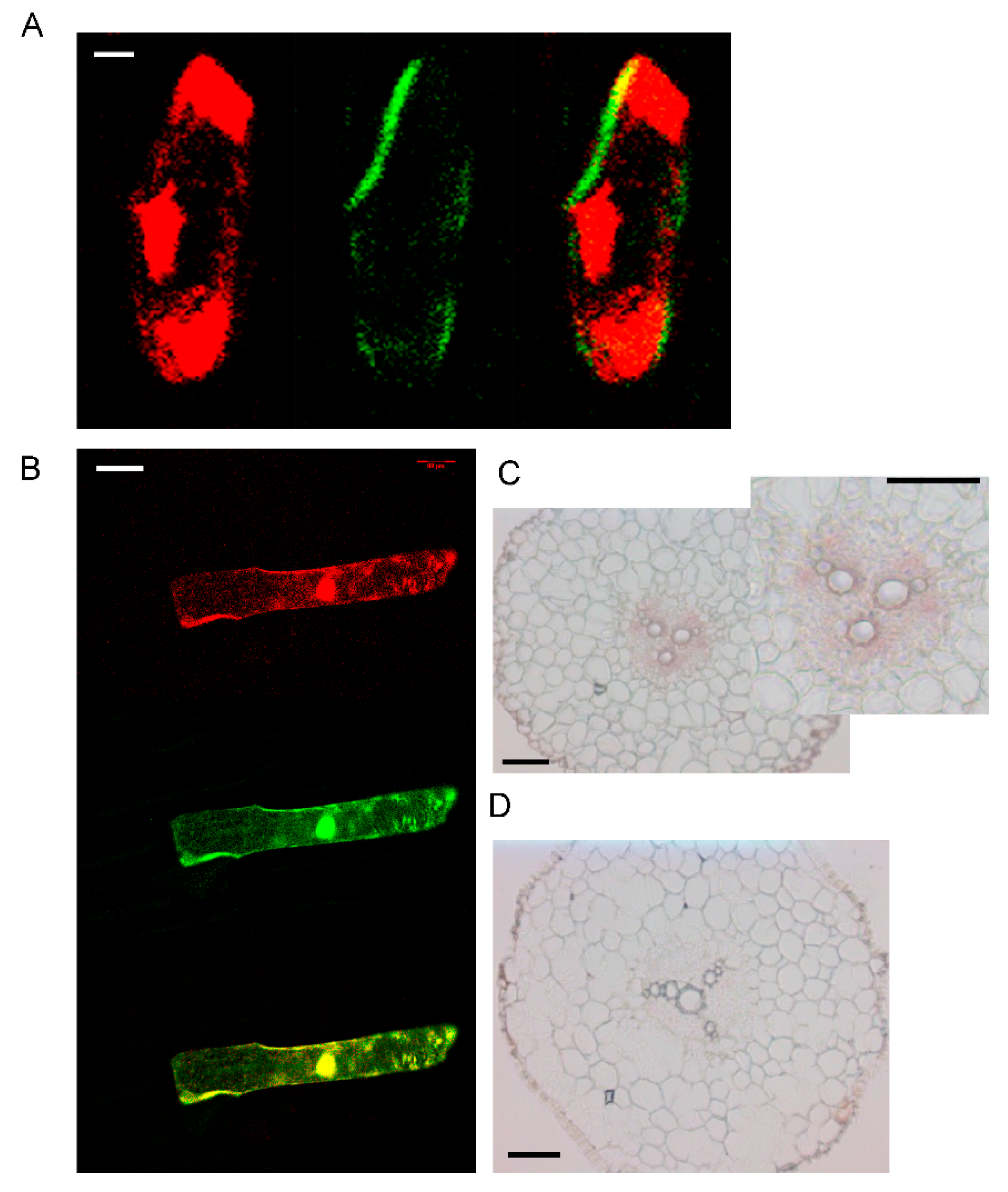
3.2. Tissue Expression and Localization of AhYSL3.1
3.3. The Subcellular Localization of AhYSL3.1
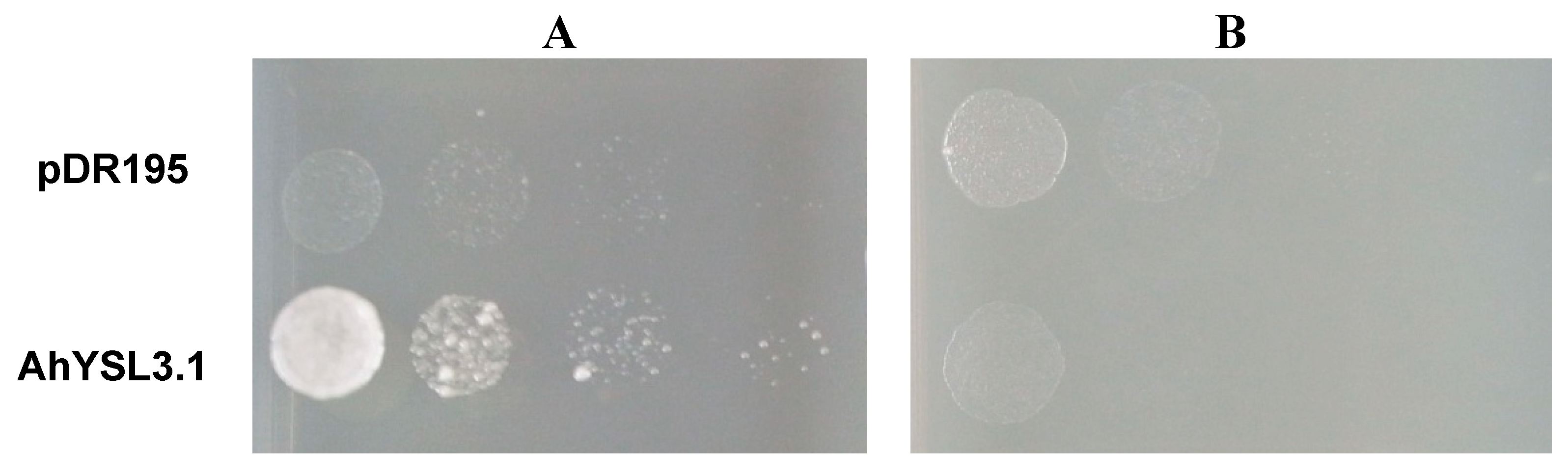
3.4. AhYSL3.1 is a Cu–NA Transporter
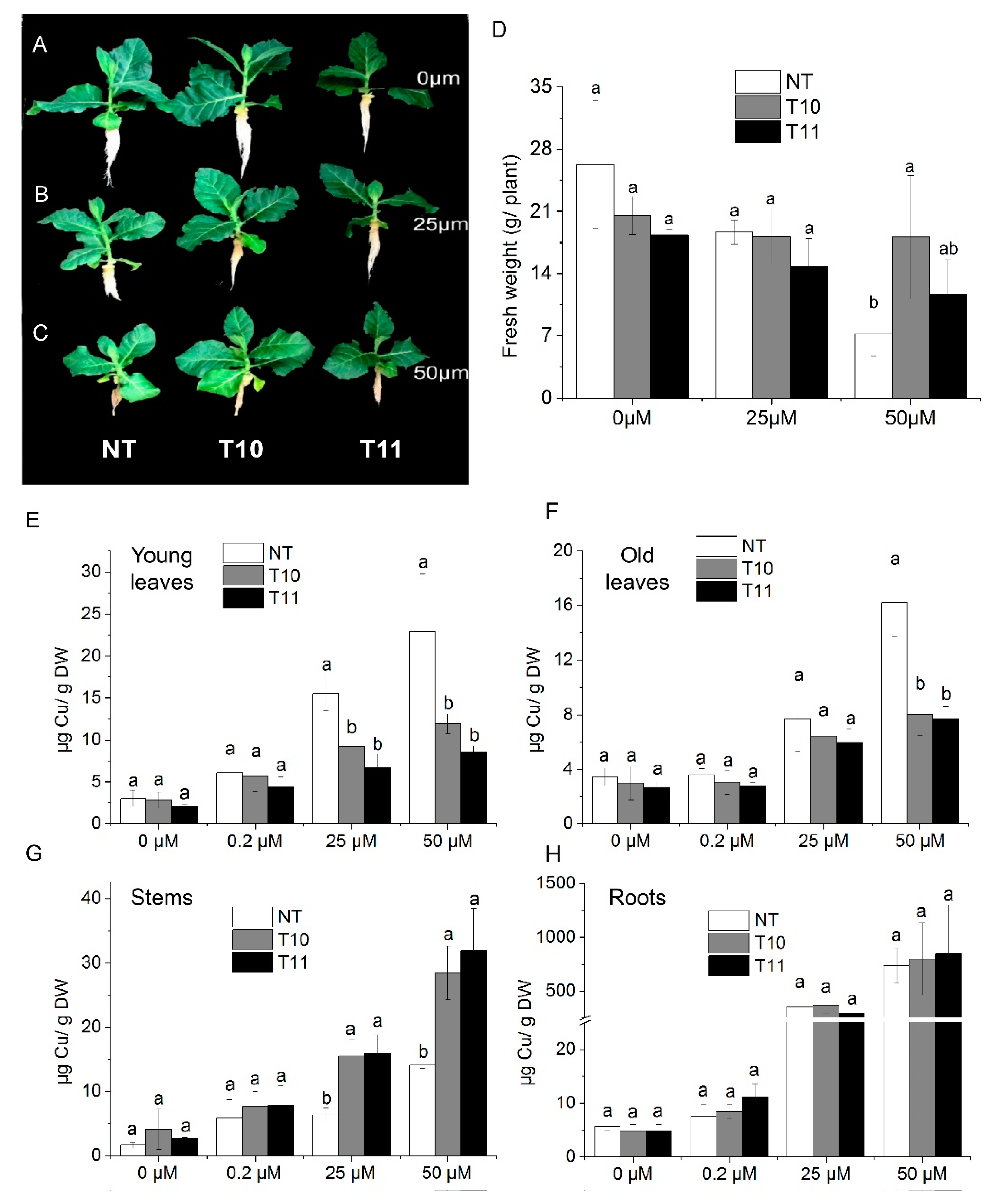
3.5. High-Level Expression of AhYSL3.1 in Tobacco Results in Tolerance to Excess Cu
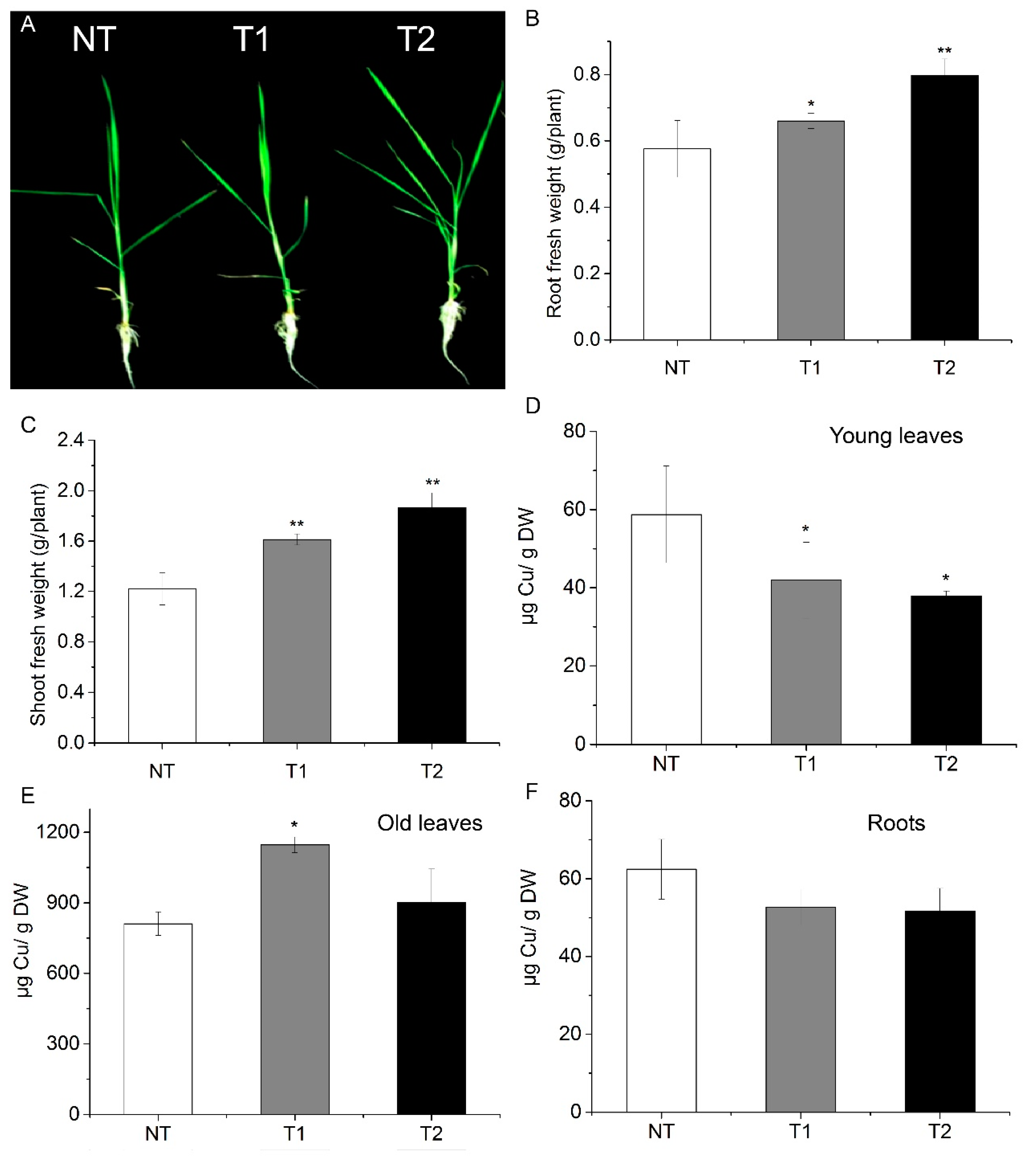
3.6. Transgenic Rice Plants Are also Tolerant to Excess Cu
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| Cu | Copper |
| CTR | Cu transporter family proteins |
| COPT | CTR-like transporters |
| HMA | The P-type heavy-metal ATPase family proteins |
| NA | Nicotianamine |
| Zn | Zinc |
| Cd | Cadmium |
| YSL | The Yellow Stripe-Like family genes |
| Fe | iron |
| SUMO/SIZ1 | Small Ubiquitin-like Modifier E3 ligase |
| DMA | Deoxymugineic acid |
| NT | Non-transgenic |
References
- Pilon, M.; Abdel-Ghany, S.E.; Cohu, C.M.; Gogolin, K.A.; Ye, H. Copper cofactor delivery in plant cells. Curr. Opin. Plant Boil. 2006, 9, 256–263. [Google Scholar] [CrossRef] [PubMed]
- Hansch, R.; Mendel, R.R. Physiological functions of mineral micronutrients (Cu, Zn, Mn, Fe, Ni, Mo, B, Cl). Curr. Opin. Plant Boil. 2009, 12, 259–266. [Google Scholar] [CrossRef] [PubMed]
- Klaumann, S.; Nickolaus, S.D.; Furst, S.H.; Starck, S.; Schneider, S.; Ekkehard Neuhaus, H.; Trentmann, O. The tonoplast copper transporter COPT5 acts as an exporter and is required for interorgan allocation of copper in Arabidopsis thaliana. New Phytol. 2011, 192, 393–404. [Google Scholar] [CrossRef] [PubMed]
- Lequeux, H.; Hermans, C.; Lutts, S.; Verbruggen, N. Response to copper excess in Arabidopsis thaliana: Impact on the root system architecture, hormone distribution, lignin accumulation and mineral profile. Plant Physiol. Biochem. 2010, 48, 673–682. [Google Scholar] [CrossRef] [PubMed]
- Adrees, M.; Ali, S.; Rizwan, M.; Ibrahim, M.; Abbas, F.; Farid, M.; Zia-Ur-Rehman, M.; Irshad, M.K.; Bharwana, S.A. The effect of excess copper on growth and physiology of important food crops: A review. Environ. Sci. Pollut. Res. Int. 2015, 22, 8148–8162. [Google Scholar] [CrossRef]
- Burkhead, J.L.; Reynolds, K.A.; Abdel-Ghany, S.E.; Cohu, C.M.; Pilon, M. Copper homeostasis. New Phytol. 2009, 182, 799–816. [Google Scholar] [CrossRef] [PubMed]
- Peñarrubia, L.; Andrés-Colás, N.; Moreno, J.; Puig, S. Regulation of copper transport in Arabidopsis thaliana: A biochemical oscillator? J. Biol. Inorg. Chem. 2010, 15, 29–36. [Google Scholar] [CrossRef]
- Sancenon, V.; Puig, S.; Mira, H.; Thiele, D.J.; Penarrubia, L. Identification of a copper transporter family in Arabidopsis thaliana. Plant Mol. Boil. 2003, 51, 577–587. [Google Scholar] [CrossRef]
- Kampfenkel, K.; Kushnir, S.; Babiychuk, E.; Inze, D.; Van Montagu, M. Molecular characterization of a putative Arabidopsis thaliana copper transporter and its yeast homologue. J. Boil. Chem. 1995, 270, 28479–28486. [Google Scholar]
- Sancenon, V.; Puig, S.; Mateu-Andres, I.; Dorcey, E.; Thiele, D.J.; Penarrubia, L. The Arabidopsis copper transporter COPT1 functions in root elongation and pollen development. J. Boil. Chem. 2004, 279, 15348–15355. [Google Scholar] [CrossRef]
- Perea-Garcia, A.; Garcia-Molina, A.; Andres-Colas, N.; Vera-Sirera, F.; Perez-Amador, M.A.; Puig, S.; Penarrubia, L. Arabidopsis copper transport protein COPT2 participates in the cross talk between iron deficiency responses and low-phosphate signaling. Plant Physiol. 2013, 162, 180–194. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Molina, A.; Andres-Colas, N.; Perea-Garcia, A.; Del Valle-Tascon, S.; Penarrubia, L.; Puig, S. The intracellular Arabidopsis COPT5 transport protein is required for photosynthetic electron transport under severe copper deficiency. Plant J. Cell Mol. Boil. 2011, 65, 848–860. [Google Scholar] [CrossRef] [PubMed]
- Yuan, M.; Li, X.; Xiao, J.; Wang, S. Molecular and functional analyses of COPT/Ctr-type copper transporter-like gene family in rice. BMC Plant Boil. 2011, 11, 69. [Google Scholar] [CrossRef] [PubMed]
- Jung, H.I.; Gayomba, S.R.; Rutzke, M.A.; Craft, E.; Kochian, L.V.; Vatamaniuk, O.K. COPT6 is a plasma membrane transporter that functions in copper homeostasis in Arabidopsis and is a novel target of SQUAMOSA promoter-binding protein-like 7. J. Boil. Chem. 2012, 287, 33252–33267. [Google Scholar] [CrossRef] [PubMed]
- Yuan, M.; Chu, Z.; Li, X.; Xu, C.; Wang, S. The bacterial pathogen Xanthomonas oryzae overcomes rice defenses by regulating host copper redistribution. Plant Cell 2010, 22, 3164–3176. [Google Scholar] [CrossRef] [PubMed]
- Tapken, W.; Ravet, K.; Pilon, M. Plastocyanin controls the stabilization of the thylakoid Cu-transporting P-type ATPase PAA2/HMA8 in response to low copper in Arabidopsis. J. Boil. Chem. 2012, 287, 18544–18550. [Google Scholar] [CrossRef] [PubMed]
- Catty, P.; Boutigny, S.; Miras, R.; Joyard, J.; Rolland, N.; Seigneurin-Berny, D. Biochemical characterization of AtHMA6/PAA1, a chloroplast envelope Cu(I)-ATPase. J. Boil. Chem. 2011, 286, 36188–36197. [Google Scholar] [CrossRef] [PubMed]
- Seigneurin-Berny, D.; Gravot, A.; Auroy, P.; Mazard, C.; Kraut, A.; Finazzi, G.; Grunwald, D.; Rappaport, F.; Vavasseur, A.; Joyard, J.; et al. HMA1, a new Cu-ATPase of the chloroplast envelope, is essential for growth under adverse light conditions. J. Boil. Chem. 2006, 281, 2882–2892. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Ghany, S.E.; Muller-Moule, P.; Niyogi, K.K.; Pilon, M.; Shikanai, T. Two P-type atpases are required for copper delivery in Arabidopsis thaliana chloroplasts. Plant Cell 2005, 17, 1233–1251. [Google Scholar] [CrossRef] [PubMed]
- Andres-Colas, N.; Sancenon, V.; Rodriguez-Navarro, S.; Mayo, S.; Thiele, D.J.; Ecker, J.R.; Puig, S.; Penarrubia, L. The Arabidopsis heavy metal P-type atpase HMA5 interacts with metallochaperones and functions in copper detoxification of roots. Plant J. Cell Mol. Boil. 2006, 45, 225–236. [Google Scholar] [CrossRef]
- Kobayashi, Y.; Kuroda, K.; Kimura, K.; Southron-Francis, J.L.; Furuzawa, A.; Kimura, K.; Iuchi, S.; Kobayashi, M.; Taylor, G.J.; Koyama, H. Amino acid polymorphisms in strictly conserved domains of a P-type ATPase HMA5 are involved in the mechanism of copper tolerance variation in Arabidopsis. Plant Physiol. 2008, 148, 969–980. [Google Scholar] [CrossRef] [PubMed]
- Deng, F.; Yamaji, N.; Xia, J.; Ma, J.F. A member of the heavy metal P-type ATPase OsHMA5 is involved in xylem loading of copper in rice. Plant Physiol. 2013, 163, 1353–1362. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.Y.; Deng, F.; Yamaji, N.; Pinson, S.R.; Fujii-Kashino, M.; Danku, J.; Douglas, A.; Guerinot, M.L.; Salt, D.E.; Ma, J.F. A heavy metal P-type ATPase OsHMA4 prevents copper accumulation in rice grain. Nat. Commun. 2016, 7, 12138. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Kim, Y.Y.; Lee, Y.; An, G. Rice P1B-type heavy-metal aATPase, OsHMA9, is a metal efflux protein. Plant Physiol. 2007, 145, 831–842. [Google Scholar] [CrossRef] [PubMed]
- Migocka, M.; Posyniak, E.; Maciaszczyk-Dziubinska, E.; Papierniak, A.; Kosieradzaka, A. Functional and biochemical characterization of cucumber genes encoding two copper ATPases CsHMA5.1 and CsHMA5.2. J. Boil. Chem. 2015, 290, 15717–15729. [Google Scholar] [CrossRef] [PubMed]
- Benes, I.; Schreiber, K.; Ripperger, H.; Kircheiss, A. The “normalizing factor” for the tomato mutant chloronerva, XXIII: Metal-complex formation by nicotianamine, a possible phytosiderophore. Cell. Mol. Life Sci. 1983, 39, 261–262. [Google Scholar]
- Anderegg, G.; Ripperger, H. The “normalizing factor” for the tomato mutant chloronerva, XXIX: Correlation between metal-complex formation and biological-activity of nicotianamine analogs. J. Chem. Soc. Chem. Comm. 1989, 647–650. [Google Scholar] [CrossRef]
- Pich, A.; Scholz, G.; Stephan, U. Iron-dependent changes of heavy metals, nicotianamine, and citrate in different plant organs and in the xylem exudate of two tomato genotypes. Nicotianamine as possible copper translocator. Plant Soil 1994, 165, 189–196. [Google Scholar] [CrossRef]
- Pich, A.; Scholz, G. Translocation of copper and other micronutrients in tomato plants (Lycopersicon esculentum mill): Nicotianamine-stimulated copper transport in the xylem. J. Exp. Bot. 1996, 47, 41–47. [Google Scholar] [CrossRef]
- DiDonato, R.J., Jr.; Roberts, L.A.; Sanderson, T.; Eisley, R.B.; Walker, E.L. Arabidopsis Yellow Stripe-Like2 (YSL2): A metal-regulated gene encoding a plasma membrane transporter of nicotianamine-metal complexes. Plant J. Cell Mol. Boil. 2004, 39, 403–414. [Google Scholar] [CrossRef]
- Schaaf, G.; Schikora, A.; Haberle, J.; Vert, G.; Ludewig, U.; Briat, J.F.; Curie, C.; von Wiren, N. A putative function for the Arabidopsis Fe-Phytosiderophore transporter homolog AtYSL2 in Fe and Zn homeostasis. Plant Cell Physiol. 2005, 46, 762–774. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.C.; Chen, Y.Y.; Tang, I.C.; Liang, H.M.; Lai, C.C.; Chiou, J.M.; Yeh, K.C. Arabidopsis SUMO E3 ligase SIZ1 is involved in excess copper tolerance. Plant Physiol. 2011, 156, 2225–2234. [Google Scholar] [CrossRef] [PubMed]
- Zheng, L.; Yamaji, N.; Yokosho, K.; Ma, J.F. YSL16 is a phloem-localized transporter of the copper-nicotianamine complex that is responsible for copper distribution in rice. Plant Cell 2012, 24, 3767–3782. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Lu, W.; Yang, Y.; Shen, Z.; Ma, J.F.; Zheng, L. OsYSL16 is required for preferential cu distribution to floral organs in rice. Plant Cell Physiol. 2018, 59, 2039–2051. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Ryoo, N.; Jeon, J.S.; Guerinot, M.L.; An, G. Activation of rice Yellow Stripe1-Like 16 (OsYSL16) enhances iron efficiency. Mol. Cells 2012, 33, 117–126. [Google Scholar] [CrossRef]
- Kakei, Y.; Ishimaru, Y.; Kobayashi, T.; Yamakawa, T.; Nakanishi, H.; Nishizawa, N.K. OsYSL16 plays a role in the allocation of iron. Plant Mol. Boil. 2012, 79, 583–594. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, T.; Nishizawa, N.K. Iron uptake, translocation, and regulation in higher plants. Annu. Rev. Plant Boil. 2012, 63, 131–152. [Google Scholar] [CrossRef]
- Xiong, H.; Kakei, Y.; Kobayashi, T.; Guo, X.; Nakazono, M.; Takahashi, H.; Nakanishi, H.; Shen, H.; Zhang, F.; Nishizawa, N.K.; et al. Molecular evidence for phytosiderophore-induced improvement of iron nutrition of peanut intercropped with maize in calcareous soil. Plant Cell Environ. 2013, 36, 1888–1902. [Google Scholar] [CrossRef]
- Xiong, H.; Kobayashi, T.; Kakei, Y.; Senoura, T.; Nakazono, M.; Takahashi, H.; Nakanishi, H.; Shen, H.; Duan, P.; Guo, X.; et al. AhNRAMP1 iron transporter is involved in iron acquisition in peanut. J. Exp. Bot. 2012, 63, 4437–4446. [Google Scholar] [CrossRef]
- Luo, M.; Dang, P.; Bausher, M.G.; Holbrook, C.C.; Lee, R.D.; Lynch, R.E.; Guo, B.Z. Identification of transcripts involved in resistance responses to leaf spot disease caused by Cercosporidium personatum in peanut (Arachis hypogaea). Phytopathology 2005, 95, 381–387. [Google Scholar] [CrossRef]
- Mizuno, D.; Higuchi, K.; Sakamoto, T.; Nakanishi, H.; Mori, S.; Nishizawa, N.K. Three nicotianamine synthase genes isolated from maize are differentially regulated by iron nutritional status. Plant Physiol. 2003, 132, 1989–1997. [Google Scholar] [CrossRef] [PubMed]
- Gietz, R.D.; Schiestl, R.H. Transforming yeast with DNA. Methods Mol. Cell. Biol. 1995, 5, 255–269. [Google Scholar]
- Kobayashi, T.; Nakayama, Y.; Takahashi, M.; Inoue, H.; Nakanishi, H.; Yoshihara, T.; Mori, S.; Nishizawa, N.K. Construction of artificial promoters highly responsive to iron deficiency. Soil Sci. Plant Nutr. 2004, 50, 1167–1175. [Google Scholar] [CrossRef]
- Kobayashi, T.; Nakayama, Y.; Itai, R.N.; Nakanishi, H.; Yoshihara, T.; Mori, S.; Nishizawa, N.K. Identification of novel cis-acting elements, IDE1 and IDE2, of the barley IDS2 gene promoter conferring iron-deficiency-inducible, root-specific expression in heterogeneous tobacco plants. Plant J. Cell Mol. Boil. 2003, 36, 780–793. [Google Scholar] [CrossRef]
- Hiei, Y.; Ohta, S.; Komari, T.; Kumashiro, T. Efficient transformation of rice (Oryza sativa L.) mediated by agrobacterium and sequence analysis of the boundaries of the t-DNA. Plant J. Cell Mol. Boil. 1994, 6, 271–282. [Google Scholar] [CrossRef]
- Helmer, G.; Casadaban, M.; Bevan, M.; Kayes, L.; Chilton, M.D. A new chimeric gene as a marker for plant transformation—The expression of Escherichia-coli β-galactosidase in sunflower and tobacco cells. Biotechnolgy 1984, 2, 520–527. [Google Scholar] [CrossRef]
- TOPCONS. Available online: http://www.topcons.net/pred/ (accessed on 5 December 2018).
- Dancis, A.; Yuan, D.S.; Haile, D.; Askwith, C.; Eide, D.; Moehle, C.; Kaplan, J.; Klausner, R.D. Molecular characterization of a copper transport protein in S. cerevisiae: An unexpected role for copper in iron transport. Cell 1994, 76, 393–402. [Google Scholar] [CrossRef]
- Pilon, M. Moving copper in plants. New Phytol. 2011, 192, 305–307. [Google Scholar] [CrossRef]
- Pena, M.M.; Puig, S.; Thiele, D.J. Characterization of the Saccharomyces cerevisiae high affinity copper transporter CTR3. J. Boil. Chem. 2000, 275, 33244–33251. [Google Scholar] [CrossRef]
- Rees, E.M.; Lee, J.; Thiele, D.J. Mobilization of intracellular copper stores by the CTR2 vacuolar copper transporter. J. Boil. Chem. 2004, 279, 54221–54229. [Google Scholar] [CrossRef]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dai, J.; Wang, N.; Xiong, H.; Qiu, W.; Nakanishi, H.; Kobayashi, T.; Nishizawa, N.K.; Zuo, Y. The Yellow Stripe-Like (YSL) Gene Functions in Internal Copper Transport in Peanut. Genes 2018, 9, 635. https://doi.org/10.3390/genes9120635
Dai J, Wang N, Xiong H, Qiu W, Nakanishi H, Kobayashi T, Nishizawa NK, Zuo Y. The Yellow Stripe-Like (YSL) Gene Functions in Internal Copper Transport in Peanut. Genes. 2018; 9(12):635. https://doi.org/10.3390/genes9120635
Chicago/Turabian StyleDai, Jing, Nanqi Wang, Hongchun Xiong, Wei Qiu, Hiromi Nakanishi, Takanori Kobayashi, Naoko K. Nishizawa, and Yuanmei Zuo. 2018. "The Yellow Stripe-Like (YSL) Gene Functions in Internal Copper Transport in Peanut" Genes 9, no. 12: 635. https://doi.org/10.3390/genes9120635
APA StyleDai, J., Wang, N., Xiong, H., Qiu, W., Nakanishi, H., Kobayashi, T., Nishizawa, N. K., & Zuo, Y. (2018). The Yellow Stripe-Like (YSL) Gene Functions in Internal Copper Transport in Peanut. Genes, 9(12), 635. https://doi.org/10.3390/genes9120635




