Genomics of the Parasitic Nematode Ascaris and Its Relatives
Abstract
1. Introduction
2. History of Ascaris Genome Assemblies
3. Ascaris Genomes
4. Ascaris Genes and Transcriptomes
5. Ascaris Small RNAs
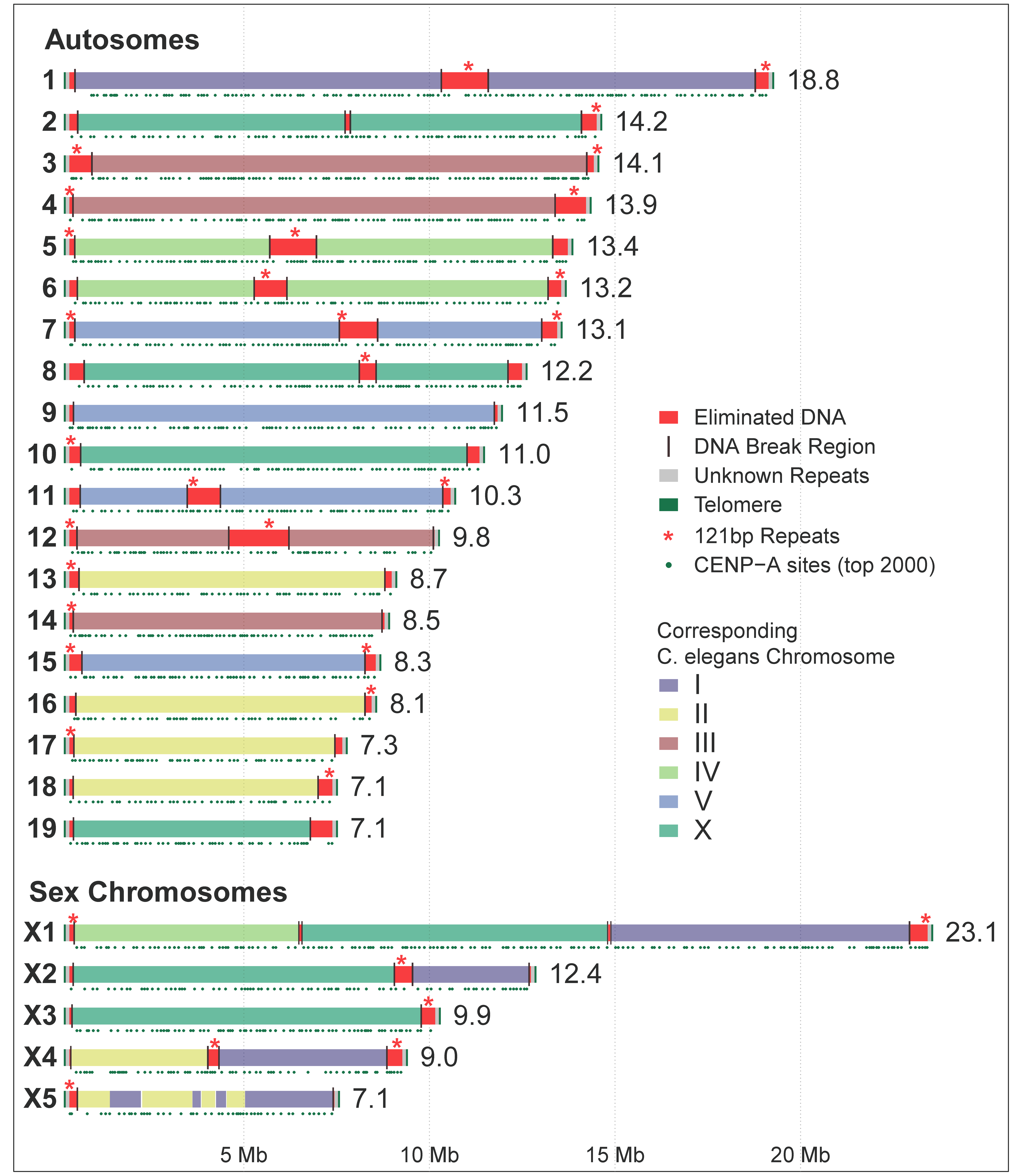
6. Ascaris Chromatin and Epigenome
7. Population Genomics of Ascaris from Pigs and Humans
8. Comparative Genomics and Ascarids Evolution
9. Future Perspectives
Supplementary Materials
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wang, J.; Davis, R.E. Ascaris. Curr. Biol. 2020, 30, R423–R425. [Google Scholar] [CrossRef] [PubMed]
- Bethony, J.; Brooker, S.; Albonico, M.; Geiger, S.M.; Loukas, A.; Diemert, D.; Hotez, P.J. Soil-transmitted helminth infections: Ascariasis, trichuriasis, and hookworm. Lancet 2006, 367, 1521–1532. [Google Scholar] [CrossRef]
- Hotez, P.J. The Unholy Trinity: The soil-transmitted helminth infections ascariasis, trichuriasis, and hookworm infection. Forgot. People Forgot. Dis. ASM Press 2013, 17–40. [Google Scholar] [CrossRef]
- Jourdan, P.M.; Lamberton, P.H.L.; Fenwick, A.; Addiss, D.G. Soil-transmitted helminth infections. Lancet 2018, 391, 252–265. [Google Scholar] [CrossRef]
- Hotez, P.J.; Brindley, P.J.; Bethony, J.M.; King, C.H.; Pearce, E.J.; Jacobson, J. Helminth infections: The great neglected tropical diseases. J. Clin. Investig. 2008, 118, 1311–1321. [Google Scholar] [CrossRef] [PubMed]
- Brooker, S. Estimating the global distribution and disease burden of intestinal nematode infections: Adding up the numbers-a review. Int. J. Parasitol. 2010, 40, 1137–1144. [Google Scholar] [CrossRef]
- Pullan, R.L.; Smith, J.L.; Jasrasaria, R.; Brooker, S.J. Global numbers of infection and disease burden of soil transmitted helminth infections in 2010. Parasit. Vectors 2014, 7, 37. [Google Scholar] [CrossRef]
- Sadaow, L.; Sanpool, O.; Phosuk, I.; Rodpai, R.; Thanchomnang, T.; Wijit, A.; Anamnart, W.; Laymanivong, S.; Aung, W.P.P.; Janwan, P.; et al. Molecular identification of Ascaris lumbricoides and Ascaris suum recovered from humans and pigs in Thailand, Lao PDR, and Myanmar. Parasitol. Res. 2018, 117, 2427–2436. [Google Scholar] [CrossRef]
- Betson, M.; Nejsum, P.; Bendall, R.P.; Deb, R.M.; Stothard, J.R. Molecular epidemiology of ascariasis: A global perspective on the transmission dynamics of Ascaris in people and pigs. J. Infect. Dis. 2014, 210, 932–941. [Google Scholar] [CrossRef]
- Miller, L.A.; Colby, K.; Manning, S.E.; Hoenig, D.; McEvoy, E.; Montgomery, S.; Mathison, B.; de Almeida, M.; Bishop, H.; Dasilva, A.; et al. Ascariasis in humans and pigs on small-scale farms, Maine, USA, 2010–2013. Emerg. Infect. Dis. 2015, 21, 332–334. [Google Scholar] [CrossRef]
- Easton, A.; Gao, S.; Lawton, S.P.; Bennuru, S.; Khan, A.; Dahlstrom, E.; Oliveira, R.G.; Kepha, S.; Porcella, S.F.; Webster, J.; et al. Molecular evidence of hybridization between pig and human Ascaris indicates an interbred species complex infecting humans. Elife 2020, 9. [Google Scholar] [CrossRef]
- Satzinger, H. Theodor and Marcella Boveri: Chromosomes and cytoplasm in heredity and development. Nat. Rev. Genet. 2008, 9, 231–238. [Google Scholar] [CrossRef] [PubMed]
- Maderspacher, F. Theodor Boveri and the natural experiment. Curr. Biol. CB 2008, 18, R279–R286. [Google Scholar] [CrossRef]
- Muller, F.; Tobler, H. Chromatin diminution in the parasitic nematodes Ascaris suum and parascaris univalens. Int. J. Parasitol. 2000, 30, 391–399. [Google Scholar] [CrossRef]
- Wang, J.; Davis, R.E. Programmed DNA elimination in multicellular organisms. Curr. Opin. Genet. Dev. 2014, 27, 26–34. [Google Scholar] [CrossRef]
- Streit, A.; Wang, J.; Kang, Y.; Davis, R.E. Gene silencing and sex determination by programmed DNA elimination in parasitic nematodes. Curr. Opin. Microbiol. 2016, 32, 120–127. [Google Scholar] [CrossRef]
- Hannon, G.J.; Maroney, P.A.; Denker, J.A.; Nilsen, T.W. Trans splicing of nematode pre-messenger RNA in vitro. Cell 1990, 61, 1247–1255. [Google Scholar] [CrossRef]
- Davis, R.E. Spliced leader RNA trans-splicing in metazoa. Parasitol. Today 1996, 12, 33–40. [Google Scholar] [CrossRef]
- Lall, S.; Friedman, C.C.; Jankowska-Anyszka, M.; Stepinski, J.; Darzynkiewicz, E.; Davis, R.E. Contribution of trans-splicing, 5′ -leader length, cap-poly(A) synergism, and initiation factors to nematode translation in an Ascaris suum embryo cell-free system. J. Biol. Chem. 2004, 279, 45573–45585. [Google Scholar] [CrossRef]
- Cohen, L.S.; Mikhli, C.; Friedman, C.; Jankowska-Anyszka, M.; Stepinski, J.; Darzynkiewicz, E.; Davis, R.E. Nematode m7GpppG and m3(2,2,7)GpppG decapping: Activities in Ascaris embryos and characterization of C. elegans scavenger DcpS. RNA 2004, 10, 1609–1624. [Google Scholar] [CrossRef]
- Davis, R.E.; Stretton, A.O. The motornervous system of Ascaris: Electrophysiology and anatomy of the neurons and their control by neuromodulators. Parasitology 1996, 113, S97–S117. [Google Scholar] [CrossRef] [PubMed]
- Stretton, A.O.; Maule, A.G. The Neurobiology of Ascaris and Other Parasitic Nematodes. Ascaris Negl. Parasite 2013, 127–152. [Google Scholar] [CrossRef]
- Rathbone, L. Oxidative metabolism in Ascaris lumbricoides from the pig. Biochem. J. 1955, 61, 574–579. [Google Scholar] [CrossRef] [PubMed]
- Beis, I.; Barrett, J. Energy metabolism in developing Ascaris lumbricoides eggs. II, The steady state content of intermediary metabolites. Dev. Biol. 1975, 42, 188–195. [Google Scholar] [CrossRef]
- Barrett, J.; Beis, I. Energy metabolism in developing Ascaris lumbricoides eggs. I. The glycolytic enzymes. Dev. Biol. 1975, 42, 181–187. [Google Scholar] [CrossRef]
- Tan, J.H.; Lautens, M.; Romanelli-Cedrez, L.; Wang, J.; Schertzberg, M.R.; Reinl, S.R.; Davis, R.E.; Shepherd, J.N.; Fraser, A.G.; Salinas, G. Alternative splicing of coq-2 controls the levels of rhodoquinone in animals. Elife 2020, 9. [Google Scholar] [CrossRef] [PubMed]
- Theriot, J.A. Worm sperm and advances in cell locomotion. Cell 1996, 84, 1–4. [Google Scholar] [CrossRef]
- Italiano, J.E., Jr.; Roberts, T.M.; Stewart, M.; Fontana, C.A. Reconstitution in vitro of the motile apparatus from the amoeboid sperm of Ascaris shows that filament assembly and bundling move membranes. Cell 1996, 84, 105–114. [Google Scholar] [CrossRef]
- Bottino, D.; Mogilner, A.; Roberts, T.; Stewart, M.; Oster, G. How nematode sperm crawl. J. Cell Sci. 2002, 115, 367–384. [Google Scholar] [PubMed]
- Roberts, T.M.; Stewart, M. Role of major sperm protein (MSP) in the protrusion and retraction of Ascaris sperm. Int. Rev. Cell Mol. Biol. 2012, 297, 265–293. [Google Scholar] [CrossRef]
- Cram, E.B. The egg-producing capacity of Ascaris lumbricoides. J. Agric. Res. 1925, 30, 977–983. [Google Scholar]
- Olsen, L.S.; Kelley, G.W.; Sen, H.G. Longevity and egg-production of Ascaris suum. Trans. Am. Microsc. Soc. 1958, 77, 380–383. [Google Scholar] [CrossRef]
- Wallace, A.; Filbin, M.E.; Veo, B.; McFarland, C.; Stepinski, J.; Jankowska-Anyszka, M.; Darzynkiewicz, E.; Davis, R.E. The nematode eukaryotic translation initiation factor 4E/G complex works with a trans-spliced leader stem-loop to enable efficient translation of trimethylguanosine-capped RNAs. Mol. Cell Biol. 2010, 30, 1958–1970. [Google Scholar] [CrossRef]
- Wang, J.; Czech, B.; Crunk, A.; Wallace, A.; Mitreva, M.; Hannon, G.J.; Davis, R.E. Deep small RNA sequencing from the nematode Ascaris reveals conservation, functional diversification, and novel developmental profiles. Genome Res. 2011, 21, 1462–1477. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Mitreva, M.; Berriman, M.; Thorne, A.; Magrini, V.; Koutsovoulos, G.; Kumar, S.; Blaxter, M.L.; Davis, R.E. Silencing of germline-expressed genes by DNA elimination in somatic cells. Dev. Cell 2012, 23, 1072–1080. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Garrey, J.; Davis, R.E. Transcription in pronuclei and one- to four-cell embryos drives early development in a nematode. Curr. Biol. 2014, 24, 124–133. [Google Scholar] [CrossRef]
- Kang, Y.; Wang, J.; Neff, A.; Kratzer, S.; Kimura, H.; Davis, R.E. Differential chromosomal localization of centromeric histone CENP-A contributes to nematode programmed DNA elimination. Cell Rep. 2016, 16, 2308–2316. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Gao, S.; Mostovoy, Y.; Kang, Y.; Zagoskin, M.; Sun, Y.; Zhang, B.; White, L.K.; Easton, A.; Nutman, T.B.; et al. Comparative genome analysis of programmed DNA elimination in nematodes. Genome Res. 2017, 27, 2001–2014. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Veronezi, G.M.B.; Kang, Y.; Zagoskin, M.; O’Toole, E.T.; Davis, R.E. Comprehensive Chromosome end remodeling during programmed DNA elimination. Curr. Biol. 2020, 30, 3397–3413.e4. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Davis, R.E. Contribution of transcription to animal early development. Transcription 2014, 5, e967602. [Google Scholar] [CrossRef]
- Shao, C.C.; Xu, M.J.; Alasaad, S.; Song, H.Q.; Peng, L.; Tao, J.P.; Zhu, X.Q. Comparative analysis of microRNA profiles between adult Ascaris lumbricoides and Ascaris suum. BMC Vet. Res. 2014, 10, 99. [Google Scholar] [CrossRef]
- Xu, M.J.; Fu, J.H.; Nisbet, A.J.; Huang, S.Y.; Zhou, D.H.; Lin, R.Q.; Song, H.Q.; Zhu, X.Q. Comparative profiling of microRNAs in male and female adults of Ascaris suum. Parasitol. Res. 2013, 112, 1189–1195. [Google Scholar] [CrossRef]
- Ma, G.; Luo, Y.; Zhu, H.; Luo, Y.; Korhonen, P.K.; Young, N.D.; Gasser, R.B.; Zhou, R. MicroRNAs of Toxocara canis and their predicted functional roles. Parasit. Vectors 2016, 9, 229. [Google Scholar] [CrossRef] [PubMed]
- Sarkies, P.; Selkirk, M.E.; Jones, J.T.; Blok, V.; Boothby, T.; Goldstein, B.; Hanelt, B.; Ardila-Garcia, A.; Fast, N.M.; Schiffer, P.M.; et al. Ancient and novel small RNA pathways compensate for the loss of piRNAs in multiple independent nematode lineages. PLoS Biol. 2015, 13, e1002061. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Tyagi, R.; Magrini, V.; Ly, A.; Jasmer, D.P.; Mitreva, M. Compartmentalization of functions and predicted miRNA regulation among contiguous regions of the nematode intestine. RNA Biol. 2017, 14, 1335–1352. [Google Scholar] [CrossRef] [PubMed]
- Jex, A.R.; Liu, S.; Li, B.; Young, N.D.; Hall, R.S.; Li, Y.; Yang, L.; Zeng, N.; Xu, X.; Xiong, Z.; et al. Ascaris suum draft genome. Nature 2011, 479, 529–533. [Google Scholar] [CrossRef] [PubMed]
- IHG Consortium. Comparative genomics of the major parasitic worms. Nat. Genet. 2019, 51, 163–174. [Google Scholar] [CrossRef] [PubMed]
- Stroehlein, A.J.; Young, N.D.; Gasser, R.B. Advances in kinome research of parasitic worms-implications for fundamental research and applied biotechnological outcomes. Biotechnol. Adv. 2018, 36, 915–934. [Google Scholar] [CrossRef]
- Zhu, X.Q.; Korhonen, P.K.; Cai, H.; Young, N.D.; Nejsum, P.; von Samson-Himmelstjerna, G.; Boag, P.R.; Tan, P.; Li, Q.; Min, J.; et al. Genetic blueprint of the zoonotic pathogen Toxocara canis. Nat. Commun. 2015, 6, 6145. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Yu, L.; Fan, H.; Huang, G.; Wu, Q.; Nie, Y.; Liu, S.; Yan, L.; Wei, F. Genomic signatures of coevolution between non-model mammals and parasitic roundworms. Mol. Biol. Evol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.B.; Zou, Y.; Zhu, X.Q.; Liu, G.H. Toxocara “omics” and the promises it holds for medicine and veterinary medicine. Adv. Parasitol. 2020, 109, 89–108. [Google Scholar] [CrossRef]
- Hansen, E.P.; Fromm, B.; Andersen, S.D.; Marcilla, A.; Andersen, K.L.; Borup, A.; Williams, A.R.; Jex, A.R.; Gasser, R.B.; Young, N.D.; et al. Exploration of extracellular vesicles from Ascaris suum provides evidence of parasite-host cross talk. J. Extracell. Vesicles 2019, 8, 1578116. [Google Scholar] [CrossRef] [PubMed]
- Gerhard, A.P.; Krucken, J.; Heitlinger, E.; Janssen, I.J.I.; Basiaga, M.; Kornas, S.; Beier, C.; Nielsen, M.K.; Davis, R.E.; Wang, J.; et al. The P-glycoprotein repertoire of the equine parasitic nematode Parascaris univalens. Sci. Rep. 2020, 10, 13586. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, M.K.; Wang, J.; Davis, R.; Bellaw, J.L.; Lyons, E.T.; Lear, T.L.; Goday, C. Parascaris univalens—A victim of large-scale misidentification? Parasitol. Res. 2014, 113, 4485–4490. [Google Scholar] [CrossRef]
- Pilotte, N.; Maasch, J.; Easton, A.V.; Dahlstrom, E.; Nutman, T.B.; Williams, S.A. Targeting a highly repeated germline DNA sequence for improved real-time PCR-based detection of Ascaris infection in human stool. PLoS Negl. Trop. Dis. 2019, 13, e0007593. [Google Scholar] [CrossRef] [PubMed]
- Tobler, H.; Smith, K.D.; Ursprung, H. Molecular aspects of chromatin elimination in Ascaris lumbricoides. Dev. Biol. 1972, 27, 190–203. [Google Scholar] [CrossRef]
- Moritz, K.B.; Roth, G.E. Complexity of germline and somatic DNA in Ascaris. Nature 1976, 259, 55–57. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, P.; Straus, N.A. Molecular characterization of Ascaris suum DNA and of chromatin diminution. Exp. Cell Res. 1978, 116, 462–466. [Google Scholar] [CrossRef]
- Roth, G.E.; Moritz, K.B. Restriction enzyme analysis of the germ line limited DNA of Ascaris suum. Chromosoma 1981, 83, 169–190. [Google Scholar] [CrossRef] [PubMed]
- Muller, F.; Walker, P.; Aeby, P.; Neuhaus, H.; Back, E.; Tobler, H. Molecular cloning and sequence analysis of highly repetitive DNA sequences contained in the eliminated genome of Ascaris lumbricoides. Prog. Clin. Biol. Res. 1982, 85 Pt A, 127–138. [Google Scholar]
- Muller, F.; Walker, P.; Aeby, P.; Neuhaus, H.; Felder, H.; Back, E.; Tobler, H. Nucleotide sequence of satellite DNA contained in the eliminated genome of Ascaris lumbricoides. Nucleic Acids Res. 1982, 10, 7493–7510. [Google Scholar] [CrossRef]
- Aeby, P.; Spicher, A.; de Chastonay, Y.; Muller, F.; Tobler, H. Structure and genomic organization of proretrovirus-like elements partially eliminated from the somatic genome of Ascaris lumbricoides. EMBO J. 1986, 5, 3353–3360. [Google Scholar] [CrossRef] [PubMed]
- Etter, A.; Aboutanos, M.; Tobler, H.; Muller, F. Eliminated chromatin of Ascaris contains a gene that encodes a putative ribosomal protein. Proc. Natl. Acad. Sci. USA 1991, 88, 1593–1596. [Google Scholar] [CrossRef]
- Spicher, A.; Etter, A.; Bernard, V.; Tobler, H.; Muller, F. Extremely stable transcripts may compensate for the elimination of the gene fert-1 from all Ascaris lumbricoides somatic cells. Dev. Biol. 1994, 164, 72–86. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.J.; Stoffel, R.; Tobler, H.; Mueller, F. A newly formed telomere in Ascaris suum does not exert a telomere position effect on a nearby gene. Mol. Cell. Biol. 1996, 16, 130–134. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bachmann-Waldmann, C.; Jentsch, S.; Tobler, H.; Muller, F. Chromatin diminution leads to rapid evolutionary changes in the organization of the germ line genomes of the parasitic nematodes A. suum and P. univalens. Mol. Biochem. Parasitol. 2004, 134, 53–64. [Google Scholar] [CrossRef]
- Jentsch, S.; Tobler, H.; Muller, F. New telomere formation during the process of chromatin diminution in Ascaris suum. Int. J. Dev. Biol. 2002, 46, 143–148. [Google Scholar] [PubMed]
- Muller, F.; Wicky, C.; Spicher, A.; Tobler, H. New telomere formation after developmentally regulated chromosomal breakage during the process of chromatin diminution in Ascaris lumbricoides. Cell 1991, 67, 815–822. [Google Scholar] [CrossRef]
- Gardner, M.J.; Hall, N.; Fung, E.; White, O.; Berriman, M.; Hyman, R.W.; Carlton, J.M.; Pain, A.; Nelson, K.E.; Bowman, S.; et al. Genome sequence of the human malaria parasite Plasmodium falciparum. Nature 2002, 419, 498–511. [Google Scholar] [CrossRef] [PubMed]
- Jones, T.; Federspiel, N.A.; Chibana, H.; Dungan, J.; Kalman, S.; Magee, B.B.; Newport, G.; Thorstenson, Y.R.; Agabian, N.; Magee, P.T.; et al. The diploid genome sequence of Candida albicans. Proc. Natl. Acad. Sci. USA 2004, 101, 7329–7334. [Google Scholar] [CrossRef]
- Nierman, W.C.; Pain, A.; Anderson, M.J.; Wortman, J.R.; Kim, H.S.; Arroyo, J.; Berriman, M.; Abe, K.; Archer, D.B.; Bermejo, C.; et al. Genomic sequence of the pathogenic and allergenic filamentous fungus Aspergillus fumigatus. Nature 2005, 438, 1151–1156. [Google Scholar] [CrossRef]
- Loftus, B.; Anderson, I.; Davies, R.; Alsmark, U.C.; Samuelson, J.; Amedeo, P.; Roncaglia, P.; Berriman, M.; Hirt, R.P.; Mann, B.J.; et al. The genome of the protist parasite Entamoeba histolytica. Nature 2005, 433, 865–868. [Google Scholar] [CrossRef] [PubMed]
- Ivens, A.C.; Peacock, C.S.; Worthey, E.A.; Murphy, L.; Aggarwal, G.; Berriman, M.; Sisk, E.; Rajandream, M.A.; Adlem, E.; Aert, R.; et al. The genome of the kinetoplastid parasite, Leishmania major. Science 2005, 309, 436–442. [Google Scholar] [CrossRef] [PubMed]
- Berriman, M.; Ghedin, E.; Hertz-Fowler, C.; Blandin, G.; Renauld, H.; Bartholomeu, D.C.; Lennard, N.J.; Caler, E.; Hamlin, N.E.; Haas, B.; et al. The genome of the African trypanosome Trypanosoma brucei. Science 2005, 309, 416–422. [Google Scholar] [CrossRef]
- Ghedin, E.; Wang, S.; Spiro, D.; Caler, E.; Zhao, Q.; Crabtree, J.; Allen, J.E.; Delcher, A.L.; Guiliano, D.B.; Miranda-Saavedra, D.; et al. Draft genome of the filarial nematode parasite Brugia malayi. Science 2007, 317, 1756–1760. [Google Scholar] [CrossRef]
- Berriman, M.; Haas, B.J.; LoVerde, P.T.; Wilson, R.A.; Dillon, G.P.; Cerqueira, G.C.; Mashiyama, S.T.; Al-Lazikani, B.; Andrade, L.F.; Ashton, P.D.; et al. The genome of the blood fluke Schistosoma mansoni. Nature 2009, 460, 352–358. [Google Scholar] [CrossRef] [PubMed]
- Mitreva, M.; Jasmer, D.P.; Zarlenga, D.S.; Wang, Z.; Abubucker, S.; Martin, J.; Taylor, C.M.; Yin, Y.; Fulton, L.; Minx, P.; et al. The draft genome of the parasitic nematode Trichinella spiralis. Nat. Genet. 2011, 43, 228–235. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez de la Rosa, P.M.; Thomson, M.; Trivedi, U.; Tracey, A.; Tandonnet, S.; Blaxter, M. A telomere-to-telomere assembly of Oscheius tipulae and the evolution of rhabditid nematode chromosomes. G3 2021, 11. [Google Scholar] [CrossRef]
- Gruetzner, F.; Ashley, T.; Rowell, D.M.; Marshall Graves, J.A. How did the platypus get its sex chromosome chain? A comparison of meiotic multiples and sex chromosomes in plants and animals. Chromosoma 2006, 115, 75–88. [Google Scholar] [CrossRef]
- Galian, J.; Hogan, J.E.; Vogler, A.P. The origin of multiple sex chromosomes in tiger beetles. Mol. Biol. Evol. 2002, 19, 1792–1796. [Google Scholar] [CrossRef][Green Version]
- Zhou, Y.; Shearwin-Whyatt, L.; Li, J.; Song, Z.; Hayakawa, T.; Stevens, D.; Fenelon, J.C.; Peel, E.; Cheng, Y.; Pajpach, F.; et al. Platypus and echidna genomes reveal mammalian biology and evolution. Nature 2021. [Google Scholar] [CrossRef] [PubMed]
- Grutzner, F.; Rens, W.; Tsend-Ayush, E.; El-Mogharbel, N.; O’Brien, P.C.; Jones, R.C.; Ferguson-Smith, M.A.; Marshall Graves, J.A. In the platypus a meiotic chain of ten sex chromosomes shares genes with the bird Z and mammal X chromosomes. Nature 2004, 432, 913–917. [Google Scholar] [CrossRef] [PubMed]
- Allen, M.A.; Hillier, L.W.; Waterston, R.H.; Blumenthal, T. A global analysis of C. elegans trans-splicing. Genome Res. 2011, 21, 255–264. [Google Scholar] [CrossRef]
- Blumenthal, T. Trans-splicing and operons in C. elegans. WormBook 2012, 1–11. [Google Scholar] [CrossRef]
- Ingolia, N.T. Ribosome profiling: New views of translation, from single codons to genome scale. Nat. Rev. Genet. 2014, 15, 205–213. [Google Scholar] [CrossRef] [PubMed]
- Vastenhouw, N.L.; Cao, W.X.; Lipshitz, H.D. The maternal-to-zygotic transition revisited. Development 2019, 146. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.T.; Bonneau, A.R.; Giraldez, A.J. Zygotic genome activation during the maternal-to-zygotic transition. Annu Rev. Cell Dev. Biol. 2014, 30, 581–613. [Google Scholar] [CrossRef]
- Tadros, W.; Lipshitz, H.D. The maternal-to-zygotic transition: A play in two acts. Development 2009, 136, 3033–3042. [Google Scholar] [CrossRef] [PubMed]
- Rosa, B.A.; Jasmer, D.P.; Mitreva, M. Genome-wide tissue-specific gene expression, co-expression and regulation of co-expressed genes in adult nematode Ascaris suum. PLoS Negl. Trop. Dis. 2014, 8, e2678. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Gao, X.; Martin, J.; Yin, Y.; Abubucker, S.; Rash, A.C.; Li, B.W.; Nash, B.; Hallsworth-Pepin, K.; Jasmer, D.P.; et al. Gene expression analysis distinguishes tissue-specific and gender-related functions among adult Ascaris suum tissues. Mol. Genet. Genom. 2013, 288, 243–260. [Google Scholar] [CrossRef] [PubMed]
- Jasmer, D.P.; Rosa, B.A.; Tyagi, R.; Mitreva, M. Omics driven understanding of the intestines of parasitic nematodes. Front. Genet. 2019, 10, 652. [Google Scholar] [CrossRef] [PubMed]
- Rosa, B.A.; Townsend, R.; Jasmer, D.P.; Mitreva, M. Functional and phylogenetic characterization of proteins detected in various nematode intestinal compartments. Mol. Cell Proteom. 2015, 14, 812–827. [Google Scholar] [CrossRef] [PubMed]
- Rosa, B.A.; McNulty, S.N.; Mitreva, M.; Jasmer, D.P. Direct experimental manipulation of intestinal cells in Ascaris suum, with minor influences on the global transcriptome. Int. J. Parasitol. 2017, 47, 271–279. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Jasmer, D.P.; Rosa, B.A.; Tyagi, R.; Bulman, C.A.; Beerntsen, B.; Urban, J.F., Jr.; Sakanari, J.; Mitreva, M. De novo identification of toxicants that cause irreparable damage to parasitic nematode intestinal cells. PLoS Negl. Trop. Dis. 2020, 14, e0007942. [Google Scholar] [CrossRef] [PubMed]
- Almeida, M.V.; Andrade-Navarro, M.A.; Ketting, R.F. Function and evolution of nematode RNAi pathways. Noncoding RNA 2019, 5, 8. [Google Scholar] [CrossRef]
- Billi, A.C.; Fischer, S.E.; Kim, J.K. Endogenous RNAi pathways in C. elegans. WormBook 2014, 1–49. [Google Scholar] [CrossRef]
- Buck, A.H.; Blaxter, M. Functional diversification of Argonautes in nematodes: An expanding universe. Biochem. Soc. Trans. 2013, 41, 881–886. [Google Scholar] [CrossRef]
- Buck, A.H.; Coakley, G.; Simbari, F.; McSorley, H.J.; Quintana, J.F.; Le Bihan, T.; Kumar, S.; Abreu-Goodger, C.; Lear, M.; Harcus, Y.; et al. Exosomes secreted by nematode parasites transfer small RNAs to mammalian cells and modulate innate immunity. Nat. Commun. 2014, 5, 5488. [Google Scholar] [CrossRef]
- Chow, F.W.; Koutsovoulos, G.; Ovando-Vazquez, C.; Neophytou, K.; Bermudez-Barrientos, J.R.; Laetsch, D.R.; Robertson, E.; Kumar, S.; Claycomb, J.M.; Blaxter, M.; et al. Secretion of an Argonaute protein by a parasitic nematode and the evolution of its siRNA guides. Nucleic Acids Res. 2019, 47, 3594–3606. [Google Scholar] [CrossRef]
- Jenuwein, T.; Allis, C.D. Translating the histone code. Science 2001, 293, 1074–1080. [Google Scholar] [CrossRef]
- Buenrostro, J.D.; Wu, B.; Chang, H.Y.; Greenleaf, W.J. ATAC-seq: A method for assaying chromatin accessibility genome-wide. Curr. Protoc. Mol. Biol. 2015, 109, 21–29. [Google Scholar] [CrossRef]
- Fukagawa, T.; Earnshaw, W.C. The centromere: Chromatin foundation for the kinetochore machinery. Dev. Cell 2014, 30, 496–508. [Google Scholar] [CrossRef]
- Black, B.E.; Cleveland, D.W. Epigenetic centromere propagation and the nature of CENP-a nucleosomes. Cell 2011, 144, 471–479. [Google Scholar] [CrossRef]
- Allshire, R.C.; Karpen, G.H. Epigenetic regulation of centromeric chromatin: Old dogs, new tricks? Nat. Rev. Genet. 2008, 9, 923–937. [Google Scholar] [CrossRef] [PubMed]
- Buchwitz, B.J.; Ahmad, K.; Moore, L.L.; Roth, M.B.; Henikoff, S. A histone-H3-like protein in C. elegans. Nature 1999, 401, 547–548. [Google Scholar] [CrossRef] [PubMed]
- Gassmann, R.; Rechtsteiner, A.; Yuen, K.W.; Muroyama, A.; Egelhofer, T.; Gaydos, L.; Barron, F.; Maddox, P.; Essex, A.; Monen, J.; et al. An inverse relationship to germline transcription defines centromeric chromatin in C. elegans. Nature 2012, 484, 534–537. [Google Scholar] [CrossRef] [PubMed]
- Criscione, C.D.; Anderson, J.D.; Sudimack, D.; Peng, W.; Jha, B.; Williams-Blangero, S.; Anderson, T.J. Disentangling hybridization and host colonization in parasitic roundworms of humans and pigs. Proc. Biol. Sci. 2007, 274, 2669–2677. [Google Scholar] [CrossRef]
- Peng, W.; Criscione, C.D. Ascariasis in people and pigs: New inferences from DNA analysis of worm populations. Infect. Genet. Evol. 2012, 12, 227–235. [Google Scholar] [CrossRef] [PubMed]
- Peng, W.; Anderson, T.J.; Zhou, X.; Kennedy, M.W. Genetic variation in sympatric Ascaris populations from humans and pigs in China. Parasitology 1998, 117 Pt 4, 355–361. [Google Scholar] [CrossRef]
- Zhou, C.; Li, M.; Yuan, K.; Hu, N.; Peng, W. Phylogeography of Ascaris lumbricoides and A. suum from China. Parasitol. Res. 2011, 109, 329–338. [Google Scholar] [CrossRef]
- Cavallero, S.; Snabel, V.; Pacella, F.; Perrone, V.; D’Amelio, S. Phylogeographical studies of Ascaris spp. based on ribosomal and mitochondrial DNA sequences. PLoS Negl. Trop. Dis. 2013, 7, e2170. [Google Scholar] [CrossRef]
- Nejsum, P.; Hawash, M.B.; Betson, M.; Stothard, J.R.; Gasser, R.B.; Andersen, L.O. Ascaris phylogeny based on multiple whole mtDNA genomes. Infect. Genet. Evol. 2017, 48, 4–9. [Google Scholar] [CrossRef] [PubMed]
- Levine, M.T.; Jones, C.D.; Kern, A.D.; Lindfors, H.A.; Begun, D.J. Novel genes derived from noncoding DNA in Drosophila melanogaster are frequently X-linked and exhibit testis-biased expression. Proc. Natl. Acad. Sci. USA 2006, 103, 9935–9939. [Google Scholar] [CrossRef] [PubMed]
- Van Oss, S.B.; Carvunis, A.R. De novo gene birth. PLoS Genet. 2019, 15, e1008160. [Google Scholar] [CrossRef] [PubMed]
- Kaessmann, H. Origins, evolution, and phenotypic impact of new genes. Genome Res. 2010, 20, 1313–1326. [Google Scholar] [CrossRef]
- Rodelsperger, C.; Ebbing, A.; Sharma, D.R.; Okumura, M.; Sommer, R.J.; Korswagen, H.C. Spatial transcriptomics of nematodes identifies sperm cells as a source of genomic novelty and rapid evolution. Mol. Biol. Evol. 2021, 38, 229–243. [Google Scholar] [CrossRef]
- Lopienska-Biernat, E.; Paukszto, L.; Jastrzebski, J.P.; Myszczynski, K.; Polak, I.; Stryinski, R. Genome-wide analysis of Anisakis simplex sensu lato: The role of carbohydrate metabolism genes in the parasite’s development. Int. J. Parasitol. 2019, 49, 933–943. [Google Scholar] [CrossRef]
- D’Amelio, S.; Lombardo, F.; Pizzarelli, A.; Bellini, I.; Cavallero, S. Advances in omic studies drive discoveries in the biology of Anisakid nematodes. Genes 2020, 11, 801. [Google Scholar] [CrossRef]
- Wang, J. Genome analysis of programmed DNA elimination in parasitic nematodes. Methods Mol. Biol. 2021, in press. [Google Scholar]
- Camp, J.G.; Platt, R.; Treutlein, B. Mapping human cell phenotypes to genotypes with single-cell genomics. Science 2019, 365, 1401–1405. [Google Scholar] [CrossRef]
- Tanay, A.; Regev, A. Scaling single-cell genomics from phenomenology to mechanism. Nature 2017, 541, 331–338. [Google Scholar] [CrossRef]
- Macaulay, I.C.; Voet, T. Single cell genomics: Advances and future perspectives. PLoS Genet. 2014, 10, e1004126. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Daugharthy, E.R.; Scheiman, J.; Kalhor, R.; Yang, J.L.; Ferrante, T.C.; Terry, R.; Jeanty, S.S.; Li, C.; Amamoto, R.; et al. Highly multiplexed subcellular RNA sequencing in situ. Science 2014, 343, 1360–1363. [Google Scholar] [CrossRef] [PubMed]
- Payne, A.C.; Chiang, Z.D.; Reginato, P.L.; Mangiameli, S.M.; Murray, E.M.; Yao, C.C.; Markoulaki, S.; Earl, A.S.; Labade, A.S.; Jaenisch, R.; et al. In situ genome sequencing resolves DNA sequence and structure in intact biological samples. Science 2020. [Google Scholar] [CrossRef]
- Howe, K.L.; Bolt, B.J.; Shafie, M.; Kersey, P.; Berriman, M. WormBase ParaSite—A comprehensive resource for helminth genomics. Mol. Biochem. Parasitol. 2017, 215, 2–10. [Google Scholar] [CrossRef] [PubMed]
- Wong, N.; John, S.; Nussenzweig, A.; Canela, A. END-seq: An unbiased, high-resolution, and genome-wide approach to map DNA double-strand breaks and resection in human cells. Methods Mol. Biol. 2021, 2153, 9–31. [Google Scholar] [CrossRef]
- Santos-Pereira, J.M.; Aguilera, A. R loops: New modulators of genome dynamics and function. Nat. Rev. Genet. 2015, 16, 583–597. [Google Scholar] [CrossRef]
- Aguilera, A.; Garcia-Muse, T. R loops: From transcription byproducts to threats to genome stability. Mol. Cell 2012, 46, 115–124. [Google Scholar] [CrossRef]
- Anderson, T.J.C.; LoVerde, P.T.; Le Clec’h, W.; Chevalier, F.D. Genetic crosses and linkage mapping in schistosome parasites. Trends Parasitol. 2018, 34, 982–996. [Google Scholar] [CrossRef] [PubMed]
- Bennuru, S.; O’Connell, E.M.; Drame, P.M.; Nutman, T.B. Mining filarial genomes for diagnostic and therapeutic targets. Trends Parasitol. 2018, 34, 80–90. [Google Scholar] [CrossRef]
- Doyle, S.R.; Cotton, J.A. Genome-wide approaches to investigate anthelmintic resistance. Trends Parasitol. 2019, 35, 289–301. [Google Scholar] [CrossRef]
- Wit, J.; Dilks, C.M.; Andersen, E.C. Complementary approaches with free-living and parasitic nematodes to understanding anthelmintic resistance. Trends Parasitol. 2021, 37, 240–250. [Google Scholar] [CrossRef] [PubMed]
- Adugna, S.; Kebede, Y.; Moges, F.; Tiruneh, M. Efficacy of mebendazole and albendazole for Ascaris lumbricoides and hookworm infections in an area with long time exposure for antihelminthes, Northwest Ethiopia. Ethiop. Med. J. 2007, 45, 301–306. [Google Scholar] [PubMed]
- Matthews, J.B. Anthelmintic resistance in equine nematodes. Int. J. Parasitol. Drugs Drug Resist. 2014, 4, 310–315. [Google Scholar] [CrossRef]
- Vercruysse, J.; Albonico, M.; Behnke, J.M.; Kotze, A.C.; Prichard, R.K.; McCarthy, J.S.; Montresor, A.; Levecke, B. Is anthelmintic resistance a concern for the control of human soil-transmitted helminths? Int. J. Parasitol. Drugs Drug Resist. 2011, 1, 14–27. [Google Scholar] [CrossRef]
- Jia, T.W.; Melville, S.; Utzinger, J.; King, C.H.; Zhou, X.N. Soil-transmitted helminth reinfection after drug treatment: A systematic review and meta-analysis. PLoS Negl. Trop. Dis. 2012, 6, e1621. [Google Scholar] [CrossRef]
- Hawdon, J.M. Controlling soil-transmitted helminths: Time to think inside the box? J. Parasitol. 2014, 100, 166–188. [Google Scholar] [CrossRef] [PubMed]
- Raney, B.J.; Dreszer, T.R.; Barber, G.P.; Clawson, H.; Fujita, P.A.; Wang, T.; Nguyen, N.; Paten, B.; Zweig, A.S.; Karolchik, D.; et al. Track data hubs enable visualization of user-defined genome-wide annotations on the UCSC Genome Browser. Bioinformatics 2014, 30, 1003–1005. [Google Scholar] [CrossRef]

| Features | Ascaris suum | Ascaris lumbricoides | Parascaris univalens | Toxocara canis | Baylisascaris schroederi | Baylisascaris ailuri | Toxascaris leonina |
|---|---|---|---|---|---|---|---|
| Major host | pig | human | horse | dog | giant panda | red panda | lion |
| Assembled bases (Mb) | 279 | 296 | 253 | 317 | 282 | 267 | 285 |
| N50 (kb) | 12,191 | 4633 | 1826 | 375 | 889 | 51 | 36 |
| Scaffold number | 109 * | 415 | 1274 | 22,857 | 2834 | 30,943 | 49,543 |
| Largest scaffold (Mb) | 23.1 | 13.2 | 5.6 | 1.9 | 5.8 | 0.5 | 0.4 |
| Protein-coding genes | 15,714 | 17,902 | 15,027 | 18,596 | 13,284 | 12,252 | 16,087 |
| Accession number | JACCHR01 | SMSY01 | NJFU01 | JPKZ01 | NA | NA | NA |
| Major technologies used | PacBio, Hi-C | Illumina, PacBio | Illumina, PacBio, BioNano | Illumina | Illumina, PacBio | Illumina | Illumina |
| Reference | Wang et al. 2020 Curr. Biol. [39] | Easton et al. 2020 eLife [11] | Wang et al. 2017 Genome Res. [38] | Zhu et al. 2015 Nat. Commun. [49] | Hu et al. 2020 Mol. Biol. Evol. [50] | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, J. Genomics of the Parasitic Nematode Ascaris and Its Relatives. Genes 2021, 12, 493. https://doi.org/10.3390/genes12040493
Wang J. Genomics of the Parasitic Nematode Ascaris and Its Relatives. Genes. 2021; 12(4):493. https://doi.org/10.3390/genes12040493
Chicago/Turabian StyleWang, Jianbin. 2021. "Genomics of the Parasitic Nematode Ascaris and Its Relatives" Genes 12, no. 4: 493. https://doi.org/10.3390/genes12040493
APA StyleWang, J. (2021). Genomics of the Parasitic Nematode Ascaris and Its Relatives. Genes, 12(4), 493. https://doi.org/10.3390/genes12040493





