t(8;21) Acute Myeloid Leukemia as a Paradigm for the Understanding of Leukemogenesis at the Level of Gene Regulation and Chromatin Programming
Abstract
1. Overview
2. What Is AML
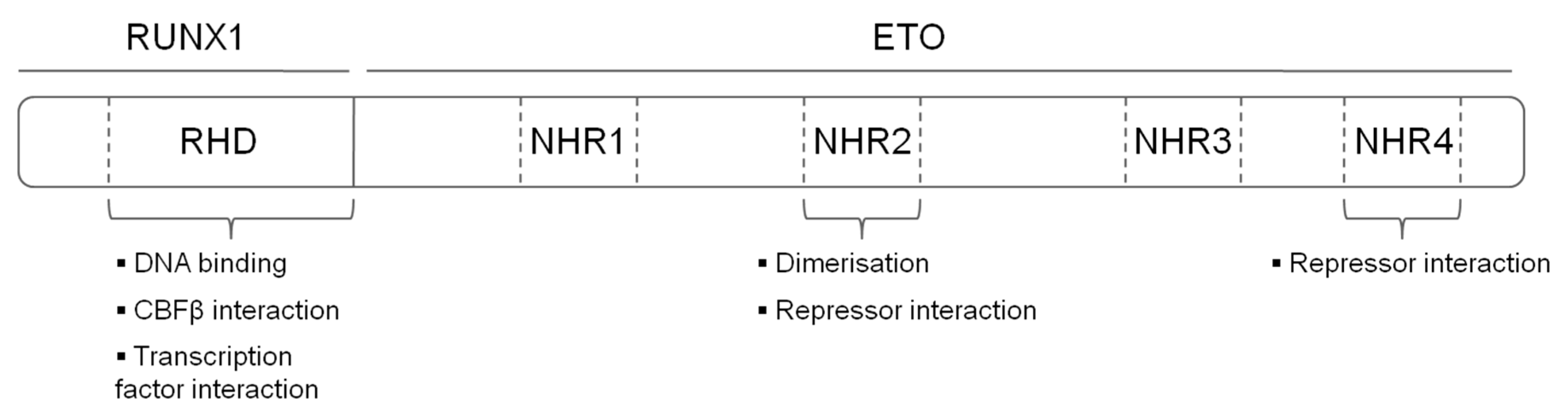
3. The Chromosomal Rearrangement t(8;21) Gives Rise to a Fusion Protein
4. Murine Model Systems Studying t(8;21) AML—RUNX1/ETO cannot Do It Alone
5. Molecular Aspects of RUNX1/ETO Function
6. Concurrent Mutations Add another Layer of Complexity to the RUNX1/ETO Cellular Network
7. System Wide Studies on Aberrant Chromatin Programming by RUNX1/ETO
8. Therapeutic Targeting of RUNX1/ETO
9. Outlook/Vision
Author Contributions
Funding
Conflicts of Interest
References
- Mrózek, K.; Bloomfield, C.D. Clinical significance of the most common chromosome translocations in adult acute myeloid leukemia. J. Natl. Cancer Inst. Monogr. 2008, 2008, 52–57. [Google Scholar] [CrossRef] [PubMed]
- Wiemels, J. Chromosomal translocations in childhood leukemia: Natural history, mechanisms, and epidemiology. J. Natl. Cancer Inst. Monogr. 2008, 2008, 87–90. [Google Scholar] [CrossRef] [PubMed]
- Speck, N.A.; Stacy, T.; Wang, Q.; North, T.; Gu, T.-L.; Miller, J.; Binder, M.; Marin-Padilla, M. Core-binding factor: A central player in hematopoiesis and leukemia. Cancer Res. 1999, 59, 1789s–1793s. [Google Scholar] [PubMed]
- Miyoshi, H.; Shimizu, K.; Kozu, T.; Maseki, N.; Kaneko, Y.; Ohki, M. t (8; 21) breakpoints on chromosome 21 in acute myeloid leukemia are clustered within a limited region of a single gene, AML1. Proc. Natl. Acad. Sci. USA 1991, 88, 10431–10434. [Google Scholar] [CrossRef] [PubMed]
- de Bruijn, M.F.; Speck, N.A. Core-binding factors in hematopoiesis and immune function. Oncogene 2004, 23, 4238–4248. [Google Scholar] [CrossRef]
- Sood, R.; Kamikubo, Y.; Liu, P. Role of RUNX1 in hematological malignancies. Blood J. Am. Soc. Hematol. 2017, 129, 2070–2082. [Google Scholar] [CrossRef] [PubMed]
- Solh, M.; Yohe, S.; Weisdorf, D.; Ustun, C. Core-binding factor acute myeloid leukemia: Heterogeneity, monitoring, and therapy. Am. J. Hematol. 2014, 89, 1121–1131. [Google Scholar] [CrossRef]
- Yzaguirre, A.D.; de Bruijn, M.F.; Speck, N.A. The role of Runx1 in embryonic blood cell formation. In RUNX Proteins in Development and Cancer; Springer: Berlin/Heidelberg, Germany, 2017; pp. 47–64. [Google Scholar]
- Link, K.A.; Chou, F.S.; Mulloy, J.C. Core binding factor at the crossroads: Determining the fate of the HSC. J. Cell. Physiol. 2010, 222, 50–56. [Google Scholar] [CrossRef]
- Odaka, Y.; Mally, A.; Elliott, L.T.; Meyers, S. Nuclear import and subnuclear localization of the proto-oncoprotein ETO (MTG8). Oncogene 2000, 19, 3584–3597. [Google Scholar] [CrossRef]
- Hug, B.A.; Lazar, M.A. ETO interacting proteins. Oncogene 2004, 23, 4270–4274. [Google Scholar] [CrossRef]
- Calabi, F.; Pannell, R.; Pavloska, G. Gene Targeting Reveals a Crucial Role forMTG8 in the Gut. Mol. Cell. Biol. 2001, 21, 5658–5666. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.-J.; Wang, Z.; Wang, L.; Jiang, Y.; Kost, N.; Soong, T.D.; Chen, W.-Y.; Tang, Z.; Nakadai, T.; Elemento, O. A stable transcription factor complex nucleated by oligomeric AML1–ETO controls leukaemogenesis. Nature 2013, 500, 93–97. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Guo, C.; Steinauer, N.; Zhang, J. New insights into transcriptional and leukemogenic mechanisms of AML1-ETO and E2A fusion proteins. Front. Biol. 2016, 11, 285–304. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lutterbach, B.; Westendorf, J.J.; Linggi, B.; Patten, A.; Moniwa, M.; Davie, J.R.; Huynh, K.D.; Bardwell, V.J.; Lavinsky, R.M.; Rosenfeld, M.G. ETO, a target of t (8; 21) in acute leukemia, interacts with the N-CoR and mSin3 corepressors. Mol. Cell. Biol. 1998, 18, 7176–7184. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Cheney, M.D.; Gaudet, J.J.; Chruszcz, M.; Lukasik, S.M.; Sugiyama, D.; Lary, J.; Cole, J.; Dauter, Z.; Minor, W. The tetramer structure of the Nervy homology two domain, NHR2, is critical for AML1/ETO’s activity. Cancer Cell 2006, 9, 249–260. [Google Scholar] [CrossRef] [PubMed]
- Wichmann, C.; Becker, Y.; Chen-Wichmann, L.; Vogel, V.; Vojtkova, A.; Herglotz, J.; Moore, S.; Koch, J.; Lausen, J.; Mäntele, W. Dimer-tetramer transition controls RUNX1/ETO leukemogenic activity. Blood J. Am. Soc. Hematol. 2010, 116, 603–613. [Google Scholar] [CrossRef]
- Hildebrand, D.; Tiefenbach, J.; Heinzel, T.; Grez, M.; Maurer, A.B. Multiple regions of ETO cooperate in transcriptional repression. J. Biol. Chem. 2001, 276, 9889–9895. [Google Scholar] [CrossRef]
- Ahn, E.-Y.; Yan, M.; Malakhova, O.A.; Lo, M.-C.; Boyapati, A.; Ommen, H.B.; Hines, R.; Hokland, P.; Zhang, D.-E. Disruption of the NHR4 domain structure in AML1-ETO abrogates SON binding and promotes leukemogenesis. Proc. Natl. Acad. Sci. USA 2008, 105, 17103–17108. [Google Scholar] [CrossRef]
- Okuda, T.; Cai, Z.; Yang, S.; Lenny, N.; Lyu, C.-j.; van Deursen, J.M.; Harada, H.; Downing, J.R. Expression of a knocked-in AML1-ETO leukemia gene inhibits the establishment of normal definitive hematopoiesis and directly generates dysplastic hematopoietic progenitors. Blood J. Am. Soc. Hematol. 1998, 91, 3134–3143. [Google Scholar]
- Okuda, T.; Van Deursen, J.; Hiebert, S.W.; Grosveld, G.; Downing, J.R. AML1, the target of multiple chromosomal translocations in human leukemia, is essential for normal fetal liver hematopoiesis. Cell 1996, 84, 321–330. [Google Scholar] [CrossRef]
- Yergeau, D.A.; Hetherington, C.J.; Wang, Q.; Zhang, P.; Sharpe, A.H.; Binder, M.; Marín-Padilla, M.; Tenen, D.G.; Speck, N.A.; Zhang, D.-E. Embryonic lethality and impairment of haematopoiesis in mice heterozygous for an AML1-ETO fusion gene. Nat. Genet. 1997, 15, 303–306. [Google Scholar] [CrossRef] [PubMed]
- Buchholz, F.; Refaeli, Y.; Trumpp, A.; Bishop, J.M. Inducible chromosomal translocation of AML1 and ETO genes through Cre/loxP-mediated recombination in the mouse. EMBO Rep. 2000, 1, 133–139. [Google Scholar] [CrossRef] [PubMed]
- Higuchi, M.; O’Brien, D.; Kumaravelu, P.; Lenny, N.; Yeoh, E.-J.; Downing, J.R. Expression of a conditional AML1-ETO oncogene bypasses embryonic lethality and establishes a murine model of human t (8; 21) acute myeloid leukemia. Cancer Cell 2002, 1, 63–74. [Google Scholar] [CrossRef]
- Rhoades, K.L.; Hetherington, C.J.; Harakawa, N.; Yergeau, D.A.; Zhou, L.; Liu, L.-Q.; Little, M.-T.; Tenen, D.G.; Zhang, D.-E. Analysis of the role of AML1-ETO in leukemogenesis, using an inducible transgenic mouse model. Blood J. Am. Soc. Hematol. 2000, 96, 2108–2115. [Google Scholar]
- Cabezas-Wallscheid, N.; Eichwald, V.; de Graaf, J.; Löwer, M.; Lehr, H.A.; Kreft, A.; Eshkind, L.; Hildebrandt, A.; Abassi, Y.; Heck, R. Instruction of haematopoietic lineage choices, evolution of transcriptional landscapes and cancer stem cell hierarchies derived from an AML1-ETO mouse model. EMBO Mol. Med. 2013, 5, 1804–1820. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Y.; Zhou, L.; Miyamoto, T.; Iwasaki, H.; Harakawa, N.; Hetherington, C.J.; Burel, S.A.; Lagasse, E.; Weissman, I.L.; Akashi, K. AML1-ETO expression is directly involved in the development of acute myeloid leukemia in the presence of additional mutations. Proc. Natl. Acad. Sci. USA 2001, 98, 10398–10403. [Google Scholar] [CrossRef] [PubMed]
- Wiemels, J.L.; Xiao, Z.; Buffler, P.A.; Maia, A.T.; Ma, X.; Dicks, B.M.; Smith, M.T.; Zhang, L.; Feusner, J.; Wiencke, J. In utero origin of t (8; 21) AML1-ETO translocations in childhood acute myeloid leukemia. Blood J. Am. Soc. Hematol. 2002, 99, 3801–3805. [Google Scholar]
- Shima, T.; Miyamoto, T.; Kikushige, Y.; Yuda, J.; Tochigi, T.; Yoshimoto, G.; Kato, K.; Takenaka, K.; Iwasaki, H.; Mizuno, S.; et al. The ordered acquisition of Class II and Class I mutations directs formation of human t(8;21) acute myelogenous leukemia stem cell. Exp. Hematol. 2014, 42, 955–965.e5. [Google Scholar] [CrossRef]
- Grisolano, J.L.; O’Neal, J.; Cain, J.; Tomasson, M.H. An activated receptor tyrosine kinase, TEL/PDGFβR, cooperates with AML1/ETO to induce acute myeloid leukemia in mice. Proc. Natl. Acad. Sci. USA 2003, 100, 9506–9511. [Google Scholar] [CrossRef]
- Schessl, C.; Rawat, V.P.; Cusan, M.; Deshpande, A.; Kohl, T.M.; Rosten, P.M.; Spiekermann, K.; Humphries, R.K.; Schnittger, S.; Kern, W. The AML1-ETO fusion gene and the FLT3 length mutation collaborate in inducing acute leukemia in mice. J. Clin. Investig. 2005, 115, 2159–2168. [Google Scholar] [CrossRef]
- Nick, H.J.; Kim, H.-G.; Chang, C.-W.; Harris, K.W.; Reddy, V.; Klug, C.A. Distinct classes of c-Kit–activating mutations differ in their ability to promote RUNX1-ETO–associated acute myeloid leukemia. Blood J. Am. Soc. Hematol. 2012, 119, 1522–1531. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.-Y.; Zhao, L.-J.; Wu, C.-F.; Liu, P.; Shi, L.; Liang, Y.; Xiong, S.-M.; Mi, J.-Q.; Chen, Z.; Ren, R. C-KIT mutation cooperates with full-length AML1-ETO to induce acute myeloid leukemia in mice. Proc. Natl. Acad. Sci. USA 2011, 108, 2450–2455. [Google Scholar] [CrossRef] [PubMed]
- Zuber, J.; Radtke, I.; Pardee, T.S.; Zhao, Z.; Rappaport, A.R.; Luo, W.; McCurrach, M.E.; Yang, M.-M.; Dolan, M.E.; Kogan, S.C. Mouse models of human AML accurately predict chemotherapy response. Genes Dev. 2009, 23, 877–889. [Google Scholar] [CrossRef] [PubMed]
- Heidenreich, O.; Krauter, J.r.; Riehle, H.; Hadwiger, P.; John, M.; Heil, G.; Vornlocher, H.-P.; Nordheim, A. AML1/MTG8 oncogene suppression by small interfering RNAs supports myeloid differentiation of t (8; 21)-positive leukemic cells. Blood J. Am. Soc. Hematol. 2003, 101, 3157–3163. [Google Scholar]
- Ptasinska, A.; Assi, S.A.; Mannari, D.; James, S.R.; Williamson, D.; Dunne, J.; Hoogenkamp, M.; Wu, M.; Care, M.; McNeill, H.; et al. Depletion of RUNX1/ETO in t(8;21) AML cells leads to genome-wide changes in chromatin structure and transcription factor binding. Leukemia 2012, 26, 1829–1841. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Shen, T.; Huynh, L.; Klisovic, M.I.; Rush, L.J.; Ford, J.L.; Yu, J.; Becknell, B.; Li, Y.; Liu, C. Interplay of RUNX1/MTG8 and DNA methyltransferase 1 in acute myeloid leukemia. Cancer Res. 2005, 65, 1277–1284. [Google Scholar] [CrossRef]
- Lin, S.; Ptasinska, A.; Chen, X.; Shrestha, M.; Assi, S.A.; Chin, P.S.; Imperato, M.R.; Aronow, B.; Zhang, J.; Weirauch, M.T. A FOXO1-induced oncogenic network defines the AML1-ETO preleukemic program. Blood J. Am. Soc. Hematol. 2017, 130, 1213–1222. [Google Scholar] [CrossRef]
- Frank, R.C.; Sun, X.; Berguido, F.J.; Jakubowiak, A.; Nimer, S.D. The t(8;21) fusion protein, AML1/ETO, transforms NIH3T3 cells and activates AP-1. Oncogene 1999, 18, 1701–1710. [Google Scholar] [CrossRef]
- Elsasser, A.; Franzen, M.; Kohlmann, A.; Weisser, M.; Schnittger, S.; Schoch, C.; Reddy, V.A.; Burel, S.; Zhang, D.E.; Ueffing, M.; et al. The fusion protein AML1-ETO in acute myeloid leukemia with translocation t(8;21) induces c-jun protein expression via the proximal AP-1 site of the c-jun promoter in an indirect, JNK-dependent manner. Oncogene 2003, 22, 5646–5657. [Google Scholar] [CrossRef]
- Mandoli, A.; Singh, A.A.; Prange, K.H.; Tijchon, E.; Oerlemans, M.; Dirks, R.; Ter Huurne, M.; Wierenga, A.T.; Janssen-Megens, E.M.; Berentsen, K. The hematopoietic transcription factors RUNX1 and ERG prevent AML1-ETO oncogene overexpression and onset of the apoptosis program in t (8; 21) AMLs. Cell Rep. 2016, 17, 2087–2100. [Google Scholar] [CrossRef]
- Ben-Ami, O.; Friedman, D.; Leshkowitz, D.; Goldenberg, D.; Orlovsky, K.; Pencovich, N.; Lotem, J.; Tanay, A.; Groner, Y. Addiction of t (8; 21) and inv (16) acute myeloid leukemia to native RUNX1. Cell Rep. 2013, 4, 1131–1143. [Google Scholar] [CrossRef] [PubMed]
- Al-Harbi, S.; Aljurf, M.; Mohty, M.; Almohareb, F.; Ahmed, S.O.A. An update on the molecular pathogenesis and potential therapeutic targeting of AML with t (8; 21)(q22; q22. 1); RUNX1-RUNX1T1. Blood Adv. 2020, 4, 229–238. [Google Scholar] [CrossRef] [PubMed]
- Schnittger, S.; Bacher, U.; Kern, W.; Haferlach, C.; Haferlach, T. JAK2 seems to be a typical cooperating mutation in therapy-related t (8; 21)/AML 1-ETO-positive AML. Leukemia 2007, 21, 183–184. [Google Scholar] [CrossRef] [PubMed]
- Renneville, A.; Roumier, C.; Biggio, V.; Nibourel, O.; Boissel, N.; Fenaux, P.; Preudhomme, C. Cooperating gene mutations in acute myeloid leukemia: A review of the literature. Leukemia 2008, 22, 915–931. [Google Scholar] [CrossRef] [PubMed]
- Forster, V.; Nahari, M.; Martinez-Soria, N.; Bradburn, A.; Ptasinska, A.; Assi, S.; Fordham, S.; McNeil, H.; Bonifer, C.; Heidenreich, O. The leukemia-associated RUNX1/ETO oncoprotein confers a mutator phenotype. Leukemia 2016, 30, 251–254. [Google Scholar] [CrossRef] [PubMed]
- Christen, F.; Hoyer, K.; Yoshida, K.; Hou, H.-A.; Waldhueter, N.; Heuser, M.; Hills, R.K.; Chan, W.; Hablesreiter, R.; Blau, O. Genomic landscape and clonal evolution of acute myeloid leukemia with t (8; 21): An international study on 331 patients. Blood J. Am. Soc. Hematol. 2019, 133, 1140–1151. [Google Scholar]
- Faber, Z.J.; Chen, X.; Gedman, A.L.; Boggs, K.; Cheng, J.; Ma, J.; Radtke, I.; Chao, J.-R.; Walsh, M.P.; Song, G. The genomic landscape of core-binding factor acute myeloid leukemias. Nat. Genet. 2016, 48, 1551–1556. [Google Scholar] [CrossRef]
- Martinez-Soria, N.; McKenzie, L.; Draper, J.; Ptasinska, A.; Issa, H.; Potluri, S.; Blair, H.J.; Pickin, A.; Isa, A.; Chin, P.S. The oncogenic transcription factor RUNX1/ETO corrupts cell cycle regulation to drive leukemic transformation. Cancer Cell 2018, 34, 626–642.e628. [Google Scholar] [CrossRef]
- Eisfeld, A.-K.; Kohlschmidt, J.; Schwind, S.; Nicolet, D.; Blachly, J.S.; Orwick, S.; Shah, C.; Bainazar, M.; Kroll, K.W.; Walker, C.J. Mutations in the CCND1 and CCND2 genes are frequent events in adult patients with t (8; 21)(q22; q22) acute myeloid leukemia. Leukemia 2017, 31, 1278–1285. [Google Scholar] [CrossRef]
- Khanna, V.; Eide, C.; Tognon, C.; Maxson, J.; Wilmot, B.; Bottomly, D.; McWeeney, S.; Druker, B.; Tyner, J. Recurrent cyclin D2 mutations in myeloid neoplasms. Leukemia 2017, 31, 2005–2008. [Google Scholar] [CrossRef]
- Monte, E.R.; Wilding, A.; Leubolt, G.; Kerbs, P.; Bagnoli, J.W.; Hartmann, L.; Hiddemann, W.; Chen-Wichmann, L.; Krebs, S.; Blum, H. ZBTB7A prevents RUNX1-RUNX1T1-dependent clonal expansion of human hematopoietic stem and progenitor cells. Oncogene 2020, 39, 3195–3205. [Google Scholar] [CrossRef] [PubMed]
- Martens, J.H.; Mandoli, A.; Simmer, F.; Wierenga, B.J.; Saeed, S.; Singh, A.A.; Altucci, L.; Vellenga, E.; Stunnenberg, H.G. ERG and FLI1 binding sites demarcate targets for aberrant epigenetic regulation by AML1-ETO in acute myeloid leukemia. Blood 2012, 120, 4038–4048. [Google Scholar] [CrossRef] [PubMed]
- Saeed, S.; Logie, C.; Francoijs, K.-J.; Frigè, G.; Romanenghi, M.; Nielsen, F.G.; Raats, L.; Shahhoseini, M.; Huynen, M.; Altucci, L.; et al. Chromatin accessibility, p300, and histone acetylation define PML-RARα and AML1-ETO binding sites in acute myeloid leukemia. Blood 2012, 120, 3058–3068. [Google Scholar] [CrossRef] [PubMed]
- Ptasinska, A.; Assi, S.A.; Martinez-Soria, N.; Imperato, M.R.; Piper, J.; Cauchy, P.; Pickin, A.; James, S.R.; Hoogenkamp, M.; Williamson, D. Identification of a dynamic core transcriptional network in t (8; 21) AML that regulates differentiation block and self-renewal. Cell Rep. 2014, 8, 1974–1988. [Google Scholar] [CrossRef] [PubMed]
- Nafria, M.; Keane, P.; Ng, E.S.; Stanley, E.G.; Elefanty, A.G.; Bonifer, C. Expression of RUNX1-ETO Rapidly Alters the Chromatin Landscape and Growth of Early Human Myeloid Precursor Cells. Cell Rep. 2020, 31, 107691. [Google Scholar] [CrossRef]
- Regha, K.; Assi, S.A.; Tsoulaki, O.; Gilmour, J.; Lacaud, G.; Bonifer, C. Developmental-stage-dependent transcriptional response to leukaemic oncogene expression. Nat. Commun. 2015, 6, 7203. [Google Scholar] [CrossRef]
- Assi, S.A.; Imperato, M.R.; Coleman, D.J.L.; Pickin, A.; Potluri, S.; Ptasinska, A.; Chin, P.S.; Blair, H.; Cauchy, P.; James, S.R.; et al. Subtype-specific regulatory network rewiring in acute myeloid leukemia. Nat. Genet. 2019, 51, 151–162. [Google Scholar] [CrossRef]
- Quinlan, A.R.; Hall, I.M. BEDTools: A flexible suite of utilities for comparing genomic features. Bioinformatics 2010, 26, 841–842. [Google Scholar] [CrossRef]
- Vangala, R.K.; Heiss-Neumann, M.S.; Rangatia, J.S.; Singh, S.M.; Schoch, C.; Tenen, D.G.; Hiddemann, W.; Behre, G. The myeloid master regulator transcription factor PU.1 is inactivated by AML1-ETO in t(8;21) myeloid leukemia. Blood 2003, 101, 270–277. [Google Scholar] [CrossRef]
- Zhang, J.; Kalkum, M.; Yamamura, S.; Chait, B.T.; Roeder, R.G. E Protein Silencing by the Leukemogenic AML1-ETO Fusion Protein. Science 2004, 305, 1286. [Google Scholar] [CrossRef]
- Wang, J.; Hoshino, T.; Redner, R.L.; Kajigaya, S.; Liu, J.M. ETO, fusion partner in t(8;21) acute myeloid leukemia, represses transcription by interaction with the human N-CoR/mSin3/HDAC1 complex. Proc. Natl. Acad. Sci. USA 1998, 95, 10860. [Google Scholar] [CrossRef] [PubMed]
- Stengel, K.R.; Ellis, J.; Spielman, C.; Bomber, M.; Hiebert, S.W. Definition of a Small Core Transcriptional Circuit Regulated by AML1-ETO. bioRxiv 2020. [Google Scholar] [CrossRef]
- Ray, D.; Kwon, S.Y.; Tagoh, H.; Heidenreich, O.; Ptasinska, A.; Bonifer, C. Lineage-inappropriate PAX5 expression in t(8;21) acute myeloid leukemia requires signaling-mediated abrogation of polycomb repression. Blood 2013, 122, 759–769. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Xing, Y.; Lu, W.; Li, S.; Tian, Z.; Xing, H.; Tang, K.; Xu, Y.; Rao, Q.; Wang, M.; et al. RUNX1 inhibits proliferation and induces apoptosis of t(8;21) leukemia cells via KLF4 mediated transactivation of P57. Haematologica 2019. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Gural, A.; Sun, X.-J.; Zhao, X.; Perna, F.; Huang, G.; Hatlen, M.A.; Vu, L.; Liu, F.; Xu, H. The leukemogenicity of AML1-ETO is dependent on site-specific lysine acetylation. Science 2011, 333, 765–769. [Google Scholar] [CrossRef] [PubMed]
- Trombly, D.J.; Whitfield, T.W.; Padmanabhan, S.; Gordon, J.A.R.; Lian, J.B.; van Wijnen, A.J.; Zaidi, S.K.; Stein, J.L.; Stein, G.S. Genome-wide co-occupancy of AML1-ETO and N-CoR defines the t(8;21) AML signature in leukemic cells. BMC Genom. 2015, 16, 309. [Google Scholar] [CrossRef]
- Chen, G.; Liu, A.; Xu, Y.; Gao, L.; Jiang, M.; Li, Y.; Lv, N.; Zhou, L.; Wang, L.; Yu, L.; et al. The RUNX1–ETO fusion protein trans-activates c-KIT expression by recruiting histone acetyltransferase P300 on its promoter. FEBS J. 2019, 286, 901–912. [Google Scholar] [CrossRef]
- Kitabayashi, I.; Yokoyama, A.; Shimizu, K.; Ohki, M. Interaction and functional cooperation of the leukemia-associated factors AML1 and p300 in myeloid cell differentiation. EMBO J. 1998, 17, 2994–3004. [Google Scholar] [CrossRef]
- Yamaguchi, Y.; Kurokawa, M.; Imai, Y.; Izutsu, K.; Asai, T.; Ichikawa, M.; Yamamoto, G.; Nitta, E.; Yamagata, T.; Sasaki, K.; et al. AML1 Is Functionally Regulated through p300-mediated Acetylation on Specific Lysine Residues. J. Biol. Chem. 2004, 279, 15630–15638. [Google Scholar] [CrossRef]
- Guo, C.; Li, J.; Steinauer, N.; Wong, M.; Wu, B.; Dickson, A.; Kalkum, M.; Zhang, J. Histone deacetylase 3 preferentially binds and collaborates with the transcription factor RUNX1 to repress AML1–ETO–dependent transcription in t(8;21) AML. J. Biol. Chem. 2020, 295, 4212–4223. [Google Scholar] [CrossRef]
- Stoner, S.A.; Liu, K.T.H.; Andrews, E.T.; Liu, M.; Arimoto, K.I.; Yan, M.; Davis, A.G.; Weng, S.; Dow, M.; Xian, S.; et al. The RUNX1-ETO target gene RASSF2 suppresses t(8;21) AML development and regulates Rac GTPase signaling. Blood Cancer J 2020, 10, 16. [Google Scholar] [CrossRef] [PubMed]
- Sinha, C.; Cunningham, L.C.; Liu, P.P. Core binding factor acute myeloid leukemia: New prognostic categories and therapeutic opportunities. Semin. Hematol. 2015, 52, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Zhang, J.; Hua, X.; Li, H.; Wang, Z.; Yang, B. clinical heterogeneity under induction with different dosages of cytarabine in core binding factor acute myeloid leukaemia. Sci. Rep. 2020, 10, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Paschka, P.; Döhner, K. Core-binding factor acute myeloid leukemia: Can we improve on HiDAC consolidation? Hematology 2013, 2013, 209–219. [Google Scholar] [CrossRef]
- Illendula, A.; Gilmour, J.; Grembecka, J.; Tirumala, V.S.S.; Boulton, A.; Kuntimaddi, A.; Schmidt, C.; Wang, L.; Pulikkan, J.A.; Zong, H.; et al. Small Molecule Inhibitor of CBFbeta-RUNX Binding for RUNX Transcription Factor Driven Cancers. EBioMedicine 2016, 8, 117–131. [Google Scholar] [CrossRef]
- Metz, A.; Schanda, J.; Grez, M.; Wichmann, C.; Gohlke, H. From determinants of RUNX1/ETO tetramerization to small-molecule protein–protein interaction inhibitors targeting acute myeloid leukemia. J. Chem. Inf. Modeling 2013, 53, 2197–2202. [Google Scholar] [CrossRef]
- Wichmann, C.; Chen, L.; Heinrich, M.; Baus, D.; Pfitzner, E.; Zörnig, M.; Ottmann, O.G.; Grez, M. Targeting the oligomerization domain of ETO interferes with RUNX1/ETO oncogenic activity in t (8; 21)-positive leukemic cells. Cancer Res. 2007, 67, 2280–2289. [Google Scholar] [CrossRef]
- Bartel, Y.; Grez, M.; Wichmann, C. Interference with RUNX1/ETO leukemogenic function by cell-penetrating peptides targeting the NHR2 oligomerization domain. Biomed Res. Int. 2013, 2013. [Google Scholar] [CrossRef]
- Marcucci, G.; Geyer, S.; Laumann, K.; Zhao, W.; Bucci, D.; Uy, G.L.; Blum, W.; Eisfeld, A.-K.; Pardee, T.S.; Wang, E.S. Combination of dasatinib with chemotherapy in previously untreated core binding factor acute myeloid leukemia: CALGB 10801. Blood Adv. 2020, 4, 696–705. [Google Scholar] [CrossRef]
- Abbas, H.A.; Alfayez, M.; Kadia, T.; Ravandi-Kashani, F.; Daver, N. Midostaurin In Acute Myeloid Leukemia: An Evidence-Based Review And Patient Selection. Cancer Manag. Res. 2019, 11, 8817. [Google Scholar] [CrossRef]
- Lo, M.-C.; Peterson, L.F.; Yan, M.; Cong, X.; Hickman, J.H.; DeKelver, R.C.; Niewerth, D.; Zhang, D.-E. JAK inhibitors suppress t (8; 21) fusion protein-induced leukemia. Leukemia 2013, 27, 2272–2279. [Google Scholar] [CrossRef] [PubMed]
- van der Kouwe, E.; Staber, P.B. RUNX1-ETO: Attacking the epigenome for genomic instable leukemia. Int. J. Mol. Sci. 2019, 20, 350. [Google Scholar] [CrossRef] [PubMed]
- Duque-Afonso, J.; Yalcin, A.; Berg, T.; Abdelkarim, M.; Heidenreich, O.; Lübbert, M. The HDAC class I-specific inhibitor entinostat (MS-275) effectively relieves epigenetic silencing of the LAT2 gene mediated by AML1/ETO. Oncogene 2011, 30, 3062–3072. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bots, M.; Verbrugge, I.; Martin, B.P.; Salmon, J.M.; Ghisi, M.; Baker, A.; Stanley, K.; Shortt, J.; Ossenkoppele, G.J.; Zuber, J. Differentiation therapy for the treatment of t (8; 21) acute myeloid leukemia using histone deacetylase inhibitors. Blood J. Am. Soc. Hematol. 2014, 123, 1341–1352. [Google Scholar]
- Esposito, M.T.; Zhao, L.; Fung, T.K.; Rane, J.K.; Wilson, A.; Martin, N.; Gil, J.; Leung, A.Y.; Ashworth, A.; So, C.W.E. Synthetic lethal targeting of oncogenic transcription factors in acute leukemia by PARP inhibitors. Nat. Med. 2015, 21, 1481. [Google Scholar] [CrossRef] [PubMed]
- Nieborowska-Skorska, M.; Paietta, E.M.; Levine, R.L.; Fernandez, H.F.; Tallman, M.S.; Litzow, M.R.; Skorski, T. Inhibition of the mutated c-KIT kinase in AML1-ETO–positive leukemia cells restores sensitivity to PARP inhibitor. Blood Adv. 2019, 3, 4050. [Google Scholar] [CrossRef] [PubMed]
- Schmoellerl, J.; Barbosa, I.A.M.; Eder, T.; Brandstoetter, T.; Schmidt, L.; Maurer, B.; Troester, S.; Pham, H.T.T.; Sagarajit, M.; Ebner, J.; et al. CDK6 is an essential direct target of NUP98 fusion proteins in acute myeloid leukemia. Blood 2020, 136, 387–400. [Google Scholar] [CrossRef]
- Uras, I.Z.; Walter, G.J.; Scheicher, R.; Bellutti, F.; Prchal-Murphy, M.; Tigan, A.S.; Valent, P.; Heidel, F.H.; Kubicek, S.; Scholl, C.; et al. Palbociclib treatment of FLT3-ITD+ AML cells uncovers a kinase-dependent transcriptional regulation of FLT3 and PIM1 by CDK6. Blood 2016, 127, 2890–2902. [Google Scholar] [CrossRef]
- Pikman, Y.; Alexe, G.; Roti, G.; Conway, A.S.; Furman, A.; Lee, E.S.; Place, A.E.; Kim, S.; Saran, C.; Modiste, R.; et al. Synergistic Drug Combinations with a CDK4/6 Inhibitor in T-cell Acute Lymphoblastic Leukemia. Clin Cancer Res 2017, 23, 1012–1024. [Google Scholar] [CrossRef]
- Yang, C.; Boyson, C.A.; Di Liberto, M.; Huang, X.; Hannah, J.; Dorn, D.C.; Moore, M.A.; Chen-Kiang, S.; Zhou, P. CDK4/6 Inhibitor PD 0332991 Sensitizes Acute Myeloid Leukemia to Cytarabine-Mediated Cytotoxicity. Cancer Res 2015, 75, 1838–1845. [Google Scholar] [CrossRef]
- Bushweller, J.H. Targeting transcription factors in cancer-from undruggable to reality. Nat. Rev. Cancer 2019, 19, 611–624. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kellaway, S.; Chin, P.S.; Barneh, F.; Bonifer, C.; Heidenreich, O. t(8;21) Acute Myeloid Leukemia as a Paradigm for the Understanding of Leukemogenesis at the Level of Gene Regulation and Chromatin Programming. Cells 2020, 9, 2681. https://doi.org/10.3390/cells9122681
Kellaway S, Chin PS, Barneh F, Bonifer C, Heidenreich O. t(8;21) Acute Myeloid Leukemia as a Paradigm for the Understanding of Leukemogenesis at the Level of Gene Regulation and Chromatin Programming. Cells. 2020; 9(12):2681. https://doi.org/10.3390/cells9122681
Chicago/Turabian StyleKellaway, Sophie, Paulynn S. Chin, Farnaz Barneh, Constanze Bonifer, and Olaf Heidenreich. 2020. "t(8;21) Acute Myeloid Leukemia as a Paradigm for the Understanding of Leukemogenesis at the Level of Gene Regulation and Chromatin Programming" Cells 9, no. 12: 2681. https://doi.org/10.3390/cells9122681
APA StyleKellaway, S., Chin, P. S., Barneh, F., Bonifer, C., & Heidenreich, O. (2020). t(8;21) Acute Myeloid Leukemia as a Paradigm for the Understanding of Leukemogenesis at the Level of Gene Regulation and Chromatin Programming. Cells, 9(12), 2681. https://doi.org/10.3390/cells9122681




