Fibronectin and Periostin as Prognostic Markers in Ovarian Cancer
Abstract
1. Introduction
2. Materials and Methods
2.1. Tissue Arrays
2.2. Clinical Samples Used for Survival Analysis
2.3. Immunohistochemistry
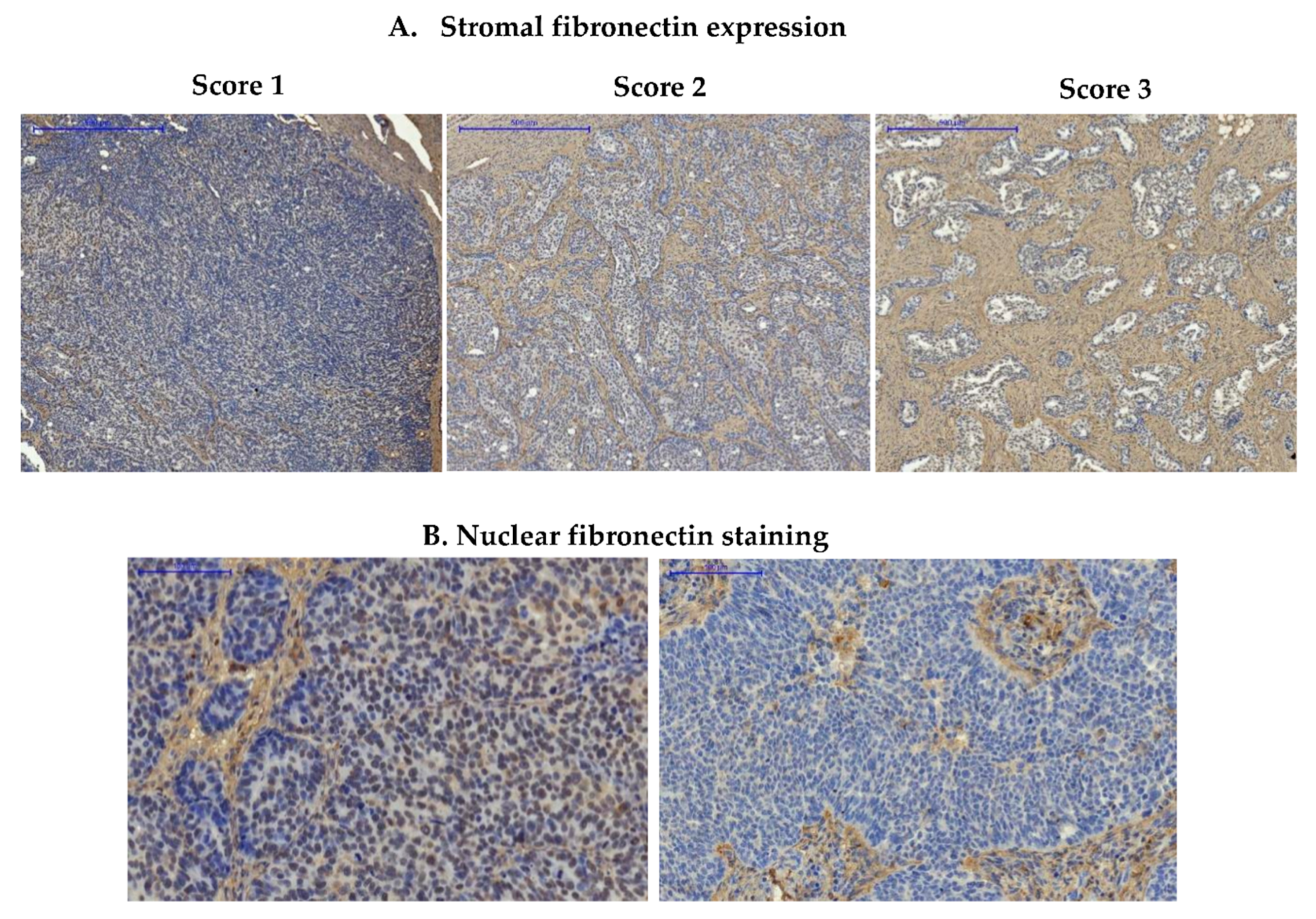
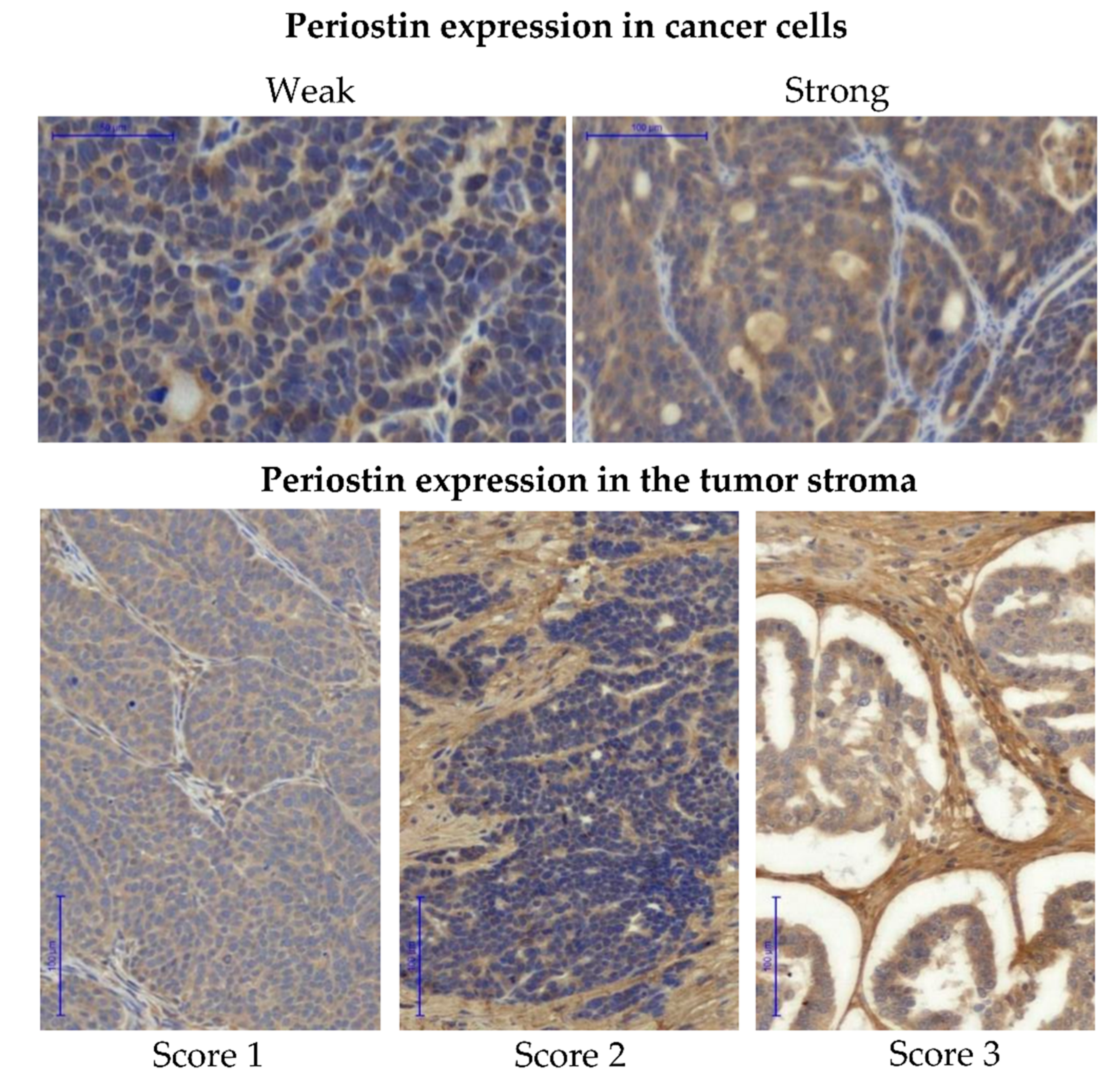
2.4. Pathological and Immunohistochemical Evaluation of the Samples
2.5. Statistical Analysis
3. Results
3.1. Fibronectin
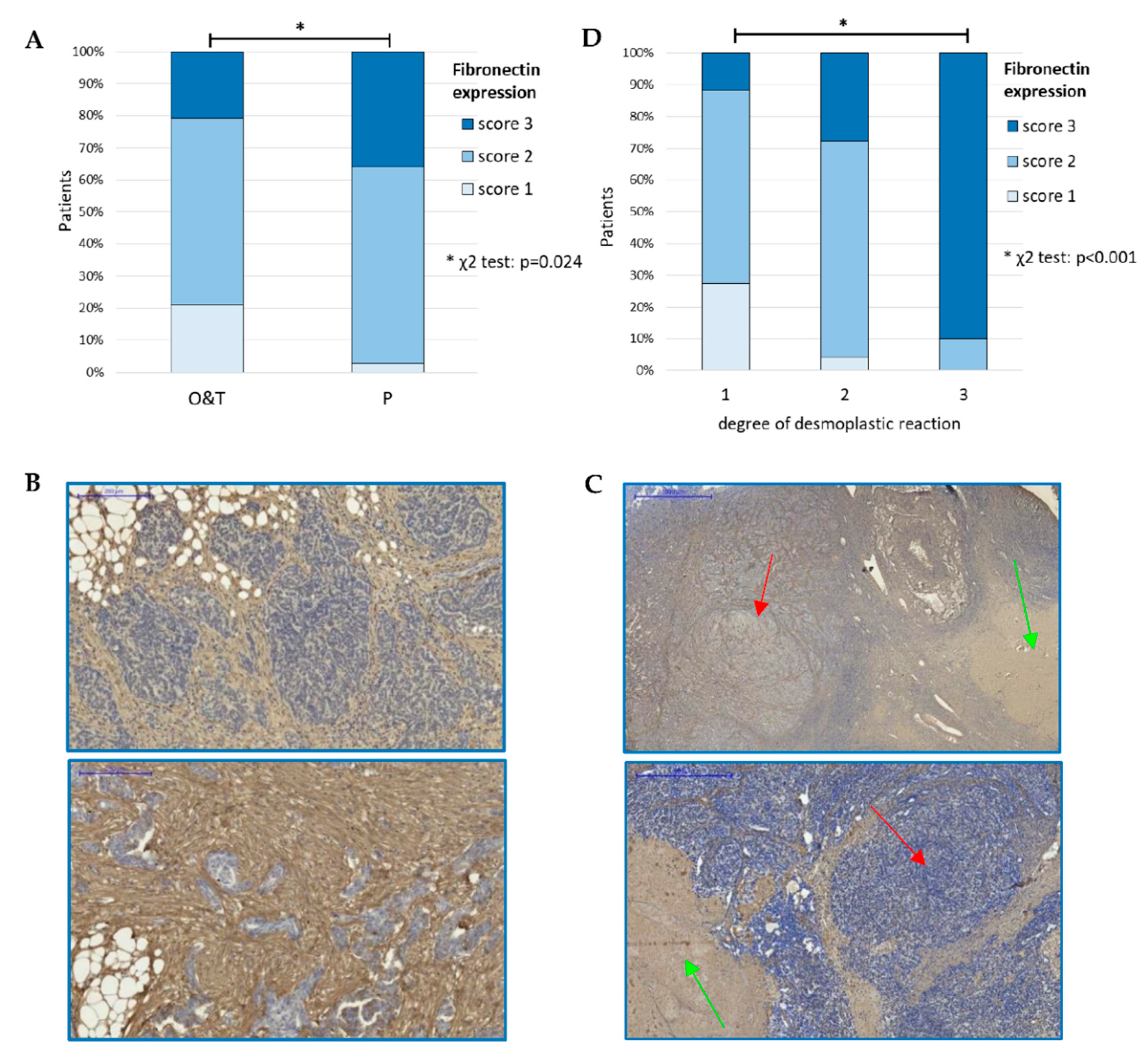
3.1.1. Expression of Fibronectin in Ovarian Cancer Samples
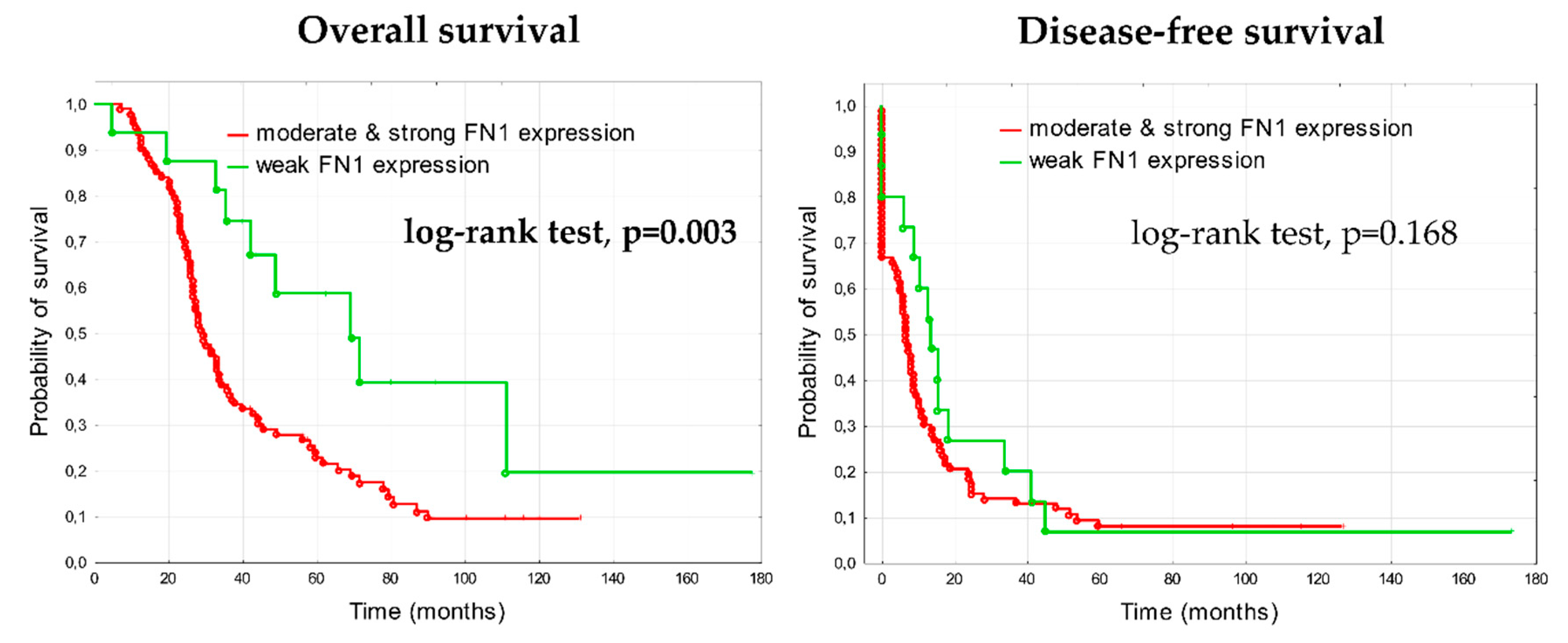
3.1.2. Prognostic Significance of Fibronectin in Advanced Ovarian Cancer
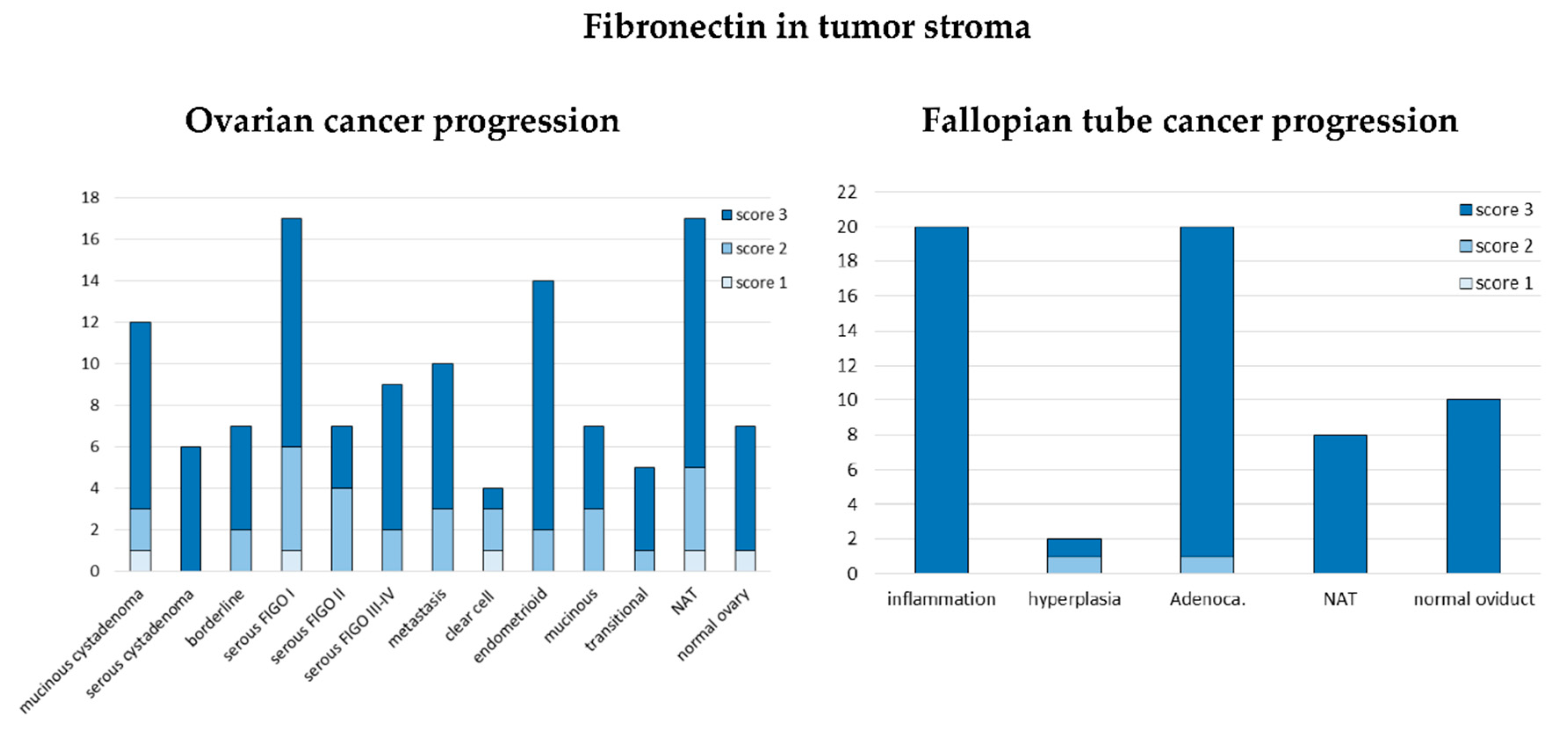
3.1.3. Expression of Fibronectin on Tissue Arrays
3.2. Periostin
3.2.1. Expression of Periostin in Advanced Ovarian Cancers
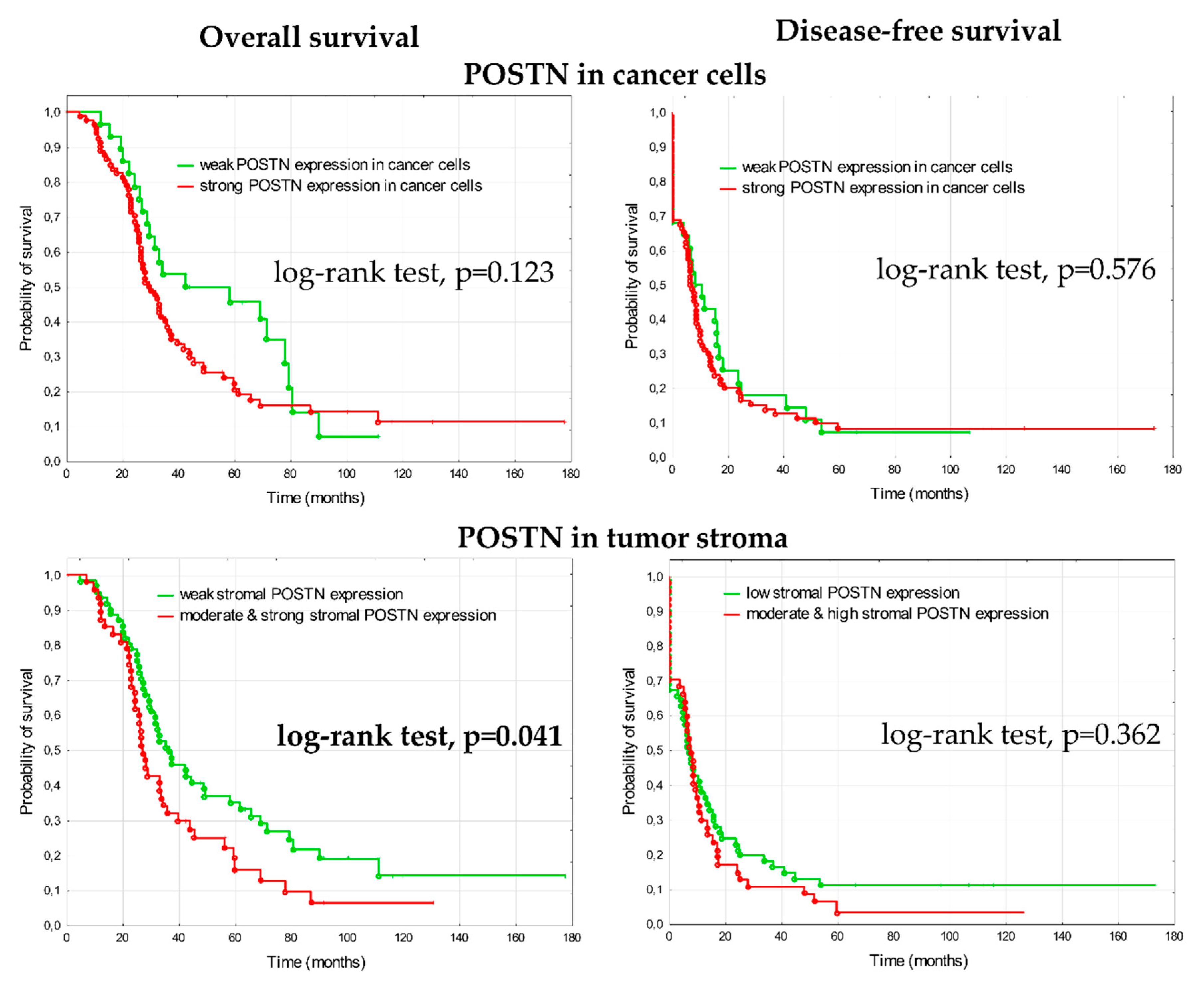
3.2.2. Prognostic Significance of Periostin in Advanced Ovarian Cancer
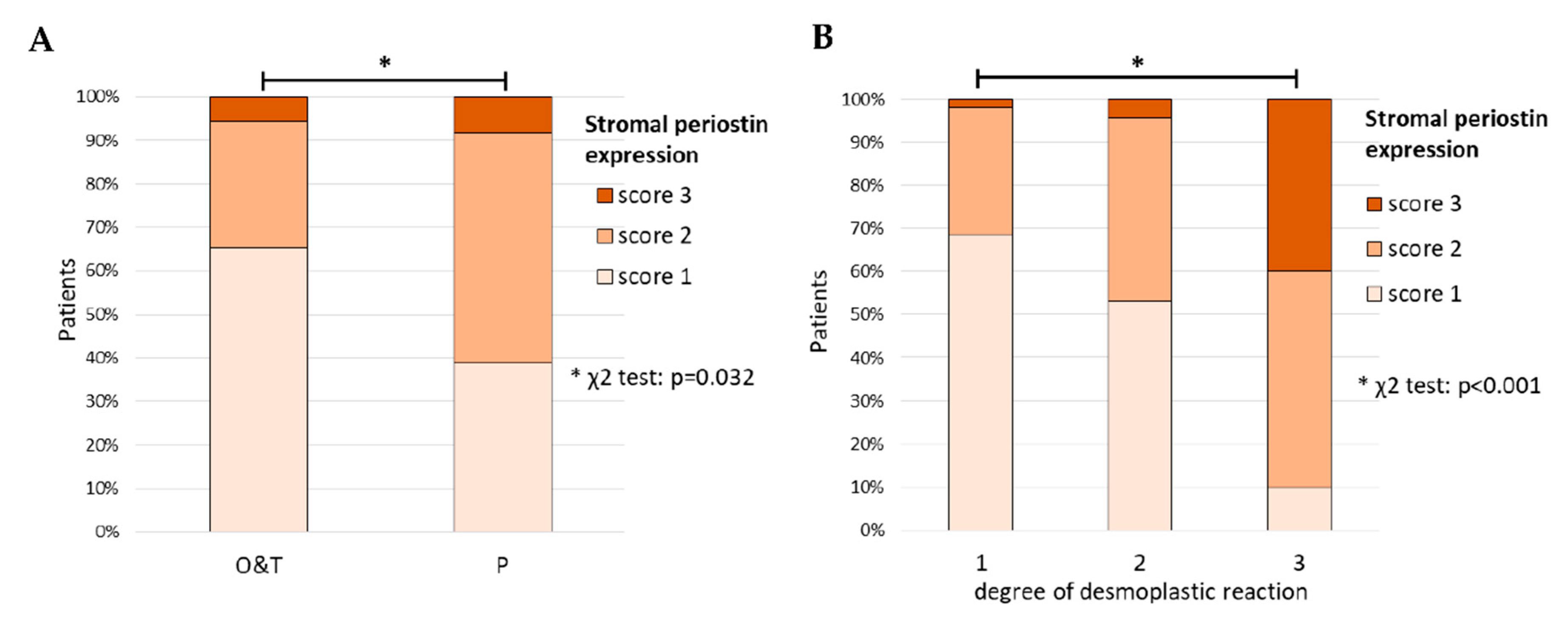
3.2.3. Expression of Periostin in Tissue Arrays
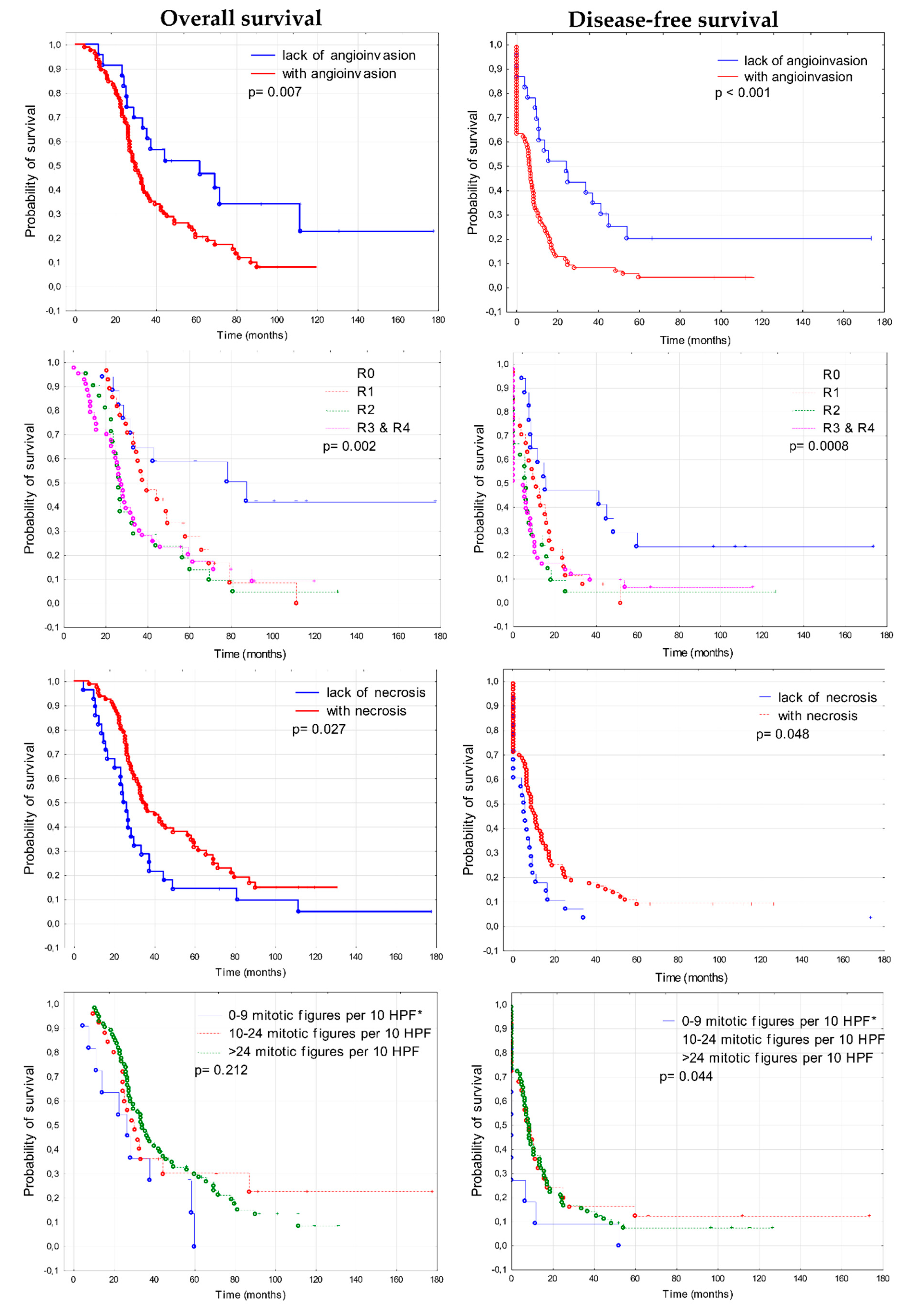
3.3. Prognostic Significance of Clinico-Pathological Features
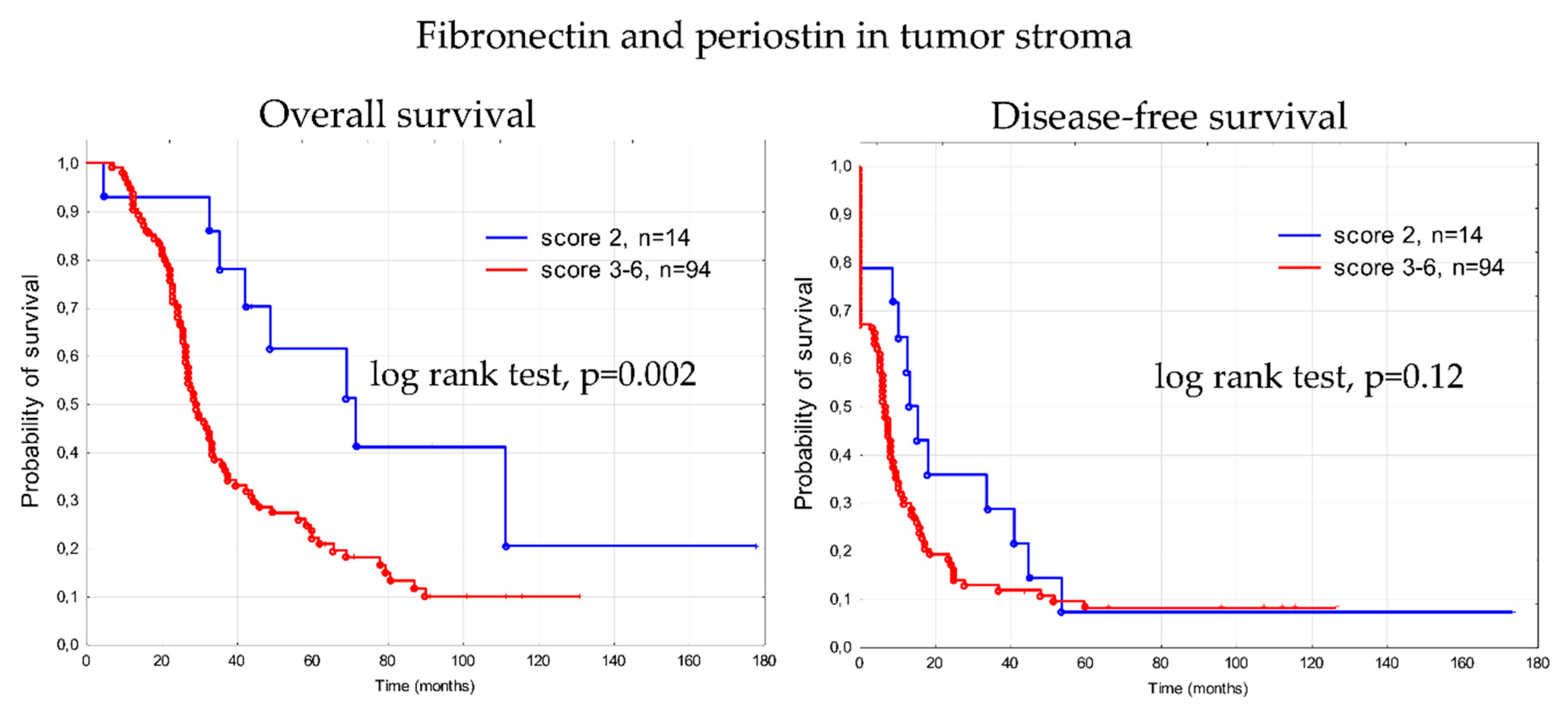
3.4. Prognostic Significance of Fibronectin 1 and Periostin (FN1&POSTN Score)
4. Discussion
4.1. The Role of Fibronectin in Ovarian Cancer
4.2. Periostin Significance in Ovarian Cancer
4.3. FN1&POSTN Score
4.4. Other Prognostic Factors
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kujawa, K.A.; Lisowska, K.M. Ovarian cancer-from biology to clinic. Postepy Hig. Med. Dosw. (Online) 2015, 69, 1275–1290. [Google Scholar] [CrossRef] [PubMed]
- Tudrej, P.; Kujawa, K.A.; Cortez, A.J.; Lisowska, K.M. Characteristics of in Vivo Model Systems for Ovarian Cancer Studies. Diagnostics 2019, 9, 120. [Google Scholar] [CrossRef] [PubMed]
- Tudrej, P.; Kujawa, K.; Cortez, A.J.; Lisowska, K.M. Characteristics of in vitro model systems for ovarian cancer studies. Oncol. Clin. Pract. 2019, 15, 246–259. [Google Scholar] [CrossRef]
- Lisowska, K.M.; Olbryt, M.; Dudaladava, V.; Pamula-Pilat, J.; Kujawa, K.; Grzybowska, E.; Jarzab, M.; Student, S.; Rzepecka, I.K.; Jarzab, B.; et al. Gene expression analysis in ovarian cancer—Faults and hints from DNA microarray study. Front. Oncol. 2014, 4, 6. [Google Scholar] [CrossRef] [PubMed]
- Lisowska, K.M.; Olbryt, M.; Student, S.; Kujawa, K.A.; Cortez, A.J.; Simek, K.; Dansonka-Mieszkowska, A.; Rzepecka, I.K.; Tudrej, P.; Kupryjanczyk, J. Unsupervised analysis reveals two molecular subgroups of serous ovarian cancer with distinct gene expression profiles and survival. J. Cancer Res. Clin. Oncol. 2016, 142, 1239–1252. [Google Scholar] [CrossRef] [PubMed]
- White, E.S.; Muro, A.F. Fibronectin splice variants: Understanding their multiple roles in health and disease using engineered mouse models. IUBMB Life 2011, 63, 538–546. [Google Scholar] [CrossRef] [PubMed]
- To, W.S.; Midwood, K.S. Plasma and cellular fibronectin: Distinct and independent functions during tissue repair. Fibrogenesis Tissue Repair 2011, 4, 21. [Google Scholar] [CrossRef]
- Singh, P.; Carraher, C.; Schwarzbauer, J.E. Assembly of fibronectin extracellular matrix. Annu. Rev. Cell Dev. Biol. 2010, 26, 397–419. [Google Scholar] [CrossRef]
- Rick, J.W.; Chandra, A.; Dalle Ore, C.; Nguyen, A.T.; Yagnik, G.; Aghi, M.K. Fibronectin in malignancy: Cancer-specific alterations, protumoral effects, and therapeutic implications. Semin. Oncol. 2019, 46, 284–290. [Google Scholar] [CrossRef]
- Lin, T.-C.; Yang, C.-H.; Cheng, L.-H.; Chang, W.-T.; Lin, Y.-R.; Cheng, H.-C. Fibronectin in Cancer: Friend or Foe. Cells 2019, 9, 27. [Google Scholar] [CrossRef]
- Zand, L.; Qiang, F.; Roskelley, C.D.; Leung, P.C.; Auersperg, N. Differential effects of cellular fibronectin and plasma fibronectin on ovarian cancer cell adhesion, migration, and invasion. In Vitro Cell. Dev. Biol. Anim. 2003, 39, 178–182. [Google Scholar] [CrossRef]
- Ricciardelli, C.; Lokman, N.A.; Ween, M.P.; Oehler, M.K. WOMEN IN CANCER THEMATIC REVIEW: Ovarian cancer-peritoneal cell interactions promote extracellular matrix processing. Endocr. Relat. Cancer 2016, 23, T155–T168. [Google Scholar] [CrossRef] [PubMed]
- Takeshita, S.; Kikuno, R.; Tezuka, K.; Amann, E. Osteoblast-specific factor 2: Cloning of a putative bone adhesion protein with homology with the insect protein fasciclin I. Biochem. J. 1993, 294, 271–278. [Google Scholar] [CrossRef]
- Horiuchi, K.; Amizuka, N.; Takeshita, S.; Takamatsu, H.; Katsuura, M.; Ozawa, H.; Toyama, Y.; Bonewald, L.F.; Kudo, A. Identification and characterization of a novel protein, periostin, with restricted expression to periosteum and periodontal ligament and increased expression by transforming growth factor beta. J. Bone Miner. Res. 1999, 14, 1239–1249. [Google Scholar] [CrossRef] [PubMed]
- Kruzynska-Frejtag, A.; Machnicki, M.; Rogers, R.; Markwald, R.R.; Conway, S.J. Periostin (an osteoblast-specific factor) is expressed within the embryonic mouse heart during valve formation. Mech. Dev. 2001, 103, 183–188. [Google Scholar] [CrossRef]
- Ratajczak-Wielgomas, K.; Dziegiel, P. The role of periostin in neoplastic processes. Folia Histochem. Cytobiol. 2015, 53, 120–132. [Google Scholar] [CrossRef]
- Gillan, L.; Matei, D.; Fishman, D.A.; Gerbin, C.S.; Karlan, B.Y.; Chang, D.D. Periostin secreted by epithelial ovarian carcinoma is a ligand for αVβ3 and αVβ5 integrins and promotes cell motility. Cancer Res. 2002, 62, 5358–5364. [Google Scholar] [PubMed]
- Choi, K.U.; Yun, J.S.; Lee, I.H.; Heo, S.C.; Shin, S.H.; Jeon, E.S.; Choi, Y.J.; Suh, D.S.; Yoon, M.S.; Kim, J.H. Lysophosphatidic acid-induced expression of periostin in stromal cells: Prognoistic relevance of periostin expression in epithelial ovarian cancer. Int. J. Cancer 2011, 128, 332–342. [Google Scholar] [CrossRef] [PubMed]
- Sterzynska, K.; Kazmierczak, D.; Klejewski, A.; Swierczewska, M.; Wojtowicz, K.; Nowacka, M.; Brazert, J.; Nowicki, M.; Januchowski, R. Expression of Osteoblast-Specific Factor 2 (OSF-2, Periostin) Is Associated with Drug Resistance in Ovarian Cancer Cell Lines. Int. J. Mol. Sci. 2019, 20. [Google Scholar] [CrossRef]
- Januchowski, R.; Zawierucha, P.; Rucinski, M.; Zabel, M. Microarray-based detection and expression analysis of extracellular matrix proteins in drugresistant ovarian cancer cell lines. Oncol. Rep. 2014, 32, 1981–1990. [Google Scholar] [CrossRef]
- Wu, Y.H.; Chang, T.H.; Huang, Y.F.; Chen, C.C.; Chou, C.Y. COL11A1 confers chemoresistance on ovarian cancer cells through the activation of Akt/c/EBPbeta pathway and PDK1 stabilization. Oncotarget 2015, 6, 23748–23763. [Google Scholar] [PubMed]
- Franke, F.E.; Von Georgi, R.; Zygmunt, M.; Munstedt, K. Association between fibronectin expression and prognosis in ovarian carcinoma. Anticancer Res. 2003, 23, 4261–4268. [Google Scholar] [PubMed]
- Demeter, A.; Sziller, I.; Csapo, Z.; Olah, J.; Keszler, G.; Jeney, A.; Papp, Z.; Staub, M. Molecular prognostic markers in recurrent and in non-recurrent epithelial ovarian cancer. Anticancer Res. 2005, 25, 2885–2889. [Google Scholar] [PubMed]
- Karlan, B.Y.; Dering, J.; Walsh, C.; Orsulic, S.; Lester, J.; Anderson, L.A.; Ginther, C.L.; Fejzo, M.; Slamon, D. POSTN/TGFBI-associated stromal signature predicts poor prognosis in serous epithelial ovarian cancer. Gynecol. Oncol. 2014, 132, 334–342. [Google Scholar] [CrossRef]
- Sung, P.L.; Jan, Y.H.; Lin, S.C.; Huang, C.C.; Lin, H.; Wen, K.C.; Chao, K.C.; Lai, C.R.; Wang, P.H.; Chuang, C.M.; et al. Periostin in tumor microenvironment is associated with poor prognosis and platinum resistance in epithelial ovarian carcinoma. Oncotarget 2015, 7, 4036–4047. [Google Scholar] [CrossRef]
- Barber, H.R.; Sommers, S.C.; Synder, R.; Kwon, T.H. Histologic and nuclear grading and stromal reactions as indices for prognosis in ovarian cancer. Am. J. Obstet. Gynecol. 1975, 121, 795–807. [Google Scholar]
- Ziolkowska-Seta, I.; Madry, R.; Kraszewska, E.; Szymanska, T.; Timorek, A.; Rembiszewska, A.; Kupryjanczyk, J. TP53, BCL-2 and BAX analysis in 199 ovarian cancer patients treated with taxane-platinum regimens. Gynecol. Oncol. 2009, 112, 179–184. [Google Scholar] [CrossRef]
- Kupryjanczyk, J.; Kraszewska, E.; Ziolkowska-Seta, I.; Madry, R.; Timorek, A.; Markowska, J.; Stelmachow, J.; Bidzinski, M.; Polish Ovarian Cancer Study, G. TP53 status and taxane-platinum versus platinum-based therapy in ovarian cancer patients: A non-randomized retrospective study. BMC Cancer 2008, 8, 27. [Google Scholar] [CrossRef]
- Yen, T.W.; Aardal, N.P.; Bronner, M.P.; Thorning, D.R.; Savard, C.E.; Lee, S.P.; Bell, R.H., Jr. Myofibroblasts are responsible for the desmoplastic reaction surrounding human pancreatic carcinomas. Surgery 2002, 131, 129–134. [Google Scholar] [CrossRef]
- Cortez, A.J.; Tudrej, P.; Kujawa, K.A.; Lisowska, K.M. Advances in ovarian cancer therapy. Cancer Chemother. Pharmacol. 2018, 81, 17–38. [Google Scholar] [CrossRef]
- Quality Indicators Complete Report: Advanced (Stage III-IV) Ovarian Cancer Surgery. Guidelines; ESGO: Praque, Czech Republic, 2015.
- Le, T.; Shahriari, P.; Hopkins, L.; Faught, W.; Fung Kee Fung, M. Prognostic significance of tumor necrosis in ovarian cancer patients treated with neoadjuvant chemotherapy and interval surgical debulking. Int. J. Gynecol. Cancer. 2006, 16, 986–990. [Google Scholar] [CrossRef] [PubMed]
- Akiyama, S.K.; Olden, K.; Yamada, K.M. Fibronectin and integrins in invasion and metastasis. Cancer Metastasis Rev. 1995, 14, 173–189. [Google Scholar] [CrossRef]
- Mitra, A.K.; Sawada, K.; Tiwari, P.; Mui, K.; Gwin, K.; Lengyel, E. Ligand-independent activation of c-Met by fibronectin and α5β1-integrin regulates ovarian cancer invasion and metastasis. Oncogene 2011, 30, 1566–1576. [Google Scholar] [CrossRef] [PubMed]
- Tan, T.Z.; Yang, H.; Ye, J.; Low, J.; Choolani, M.; Tan, D.S.; Thiery, J.P.; Huang, R.Y. CSIOVDB: A microarray gene expression database of epithelial ovarian cancer subtype. Oncotarget 2015, 6, 43843–43852. [Google Scholar] [CrossRef] [PubMed]
- Kenny, H.A.; Chiang, C.Y.; White, E.A.; Schryver, E.M.; Habis, M.; Romero, I.L.; Ladanyi, A.; Penicka, C.V.; George, J.; Matlin, K.; et al. Mesothelial cells promote early ovarian cancer metastasis through fibronectin secretion. J. Clin. Investig. 2014, 124, 4614–4628. [Google Scholar] [CrossRef]
- Yeung, T.L.; Sheng, J.; Leung, C.S.; Li, F.; Kim, J.; Ho, S.Y.; Matzuk, M.M.; Lu, K.H.; Wong, S.T.C.; Mok, S.C. Systematic Identification of Druggable Epithelial-Stromal Crosstalk Signaling Networks in Ovarian Cancer. J. Natl. Cancer Inst. 2019, 111, 272–282. [Google Scholar] [CrossRef]
- Tan, T.Z.; Miow, Q.H.; Huang, R.Y.; Wong, M.K.; Ye, J.; Lau, J.A.; Wu, M.C.; Bin Abdul Hadi, L.H.; Soong, R.; Choolani, M.; et al. Functional genomics identifies five distinct molecular subtypes with clinical relevance and pathways for growth control in epithelial ovarian cancer. EMBO Mol. Med. 2013, 5, 1051–1066. [Google Scholar] [CrossRef]
- Yip, P.; Chen, T.H.; Seshaiah, P.; Stephen, L.L.; Michael-Ballard, K.L.; Mapes, J.P.; Mansfield, B.C.; Bertenshaw, G.P. Comprehensive serum profiling for the discovery of epithelial ovarian cancer biomarkers. PLoS ONE 2011, 6, e29533. [Google Scholar] [CrossRef]
- Wang, J.P.; Hielscher, A. Fibronectin: How Its Aberrant Expression in Tumors May Improve Therapeutic Targeting. J. Cancer 2017, 8, 674–682. [Google Scholar] [CrossRef]
- Ronca, R.; Sozzani, S.; Presta, M.; Alessi, P. Delivering cytokines at tumor site: The immunocytokine-conjugated anti-EDB-fibronectin antibody case. Immunobiology 2009, 214, 800–810. [Google Scholar] [CrossRef]
- Johannsen, M.; Spitaleri, G.; Curigliano, G.; Roigas, J.; Weikert, S.; Kempkensteffen, C.; Roemer, A.; Kloeters, C.; Rogalla, P.; Pecher, G.; et al. The tumour-targeting human L19-IL2 immunocytokine: Preclinical safety studies, phase I clinical trial in patients with solid tumours and expansion into patients with advanced renal cell carcinoma. Eur. J. Cancer 2010, 46, 2926–2935. [Google Scholar] [CrossRef] [PubMed]
- Spitaleri, G.; Berardi, R.; Pierantoni, C.; De Pas, T.; Noberasco, C.; Libbra, C.; Gonzalez-Iglesias, R.; Giovannoni, L.; Tasciotti, A.; Neri, D.; et al. Phase I/II study of the tumour-targeting human monoclonal antibody-cytokine fusion protein L19-TNF in patients with advanced solid tumours. J. Cancer Res. Clin. Oncol. 2013, 139, 447–455. [Google Scholar] [CrossRef] [PubMed]
- Morra, L.; Moch, H. Periostin expression and epithelial-mesenchymal transition in cancer: A review and an update. Virchows Arch. 2011, 459, 465–475. [Google Scholar] [CrossRef] [PubMed]
- Cui, D.; Huang, Z.; Liu, Y.; Ouyang, G. The multifaceted role of periostin in priming the tumor microenvironments for tumor progression. Cell. Mol. Life Sci. 2017, 74, 4287–4291. [Google Scholar] [CrossRef] [PubMed]
- Ryner, L.; Guan, Y.; Firestein, R.; Xiao, Y.; Choi, Y.; Rabe, C.; Lu, S.; Fuentes, E.; Huw, L.Y.; Lackner, M.R.; et al. Upregulation of Periostin and Reactive Stroma Is Associated with Primary Chemoresistance and Predicts Clinical Outcomes in Epithelial Ovarian Cancer. Clin. Cancer Res. 2015, 21, 2941–2951. [Google Scholar] [CrossRef]
- Kim, S.S.; Nikoloudaki, G.E.; Michelsons, S.; Creber, K.; Hamilton, D.W. Fibronectin synthesis, but not alpha-smooth muscle expression, is regulated by periostin in gingival healing through FAK/JNK signaling. Sci. Rep. 2019, 9, 2708. [Google Scholar] [CrossRef]
- Colombo, N.; Sessa, C.; du Bois, A.; Ledermann, J.; McCluggage, W.G.; McNeish, I.; Morice, P.; Pignata, S.; Ray-Coquard, I.; Vergote, I.; et al. ESMO-ESGO consensus conference recommendations on ovarian cancer: Pathology and molecular biology, early and advanced stages, borderline tumours and recurrent diseasedagger. Ann. Oncol. 2019, 30, 672–705. [Google Scholar] [CrossRef]

| Characteristics | (Total) | Number of Samples | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Residual tumor 1 | (108) | R0 | 17 | R1 | 27 | R2 | 21 | R3 | 43 |
| CHT response (acc. to RECIST) 2 | (108) | CR | 74 | PR | 31 | NC | 1 | P | 2 |
| Histopathological type | (108) | serous | 97 | undifferentiated | 10 | other | 1 | ||
| Tumor grade | (108) | G2 | 2 | G3 | 76 | G4 * | 30 | ||
| Platinum sensitivity 3 | (108) | Highly sensitive | 22 | Moderately sensitive | 42 | Resistant | 44 | ||
| TP53 accumulation | (108) | Yes | 68 | No | 40 | ||||
| Age | (108) | ≤54 years | 56 | >54 years | 52 | ||||
| Stromal FN1 | StroMal POSTN | FN1&POSTN Score | No. of Samples | Median OS Months |
|---|---|---|---|---|
| 1 | 1 | 2 | 14 | 55.88 (range 4.8–77.83) |
| 1 | 2 | 3 | 2 | 30.92 (range 10.7–119.93) |
| 2 | 1 | 42 | ||
| 2 | 2 | 4 | 21 | 28.77 (range 7.3–100.83) |
| 1 | 3 | 0 | ||
| 3 | 1 | 5 | ||
| 2 | 3 | 5 | 1 | 26.55 (range 11.53–91.53) |
| 3 | 2 | 17 | ||
| 3 | 3 | 6 | 6 | 42.23 (range 9.87–131.17) |
| Feature | Univariate Analysis | Multivariate Analysis | ||
|---|---|---|---|---|
| HR (95% CI) | p Value | HR (95% CI) | p Value | |
| FN1 & POSTN | ||||
| Score 3–6 vs. Score 2 | 2.54 (1.22–5.28) | 0.012 | 2.16 (1.02–4.60) | 0.044 |
| Angioinvasion | ||||
| Present vs. Absent | 1.98 (1.13–3.47) | 0.017 | 1.76 (0.99–3.15) | 0.054 |
| Necrosis | ||||
| Present vs. Absent | 0.57 (0.36–0.89) | 0.016 | 0.51 (0.32–0.82) | 0.006 |
| Residual Disease | ||||
| Present vs. Absent | 2.87 (1.43–5.76) | 0.003 | 2.39 (1.18–4.83) | 0.015 |
| Feature | Univariate Analysis | Multivariate Analysis | ||
|---|---|---|---|---|
| HR (95% CI) | p Value | HR (95% CI) | p Value | |
| FN1&POSTN | ||||
| Score 2 vs. Score 3–6 | 1.46 (0.81–2.63) | 0.203 | 1.02 (0.55–1.90) | 0.93 |
| Angioinvasion | ||||
| Present vs. Absent | 2.27 (1.35–3.83) | 0.002 | 2.48 (1.42–4.32) | 0.001 |
| Necrosis | ||||
| Present vs. Lack | 0.62 (0.39–0.97) | 0.038 | 0.55 (0.34–0.88) | 0.013 |
| Residual Disease | ||||
| Present vs. Lack | 2.34 (1.29–4.25) | 0.005 | 2.17 (1.18–3.99) | 0.012 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kujawa, K.A.; Zembala-Nożyńska, E.; Cortez, A.J.; Kujawa, T.; Kupryjańczyk, J.; Lisowska, K.M. Fibronectin and Periostin as Prognostic Markers in Ovarian Cancer. Cells 2020, 9, 149. https://doi.org/10.3390/cells9010149
Kujawa KA, Zembala-Nożyńska E, Cortez AJ, Kujawa T, Kupryjańczyk J, Lisowska KM. Fibronectin and Periostin as Prognostic Markers in Ovarian Cancer. Cells. 2020; 9(1):149. https://doi.org/10.3390/cells9010149
Chicago/Turabian StyleKujawa, Katarzyna Aleksandra, Ewa Zembala-Nożyńska, Alexander Jorge Cortez, Tomasz Kujawa, Jolanta Kupryjańczyk, and Katarzyna Marta Lisowska. 2020. "Fibronectin and Periostin as Prognostic Markers in Ovarian Cancer" Cells 9, no. 1: 149. https://doi.org/10.3390/cells9010149
APA StyleKujawa, K. A., Zembala-Nożyńska, E., Cortez, A. J., Kujawa, T., Kupryjańczyk, J., & Lisowska, K. M. (2020). Fibronectin and Periostin as Prognostic Markers in Ovarian Cancer. Cells, 9(1), 149. https://doi.org/10.3390/cells9010149






