Molecular Imaging of the Dopamine Transporter
Abstract
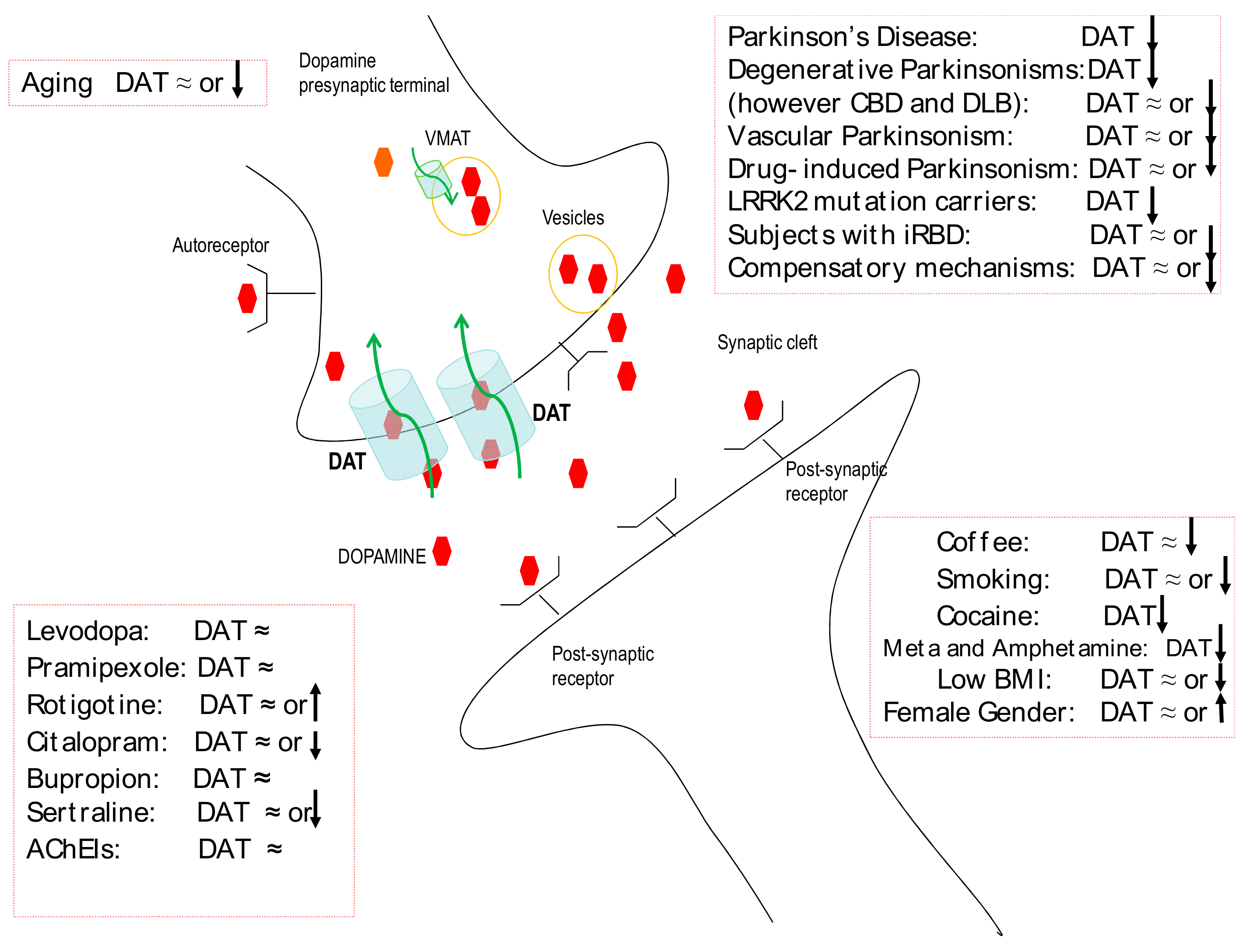
1. Dopamine Transporter Imaging: Role in Diagnosis
2. Dopamine Transporter Imaging: Drug and Habit Interferences
3. Dopamine Transporter Imaging: Relationship with Nigral Cell Counts
4. Dopamine Transporter Imaging: A Window of Compensatory Mechanisms
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Dickson, D.W. Neuropathology of Parkinson disease. Parkinsonism Relat. Disord. 2018, 46, S30–S33. [Google Scholar]
- Sulzer, D. Multiple hit hypothesis for dopamine neuron loss in Parkinson’s disease. Trends Neurosci. 2007, 30, 244–250. [Google Scholar] [CrossRef] [PubMed]
- Uhl, G.R. Dopamine transporter: Basic science and human variation of a key molecule for dopaminergic function, locomotion, and parkinsonism. Mov. Disord. 2003, 18 (Suppl. 7), S71–S80. [Google Scholar] [CrossRef]
- Sulzer, D.; Cragg, S.J.; Rice, M.E. Striatal dopamine neurotransmission: Regulation of release and uptake. Basal Ganglia 2016, 6, 123–148. [Google Scholar] [CrossRef] [PubMed]
- Mulvihill, K.G. Presynaptic regulation of dopamine release: Role of the DAT and VMAT2 transporters. Neurochem. Int. 2019, 122, 94–105. [Google Scholar] [CrossRef] [PubMed]
- Benamer, H.T.S.; Patterson, J.; Wyper, D.J.; Hadley, D.L.; Macphee, G.J.A.; Grosset, D.G. Correlation of Parkinsons disease severity and duration with [123I]FP-CIT SPECT striatal uptake. Mov. Disord. 2000, 15, 692–698. [Google Scholar] [CrossRef]
- Spiegel, J.; Hellwig, D.; Samnick, S.; Jost, W.; Möllers, M.O.; Fassbender, K.; Kirsch, C.M.; Dillmann, U. Striatal FP-CIT uptake differs in the subtypes of early Parkinson’s disease. J. Neural Transm. 2007, 114, 331–335. [Google Scholar] [CrossRef] [PubMed]
- Rossi, C.; Frosini, D.; Volterrani, D.; De Feo, P.; Unti, E.; Nicoletti, V.; Kiferle, L.; Bonuccelli, U.; Ceravolo, R. Differences in nigro-striatal impairment in clinical variants of early Parkinson’s disease: Evidence from a FP-CIT SPECT study. Eur. J. Neurol. 2010, 17, 626–630. [Google Scholar] [CrossRef]
- Booij, T.; Tissingh, G.; Boer, G.; Speelman, J.D.; Stoof, J.C.; Janssen, A.G.; Wolters, E.C.; van Royen, E.A. [123I]FP-SPECT shows a pronounced decline of striatal dopamine transporter labelling in early and advanced Parkinson’s disease. J. Neurol. Neurosurg. Psychiatry 1997, 62, 133–140. [Google Scholar] [CrossRef]
- Mozley, P.D.; Schneider, J.S.; Acton, P.D.; Plössl, K.; Stern, M.B.; Siderowf, A.; Leopold, N.A.; Li, P.Y.; Alavi, A.; Kung, H.F. Binding of [99mTc- ]TRODAT-1 to dopamine transporters in patients with Parkinson’s disease and in healthy volunteers. J. Nucl. Med. 2000, 41, 584–589. [Google Scholar]
- Marek, K.; Innis, R.B.; Van Dyck, C.H.; Fussell, B.; Early, M.; Eberly, S.; Oakes, D.; Seibyl, J. [123I]beta-CIT SPECT imaging assessment of the rate of Parkinson’s disease progression. Neurology 2001, 57, 2089–2094. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Im, J.H.; Yang, S.O.; Moon, D.H.; Ryu, J.S.; Bong, J.K.; Nam, K.P.; Cheon, J.H.; Lee, M.C.; Lee, H.K. Imaging and quantitation of dopamine transporters with iodine-123-IPT in normal and Parkinson’s disease subjects. J. Nucl. Med. 1997, 38, 1703–1711. [Google Scholar] [PubMed]
- Tatsch, K.; Poepperl, G. Nigrostriatal dopamine terminal imaging with dopamine transporter SPECT: An update. J. Nucl. Med. 2013, 54, 1331–1338. [Google Scholar] [CrossRef] [PubMed]
- Saeed, U.; Compagnone, J.; Aviv, R.I.; Strafella, A.P.; Black, S.E.; Lang, A.E.; Masellis, M. Imaging biomarkers in Parkinson’s disease and Parkinsonian syndromes: Current and emerging concepts. Transl. Neurodegener. 2017, 6, 8. [Google Scholar] [CrossRef] [PubMed]
- Bajaj, N.; Hauser, R.A.; Grachev, I.D. Clinical utility of dopamine transporter single photon emission CT (DaT-SPECT) with (123I) ioflupane in diagnosis of parkinsonian syndromes. J. Neurol. Neurosurg. Psychiatry 2013, 84, 1288–1295. [Google Scholar] [CrossRef] [PubMed]
- Gerschlager, W.; Bencsits, G.; Pirker, W.; Bloem, B.R.; Asenbaum, S.; Prayer, D.; Zijlmans, J.C.; Hoffmann, M.; Brücke, T. [123I]beta-CIT SPECT distinguishes vascular parkinsonism from Parkinson’s disease. Mov. Disord. 2002, 17, 518–523. [Google Scholar] [CrossRef]
- Lorberboym, M.; Djaldetti, R.; Melamed, E.; Sadeh, M.; Lampl, Y. 123I-FP-CIT SPECT imaging of dopamine transporters in patients with cerebrovascular disease and clinical diagnosis of vascular parkinsonism. J. Nucl. Med. 2004, 45, 1688–1693. [Google Scholar] [PubMed]
- Antonini, A.; Vitale, C.; Barone, P.; Cilia, R.; Righini, A.; Bonuccelli, U.; Abbruzzese, G.; Ramat, S.; Petrone, A.; Quatrale, R.; et al. The relationship between cerebral vascular disease and parkinsonism: The VADO study. Parkinsonism Relat. Disord. 2012, 18, 775–780. [Google Scholar] [CrossRef]
- Benítez-Rivero, S.; Marín-Oyaga, V.A.; García-Solís, D.; Huertas-Fernández, I.; García-Gómez, F.J.; Jesús, S.; Cáceres, M.T.; Carrillo, F.; Ortiz, A.M.; Carballo, M.; et al. Clinical features and 123I-FP-CIT SPECT imaging in vascular parkinsonism and Parkinson’s disease. J. Neurol. Neurosurg. Psychiatry 2013, 84, 122–129. [Google Scholar] [CrossRef]
- Tolosa, E.; Coelho, M.; Gallardo, M. DAT imaging in drug-induced and psychogenic parkinsonism. Mov. Disord. 2003, 18 (Suppl. 7), 28–33. [Google Scholar] [CrossRef]
- Lorberboym, M.; Treves, T.A.; Melamed, E.; Lampl, Y.; Hellmann, M.; Djaldetti, E. [123I]-FP/CIT SPECT imaging for distinguishing drug-induced parkinsonism from Parkinson’s disease. Mov. Disord. 2006, 21, 510–514. [Google Scholar] [CrossRef] [PubMed]
- Tinazzi, M.; Cipriani, A.; Matinella, A.; Cannas, A.; Solla, P.; Nicoletti, A.; Zappia, M.; Morgante, L.; Morgante, F.; Pacchetti, C.; et al. [123I]FP-CIT single photon emission computed tomography findings in drug-induced Parkinsonism. Schizophr. Res. 2012, 139, 40–45. [Google Scholar] [CrossRef] [PubMed]
- Ceravolo, R.; Antonini, A.; Volterrani, D.; Rossi, C.; Kiferle, L.; Frosini, D.; Lucetti, C.; Isaias, I.U.; Benti, R.; Murri, L.; et al. Predictive value of nigrostriatal dysfunction in isolated tremor: A clinical and SPECT study. Mov. Disord. 2008, 23, 2049–2054. [Google Scholar] [CrossRef] [PubMed]
- Novellino, F.; Arabia, G.; Bagnato, A.; Cascini, G.L.; Salsone, M.; Nicoletti, G.; Messina, D.; Morelli, M.; Paglionico, S.; Giofrè, L.; et al. Combined use of DAT-SPECT and cardiac MIBG scintigraphy in mixed tremors. Mov. Disord. 2009, 24, 2242–2248. [Google Scholar] [CrossRef] [PubMed]
- Arabia, G.; Novellino, F.; Morelli, M.; Paglionico, S.; Salsone, M.; Giofrè, L.; Pucci, F.; Bagnato, A.; Cascini, G.L.; Nicoletti, G.; et al. Mixed tremors with integrity of nigrostriatal system: A clinical and DAT-SPECT follow-up study. Mov. Disord. 2010, 25, 662–664. [Google Scholar] [CrossRef] [PubMed]
- De Verdal, M.; Renard, D.; Collombier, L.; Boudousq, V.; Kotzki, P.O.; Labauge, P.; Castelnovo, G. I123-FP-CIT single-photon emission tomography in patients with long-standing mixed tremor. Eur. J. Neurol. 2013, 20, 382–388. [Google Scholar] [CrossRef] [PubMed]
- Postuma, R.B.; Berg, D.; Stern, M.; Poewe, W.; Olanow, C.W.; Oertel, W.; Obeso, J.; Marek, K.; Litvan, I.; Lang, A.E.; et al. MDS clinical diagnostic criteria for Parkinson’s disease. Mov. Disord. 2015, 30, 1591–1601. [Google Scholar] [CrossRef]
- Erro, R.; Schneider, S.A.; Stamelou, M.; Quinn, N.P.; Bhatia, K.P. What do patients with scans without evidence of dopaminergic deficit (SWEDD) have? New evidence and continuing controversies. J. Neurol. Neurosurg. Psychiatry 2016, 87, 319–323. [Google Scholar] [CrossRef]
- Sixel-Döring, F.; Liepe, K.; Mollenhauer, B.; Trautmann, E.; Trenkwalder, C. The role of 123I-FP-CIT-SPECT in the differential diagnosis of Parkinson and tremor syndromes: A critical assessment of 125 cases. J. Neurol. 2011, 258, 2147–2154. [Google Scholar] [CrossRef]
- Marshall, V.L.; Patterson, J.; Hadley, D.M.; Grosset, K.A.; Grosset, D.G. Two-year follow-up in 150 consecutive cases with normal dopamine transporter imaging. Nucl. Med. Commun. 2006, 27, 933–937. [Google Scholar] [CrossRef]
- Wyman-Chick, K.A.; Martin, P.K.; Minar, M.; Schroeder, R.W. Cognition in patients with a clinical diagnosis of Parkinson disease and scans without evidence of dopaminergic deficit (SWEDD): 2-year follow-up. Cogn. Behav. Neurol. 2016, 29, 190–196. [Google Scholar] [CrossRef] [PubMed]
- Mille, E.; Levin, J.; Brendel, M.; Zach, C.; Barthel, H.; Sabri, O.; Bötzel, K.; Bartenstein, P.; Danek, A.; Rominger, A. Cerebral glucose metabolism and dopaminergic function in patients with corticobasal syndrome. J. Neuroimaging 2017, 27, 255–261. [Google Scholar] [CrossRef] [PubMed]
- McKeith, I.; O’Brien, J.; Walker, Z.; Tatsch, K.; Booij, J.; Darcourt, J.; Padovani, A.; Giubbini, R.; Bonuccelli, U.; Volterrani, D.; et al. Sensitivity and specificity of dopamine transporter imaging with 123I-FP-CIT SPECT in dementia with Lewy bodies: A phase III, multicentre study. Lancet Neurol. 2007, 6, 305–313. [Google Scholar] [CrossRef]
- Thomas, A.J.; Attems, J.; Colloby, S.J.; O’Brien, J.T.; McKeith, I.; Walker, R.; Lee, L.; Burn, D.; Lett, D.J.; Walker, Z. Autopsy validation of 123I-FP-CIT dopaminergic neuroimaging for the diagnosis of DLB. Neurology 2017, 88, 276–283. [Google Scholar] [CrossRef] [PubMed]
- Cilia, R.; Rossi, C.; Frosini, D.; Volterrani, D.; Siri, C.; Pagni, C.; Benti, R.; Pezzoli, G.; Bonuccelli, U.; Antonini, A.; et al. Dopamine transporter SPECT imaging in corticobasal syndrome. PLos ONE 2011, 6, e18301. [Google Scholar] [CrossRef] [PubMed]
- Ceravolo, R.; Rossi, C.; Cilia, R.; Tognoni, G.; Antonini, A.; Volterrani, D.; Bonuccelli, U. Evidence of delayed nigrostriatal dysfunction in corticobasal syndrome: A SPECT follow-up study. Parkinsonism Relat. Disord. 2013, 19, 557–559. [Google Scholar] [CrossRef] [PubMed]
- Pirker, S.; Perju-Dumbrava, L.; Kovacs, G.G.; Traub-Weidinger, T.; Pirker, W. Progressive dopamine transporter binding loss in autopsy-confirmed corticobasal degeneration. J. Parkinsons Dis. 2015, 5, 907–912. [Google Scholar] [CrossRef] [PubMed]
- Van der Zande, J.J.; Booij, J.; Scheltens, P.; Raijmakers, P.G.; Lemstra, A.W. [(123)]FP-CIT SPECT scans initially rated as normal became abnormal over time in patients with probable dementia with Lewy bodies. Eur. J. Nucl. Med. Mol. Imaging 2016, 43, 1060–1066. [Google Scholar] [CrossRef]
- Filippi, L.; Manni, C.; Pierantozzi, M.; Brusa, L.; Danieli, R.; Stanzione, P.; Schillaci, O. 123I-FP-CIT in progressive supranuclear palsy and in Parkinson’s disease: A SPECTsemiquantitative study. Nucl. Med. Commun. 2006, 27, 381–386. [Google Scholar] [CrossRef]
- Perju-Dumbrava, L.D.; Kovacs, G.G.; Pirker, S.; Jellinger, K.; Hoffmann, M.; Asenbaum, S.; Pirker, W. Dopamine transporter imaging in autopsy-confirmed Parkinson’s disease and multiple system atrophy. Mov. Disord. 2012, 27, 65–71. [Google Scholar] [CrossRef]
- Matesan, M.; Gaddikeri, S.; Longfellow, K.; Miyaoka, R.; Elojeimy, S.; Elman, S.; Hu, S.C.; Minoshima, S.; Lewis, D. I-123 DaTscan SPECT Brain Imaging in Parkinsonian Syndromes: Utility of the Putamen-to-Caudate Ratio. J. Neuroimaging 2018, 28, 629–634. [Google Scholar] [CrossRef]
- Kahraman, D.; Eggers, C.; Schicha, H.; Timmermann, L.; Schmidt, M. Visual assessment of dopaminergic degeneration pattern in 123I-FP-CIT SPECT differentiates patients with atypical parkinsonian syndromes and idiopathic Parkinson’s disease. J. Neurol. 2012, 259, 251–260. [Google Scholar] [CrossRef] [PubMed]
- Davidsson, A.; Georgiopoulos, C.; Dizdar, N.; Granerus, G.; Zachrisson, H. Comparison between visual assessment of dopaminergic degeneration pattern and semi-quantitative ratio calculations in patients with Parkinson’s disease and atypical parkinsonian syndromes using DaTSCAN (R) SPECT. Ann. Nucl. Med. 2014, 28, 851–859. [Google Scholar] [CrossRef] [PubMed]
- Brooks, D.J. Imaging of genetic and degenerative disorders primarily causing Parkinsonism. Handb. Clin. Neurol. 2016, 135, 493–505. [Google Scholar] [PubMed]
- Torres, G.E. The dopamine transporter proteome. J. Neurochem. 2006, 97 (Suppl 1), 3–10. [Google Scholar] [CrossRef]
- Ikawa, K.; Watanabe, A.; Kaneno, S.; Toru, M. Modulation of [3H]mazindol binding sites in rat striatum by dopaminergic agents. Eur. J. Pharmacol. 1993, 250, 261–266. [Google Scholar] [CrossRef]
- Rioux, L.; Frohna, P.A.; Joyce, J.N.; Schneider, J.S. The effects of chronic levodopa treatment on pre- and postsynaptic markers of dopaminergic function in striatum of parkinsonian monkeys. Mov. Disord. 1997, 12, 148–158. [Google Scholar] [CrossRef]
- Murer, M.G.; Dziewczapolski, G.; Menalled, L.B.; García, M.C.; Agid, Y.; Gershanik, O.; Raisman-Vozari, R. Chronic levodopa is not toxic for remaining dopamine neurons, but instead promotes their recovery, in rats with moderate nigrostriatal lesions. Ann. Neurol. 1998, 43, 561–575. [Google Scholar] [CrossRef]
- Gnanalingham, K.K.; Robertson, R.G. The effects of chronic continuous versus intermittent levodopa treatments on striatal and extrastriatal D1 and D2 dopamine receptors and dopamine uptake sites in the 6-hydroxydopamine lesioned rat–an autoradiographic study. Brain Res. 1994, 640, 185–194. [Google Scholar] [CrossRef]
- Innis, R.B.; Marek, K.L.; Sheff, K.; Zoghbi, S.; Castronuovo, J.; Feigin, A.; Seibyl, J.P. Effect of treatment with Levodopa-Carbidopa or L-Selegiline on striatal dopamine transporter SPECT imaging with I [123]Beta-CIT. Mov. Disord. 1999, 14, 436–442. [Google Scholar] [CrossRef]
- Guttman, M.; Stewart, D.; Hussey, D.; Wilson, A.; Houle, S.; Kish, S. Influence of L-Dopa and pramipexole on striatal dopamine transporter in early PD. Neurology 2001, 56, 1559–1564. [Google Scholar] [CrossRef] [PubMed]
- Ahlskog, J.E.; Uitti, R.J.; O’Connor, M.K.; Maraganore, D.M.; Matsumoto, J.Y.; Stark, K.F.; Turk, M.F.; Burnett, O.L. The effect of dopamine agonist therapy on dopamine transporter imaging in Parkinson’s disease. Mov. Disord. 1999, 14, 940–946. [Google Scholar] [CrossRef]
- Parkinson Study Group. Dopamine transporter brain imaging to assess the effects of pramipexole vs. levodopa on Parkinson disease progression. JAMA 2002, 287, 1653–1661. [Google Scholar] [CrossRef] [PubMed]
- Bolan, E.A.; Kivell, B.; Jaligam, V.; Oz, M.; Jayanthi, L.D.; Han, Y.; Sen, N.; Urizar, E.; Gomes, I.; Devi, L.A.; et al. D2 receptors regulate dopamine transporter function via an extracellular signal-regulated kinases 1 and 2-dependent and phosphoinositide 3 kinase-independent mechanism. Mol. Pharmacol. 2007, 7, 1222–1232. [Google Scholar] [CrossRef] [PubMed]
- Castro-Hernandez, J.; Afonso-Oramas, D.; Cruz-Munos, I.; Salas-Hernández, J.; Barroso-Chinea, P.; Moratalla, R.; Millan, M.J.; González-Hernández, T. Prolonged treatment with pramipexole promotes physical interaction of striatal dopamine D3 autoreceptors with dopamine transporters to reduce dopamine uptake. Neurobiol. Dis. 2015, 74, 325–335. [Google Scholar] [CrossRef] [PubMed]
- Stouffer, M.A.; Ali, S.; Reith, M.E.A.; Patel, J.C.; Sarti, F.; Carr, K.D.; Rice, M.E. SKF-83566, a D1 dopamine receptor antagonist, inhibits the dopamine transporter. J. Neurochem. 2011, 118, 714–720. [Google Scholar] [CrossRef] [PubMed]
- Rossi, C.; Genovesi, D.; Marzullo, P.; Giorgetti, A.; Filidei, E.; Corsini, G.U.; Bonuccelli, U.; Ceravolo, R. Striatal dopamine transporter modulation after rotigotine: Results from a pilot single-photon emission computed tomography study in a group of early stage Parkinson disease patients. Clin. Neuropharmacol. 2017, 40, 34–36. [Google Scholar] [CrossRef]
- Ikeda, K.; Ebina, J.; Kawabe, K.; Iwasaki, Y. Dopamine Transporter Imaging in Parkinson Disease: Progressive Changes and Therapeutic Modification after Anti-parkinsonian Medications. Intern. Med. 2019. Epub ahead of print. [Google Scholar] [CrossRef]
- Kugaya, A.; Seneca, N.M.; Snyder, P.J.; Williams, S.A.; Malison, R.T.; Baldwin, R.M.; Seibyl, J.P.; Innis, R.B. Changes in human in vivo serotonin and dopamine transporter availabilities during chronic antidepressant administration. Neuropsychopharmacology 2003, 28, 413–420. [Google Scholar] [CrossRef]
- Wu, C.K.; Chin Chen, K.; See Chen, P.; Chiu, N.T.; Yeh, T.L.; Lee, I.H.; Yang, Y.K. No changes in striatal dopamine transporter in antidepressant-treated patients with major depression. Int. Clin. Psychopharmacol. 2013, 28, 141–144. [Google Scholar] [CrossRef]
- Taylor, J.P.; Colloby, S.J.; McKeith, I.G.; Burn, D.J.; Williams, D.; Patterson, J.; O’Brien, J.T. Cholinesterase inhibitor use does not significantly influence the ability of 123I-FP-CIT imaging to distinguish Alzheimer’s disease from dementia with Lewy bodies. J. Neurol. Neurosurg. Psychiatry 2007, 78, 1069–1071. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Gigante, A.F.; Asabella, A.N.; Iliceto, G.; Martino, T.; Ferrari, C.; Defazio, G.; Rubini, G. Chronic coffee consumption and striatal DAT-SPECT findings in Parkinson’s disease. Neurol. Sci. 2018, 39, 551–555. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.K.; Yao, W.J.; Yeh, T.L.; Lee, I.H.; Chen, P.S.; Lu, R.B.; Chiu, N.T. Decreased dopamine transporter availability in male smokers—A dual isotope SPECT study. Prog. Neuropsychopharmacol. Biol. Psychiatry 2008, 32, 274–279. [Google Scholar] [CrossRef] [PubMed]
- Ashok, A.H.; Mizuno, Y.; Howes, O.D. Tobacco smoking and dopaminergic function in humans: A meta-analysis of molecular imaging studies. Psychopharmacology 2019, 236, 1119–1129. [Google Scholar] [CrossRef] [PubMed]
- Proebstl, L.; Kamp, F.; Manz, K.; Krause, D.; Adorjan, K.; Pogarell, O.; Koller, G.; Soyka, M.; Falkai, P.; Kambeitz, J. Effects of stimulant drug use on the dopaminergic system: A systematic review and meta-analysis of in vivo neuroimaging studies. Eur. Psychiatry 2019, 59, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Bezard, E.; Dovero, S.; Prunier, C.; Ravenscroft, P.; Chalon, S.; Guilloteau, D.; Crossman, A.R.; Bioulac, B.; Brotchie, J.M.; Gross, C.E. Relationship between the appearance of symptoms and the level of nigrostriatal degeneration in a progressive 1-methyl-4-phenyl-1,2,3,6 tetrahydropyridinelesioned macaque model of Parkinson’s disease. J. Neurosci. 2001, 21, 6853–6861. [Google Scholar] [CrossRef] [PubMed]
- Colloby, S.; McParland, S.; O’Brien, J.T.; Attems, J. Neuropathological correlates of dopaminergic imaging in Alzheimer’s disease and Lewy body dementias. Brain 2012, 135, 2798–2808. [Google Scholar] [CrossRef] [PubMed]
- Kraemmer, J.; Kovacs, G.G.; Perju-Dumbrava, L.; Pirker, S.; Traub-Weidinger, T.; Pirker, W. Correlation of striatal dopamine transporter imaging with post mortem substantia nigra cell counts. Mov. Disord. 2014, 29, 1767–1773. [Google Scholar] [CrossRef]
- Karimi, M.; Tian, L.; Brown, C.A.; Flores, H.P.; Loftin, S.K.; Videen, T.O.; Moerlein, S.M.; Perlmutter, J.S. Validation of nigrostriatal positron emission tomography measures: Critical limits. Ann. Neurol. 2013, 73, 390–396. [Google Scholar] [CrossRef]
- Saari, L.; Kivinen, K.; Gardberg, M.; Joutsa, J.; Noponen, T.; Kaasinen, V. Dopamine transporter imaging does not predict the number of nigral neurons in Parkinson disease. Neurology 2017, 88, 1461–1467. [Google Scholar] [CrossRef]
- Sasaki, M.; Shibata, E.; Tohyama, K.; Takahashi, J.; Otsuka, K.; Tsuchiya, K.; Takahashi, S.; Ehara, S.; Terayama, Y.; Sakai, A. Neuromelanin magnetic resonance imaging of locus ceruleus and substantia nigra in Parkinson’s disease. Neuroreport 2006, 17, 1215–1218. [Google Scholar] [CrossRef] [PubMed]
- Ohtsuka, C.; Sasaki, M.; Konno, K.; Koide, M.; Kato, K.; Takahashi, J.; Takahashi, S.; Kudo, K.; Yamashita, F.; Terayama, Y. Changes in substantia nigra and locus coeruleus in patients with early-stage Parkinson’s disease using neuromelanin-sensitive MR imaging. Neurosci. Lett. 2013, 29, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Ogisu, K.; Kudo, K.; Sasaki, M.; Sakushima, K.; Yabe, I.; Sasaki, H.; Terae, S.; Nakanishi, M.; Shirato, H. 3D neuromelanin-sensitive magnetic resonance imaging with semi-automated volume measurement of the sub stantia nigra pars compacta for diagnosis of Parkinson’s disease. Neuroradiology 2013, 55, 719–724. [Google Scholar] [CrossRef] [PubMed]
- Ohtsuka, C.; Sasaki, M.; Konno, K.; Kato, K.; Takahashi, J.; Yamashita, F.; Terayama, Y. Differentiation of early-stage parkinsonisms using neuromelanin-sensitive magnetic resonance imaging. Parkinsonism Relat. Disord. 2014, 20, 755–760. [Google Scholar] [CrossRef] [PubMed]
- Reimao, S.; Pita Lobo, P.; Neutel, D.; Correia Guedes, L.; Coelho, M.; Rosa, M.M.; Ferreira, J.; Abreu, D.; Gonçalves, N.; Morgado, C.; et al. Substantia nigra neuromelanin magnetic resonance imaging in de novo Parkinson’s disease patients. Eur. J. Neurol. 2015, 22, 540–546. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, S.T.; Xing, Y.; Tomar, P.; Bajaj, N.; Auer, D.P. In vivo assessment of brainstem depigmentation in Parkinson disease: Potential as a severity marker for multicenter studies. Radiology 2017, 283, 789–798. [Google Scholar] [CrossRef]
- Prasad, S.; Stezin, A.; Lenka, A.; George, L.; Saini, J.; Yadav, R.; Pal, P.K. Three-dimensional neuromelanin-sensitive magnetic resonance imaging of the substantia nigra in Parkinson’s disease. Eur. J. Neurol. 2018, 25, 680–686. [Google Scholar] [CrossRef]
- Pavese, N.; Tai, Y.F. Nigrosome Imaging and Neuromelanin Sensitive MRI in Diagnostic Evaluation of Parkinsonism. Mov. Disord. Clin. Pract. 2018, 22, 131–140. [Google Scholar] [CrossRef]
- Reimao, S.; Pita Lobo, P.; Neutel, D.; Guedes, L.C.; Coelho, M.; Rosa, M.M.; Azevedo, P.; Ferreira, J.; Abreu, D.; Gonçalves, N.; et al. Substantia nigra neuromelanin-MR imaging differentiates essential tremor from Parkinson’s disease. Mov. Disord. 2015, 30, 953–959. [Google Scholar] [CrossRef]
- Kashihara, K.; Shinya, T.; Higaki, F. Neuromelanin magnetic resonance imaging of nigral volume loss in patients with Parkinson’s disease. J. Clin. Neurosci. 2011, 18, 1093–1096. [Google Scholar] [CrossRef]
- Miyoshi, F.; Ogawa, T.; Kitao, S.I.; Kitayama, M.; Shinohara, Y.; Takasugi, M.; Fujii, S.; Kaminou, T. Evaluation of Parkinson disease and Alzheimer disease with the use of neuromelanin MR imaging and (123)Imetaiodobenzylguanidine scintigraphy. AJNR Am. J. Neuroradiol. 2013, 34, 2113–2118. [Google Scholar] [CrossRef]
- Kitao, S.; Matsusue, E.; Fujii, S.; Miyoshi, F.; Kaminou, T.; Kato, S.; Ito, H.; Ogawa, T. Correlation between pathology and neuromelanin MR imaging in Parkinson’s disease and dementia with Lewy bodies. Neuroradiology 2013, 55, 947–953. [Google Scholar] [CrossRef] [PubMed]
- Martín-Bastida, A.; Lao-Kaim, N.P.; Roussakis, A.A.; Searle, G.E.; Xing, Y.; Gunn, R.N.; Schwarz, S.T.; Barker, R.A.; Auer, D.P.; Piccini, P. Relationship between neuromelanin and dopamine terminals within the Parkinson’s nigrostriatal system. Brain 2019. Epub ahead of print. [Google Scholar]
- Kuya, K.; Shinohara, Y.; Miyoshi, F.; Fujii, S.; Tanabe, Y.; Ogawa, T. Correlation between neuromelanin-sensitive MR imaging and (123)I-FP-CIT SPECT in patients with parkinsonism. Neuroradiology 2016, 58, 351–356. [Google Scholar] [CrossRef] [PubMed]
- Isaias, I.U.; Trujillo, P.; Summers, P.; Marotta, G.; Mainardi, L.; Pezzoli, G.; Zecca, L.; Costa, A. Neuromelanin imaging and dopaminergic loss in Parkinson’s disease. Front. Aging Neurosci. 2016, 8, 196. [Google Scholar] [CrossRef] [PubMed]
- Lehericy, S.; Sharman, M.A.; Dos Santos, C.L.; Paquin, R.; Gallea, C. Magnetic resonance imaging of the substantia nigra in Parkinson’s disease. Mov. Disord. 2012, 27, 822–830. [Google Scholar] [CrossRef] [PubMed]
- Cosottini, M.; Frosini, D.; Pesaresi, I.; Costagli, M.; Biagi, L.; Ceravolo, R.; Bonuccelli, U.; Tosetti, M. MR imaging of the substantia nigra at 7 T enables diagnosis of Parkinson disease. Radiology 2014, 271, 831–838. [Google Scholar] [CrossRef]
- Blazejewska, A.I.; Schwarz, S.T.; Pitiot, A.; Stephenson, M.C.; Lowe, J.; Bajaj, N.; Bowtell, R.W.; Auer, D.P.; Gowland, P.A. Visualization of nigrosome 1 and its loss in PD: Pathoanatomical correlation and in vivo 7 T MRI. Neurology 2013, 81, 534–540. [Google Scholar] [CrossRef]
- Lehericy, S.; Vaillancourt, D.E.; Seppi, K.; Monchi, O.; Rektorova, I.; Antonini, A.; McKeown, M.J.; Masellis, M.; Berg, D.; Rowe, J.B.; et al. The role of high-field magnetic resonance imaging in parkinsonian disorders: Pushing the boundaries forward. Mov. Disord. 2017, 32, 510–525. [Google Scholar] [CrossRef]
- Postuma, R.B.; Berg, D. Advances in markers of prodromal Parkinson disease. Nat. Rev. Neurol. 2016, 12, 622–634. [Google Scholar] [CrossRef]
- Iranzo, A.; Lomeña, F.; Stockner, H.; Valldeoriola, F.; Vilaseca, I.; Salamero, M.; Molinuevo, J.L.; Serradell, M.; Duch, J.; Pavía, J.; et al. Decreased striatal dopamine transporter uptake and substantia nigra hyperechogenicity as risk markers of synucleinopathy in patients with idiopathic rapid-eye-movement sleep behaviour disorder: A prospective study. Lancet Neurol. 2010, 9, 1070–1077. [Google Scholar] [CrossRef]
- Li, Y.; Kang, W.; Yang, Q.; Zhang, L.; Zhang, L.; Dong, F.; Chen, S.; Liu, J. Predictive markers for early conversion of iRBD to neurodegenerative synucleinopathy diseases. Neurology 2017, 88, 1493–1500. [Google Scholar] [CrossRef] [PubMed]
- Iranzo, A.; Santamaria, J.; Valldeoriola, F.; Serradell, M.; Salamero, M.; Gaig, C.; Niñerola-Baizán, A.; Sánchez-Valle, R.; Lladó, A.; De Marzi, R.; et al. Dopamine transporter imaging deficit predicts early transition to synucleinopathy in idiopathic REM sleep behavior disorder. Ann. Neurol. 2017, 82, 419–428. [Google Scholar] [CrossRef] [PubMed]
- Ceravolo, R.; Antonini, A.; Frosini, D.; De Iuliis, A.; Weis, L.; Cecchin, D.; Tosetti, M.; Bonuccelli, U.; Cosottini, M. Nigral anatomy and striatal denervation in genetic parkinsonism: A family report. Mov. Disord. 2015, 30, 1148–1149. [Google Scholar] [CrossRef] [PubMed]
- De Marzi, R.; Seppi, K.; Hogl, B.; Müller, C.; Scherfler, C.; Stefani, A.; Iranzo, A.; Tolosa, E.; Santamarìa, J.; Gizewski, E.; et al. Loss of dorsolateral nigral hyperintensity on 3.0 tesla susceptibility-weighted imaging in idiopathic rapid eye movement sleep behavior disorder. Ann. Neurol. 2016, 79, 1026–1030. [Google Scholar] [CrossRef] [PubMed]
- Bae, Y.J.; Kim, J.M.; Kim, E.; Lee, K.M.; Kang, S.Y.; Park, H.S.; Kim, K.J.; Kim, Y.E.; Oh, E.S.; Yun, J.Y.; et al. Loss of nigral hyperintensity on 3 Tesla MRI of parkinsonism: Comparison with (123) I-FP-CIT SPECT. Mov. Disord. 2016, 31, 684–692. [Google Scholar] [CrossRef] [PubMed]
- Frosini, D.; Ceravolo, R.; Tosetti, M.; Bonuccelli, U.; Cosottini, M. Nigral involvement in atypical parkinsonisms: Evidence from a pilot study with ultra-high field MRI. J. Neural Transm. 2016, 123, 509–513. [Google Scholar] [CrossRef] [PubMed]
- Frosini, D.; Cosottini, M.; Donatelli, G.; Costagli, M.; Biagi, L.; Pacchetti, C.; Terzaghi, M.; Cortelli, P.; Arnaldi, D.; Bonanni, E.; et al. Seven tesla MRI of the substantia nigra in patients with rapid eye movement sleep behavior disorder. Parkinsonism Relat. Disord. 2017, 43, 105–109. [Google Scholar] [CrossRef]
- Lee, C.S.; Samii, A.; Sossi, V.; Ruth, T.J.; Schulzer, M.; Holden, J.E.; Wudel, J.; Pal, P.K.; de la Fuente-Fernandez, R.; Calne, D.B.; et al. In vivo positron emission tomographic evidence for compensatory changes in presynaptic dopaminergic nerve terminals in Parkinson’s disease. Ann. Neurol. 2000, 47, 493–503. [Google Scholar] [CrossRef]
- Schirinzi, T.; Madeo, G.; Martella, G.; Maltese, M.; Picconi, B.; Calabresi, P.; Pisani, A. Early synaptic dysfunction in Parkinson’s disease: Insights from animal models. Mov. Disord. 2016, 31, 802–813. [Google Scholar] [CrossRef]
- Cheng, H.C.; Ulane, C.M.; Burke, R.E. Clinical progression in Parkinson disease and the neurobiology of axons. Ann. Neurol. 2010, 67, 715–725. [Google Scholar] [CrossRef] [PubMed]
- Dijkstra, A.A.; Ingrassia, A.; de Menezes, R.X.; van Kesteren, R.E.; Rozemuller, A.J.; Heutink, P.; van de Berg, W.D. Evidence for Immune Response, Axonal Dysfunction and Reduced Endocytosis in the Substantia Nigra in Early Stage Parkinson’s Disease. PLos ONE 2015, 10, e0128651. [Google Scholar] [CrossRef] [PubMed]
- Hornykiewicz, O. Biochemical aspects of Parkinson’s disease. Neurology 1998, 51, S2–S9. [Google Scholar] [CrossRef] [PubMed]
- Sian-Hulsmann, J.; Monoranu, C.; Strobel, S.; Riederer, P. Lewy bodies: A spectator or salient killer? Cns Neurol. Disord. Drug Targets 2015, 14, 947–955. [Google Scholar] [CrossRef] [PubMed]
- Simuni, T.; Siderowf, A.; Lasch, S.; Coffey, C.S.; Caspell-Garcia, C.; Jennings, D.; Tanner, C.M.; Trojanowski, J.Q.; Shaw, L.M.; Seibyl, J.; et al. Parkinson’s Progression Marker Initiative. Longitudinal change of clinical and biological measures in early Parkinson’s disease: Parkinson’s Progression Markers Initiative cohort. Mov. Disord. 2018, 33, 771–782. [Google Scholar] [CrossRef] [PubMed]
- Kordower, J.H.; Olanow, C.W.; Dodiya, H.B.; Chu, Y.; Beach, T.G.; Adler, C.H.; Halliday, G.M.; Bartus, R.T. Disease duration and the integrity of the nigrostriatal system in Parkinson’s disease. Brain 2013, 136, 2419–2431. [Google Scholar] [CrossRef] [PubMed]
- Bridi, J.C.; Hirth, F. Mechanisms of α-Synuclein Induced Synaptopathy in Parkinson’s Disease. Front. Neurosci. 2018, 12, 80. [Google Scholar] [CrossRef]
- Phan, J.A.; Stokholm, K.; Zareba-Paslawska, J.; Jakobsen, S.; Vang, K.; Gjedde, A.; Landau, A.M.; Romero-Ramos, M. Early synaptic dysfunction induced by α-synuclein in a rat model of Parkinson’s disease. Sci. Rep. 2017, 7, 6363. [Google Scholar] [CrossRef]
- Ross, G.W.; Petrovitch, H.; Abbott, R.D.; Nelson, J.; Markesbery, W.; Davis, D.; Hardman, J.; Launer, L.; Masaki, K.; Tanner, C.M.; et al. Parkinsonian signs and substantia nigra neuron density in decendents elders without PD. Ann. Neurol. 2004, 56, 532–539. [Google Scholar] [CrossRef]
- Dorsey, E.R.; Sherer, T.; Okun, M.S.; Bloem, B.R. The Emerging Evidence of the Parkinson Pandemic. J. Parkinsons Dis. 2018, 8, S3–S8. [Google Scholar] [CrossRef]
- Tysnes, O.B.; Storstein, A. Epidemiology of Parkinson’s disease. J. Neural Transm. 2017, 124, 901–905. [Google Scholar] [CrossRef] [PubMed]
- Kempster, P.A.; O’Sullivan, S.S.; Holton, J.L.; Revesz, T.; Lees, A.J. Relationships between age and late progression of Parkinson’s disease: A clinico-pathological study. Brain 2010, 133, 1755–1762. [Google Scholar] [CrossRef] [PubMed]
- Kish, S.J.; Shannak, K.; Rajput, A.; Deck, J.H.; Hornykiewicz, O. Aging produces a specific pattern of striatal dopamine loss: Implications for the etiology of idiopathic Parkinson’s disease. J. Neurochem. 1992, 58, 642–648. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Muros, I.; Afonso-Oramas, D.; Abreu, P.; Pérez-Delgado, M.M.; Rodríguez, M.; González-Hernández, T. Aging effects on the dopamine transporter expression and compensatory mechanisms. Neurobiol. Aging 2009, 30, 973–986. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.S.; Kim, S.J.; Oh, S.J.; Kim, H.O.; Yun, S.C.; Doudet, D.; Kim, J.S. Uneven age effects of [(18)F]FP-CIT binding in the striatum of Parkinson’s disease. Ann. Nucl. Med. 2014, 28, 874–879. [Google Scholar] [CrossRef]
- Kaasinen, V.; Joutsa, J.; Noponen, T.; Johansson, J.; Seppänen, M. Effects of aging and gender on striatal and extrastriatal [123I]FP-CIT binding in Parkinson’s disease. Neurobiol. Aging 2015, 36, 1757–1763. [Google Scholar] [CrossRef] [PubMed]
- Darbin, O. The aging striatal dopamine function. Parkinsonism Relat. Disord. 2012, 18, 426–432. [Google Scholar] [CrossRef]
- Iacono, D.; Geraci-Erck, M.; Rabin, M.L.; Adler, C.H.; Serrano, G.; Beach, T.G.; Kurlan, R. Parkinson disease and incidental Lewy body disease: Just a question of time? Neurology 2015, 85, 1670–1679. [Google Scholar] [CrossRef]
- Gaig, C.; Tolosa, E. When does Parkinson’s disease begin? Mov. Disord. 2009, 24 (Suppl. 2), S656–S664. [Google Scholar] [CrossRef]
- Piccini, P.; Morrish, P.K.; Turjanski, N.; Sawle, G.V.; Burn, D.J.; Weeks, R.A.; Mark, M.H.; Maraganore, D.M.; Lees, A.J.; Brooks, D.J. Dopaminergic function in familial Parkinson’s disease: A clinical and [18F]dopa positron emission tomography study. Ann. Neurol. 1997, 41, 222–229. [Google Scholar] [CrossRef]
- Piccini, P.; Burn, D.J.; Ceravolo, R.; Maraganore, D.; Brooks, D.J. The role of inheritance in sporadic Parkinson’s disease: Evidence from a longitudinal study of dopaminergic function in twins. Ann. Neurol. 1999, 45, 577–582. [Google Scholar] [CrossRef]
- Adams, J.R.; van Netten, H.; Schulzer, M.; Mak, E.; Mckenzie, J.; Strongosky, A.; Sossi, V.; Ruth, T.J.; Lee, C.S.; Farrer, M.; et al. PET inLRRK2 mutations: Comparison to sporadic Parkinson’s disease and evidence for presymptomatic compensation. Brain 2005, 128, 2777–2785. [Google Scholar] [CrossRef] [PubMed]
- Beach, T.G.; Adler, C.H.; Lue, L.; Sue, L.I.; Bachalakuri, J.; Henry-Watson, J.; Sasse, J.; Boyer, S.; Shirohi, S.; Brooks, R.; et al. Unified staging system for Lewy body disorders: Correlation with nigrostriatal degeneration, cognitive impairment and motor dysfunction. Acta Neuropathol. 2009, 117, 613–634. [Google Scholar] [CrossRef] [PubMed]
- Greffard, S.; Verny, M.; Bonnet, A.M.; Beinis, J.Y.; Gallinari, C.; Meaume, S.; Piette, F.; Hauw, J.J.; Duyckaerts, C. Motor score of the Unified Parkinson Disease Rating Scale as a good predictor of Lewy body-associated neuronal loss in the substantia nigra. Arch. Neurol. 2006, 63, 584–588. [Google Scholar] [CrossRef] [PubMed]
- Zigmond, M.J.; Abercrombie, E.D.; Berger, T.W.; Grace, A.A.; Stricker, E.M. Compensations after lesions of central dopaminergic neurons: Some clinical and basic implications. Trends Neurosci. 1990, 13, 290–296. [Google Scholar] [CrossRef]
- Obeso, J.A.; Rodriguez-Oroz, M.C.; Lanciego, J.L.; Diaz, M.R.; Rodriguez Diaz, M. How does Parkinson’s disease begin? The role of compensatory mechanisms. Trends Neurosci. 2004, 27, 125–127. [Google Scholar] [CrossRef] [PubMed]
- Kagi, G.; Bhatia, K.P.; Tolosa, E. The role of DAT-SPECT in movement disorders. J. Neurol. Neurosurg. Psychiatry 2010, 81, 5–12. [Google Scholar] [CrossRef]
- Bezard, E.; Gross, C.E.; Brotchie, J.M. Presymptomatic compensation in Parkinson’s disease is not dopamine-mediated. Trends Neurosci. 2003, 26, 215–221. [Google Scholar] [CrossRef]
- Brotchie, J.; Fitzer-Attas, C. Mechanisms compensating for dopamine loss in early Parkinson disease. Neurology 2009, 72 (Suppl. 7), S32–S38. [Google Scholar] [CrossRef]
- Blesa, J.; Trigo-Damas, I.; Dileone, M.; Del Rey, N.L.; Hernandez, L.F.; Obeso, J.A. Compensatory mechanisms in Parkinson’s disease: Circuits adaptations and role in disease modification. Exp. Neurol. 2017, 298, 148–161. [Google Scholar] [CrossRef]
- De la Fuente-Fernández, R.; Pal, P.K.; Vingerhoets, F.J.; Kishore, A.; Schulzer, M.; Mak, E.K.; Ruth, T.J.; Snow, B.J.; Calne, D.B.; Stoessl, A.J. Evidence for impaired presynaptic dopamine function in parkinsonian patients with motor fluctuations. J. Neural Transm. 2000, 107, 49–57. [Google Scholar] [CrossRef] [PubMed]
- De la Fuente-Fernàndez, R.; Lu, J.Q.; Sossi, V.; Jivan, S.; Schulzer, M.; Holden, J.E.; Lee, C.S.; Ruth, T.J.; Calne, D.B.; Stoessl, A.J. Biochemical variations in the synaptic level of dopamine precede motor fluctuations in Parkinson’s disease: PET evidence of increased dopamine turnover. Ann. Neurol. 2001, 49, 298–303. [Google Scholar] [CrossRef] [PubMed]
- De la Fuente-Fernàndez, R.; Sossi, V.; Huang, Z.; Furtado, S.; Lu, J.Q.; Calne, D.B.; Ruth, T.J.; Stoessl, A.J. Levodopa induced changes in synaptic dopamine levels increase with progression of Parkinson’s disease: Implications for dyskinesias. Brain 2004, 127, 2747–2754. [Google Scholar] [CrossRef] [PubMed]
- De la Fuente-Fernàndez, R.; Schulzer, M.; Mak, E.; Calne, D.B.; Stoessl, A.J. Presynaptic mechanisms of motor fluctuations in Parkinson’s disease: A probabilistic model. Brain 2004, 127, 888–899. [Google Scholar] [CrossRef] [PubMed]
- Sossi, V.; de la Fuente-Fernández, R.; Schulzer, M.; Troiano, A.R.; Ruth, T.J.; Stoessl, A.J. Dopamine transporter relation to dopamine turnover in Parkinson’s disease: A positron emission tomography study. Ann. Neurol. 2007, 62, 468–474. [Google Scholar] [CrossRef] [PubMed]
- Stoessl, A.J. Central pharmacokinetics of levodopa: Lessons from imaging studies. Mov. Disord. 2015, 30, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Troiano, A.R.; de la Fuente-Fernàndez, R.; Sossi, V.; Schulzer, M.; Mak, E.; Ruth, T.J.; Stoessl, A.J. PET demonstrates reduced dopamine transporter expression in PD with dyskinesias. Neurology 2009, 72, 1211–1216. [Google Scholar] [CrossRef] [PubMed]
- Hong, J.Y.; Oh, J.S.; Lee, I.; Sunwoo, M.K.; Ham, J.H.; Lee, J.E.; Sohn, Y.H.; Kim, J.S.; Lee, P.H. Presynaptic dopamine depletion predicts levodopa-induced dyskinesia in de novo Parkinson disease. Neurology 2014, 82, 1597–1604. [Google Scholar] [CrossRef]
- Löhle, M.; Mende, J.; Wolz, M.; Beuthien-Baumann, B.; Oehme, L.; van den Hoff, J.; Kotzerke, J.; Reichmann, H.; Storch, A. Putaminal dopamine turnover in de novo Parkinson disease predicts later motor complications. Neurology 2016, 86, 231–240. [Google Scholar] [CrossRef]
- Tomas, D.; Stanic, D.; Chua, H.K.; White, K.; Boon, W.C.; Horne, M. Restoration of the Dopamine Transporter through Cell Therapy Improves Dyskinesia in a Rat Model of Parkinson’s Disease. PLos ONE 2016, 11, e0153424. [Google Scholar] [CrossRef]
- Yoo, H.S.; Chung, S.J.; Chung, S.J.; Moon, H.; Oh, J.S.; Kim, J.S.; Hong, J.Y.; Ye, B.S.; Sohn, Y.H.; Lee, P.H. Presynaptic dopamine depletion determines the timing of levodopa-induced dyskinesia onset in Parkinson’s disease. Eur. J. Nucl. Med. Mol. Imaging 2018, 45, 423–443. [Google Scholar] [CrossRef] [PubMed]
- Nandhagopal, R.; Kuramoto, L.; Schulzer, M.; Mak, E.; Cragg, J.; Lee, C.S.; McKenzie, J.; McCormick, S.; Samii, A.; Troiano, A.; et al. Longitudinal progression of sporadic Parkinson’s disease: A multitracer positron emission tomography study. Brain 2009, 132, 2970–2979. [Google Scholar] [CrossRef]
- Chung, S.J.; Lee, Y.; Oh, J.S.; Kim, J.S.; Lee, P.H.; Sohn, Y.H. Putaminal dopamine depletion in de novo Parkinson’s disease predicts future development of wearing-off. Parkinsonism Relatdisord. 2018, 53, 96–100. [Google Scholar] [CrossRef] [PubMed]
- Linazasoro, G.; Van Blercom, N.; Bergaretxe, A.; Iñaki, F.M.; Laborda, E.; Ruiz Ortega, J.A. Levodopa-induced dyskinesias in Parkinson disease are independent of the extent of striatal dopaminergic denervation: A pharmacological and SPECT study. Clin. Neuropharmacol. 2009, 32, 326–329. [Google Scholar] [CrossRef]
- Djaldetti, R.; Rigbi, A.; Greenbaum, L.; Reiner, J.; Lorberboym, M. Can early dopamine transporter imaging serve as a predictor of Parkinson’s disease progression and late motor complications? J. Neurol. Sci. 2018, 390, 255–260. [Google Scholar] [CrossRef] [PubMed]
- Calabresi, P.; Di Filippo, M.; Ghiglieri, V.; Tambasco, N.; Picconi, B. Levodopa-induced dyskinesias in patients with Parkinson’s disease: Filling the bench-to-bedside gap. Lancet Neurol. 2010, 9, 1106–1117. [Google Scholar] [CrossRef]
- Ravina, B.; Marek, K.; Eberly, S.; Oakes, D.; Kurlan, R.; Ascherio, A.; Beal, F.; Beck, J.; Flagg, E.; Galpern, W.R.; et al. Dopamine transporter imaging is associated with long-term outcomes in Parkinson’s disease. Mov. Disord. 2012, 27, 1392–1397. [Google Scholar] [CrossRef]
- Erro, R.; Pappatà, S.; Amboni, M.; Vicidomini, C.; Longo, K.; Santangelo, G.; Picillo, M.; Vitale, C.; Moccia, M.; Giordano, F.; et al. Anxiety is associated with striatal dopamine transporter availability in newly diagnosed untreated Parkinson’s disease patients. Parkinsonism Relat. Disord. 2012, 18, 1034–1038. [Google Scholar] [CrossRef]
- Kiferle, L.; Ceravolo, R.; Giuntini, M.; Linsalata, G.; Puccini, G.; Volterrani, D.; Bonuccelli, U. Caudate dopaminergic denervation and visual hallucinations: Evidence from a 123I-FP-CIT SPECT study. Parkinsonism Relat. Disord. 2014, 20, 761–765. [Google Scholar] [CrossRef]
- Santangelo, G.; Vitale, C.; Picillo, M.; Cuoco, S.; Moccia, M.; Pezzella, D.; Erro, R.; Longo, K.; Vicidomini, C.; Pellecchia, M.T.; et al. Apathy and striatal dopamine transporter levels in de-novo, untreated Parkinson’s disease patients. Parkinsonism Relat. Disord. 2015, 21, 489–493. [Google Scholar] [CrossRef]
- Kim, R.; Lee, J.; Kim, Y.; Kim, A.; Jang, M.; Kim, H.J.; Jeon, B.; Kang, U.J.; Fahn, S. Presynaptic striatal dopaminergic depletion predicts the later development of freezing of gait in de novo Parkinson’s disease: An analysis of the PPMI cohort. Parkinsonism Relat. Disord. 2018, 51, 49–54. [Google Scholar] [CrossRef] [PubMed]
- Kim, R.; Jun, J.S. Association of autonomic symptoms with presynaptic striatal dopamine depletion in drug-naive Parkinson’s disease: An analysis of the PPMI data. Auton. Neurosci. 2019, 216, 59–62. [Google Scholar] [CrossRef] [PubMed]
- Latourelle, J.C.; Beste, M.T.; Hadzi, T.C.; Miller, R.E.; Oppenheim, J.N.; Valko, M.P.; Wuest, D.M.; Church, B.W.; Khalil, I.G.; Hayete, B.; et al. Large-scale identification of clinical and genetic predictors of motor progression in patients with newly diagnosed Parkinson’s disease: A longitudinal cohort study and validation. Lancet Neurol. 2017, 16, 908–916. [Google Scholar] [CrossRef]
- Li, W.; Lao-Kaim, N.P.; Roussakis, A.A.; Martín-Bastida, A.; Valle-Guzman, N.; Paul, G.; Loane, C.; Widner, H.; Politis, M.; Foltynie, T. 11 C-PE2I and 18 F-Dopa PET for assessing progression rate in Parkinson’s: A longitudinal study. Mov. Disord. 2018, 33, 117–127. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.J.; Oh, J.S.; Ham, J.H.; Lee, D.H.; Lee, I.; Sohn, Y.H.; Kim, J.S.; Lee, P.H. Association of body mass index and the depletion of nigrostriatal dopamine in Parkinson’s disease. Neurobiol. Aging 2016, 38, 197–204. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Palermo, G.; Ceravolo, R. Molecular Imaging of the Dopamine Transporter. Cells 2019, 8, 872. https://doi.org/10.3390/cells8080872
Palermo G, Ceravolo R. Molecular Imaging of the Dopamine Transporter. Cells. 2019; 8(8):872. https://doi.org/10.3390/cells8080872
Chicago/Turabian StylePalermo, Giovanni, and Roberto Ceravolo. 2019. "Molecular Imaging of the Dopamine Transporter" Cells 8, no. 8: 872. https://doi.org/10.3390/cells8080872
APA StylePalermo, G., & Ceravolo, R. (2019). Molecular Imaging of the Dopamine Transporter. Cells, 8(8), 872. https://doi.org/10.3390/cells8080872



