Impact of the Recipient’s Pre-Treatment Blood Lymphocyte Count on Intended and Unintended Effects of Anti-T-Lymphocyte Globulin in Allogeneic Hematopoietic Stem Cell Transplantation
Abstract
1. Introduction
2. Materials and Methods
2.1. Definitions and Grading
2.2. Statistical Methods
3. Results
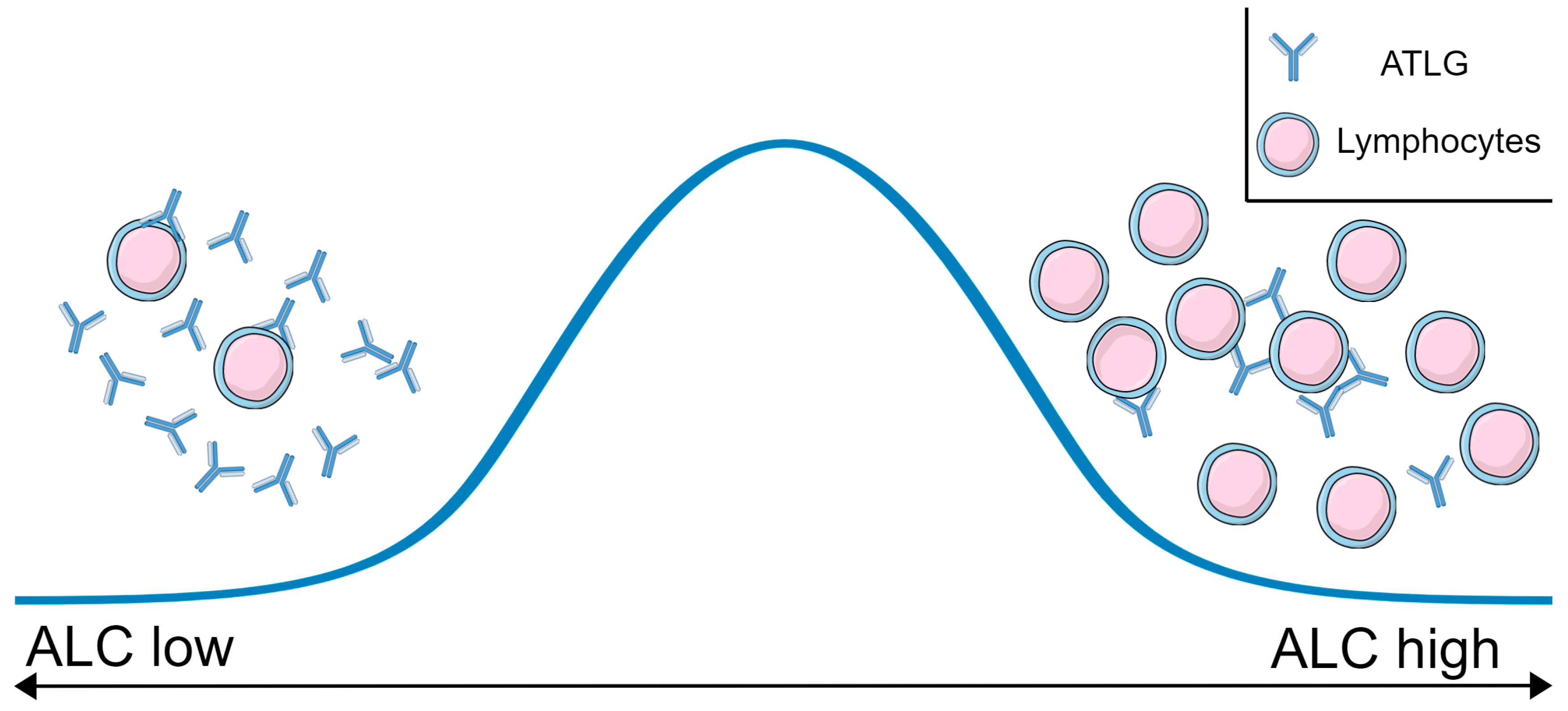
3.1. Determination of the Recipient’s ALC before First ATLG Administration
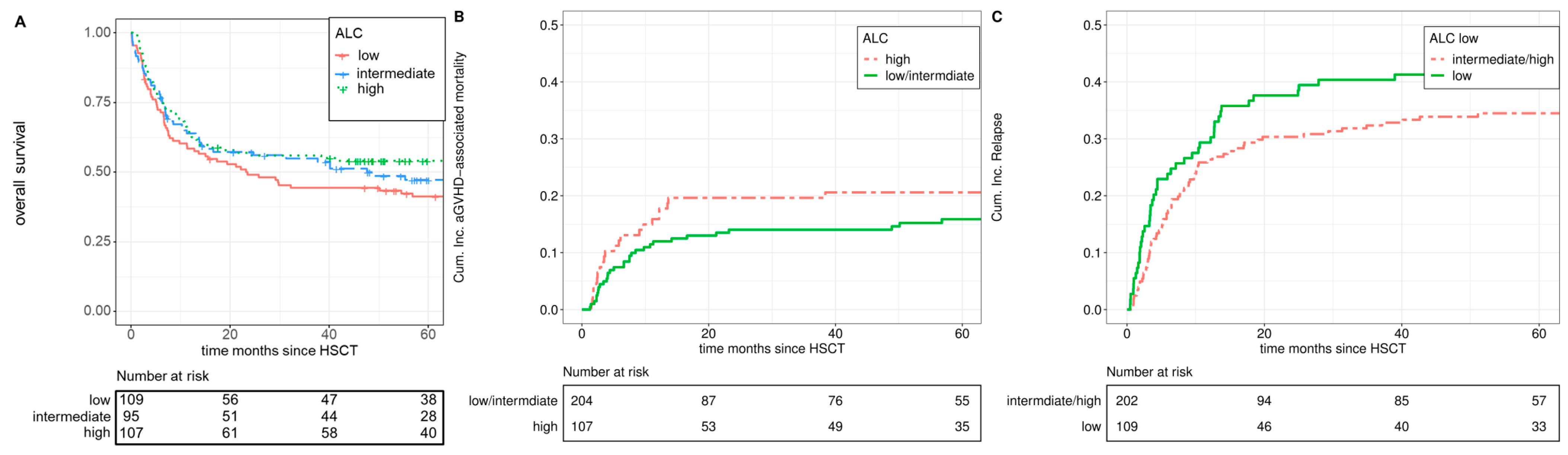
3.2. Acute GVHD Grade III-IV, aGVHD-Associated Mortality, and Chronic GVHD
3.3. Overall Survival, Non-Relapse Mortality, Incidence of Relapse, and Progression-Free Survival
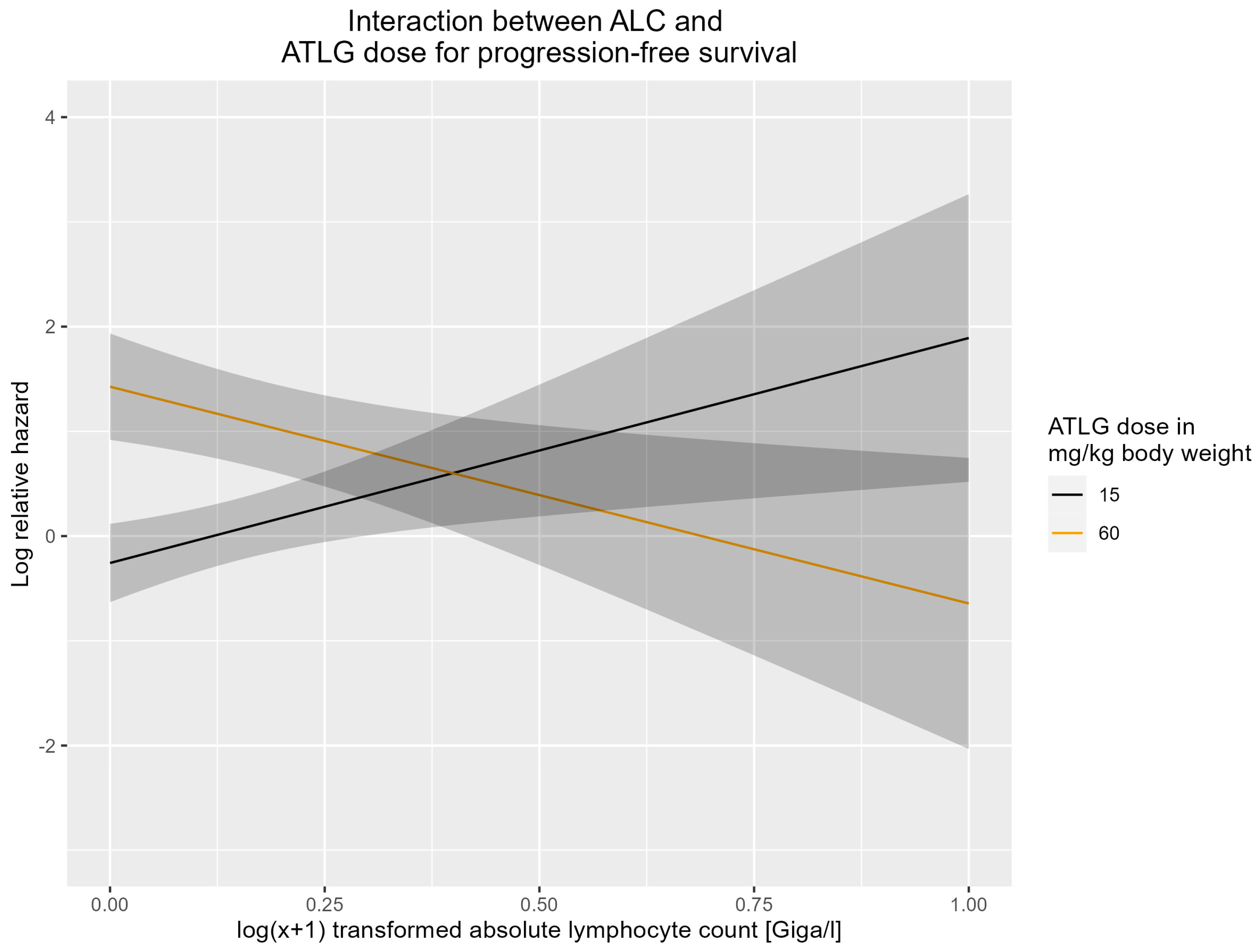
3.4. Interaction between ATLG Dose and ALC
3.5. Subgroup Analyses of Conditioning Intensity, Donor Relationship, and ATLG Dose Levels
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hale, G.; Jacobs, P.; Wood, L.; Fibbe, W.E.; Barge, R.; Novitzky, N.; Toit, C.; Abrahams, L.; Thomas, V.; Bunjes, D.; et al. CD52 antibodies for prevention of graft-versus-host disease and graft rejection following transplantation of allogeneic peripheral blood stem cells. Bone Marrow Transplant. 2000, 26, 69–76. [Google Scholar] [CrossRef]
- Finke, J.; Bethge, W.A.; Schmoor, C.; Ottinger, H.D.; Stelljes, M.; Zander, A.R.; Volin, L.; Ruutu, T.; Heim, D.A.; Schwerdtfeger, R.; et al. Standard graft-versus-host disease prophylaxis with or without anti-T-cell globulin in haematopoietic cell transplantation from matched unrelated donors: A randomised, open-label, multicentre phase 3 trial. Lancet Oncol. 2009, 10, 855–864. [Google Scholar] [CrossRef] [PubMed]
- Bacigalupo, A.; Lamparelli, T.; Bruzzi, P.; Guidi, S.; Alessandrino, P.E.; di Bartolomeo, P.; Oneto, R.; Bruno, B.; Barbanti, M.; Sacchi, N.; et al. Antithymocyte globulin for graft-versus-host disease prophylaxis in transplants from unrelated donors: 2 randomized studies from Gruppo Italiano Trapianti Midollo Osseo (GITMO). Blood 2001, 98, 2942–2947. [Google Scholar] [CrossRef] [PubMed]
- Walker, I.; Panzarella, T.; Couban, S.; Couture, F.; Devins, G.; Elemary, M.; Gallagher, G.; Kerr, H.; Kuruvilla, J.; Lee, S.J.; et al. Pretreatment with anti-thymocyte globulin versus no anti-thymocyte globulin in patients with haematological malignancies undergoing haemopoietic cell transplantation from unrelated donors: A randomised, controlled, open-label, phase 3, multicentre trial. Lancet Oncol. 2016, 17, 164–173. [Google Scholar] [CrossRef] [PubMed]
- Kröger, N.; Solano, C.; Wolschke, C.; Bandini, G.; Patriarca, F.; Pini, M.; Nagler, A.; Selleri, C.; Risitano, A.; Messina, G.; et al. Antilymphocyte Globulin for Prevention of Chronic Graft-versus-Host Disease. N. Engl. J. Med. 2016, 374, 43–53. [Google Scholar] [CrossRef]
- Clausen, J.; Böhm, A.; Straßl, I.; Stiefel, O.; Buxhofer-Ausch, V.; Machherndl-Spandl, S.; König, J.; Schmidt, S.; Steitzer, H.; Danzer, M.; et al. HLA-C KIR-Ligands Determine the Impact of Anti-Thymocyte Globulin (ATG) on Graft versus Host and Graft versus Leukemia Effects Following Hematopoietic Stem Cell Transplantation. Biomedicines 2017, 5, 13. [Google Scholar] [CrossRef]
- Soiffer, R.J.; Kim, H.T.; McGuirk, J.; Horwitz, M.E.; Johnston, L.; Patnaik, M.M.; Rybka, W.; Artz, A.; Porter, D.L.; Shea, T.C.; et al. Prospective, Randomized, Double-Blind, Phase III Clinical Trial of Anti-T-Lymphocyte Globulin to Assess Impact on Chronic Graft-Versus-Host Disease-Free Survival in Patients Undergoing HLA-Matched Unrelated Myeloablative Hematopoietic Cell Transplantation. J. Clin. Oncol. 2017, 35, 4003–4011. [Google Scholar] [CrossRef]
- Penack, O.; Marchetti, M.; Ruutu, T.; Aljurf, M.; Bacigalupo, A.; Bonifazi, F.; Ciceri, F.; Cornelissen, J.; Malladi, R.; Duarte, R.F.; et al. Prophylaxis and management of graft versus host disease after stem-cell transplantation for haematological malignancies: Updated consensus recommendations of the European Society for Blood and Marrow Transplantation. Lancet Haematol. 2020, 7, e157–e167. [Google Scholar] [CrossRef]
- Turki, A.T.; Klisanin, V.; Bayraktar, E.; Kordelas, L.; Trenschel, R.; Ottinger, H.; Steckel, N.K.; Tsachakis-Mück, N.; Leserer, S.; Ditschkowski, M.; et al. Optimizing anti-T-lymphocyte globulin dosing to improve long-term outcome after unrelated hematopoietic cell transplantation for hematologic malignancies. Am. J. Transplant. 2020, 20, 677–688. [Google Scholar] [CrossRef]
- Admiraal, R.; Nierkens, S.; de Witte, M.A.; Petersen, E.J.; Fleurke, G.-J.; Verrest, L.; Belitser, S.V.; Bredius, R.G.M.; Raymakers, R.A.P.; Knibbe, C.A.J.; et al. Association between anti-thymocyte globulin exposure and survival outcomes in adult unrelated haemopoietic cell transplantation: A multicentre, retrospective, pharmacodynamic cohort analysis. Lancet Haematol. 2017, 4, e183–e191. [Google Scholar] [CrossRef]
- Shiratori, S.; Ohigashi, H.; Ara, T.; Yasumoto, A.; Goto, H.; Nakagawa, M.; Sugita, J.; Onozawa, M.; Kahata, K.; Endo, T.; et al. High lymphocyte counts before antithymocyte globulin administration predict acute graft-versus-host disease. Ann. Hematol. 2021, 100, 1321–1328. [Google Scholar] [CrossRef] [PubMed]
- Woo, G.-U.; Hong, J.; Kim, H.; Byun, J.M.; Koh, Y.; Shin, D.-Y.; Kim, I.; Yoon, S.-S. Preconditioning Absolute Lymphocyte Count and Transplantation Outcomes in Matched Related Donor Allogeneic Hematopoietic Stem Cell Transplantation Recipients with Reduced-Intensity Conditioning and Antithymocyte Globulin Treatment. Biol. Blood Marrow Transplant. 2020, 26, 1855–1860. [Google Scholar] [CrossRef] [PubMed]
- Marsh, R.A.; Fukuda, T.; Emoto, C.; Neumeier, L.; Khandelwal, P.; Chandra, S.; Teusink-Cross, A.; Vinks, A.A.; Mehta, P.A. Pretransplant absolute lymphocyte counts impact the pharmacokinetics of alemtuzumab. Biol. Blood Marrow Transplant. 2017, 23, 635–641. [Google Scholar] [CrossRef] [PubMed]
- Giralt, S.; Ballen, K.; Rizzo, D.; Bacigalupo, A.; Horowitz, M.; Pasquini, M.; Sandmaier, B. Reduced-intensity conditioning regimen workshop: Defining the dose spectrum. Report of a workshop convened by the center for international blood and marrow transplant research. Biol. Blood Marrow Transplant. 2009, 15, 367–369. [Google Scholar] [CrossRef]
- Armand, P.; Kim, H.T.; Logan, B.R.; Wang, Z.; Alyea, E.P.; Kalaycio, M.E.; Maziarz, R.T.; Antin, J.H.; Soiffer, R.J.; Weisdorf, D.J.; et al. Validation and refinement of the Disease Risk Index for allogeneic stem cell transplantation. Blood 2014, 123, 3664–3671. [Google Scholar] [CrossRef]
- Przepiorka, D.; Weisdorf, D.; Martin, P.; Klingemann, H.G.; Beatty, P.; Hows, J.; Thomas, E.D. 1994 consensus conference on acute GVHD grading. Bone Marrow Transplant. 1995, 15, 825–828. [Google Scholar]
- Shulman, H.M.; Sullivan, K.M.; Weiden, P.L.; McDonald, G.B.; Striker, G.E.; Sale, G.E.; Hackman, R.; Tsoi, M.-S.; Storb, R.; Donnall Thomas, E. Chronic graft-versus-host syndrome in man. Am. J. Med. 1980, 69, 204–217. [Google Scholar] [CrossRef]
- Filipovich, A.H.; Weisdorf, D.; Pavletic, S.; Socie, G.; Wingard, J.R.; Lee, S.J.; Martin, P.; Chien, J.; Przepiorka, D.; Couriel, D.; et al. National Institutes of Health consensus development project on criteria for clinical trials in chronic graft-versus-host disease: I. Diagnosis and staging working group report. Biol. Blood Marrow Transplant. 2005, 11, 945–956. [Google Scholar] [CrossRef]
- Kennedy, V.E.; Chen, H.; Savani, B.N.; Greer, J.; Kassim, A.A.; Engelhardt, B.G.; Goodman, S.; Sengsayadeth, S.; Chinratanalab, W.; Jagasia, M. Optimizing antithymocyte globulin dosing for unrelated donor allogeneic hematopoietic cell transplantation based on recipient absolute lymphocyte count. Biol. Blood Marrow Transplant. 2018, 24, 150–155. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2013. [Google Scholar]
- Heelan, F.; Mallick, R.; Bryant, A.; Radhwi, O.; Atkins, H.; Huebsch, L.; Bredeson, C.; Allan, D.; Kekre, N. Does Lymphocyte Count Impact Dosing of Anti-Thymocyte Globulin in Unrelated Donor Stem Cell Transplantation? Biol. Blood Marrow Transplant. 2020, 26, 1298–1302. [Google Scholar] [CrossRef]
- Seo, J.; Shin, D.-Y.; Koh, Y.; Kim, I.; Yoon, S.-S.; Min Byun, J.; Hong, J. Association between preconditioning absolute lymphocyte count and transplant outcomes in patients undergoing matched unrelated donor allogeneic hematopoietic stem cell transplantation with reduced-intensity conditioning and anti-thymocyte globulin. Ther. Adv. Hematol. 2021, 12, 20406207211063784. [Google Scholar] [CrossRef] [PubMed]
- Sheth, V.; Kennedy, V.; de Lavallade, H.; Mclornan, D.; Potter, V.; Engelhardt, B.G.; Savani, B.; Chinratanalab, W.; Goodman, S.; Greer, J.; et al. Differential Interaction of Peripheral Blood Lymphocyte Counts (ALC) with Different in vivo Depletion Strategies in Predicting Outcomes of Allogeneic Transplant: An International 2 Center Experience. Front. Oncol. 2019, 9, 623. [Google Scholar] [CrossRef] [PubMed]
- Modi, D.; Kim, S.; Surapaneni, M.; Ayash, L.; Ratanatharathorn, V.; Uberti, J.P.; Deol, A. Absolute lymphocyte count on the first day of thymoglobulin predicts relapse-free survival in matched unrelated peripheral blood stem cell transplantation. Leuk. Lymphoma 2020, 61, 3137–3145. [Google Scholar] [CrossRef] [PubMed]
- Grasso, A.G.; Simeone, R.; Maestro, A.; Zanon, D.; Maximova, N. Pre-Transplant Total Lymphocyte Count Determines Anti-Thymocyte Globulin Exposure, Modifying Graft-versus-Host Disease Incidence and Post-Transplant Thymic Restoration: A Single-Center Retrospective Study. J. Clin. Med. 2023, 12, 730. [Google Scholar] [CrossRef]
- Admiraal, R.; van Kesteren, C.; Jol-van der Zijde, C.M.; Lankester, A.C.; Bierings, M.B.; Egberts, T.C.G.; van Tol, M.J.D.; Knibbe, C.A.J.; Bredius, R.G.M.; Boelens, J.J. Association between anti-thymocyte globulin exposure and CD4+ immune reconstitution in paediatric haemopoietic cell transplantation: A multicentre, retrospective pharmacodynamic cohort analysis. Lancet Haematol. 2015, 2, e194–e203. [Google Scholar] [CrossRef]
- Nikoloudis, A.; Strassl, I.; Binder, M.; Stiefel, O.; Wipplinger, D.; Milanov, R.; Aichinger, C.; Kaynak, E.; Machherndl-Spandl, S.; Buxhofer-Ausch, V.; et al. Comparison of Benefits and Risks Associated with Anti-T-Lymphocyte Globulin (ATLG) Serotherapy in Methotrexate (MTX)- versus Mycophenolate Mofetil (MMF)-Based Hematopoietic Stem Cell Transplantation. Transplantology 2023, 4, 22–37. [Google Scholar] [CrossRef]
| Cohort Characteristics | ||
|---|---|---|
| Factor | Group | Overall |
| n | 311 | |
| age donor (median [range]) | 35.00 [18.00, 70.20] | |
| age recipient (median [range]) | 50.60 [18.00, 73.00] | |
| ATG type (%) | ATLG | 311 (100.0) |
| ATLG dose (median [range]) | 30.00 [10.00, 60.00] | |
| ALC (median [range]) 1 | 0.07 [0.00, 13.40] | |
| ALC group (%) | lower third (tertile) | 109 (35.0) |
| middle third (tertile) | 95 (30.5) | |
| upper third (tertile) | 107 (34.4) | |
| CMV IgG status donor (%) | no | 192 (61.7) |
| yes | 119 (38.3) | |
| CMV IgG status recipient (%) | no | 150 (48.2) |
| yes | 161 (51.8) | |
| disease risk (%) | low | 104 (33.4) |
| intermediate | 81 (26.0) | |
| high | 126 (40.5) | |
| conditioning (%) | MAC 2 | 166 (53.4) |
| RIC 3 | 145 (46.6) | |
| HLA matching (%) | 10/10 matched | 239 (76.8) |
| 9/10 matched | 72 (23.2) | |
| GVHD prophylaxis (%) | CNI/MMF or other 4 | 233 (74.9) |
| CNI/MTX 5 | 78 (25.1) | |
| donor type (%) | matched related | 71 (22.8) |
| unrelated | 240 (77.2) | |
| sex mismatched (%) 6 | no | 243 (78.1) |
| yes | 68 (21.9) | |
| graft source (%) | bone marrow | 16 (5.1) |
| PBSC 7 | 294 (94.9) | |
| diagnosis (%) | MDS or MPN | 57 (18.3) |
| AML | 146 (46.9) | |
| lymphoma or myeloma | 47 (15.1) | |
| ALL | 51 (16.4) | |
| nonmalignant | 10 (3.2) | |
| Multivariate analyses of the impact of absolute lymphocyte count on study endpoints | ||
| Endpoint | (s)HR | p-value |
| overall survival * time_TX, ATLG_dose, relation, age_d, age_r, DR, mm, MTX_bsd, source, maj_AB0mm | ||
| ALC low/intermediate/high | 1.19 | 0.38 |
| progression-free survival * time_TX, DR, ATLG_dose, relation, cond, age_d, age_r, DR, cmv_d, cmv_r, mm, MTX_bsd, sex_mm, source, maj_AB0mm, min_AB0mm, mnc | ||
| ALC low/intermediate/high | 0.99 | 0.95 |
| non-relapse mortality * time_TX, ATLG_dose, DR, cmv_d, cmv_r, age_r, mm, relation, source, maj_AB0mm, mnc | ||
| ALC high | 1.41 | 0.16 |
| aGVHD-associated mortality * time_TX, lctest_Dummy, age_r, mm, relation, min_AB0mm, mnc | ||
| ALC high | 1.81 | 0.04 |
| Relapse * time_TX, ATLG_dose, age_d, age_r, DR, cmv_d, cmv_r, cond, mm, MTX_bsd, relation, sex_mm, maj_AB0mm, min_AB0mm, mnc | ||
| ALC low | 1.06 | 0.76 |
| aGVHD 2–4 * cond, DR, mnc, age_r, time_TX, ATLG_dose, relation | ||
| ALC high | 1.24 | 0.20 |
| aGVHD 3–4 * DR, mm, source, mnc | ||
| ALC high | 1.81 | 0.01 |
| cGVHD moderate/severe or extensive * ATLG_dose, DR, relation, min_AB0mm | ||
| ALC high | 1.60 | 0.06 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nikoloudis, A.; Buxhofer-Ausch, V.; Aichinger, C.; Binder, M.; Hasengruber, P.; Kaynak, E.; Wipplinger, D.; Milanov, R.; Strassl, I.; Stiefel, O.; et al. Impact of the Recipient’s Pre-Treatment Blood Lymphocyte Count on Intended and Unintended Effects of Anti-T-Lymphocyte Globulin in Allogeneic Hematopoietic Stem Cell Transplantation. Cells 2023, 12, 1831. https://doi.org/10.3390/cells12141831
Nikoloudis A, Buxhofer-Ausch V, Aichinger C, Binder M, Hasengruber P, Kaynak E, Wipplinger D, Milanov R, Strassl I, Stiefel O, et al. Impact of the Recipient’s Pre-Treatment Blood Lymphocyte Count on Intended and Unintended Effects of Anti-T-Lymphocyte Globulin in Allogeneic Hematopoietic Stem Cell Transplantation. Cells. 2023; 12(14):1831. https://doi.org/10.3390/cells12141831
Chicago/Turabian StyleNikoloudis, Alexander, Veronika Buxhofer-Ausch, Christoph Aichinger, Michaela Binder, Petra Hasengruber, Emine Kaynak, Dagmar Wipplinger, Robert Milanov, Irene Strassl, Olga Stiefel, and et al. 2023. "Impact of the Recipient’s Pre-Treatment Blood Lymphocyte Count on Intended and Unintended Effects of Anti-T-Lymphocyte Globulin in Allogeneic Hematopoietic Stem Cell Transplantation" Cells 12, no. 14: 1831. https://doi.org/10.3390/cells12141831
APA StyleNikoloudis, A., Buxhofer-Ausch, V., Aichinger, C., Binder, M., Hasengruber, P., Kaynak, E., Wipplinger, D., Milanov, R., Strassl, I., Stiefel, O., Machherndl-Spandl, S., Petzer, A., Weltermann, A., & Clausen, J. (2023). Impact of the Recipient’s Pre-Treatment Blood Lymphocyte Count on Intended and Unintended Effects of Anti-T-Lymphocyte Globulin in Allogeneic Hematopoietic Stem Cell Transplantation. Cells, 12(14), 1831. https://doi.org/10.3390/cells12141831






