SGK1 Target Genes Involved in Heart and Blood Vessel Functions in PC12 Cells
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Line and Reagents
2.2. Real-Time PCR Assay
2.3. SDS-PAGE and Western Blotting
2.4. RNA-Seq and DEG Identification
2.5. Gene Enrichment Analysis
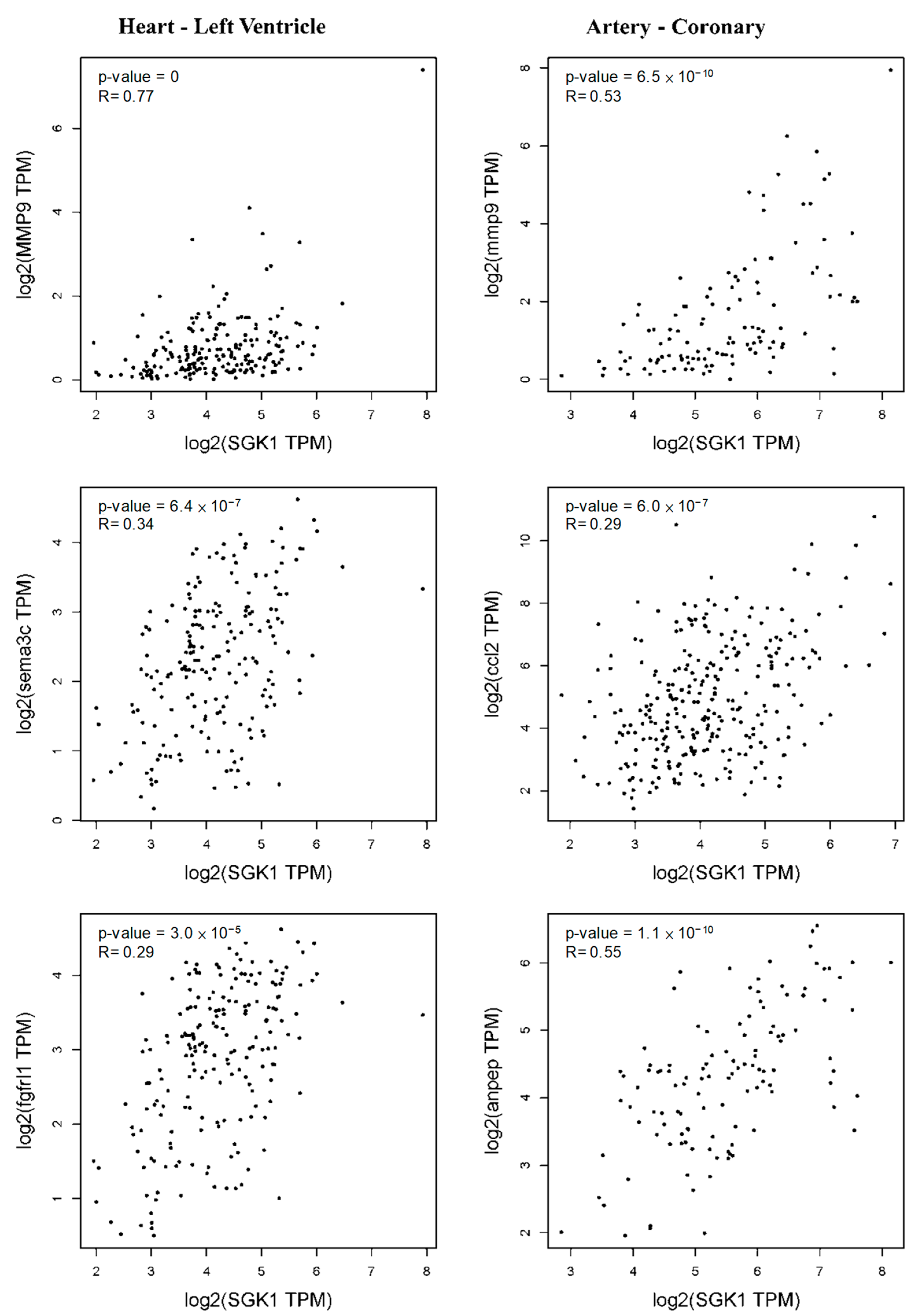
2.6. GEPIA2 Correlation Analysis
2.7. Statistical Analysis
3. Results
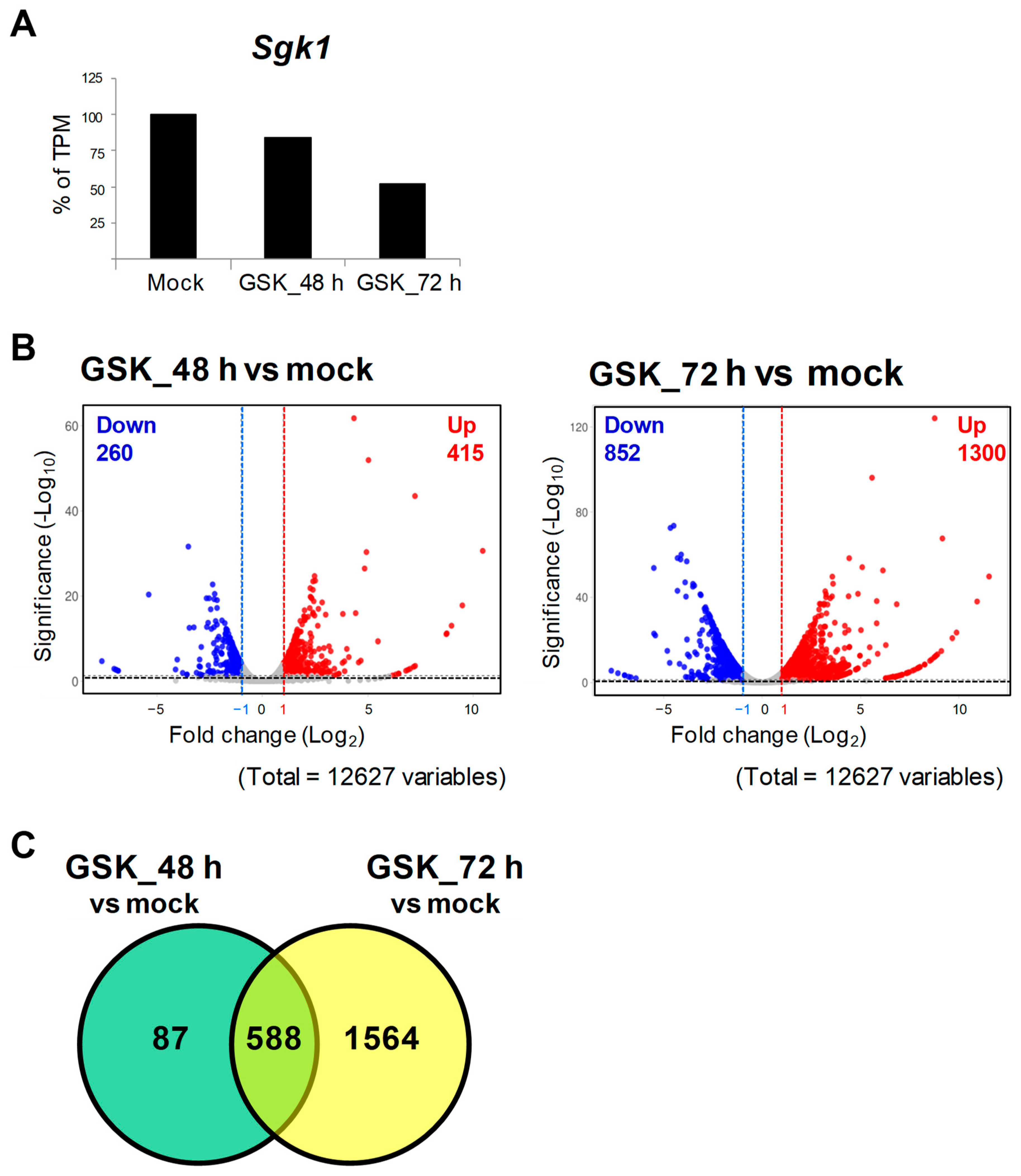
3.1. SGK1 Inhibitor GSK650394 Inhibits SGK1 Expression in PC12 Cells
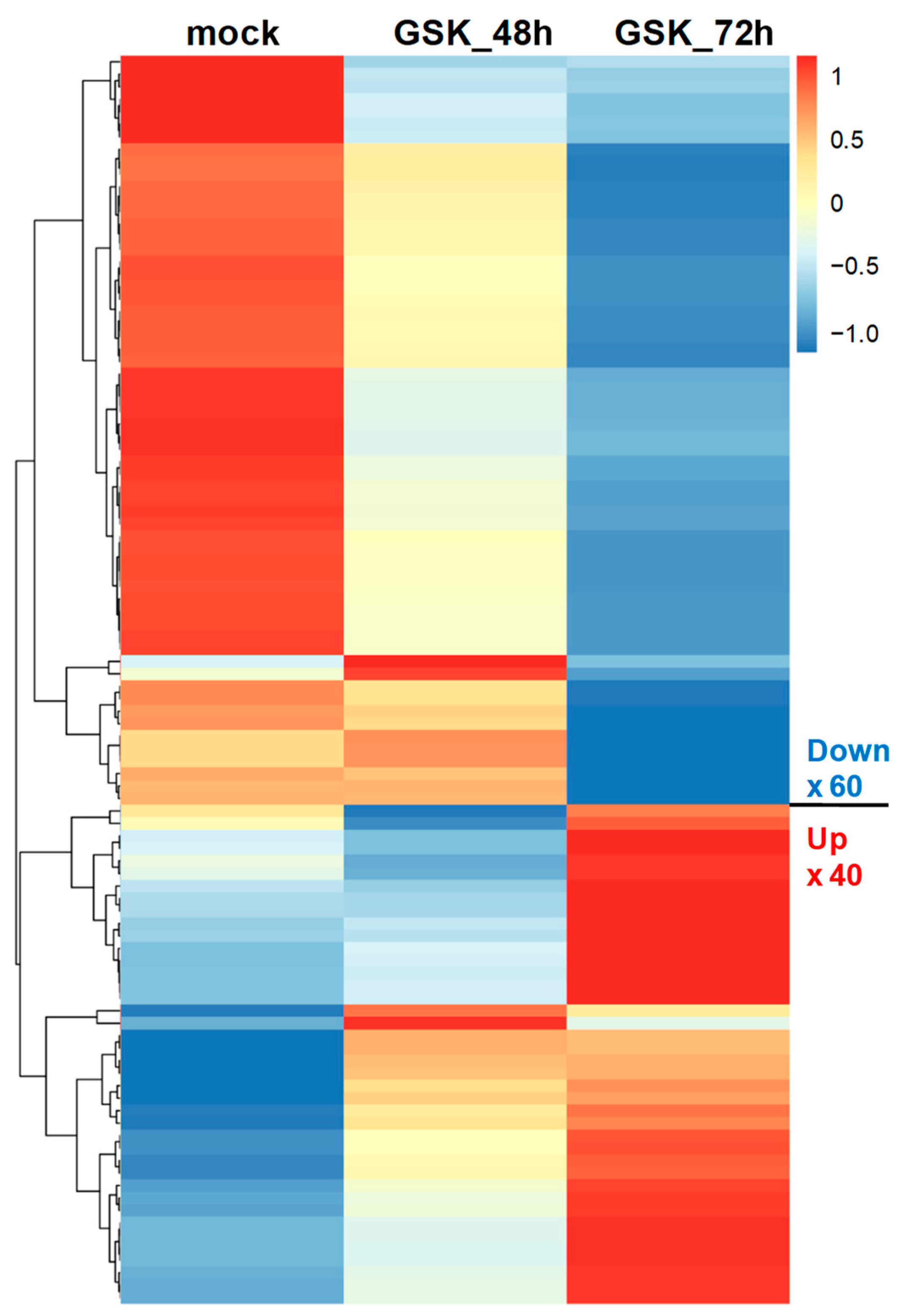
3.2. RNA-Seq and DEG Analysis of GSK650394-Treated PC12 Cells
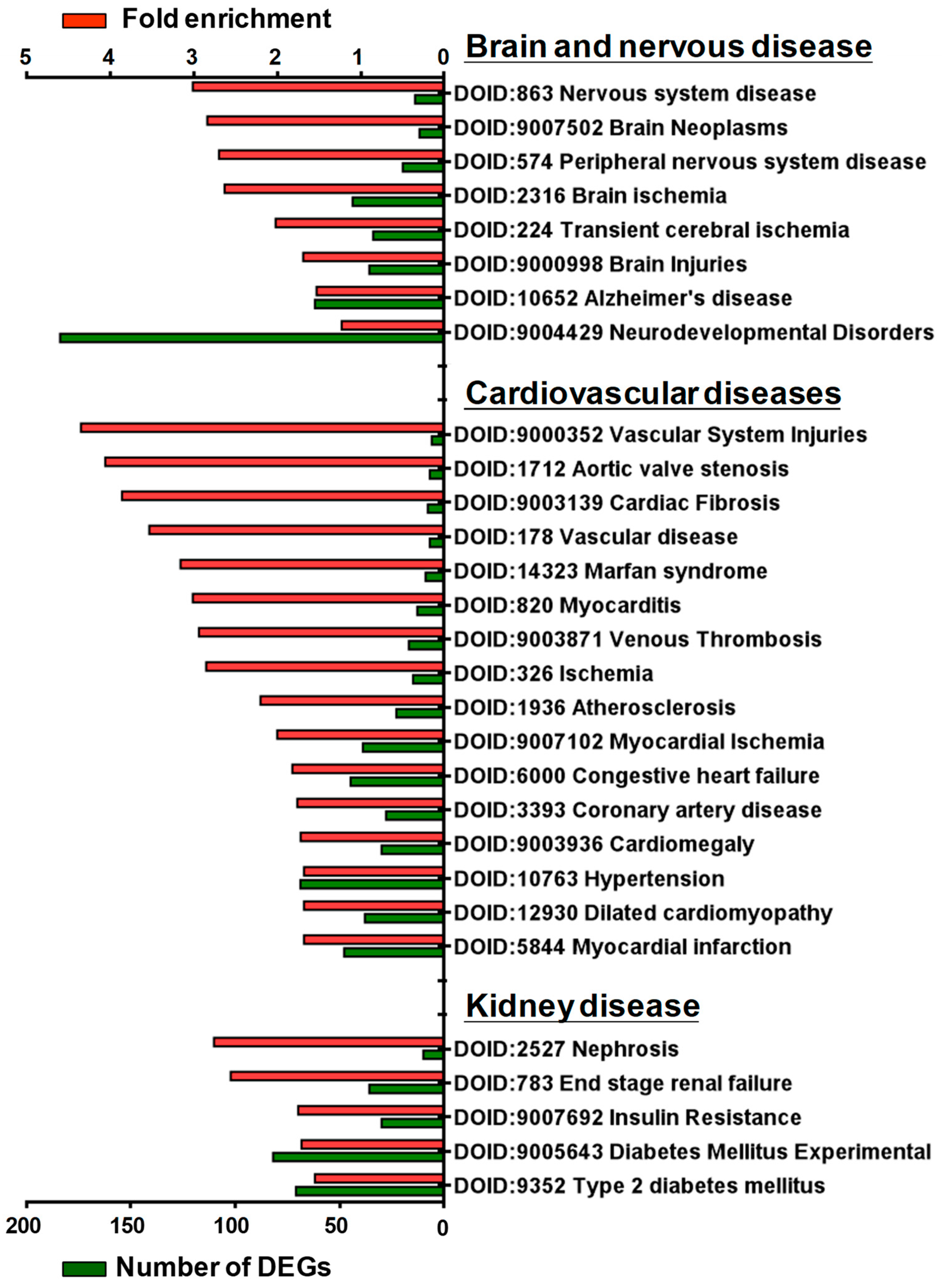
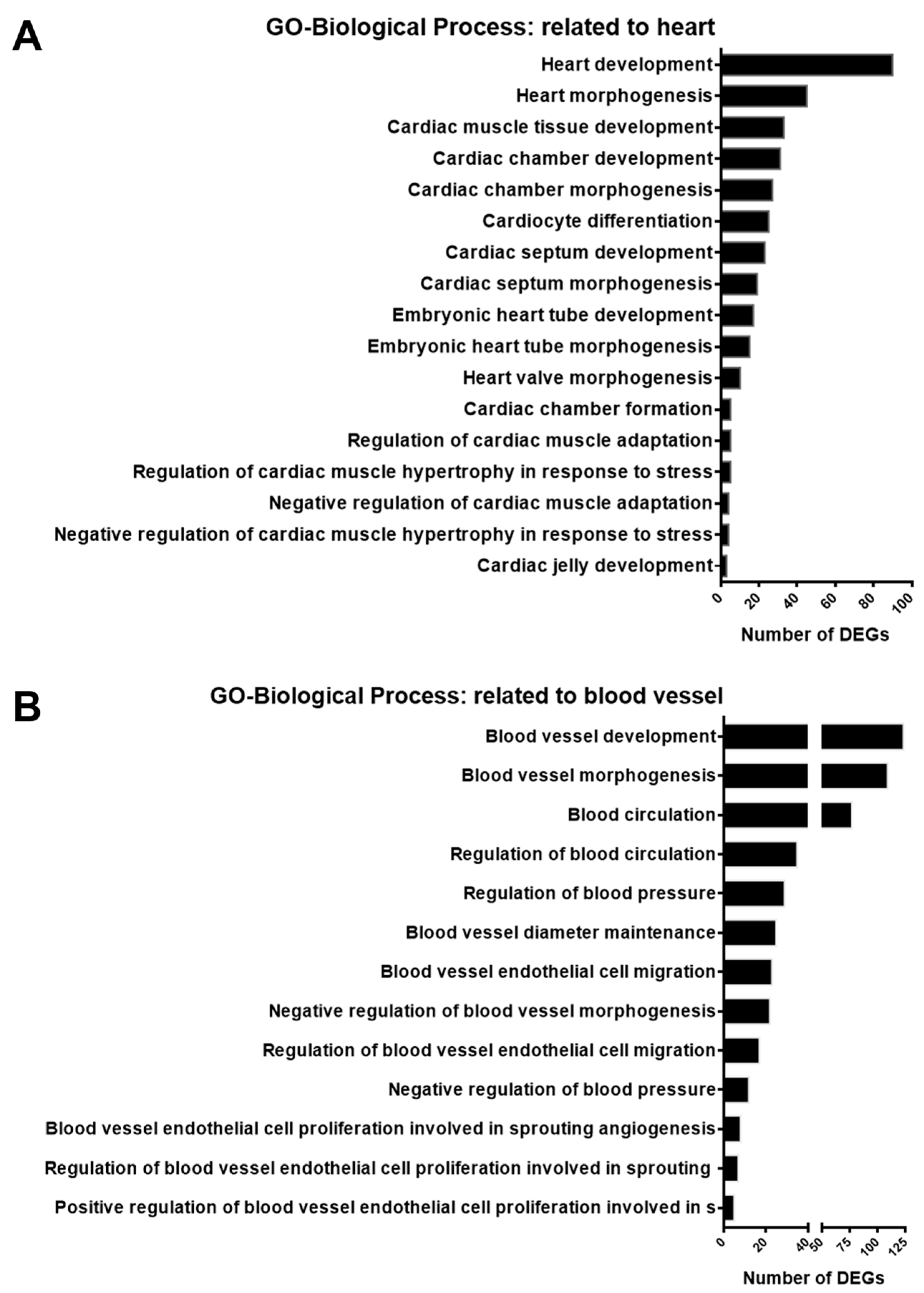
3.3. Gene Enrichment Analysis of DEGs in GSK650394-Treated PC12 Cells
3.4. Validation of Selected DEGs Related to Heart and Blood Vessel Functions
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Webster, M.K.; Goya, L.; Ge, Y.; Maiyar, A.C.; Firestone, G.L. Characterization of sgk, a novel member of the serine/threonine protein kinase gene family which is transcriptionally induced by glucocorticoids and serum. Mol. Cell. Biol. 1993, 13, 2031–2040. [Google Scholar] [CrossRef] [PubMed]
- Lang, F.; Böhmer, C.; Palmada, M.; Seebohm, G.; Strutz-Seebohm, N.; Vallon, V. (Patho)physiological significance of the serum- and glucocorticoid-inducible kinase isoforms. Physiol. Rev. 2006, 86, 1151–1178. [Google Scholar] [CrossRef] [PubMed]
- Lang, F.; Artunc, F.; Vallon, V. The physiological impact of the serum and glucocorticoid-inducible kinase SGK1. Curr. Opin. Nephrol. Hypertens. 2009, 18, 439–448. [Google Scholar] [CrossRef] [PubMed]
- Lang, F.; Shumilina, E. Regulation of ion channels by the serum- and glucocorticoid-inducible kinase SGK1. FASEB J. 2013, 27, 3–12. [Google Scholar] [CrossRef]
- von Wowern, F.; Berglund, G.; Carlson, J.; Månsson, H.; Hedblad, B.; Melander, O. Genetic variance of SGK-1 is associated with blood pressure, blood pressure change over time and strength of the insulin-diastolic blood pressure relationship. Kidney Int. 2005, 68, 2164–2172. [Google Scholar] [CrossRef]
- Lang, F.; Strutz-Seebohm, N.; Seebohm, G.; Lang, U.E. Significance of SGK1 in the regulation of neuronal function. J. Physiol. 2010, 588, 3349–3354. [Google Scholar] [CrossRef]
- Schoenebeck, B.; Bader, V.; Zhu, X.R.; Schmitz, B.; Lübbert, H.; Stichel, C.C. Sgk1, a cell survival response in neurodegenerative diseases. Mol. Cell. Neurosci. 2005, 30, 249–264. [Google Scholar] [CrossRef]
- Sierra-Ramos, C.; Velazquez-Garcia, S.; Vastola-Mascolo, A.; Hernández, G.; Faresse, N.; Alvarez de la Rosa, D. SGK1 activation exacerbates diet-induced obesity, metabolic syndrome and hypertension. J. Endocrinol. 2020, 244, 149–162. [Google Scholar] [CrossRef]
- Sang, Y.; Kong, P.; Zhang, S.; Zhang, L.; Cao, Y.; Duan, X.; Sun, T.; Tao, Z.; Liu, W. SGK1 in Human Cancer: Emerging Roles and Mechanisms. Front. Oncol. 2020, 10, 608722. [Google Scholar] [CrossRef]
- Sierra-Ramos, C.; Velazquez-Garcia, S.; Keskus, A.G.; Vastola-Mascolo, A.; Rodríguez-Rodríguez, A.E.; Luis-Lima, S.; Hernández, G.; Navarro-González, J.F.; Porrini, E.; Konu, O.; et al. Increased SGK1 activity potentiates mineralocorticoid/NaCl-induced kidney injury. Am. J. Physiol. Ren. Physiol. 2021, 320, F628–F643. [Google Scholar] [CrossRef]
- Busjahn, A.; Seebohm, G.; Maier, G.; Toliat, M.R.; Nürnberg, P.; Aydin, A.; Luft, F.C.; Lang, F. Association of the serum and glucocorticoid regulated kinase (sgk1) gene with QT interval. Cell. Physiol. Biochem. Int. J. Exp. Cell. Physiol. Biochem. Pharmacol. 2004, 14, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Das, S.; Aiba, T.; Rosenberg, M.; Hessler, K.; Xiao, C.; Quintero, P.A.; Ottaviano, F.G.; Knight, A.C.; Graham, E.L.; Boström, P.; et al. Pathological role of serum- and glucocorticoid-regulated kinase 1 in adverse ventricular remodeling. Circulation 2012, 126, 2208–2219. [Google Scholar] [CrossRef] [PubMed]
- Medina-Jover, F.; Gendrau-Sanclemente, N.; Viñals, F. SGK1 is a signalling hub that controls protein synthesis and proliferation in endothelial cells. FEBS Lett. 2020, 594, 3200–3215. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.L.; Tsai, M.C.; Hsu, W.L.; Lee, E.H. SGK protein kinase facilitates the expression of long-term potentiation in hippocampal neurons. Learn. Mem. 2006, 13, 114–118. [Google Scholar] [CrossRef]
- Anacker, C.; Cattaneo, A.; Musaelyan, K.; Zunszain, P.A.; Horowitz, M.; Molteni, R.; Luoni, A.; Calabrese, F.; Tansey, K.; Gennarelli, M.; et al. Role for the kinase SGK1 in stress, depression, and glucocorticoid effects on hippocampal neurogenesis. Proc. Natl. Acad. Sci. USA 2013, 110, 8708–8713. [Google Scholar] [CrossRef]
- Celano, C.M.; Freudenreich, O.; Fernandez-Robles, C.; Stern, T.A.; Caro, M.A.; Huffman, J.C. Depressogenic effects of medications: A review. Dialogues Clin. Neurosci. 2011, 13, 109–125. [Google Scholar] [CrossRef]
- Dattilo, V.; Amato, R.; Perrotti, N.; Gennarelli, M. The Emerging Role of SGK1 (Serum- and Glucocorticoid-Regulated Kinase 1) in Major Depressive Disorder: Hypothesis and Mechanisms. Front. Genet. 2020, 11, 826. [Google Scholar] [CrossRef]
- Meng, P.; Zhu, Q.; Yang, H.; Liu, D.; Lin, X.; Liu, J.; Fan, J.; Liu, X.; Su, W.; Liu, L.; et al. Leonurine promotes neurite outgrowth and neurotrophic activity by modulating the GR/SGK1 signaling pathway in cultured PC12 cells. Neuroreport 2019, 30, 247–254. [Google Scholar] [CrossRef]
- Hadaya, J.; Ardell, J.L. Autonomic Modulation for Cardiovascular Disease. Front. Physiol. 2020, 11, 617459. [Google Scholar] [CrossRef]
- Greene, L.A.; Tischler, A.S. Establishment of a noradrenergic clonal line of rat adrenal pheochromocytoma cells which respond to nerve growth factor. Proc. Natl. Acad. Sci. USA 1976, 73, 2424–2428. [Google Scholar] [CrossRef]
- Schriml, L.M.; Munro, J.B.; Schor, M.; Olley, D.; McCracken, C.; Felix, V.; Baron, J.A.; Jackson, R.; Bello, S.M.; Bearer, C.; et al. The Human Disease Ontology 2022 update. Nucleic Acids Res. 2022, 50, D1255–D1261. [Google Scholar] [CrossRef]
- Lang, F.; Stournaras, C. Serum and glucocorticoid inducible kinase, metabolic syndrome, inflammation, and tumor growth. Hormones 2013, 12, 160–171. [Google Scholar] [CrossRef] [PubMed]
- Lang, F.; Stournaras, C.; Zacharopoulou, N.; Voelkl, J.; Alesutan, I. Serum- and glucocorticoid-inducible kinase 1 and the response to cell stress. Cell Stress 2018, 3, 1–8. [Google Scholar] [CrossRef]
- Yang, Y.C.; Lin, C.H.; Lee, E.H. Serum- and glucocorticoid-inducible kinase 1 (SGK1) increases neurite formation through microtubule depolymerization by SGK1 and by SGK1 phosphorylation of tau. Mol. Cell. Biol. 2006, 26, 8357–8370. [Google Scholar] [CrossRef]
- Liu, T.; Yu, T.; Hu, H.; He, K. Knockdown of the long non-coding RNA HOTTIP inhibits colorectal cancer cell proliferation and migration and induces apoptosis by targeting SGK1. Biomed. Pharmacother. 2018, 98, 286–296. [Google Scholar] [CrossRef]
- Schmidt, E.M.; Kraemer, B.F.; Borst, O.; Münzer, P.; Schönberger, T.; Schmidt, C.; Leibrock, C.; Towhid, S.T.; Seizer, P.; Kuhl, D.; et al. SGK1 sensitivity of platelet migration. Cell. Physiol. Biochem. Int. J. Exp. Cell. Physiol. Biochem. Pharmacol. 2012, 30, 259–268. [Google Scholar] [CrossRef]
- Walker-Allgaier, B.; Schaub, M.; Alesutan, I.; Voelkl, J.; Geue, S.; Münzer, P.; Rodríguez, J.M.; Kuhl, D.; Lang, F.; Gawaz, M.; et al. SGK1 up-regulates Orai1 expression and VSMC migration during neointima formation after arterial injury. Thromb. Haemost. 2017, 117, 1002–1005. [Google Scholar] [CrossRef]
- Kleinewietfeld, M.; Manzel, A.; Titze, J.; Kvakan, H.; Yosef, N.; Linker, R.A.; Muller, D.N.; Hafler, D.A. Sodium chloride drives autoimmune disease by the induction of pathogenic TH17 cells. Nature 2013, 496, 518–522. [Google Scholar] [CrossRef]
- Catela, C.; Kratsios, P.; Hede, M.; Lang, F.; Rosenthal, N. Serum and glucocorticoid-inducible kinase 1 (SGK1) is necessary for vascular remodeling during angiogenesis. Dev. Dyn. 2010, 239, 2149–2160. [Google Scholar] [CrossRef] [PubMed]
- Zarrinpashneh, E.; Poggioli, T.; Sarathchandra, P.; Lexow, J.; Monassier, L.; Terracciano, C.; Lang, F.; Damilano, F.; Zhou, J.Q.; Rosenzweig, A.; et al. Ablation of SGK1 impairs endothelial cell migration and tube formation leading to decreased neo-angiogenesis following myocardial infarction. PLoS ONE 2013, 8, e80268. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.J.; Zhang, P.; Li, L.; Chen, S.X.; Joines, A.; Wu, D.; Wong, M.; Shahan, J.; Golden, T.; Wu, N.; et al. Differential gene expression profile analysis in corticosterone-treated PC12 cells. Int. J. Clin. Exp. Pathol. 2018, 11, 3097–3103. [Google Scholar] [PubMed]
- Halade, G.V.; Jin, Y.F.; Lindsey, M.L. Matrix metalloproteinase (MMP)-9: A proximal biomarker for cardiac remodeling and a distal biomarker for inflammation. Pharmacol. Ther. 2013, 139, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Matschke, V.; Theiss, C.; Hollmann, M.; Schulze-Bahr, E.; Lang, F.; Seebohm, G.; Strutz-Seebohm, N. NDRG2 phosphorylation provides negative feedback for SGK1-dependent regulation of a kainate receptor in astrocytes. Front. Cell. Neurosci. 2015, 9, 387. [Google Scholar] [CrossRef]
- Murray, J.T.; Campbell, D.G.; Morrice, N.; Auld, G.C.; Shpiro, N.; Marquez, R.; Peggie, M.; Bain, J.; Bloomberg, G.B.; Grahammer, F.; et al. Exploitation of KESTREL to identify NDRG family members as physiological substrates for SGK1 and GSK3. Biochem. J. 2004, 384, 477–488. [Google Scholar] [CrossRef]
- Maruyama, Y.; Ono, M.; Kawahara, A.; Yokoyama, T.; Basaki, Y.; Kage, M.; Aoyagi, S.; Kinoshita, H.; Kuwano, M. Tumor growth suppression in pancreatic cancer by a putative metastasis suppressor gene Cap43/NDRG1/Drg-1 through modulation of angiogenesis. Cancer Res. 2006, 66, 6233–6242. [Google Scholar] [CrossRef]
- Kim, A.; Kim, M.J.; Yang, Y.; Kim, J.W.; Yeom, Y.I.; Lim, J.S. Suppression of NF-kappaB activity by NDRG2 expression attenuates the invasive potential of highly malignant tumor cells. Carcinogenesis 2009, 30, 927–936. [Google Scholar] [CrossRef]
- Vieira, J.R.; Shah, B.; Dupraz, S.; Paredes, I.; Himmels, P.; Schermann, G.; Adler, H.; Motta, A.; Gärtner, L.; Navarro-Aragall, A.; et al. Endothelial PlexinD1 signaling instructs spinal cord vascularization and motor neuron development. Neuron 2022, 110, 4074–4089.E6. [Google Scholar] [CrossRef]
- Epstein, J.A.; Aghajanian, H.; Singh, M.K. Semaphorin signaling in cardiovascular development. Cell Metab. 2015, 21, 163–173. [Google Scholar] [CrossRef] [PubMed]
- Roque, C.; Baltazar, G. G protein-coupled estrogen receptor 1 (GPER) activation triggers different signaling pathways on neurons and astrocytes. Neural Regen. Res. 2019, 14, 2069–2070. [Google Scholar] [CrossRef]
- Gohar, E.Y. G protein-coupled estrogen receptor 1 as a novel regulator of blood pressure. Am. J. Physiol. Ren. Physiol. 2020, 319, F612–F617. [Google Scholar] [CrossRef] [PubMed]
- Duan, L.; Zhang, X.D.; Miao, W.Y.; Sun, Y.J.; Xiong, G.; Wu, Q.; Li, G.; Yang, P.; Yu, H.; Li, H.; et al. PDGFRβ Cells Rapidly Relay Inflammatory Signal from the Circulatory System to Neurons via Chemokine CCL2. Neuron 2018, 100, 183–200.E8. [Google Scholar] [CrossRef] [PubMed]
- Roblek, M.; Protsyuk, D.; Becker, P.F.; Stefanescu, C.; Gorzelanny, C.; Glaus Garzon, J.F.; Knopfova, L.; Heikenwalder, M.; Luckow, B.; Schneider, S.W.; et al. CCL2 Is a Vascular Permeability Factor Inducing CCR2-Dependent Endothelial Retraction during Lung Metastasis. Mol. Cancer Res. 2019, 17, 783–793. [Google Scholar] [CrossRef] [PubMed]
- Park, H.J.; Hong, M.; Bronson, R.T.; Israel, M.A.; Frankel, W.N.; Yun, K. Elevated Id2 expression results in precocious neural stem cell depletion and abnormal brain development. Stem Cells 2013, 31, 1010–1021. [Google Scholar] [CrossRef] [PubMed]
- Gratze, P.; Dechend, R.; Stocker, C.; Park, J.K.; Feldt, S.; Shagdarsuren, E.; Wellner, M.; Gueler, F.; Rong, S.; Gross, V.; et al. Novel role for inhibitor of differentiation 2 in the genesis of angiotensin II-induced hypertension. Circulation 2008, 117, 2645–2656. [Google Scholar] [CrossRef] [PubMed]




| Symbol | Description | Forward Primer Sequences (5′ > 3′) | Reverse Primer Sequences (5′ > 3′) |
|---|---|---|---|
| Sgk1 | serum/glucocorticoid regulated kinase 1 | TTCTGTGGCACGCCTGAGTA | GGTCGTACGGCTGCTTATGG |
| Gapdh | glyceraldehyde-3-phosphate dehydrogenase | GGCAAGTTCAACGGCACAGT | ATGGGTTTCCCGTTGATGAC |
| Adamts1 | ADAM metallopeptidase with thrombospondin type 1 motif, 1 | CAGGGCGCGGGAAAG | GGACACAAATCGCTTCTTCCTT |
| Anpep | alanyl aminopeptidase, membrane | CCAAGGGTTTCTACATTTCCAAGA | CGGCTGCCACACCTAACAG |
| Atp1a1 | ATPase Na+/K+ transporting subunit alpha 1 | CATCTTCCTCATTGGTATCATCGT | TGACGGTGGCCAGCAAA |
| Atp2b4 | ATPase plasma membrane Ca2+ transporting 4 | GGGCTGCAGAGTCGCATT | TGTATAAGCTGACCATTCCGGATA |
| Bmp2 | bone morphogenetic protein 2 | ACGACGTCCTCAGCGAGTTT | GGTCTCTGCTTCAGGCCAAA |
| Ccl2 | C-C motif chemokine ligand 2 | CACGCTTCTGGGCCTGTT | TGAGACAGCACGTGGATGCT |
| Col2a1 | collagen type II alpha 1 chain | GACTGGCGAGTCTTGCGTCTA | CTTGCTGCTCCACCAGTTCTT |
| Dll1 | delta-like canonical Notch ligand 1 | CTGCACTGACCCCATTTGTCT | CCCGGTTTGTCACAATATCCA |
| Dll4 | delta-like canonical Notch ligand 4 | GAATAGCGGCAGTGGTCGTAA | GGTTCCCGGCCAGGTAAA |
| Errfi1 | ERBB receptor feedback inhibitor 1 | CGTGCCTTCCGATTTCAAA | CGCACCCACGGAAGCTT |
| Fgfrl1 | fibroblast growth factor receptor-like 1 | CCCACCACCGTTGACCAT | TCCAGCCACTGTGGATCGT |
| Foxo1 | forkhead box O1 | CCTCGAACCAGCTCAAACG | CTGCTCTGTCATGATGGGAGAA |
| Fzd7 | frizzled class receptor 7 | GTGCACGGATATTGCCTACAAC | CCCGCGTCCTCTTGGTT |
| Gja1 | gap junction protein alpha 1 | GGCCTTCCTGCTCATCCA | AGGTGTAGACCGCGCTCAA |
| Gper1 | G protein-coupled estrogen receptor 1 | CCTGGACGAGCAGTACTACGATATC | TGATCTGCAGGAAGAGGGACAT |
| Gucy1a1 | guanylate cyclase 1 soluble subunit alpha 1 | TGCTTCTCCCCGGTATCATT | TCCACGTGGCTTTCGTACAG |
| Hmox1 | heme oxygenase 1 | TGATGGCCTCCTTGTACCATATC | GGTTCTGCTTGTTTCGCTCTATC |
| Hrh3 | histamine receptor H3 | TCGCCATCTCCGACTTCCT | GTCAGCACATAGGGTACGTACAATG |
| Id2 | inhibitor of DNA binding 2 | CGGTGAGGTCCGTTAGGAAA | CGGGAGATGCCCAAGCT |
| Id3 | inhibitor of DNA binding 3 | TTAGCCTCTTGGACGACATGAA | GGACTCCCGGCACCAGTT |
| Mmp2 | matrix metallopeptidase 2 | ACGCTTTTCTCGAATCCATGA | ATGCTCCCATCGACCAAAGT |
| Mmp9 | matrix metallopeptidase 9 | TTCGAAGGCGACCTCAAGTG | TTCGGTGTAGCTTTGGATCCA |
| Ncald | neurocalcin delta | CATAGCCCTGAGTGTGACATCAA | ACATGCTGAAGGCCCACTTC |
| Nisch | Nischarin | CCCTGGAAGGCGTACACACT | AAGTTGCCTGCCAGGTTTAGG |
| Npy1r | neuropeptide Y receptor Y1 | TGCTGGTCGCAGTCATGTGT | CCCAGTGGTCCATCAGTGTGT |
| Nr2f2 | nuclear receptor subfamily 2 group F member 2 | CAGCACCACCGCAACCA | CCCACTTTGAGGCACTTTTTG |
| Pik3r1 | phosphoinositide-3-kinase regulatory subunit 1 | GCAAGAGCCCTCTCTGAAATTTT | AGAGCTGGCTGCTGGGAAT |
| Ppara | peroxisome proliferator activated receptor alpha | GCTAAAGCTGGCGTACGACAA | TGTTCCGGTTCTTTTTCTGAATC |
| Sema3c | semaphorin 3C | CATTTGCGTCCAAGGATCATC | CGAAGCTCATCGAATGTTAAATACA |
| Slit2 | slit guidance ligand 2 | TTCCGTGGTGCAGTTGACAT | CCATCTTCAATGCAGCTGATCT |
| Smad3 | SMAD family member 3 | GGGCCTGCTGTCCAATGTT | AATGTGCCGCCTTGTAAGCT |
| Smo | smoothened, frizzled class receptor | ACGAGGGTGGCCTGACTTT | GGACAGCCTTCAGGGAAGTG |
| Smtn | smoothelin | AACCCCTCTTGCAGGATCATC | CGCACTCGGTCTCTCACAGA |
| Sod2 | superoxide dismutase 2 | GGGCTGGCTTGGCTTCA | AGCAGGCGGCAATCTGTAA |
| Tbr1 | T-box brain transcription factor 1 | TGCAAGGAAACCGGGTCTAT | CATCCAGTGAGCCCCAGTGT |
| Tbx2 | T-box transcription factor 2 | ATCGTGCGAGCCAACGA | GGAAGACATAGGTGCGAAAGGT |
| Tbx3 | T-box transcription factor 3 | AAGGAGACCGGAACTTCTGATG | CCTGGGCAAAGCAGTTGAA |
| tbxa2r | thromboxane A2 receptor | GCCACATGGAACCAGATCTTG | TGCAGGCGTCGAAGCA |
| Tnf | tumor necrosis factor | TGATCGGTCCCAACAAGGA | GGGCCATGGAACTGATGAGA |
| Trpc3 | transient receptor potential cation channel subfamily C member 3 | CCTTGGGCCTTCCATTCCT | CCCCAGCCTGCTACAAGGT |
| Vegfc | vascular endothelial growth factor C | GGCCCCAAACCAGTCACA | ACATGCACCGGCAGGAA |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, Y.-H.; Sun, C.-C.; Chen, P.-M.; Chen, H.-H. SGK1 Target Genes Involved in Heart and Blood Vessel Functions in PC12 Cells. Cells 2023, 12, 1641. https://doi.org/10.3390/cells12121641
Li Y-H, Sun C-C, Chen P-M, Chen H-H. SGK1 Target Genes Involved in Heart and Blood Vessel Functions in PC12 Cells. Cells. 2023; 12(12):1641. https://doi.org/10.3390/cells12121641
Chicago/Turabian StyleLi, Yu-He, Chia-Cheng Sun, Po-Ming Chen, and Hsin-Hung Chen. 2023. "SGK1 Target Genes Involved in Heart and Blood Vessel Functions in PC12 Cells" Cells 12, no. 12: 1641. https://doi.org/10.3390/cells12121641
APA StyleLi, Y.-H., Sun, C.-C., Chen, P.-M., & Chen, H.-H. (2023). SGK1 Target Genes Involved in Heart and Blood Vessel Functions in PC12 Cells. Cells, 12(12), 1641. https://doi.org/10.3390/cells12121641






